Impact of Bacterial Cellulose Nanocrystals-Gelatin/Cinnamon Essential Oil Emulsion Coatings on the Quality Attributes of ‘Red Delicious’ Apples
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials and Chemicals
2.2. Coating Preparation
2.3. Sample Preparation
2.4. Sample Characterization
2.4.1. Weight Loss (WL)
2.4.2. Respiration Rate
2.4.3. C2H4 Production Rate
2.4.4. Flesh Firmness
2.4.5. Color Measurement
2.4.6. Total Soluble Solids (TSS), Total Acidity (TA), pH, and Vitamin C
2.4.7. Polyphenol Oxidase (PPO) and Peroxidase (POD) Activity
2.4.8. Sensory Analysis
2.5. Statistical Analysis
3. Results
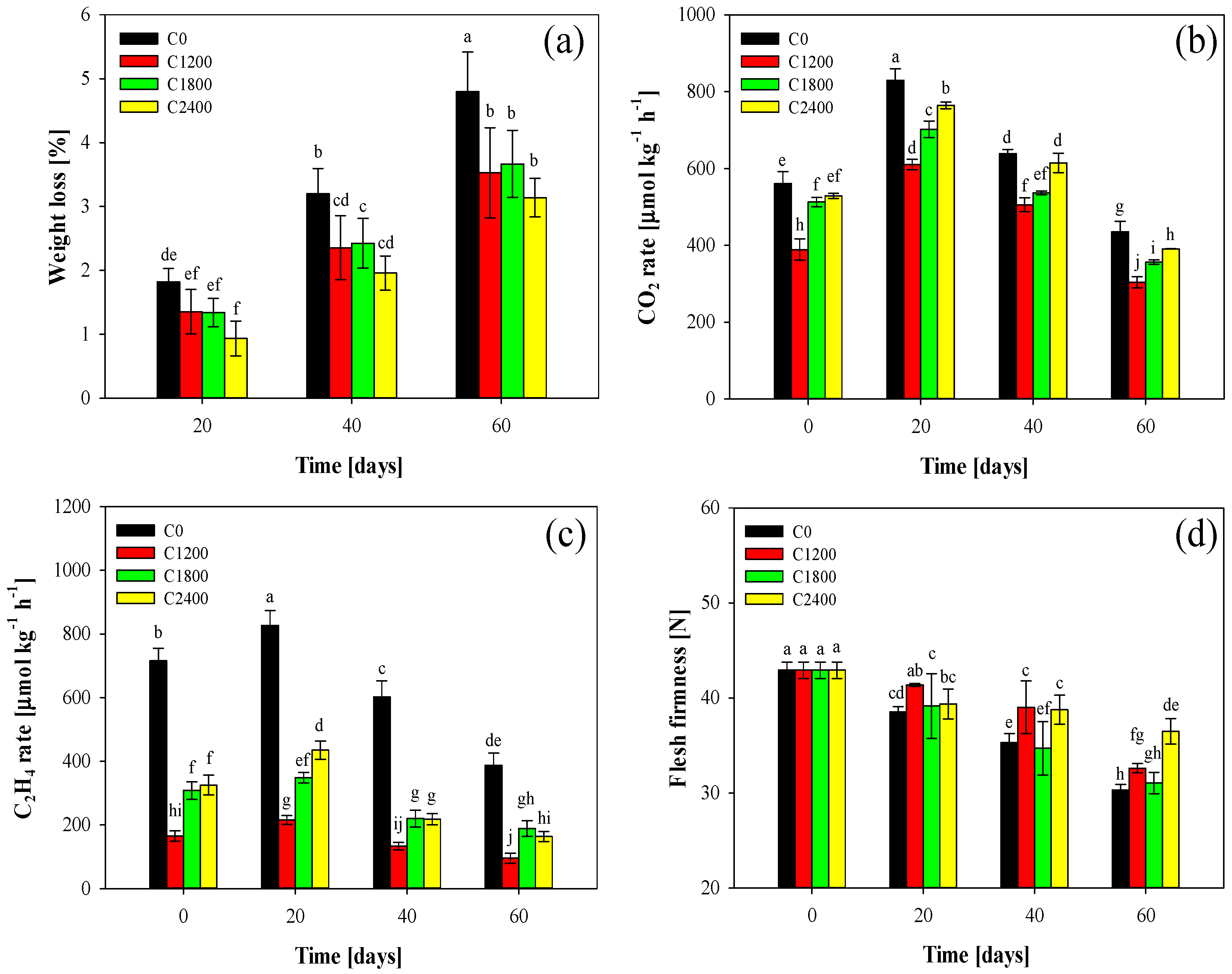
3.1. Weight Loss
3.2. Respiration Rate and c2h4 Production
3.3. Flesh Firmness
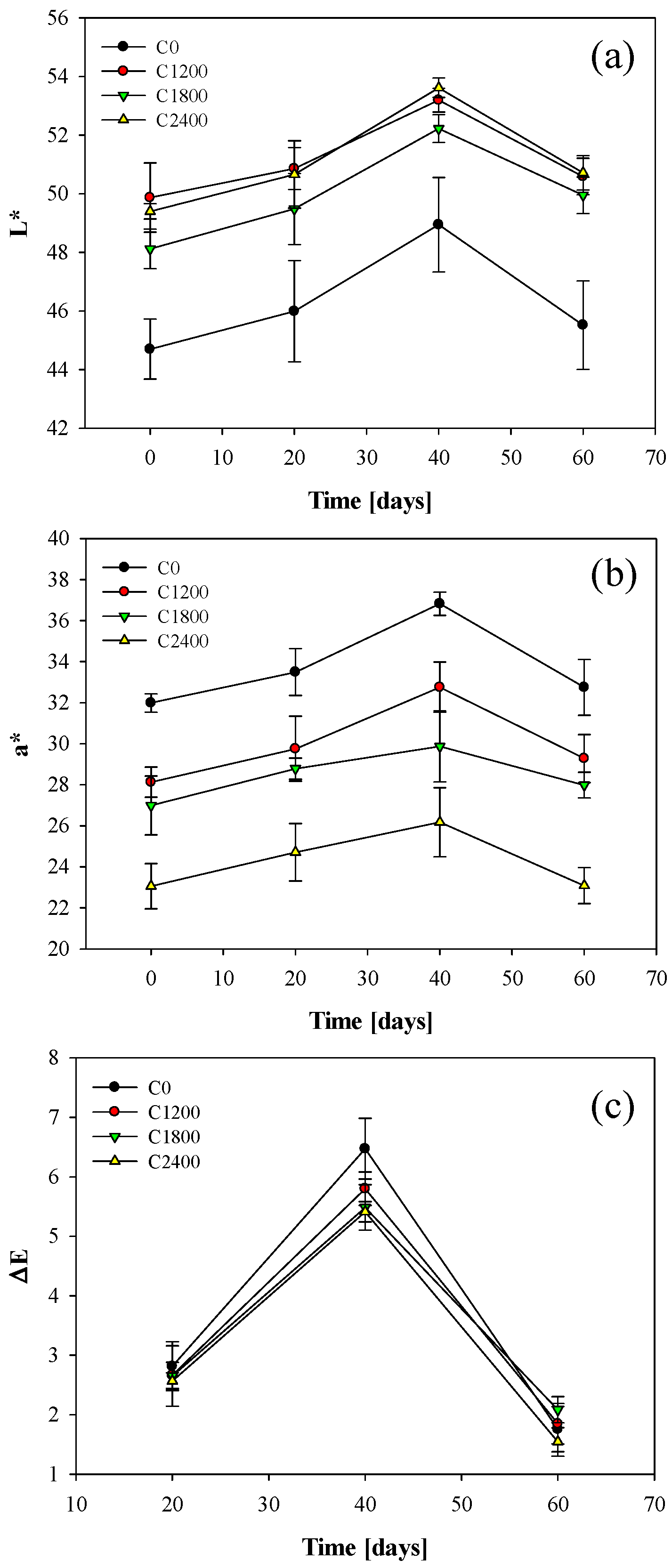
3.4. Color Measurements
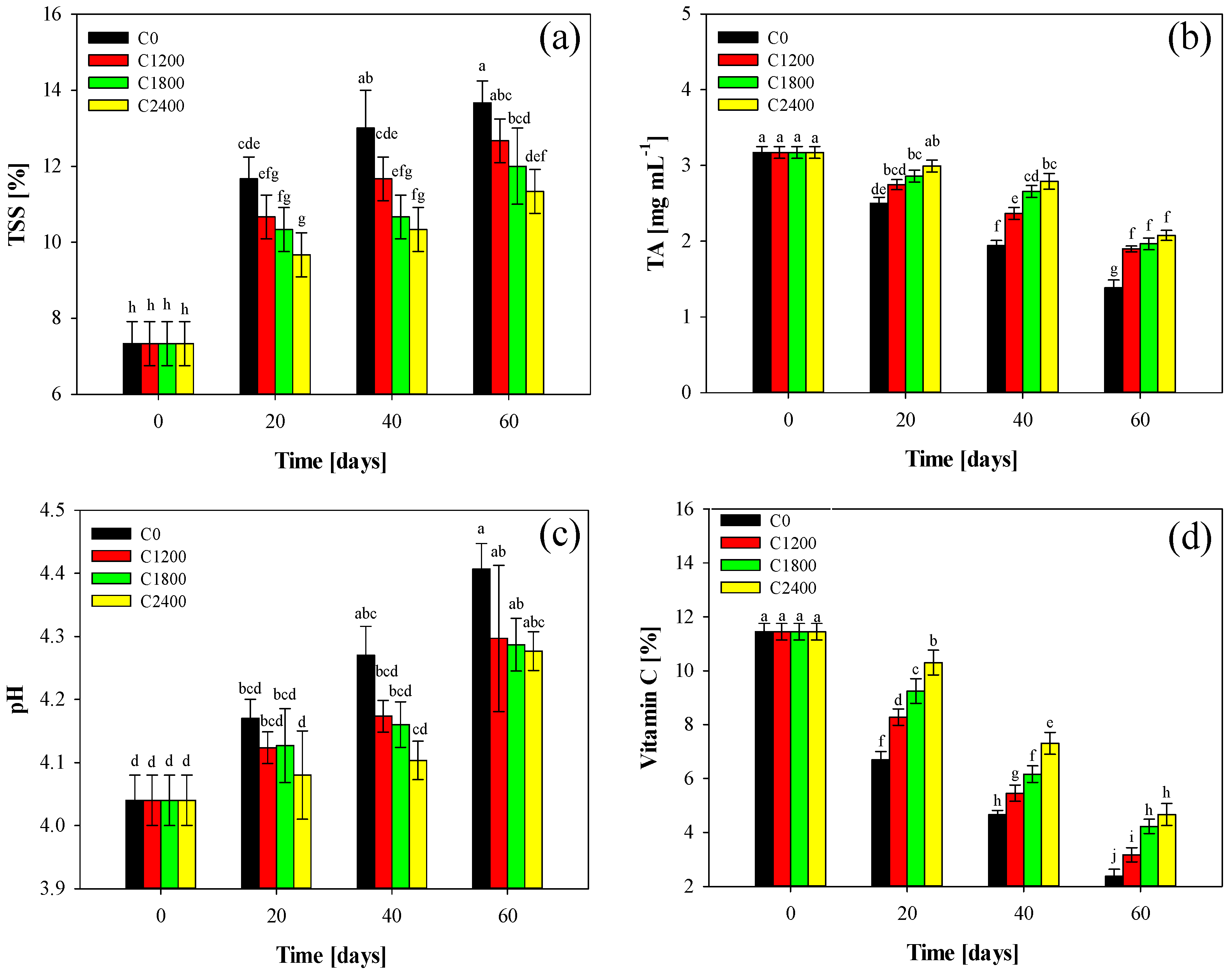
3.5. TSS, TA, pH, and Vitamin C
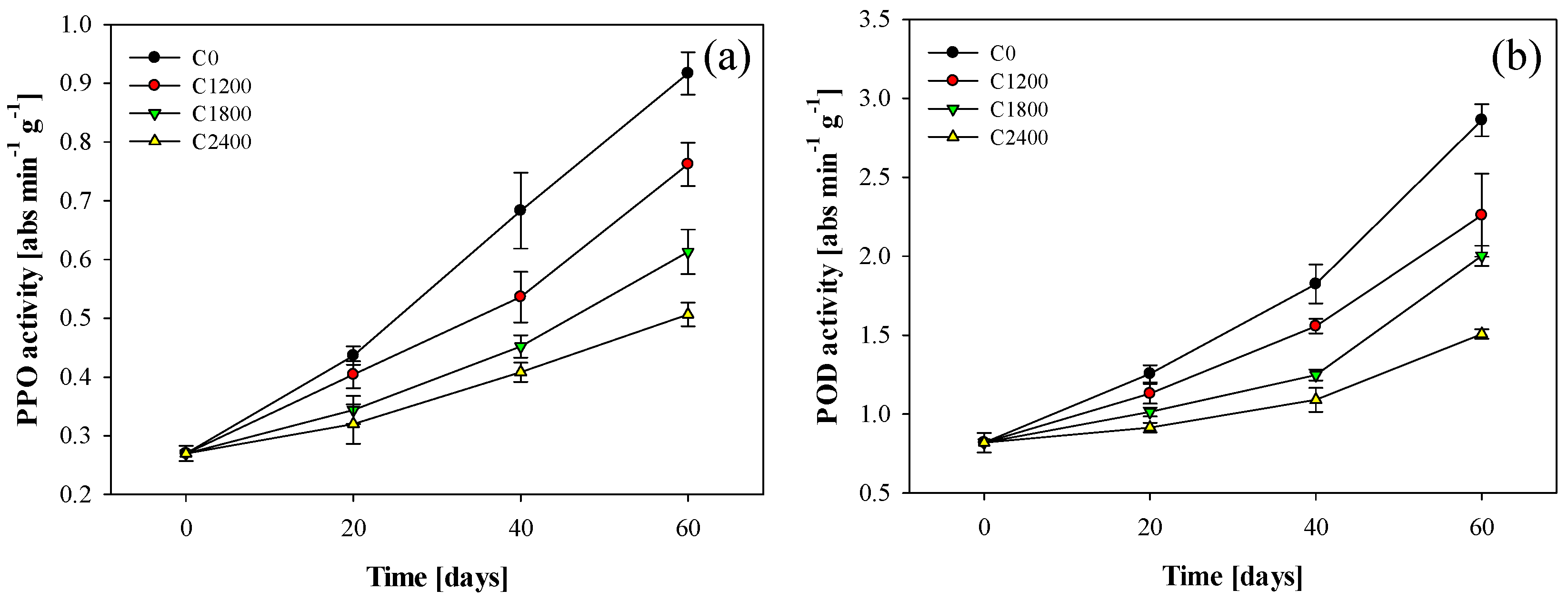
3.6. PPO and POD Activity
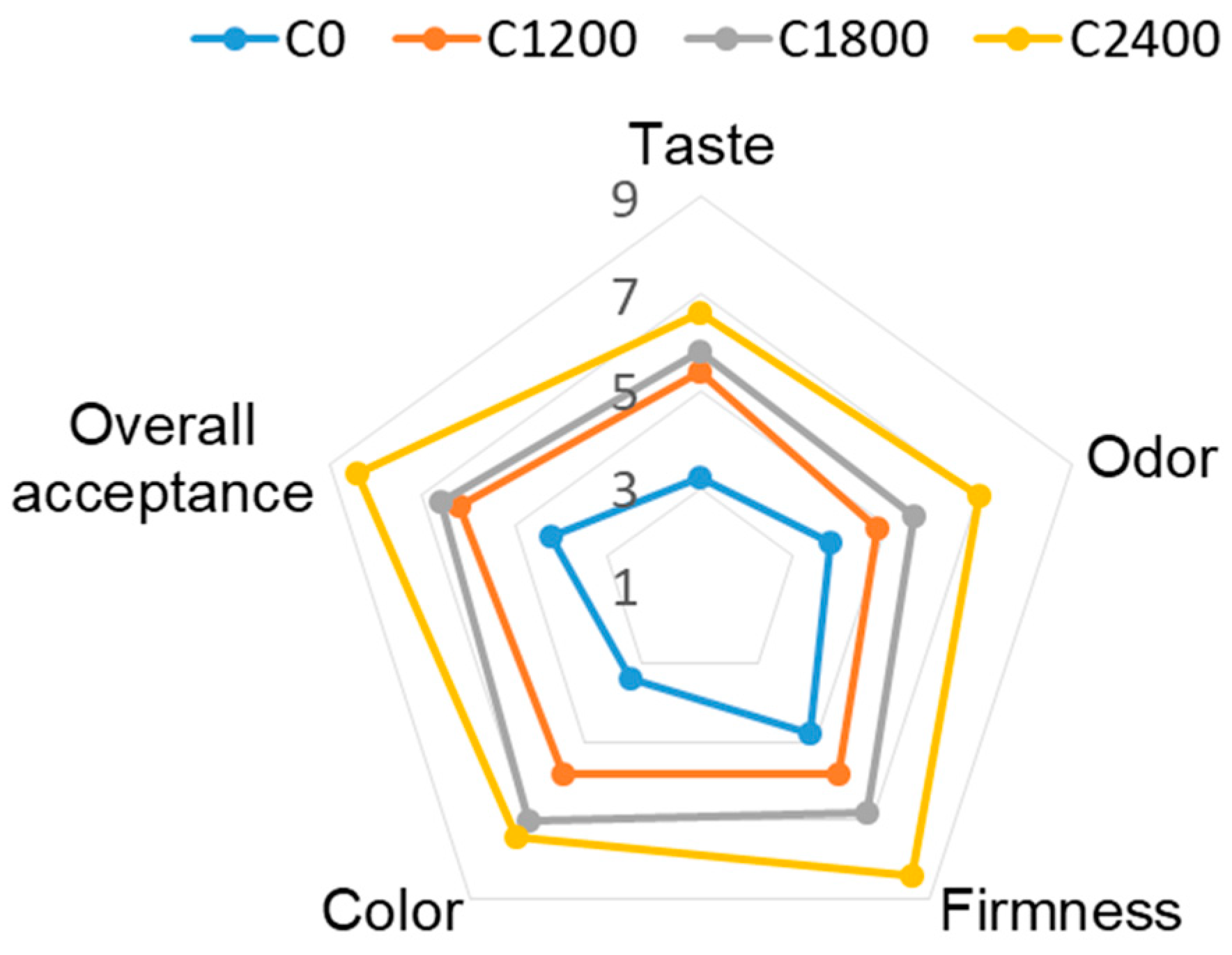
3.7. Sensory Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, L.Z.; Liu, L.; Holmes, J.; Kerry, J.F.; Kerry, J.P. Assessment of film-forming potential and properties of protein and polysaccharide-based biopolymer films. Int. J. Food Sci. Technol. 2007, 42, 1128–1138. [Google Scholar] [CrossRef]
- Mditshwa, A.; Fawole, O.A.; Opara, U.L. Recent developments on dynamic controlled atmosphere storage of apples—A review. Food Packag. Shelf Life 2018, 16, 59–68. [Google Scholar] [CrossRef]
- Prusky, D. Reduction of the incidence of postharvest quality losses, and future. Food Secur. 2011, 3, 463–474. [Google Scholar] [CrossRef]
- Sahraei Khosh Gardesh, A.; Badii, F.; Hashemi, M.; Ardakani, A.Y.; Maftoonazad, N.; Gorji, A.M. Effect of nanochitosan based coating on climacteric behavior and postharvest shelf-life extension of apple cv. Golab Kohanz. LWT 2016, 70, 33–40. [Google Scholar] [CrossRef]
- Maftoonazad, N.; Ramaswamy, H.S. Postharvest shelf-life extension of avocados using methyl cellulose-based coating. LWT 2005, 38, 617–624. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Tapia, M.S.; Martín-Belloso, O. Using polysaccharide-based edible coatings to maintain quality of fresh-cut Fuji apples. LWT 2008, 41, 139–147. [Google Scholar] [CrossRef]
- Maftoonazad, N.; Ramaswamy, H.S. Effect of pectin-based coating on the kinetics of quality change associated with stored avocados. J. Food Process. Preserv. 2008, 32, 621–643. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Abdou, E.S. Chitosan based edible films and coatings: A review. Mater. Sci. Eng. C 2013, 33, 1819–1841. [Google Scholar] [CrossRef]
- Fakhouri, F.M.; Martelli, S.M.; Caon, T.; Velasco, J.I.; Mei, L.H.I. Edible films and coatings based on starch/gelatin: Film properties and effect of coatings on quality of refrigerated Red Crimson grapes. Postharvest Biol. Technol. 2015, 109, 57–64. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Food applications of emulsion-based edible films and coatings. Trends Food Sci. Technol. 2015, 45, 273–283. [Google Scholar] [CrossRef]
- Salvia-Trujillo, L.; Rojas-Graü, M.A.; Soliva-Fortuny, R.; Martín-Belloso, O. Use of antimicrobial nanoemulsions as edible coatings: Impact on safety and quality attributes of fresh-cut Fuji apples. Postharvest Biol. Technol. 2015, 105, 8–16. [Google Scholar] [CrossRef]
- De León-Zapata, M.A.; Ventura-Sobrevilla, J.M.; Salinas-Jasso, T.A.; Flores-Gallegos, A.C.; Rodríguez-Herrera, R.; Pastrana-Castro, L.; Rua-Rodríguez, M.L.; Aguilar, C.N. Changes of the shelf life of candelilla wax/tarbush bioactive based-nanocoated apples at industrial level conditions. Sci. Hortic. 2018, 231, 43–48. [Google Scholar] [CrossRef]
- Ncama, K.; Magwaza, L.S.; Mditshwa, A.; Tesfay, S.Z. Plant-based edible coatings for managing postharvest quality of fresh horticultural produce: A review. Food Packag. Shelf Life 2018, 16, 157–167. [Google Scholar] [CrossRef]
- Sapper, M.; Palou, L.; Pérez-Gago, M.B.; Chiralt, A. Antifungal starch–gellan edible coatings with thyme essential oil for the postharvest preservation of apple and persimmon. Coatings 2019, 9, 333. [Google Scholar] [CrossRef]
- Sapper, M.; Martin-Esparza, M.E.; Chiralt, A.; Gonzalez Martinez, C. Antifungal polyvinyl alcohol coatings incorporating carvacrol for the postharvest preservation of golden delicious apple. Coatings 2020, 10, 1027. [Google Scholar] [CrossRef]
- Ali, U.; Basu, S.; Mazumder, K. Improved postharvest quality of apple (Rich Red) by composite coating based on arabinoxylan and β-glucan stearic acid ester. Int. J. Biol. Macromol. 2020, 151, 618–627. [Google Scholar] [CrossRef]
- Ali, U.; Kanwar, S.; Yadav, K.; Basu, S.; Mazumder, K. Effect of arabinoxylan and β-glucan stearic acid ester coatings on post-harvest quality of apple (Royal Delicious). Carbohydr. Polym. 2019, 209, 338–349. [Google Scholar] [CrossRef]
- de Oliveira Alves Sena, E.; Oliveira da Silva, P.S.; de Aragão Batista, M.C.; Alonzo Sargent, S.; Ganassali de Oliveira Junior, L.F.; Almeida Castro Pagani, A.; Gutierrez Carnelossi, M.A. Calcium application via hydrocooling and edible coating for the conservation and quality of cashew apples. Sci. Hortic. 2019, 256, 108531. [Google Scholar] [CrossRef]
- Rashid, F.; Ahmed, Z.; Ameer, K.; Amir, R.M.; Khattak, M. Optimization of polysaccharides-based nanoemulsion using response surface methodology and application to improve postharvest storage of apple (Malus domestica). J. Food Meas. Charact. 2020, 14, 2676–2688. [Google Scholar] [CrossRef]
- Zhang, W.; Li, X.; Jiang, W. Development of antioxidant chitosan film with banana peels extract and its application as coating in maintaining the storage quality of apple. Int. J. Biol. Macromol. 2020, 154, 1205–1214. [Google Scholar] [CrossRef]
- Thakur, R.; Pristijono, P.; Scarlett, C.J.; Bowyer, M.; Singh, S.P.; Vuong, Q.V. Starch-based edible coating formulation: Optimization and its application to improve the postharvest quality of “Cripps pink” apple under different temperature regimes. Food Packag. Shelf Life 2019, 22, 100409. [Google Scholar] [CrossRef]
- Madanipour, S.; Alimohammadi, M.; Rezaie, S.; Nabizadeh, R.; Jahed Khaniki, G.; Hadi, M.; Yousefi, M.; Madihi Bidgoli, S.; Yousefzadeh, S. Influence of postharvest application of chitosan combined with ethanolic extract of liquorice on shelflife of apple fruit. J. Environ. Health Sci. Eng. 2019, 17, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Jahanshahi, B.; Jafari, A.; Vazifeshenas, M.R.; Gholamnejad, J. A novel edible coating for apple fruits. JHPR 2018, 1, 63–72. [Google Scholar] [CrossRef]
- Khalifa, I.; Barakat, H.; El-Mansy, H.A.; Soliman, S.A. Preserving apple (Malus domestica var. anna) fruit bioactive substances using olive wastes extract-chitosan film coating. Inf. Process. Agric. 2017, 4, 90–99. [Google Scholar] [CrossRef]
- Razavi, M.S.; Golmohammadi, A.; Nematollahzadeh, A.; Fiori, F.; Rovera, C.; Farris, S. Preparation of cinnamon essential oil emulsion by bacterial cellulose nanocrystals and fish gelatin. Food Hydrocoll. 2020, 109, 106111. [Google Scholar] [CrossRef]
- Razavi, M.S.; Golmohammadi, A.; Nematollahzadeh, A.; Rovera, C.; Farris, S. Cinnamon essential oil encapsulated into a fish gelatin-bacterial cellulose nanocrystals complex and active films thereof. Food Biophys. 2021, 17, 38–46. [Google Scholar] [CrossRef]
- Jung, J.; Deng, Z.; Zhao, Y. Mechanisms and performance of cellulose nanocrystals Pickering emulsion chitosan coatings for reducing ethylene production and physiological disorders in postharvest ‘Bartlett’ pears (Pyrus communis L.) during cold storage. Food Chem. 2020, 309, 125693. [Google Scholar] [CrossRef]
- Souza Vieira da Silva, I.; Soares Prado, N.; Gontijo de Melo, P.; Campion Arantes, D.; Zeni Andrade, M.; Otaguro, H.; Pasquini, D. Edible coatings based on apple pectin, cellulose nanocrystals, and essential oil of lemongrass: Improving the quality and shelf life of strawberries (Fragaria ananassa). J. Renew. Mater. 2019, 7, 73–87. [Google Scholar] [CrossRef]
- Deng, Z.; Jung, J.; Simonsen, J.; Zhao, Y. Cellulose nanocrystals Pickering emulsion incorporated chitosan coatings for improving storability of postharvest Bartlett pears (Pyrus communis) during long-term cold storage. Food Hydrocoll. 2018, 84, 229–237. [Google Scholar] [CrossRef]
- Deng, Z.; Jung, J.; Simonsen, J.; Wang, Y.; Zhao, Y. Cellulose nanocrystal reinforced chitosan coatings for improving the storability of postharvest pears under both ambient and cold storages. J. Food Sci. 2017, 82, 453–462. [Google Scholar] [CrossRef]
- Fakhouri, F.M.; Casari, A.C.A.; Mariano, M.; Yamashita, F.; Mei, L.H.I.; Soldi, V.; Martelli, S.M. Effect of a gelatin-based edible coating containing cellulose nanocrystals (CNC) on the quality and nutrient retention of fresh strawberries during storage. IOP Conf. Ser. Mater. Sci. Eng. 2014, 64, 012024. [Google Scholar] [CrossRef]
- Dong, F.; Li, S.; Liu, Z.; Zhu, K.; Wang, X.; Jin, C. Improvement of quality and shelf life of strawberry with nanocellulose/chitosan composite coatings. Bangladesh J. Bot. 2015, 44, 709–717. [Google Scholar]
- Azeredo, H.M.C.; Miranda, K.W.E.; Ribeiro, H.L.; Rosa, M.F.; Nascimento, D.M. Nanoreinforced alginate–acerola puree coatings on acerola fruits. J. Food Eng. 2012, 113, 505–510. [Google Scholar] [CrossRef]
- Jung, J.; Deng, Z.; Zhao, Y. A review of cellulose nanomaterials incorporated fruit coatings with improved barrier property and stability: Principles and applications. J. Food Process Eng. 2019, 43, e13344. [Google Scholar] [CrossRef]
- Rovera, C.; Ghaani, M.; Santo, N.; Trabattoni, S.; Olsson, R.T.; Romano, D.; Farris, S. Enzymatic hydrolysis in the green production of bacterial cellulose nanocrystals. ACS Sustain. Chem. Eng. 2018, 6, 7725–7734. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 18th ed.; The Association Official Analytical Chemistry: Arelington, VA, USA, 2006. [Google Scholar]
- Fonseca, S.C.; Oliveira, F.A.R.; Brecht, J.K. Modelling respiration rate of fresh fruits and vegetables for modified atmosphere packages: A review. J. Food Eng. 2002, 52, 99–119. [Google Scholar] [CrossRef]
- Iqbal Durrani, A.; Hun Nasar, S.; Bashir, U.; Khan Durrani, A.; Munawar, A. Preparation, characterization and application of vitamin-e fortified nanocoatings on fresh-cut apples. J. Eng. Technol. 2014, 21, 46–59. [Google Scholar]
- Barrios, S.; Lema, P.; Lareo, C. Modeling Respiration Rate of Strawberry (cv. San Andreas) for Modified Atmosphere Packaging Design. Int. J. Food Prop. 2014, 17, 2039–2051. [Google Scholar] [CrossRef]
- Mendoza, R.; Castellanos, D.A.; García, J.C.; Vargas, J.C.; Herrera, A.O. Ethylene production, respiration and gas exchange modelling in modified atmosphere packaging for banana fruits. Int. J. Food Sci. Technol. 2016, 51, 777–788. [Google Scholar] [CrossRef]
- Imanpanah, H.; Kasraei, M.; Raoufat, M.H.; Nejadi, J. Development and evaluation of a portable apparatus for bioyield detection: A case study with apple and peach fruits. Int. J. Food Prop. 2015, 18, 1434–1445. [Google Scholar] [CrossRef]
- Sadler, G.D.; Murphy, P.A. pH and titratable acidity. In Food Analysis, 4th ed.; Nielsen, S.S., Ed.; Springer: New York, NY, USA, 2010; pp. 219–238. [Google Scholar] [CrossRef]
- Eshghi, S.; Hashemi, M.; Mohammadi, A.; Badii, F.; Mohammadhoseini, Z.; Ahmadi, K. Effect of nanochitosan-based coating with and without copper loaded on physicochemical and bioactive components of fresh strawberry fruit (Fragaria x ananassa Duchesne) during storage. Food Bioproc. Technol. 2014, 7, 2397–2409. [Google Scholar] [CrossRef]
- Terefe, N.; Matthies, K.; Simons, L.; Versteeg, C. Combined high pressure-mild temperature processing for optimal retention of physical and nutritional quality of strawberries (Fragaria ananassa). Innov. Food Sci. Emerg. Technol. 2009, 10, 97–307. [Google Scholar] [CrossRef]
- Meilgaard, M.; Civille, G.V.; Carr, B.T. Overall difference tests: Does a sensory difference exist between samples. In Sensory Evaluation Techniques, 3rd ed.; Meilgaard, M., Civille, G.V., Carr, B.T., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 63–104. [Google Scholar] [CrossRef]
- Mangaraj, S.; Sadawarti, M.J.; Prasad, S. Assessment of quality of pears stored in laminated modified atmosphere packages. Int. J. Food Prop. 2011, 14, 1110–1123. [Google Scholar] [CrossRef]
- Deng, Z.; Jung, J.; Simonsen, J.; Zhao, Y. Cellulose nanomaterials emulsion coatings for controlling physiological activity, modifying surface morphology, and enhancing storability of postharvest bananas (Musa acuminate). Food Chem. 2017, 232, 359–368. [Google Scholar] [CrossRef]
- Valero, D.; Díaz-Mula, H.M.; Zapata, P.J.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M. Effects of alginate edible coating on preserving fruit quality in four plum cultivars during postharvest storage. Postharvest Biol. Technol. 2013, 77, 1–6. [Google Scholar] [CrossRef]
- Cosgrove, D.J. Expansive growth of plant cell walls. Plant Physiol. Biochem. 2000, 38, 109–124. [Google Scholar] [CrossRef]
- Brummell, D.A.; Harpster, M.H. Cell wall metabolism in fruit softening and quality and its manipulation in transgenic plants. Plant Mol. Biol. 2001, 47, 311–339. [Google Scholar] [CrossRef]
- Dabesor, P.A.; Sanni, D.M.; Kolawole, A.O.; Enujiugha, V.N.; Lawal, O.T.; Edeh, A.T. Changes in physicochemical properties and enzymes associated with ripening of snake tomato (Trichosanthes Cucumerina L.) fruit. Biocatal. Agric. Biotechnol. 2022, 40, 102313. [Google Scholar] [CrossRef]
- López_Gómez, A.; Navarro-Martínez, A.; Martínez-Hernández, G.B. Active paper sheets including nanoencapsulated essential oils: A green packaging technique to control ethylene production and maintain quality in fresh horticultural products—A case study on flat peaches. Foods 2020, 9, 1904. [Google Scholar] [CrossRef]
- Han, C.; Zhao, Y.; Leonard, S.W.; Traber, M.G. Edible coatings to improve storability and enhance nutritional value of fresh and frozen strawberries (Fragaria x ananassa) and raspberries. Postharvest Biol. Technol. 2004, 33, 67–78. [Google Scholar] [CrossRef]
- Zambrano-Zaragoza, M.L.; Mercado-Silva, E.; Del Real L, A.; Gutiérrez-Cortez, E.; Cornejo-Villegas, M.A.; Quintanar-Guerrero, D. The effect of nano-coatings with α-tocopherol and xanthan gum on shelf-life and browning index of fresh-cut “Red Delicious” apples. Innov. Food Sci. Emerg. Technol. 2014, 22, 188–196. [Google Scholar] [CrossRef]
- Pourdarbani, R.; Sabzi, S.; Kalantari, D.; Karimzadeh, R.; Ilbeygi, E.; Arribas, J.I. Automatic non-destructive video estimation of maturation levels in Fuji apple (Malus Malus pumila) fruit in orchard based on colour (Vis) and spectral (NIR) data. Biosyst. Eng. 2020, 195, 136–151. [Google Scholar] [CrossRef]
- Bodbodak, S.; Rafiee, Z. Recent trends in active packaging in fruits and vegetables. In Eco-Friendly Technology for Postharvest Produce Quality; Siddiqui, M.W., Ed.; Academic Press Inc.: Cambridge, MA, USA, 2016; pp. 77–125. [Google Scholar]
- Ahlawat, Y.; Liu, T. Varied Expression of senescence-associated and ethylene-related genes during postharvest storage of Brassica vegetables. Int. J. Mol. Sci. 2021, 22, 839. [Google Scholar] [CrossRef]
- Watkins, C.B. Principles and Practices of Postharvest Handling and Stress. In Apples: Botany, Production and Uses, 1st ed.; Ferree, D.C., Warrington, I.J., Eds.; CAB Publishing: Chesterfield, UK, 2003; pp. 585–614. [Google Scholar] [CrossRef]
- Thammawong, M.; Arakawa, O. Starch to sugar conversion in “tsugaru” apples under ethylene and 1-methylcyclopropene treatments. J. Agric. Sci. Technol. 2010, 12, 617–626. [Google Scholar]
- Tanda-Palmu, P.S.; Grosso, C.R.F. Effect of edible wheat gluten-based films and coatings on refrigerated strawberry (Fragaria ananassa) quality. Postharvest Biol. Technol. 2005, 36, 199–208. [Google Scholar] [CrossRef]
- Treviño-Garza, M.Z.; García, S.; del Socorro Flores-González, M.; Arévalo-Niño, K. Edible active coatings based on pectin, pullulan, and chitosan increase quality and shelf life of strawberries (Fragaria ananassa). J. Food Sci. 2015, 80, 1823–1830. [Google Scholar] [CrossRef] [PubMed]
- Conforti, F.D.; Totty, J.A. Effect of three lipid/hydrocolloid coatings on shelf-life stability of Golden Delicious apples. Int. J. Food Sci. Technol. 2007, 42, 1101–1106. [Google Scholar] [CrossRef]
- Sumedrea, D.; Florea, A.; Sumedrea, M.; Asănica, A.; Coman, R.; Militaru, M.; Chiţu, E.; Butac, M.; Marin, F.C.; Calinescu, M. Influence of different storage methods on apples chemical proprieties. Agric. Life 2018, 1, 316–321. [Google Scholar] [CrossRef][Green Version]
- Aslmoshtaghi, E.; Shahsavar, A. Peroxidase, polyphenol oxidase and protein changes in olives during adventitious root formation. Trakia J. Sci. 2016, 14, 176–182. [Google Scholar] [CrossRef]
- Deutch, C.E. Browning in apples: Exploring the biochemical basis of an easily-observable phenotype. Biochem. Mol. Biol. Educ. 2017, 46, 76–82. [Google Scholar] [CrossRef]
- Hutabarat, O.S.; Halbwirth, H. Polyphenol oxidase and peroxidase activity in apple: Dependency on cultivar and fruit processing. IOP Conf. Ser. Earth Environ. Sci. 2019, 355, 012106. [Google Scholar] [CrossRef]
- Minibayeva, F.; Beckett, R.P.; Kranner, I. Roles of apoplastic peroxidases in plant response to wounding. Phytochemistry 2015, 112, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, K.S.; de Andrade, M.T.; Coelho, D.G.; de Sousa, A.E.D.; de Melo Neto, D.F.; de Brito, F.A.L.; da Silva, R.M.; do Nascimento Simões, A. Baby Cassava: An Alternative Marketing Strategy for Freshly Cut Cassava. In Cassava, 1st ed.; Waisundara, V.Y., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Othman, O.C. Polyphenoloxidase and perioxidase activity during open air ripening storage of pineapple (Ananas comosus L.), mango (Mangifera indica) and papaya (Carica papaya) fruits grown in Dar es Salaam, Tanzania. Tanz. J. Sci. 2012, 38, 84–94. [Google Scholar]
- Buendía−Moreno, L.; Sánchez−Martínez, M.J.; Antolinos, V.; Ros−Chumillas, M.; Navarro−Segura, L.; Soto−Jover, S.; Martínez−Hernández, G.B.; López−Gómez, A. Active cardboard box with a coating including essential oils entrapped within cyclodextrins and/or halloysite nanotubes. A case study for fresh tomato storage. Food Control 2020, 107, 106763. [Google Scholar] [CrossRef]
- Can, Z.; Dincer, B.; Sahin, H.; Baltas, N.; Yildiz, O.; Kolayli, S. Polyphenol oxidase activity and antioxidant properties of Yomra apple (Malus Communis L.) from Turkey. J. Enzym. Inhib. Med. Chem. 2014, 29, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Jameel Jhalegar, M.D.; Sharma, R.R.; Singh, D. In vitro and In vivo activity of essential oils against major postharvest pathogens of Kinnow (Citrus nobilis × C. deliciosa) mandarin. J. Food Sci. Technol. 2015, 52, 2229–2237. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, R.; ValizadehKaji, B.; Khadivi, A.; Shahrjerdi, I. Effect of chitosan and thymol essential oil on quality maintenance and shelf life extension of peach fruits cv. ‘Zaferani’. J. Hortic. Postharvest Res. 2019, 2, 143–156. [Google Scholar] [CrossRef]
- Valente, J.; Almeida, R.; Kooistra, L. A Comprehensive Study of the Potential Application of Flying Ethylene-Sensitive Sensors for Ripeness Detection in Apple Orchards. Sensors 2019, 19, 372. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sadat Razavi, M.; Golmohammadi, A.; Nematollahzadeh, A.; Ghanbari, A.; Davari, M.; Rovera, C.; Carullo, D.; Farris, S. Impact of Bacterial Cellulose Nanocrystals-Gelatin/Cinnamon Essential Oil Emulsion Coatings on the Quality Attributes of ‘Red Delicious’ Apples. Coatings 2022, 12, 741. https://doi.org/10.3390/coatings12060741
Sadat Razavi M, Golmohammadi A, Nematollahzadeh A, Ghanbari A, Davari M, Rovera C, Carullo D, Farris S. Impact of Bacterial Cellulose Nanocrystals-Gelatin/Cinnamon Essential Oil Emulsion Coatings on the Quality Attributes of ‘Red Delicious’ Apples. Coatings. 2022; 12(6):741. https://doi.org/10.3390/coatings12060741
Chicago/Turabian StyleSadat Razavi, Mahsa, Abdollah Golmohammadi, Ali Nematollahzadeh, Alireza Ghanbari, Mahdi Davari, Cesare Rovera, Daniele Carullo, and Stefano Farris. 2022. "Impact of Bacterial Cellulose Nanocrystals-Gelatin/Cinnamon Essential Oil Emulsion Coatings on the Quality Attributes of ‘Red Delicious’ Apples" Coatings 12, no. 6: 741. https://doi.org/10.3390/coatings12060741
APA StyleSadat Razavi, M., Golmohammadi, A., Nematollahzadeh, A., Ghanbari, A., Davari, M., Rovera, C., Carullo, D., & Farris, S. (2022). Impact of Bacterial Cellulose Nanocrystals-Gelatin/Cinnamon Essential Oil Emulsion Coatings on the Quality Attributes of ‘Red Delicious’ Apples. Coatings, 12(6), 741. https://doi.org/10.3390/coatings12060741







