Fabrication by Spin-Coating and Optical Characterization of Poly(styrene-co-acrylonitrile) Thin Films
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication and Processing
2.3. Characterization
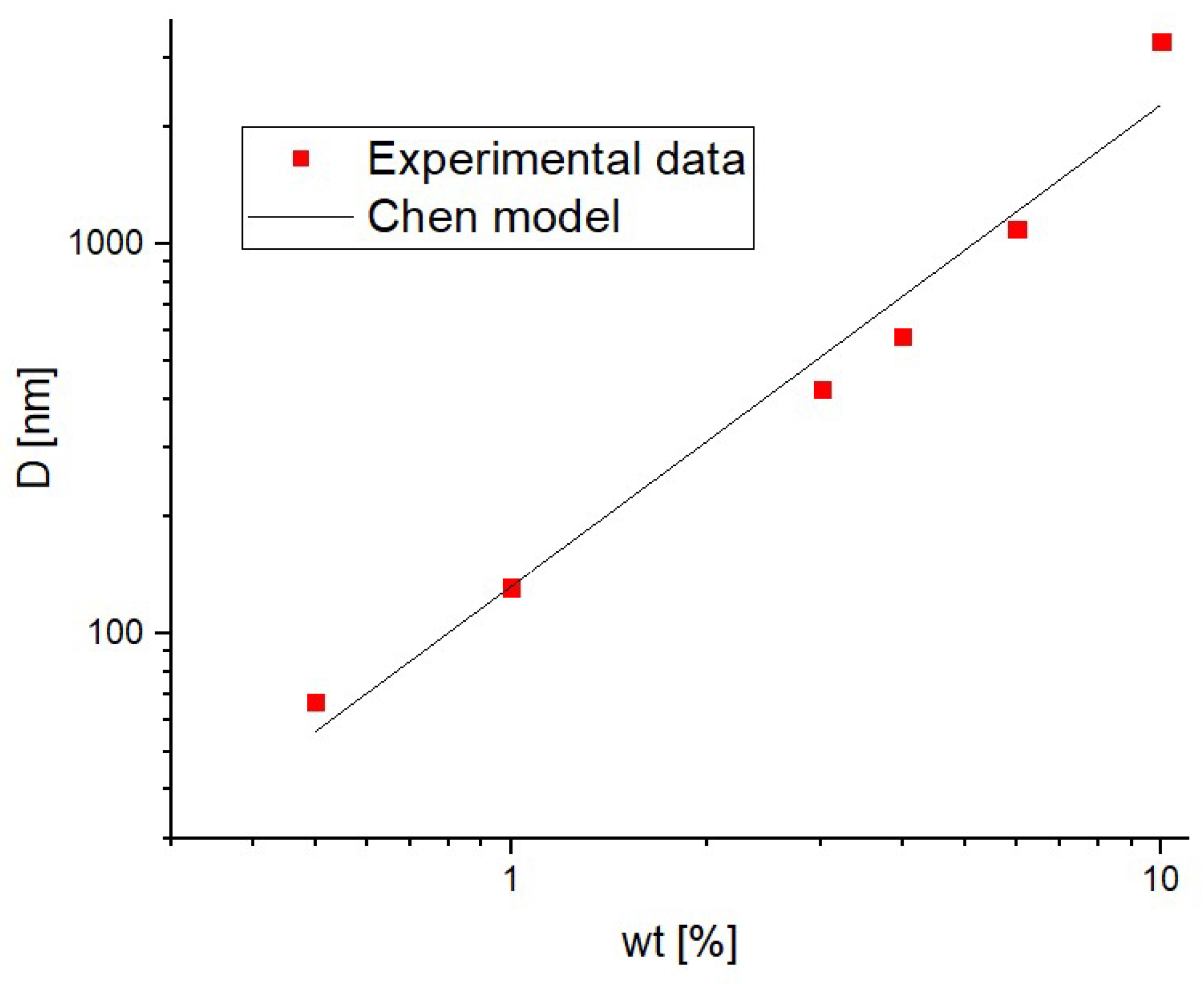
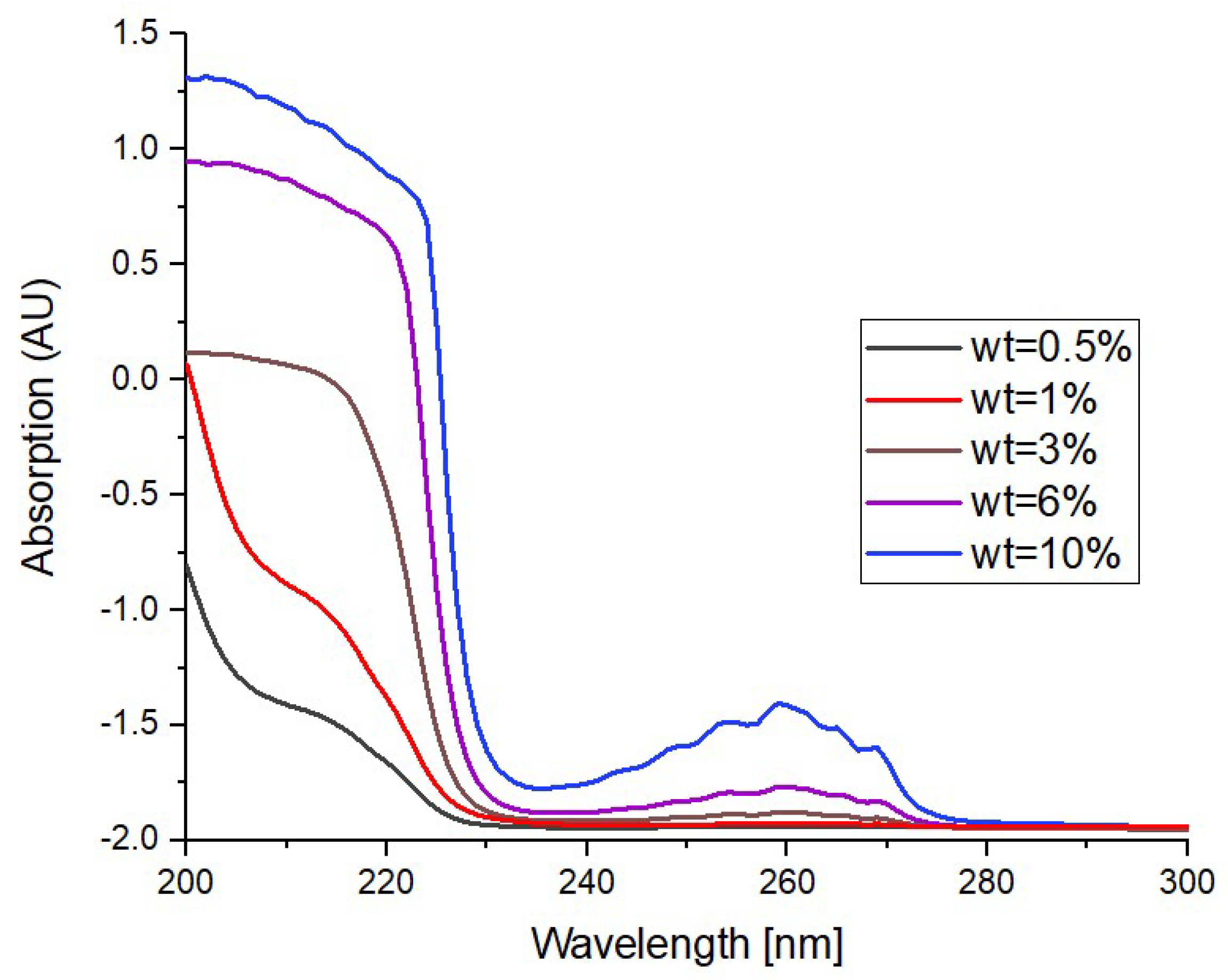
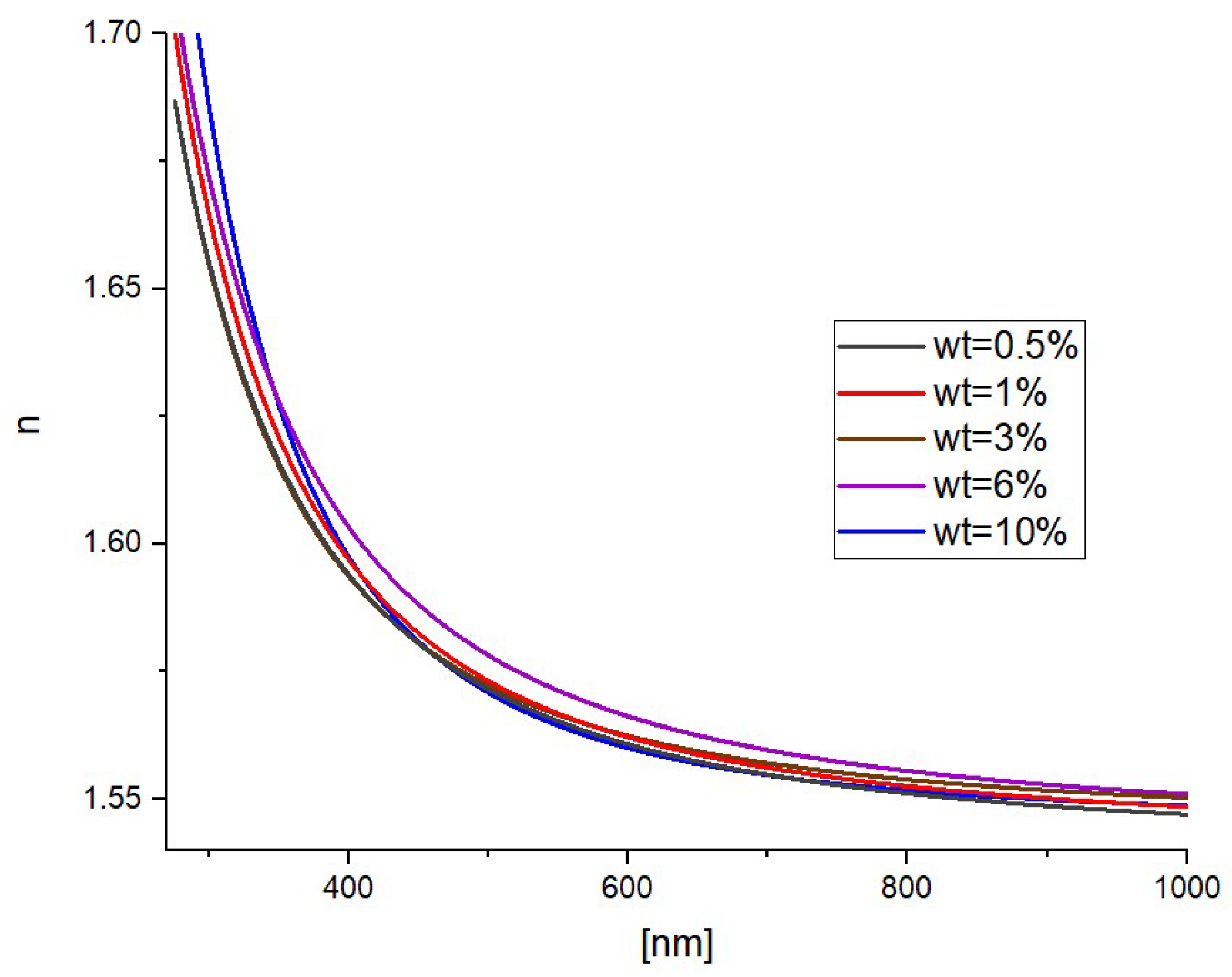
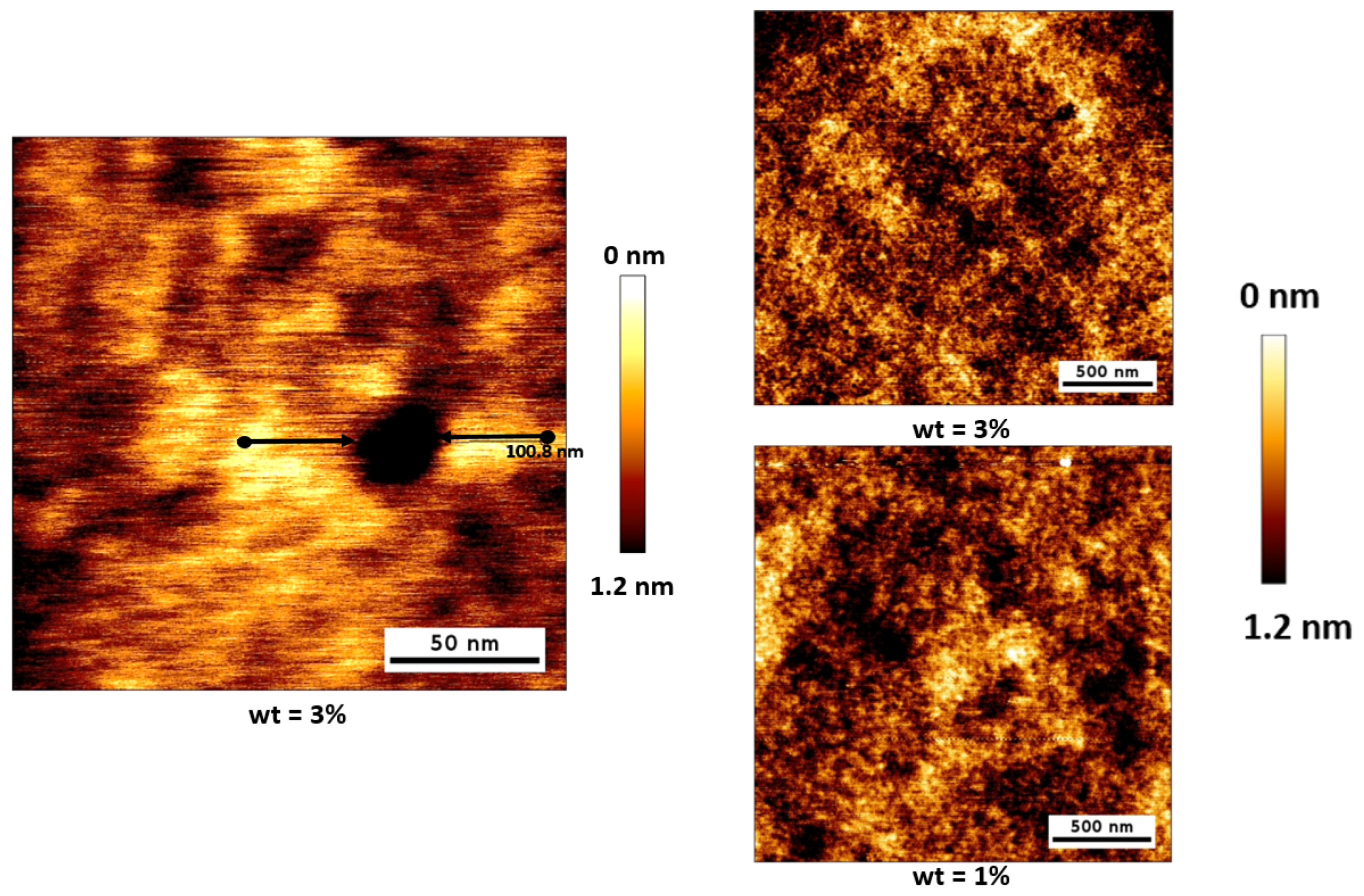
3. Results and Discussion
3.1. As Fabricated Samples
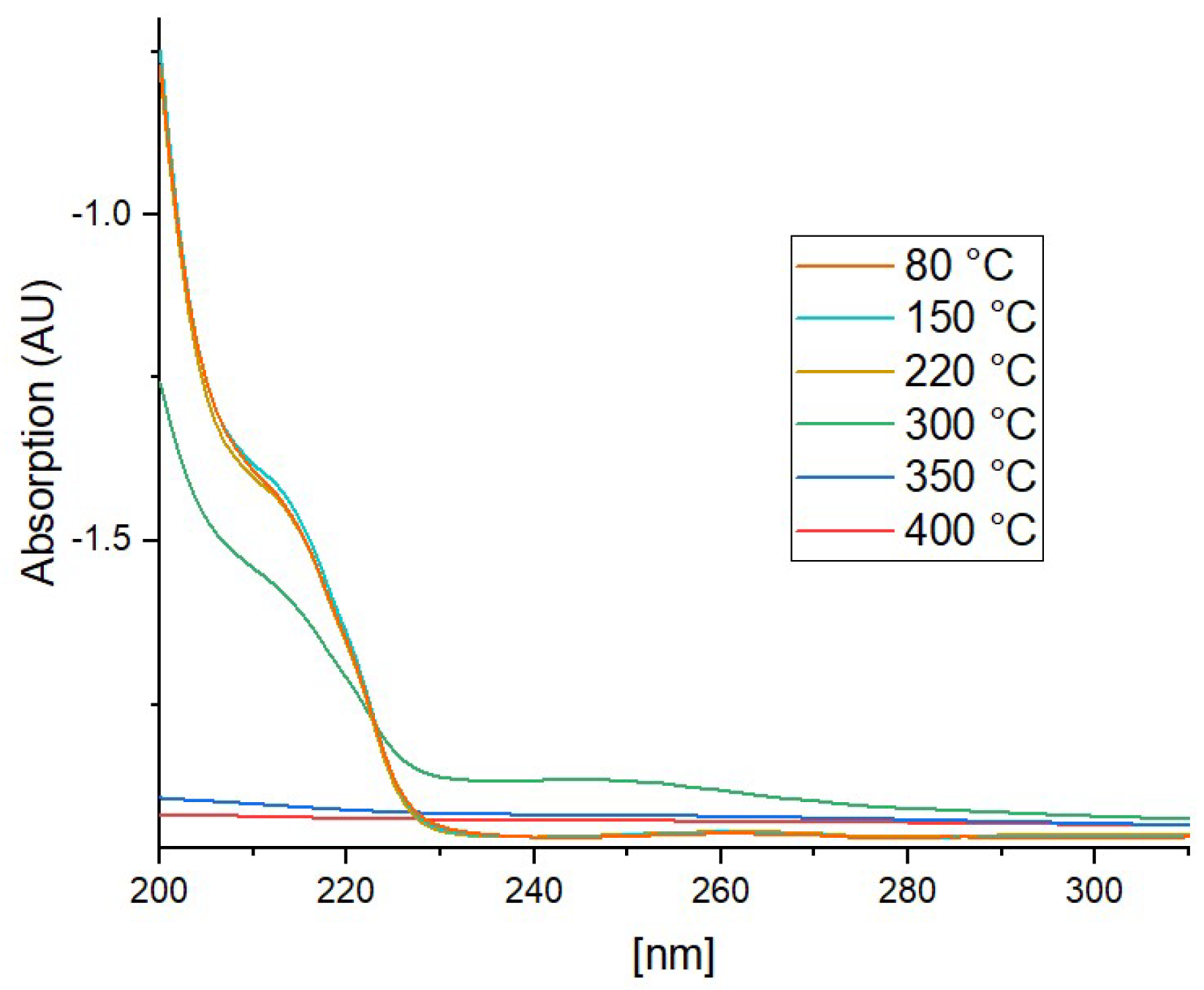
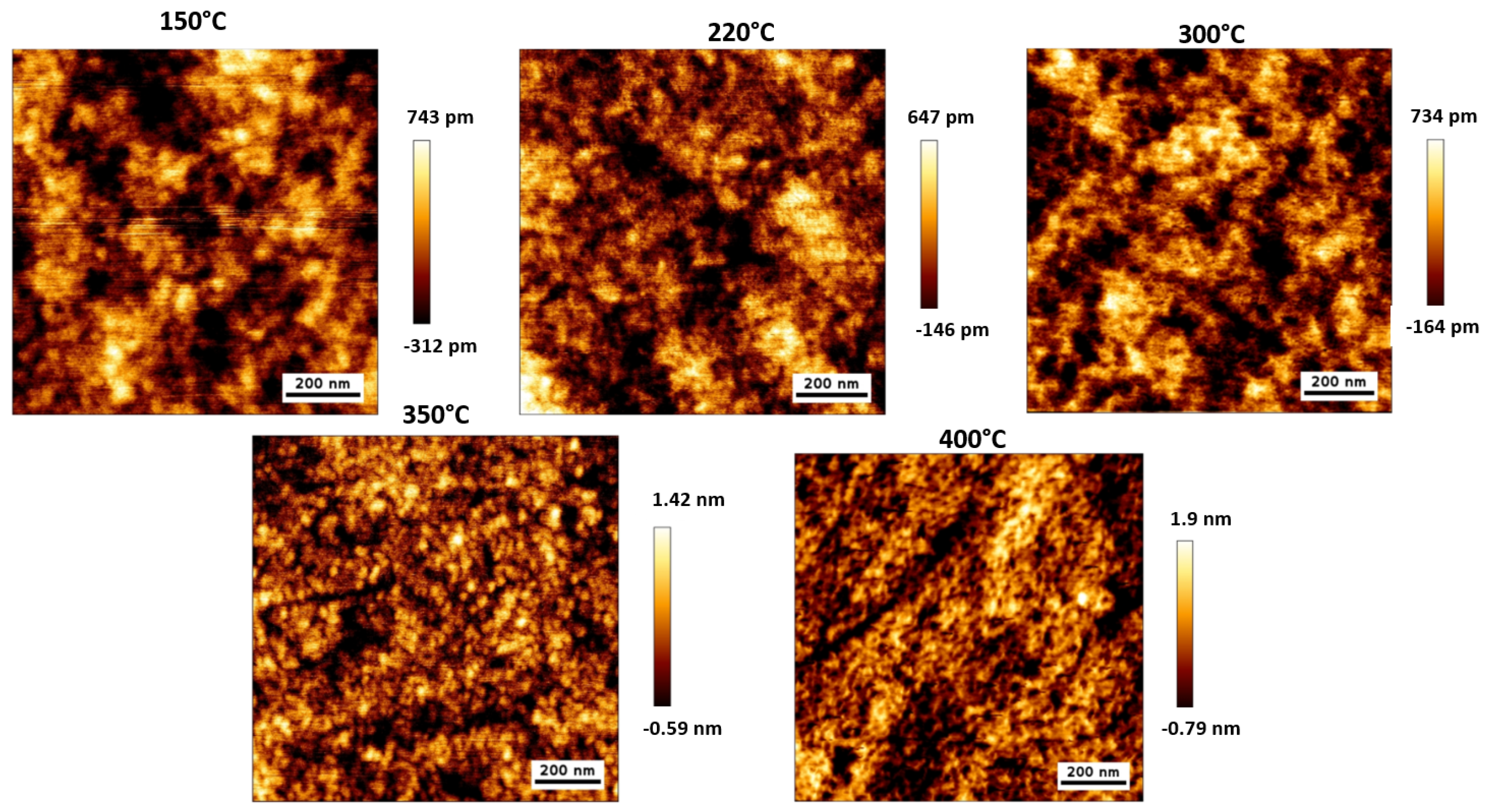
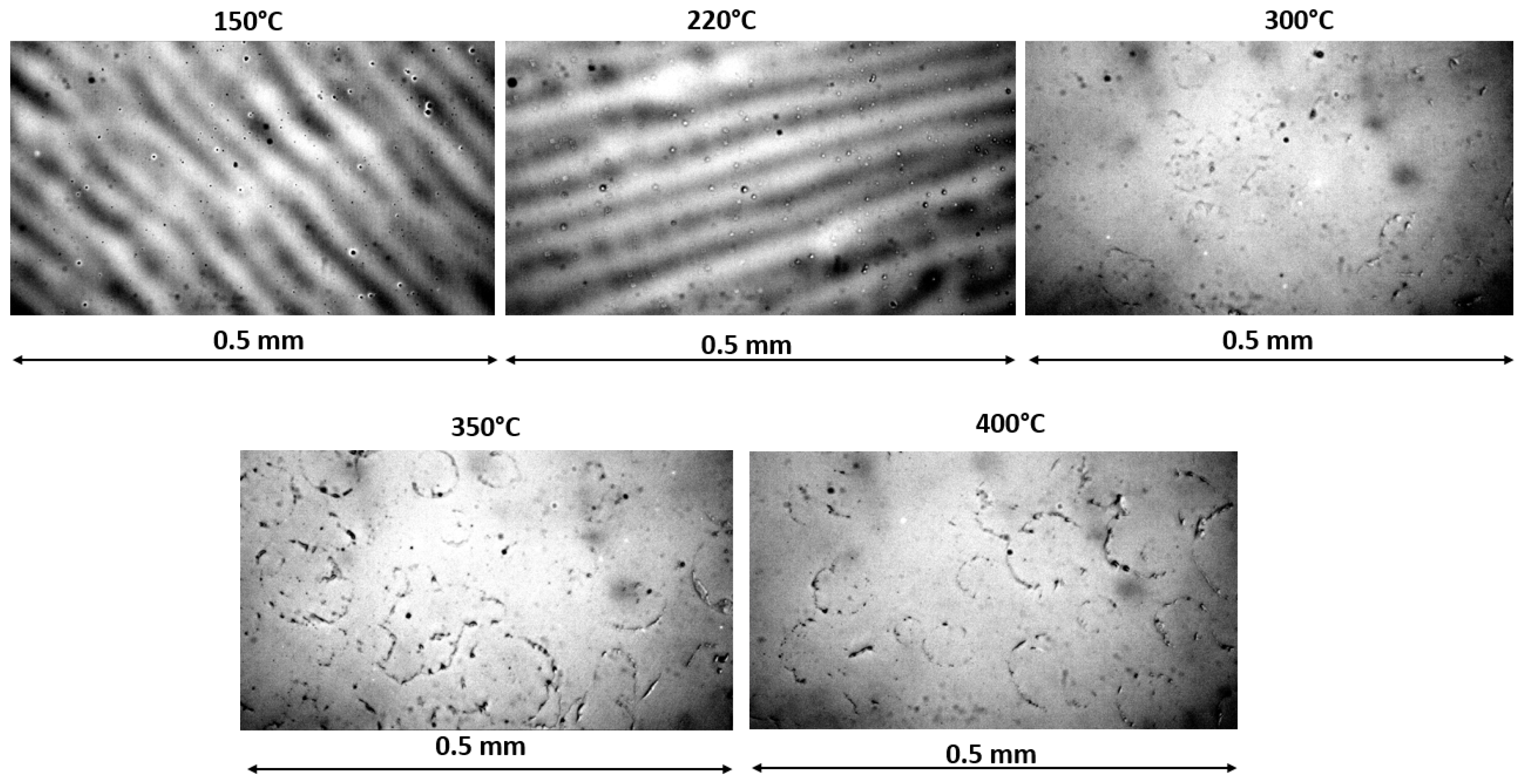
3.2. Effect of Thermal Annealing
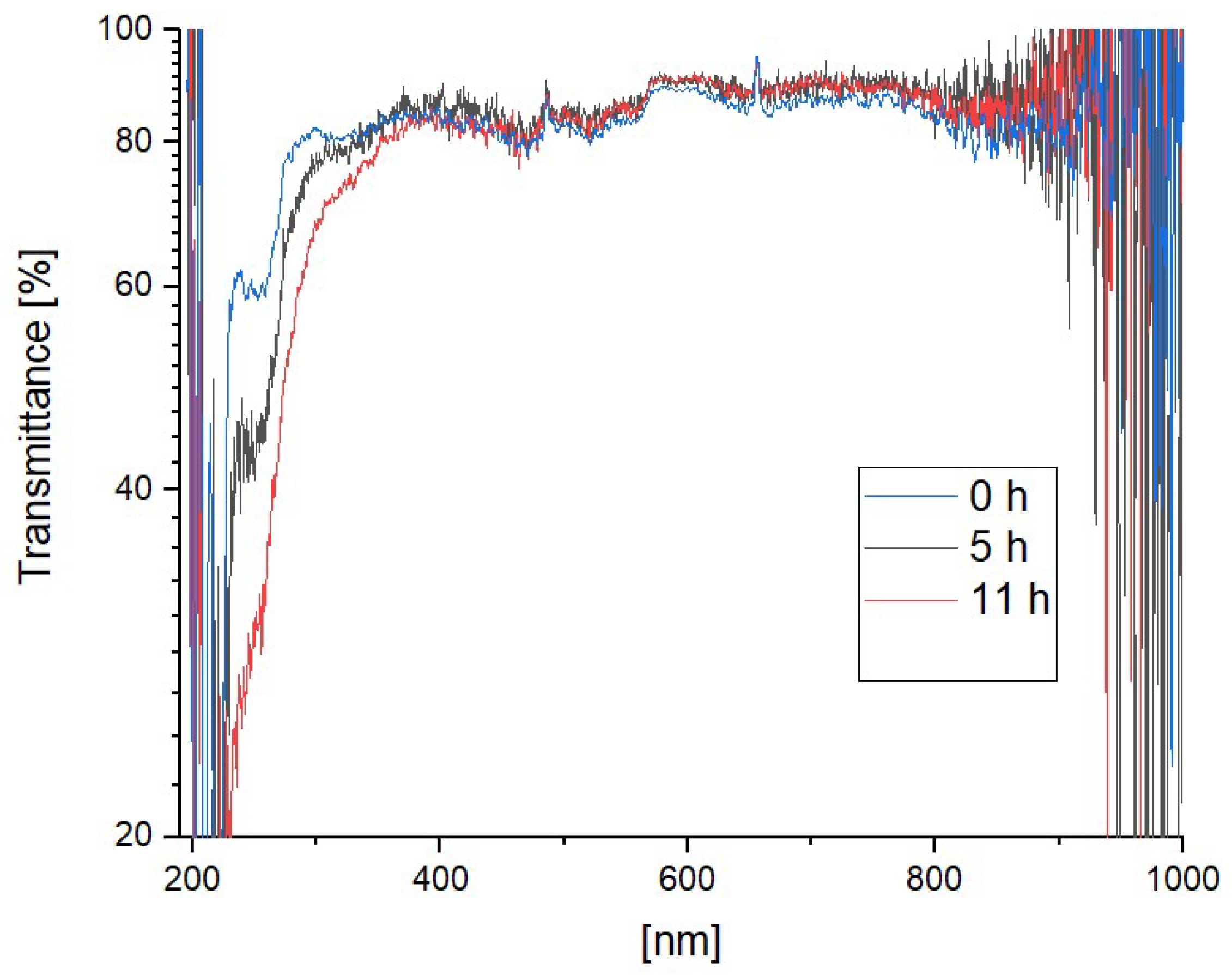
3.3. Photodegradation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Knausz, M.; Oreski, G.; Schmidt, M.; Guttmann, P.; Berger, K.; Voronko, Y.; Eder, G.; Koch, T.; Pinter, G. Thermal expansion behavior of solar cell encapsulation materials. Polym. Test. 2015, 44, 160–167. [Google Scholar] [CrossRef]
- Peike, C.; Hädrich, I.; Weiß, K.A.; Dürr, I.; Ise, F. Overview of PV module encapsulation materials. Photovoltaics Int. 2013, 19, 85–92. [Google Scholar]
- Hussein, A.M.; Dannoun, E.; Aziz, S.B.; Brza, M.A.; Abdulwahid, R.T.; Hussen, S.A.; Rostam, S.; Mustafa, D.M.; Muhammad, D.S. Steps toward the band gap identification in polystyrene based solid polymer nanocomposites integrated with tin titanate nanoparticles. Polymers 2020, 12, 2320. [Google Scholar] [CrossRef] [PubMed]
- Rochat, S.; Swager, T.M. Conjugated amplifying polymers for optical sensing applications. ACS Appl. Mater. Interfaces 2013, 5, 4488–4502. [Google Scholar] [CrossRef]
- Moliton, A.; Hiorns, R.C. Review of electronic and optical properties of semiconducting π-conjugated polymers: Applications in optoelectronics. Polym. Int. 2004, 53, 1397–1412. [Google Scholar] [CrossRef]
- El-Aassar, M.R.; Masoud, M.S.; Elkady, M.F.; Elzain, A.A. Synthesis, optimization, and characterization of poly (Styrene-co-Acrylonitrile) copolymer prepared via precipitation polymerization. Adv. Polym. Technol. 2018, 37, 2021–2029. [Google Scholar] [CrossRef]
- Mailhot, B.; Gardette, J.L. Mechanism of poly (styrene-co-acrylonitrile) photooxidation. Polym. Degrad. Stab. 1994, 44, 237–247. [Google Scholar] [CrossRef]
- Cho, K.; Lee, J.; Xing, P. Enzymatic degradation of blends of poly (ε-caprolactone) and poly (styrene-co-acrylonitrile) by Pseudomonas lipase. J. Appl. Polym. Sci. 2002, 83, 868–879. [Google Scholar] [CrossRef]
- Bedjaoui, K.; Krache, R.; Marcos-Fernández, A.; Guessoum, M. The effect of compatibilizer SEBS on the mechanical, morphological and thermal properties of the polystyrene/poly (styrene-co-acrylonitrile) copolymer blends. Mater. Res. Express 2019, 6, 105334. [Google Scholar] [CrossRef]
- Cárdenas, T.G.; Acuña, E.J.; Carbacho, H.H.; Rodríguez, B.M.; Tagle, D.L.H. Thermal Studies of Metal Poly (Styrene-Co-Acrylonitrile). Part 14. Int. J. Polym. Mater. 1994, 26, 199–206. [Google Scholar] [CrossRef]
- Mu, S.Y.; Guo, J.; Gong, Y.M.; Zhang, S.; Yu, Y. Synthesis and thermal properties of poly (styrene-co-acrylonitrile)-graft-polyethylene glycol copolymers as novel solid–solid phase change materials for thermal energy storage. Chin. Chem. Lett. 2015, 26, 1364–1366. [Google Scholar] [CrossRef]
- Benali, S.; Olivier, A.; Brocorens, P.; Bonnaud, L.; Alexandre, M.; Bourbigot, S.; Espuche, E.; Gouanve, F.; Lazzaroni, R.; Dubois, P. Fire and gas barrier properties of poly (styrene-co-acrylonitrile) nanocomposites using polycaprolactone/clay nanohybrid based-masterbatch. Adv. Mater. Sci. Eng. 2008, 2008, 394235. [Google Scholar] [CrossRef] [Green Version]
- Wen, F.; Xu, Z.; Xia, W.; Ye, H.; Wei, X.; Zhang, Z. High-energy-density poly (styrene-co-acrylonitrile) thin films. J. Electron. Mater. 2013, 42, 3489–3493. [Google Scholar] [CrossRef]
- Khan, Z.; Baloch, M.K. Investigating the miscibility of polystyrene/poly (styrene-co-acrylonitrile) blend in tetrahydrofuran by simple physical techniques at various temperatures. Polym. Bull. 2013, 70, 2015–2033. [Google Scholar] [CrossRef]
- Svoboda, P.; Svobodova, D.; Chiba, T.; Inoue, T. Competition of phase dissolution and crystallization in poly (ε-caprolactone)/poly (styrene-co-acrylonitrile) blend. Eur. Polym. J. 2008, 44, 329–341. [Google Scholar] [CrossRef]
- Ghawana, K.; Singh, S.; Tripathi, K. Determination of waveguide parameters of acrylonitrile-based polymer optical waveguides. J. Opt. 1998, 29, 265. [Google Scholar] [CrossRef]
- Yang, J.; Cao, Q.; He, Z.; Pu, X.; Li, T.; Gao, B.; Li, X. The poly (styrene-co-acrylonitrile) polymer assisted preparation of high-performance inverted perovskite solar cells with efficiency exceeding 22%. Nano Energy 2021, 82, 105731. [Google Scholar] [CrossRef]
- Wen, G.; Li, X.; Liao, Y.; An, L. Surface phase separations of PMMA/SAN blends investigated by atomic force microscopy. Polymer 2003, 44, 4035–4045. [Google Scholar] [CrossRef]
- Yukioka, S.; Inoue, T. Ellipsometric studies on immiscible polymer-polymer interfaces. Polymer 1993, 34, 1256–1259. [Google Scholar] [CrossRef]
- Reiter, G. Dewetting of thin polymer films. Phys. Rev. Lett. 1992, 68, 75. [Google Scholar] [CrossRef]
- Ammar, M.; Napierala, C.; Laffez, P. Infrared thermochromic behaviour of a composite Sm0. 65Ca0. 35MnO3–poly (styrene-co-acrylonitrile) film. Smart Mater. Struct. 2009, 18, 055002. [Google Scholar] [CrossRef]
- El Wafa, A.M.A.; Okada, S.; Nakanishi, H. Poling and its relaxation studies of polycarbonate and poly (styrene-co-acrylonitrile) doped by a nonlinear optical chromophore. Dyes Pigment. 2006, 69, 239–244. [Google Scholar] [CrossRef]
- Hsu, W.P. Effect of tacticity of poly (methyl methacrylate) on the miscibility with poly (styrene-co-acrylonitrile). J. Appl. Polym. Sci. 1999, 74, 2894–2899. [Google Scholar]
- Chen, B. Investigation of the solvent-evaporation effect on spin coating of thin films. Polym. Eng. Sci. 1983, 23, 399–403. [Google Scholar] [CrossRef]
- Weill, A.; Dechenaux, E. The spin-coating process mechanism related to polymer solution properties. Polym. Eng. Sci. 1988, 28, 945–948. [Google Scholar] [CrossRef]
- Lawrence, C. The mechanics of spin coating of polymer films. Phys. Fluids 1988, 31, 2786–2795. [Google Scholar] [CrossRef]
- Tyona, M. A theoritical study on spin coating technique. Adv. Mater. Res. 2013, 2, 195. [Google Scholar] [CrossRef] [Green Version]
- Hall, D.B.; Underhill, P.; Torkelson, J.M. Spin coating of thin and ultrathin polymer films. Polym. Eng. Sci. 1998, 38, 2039–2045. [Google Scholar] [CrossRef]
- Nosidlak, N.; Dulian, P.; Mierzwiński, D.; Jaglarz, J. The Determination of the Electronic Parameters of Thin Amorphous Organic Films by Ellipsometric and Spectrophotometric Study. Coatings 2020, 10, 980. [Google Scholar] [CrossRef]
- Horcas, I.; Fernández, R.; Gomez-Rodriguez, J.; Colchero, J.; Gómez-Herrero, J.; Baro, A. WSXM: A software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 2007, 78, 013705. [Google Scholar] [CrossRef] [PubMed]
- Chapman, N.; Chapman, M.; Euler, W.B. Modeling of Poly (methylmethacrylate) Viscous Thin Films by Spin-Coating. Coatings 2021, 11, 198. [Google Scholar] [CrossRef]
- Li, T.; Zhou, C.; Jiang, M. UV absorption spectra of polystyrene. Polym. Bull. 1991, 25, 211–216. [Google Scholar] [CrossRef]
- Rasmussen, S. Low-bandgap polymers. In Encyclopedia of Polymeric Nanomaterials; Springer: Berlin/Heidelberg, Germany, 2015; pp. 1155–1166. [Google Scholar]
- Hansen, C. The Three Dimensional Solubility Parameter and Solvent Diffusion Coefficient; Danish Technical Press: Copenhagen, Denmark, 1967. [Google Scholar]
- Anandhan, S.; De, P.; De, S.; Bandyopadhyay, S.; Bhowmick, A. Mapping of thermoplastic elastomeric nitrile rubber/poly (styrene-co-acrylonitrile) blends using tapping mode atomic force microscopy and transmission electron microscopy. J. Mater. Sci. 2003, 38, 2793–2801. [Google Scholar] [CrossRef]
- Birnie, D.P., III. A model for drying control cosolvent selection for spin-coating uniformity: The thin film limit. Langmuir 2013, 29, 9072–9078. [Google Scholar] [CrossRef] [PubMed]
- Ohlídal, I.; Vohánka, J.; Čermák, M. Optics of Inhomogeneous Thin Films with Defects: Application to Optical Characterization. Coatings 2021, 11, 22. [Google Scholar] [CrossRef]
- Singh, R.; Verma, K.; Singh, T.; Barman, P.; Sharma, D. UV shielding with visible transparency based properties of poly (styrene-co-acrylonitrile)/Ag doped ZnO nanocomposite. Mater. Res. Express 2018, 5, 025035. [Google Scholar] [CrossRef]
- Broglia, M.F.; Acevedo, D.F.; Langheinrich, D.; Perez-Hernandez, H.R.; Barbero, C.A.; Lasagni, A.F. Rapid fabrication of periodic patterns on poly (styrene-co-acrylonitrile) surfaces using direct laser interference patterning. Int. J. Polym. Sci. 2015, 2015, 721035. [Google Scholar] [CrossRef]
- Tikhonravov, A.V.; Amotchkina, T.V.; Trubetskov, M.K.; Francis, R.J.; Janicki, V.; Sancho-Parramon, J.; Zorc, H.; Pervak, V. Optical characterization and reverse engineering based on multiangle spectroscopy. Appl. Opt. 2012, 51, 245–254. [Google Scholar] [CrossRef] [Green Version]

| Temperature (°C) | Thickness (nm) | Refractive Index at 500 nm |
|---|---|---|
| 80 | 67.1 ± 0.2 | 1.571 ± 0.001 |
| 150 | 64.6 ± 0.2 | 1.574 ± 0.001 |
| 220 | 67.4 ± 0.2 | 1.573 ± 0.001 |
| 300 | 51.7 ± 0.3 | 1.584 ± 0.002 |
| 350 | 36.8 ± 0.8 | 1.516 ± 0.002 |
| 400 | 36.3 ± 1.9 | 1.491 ± 0.003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hedl, E.; Fabijanić, I.; Šrut Rakić, I.; Vadla, I.; Sancho-Parramon, J. Fabrication by Spin-Coating and Optical Characterization of Poly(styrene-co-acrylonitrile) Thin Films. Coatings 2021, 11, 1015. https://doi.org/10.3390/coatings11091015
Hedl E, Fabijanić I, Šrut Rakić I, Vadla I, Sancho-Parramon J. Fabrication by Spin-Coating and Optical Characterization of Poly(styrene-co-acrylonitrile) Thin Films. Coatings. 2021; 11(9):1015. https://doi.org/10.3390/coatings11091015
Chicago/Turabian StyleHedl, Elizabeth, Ivana Fabijanić, Iva Šrut Rakić, Ivan Vadla, and Jordi Sancho-Parramon. 2021. "Fabrication by Spin-Coating and Optical Characterization of Poly(styrene-co-acrylonitrile) Thin Films" Coatings 11, no. 9: 1015. https://doi.org/10.3390/coatings11091015
APA StyleHedl, E., Fabijanić, I., Šrut Rakić, I., Vadla, I., & Sancho-Parramon, J. (2021). Fabrication by Spin-Coating and Optical Characterization of Poly(styrene-co-acrylonitrile) Thin Films. Coatings, 11(9), 1015. https://doi.org/10.3390/coatings11091015






