Curcumin: Modern Applications for a Versatile Additive
Abstract
1. Introduction
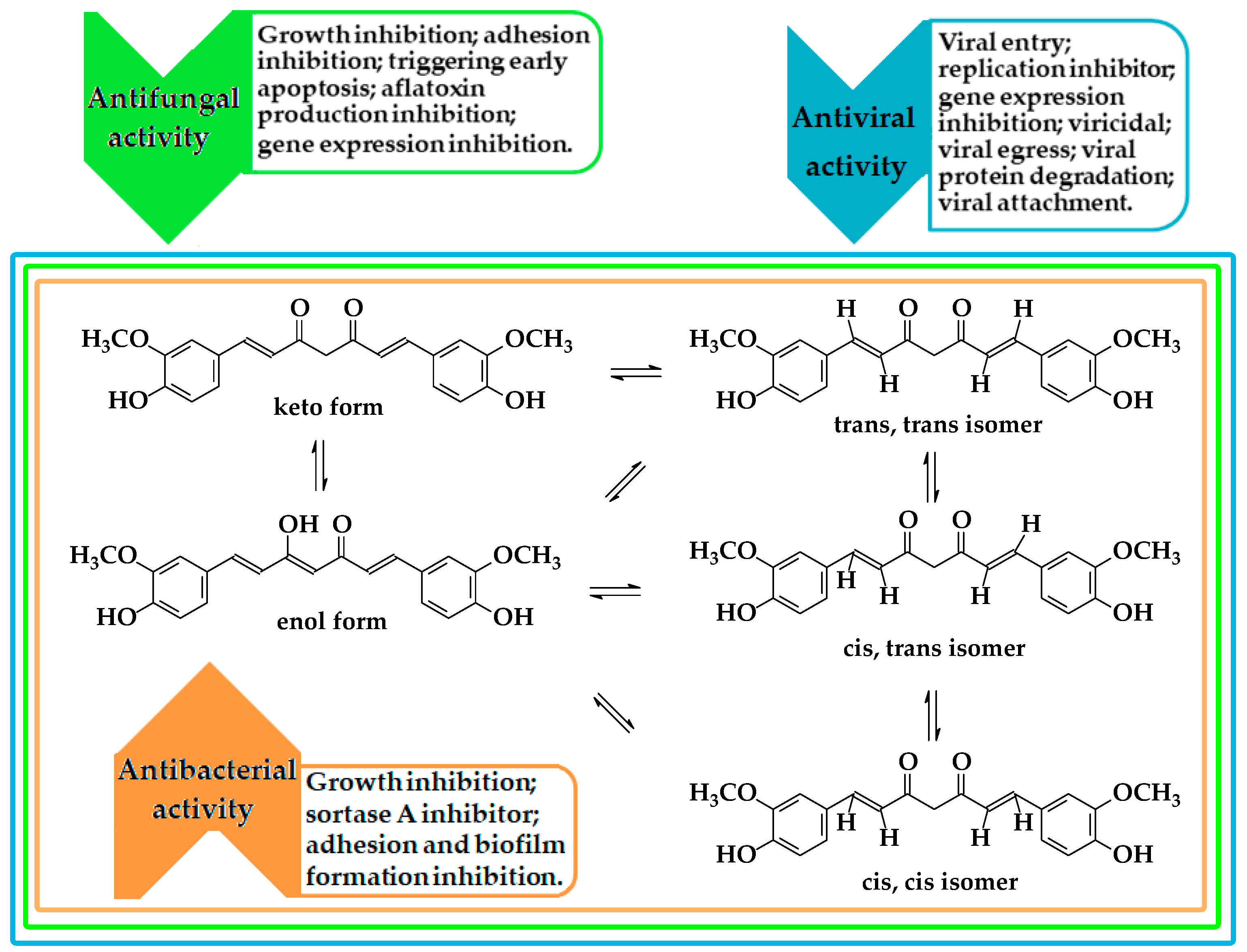
2. Methods of Obtaining and Antiviral, Antifungal, and Antibacterial Activity
| No. crt | Turmeric Species (Type of Sample) | Methods | Curcuminoids Yield % (mg/g) or Particle Size (mm) | Ref. |
|---|---|---|---|---|
| 1. | Curcuma longa (dried turmeric powder) | Surfactant-free microemulsion | (1.92) | [3] |
| 2. | Curcuma longa (dried turmeric powder) | Soxhlet extraction ternary system | 76.82 (88.96) 15.48 | [2,8] [15] |
| 3. | Curcuma longa (dried turmeric powder) | Ultrasound | (67.15) | [8] |
| 4. | Curcuma longa (dried turmeric powder) | Ultrasound/deep eutectic solvents | (58.87) | [9] |
| 5. | Curcuma longa (dried turmeric powder) | Microwave | 88(105.3) | [6] |
| 6. | Dried rhizomes of C. longa (Rajapurivariety) | Supercritical fluid extraction | (1.68) | [7] |
| 7. | Curcuma aromatic Salisb | Soxhlet extraction | 8.34 | [10] |
| 8. | Curcuma aromatic Salisb | Supercritical carbon dioxide | 7.54 | [10] |
| 9. | Curcuma xanthorrhizaRoxb | Solvent extraction | 4.98 | [3] |
| 10. | Curcuma longa (dried turmeric powder) | Pressurized liquid extraction | 0.125–0.45 | [11] |
| 11. | Curcuma longa (fresh turmeric) | Solid phase | - | [13] |
| 12. | Curcuma longa (extract the essential oil) | Subcritical water extraction | 90.19 | [14] |
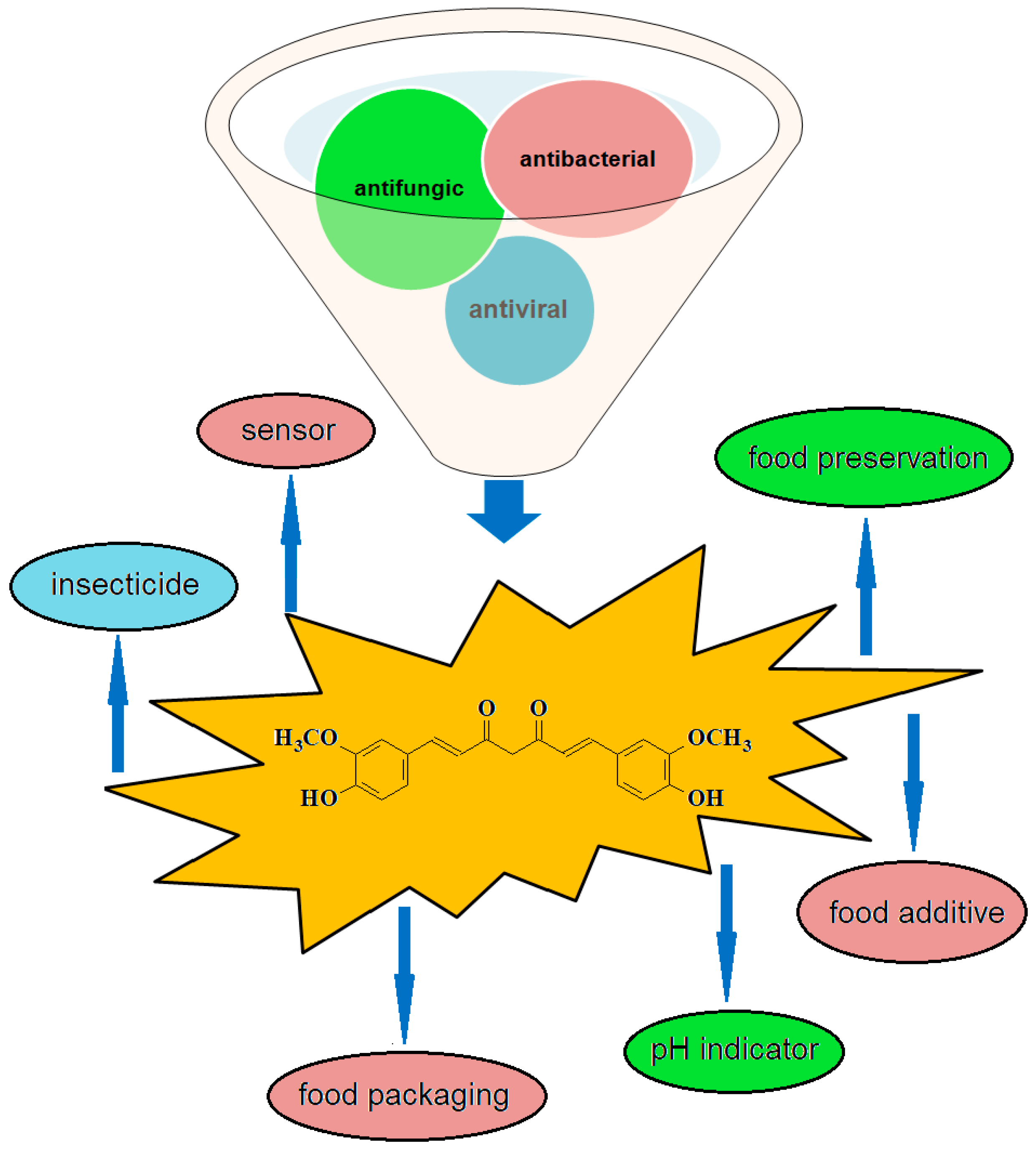
3. Additive for the Prevention of Spoilage, Safety and Quality of Food
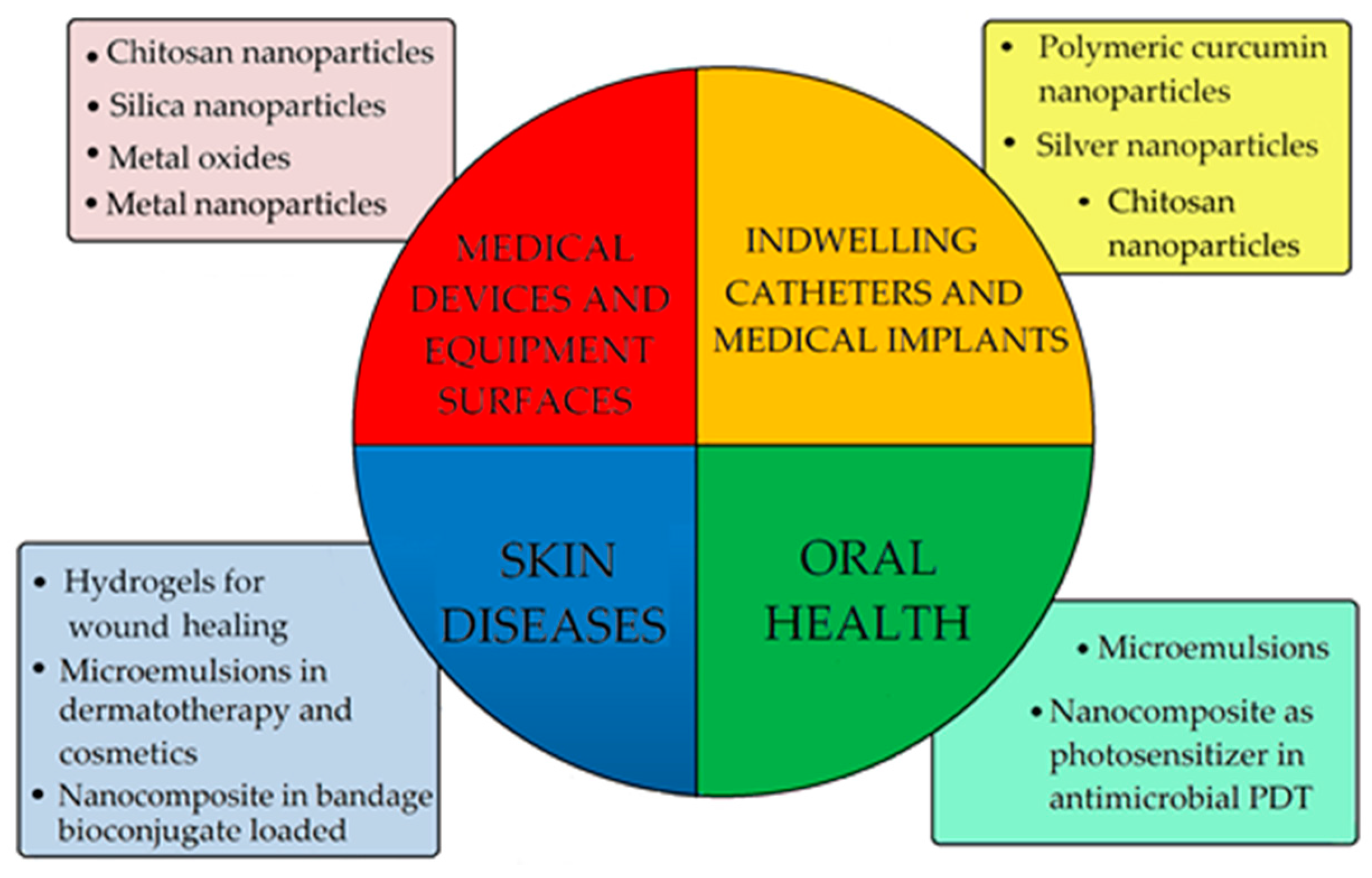
4. Additive for Health Care Products
| No. crt | Polymer Support | Target Microorganisms/Potential Application | Ref. |
|---|---|---|---|
| 1. | chitosan/pluronic membranes | S. aureus, P. aeruginosa/healing applications | [150] |
| 2. | chitosan/polycaprolactone | S. aureus/healing properties | [151] |
| 3. | hyaluronic acid modified pullulan polymers | E. coli, S. aureus/accelerating skin wound healing | [152] |
| 4 | lactide-co-glycolide /chitosan/β-cyclodextrin/poly(vinyl alcohol) | blend films | [153,154] |
| 5. | chitosan-collagen, gelatin, sodium alginate | S. aureus, E. coli | [155,156,157] |
| 6. | polyvinyl pyrrolidone(PVP)-cerium nitrate hexahydrate | S. aureus, E. coli/ dressing material-anti-scar property | [156] |
| 7. | cellulose hydroxypropyl-β-cyclodextrin-silver nanoparticle | S. aureus, P. aeruginosa, C. auris/healing properties | [158] |
| 8. | Metal oxides-NPs-cotton | MERSA, S. aureus, E. coli | [159,160,161,162] |
| 9. | cellulose-zinc oxide | S. aureus, T. rubrum/skin infection | [161] |
| 10. | graphene oxide | S. aureus, E. coli | [163] |
| 11. | 3-methyl-1-(hexadecyloxycarbonylmethyl)imidazolium bromide | hydrogel used for the wound healing | [164] |
| 12. | sodium alginate | wound healing applications | [165] |
| 15. | thiocarbohydrazide gelatin nanofibers | E. coli/wound healing applications | [166] |
| 16. | bacterial nanocellulose | S. aureus, E. coli | [167] |
| 17. | oleic acid based polymeric bandage | wound healing | [168] |
5. Future Developments of Curcumin-Based Materials
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO. Compendium of Food Additive Specifications. Available online: Fao.org/fileadmin/user_upload/jecfa_additives/docs/Monograph1/Additive-140 (accessed on 1 February 2021).
- Pawar, H.A.; Gavasane, A.J.; Choudhary, P.D. A novel and simple approach for extraction and isolation of curcuminoids from Turmeric rhizomes. Nat. Prod. Chem. Res. 2018, 6, 300. [Google Scholar] [CrossRef]
- Othman, A.F.M.; Rukayadi, Y.; Radu, S. Inhibition of Pseudomonas aeruginosa Quorum Sensing by Curcuma xanthorrhiza Roxb. Extract. J. Pure Appl. Microbiol. 2019, 13, 1335–1347. [Google Scholar] [CrossRef]
- Ibáñez, M.D.; Blázquez, M.A. Curcuma longa L. Rhizome Essential Oil from Extraction to its Agri-Food Applications. A Review. Plants 2020, 10, 44. [Google Scholar] [CrossRef] [PubMed]
- Yadav, D.K.; Sharma, K.; Dutta, A.; Kundu, A.; Awasthi, A. Purity evaluation of curcuminoids in the Turmeric extract obtained by accelerated solvent extraction. J. AOAC Int. 2017, 100, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Panichayupakaranant, P.; Lateh, L.; Yuenyongsawad, S.; Chen, H. A green method for preparation of curcuminoid-rich Curcuma longa extract and evaluation of its anticancer activity. Pharmacogn. Mag. 2019, 15, 730. [Google Scholar] [CrossRef]
- Nagavekar, N.; Singhal, R.S. Supercritical fluid extraction of Curcuma longa and Curcuma amada oleoresin: Optimization of extraction conditions, extract profiling, and comparison of bioactivities. Ind. Crop. Prod. 2019, 134, 134–145. [Google Scholar] [CrossRef]
- Patil, S.S.; Rathod, V.K. Synergistic Effect of Ultrasound and Three Phase Partitioning for the Extraction of Curcuminoids from Curcuma longa and its Bioactivity Profile. Process. Biochem. 2020, 93, 85–93. [Google Scholar] [CrossRef]
- Patil, S.S.; Pathak, A.; Rathod, V.K. Optimization and kinetic study of ultrasound assisted deep eutectic solvent based extraction: A greener route for extraction of curcuminoids from Curcuma longa. Ultrason. Sonochem. 2021, 70, 105267. [Google Scholar] [CrossRef]
- Vijayan, U.K.; Varakumar, S.; Singhal, R.S. A comparative account of extraction of oleoresin from Curcuma aromatica Salisb by solvent and supercritical carbon dioxide: Characterization and bioactivities. LWT 2019, 116, 108564. [Google Scholar] [CrossRef]
- Chao, I.-C.; Wang, C.-M.; Marcotullio, M.C.; Lin, L.-G.; Ye, W.-C.; Zhang, Q.-W. Simultaneous Quantification of Three Curcuminoids and Three Volatile Components of Curcuma longa Using Pressurized Liquid Extraction and High-Performance Liquid Chromatography. Molecules 2018, 23, 1568. [Google Scholar] [CrossRef]
- Zielińska, A.; Alves, H.; Marques, V.; Durazzo, A.; Lucarini, M.; Alves, T.; Morsink, M.; Willemen, N.; Eder, P.; Chaud, M.; et al. Properties, Extraction Methods, and Delivery Systems for Curcumin as a Natural Source of Beneficial Health Effects. Medicina 2020, 56, 336. [Google Scholar] [CrossRef]
- Thongchai, W.; Fukngoen, P. Synthesis of curcuminoid-imprinted polymers applied to the solid-phase extraction of curcuminoids from turmeric samples. J. Pharm. Anal. 2018, 8, 60–68. [Google Scholar] [CrossRef]
- Mottahedin, P.; Asl, A.H.; Khajenoori, M. Extraction of Curcumin and Essential Oil from Curcuma longa L. by Subcritical Water via Response Surface Methodology. J. Food Process. Preserv. 2016, 41, e13095. [Google Scholar] [CrossRef]
- Degot, P.; Huber, V.; El Maangar, A.; Gramüller, J.; Rohr, L.; Touraud, D.; Zemb, T.; Gschwind, R.M.; Kunz, W. Triple role of sodium salicylate in solubilization, extraction, and stabilization of curcumin from Curcuma longa. J. Mol. Liq. 2021, 329, 115538. [Google Scholar] [CrossRef]
- Theppawong, A.; van de Walle, T.; Grootaert, C.; Bultinck, M.; Desmet, T.; van Camp, J.; D’Hooghe, M. Synthesis of Novel Aza-aromatic Curcuminoids with Improved Biological Activities towards Various Cancer Cell Lines. Chem. Open 2018, 7, 381–392. [Google Scholar] [CrossRef]
- Alneyadi, S.S.; Amer, N.; Thomas, T.G.; Al Ajeil, R.; Breitener, P.; Munawar, N. Synthesis, Characterization, and Antioxidant Activity of Some 2-Methoxyphenols derivatives. Heterocycl. Commun. 2020, 26, 112–122. [Google Scholar] [CrossRef]
- Insuasty, D.; Castillo, J.; Becerra, D.; Rojas, H.; Abonia, R. Synthesis of Biologically Active Molecules through Multicomponent Reactions. Molecules 2020, 25, 505. [Google Scholar] [CrossRef] [PubMed]
- Raduly, M.F.; Raditoiu, V.; Raditoiu, A.; Wagner, L.E.; Amariutei, V.; Darvaru, G.A. Facile Synthesis of Curcumin and Curcuminoid-like Derivatives at Microwaves. Rev. Chim. 2018, 69, 1327–1331. [Google Scholar] [CrossRef]
- Tantriasa, L.D.; Anwar, C.; Astuti, E. Synthesis of Curcumin Analogs under Ultrasound Irradiation for Inhibiting α-Amylase. Mater. Sci. Forum 2019, 948, 115–119. [Google Scholar] [CrossRef]
- Ausili, A.; Gómez-Murcia, V.; Candel, A.M.; Beltrán, A.; Torrecillas, A.; He, L.; Jiang, Y.; Zhang, S.; Teruel, J.A.; Gómez-Fernández, J.C. A comparison of the location in membranes of curcumin and curcumin-derived bivalent compounds with potential neuroprotective capacity for Alzheimer’s disease. Colloids Surf. B Biointerfaces 2021, 199, 111525. [Google Scholar] [CrossRef]
- Li, S.; Lu, M.; Hu, R.; Tang, T.; Hou, K.; Liu, Y. Dyeing ramie fabrics with curcumin in NaOH/Urea solution at low temperature. Cloth. Text. Res. J. 2019, 37, 66–79. [Google Scholar] [CrossRef]
- Mejri, A.; Mars, A.; Elfil, H.; Hamzaoui, A.H. Reduced graphene oxide nanosheets modified with nickel disulfide and curcumin nanoparticles for non-enzymatic electrochemical sensing of methyl parathion and 4-nitrophenol. Microchim. Acta 2019, 186, 704. [Google Scholar] [CrossRef]
- Mejri, A.; Mars, A.; Elfil, H.; Hamzaoui, A.H. Voltammetric simultaneous quantification of p-nitrophenol and hydrazine by using magnetic spinel FeCO2O4 nanosheets on reduced graphene oxide layers modified with curcumin-stabilized silver nanoparticles. Microchim. Acta 2019, 186, 561. [Google Scholar] [CrossRef] [PubMed]
- Prabu, S.; Mohamad, S. Curcumin/beta-cyclodextrin inclusion complex as a new “turn-off” fluorescent sensor system for sensitive recognition of mercury ion. J. Mol. Struct. 2020, 1204, 127528. [Google Scholar] [CrossRef]
- Jennings, M.; Parks, R. Curcumin as an Antiviral Agent. Viruses 2020, 12, 1242. [Google Scholar] [CrossRef]
- Praditya, D.; Kirchhoff, L.; Brüning, J.; Rachmawati, H.; Steinmann, J.; Steinmann, E. Anti-infective Properties of the Golden Spice Curcumin. Front. Microbiol. 2019, 10, 912. [Google Scholar] [CrossRef]
- Wen, C.C.; Kuo, Y.H.; Jan, J.T.; Liang, P.H.; Wang, S.Y.; Liu, H.G.; Lee, C.K.; Chang, S.T.; Kuo, C.J.; Lee, S.S. Specific plant terpenoids and lignoids possess potent antiviral activities against severe acute respiratory syndrome coronavirus. J. Med. Chem. 2007, 50, 4087–4095. [Google Scholar] [CrossRef]
- Ting, D.; Dong, N.; Fang, L.; Lu, J.; Bi, J.; Xiao, S.; Han, H. Multisite Inhibitors for Enteric Coronavirus: Antiviral Cationic Carbon Dots Based on Curcumin. ACS Appl. Nano Mater. 2018, 1, 5451–5459. [Google Scholar] [CrossRef]
- Ghorbanzadeh, R.; Assadian, H.; Chiniforush, N.; Parker, S.; Pourakbari, B.; Ehsani, B.; Alikhani, M.Y.; Bahador, A. Modulation of virulence in Enterococcus faecalis cells surviving antimicrobial photodynamic inactivation with reduced graphene oxide-curcumin: An ex vivo biofilm model. Photodiagn. Photodyn. Ther. 2020, 29, 101643. [Google Scholar] [CrossRef]
- Global Curcumin Market 2021 Covid 19 Analysis with Top Industry. Available online: www.openpr.com/news/2169421/curcumin-market-with-covid-19-impact-analysis-top-industry (accessed on 27 March 2021).
- Curcumin Market Size, Share & Trends Analysis Report by Application (Pharmaceutical, Food, Cosmetics), by Region (North America, Europe, Asia Pacific, Central & South America, Middle East & Africa), and Segment Forecasts, 2020–2027. Available online: www.researchandmarkets.com/reports/4613416 (accessed on 17 January 2021).
- Curcumin Market Size Worth $1.30 Billion by 2025|CAGR 12.3%: Grand View Research, Inc. Available online: www.prnewswire.com/news-releases/curcumin-market-size-worth-1-30-billion-by-2025-cagr-12-3-grand-view-research-inc--811278562 (accessed on 17 January 2021).
- Curcumin Market by Application (Pharmaceutical, Food & Beverage, Cosmetic, Others)—Global Opportunity Analysis and Industry Forecast, 2018–2025. Available online: www.alliedmarketresearch.com/curcumin-market (accessed on 17 January 2021).
- Curcumin Market Growing Rapidly with Prominent Players Update (2022–2031)||Synthite Ind, Sabinsa, Indena. Available online: www.marketwatch.com/press-release/curcumin-market-growing-rapidly-with-prominent-players-update2022-2031-synthite-ind-sabinsa-indena-2021-01-27 (accessed on 17 January 2021).
- Curcumin Market Headed To $104.19 Mn by 2025 at 6.5% CAGR: The Demand is Expected to Surge During the Covid-19 Pandemic. Available online: www.einnews.com/pr_news/529267704/curcumin-market-headed-to-104-19-mn-by-2025-at-6-5-cagr-the-demand-is-expected-to-surge-during-the-covid-19-pandemic (accessed on 17 January 2021).
- Supply of Spices and Herbs Seriously Affected by COVID-19. Available online: www.cbi.eu/news/supply-spices-herbs-seriously-affected-covid-19 (accessed on 27 March 2021).
- Consumer Sentiment and Behavior Continue to Reflect the Uncertainty of the COVID-19 crisis. Available online: www.mckinsey.com/business-functions/marketing-and-sales/our-insights/a-global-view-of-how-consumer-behavior-is-changing-amid-covid-19# (accessed on 27 March 2021).
- Responding to Consumer Trends in the New Reality. Available online: www.assets.kpmg/content/dam/kpmg/xx/pdf/2020/11/consumers-new-reality (accessed on 27 March 2021).
- Hoekstra, J.C.; Leefang, P.S.H. Marketing in the era of COVID 19. Ital. J. Mark. 2020, 249–260. [Google Scholar] [CrossRef]
- Shetty, T.; Dubey, A.; Ravi, G.S.; Hebbar, S.; Shastry, C.S.; Charyulu, N. Antifungal and antioxidant therapy for the treatment of fungal infection with microemulsion gel containing curcumin and vitamin C. Asian J. Pharm. 2017, 11, 717–725. [Google Scholar]
- Sarwar, S.; Netzel, G.; Netzel, M.E.; Mereddy, R.; Phan, A.D.T.; Hong, H.T.; Cozzolino, D.; Sultanbawa, Y. Impact of Curcumin-Mediated Photosensitization on Fungal Growth, Physicochemical Properties and Nutritional Composition in Australian Grown Strawberry. Food Anal. Methods 2021, 14, 465–472. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Sun, X.-Z.; Wang, N. A thermosensitive textile-based drug delivery system for treating UVB-induced damage. Cellulose 2020, 27, 8329–8339. [Google Scholar] [CrossRef]
- Degot, P.; Huber, V.; Hofmann, E.; Hahn, M.; Touraud, D.; Kunz, W. Solubilization and extraction of curcumin from Curcuma Longa using green, sustainable, and food-approved surfactant-free microemulsions. Food Chem. 2021, 336, 127660. [Google Scholar] [CrossRef]
- Degot, P.; Huber, V.; Touraud, D.; Kunz, W. Curcumin extracts from Curcuma Longa—Improvement of concentration, purity, and stability in food-approved and water-soluble surfactant-free microemulsions. Food Chem. 2021, 339, 128140. [Google Scholar] [CrossRef]
- Jeliński, T.; Przybyłek, M.; Cysewski, P. Natural Deep Eutectic Solvents as Agents for Improving Solubility, Stability and Delivery of Curcumin. Pharm. Res. 2019, 36, 1–10. [Google Scholar] [CrossRef]
- Liu, Y.; Li, J.; Fu, R.; Zhang, L.; Wang, D.; Wang, S. Enhanced extraction of natural pigments from Curcuma longa L. using natural deep eutectic solvents. Ind. Crop. Prod. 2019, 140, 111620. [Google Scholar] [CrossRef]
- Doldolova, K.; Bener, M.; Lalikoğlu, M.; Aşçı, Y.S.; Arat, R.; Apak, R. Optimization and modeling of microwave-assisted extraction of curcumin and antioxidant compounds from turmeric by using natural deep eutectic solvents. Food Chem. 2021, 353, 129337. [Google Scholar] [CrossRef]
- Praveen, A.N.K.; Madhyastha, S. Role of cotton and Turmeric smoke as a potential treatment of COVID-19. Int. J. Adv. Res. Innov. Tech. 2020, 6, 287–290. [Google Scholar]
- Saraswat, J.; Singh, P.; Patel, R. A computational approach for the screening of potential antiviral compounds against SARS-CoV-2 protease: Ionic liquid vs. herbal and natural compounds. J. Mol. Liq. 2021, 326, 115298. [Google Scholar] [CrossRef]
- Momo, E.J.; Sen, M.L.; Nguemezi, S.T.; Youassi, O.Y.; Mounbain, F.; Sameza, M.L.; Tchoumbougnang, F.; Jazet, P.M. Chemical Composition of the Essential Oil of Curcuma longa and Evaluation of the Antifungal Activity on Rhizopus stolonifer and Penicillium sp. Responsible Fungi for Post-harvest Rot of Dioscorea rotoundata in Cameroon. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 797–808. [Google Scholar] [CrossRef]
- Gámez-Espinosa, E.; Anaya, M.; Borges, P.; Crespo, D.M.B. Antifungal effects of Curcuma longa L. essential oil against pathogenic strains isolated from indoor air. Aerobiologia 2021, 37, 119–126. [Google Scholar] [CrossRef]
- Marchi, L.B.; Dornellas, F.C.; Polonioc, J.C.; Pamphile, J.A.; Monteiro, A.R.G.; Gonçalves, O.H.; Perdoncini, M.R.F.G. Antifungal activity of Curcuma longa L. (Zingiberaceae) against degrading Filamentous Fungi. Chem. Eng. Trans. 2019, 75, 319–324. [Google Scholar] [CrossRef]
- Kazi, H.A.; Channa, T.; Unarb, A.A.; Unarc, K.; Sabzoib, W.; Perveenc, S.; Mangid, A.A.; Ahmer, A. Pharmaceutical formulation of Garlic and Turmeric dried crude extract and their synergistic antifungal activity and safety. Iran. J. Pharm. Sci. 2018, 14, 75–82. [Google Scholar]
- Jiang, B.-C.; Shen, J.-Y.; Wu, J.; Lu, R.-Y.; Zheng, W.; Dong, J.-X.; Yan, L.; Jin, Y.-S. In vitro antifungal activity of 163 extracts from traditional Chinese medicine herbs. Eur. J. Integr. Med. 2020, 39, 101213. [Google Scholar] [CrossRef]
- De Paula, R.L.; Maniglia, B.C.; Assis, O.B.G.; Tapia-Blácido, D.R. Evaluation of the turmeric dye extraction residue in the formation of protective coating on fresh bananas (Musa acuminata cv. ’Maçã’). J. Food Sci. Technol. 2018, 55, 3212–3220. [Google Scholar] [CrossRef]
- Abdelgaleil, S.A.M.; Zoghroban, A.A.M.; El-Bakry, A.M.; Kassem, S.M.I. Insecticidal and antifungal activities of crude extracts and pure compounds from rhizomes of Curcuma longa L. (Zingiberaceae). J. Agr. Sci. Tech. 2019, 21, 1049–1061. [Google Scholar]
- Olszewska, M.A.; Gędas, A.; Simões, M. Antimicrobial polyphenol-rich extracts: Applications and limitations in the food industry. Food Res. Int. 2020, 134, 109214. [Google Scholar] [CrossRef]
- Solano, A.A.N.; Aguirre, P.C.; Vargas, L.O.S.; Navarro, J.B.R.; Escalona, J.R.B. Antimicrobial effect of curcumin on Enterococcus faecalis, Escherichia coli, Staphylococcus aureus and Candida albicans. Nova Sci. 2020, 25, 1–12. [Google Scholar]
- Phuna, Z.X.; Yu, J.K.E.; Tee, J.Y. In vitro evaluation of nanoemulsions of curcumin, piperine and tualang honey as antifungal agents for Candida species. J. Appl. Biotechnol. Rep. 2020, 7, 190–198. [Google Scholar] [CrossRef]
- Sathiyabama, M.; Indhumathi, M.; Amutha, T. Preparation and characterization of curcumin functionalized copper nanoparticles and their application enhances disease resistance in chickpea against wilt pathogen. Biocatal. Agric. Biotechnol. 2020, 29, 101823. [Google Scholar] [CrossRef]
- Jung, S.; Cui, Y.; Barnes, M.; Satam, C.; Zhang, S.; Chowdhury, R.A.; Adumbumkulath, A.; Sahin, O.; Miller, C.; Sajadi, S.M.; et al. Multifunctional Bio-Nanocomposite Coatings for Perishable Fruits. Adv. Mater. 2020, 32, e1908291. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Misba, L.; Khan, A.U. Antibiotics versus biofilm: An emerging battleground in microbial communities. Antimicrob. Resist. Infect. Control. 2019, 8, 1–10. [Google Scholar] [CrossRef]
- Abebe, G.M. The Role of Bacterial Biofilm in Antibiotic Resistance and Food Contamination. Int. J. Microbiol. 2020, 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Raorane, C.J.; Lee, J.-H.; Kim, Y.-G.; Rajasekharan, S.K.; García-Contreras, R.; Lee, J. Antibiofilm and Antivirulence Efficacies of Flavonoids and Curcumin Against Acinetobacter baumannii. Front. Microbiol. 2019, 10, 990. [Google Scholar] [CrossRef] [PubMed]
- Baishya, R.; Banerjee, S. In-Vitro antibiofilm activity of selected medicinal plants against Staphylococcus aureus biofilm on chitin flakes as substrate. Int. J. Res. Pharm. Sci. 2020, 11, 1595–1603. [Google Scholar] [CrossRef][Green Version]
- Samiappan, S.C.; Pandiyan, R.; Palanisamy, S.; Ramalingam, S.; Saravanan, R.; Hameed, S.A. Targeting the Extracellular Polysaccharide Production (EPS) by Biofilm Forming Bacteria from Orthodontic Brackets and Wires through Antiquorum Sensing Action of Bioactive Compounds from Curcuma longa and Zingiber officinale. Biomed. Pharmacol. J. 2020, 13, 1037–1045. [Google Scholar] [CrossRef]
- Bali, E.B.; Türkmen, K.E.; Erdönmez, D.; Sağlam, N. Comparative Study of Inhibitory Potential of Dietary Phytochemicals Against Quorum Sensing Activity of and Biofilm Formation by Chromobacterium violaceum 12472, and Swimming and Swarming Behaviour of Pseudomonas aeruginosa PAO1. Food Technol. Biotechnol. 2019, 57, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Li, X.; Lin, H.; Zhou, Y. Curcumin as a Promising Antibacterial Agent: Effects on Metabolism and Biofilm Formation in S. mutans. BioMed Res. Int. 2018, 2018, 1–11. [Google Scholar] [CrossRef]
- Fakhrullina, G.; Khakimova, E.; Akhatova, F.; Lazzara, G.; Parisi, F.; Fakhrullin, R.F. Selective Antimicrobial Effects of Curcumin@Halloysite Nanoformulation: A Caenorhabditis elegans Study. ACS Appl. Mater. Interfaces 2019, 11, 23050–23064. [Google Scholar] [CrossRef]
- Gawish, S.M.; Mashaly, H.M.; Helmy, H.M.; Ramadan, A.M.; Farouk, R. Effect of Mordant on UV Protection and Antimicrobial Activity of Cotton, Wool, Silk and Nylon Fabrics Dyed with Some Natural Dyes. J. Nanomed. Nanotechnol. 2017, 8. [Google Scholar] [CrossRef]
- Xue, B.; Huang, J.; Zhang, H.; Li, B.; Xu, M.; Zhang, Y.; Xie, M.; Li, X. Micronized curcumin fabricated by supercritical CO2 to improve antibacterial activity against Pseudomonas aeruginosa. Artif. Cells Nanomed. Biotechnol. 2020, 48, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Varaprasad, K.; Vimala, K.; Ravindra, S.; Reddy, N.N.; Reddy, G.V.S.; Raju, K.M. Fabrication of silver nanocomposite films impregnated with curcumin for superior antibacterial applications. J. Mater. Sci. Mater. Med. 2011, 22, 1863–1872. [Google Scholar] [CrossRef]
- Versace, D.-L.; Moran, G.; Belqat, M.; Spangenberg, A.; Meallet-Renault, R.; Abbad-Andaloussi, S.; Brezova, V.; Malval, J.-P. Highly Virulent Bactericidal Effects of Curcumin-Based μ-Cages Fabricated by Two-Photon Polymerization. ACS Appl. Mater. Interfaces 2020, 12, 5050–5057. [Google Scholar] [CrossRef]
- Saha, T.; Kumar, P.; Sepay, N.; Ganguly, D.; Tiwari, K.; Mukhopadhyay, K.; Das, S. Multitargeting Antibacterial Activity of a Synthesized Mn2+ Complex of Curcumin on Gram-Positive and Gram-Negative Bacterial Strains. ACS Omega 2020, 5, 16342–16357. [Google Scholar] [CrossRef]
- Singh, A.K.; Mishra, H.; Firdaus, Z.; Yadav, S.; Aditi, P.; Nandy, N.; Sharma, K.; Bose, P.; Pandey, A.K.; Chauhan, B.S.; et al. MoS2-Modified Curcumin Nanostructures: The Novel Theranostic Hybrid Having Potent Antibacterial and Antibiofilm Activities against Multidrug-Resistant Hypervirulent Klebsiella pneumoniae. Chem. Res. Toxicol. 2019, 32, 1599–1618. [Google Scholar] [CrossRef] [PubMed]
- Angelini, G.; Pasc, A.; Gasbarri, C. Curcumin in silver nanoparticles aqueous solution: Kinetics of keto-enol tautomerism and effects on AgNPs. Colloids Surf. A Physicochem. Eng. Asp. 2020, 603, 125235. [Google Scholar] [CrossRef]
- El Hamid, A.; Hajer, M.; Aziz, A.; Mohamed, S.; Naeem, A.; Fatma, M. Antimicrobial efficacy of nanopropolis coated vs. silver-curcumin nanoparticles coated gutta-percha Points on various microbial species. Egypt. Dent. J. 2020, 66, 1893–1902. [Google Scholar] [CrossRef]
- Gholami, M.; Zeighami, H.; Bikas, R.; Heidari, A.; Rafiee, F.; Haghi, F. Inhibitory activity of metal-curcumin complexes on quorum sensing related virulence factors of Pseudomonas aeruginosa PAO1. AMB Express 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Masoule, S.F.; Pourhajibagher, M.; Safari, J.; Khoobi, M. Base-free green synthesis of copper (II) oxide nanoparticles using highly cross-linked poly(curcumin) nanospheres: Synergistically improved antimicrobial activity. Res. Chem. Intermed. 2019, 45, 4449–4462. [Google Scholar] [CrossRef]
- Huang, G.; Yan, Y.; Xu, D.; Wu, J.; Xu, C.; Fu, L.; Lin, B. Curcumin-loaded nanoMOFs@CMFP: A biological preserving paste with antibacterial properties and long-acting, controllable release. Food Chem. 2021, 337, 127987. [Google Scholar] [CrossRef] [PubMed]
- Kumbar, V.M.; Peram, M.R.; Kugaji, M.S.; Shah, T.; Patil, S.P.; Muddapur, U.M.; Bhat, K.G. Effect of curcumin on growth, biofilm formation and virulence factor gene expression of Porphyromonas gingivalis. Odontology 2021, 109, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Pan, T.; Lin, H.; Zhou, Y. The enhancing antibiofilm activity of curcumin on Streptococcus mutans strains from severe early childhood caries. BMC Microbiol. 2020, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mitsuwan, W.; Sangkanu, S.; Romyasamit, C.; Kaewjai, C.; Jimoh, T.O.; Pereira, M.D.L.; Siyadatpanah, A.; Kayesth, S.; Nawaz, M.; Rahmatullah, M.; et al. Curcuma longa rhizome extract and Curcumin reduce the adhesion of Acanthamoeba triangularis trophozoites and cysts in polystyrene plastic surface and contact lens. Int. J. Parasitol. Drugs Drug Resist. 2020, 14, 218–229. [Google Scholar] [CrossRef]
- Shariati, A.; Asadian, E.; Fallah, F.; Azimi, T.; Hashemi, A.; Sharahi, J.Y.; Moghadam, M.T. Evaluation of Nano-curcumin effects on expression levels of virulence genes and biofilm production of multidrug-resistant Pseudomonas aeruginosa isolated from burn wound infection in Tehran, Iran. Infect. Drug Resist. 2019, 12, 2223–2235. [Google Scholar] [CrossRef]
- Kumari, A.; Guliani, A.; Shukla, A.K.; Kumar, S.; Acharya, A. Green surfactant based synthesis of curcumin loaded poly lactic-co-glycolic acid nanoparticles with enhanced solubility, photo-stability and anti-biofilm activity. J. Drug Deliv. Sci. Technol. 2020, 59, 101884. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, J.; Huang, L.; Jing, J.; Wang, N.; Wang, L. Curcumin encapsulation and protection based on lysozyme nanoparticles. Food Sci. Nutr. 2019, 7, 2702–2707. [Google Scholar] [CrossRef]
- Lewińska, A.; Jaromin, A.; Jezierska, J. Role of architecture of N-oxide surfactants in the design of nanoemulsions for Candida skin infection. Colloids Surf. B Biointerfaces 2020, 187, 110639. [Google Scholar] [CrossRef]
- Soumya, K.R.; Jishma, P.; Dhivya, R.; Annaraj, J.; Sugathan, S.; Mathew, J.; Radhakrishnan, E.K. Role of Nanocurcumin as a Surface Modifying Agent with Excellent Preventive Effect on Device-Related CoNS Infections. Proc. Natl. Acad. Sci. USA India Sect. B Boil. Sci. 2019, 90, 29–35. [Google Scholar] [CrossRef]
- Yakub, G.; Toncheva, A.; Kussovski, V.; Toshkova, R.; Georgieva, A.; Nikolova, E.; Manolova, N.; Rashkov, I. Curcumin-PVP Loaded Electrospun Membranes with Conferred Antibacterial and Antitumoral Activities. Fibers Polym. 2020, 21, 55–65. [Google Scholar] [CrossRef]
- Montoya-Villegasa, K.A.; Ramirez-Jimeneza, A.; Zizumbo-Lopeza, A.; Perez-Sicairosa, S.; Leal-Acevedoc, B.; Bucioc, E.; Licea-Claverie, A. Controlled surface modification of silicone rubber by gamma-irradiation followed by RAFT grafting polymerization. Eur. Polym. J. 2020, 134, 109817. [Google Scholar] [CrossRef]
- Singh, A.K.; Prakash, P.; Singh, R.; Nandy, N.; Firdaus, Z.; Bansal, M.; Singh, R.K.; Srivastava, A.; Roy, J.K.; Mishra, B. Curcumin Quantum Dots mediated degradation of bacterial biofilms. Front. Microbiol. 2017, 8, 1517. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Moser, D.; Han, F.; Leonhard, M.; Schneider-Stickler, B.; Tan, Y. Preparation and antibiofilm studies of curcumin loaded chitosan nanoparticles against polymicrobial biofilms of Candida albicans and Staphylococcus aureus. Carbohydr. Polym. 2020, 241, 116254. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Patlan, D.; Solis-Cruz, B.; Cano-Vega, M.A.; Beyssac, E.; Garrait, G.; Hernandez-Velasco, X.; Lopez-Arellano, R.; Tellez, G.; Rivera-Rodriguez, G.R. Development of Chitosan and Alginate Nanocapsules to Increase the Solubility, Permeability and Stability of Curcumin. J. Pharm. Innov. 2019, 14, 132–140. [Google Scholar] [CrossRef]
- Luo, L.; Wu, Y.; Liu, C.; Zou, Y.; Huang, L.; Liang, Y.; Ren, J.; Liu, Y.; Lin, Q. Elaboration and characterization of curcumin-loaded soy soluble polysaccharide (SSPS)-based nanocarriers mediated by antimicrobial peptide nisin. Food Chem. 2021, 336, 127669. [Google Scholar] [CrossRef]
- Vasudevan, S.; Prabhune, A.A. Photophysical studies on curcumin-sophorolipid nanostructures: Applications in quorum quenching and imaging. R. Soc. Open Sci. 2018, 5, 170865. [Google Scholar] [CrossRef]
- Oves, M.; Rauf, M.A.; Ansari, M.O.; Khan, A.A.P.; Qari, H.A.; Alajmi, M.F.; Sau, S.; Iyer, A.K. Graphene Decorated Zinc Oxide and Curcumin to Disinfect the Methicillin-Resistant Staphylococcus aureus. Nanomaterials 2020, 10, 1004. [Google Scholar] [CrossRef] [PubMed]
- Prateeksha; Rao, C.V.; Das, A.K.; Barik, S.K.; Singh, B.N. ZnO/Curcumin Nanocomposites for Enhanced Inhibition of Pseudomonas aeruginosa Virulence via LasR-RhlR Quorum Sensing Systems. Mol. Pharm. 2019, 16, 3399–3413. [Google Scholar] [CrossRef]
- Hami, Z. Coating Iron Oxide Nanoparticles with Chitosan for Targeted Delivery of Nanocurcumin. Ann. Mil. Health Sci. Res. 2020, 18, 103657. [Google Scholar] [CrossRef]
- Yang, Q.-Q.; Farha, A.K.; Kim, G.; Gul, K.; Gan, R.-Y.; Corke, H. Antimicrobial and anticancer applications and related mechanisms of curcumin-mediated photodynamic treatments. Trends Food Sci. Technol. 2020, 97, 341–354. [Google Scholar] [CrossRef]
- Abdulrahman, H.; Misba, L.; Ahmad, S.; Khan, A.U. Curcumin induced photodynamic therapy mediated suppression of quorum sensing pathway of Pseudomonas aeruginosa: An approach to inhibit biofilm in vitro. Photodiagn. Photodyn. Ther. 2020, 30, 101645. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Chen, B.; Li, H.; Zeng, Q.-H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Enhanced antibacterial and antibiofilm functions of the curcumin-mediated photodynamic inactivation against Listeria monocytogenes. Food Control 2020, 108, 106886. [Google Scholar] [CrossRef]
- Pan, H.; Wang, D.; Zhang, F. In vitro antimicrobial effect of curcumin-based photodynamic therapy on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans. Photodiagn. Photodyn. Ther. 2020, 32, 102055. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.P.; Santos, M.S.; Rodrigues, P.L.F.; Araújo, T.S.D.; de Oliveira, J.M.; Rosa, L.P.; Bagnato, V.S.; da Silva, F.C. Photodynamic therapry with curcumin in the reduction of enterococcus faecalis biofilm in bone cavity: rMicrobiological and spectral fluorescense analysis. Photodiagn. Photodyn. Ther. 2021, 33, 102084. [Google Scholar] [CrossRef]
- Chen, L.; Song, Z.; Zhi, X.; Du, B. Photoinduced Antimicrobial Activity of Curcumin-Containing Coatings: Molecular Interaction, Stability and Potential Application in Food Decontamination. ACS Omega 2020, 5, 31044–31054. [Google Scholar] [CrossRef]
- Rocha, M.P.; Ruela, A.L.; Rosa, L.P.; Santos, G.P.; Rosa, F.C. Antimicrobial photodynamic therapy in dentistry using an oil-in-water microemulsion with curcumin as a mouthwash. Photodiagnosis Photodyn. Ther. 2020, 32, 101962. [Google Scholar] [CrossRef]
- Nima, G.; Soto-Montero, J.; Alves, L.A.; Mattos-Graner, R.O.; Giannini, M. Photodynamic inactivation of Streptococcus mutans by curcumin in combination with EDTA. Dent. Mater. 2021, 37, e1–e14. [Google Scholar] [CrossRef]
- Mirzahosseinipour, M.; Khorsandi, K.; Hosseinzadeh, R.; Ghazaeian, M.; Shahidi, F.K. Antimicrobial photodynamic and wound healing activity of curcumin encapsulated in silica nanoparticles. Photodiagn. Photodyn. Ther. 2020, 29, 101639. [Google Scholar] [CrossRef]
- Ahmadi, H.; Haddadi-Asl, V.; Ghafari, H.-A.; Ghorbanzadeh, R.; Mazlum, Y.; Bahador, A. Shear bond strength, adhesive remnant index, and anti-biofilm effects of a photoexcited modified orthodontic adhesive containing curcumin doped poly lactic-co-glycolic acid nanoparticles: An ex-vivo biofilm model of S. mutans on the enamel slab bonded brackets. Photodiag. Photodyn. Ther. 2020, 30, 101674. [Google Scholar] [CrossRef]
- Zago, L.H.D.P.; de Annunzio, S.R.; de Oliveira, K.T.; Barbugli, P.A.; Valdes, B.R.; Feres, M.; Fontana, C.R. Antimicrobial photodynamic therapy against metronidazole-resistant dental plaque bactéria. J. Photochem. Photobiol. B Biol. 2020, 209, 111903. [Google Scholar] [CrossRef] [PubMed]
- Pourhajibagher, M.; Plotino, G.; Chiniforush, N.; Bahador, A. Dual wavelength irradiation antimicrobial photodynamic therapy using indocyanine green and metformin doped with nano-curcumin as an efficient adjunctive endodontic treatment modality. Photodiag. Photodyn. Ther. 2020, 29, 101628. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Hodjat, M.; Bahador, A. Sonodynamic excitation of nanomicelle curcumin for eradication of Streptococcus mutans under sonodynamic antimicrobial chemotherapy: Enhanced anti-caries activity of nanomicelle curcumin. Photodiag. Photodyn. Ther. 2020, 30, 101780. [Google Scholar] [CrossRef] [PubMed]
- Kali, A.; Bhuvaneshwar, D.; Charles, P.M.V.; Seetha, K.S. Antibacterial synergy of curcumin with antibiotics against biofilm producing clinical bacterial isolates. J. Basic Clin. Pharm. 2016, 7, 93–96. [Google Scholar] [CrossRef]
- Hamzah, H.; Hertiani, T.; Pratiwi, S.U.T.; Nuryastuti, T. Inhibitory activity and degradation of curcumin as Anti-Biofilm Polymicrobial on Catheters. Int. J. Res. Pharm. Sci. 2020, 11, 830–835. [Google Scholar] [CrossRef][Green Version]
- Hosseini, A.; Nejadsattari, T.; Zargar, M. In Vitro Anti-Biofilm Activity of Curcumin Nanoparticles in Acinetobacter baumannii: A Culture-Based and Molecular Approach. Arch. Clin. Infect. Dis. 2019, 14, 83263. [Google Scholar] [CrossRef]
- Yadav, S.; Singh, A.K.; Agrahari, A.K.; Sharma, K.; Singh, A.S.; Gupta, M.K.; Tiwari, V.K.; Prakash, P. Making of water soluble curcumin to potentiate conventional antimicrobials by inducing apoptosis-like phenomena among drug-resistant bacteria. Sci. Rep. 2020, 10, 1–22. [Google Scholar] [CrossRef]
- Adlia, A.; Tomagola, I.; Damayanti, S.; Mulya, A.; Rachmawati, H. Antifibrotic Activity and In Ovo Toxicity Study of Liver-Targeted Curcumin-Gold Nanoparticle. Sci. Pharm. 2018, 86, 41. [Google Scholar] [CrossRef] [PubMed]
- Muniyappan, N.; Pandeeswaran, M.; Amalraj, A. Green synthesis of gold nanoparticles using Curcuma pseudomontana isolated curcumin: Its characterization, antimicrobial, antioxidant and anti-inflammatory activities. Environ. Chem. Ecotoxicol. 2021, 3, 117–124. [Google Scholar] [CrossRef]
- Chuacharoen, T.; Sabliov, C.M. Comparative effects of curcumin when delivered in a nanoemulsion or nanoparticle form for food applications: Study on stability and lipid oxidation inhibition. LWT 2019, 113, 108319. [Google Scholar] [CrossRef]
- Marcolino, V.A.; Zanin, G.M.; Durrant, L.R.; Benassi, M.D.T.; Matioli, G. Interaction of Curcumin and Bixin with β-Cyclodextrin: Complexation Methods, Stability, and Applications in Food. J. Agric. Food Chem. 2011, 59, 3348–3357. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Liaqat, I.; Hyder, M.Z.; Akhtar, S.; Bhatti, A.H.; Butt, S.B.; Imran, Z.; Yasmin, T.; Abbas, S. Elucidation of larvicidal potential of metallic and environment friendly food-grade nanostructures against Aedes albopictus. Environ. Geochem. Health 2020, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Olivo, P.M.; Scapim, M.R.D.S.; Miazaki, J.; Madrona, G.S.; Maia, L.F.; Rodrigues, B.M.; Pozza, M.S.D.S. Sodium alginate with turmeric coating for ripened cheeses. J. Food Sci. Technol. 2020, 57, 2364–2369. [Google Scholar] [CrossRef] [PubMed]
- Matche, R.S.; Anup, G.J.; Mrudula, G. Development of Biodegradable Films from Marine Ingredients Incorporated with Natural Antimicrobial Agents for Food Packaging. J. Packag. Technol. Res. 2020, 4, 45–55. [Google Scholar] [CrossRef]
- Dalvandi, F.; Almasi, H.; Ghanbarzadeh, B.; Hosseini, H.; Khosroshahi, N.K. Effect of vacuum packaging and edible coating containing black pepper seeds and turmeric extracts on shelf life extension of chicken breast fillets. JFBE 2020, 3, 69–78. [Google Scholar]
- Li, T.; Zhao, Y.; Matthews, K.; Gao, J.; Hao, J.; Wang, S.; Han, J.; Jia, Y. Antibacterial activity against Staphylococcus aureus of curcumin-loaded chitosan spray coupled with photodynamic treatment. LWT 2020, 134, 110073. [Google Scholar] [CrossRef]
- Luo, X.; Lim, L.-T. Curcumin-loaded electrospun nonwoven as a colorimetric indicator for volatile amines. LWT 2020, 128, 109493. [Google Scholar] [CrossRef]
- Balbinot-Alfaro, E.; Craveiro, D.V.; Lima, K.O.; Costa, H.L.G.; Lopes, D.R.; Prentice, C. Intelligent Packaging with pH Indicator Potential. Food Eng. Rev. 2019, 11, 235–244. [Google Scholar] [CrossRef]
- Halonen, N.; Pálvölgyi, P.S.; Bassani, A.; Fiorentini, C.; Nair, R.; Spigno, G.; Kordas, K. Bio-Based Smart Materials for Food Packaging and Sensors—A Review. Front. Mater. 2020, 7, 82. [Google Scholar] [CrossRef]
- Ma, X.; Chen, Y.; Huang, J.; Lv, P.; Hussain, T.; Wei, Q. In Situ formed active and intelligent bacterial cellulose/cotton fiber composite containing curcumin. Cellulose 2020, 27, 1–12. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Antioxidant and antimicrobial poly (vinyl alcohol)-based films incorporated with grapefruit seed extract and curcumin. J. Environ. Chem. Eng. 2021, 9, 104694. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Carboxymethyl cellulose-based antioxidant and antimicrobial active packaging film incorporated with curcumin and zinc oxide. Int. J. Biol. Macromol. 2020, 148, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Gao, R.; Wang, L.; Xu, M.; Yuan, Y.; Ma, L.; Wan, Z.; Yang, X. Nanocomposites of Bacterial Cellulose Nanofibrils and Zein Nanoparticles for Food Packaging. ACS Appl. Nano Mater. 2020, 3, 2899–2910. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Guo, M.; Jin, T.Z.; Arabi, S.A.; He, Q.; Ismail, B.B.; Hu, Y.; Liu, D. Antimicrobial and UV Blocking Properties of Composite Chitosan Films with Curcumin Grafted Cellulose Nanofiber. Food Hydrocoll. 2021, 112, 106337. [Google Scholar] [CrossRef]
- Zhou, Q.; Gao, Y.; Wang, X.; Liu, R.; Du, P.; Wang, X.; Zhang, X.; Lu, S.; Wang, Z.; Shi, Q.; et al. Nosocomial infections among patients with COVID-19, SARS and MERS: A rapid review and meta-analysis. Ann. Transl. Med. 2020, 8, 629. [Google Scholar] [CrossRef] [PubMed]
- Bardi, T.; Pintado, V.; Gomez-Rojo, M.; Escudero-Sanchez, R.; Lopez, A.A.; Diez-Remesal, Y.; Castro, N.M.; Ruiz-Garbajosa, P.; Pestaña, D. Nosocomial infections associated to COVID-19 in the intensive care unit: Clinical characteristics and outcome. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 495–502. [Google Scholar] [CrossRef]
- Wake, R.M.; Morgan, M.; Choi, J.; Winn, S. Reducing nosocomial transmission of COVID-19: Implementation of a COVID-19 triage system. Clin. Med. 2020, 20, e141–e145. [Google Scholar] [CrossRef]
- Abbas, M.; Nunes, T.R.; Martischang, R.; Zingg, W.; Iten, A.; Pittet, D.; Harbarth, S. Nosocomial transmission and outbreaks of coronavirus disease 2019: The need to protect both patients and healthcare workers. Antimicrob. Resist. Infect. Control 2021, 10, 1–13. [Google Scholar] [CrossRef]
- Spice It Up: 7 Turmeric Benefits For Skin. Available online: www.neutrogena.com/the-bar/spice-it-up-7-turmeric-benefits-for-skin.html (accessed on 17 January 2021).
- Turmeric. Available online: www.lorealparisusa.com/ingredient-library/turmeric.aspx (accessed on 17 January 2021).
- Zheng, Y.; Pan, C.; Zhang, Z.; Luo, W.; Liang, X.; Shi, Y.; Liang, L.; Zheng, X.; Zhang, L.; Du, Z. Antiaging effect of Curcuma longa L. essential oil on ultraviolet-irradiated skin. Microchem. J. 2020, 154, 104608. [Google Scholar] [CrossRef]
- Sienkiewicz, N.; Członka, S.; Kairyte, A.; Vaitkus, S. Curcumin as a natural compound in the synthesis of rigid polyurethane foams with enhanced mechanical, antibacterial and anti-ageing properties. Polym. Test. 2019, 79, 106046. [Google Scholar] [CrossRef]
- Vater, C.; Hlawaty, V.; Werdenits, P.; Cichoń, M.A.; Klang, V.; Elbe-Bürger, A.; Wirth, M.; Valenta, C. Effects of lecithin-based nanoemulsions on skin: Short-time cytotoxicity MTT and BrdU studies, skin penetration of surfactants and additives and the delivery of curcumin. Int. J. Pharm. 2020, 580, 119209. [Google Scholar] [CrossRef] [PubMed]
- Rapalli, V.K.; Kaul, V.; Waghule, T.; Gorantla, S.; Sharma, S.; Roy, A.; Dubey, S.K.; Singhvi, G. Curcumin loaded nanostructured lipid carriers for enhanced skin retained topical delivery: Optimization, scale-up, in-vitro characterization and assessment of ex-vivo skin deposition. Eur. J. Pharm. Sci. 2020, 152, 105438. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Z.; Xiao, L.; Ding, Z.; He, J.; Lu, G.; Lu, Q.; Kaplan, D.L. Natural Nanofiber Shuttles for Transporting Hydrophobic Cargo into Aqueous Solutions. Biomacromolecules 2019, 21, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.T.; Lee, S.; Kim, J.S.; Jeong, J.; Jeon, B.S.; Lee, J.W.; Kim, J.H.; Kim, J. Highly Stable and Fine-Textured Hybrid Microspheres for Entrapment of Cosmetic Active Ingredients. ACS Omega 2020, 5, 29577–29584. [Google Scholar] [CrossRef] [PubMed]
- Al-Akayleh, F.; Al-Naji, I.; Adwan, S.; Al-Remawi, M.; Shubair, M. Enhancement of Curcumin Solubility Using a Novel Solubilizing Polymer Soluplus®. J. Pharm. Innov. 2020, 1–13. [Google Scholar] [CrossRef]
- Wang, F.; Li, Y.; Yu, L.; Zhu, J.; Zhang, F.; Linhardt, R.J. Amphiphilic mPEG-Modified Oligo-Phenylalanine Nanoparticles Chemoenzymatically Synthesized via Papain. ACS Omega 2020, 5, 30336–30347. [Google Scholar] [CrossRef]
- Enumo, A.; Argenta, D.F.; Bazzo, G.C.; Caon, T.; Stulzer, H.K.; Parize, A.L. Development of curcumin-loaded chitosan/pluronic membranes for wound healing applications. Int. J. Biol. Macromol. 2020, 163, 167–179. [Google Scholar] [CrossRef]
- Huang, Y.; Dan, N.; Dan, W.; Zhao, W. Reinforcement of Polycaprolactone/Chitosan with Nanoclay and Controlled Release of Curcumin for Wound Dressing. ACS Omega 2019, 4, 22292–22301. [Google Scholar] [CrossRef]
- Duan, Y.; Li, K.; Wang, H.; Wu, T.; Zhao, Y.; Li, H.; Tang, H.; Yang, W. Preparation and evaluation of curcumin grafted hyaluronic acid modified pullulan polymers as a functional wound dressing material. Carbohydr. Polym. 2020, 238, 116195. [Google Scholar] [CrossRef]
- Liao, H.T.; Lai, Y.-T.; Kuo, C.-Y.; Chen, J.-P. A bioactive multi-functional heparin-grafted aligned poly(lactide-co-glycolide)/curcumin nanofiber membrane to accelerate diabetic wound healing. Mater. Sci. Eng. C 2021, 120, 111689. [Google Scholar] [CrossRef]
- Kaolaor, A.; Phunpee, S.; Ruktanonchai, U.R.; Suwantong, O. Effects of β-cyclodextrin complexation of curcumin and quaternization of chitosan on the properties of the blend films for use as wound dressings. J. Polym. Res. 2019, 26, 43. [Google Scholar] [CrossRef]
- Mohanty, C.; Pradhan, J. A human epidermal growth factor-curcumin bandage bioconjugate loaded with mesenchymal stem cell for in vivo diabetic wound healing. Mater. Sci. Eng. C 2020, 111, 110751. [Google Scholar] [CrossRef]
- Pandey, V.K.; Ajmal, G.; Upadhyay, S.N.; Mishra, P.K. Nano-fibrous scaffold with curcumin for anti-scar wound healing. Int. J. Pharm. 2020, 589, 119858. [Google Scholar] [CrossRef]
- Naghshineh, N.; Tahvildari, K.; Nozari, M. Preparation of Chitosan, Sodium Alginate, Gelatin and Collagen Biodegradable Sponge Composites and their Application in Wound Healing and Curcumin Delivery. J. Polym. Environ. 2019, 27, 2819–2830. [Google Scholar] [CrossRef]
- Gupta, A.; Briffa, S.M.; Swingler, S.; Gibson, H.; Kannappan, V.; Adamus, G.; Kowalczuk, M.M.; Martin, C.; Radecka, I. Synthesis of Silver Nanoparticles Using Curcumin-Cyclodextrins Loaded into Bacterial Cellulose-Based Hydrogels for Wound Dressing Applications. Biomacromolecules 2020, 21, 1802–1811. [Google Scholar] [CrossRef] [PubMed]
- El-Nahhal, I.M.; Salem, J.; Anbar, R.; Kodeh, F.S.; Elmanama, A. Preparation and antimicrobial activity of ZnO-NPs coated cotton/starch and their functionalized ZnO-Ag/cotton and Zn (II) curcumin/cotton materials. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, C.; Varaprasad, K.; Akbari-Fakhrabadi, A.; Hameed, A.S.H.; Sadiku, R. Biomolecule chitosan, curcumin and ZnO-based antibacterial nanomaterial, via a one-pot process. Carbohydr. Polym. 2020, 249, 116825. [Google Scholar] [CrossRef]
- Anagha, B.; George, D.; Maheswari, P.U.; Begum, K.M.M.S. Biomass derived antimicrobial hybrid cellulose hydrogel with green ZnO nanoparticles for curcumin delivery and its kinetic modelling. J. Polym. Environ. 2019, 27, 2054–2067. [Google Scholar] [CrossRef]
- Marulasiddeshwara, R.; Jyothi, M.; Soontarapa, K.; Keri, R.S.; Velmurugan, R. Nonwoven fabric supported, chitosan membrane anchored with curcumin/TiO2 complex: Scaffolds for MRSA infected wound skin reconstruction. Int. J. Biol. Macromol. 2020, 144, 85–93. [Google Scholar] [CrossRef]
- Konwar, A.; Kandimalla, R.; Kalita, S.; Chowdhury, D. Approach to Fabricate a Compact Cotton Patch without Weaving: A Smart Bandage Material. ACS Sustain. Chem. Eng. 2018, 6, 5806–5817. [Google Scholar] [CrossRef]
- Kuddushi, M.; Patel, N.K.; Gawali, S.L.; Mata, J.P.; Montes-Campos, H.; Varela, L.M.; Hassan, P.A.; Malek, N.I. Thermo-switchable de novo ionogel as metal absorbing and curcumin loaded smart bandage material. J. Mol. Liq. 2020, 306, 112922. [Google Scholar] [CrossRef]
- Ahmed, S.S.Z.; Balu, N.; Khader, S.Z.A.; Mahboob, M.R.; Lakshmanan, S.O.; Vetrivel, M. Fabrication and evaluation of bamboo fabric coated with extracts of Curcuma longa, Centella asiatica and Azadirachta indica as a wound dressing material. Adv. Tradit. Med. 2021, 21, 83–95. [Google Scholar] [CrossRef]
- Kulkarni, A.S.; Gurav, D.D.; Khan, A.A.; Shinde, V.S. Curcumin loaded nanofibrous mats for wound healing application. Colloids Surf. B Biointerfaces 2020, 189, 110885. [Google Scholar] [CrossRef]
- Tangsatianpan, V.; Torgbo, S.; Sukyai, P. Release Kinetic Model and Antimicrobial Activity of Freeze-Dried Curcumin-loaded Bacterial Nanocellulose Composite. Polym. Sci. Ser. A 2020, 62, 218–227. [Google Scholar] [CrossRef]
- Mohanty, C.; Das, M.; Sahoo, S.K. Sustained Wound Healing Activity of Curcumin Loaded Oleic Acid Based Polymeric Bandage in a Rat Model. Mol. Pharm. 2012, 9, 2801–2811. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, X.; Pi, C.; Yang, H.; Zheng, X.; Zhao, L.; Wei, Y. Review of Curcumin Physicochemical Targeting Delivery System. Int. J. Nanomed. 2020, 15, 9799–9821. [Google Scholar] [CrossRef]
- Zheng, B.; McClements, D.J. Formulation of More Efficacious Curcumin Delivery Systems Using Colloid Science: Enhanced Solubility, Stability, and Bioavailability. Molecules 2020, 25, 2791. [Google Scholar] [CrossRef]
- Al-Soliemy, A.; Al-Zahrani, F. Synthesis of novel disperse dyes based on curcumin for the creation of antibacterial polyester fabrics. Pigment. Resin Technol. 2019, 48, 502–507. [Google Scholar] [CrossRef]
- Atlan, M.; Neman, J. Targeted Transdermal Delivery of Curcumin for Breast Cancer Prevention. Int. J. Environ. Res. Public Health 2019, 16, 4949. [Google Scholar] [CrossRef]
- Sharma, N.; Nehru, B. Curcumin affords neuroprotection and inhibits α-synuclein aggregation in lipopolysaccharide-induced Parkinson’s disease model. Inflammopharmacology 2018, 26, 349–360. [Google Scholar] [CrossRef]
- Ferreira, N.; Saraiva, M.J.; Almeida, M.R. Uncovering the Neuroprotective Mechanisms of Curcumin on Transthyretin Amyloidosis. Int. J. Mol. Sci. 2019, 20, 1287. [Google Scholar] [CrossRef]
- Lin, X.; Watanabe, K.; Kuragano, M.; Tokuraku, K. Aggregation of Mouse Serum Amyloid A Protein Was Promoted by Amyloid-Enhancing Factors with the More Genetically Homologous Serum Amyloid A. Int. J. Mol. Sci. 2021, 22, 1036. [Google Scholar] [CrossRef]
- Gao, C.; Chu, X.; Gong, W.; Zheng, J.; Xie, X.; Wang, Y.; Yang, M.; Li, Z.; Gao, C.; Yang, Y. Neuron tau-targeting biomimetic nanoparticles for curcumin delivery to delay progression of Alzheimer’s disease. J. Nanobiotechnol. 2020, 18, 1–23. [Google Scholar] [CrossRef]
- Mansouri, K.; Rasoulpoor, S.; Daneshkhah, A.; Abolfathi, S.; Salari, N.; Mohammadi, M.; Shabani, S. Clinical effects of curcumin in enhancing cancer therapy: A systematic review. BMC Cancer 2020, 20, 1–11. [Google Scholar] [CrossRef]
- Rizeq, B.; Gupta, I.; Ilesanmi, J.; Alsafran, M.; Rahman, M.; Ouhtit, A. The Power of Phytochemicals Combination in Cancer Chemoprevention. J. Cancer 2020, 11, 4521–4533. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, J.; Jiang, B.; Guo, J. The roles of curcumin in regulating the tumor immunosuppressive microenvironment (Review). Oncol. Lett. 2020, 19, 3059–3070. [Google Scholar] [CrossRef]
- Willenbacher, E.; Khan, S.Z.; Mujica, S.C.A.; Trapani, D.; Hussain, S.; Wolf, D.; Willenbacher, W.; Spizzo, G.; Seeber, A. Curcumin: New Insights into an Ancient Ingredient against Cancer. Int. J. Mol. Sci. 2019, 20, 1808. [Google Scholar] [CrossRef]
- Gbenga, A.A.; Mwakikunga, A. Lopinavir and curcumin directly alters BAX/BCL2 and VEGF165b mRNA levels to suppress human squamous cervical carcinoma cell growth. Int. J. Morphol. 2019, 37, 584–591. [Google Scholar] [CrossRef]
- Shaikh, S.; Shaikh, J.; Naba, Y.S.; Doke, K.; Ahmed, K.; Yusufi, M. Curcumin: Reclaiming the lost ground against cancer resistance. Cancer Drug Resist. 2021, 4. [Google Scholar] [CrossRef]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X. A Review of Curcumin and Its Derivatives as Anticancer Agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef] [PubMed]
- Manzanares-Guevara, L.A.; Licea-Claverie, A.; Oroz-Parra, I.; Bernaldez-Sarabia, J.; Diaz-Castillo, F.; Licea-Navarro, A.F. Smart Nanoformulation Based on Stimuli-Responsive Nanogels and Curcumin: Promising Therapy against Colon Cancer. ACS Omega 2020, 5, 9171–9184. [Google Scholar] [CrossRef]
- Pandey, A.; Chaturvedi, M.; Mishra, S.; Kumar, P.; Somvanshi, P.; Chaturvedi, R. Reductive metabolites of curcumin and their therapeutic effects. Heliyon 2020, 6, e05469. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-B.; Luo, D.-D.; Xie, J.-H.; Xian, Y.-F.; Lai, Z.-Q.; Liu, Y.-H.; Liu, W.-H.; Chen, J.-N.; Lai, X.-P.; Lin, Z.-X.; et al. Curcumin’s Metabolites, Tetrahydrocurcumin and Octahydrocurcumin, Possess Superior Anti-inflammatory Effects in vivo Through Suppression of TAK1-NF-κB Pathway. Front. Pharmacol. 2018, 9, 1181. [Google Scholar] [CrossRef]
- Hadizadeh-Bazaz, M.; Vaezi, G.; Khaksari, M.; Hojati, V. Curcumin attenuates spatial memory impairment by anti-oxidative, anti-apoptosis, and anti-inflammatory mechanism against methamphetamine neurotoxicity in male Wistar rats: Histological and biochemical changes. NeuroToxicology 2021, 84, 208–217. [Google Scholar] [CrossRef]
- Mandal, M.; Jaiswal, P.; Mishra, A. Curcumin loaded nanoparticles reversed monocrotophos induced motor impairment and memory deficit: Role of oxidative stress and intracellular calcium level. J. Drug Deliv. Sci. Technol. 2020, 56, 101559. [Google Scholar] [CrossRef]
- Laabbar, W.; Abbaoui, A.; Elgot, A.; Mokni, M.; Amri, M.; Masmoudi-Kouki, O.; Gamrani, H. Aluminum induced oxidative stress, astrogliosis and cell death in rat astrocytes, is prevented by curcumin. J. Chem. Neuroanat. 2021, 112, 101915. [Google Scholar] [CrossRef]
- Hussein, Y.A.; Al-Sarraf, A.M.; Alfalluji, W.L. Modulation of oxidative stress, inflammatory and apoptotic response by curcumin against cerebral ischemia reperfusion injury in a mouse model. Interdiscip. Neurosurg. 2020, 21, 100741. [Google Scholar] [CrossRef]
- Ahmadabady, S.; Beheshti, F.; Shahidpour, F.; Khordad, E.; Hosseini, M. A protective effect of curcumin on cardiovascular oxidative stress indicators in systemic inflammation induced by lipopolysaccharide in rats. Biochem. Biophys. Rep. 2021, 25, 100908. [Google Scholar] [CrossRef]
- Prathipati, B.; Rohini, P.; Kola, P.K.; Danduga, R.C.S.R. Neuroprotective effects of curcumin loaded solid lipid nanoparticles on homocysteine induced oxidative stress in vascular dementia. CRBS 2021, 2, 100029. [Google Scholar]
- Fadeel, D.A.A.; Kamel, R.; Fadel, M. PEGylated lipid nanocarrier for enhancing photodynamic therapy of skin carcinoma using curcumin: In-Vitro/In-Vivo studies and histopatho-logical examination. Sci. Rep. 2020, 10, 10435. [Google Scholar] [CrossRef]
- Baghdan, E.; Duse, L.; Schüer, J.J.; Pinnapireddy, S.R.; Pourasghar, M.; Schäfer, J.; Schneider, M.; Bakowsky, U. Development of inhalable curcumin loaded Nano-in-Microparticles for bronchoscopic photodynamic therapy. Eur. J. Pharm. Sci. 2019, 132, 63–71. [Google Scholar] [CrossRef]
- Su, W.; Wei, T.; Lu, M.; Meng, Z.; Chen, X.; Jing, J.; Li, J.; Yao, W.; Zhu, H.; Fu, T. Treatment of metastatic lung cancer via inhalation administration of curcumin composite particles based on mesoporous silica. Eur. J. Pharm. Sci. 2019, 134, 246–255. [Google Scholar] [CrossRef]
- Kazantzis, C.; Koutsonikoli, K.; Mavroidi, B.; Zachariadis, M.; Alexiou, P.; Pelecanou, M.; Politopoulos, K.; Alexandratou, E.; Sagnou, M. Curcumin derivatives as photosensitizers in photodynamic therapy: Photophysical properties and in vitro studies with prostate cancer cells. Photochem. Photobiol. Sci. 2020, 19, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Kamel, A.E.; Fadel, M.; Louis, D. Curcumin-loaded nanostructured lipid carriers prepared using Peceol™ and olive oil in photodynamic therapy: Development and application in breast cancer cell line. Int. J. Nanomed. 2019, 14, 5073–5085. [Google Scholar] [CrossRef] [PubMed]
- Ambreen, G.; Duse, L.; Tariq, I.; Ali, U.; Ali, S.; Pinnapireddy, S.R.; Bette, M.; Bakowsky, U.; Mandic, R. Sensitivity of Papilloma Virus-Associated Cell Lines to Photodynamic Therapy with Curcumin-Loaded Liposomes. Cancers 2020, 12, 3278. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.-H.; Yu, K.-H.; Huang, Y.-C.; Lee, C.-I. EGFR-targeted photodynamic therapy by curcumin-encapsulated chitosan/TPP nanoparticles. Int. J. Nanomed. 2018, 13, 903–916. [Google Scholar] [CrossRef]
- Kuang, G.; Zhang, Q.; He, S.; Liu, Y. Curcumin-loaded PEGylated mesoporous silica nanoparticles for effective photodynamic therapy. RSC Adv. 2020, 10, 24624–24630. [Google Scholar] [CrossRef]
- Bhuyan, T.; Singh, A.K.; Ghosh, S.S.; Bandyopadhyay, D. Magnetotactic curcumin iButtonbots as efficient bactericidal agents. Bull. Mater. Sci. 2020, 43, 1–10. [Google Scholar] [CrossRef]
- Singh, R.; Verma, K.; Kumar, R. Core–shell Ag-ZnO/Curcumin nanocomposite having optically active, thermally stable, hydrophilic surfaces for self cleaning applications. Appl. Phys. A 2020, 126, 1–15. [Google Scholar] [CrossRef]
- Ishak, N.A.; Hamidon, T.S.; Zi-Hui, T.; Hussin, M.H. Extracts of curcumin-incorporated hybrid sol–gel coatings for the corrosion mitigation of mild steel in 0.5 M HCl. J. Coatings Technol. Res. 2020, 17, 1515–1535. [Google Scholar] [CrossRef]
- Kashif, M.; Ngaini, Z.; Harry, A.V.; Vekariya, R.L.; Ahmad, A.; Zuo, Z.; Sahari, S.K.; Hussain, S.; Khan, Z.A.; Alarifi, A. An experimental and DFT study on novel dyes incorporated with natural dyes on titanium dioxide (TiO2) towards solar cell application. Appl. Phys. A 2020, 126, 1–13. [Google Scholar] [CrossRef]
- Sakshi; Pathak, N.K.; Swain, B.C.; Tripathy, U. Analyzing nonlinear trends in curcumin: A comparative study. Opt. Laser Technol. 2020, 121, 105822. [Google Scholar] [CrossRef]
- Zhu, C.; Li, R.; Chen, X.; Chalmers, E.; Liu, X.; Wang, Y.; Xu, B.B.; Liu, X. Ultraelasticyarns from curcumin-assisted ELD toward wearable human–machine interface textiles. Adv. Sci. 2020, 7, 2002009. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raduly, F.M.; Raditoiu, V.; Raditoiu, A.; Purcar, V. Curcumin: Modern Applications for a Versatile Additive. Coatings 2021, 11, 519. https://doi.org/10.3390/coatings11050519
Raduly FM, Raditoiu V, Raditoiu A, Purcar V. Curcumin: Modern Applications for a Versatile Additive. Coatings. 2021; 11(5):519. https://doi.org/10.3390/coatings11050519
Chicago/Turabian StyleRaduly, Florentina Monica, Valentin Raditoiu, Alina Raditoiu, and Violeta Purcar. 2021. "Curcumin: Modern Applications for a Versatile Additive" Coatings 11, no. 5: 519. https://doi.org/10.3390/coatings11050519
APA StyleRaduly, F. M., Raditoiu, V., Raditoiu, A., & Purcar, V. (2021). Curcumin: Modern Applications for a Versatile Additive. Coatings, 11(5), 519. https://doi.org/10.3390/coatings11050519










