Protection of Stone Monuments Using a Brushing Treatment with Ammonium Oxalate
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
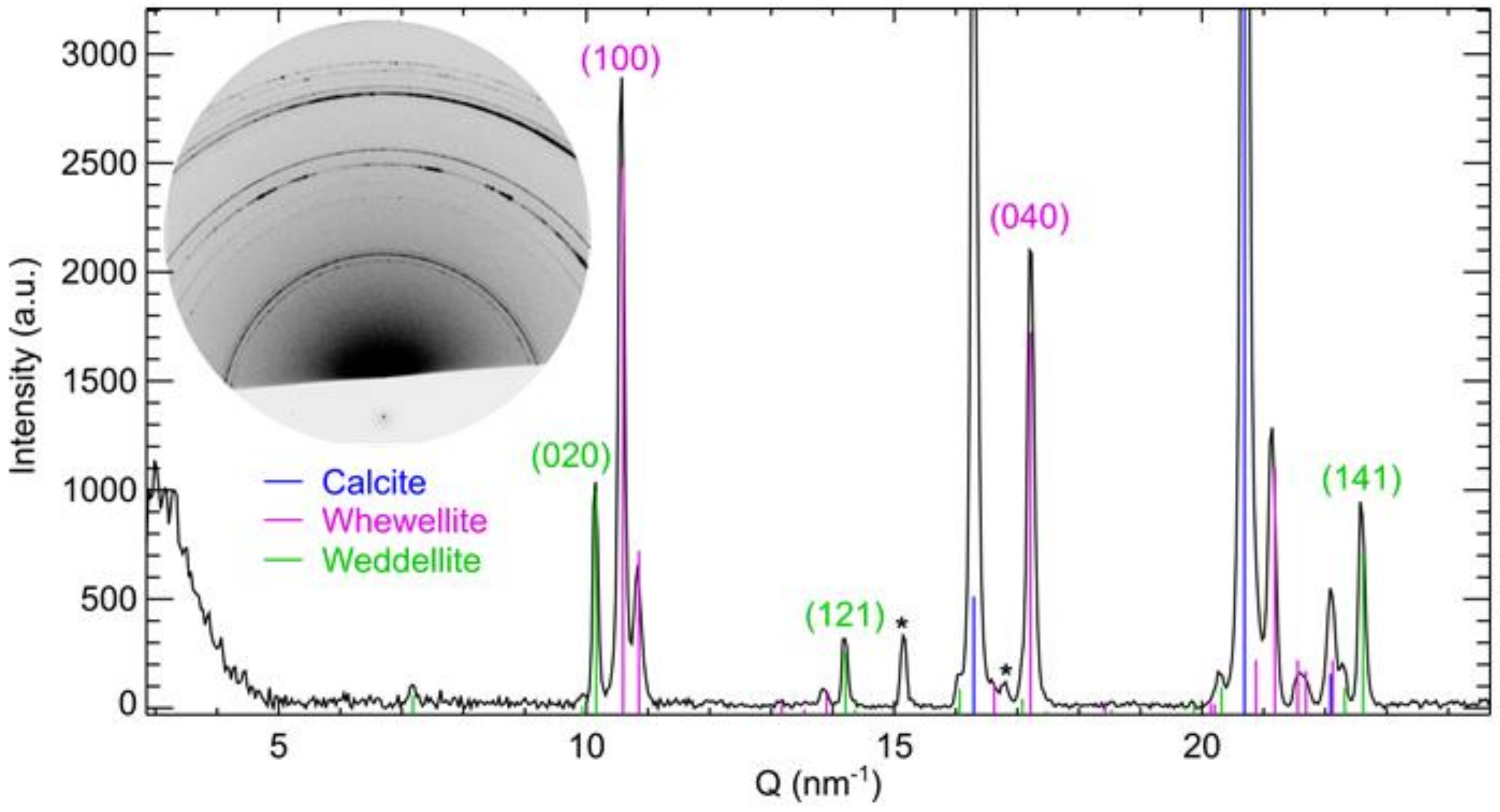
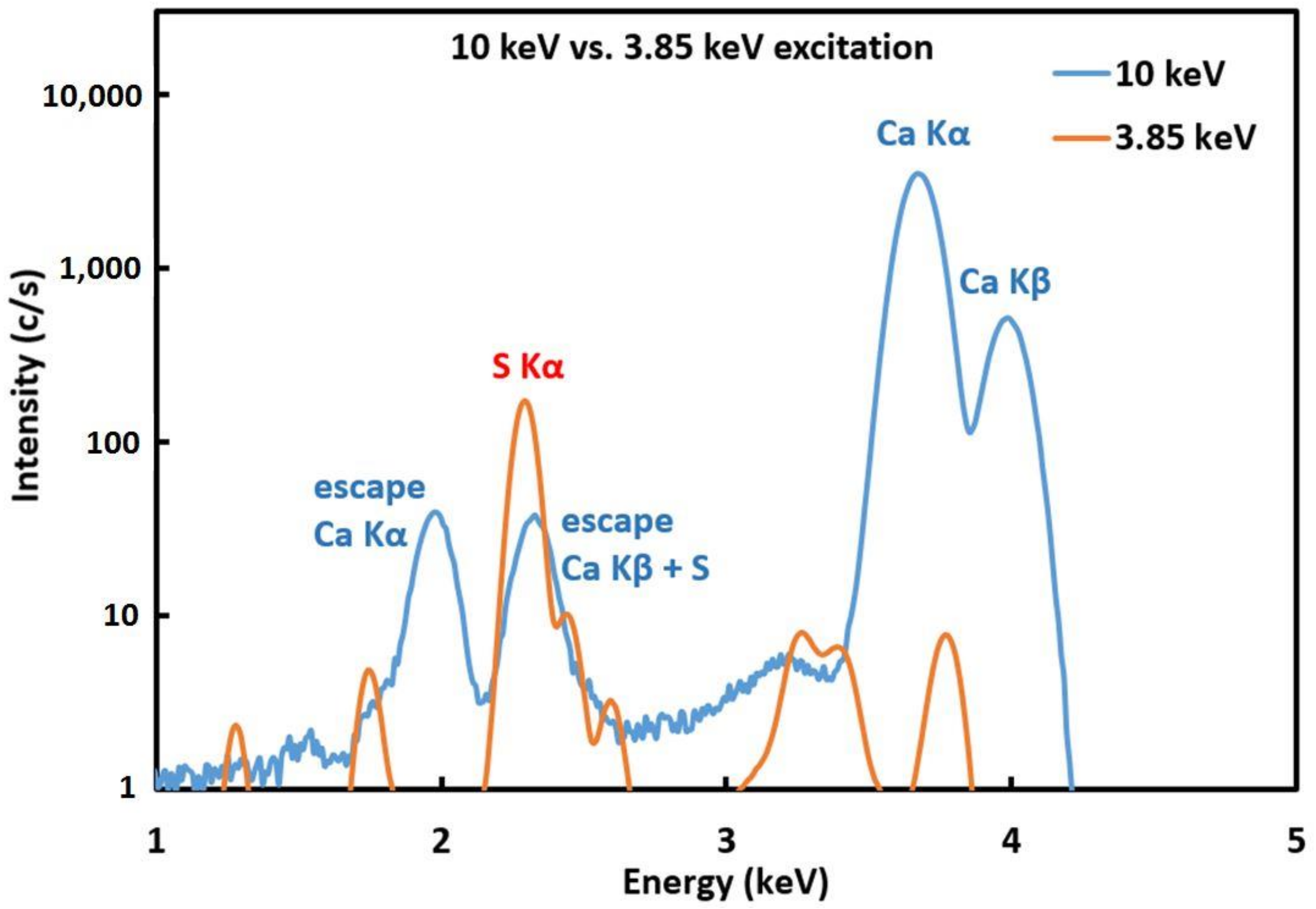
2.3. Analytical Techniques
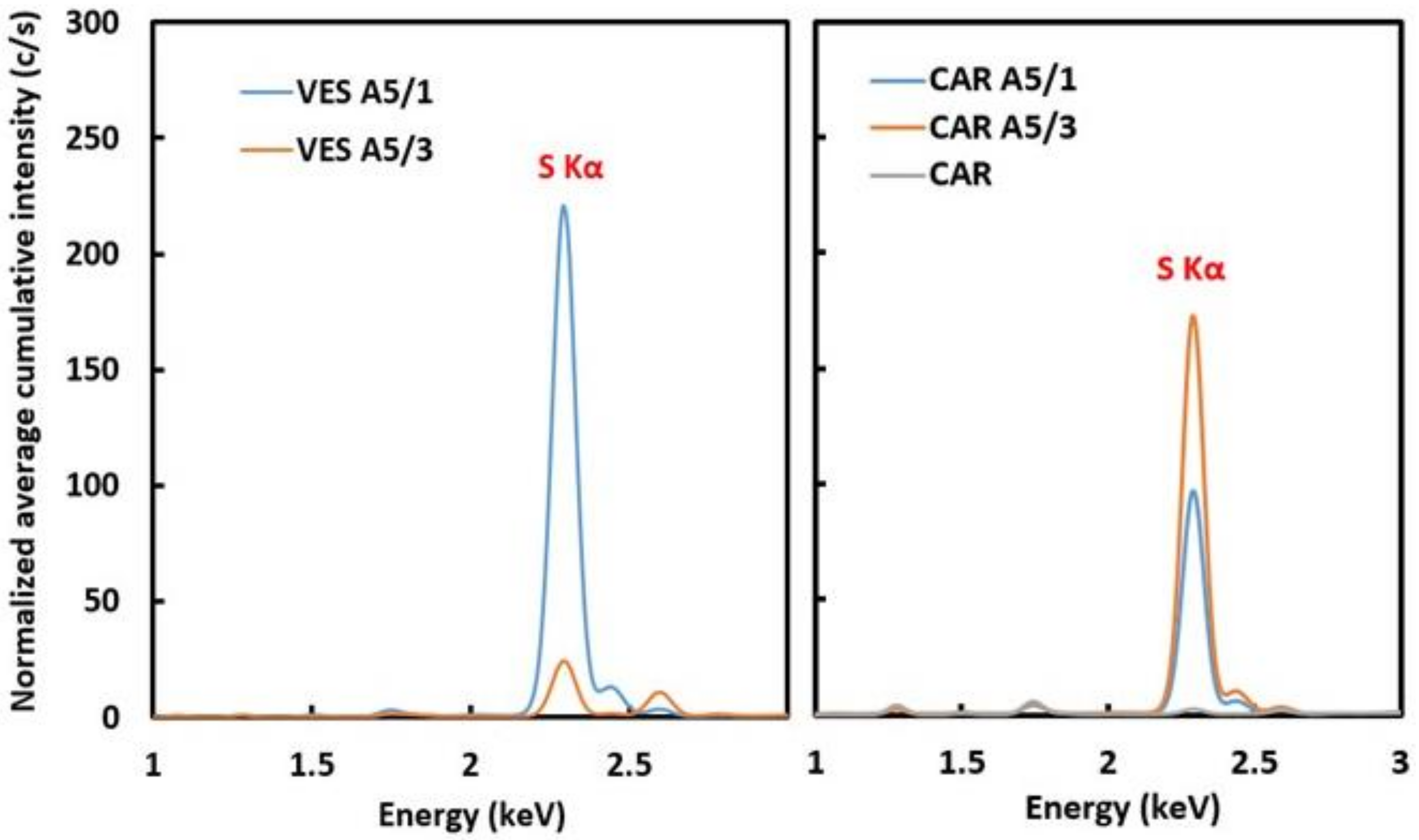
3. Results and Discussion
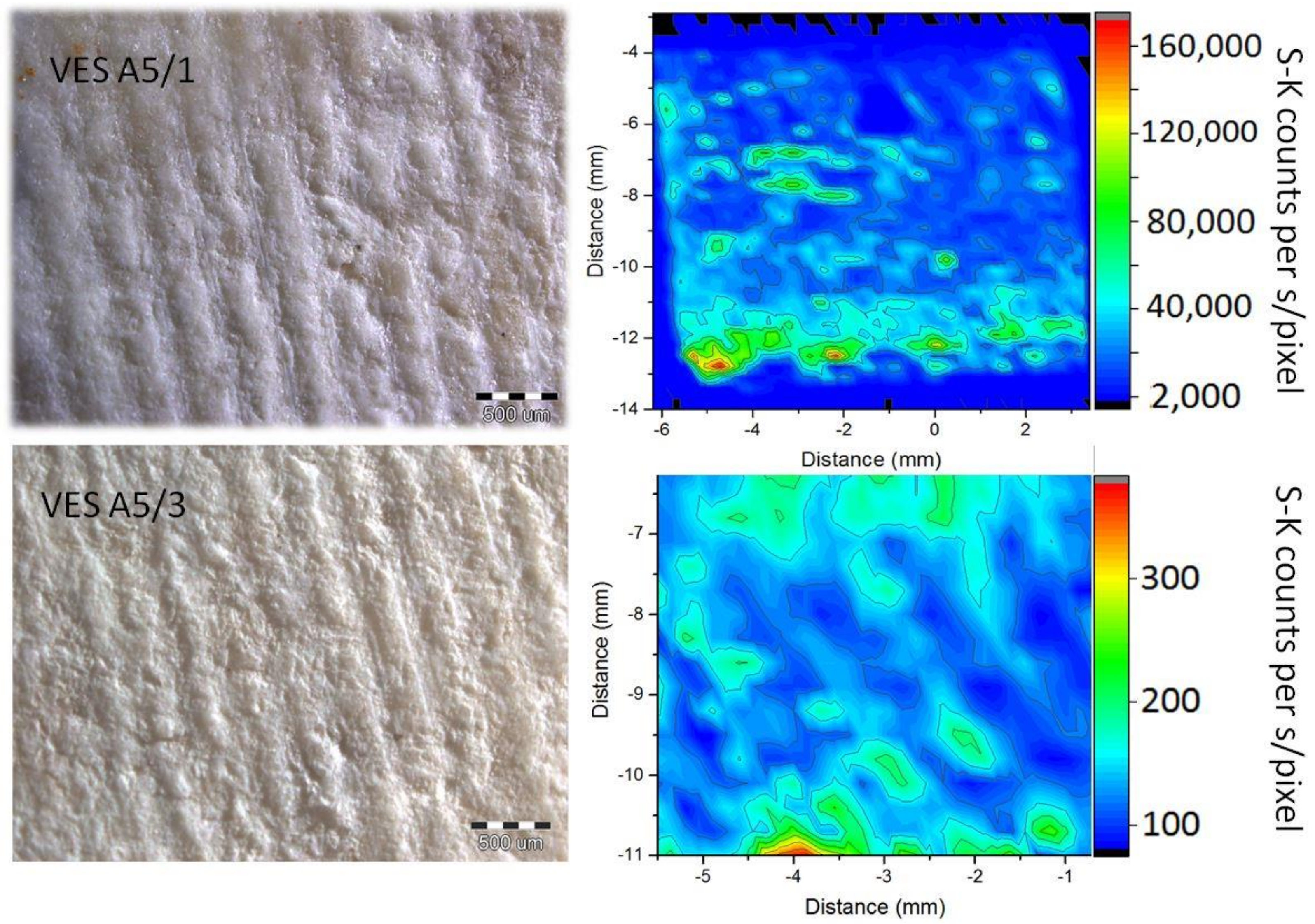
3.1. Colorimetric Analysis of Stone Surface
3.2. Thickness of Oxalate Protective Layers
3.3. Efficiency of Oxalate Protective Layers
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brady, P.V. Physics and Chemistry of Mineral Surfaces; CRC Press: New York, NY, USA, 1996. [Google Scholar]
- Naidu, S.; Blair, J.; Scherer, G.W. Acid-Resistant Coatings on Marble. J. Am. Ceram. Soc. 2016, 99, 3421–3428. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Franzoni, E.; Scherer, G.W. Calcium phosphate coatings for marble conservation: Influence of eth-anol and isopropanol addition to the precipitation medium on the coating microstructure and performance. Corros. Sci. 2018, 136, 255–267. [Google Scholar] [CrossRef]
- Carretti, E.; Dei, L. Physicochemical characterization of acrylic polymeric resins coating porous materials of artistic interest. Prog. Org. Coat. 2004, 49, 282–289. [Google Scholar] [CrossRef]
- Sabatini, V.; Pargoletti, E.; Comite, V.; Ortenzi, M.A.; Fermo, P.; Gulotta, D.; Cappelletti, G. Towards Novel Fluorinated Methacrylic Coatings for Cultural Heritage: A Combined Polymers and Surfaces Chemistry Study. Polymers 2019, 11, 1190. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, V.; Pargoletti, E.; Longoni, M.; Farina, H.; Ortenzi, M.A.; Cappelletti, G. Stearyl methacrylate co-polymers: Towards new polymer coatings for mortars protection. Appl. Surf. Sci. 2019, 488, 213–220. [Google Scholar] [CrossRef]
- Hansen, E.F.; Doehne, E.; Fidler, J.M.; Larson, J.D.; Martin, B.R.; Matteini, M.; Rodriguez-Navarro, C.; Pardo, E.S.; Price, C.; De Tagle, A.; et al. A review of selected inorganic consolidants and protective treatments for porous calcareous materials. Stud. Conserv. 2003, 48, 13–25. [Google Scholar] [CrossRef]
- Matteini, M. Inorganic treatments for the consolidation and protection of stone artefacts. Conserv. Sci. Cult. Herit. 2008, 8, 13–27. [Google Scholar]
- Dreyfuss, T. Interactions on site between powdering porous limestone, natural salt mixtures and applied ammonium oxalate. Herit. Sci. 2019, 7, 5. [Google Scholar] [CrossRef]
- Maiore, L.; Aragoni, M.C.; Carcangiu, G.; Cocco, O.; Isaia, F.; Lippolis, V.; Meloni, P.; Murru, A.; Tuveri, E.; Arca, M. Synthesis, characterization and DFT-modeling of novel agents for the protection and restoration of historical calcareous stone substrates. J. Colloid Interface Sci. 2015, 448, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Cara, A.; Ruiz-Agudo, E.; Rodriguez-Navarro, C. Effectiveness of oxalic acid treatments for the protection of marble surfaces. Mater. Des. 2017, 115, 82–92. [Google Scholar] [CrossRef]
- Sassoni, E. Hydroxyapatite and Other Calcium Phosphates for the Conservation of Cultural Heritage: A Review. Materials 2018, 11, 557. [Google Scholar] [CrossRef] [PubMed]
- Mudronja, D.; Vanmeert, F.; Hellemans, K.; Fazinic, S.; Janssens, K.; Tibljas, D.; Rogosic, M.; Jakovljevic, S. Efficiency of applying ammonium oxalate for protection of monumental limestone by poultice, immersion and brushing methods. Appl. Phys. A 2012, 111, 109–119. [Google Scholar] [CrossRef]
- Vanmeert, F.; Mudronja, D.; Fazinić, S.; Janssens, K.; Tibljas, D. Semi-quantitative analysis of the formation of a calcium oxalate protective layer for monumental limestone using combined micro-XRF and micro-XRPD. X-ray Spectrom. 2013, 42, 256–261. [Google Scholar] [CrossRef]
- Mahy, M.; Van Eycken, L.; Oosterlinck, A. Evaluation of uniform color spaces developed after the adoption of CIELAB and CIELUV. Color Res. Appl. 1994, 19, 105–121. [Google Scholar] [CrossRef]
- Hardeberg, J.Y. Acquisition and Reproduction of Color Images: Colorimetric and Multispectral Approaches; Universal-Publishers: Irvine, CA, USA, 2001; ISBN 1-58112-135-0. [Google Scholar]
- De Nolf, W.; Vanmeert, F.; Janssens, K. XRDUA: Crystalline phase distribution maps by two-dimensional scanning and tomo-graphic (micro) X-ray powder diffraction. J. Appl. Crystallogr. 2014, 47, 1107–1117. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Franzoni, E.; Scherer, G.W. New insights on protective treatments for marble by FIB-SEM. IOP Conf. Ser. Mater. Sci. Eng. 2018, 364, 012092. [Google Scholar] [CrossRef]
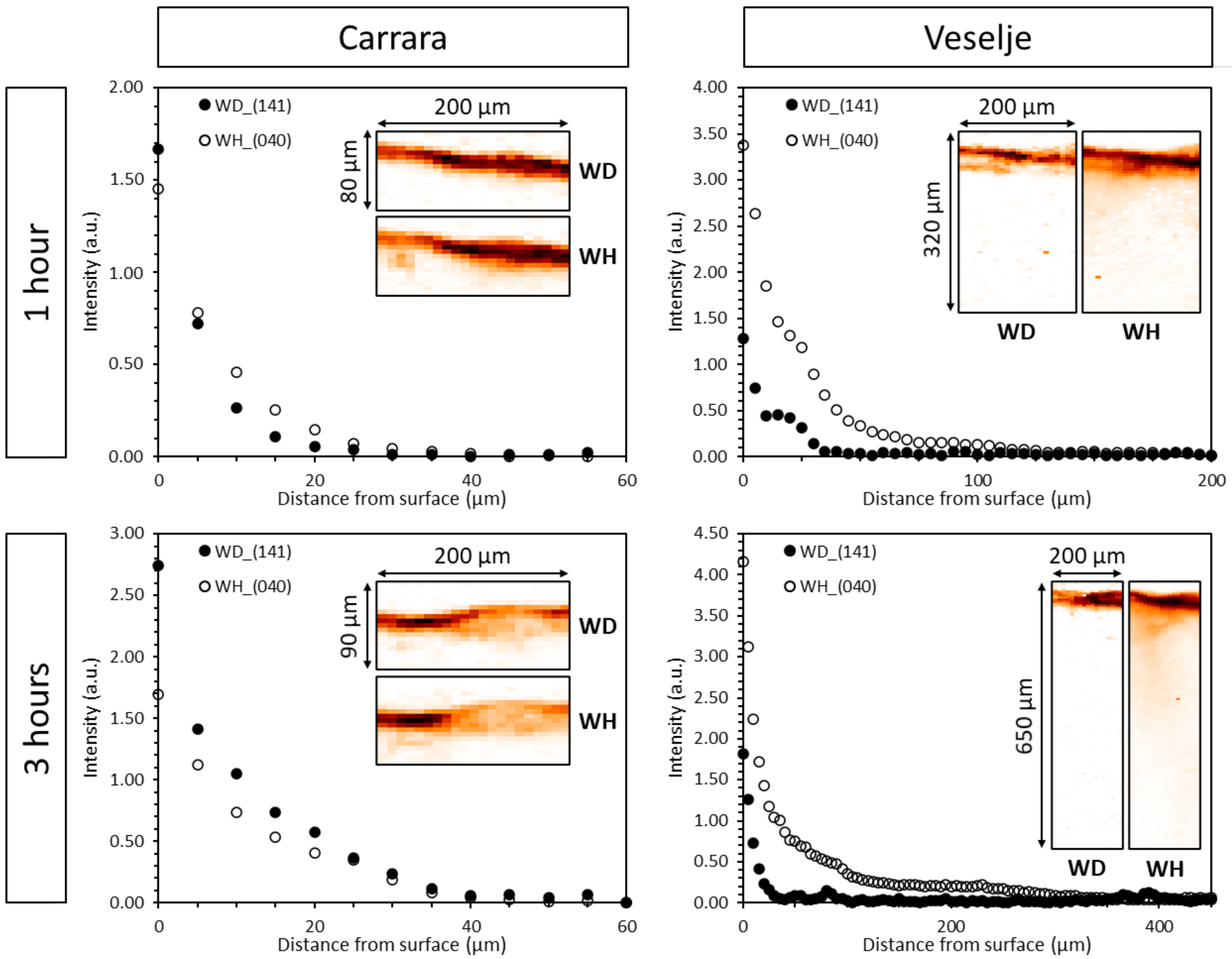
| Oxalate Layer Thickness (µm) | |||||
| WD (020) | WD (121) | WD (141) | WH (100) | WH (040) | |
| CAR_A5_1 | 6 (2) | 6 (2) | 6 (2) | 10 (7) | 8 (3) |
| CAR_A5_3 | 11 (9) | 11 (8) | 13 (9) | 18 (9) | 17 (8) |
| VES_A5_1 | 11 (9) | 12 (9) | 13 (9) | 17 (8) | 15 (7) |
| VES_A5_3 | 9 (3) | 10 (3) | 11 (3) | 12 (3) | 11 (2) |
| Oxalate Penetration Depth (µm) | |||||
| WD (020) | WD (121) | WD (141) | WH (100) | WH (040) | |
| CAR_A5_1 | 12 (2) | 11 (3) | 12 (3) | 21 (6) | 21 (8) |
| CAR_A5_3 | 25 (10) | 24 (7) | 28 (7) | 32 (5) | 29 (5) |
| VES_A5_1 | 21 (10) | 17 (9) | 29 (6) | 80 (27) | 85 (22) |
| VES_A5_3 | 19 (7) | 20 (5) | 22 (5) | 212 (93) | 188 (78) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mudronja, D.; Vanmeert, F.; Fazinic, S.; Janssens, K.; Tibljas, D.; Desnica, V. Protection of Stone Monuments Using a Brushing Treatment with Ammonium Oxalate. Coatings 2021, 11, 379. https://doi.org/10.3390/coatings11040379
Mudronja D, Vanmeert F, Fazinic S, Janssens K, Tibljas D, Desnica V. Protection of Stone Monuments Using a Brushing Treatment with Ammonium Oxalate. Coatings. 2021; 11(4):379. https://doi.org/10.3390/coatings11040379
Chicago/Turabian StyleMudronja, Domagoj, Frederik Vanmeert, Stjepko Fazinic, Koen Janssens, Darko Tibljas, and Vladan Desnica. 2021. "Protection of Stone Monuments Using a Brushing Treatment with Ammonium Oxalate" Coatings 11, no. 4: 379. https://doi.org/10.3390/coatings11040379
APA StyleMudronja, D., Vanmeert, F., Fazinic, S., Janssens, K., Tibljas, D., & Desnica, V. (2021). Protection of Stone Monuments Using a Brushing Treatment with Ammonium Oxalate. Coatings, 11(4), 379. https://doi.org/10.3390/coatings11040379







