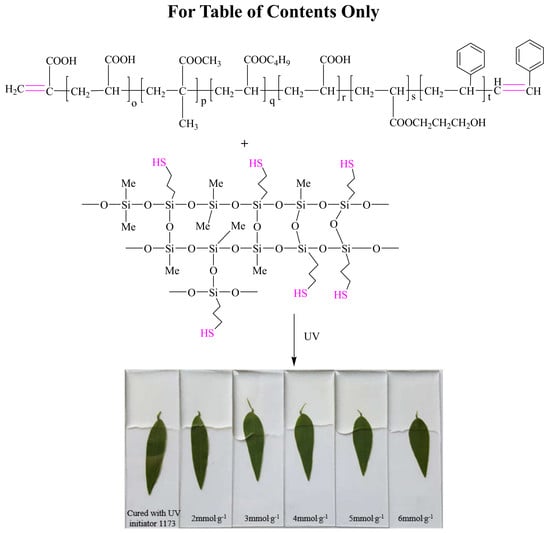
An Easy Fabrication Method to Prepare Inexpensive UV–Cured Transparent Silicone Modified Polyacrylate Coatings with Good Adhesion and UV Resistance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Copolymers of Acrylate Monomers and Styrene
2.3. Preparation of Transparent Materials
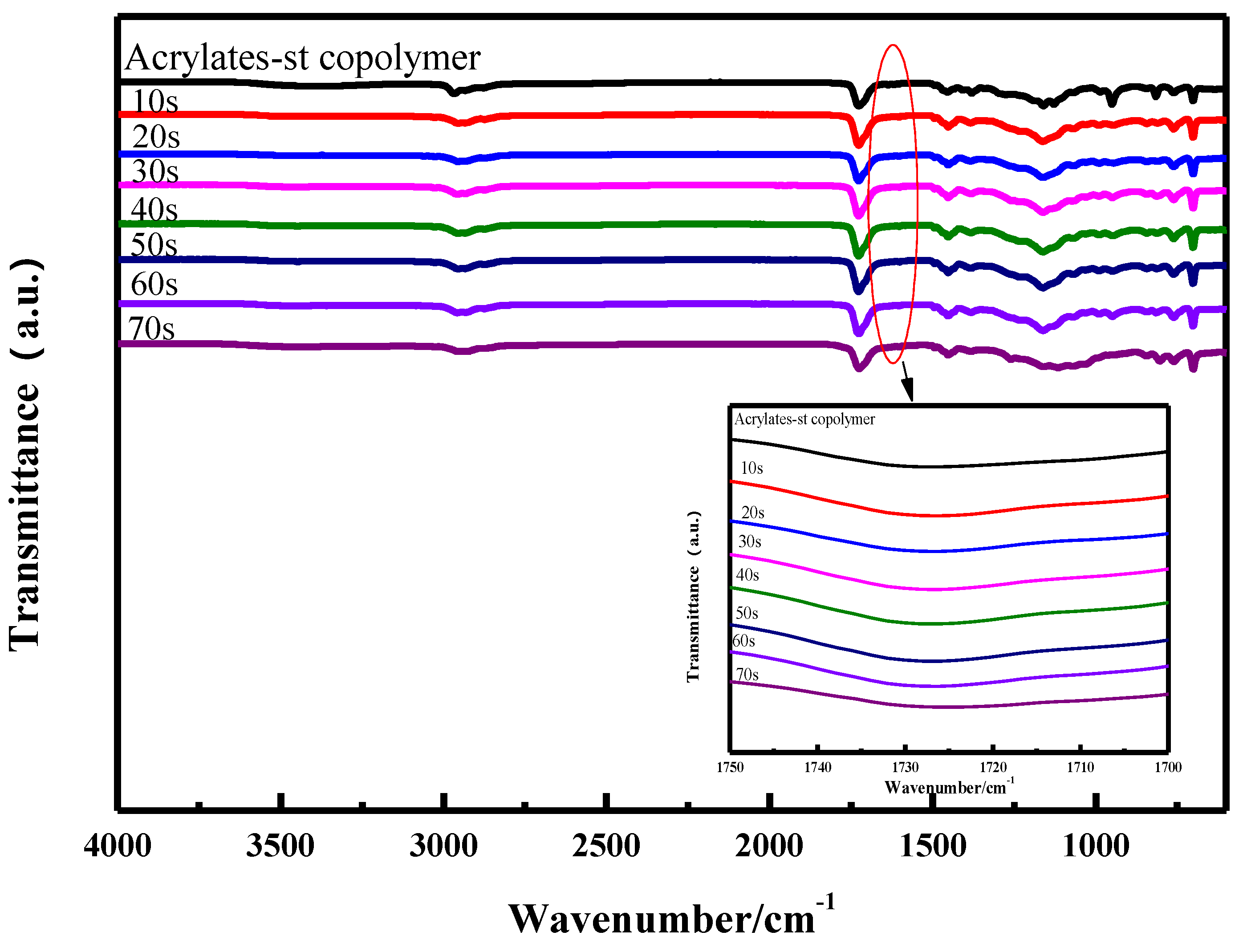
2.4. Characterization
3. Results and Discussion
3.1. UV Curing Behavior
3.1.1. Curing Time
3.1.2. The Amount of Styrene to Prepare the Copolymers
3.1.3. The Thiol Content of Silicone Resin
3.1.4. The Amount of Thiol Silicone Resin
3.2. Performance of UV–cured Coatings
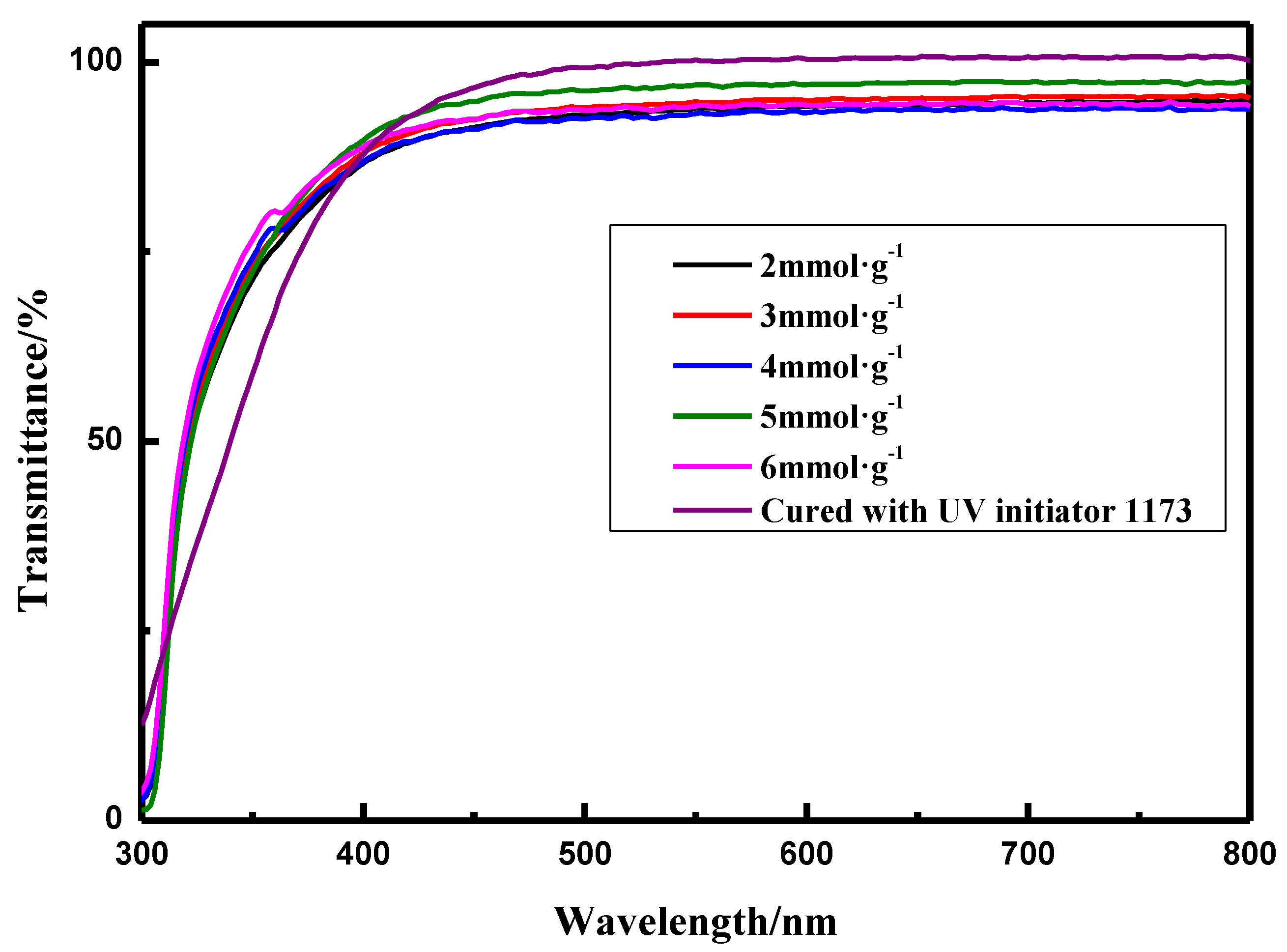
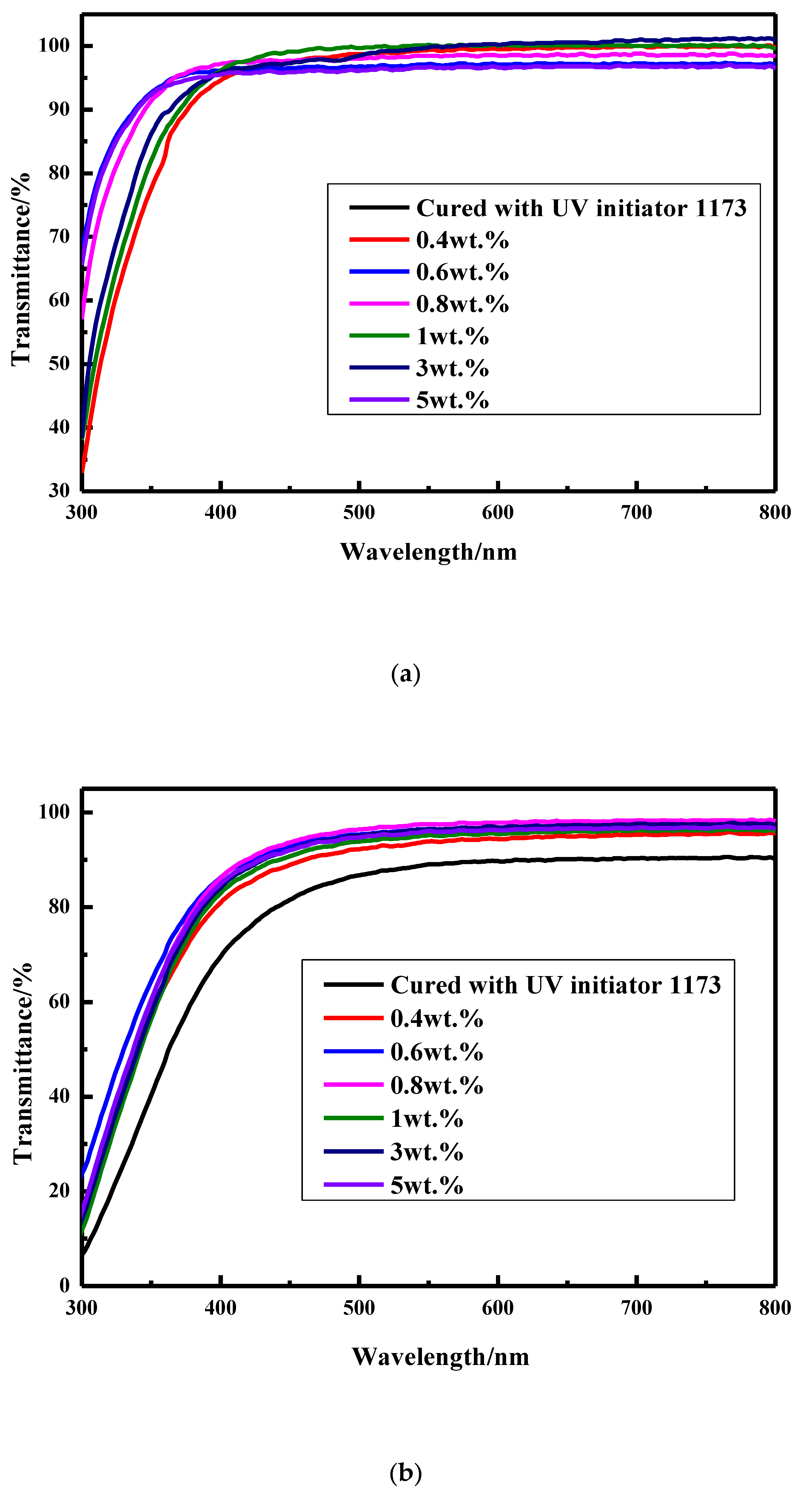
3.2.1. Transparency
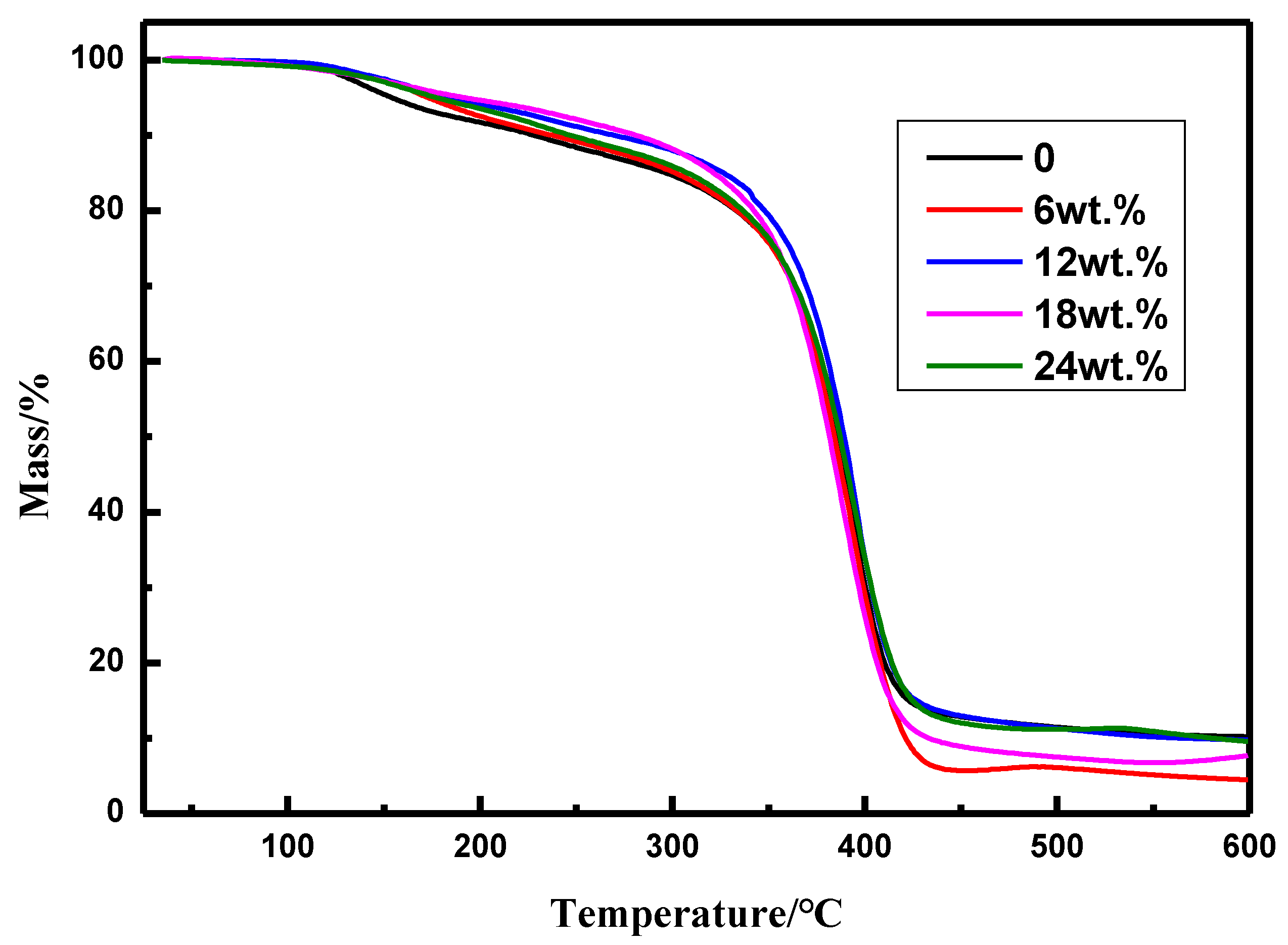
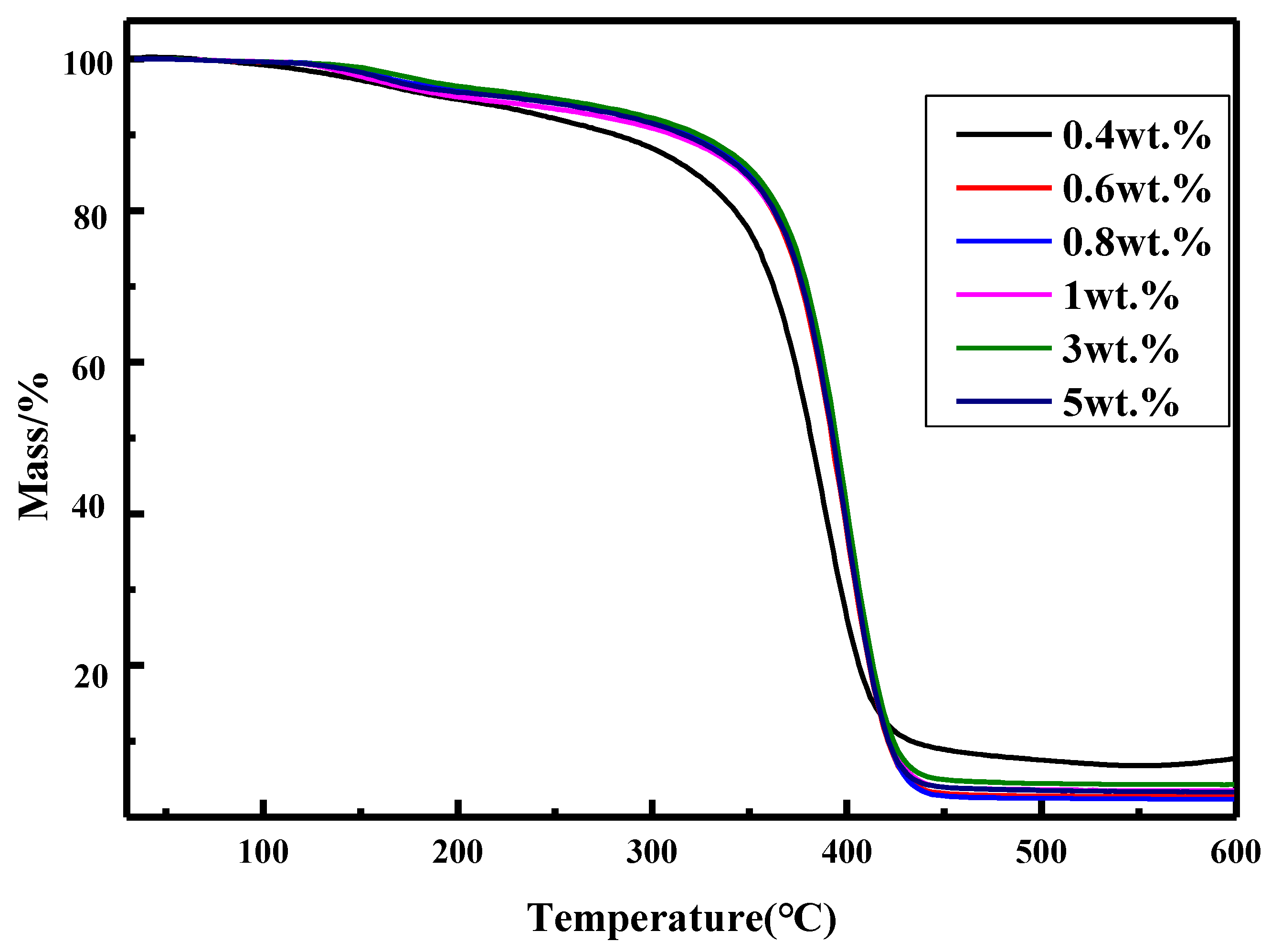
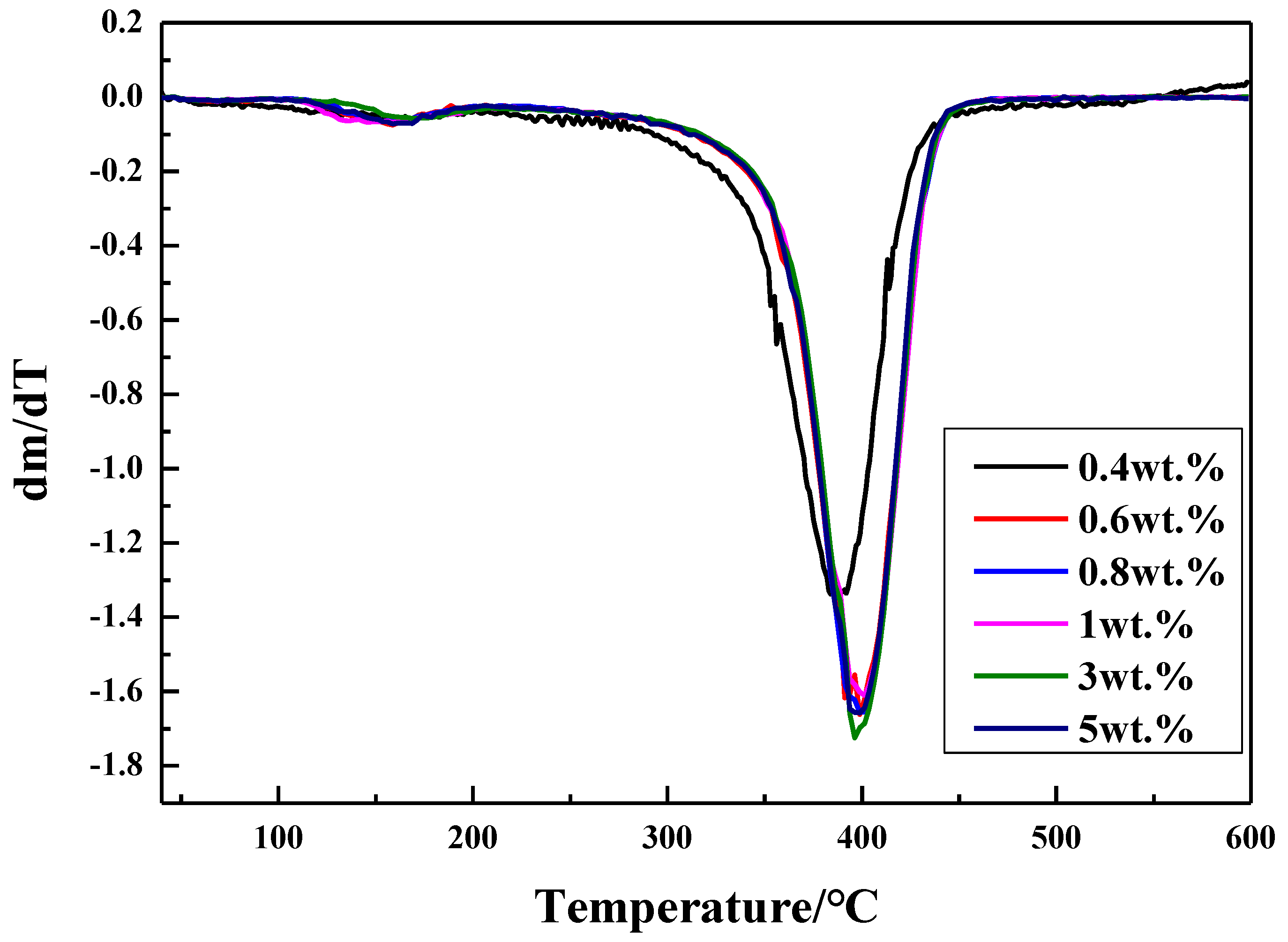
3.2.2. Thermal Stability
3.2.3. Adhesive Property
3.2.4. UV Resistance for the Coatings
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Jiao, X.J.; Zhang, T.X.; Cheng, F.; Fan, Y.X.; Liu, J.L.; Lai, G.Q.; Wu, Y.F.; Yang, X.F. UV-cured coatings prepared with sulfhydryl terminated branched polyurethane and allyl terminated hyperbranched polycarbosilane. Coatings 2020, 10, 350. [Google Scholar] [CrossRef]
- Liu, J.L.; Jiao, X.J.; Cheng, F.; Fan, Y.X.; Wu, Y.F.; Yang, X.F. Fabrication and performance of UV cured transparent silicone modified polyurethane–acrylate coatings with high hardness, good thermal stability and adhesion. Prog. Org. Coat. 2020, 144, 10567. [Google Scholar] [CrossRef]
- Wu, Y.F.; Liu, J.L.; Jiao, X.J.; Cheng, F.; Lai, G.Q.; Yang, X.F. UV–cured transparent flexible silicone materials with high tensile strength. ACS Omega 2020, 5, 6199–6206. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.Q.; Liu, W.Q.; Wang, H.L.; Sun, Y.; Wang, S.J. A novel mono-methacryloyloxy terminated fluorinated macromonomer used for the modification of UV curable acrylic copolymers. J. Appl. Polym. Sci. 2016, 133, 43116. [Google Scholar] [CrossRef]
- Yu, F.Y.; Gao, J.; Liu, C.P.; Chen, Y.K.; Zhong, G.; Hodges, C.; Chen, M.F.; Zhang, H.G. Preparation and UV aging of nano–SiO2/fluorinated polyacrylate polyurethane hydrophobic composite coating. Prog. Org. Coat. 2020, 141, 105556. [Google Scholar] [CrossRef]
- Rengasamy, S.; Mannari, V. Development of soy–based UV–curable acrylate oligomers and study of their film properties. Prog. Org. Coat. 2013, 76, 78–85. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, C.P.; Gao, J.; Chen, Y.K.; Yu, F.Y.; Chen, M.F.; Zhang, H.G. A novel hydrophobic coating film of water–borne fluoro-silicon polyacrylate polyurethane with properties governed by surface self–segregation. Prog. Org. Coat. 2019, 134, 134–144. [Google Scholar] [CrossRef]
- Darras, V.; Fichet, O.; Perrot, F.; Boileau, S.; Teyssie, D. Polysiloxane—poly(fluorinated acrylate) interpenetrating polymer networks: Synthesis and characterization. Polymer 2007, 48, 687–695. [Google Scholar] [CrossRef]
- Lowe, A.B.; Hoyle, C.E.; Bowman, C.N. Thiol–yne click chemistry: A powerful and versatile methodology for materials synthesis. J. Mater. Chem. 2010, 20, 4745–4750. [Google Scholar] [CrossRef]
- Rafael, A.; Pilar, J.M.; Jaime, G.R.; María–José, B.; Ángel, M. Thiol–ene click chemistry towards easy microarraying of half–antibodies. Chem. Commun. 2018, 54, 6144–6147. [Google Scholar] [CrossRef] [PubMed]
- Nakabayashi, K.; Sobu, S.; Kosuge, Y.; Mori, H. Synthesis and nanoimprinting of high refractive index and highly transparent polythioethers based on thiol–ene click chemistry. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 2175–2182. [Google Scholar] [CrossRef]
- Miao, J.T.; Yuan, L.; Guan, Q.B.; Liang, G.Z.; Gu, A.J. Water–phase synthesis of a biobased allyl compound for building UV–curable flexible thiol−ene polymer networks with high mechanical strength and transparency. ACS Sustain. Chem. Eng. 2018, 6, 7902–7909. [Google Scholar] [CrossRef]
- Childress, K.K.; Alim, M.D.; Hernandez, J.J.; Stansbury, J.W.; Bowman, C.N. Additive manufacture of lightly crosslinked semicrystalline thiol–enes for enhanced mechanical performance. Polym. Chem. 2020, 11, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.F.; Shao, Q.; Yang, L.L.; Zhu, X.B.; Hua, X.L.; Zheng, Q.L.; Song, G.X.; Lai, G.Q. Preparation and performance of high refractive index silicone resin–type materials for the packaging of light–emitting diodes. J. Appl. Polym. Sci. 2013, 127, 1717–1724. [Google Scholar] [CrossRef]
- Oh, Y.S.; Yoon, I.S.; Lee, C.; Kim, S.H.; Ju, B.K.; Hong, J.M. Selective photonic sintering of Ag flakes embedded in silicone elastomers to fabricate stretchable conductors. J. Mater. Chem. C 2017, 5, 11733–11740. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, J.W.; Ko, H.H. Transparent and flexible surface–enhanced raman scattering (SERS) sensors based on gold nanostar arrays embedded in silicone rubber film. ACS Appl. Mater. Int. 2017, 9, 44088–44095. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.R.; Yang, W.; Zhang, Q.H.; Hui, H.H.; Wei, Y.W.; Wang, J.; Xu, Q.; Lei, X.Y.; Chen, J.J.; Zhu, J.L. Fabrication of UV–curable silicone coating with high transmittance and laser–induced damage threshold for high–power laser system. J. Sol Gel Sci. Technol. 2018, 88, 249–254. [Google Scholar] [CrossRef]
- Afshar, A.; Mihut, D.; Baqersad, J.; Hill, S. Study of metallic thin films on epoxy matrix as protective barrier to ultraviolet radiation. Surf. Coat. Tech. 2019, 367, 41–48. [Google Scholar] [CrossRef]







| Entry | Curing Time/s | Pencil Hardness | Water contact Angle/° | Water Absorption/wt.% |
|---|---|---|---|---|
| 1 | 10 | H | 81.9 | 0.12 |
| 2 | 20 | 2 H | 76.8 | 0.11 |
| 3 | 30 | 3 H | 81.6 | 0.08 |
| 4 | 40 | 4 H | 83.5 | 0.10 |
| 5 | 50 | 5 H | 80.8 | 0.13 |
| 6 | 60 | 6 H | 83.4 | 0.02 |
| 7 | 70 | 6 H | 85.6 | 0.09 |
| Entry | The Amount of Styrene/% | Pencil Hardness | Water Contact Angle/° | Water Absorption/wt.% |
|---|---|---|---|---|
| 1 | 0 | 4B | 82.4 | 0.07 |
| 2 | 6.0 | 2B | 84.9 | 0.09 |
| 3 | 12.0 | H | 86.0 | 0.13 |
| 4 | 18.0 | 3H | 86.6 | 0.12 |
| 5 | 24.0 | 4H | 89.7 | 0.15 |
| Entry | Thiol Content of Silicone Resins/mmol g−1 | Pencil Hardness | Water Contact Angle/° | Water Absorption/wt.% |
|---|---|---|---|---|
| 1 | 2 | H | 74.7 | 0.13 |
| 2 | 3 | 2 H | 74.6 | 0.03 |
| 3 | 4 | 2 H | 87.9 | 0.01 |
| 4 | 5 | 4 H | 83.5 | 0.10 |
| 5 | 6 | 2 H | 92.7 | 0.11 |
| Entry | Thiol Amount of Thiol Silicone Resins/% | Pencil Hardness | Water Contact Angle/° | Water Absorption/wt.% |
|---|---|---|---|---|
| 1 | 0 a | 2B | 71.7 | 0.17 |
| 2 | 0.4 | F | 69.1 | 0.16 |
| 3 | 0.6 | H | 68.7 | 0.10 |
| 4 | 0.8 | H | 71.0 | 0.11 |
| 5 | 1 | 4H | 83.5 | 0.10 |
| 6 | 3 | 3H | 79.3 | 0.12 |
| 7 | 5 | 3H | 82.5 | 0.12 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, F.; Fan, Y.; Zhang, L.; Jiao, X.; Lai, G.; Hua, X.; Yang, X. An Easy Fabrication Method to Prepare Inexpensive UV–Cured Transparent Silicone Modified Polyacrylate Coatings with Good Adhesion and UV Resistance. Coatings 2020, 10, 675. https://doi.org/10.3390/coatings10070675
Cheng F, Fan Y, Zhang L, Jiao X, Lai G, Hua X, Yang X. An Easy Fabrication Method to Prepare Inexpensive UV–Cured Transparent Silicone Modified Polyacrylate Coatings with Good Adhesion and UV Resistance. Coatings. 2020; 10(7):675. https://doi.org/10.3390/coatings10070675
Chicago/Turabian StyleCheng, Fei, Yunxin Fan, Lu Zhang, Xiaojiao Jiao, Guoqiao Lai, Xilin Hua, and Xiongfa Yang. 2020. "An Easy Fabrication Method to Prepare Inexpensive UV–Cured Transparent Silicone Modified Polyacrylate Coatings with Good Adhesion and UV Resistance" Coatings 10, no. 7: 675. https://doi.org/10.3390/coatings10070675
APA StyleCheng, F., Fan, Y., Zhang, L., Jiao, X., Lai, G., Hua, X., & Yang, X. (2020). An Easy Fabrication Method to Prepare Inexpensive UV–Cured Transparent Silicone Modified Polyacrylate Coatings with Good Adhesion and UV Resistance. Coatings, 10(7), 675. https://doi.org/10.3390/coatings10070675