Chromium Diffusion Coating on an ODS Ferritic-Martensitic Steel and Its Oxidation Behavior in Air and Steam Environments
Abstract
1. Introduction
2. Materials and Methods
2.1. Material and Surface Modification Procedure
2.2. High Temperature Oxidation Test in Air and Steam Environments
3. Results and Discussion
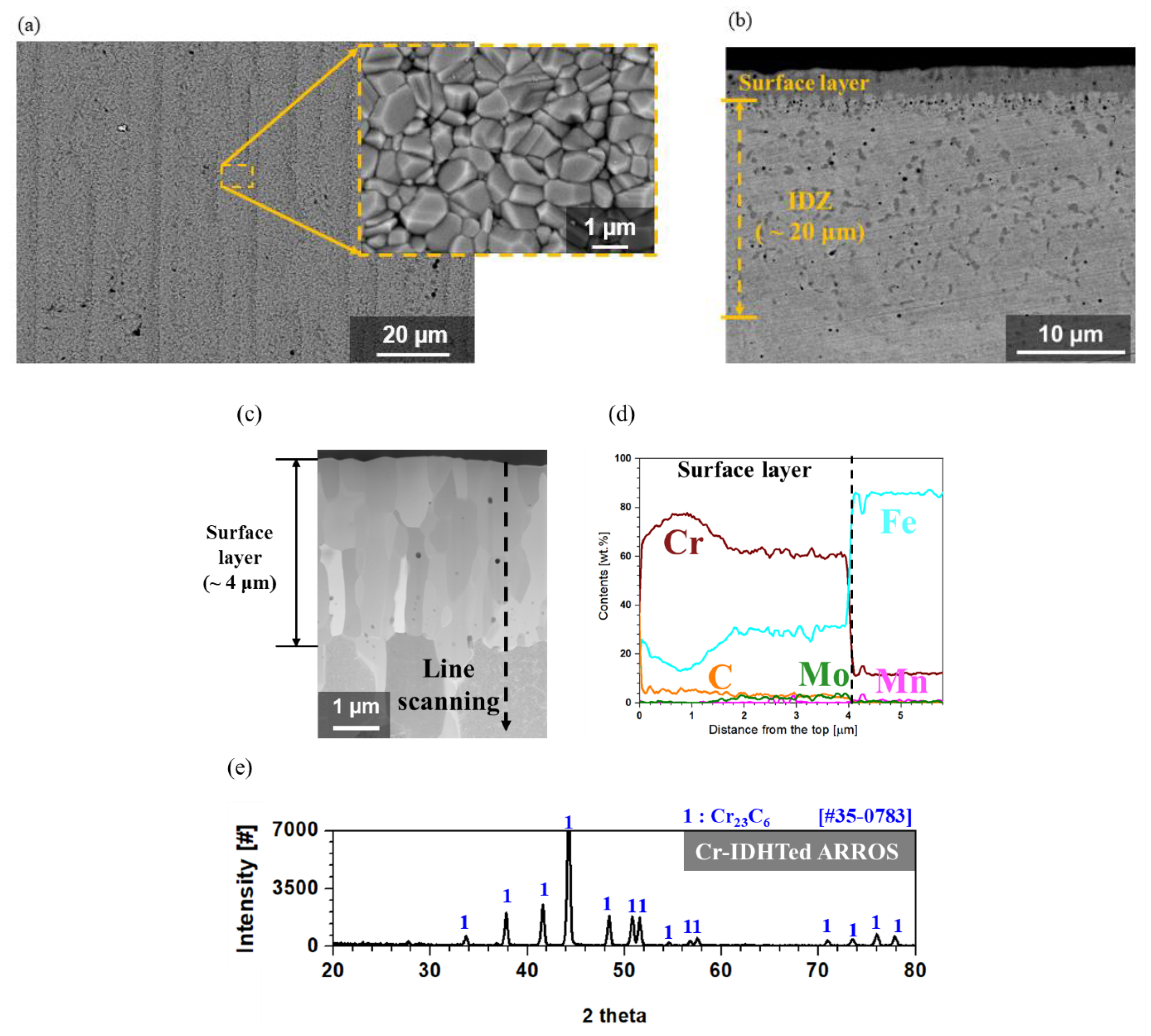
3.1. Characterization of the Surface-Modified Layer
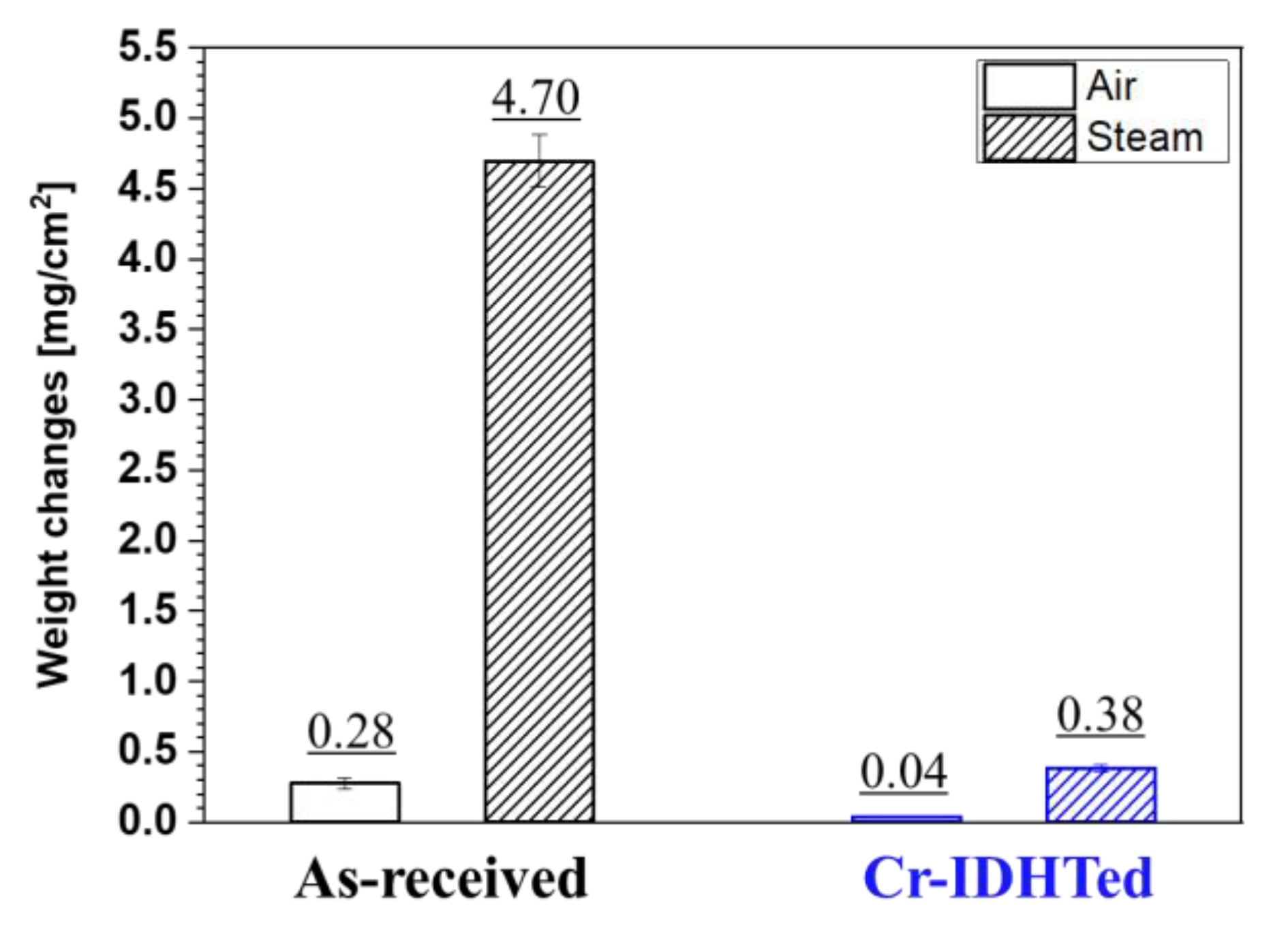
3.2. Weight Changes after Air and Steam Oxidation Test
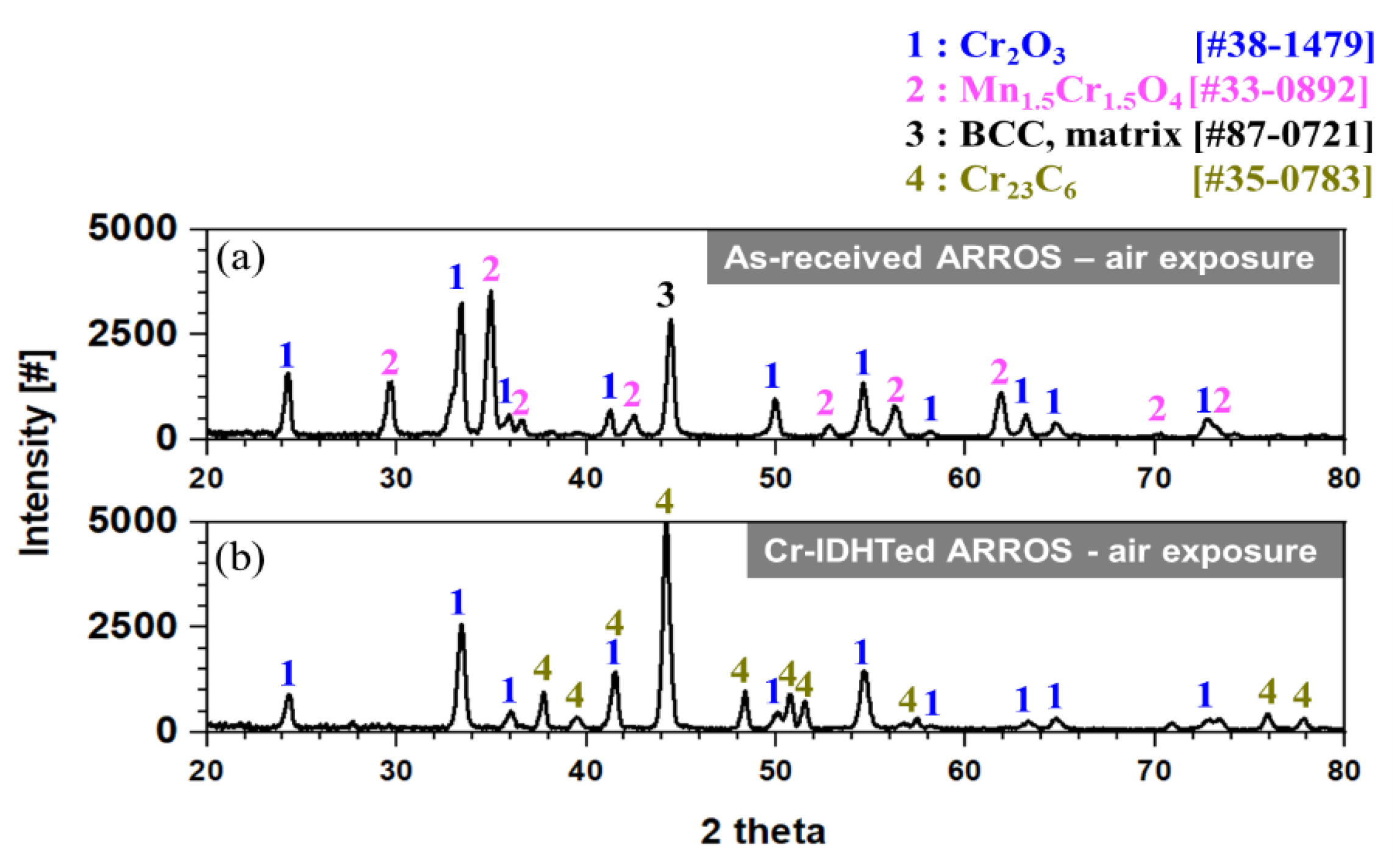
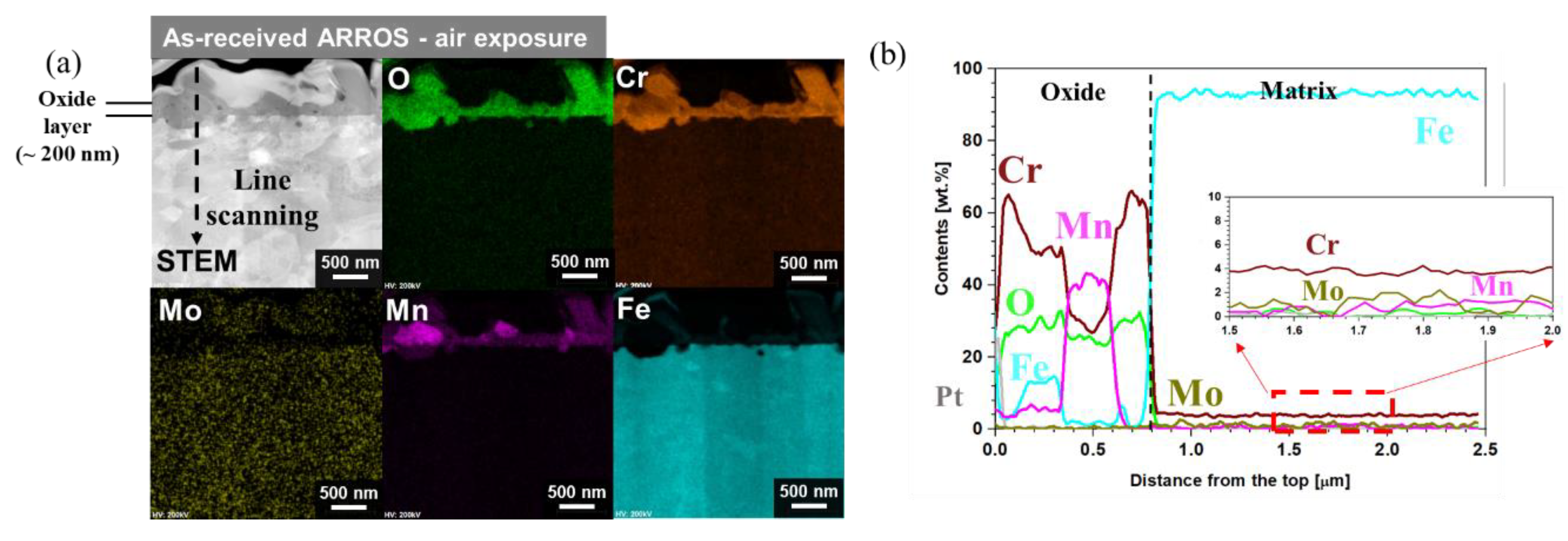
3.3. Oxidation Behavior in Air Environment
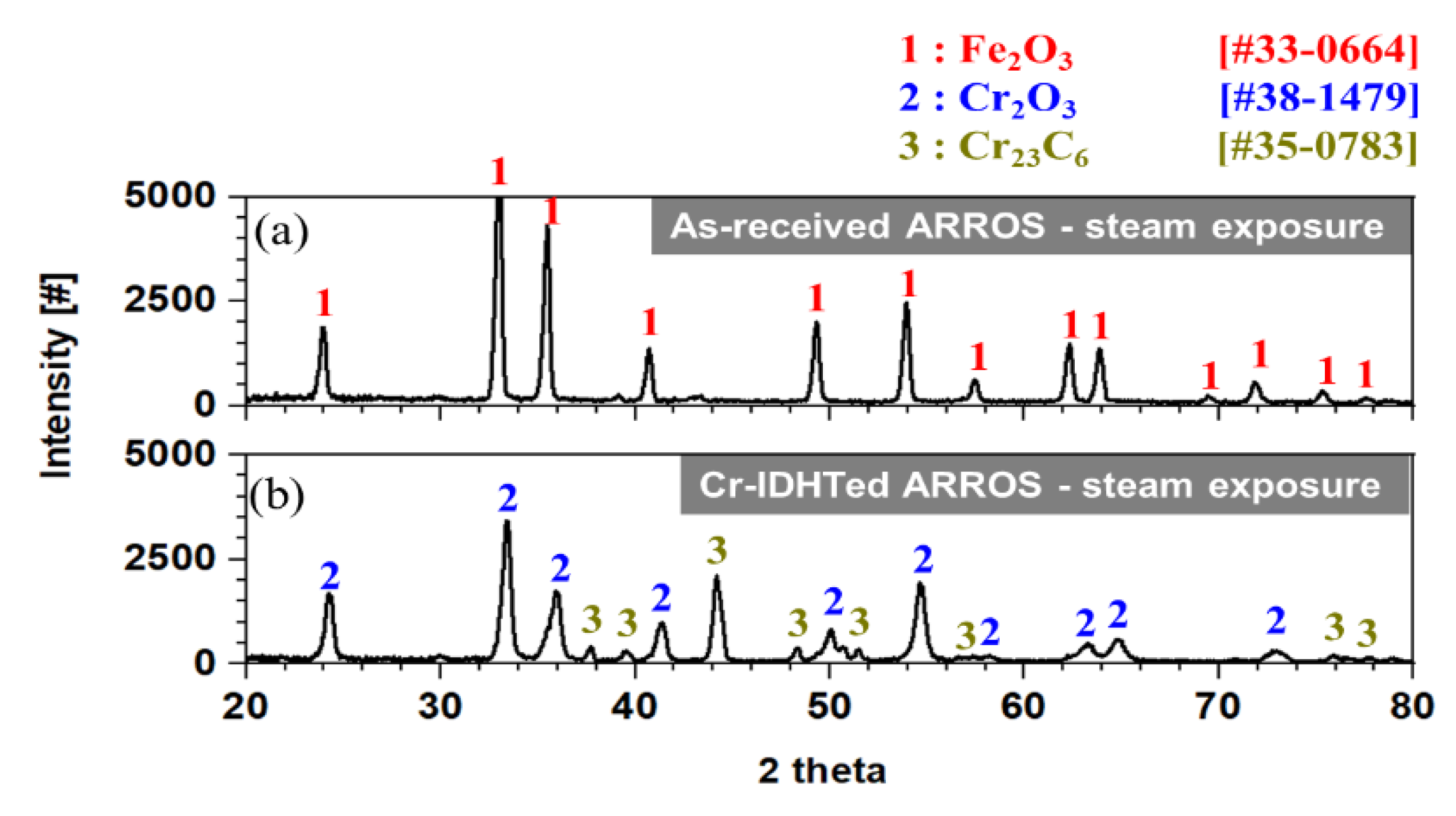
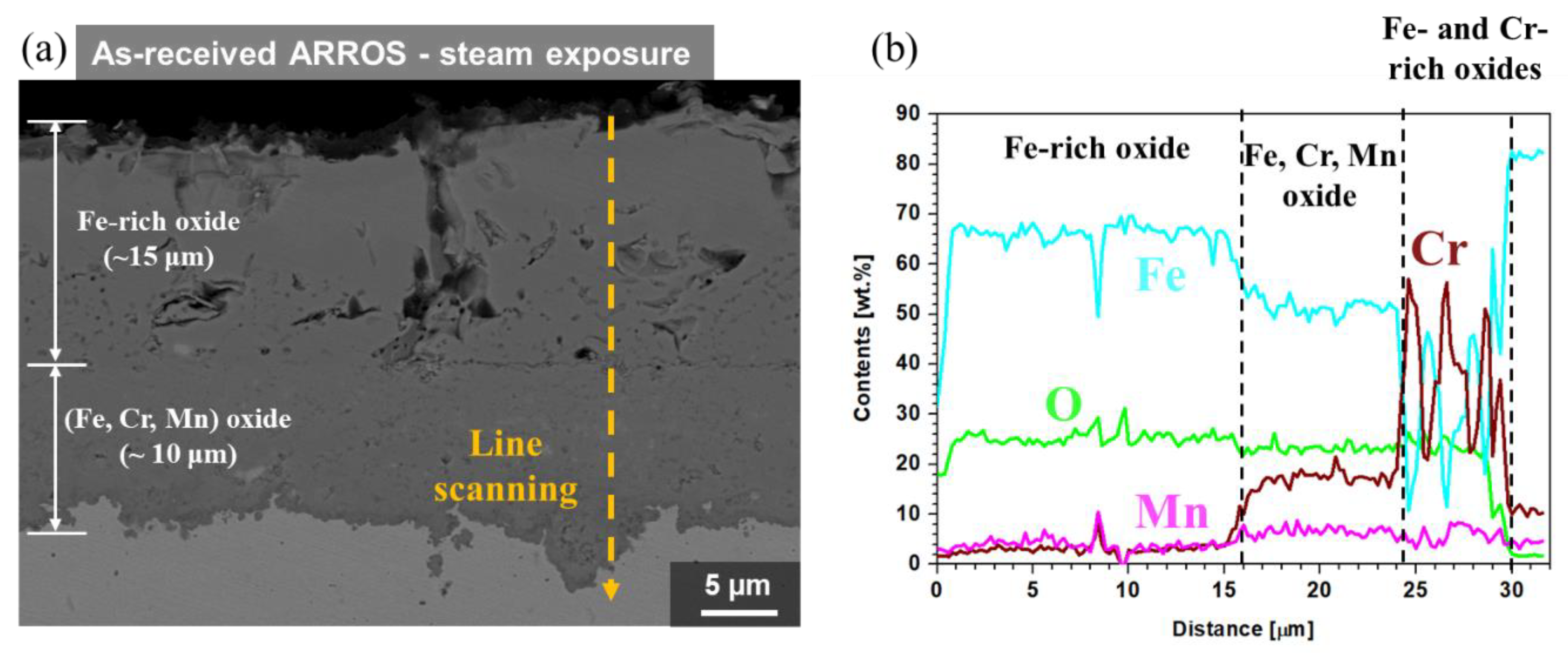
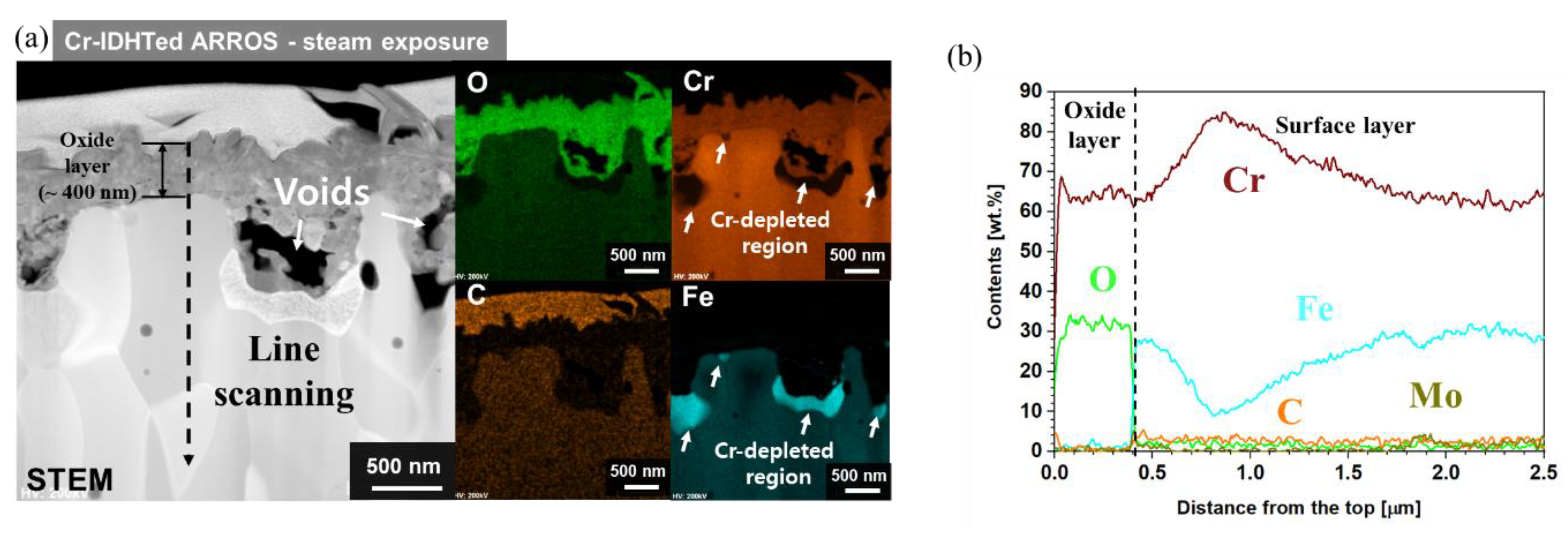
3.4. Oxidation Behavior in Steam Environment
3.5. Effect of Environments on the Oxidation Behaivor of the Cr-IDHTed Layer
4. Conclusions
- The surface-modified specimens showed enhanced oxidation resistance in both air and steam environments showing much smaller weight gains compared to the as-received specimens;
- In an air environment, while the as-received specimens formed Cr2O3 and (Mn, Cr)3O4 nodules and a thin chromia layer, the Cr-IDHTed specimen showed only thin chromia as the surface layer prevented outward diffusion of Mn from the matrix;
- In a steam environment, while thick oxide layers consisted of outer Fe-rich oxide layer and inner (Fe, Cr, Mn) oxide layer was formed on the as-received specimen, a thin and continuous chromia layer was formed on the Cr-IDHTed one because of enough Cr reservoir in the surface layer;
- In the surface layer of the Cr-IDHTed specimen, larger voids and finer grains were observed after oxidation test in steam. Rapid inward diffusion of oxygen to the oxide/surface layer interface in steam environment was responsible for higher weight gain and larger void formation.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gianfrancesco, A.D. The fossil fuel power plants technology. In Materials for Ultra-Supercritical and Advanced Ultra-Supercritical Power Plants, 1st ed.; Gianfrancesco, A.D., Ed.; Woodhead Publishing: Duxford, UK, 2017; pp. 1–49. [Google Scholar]
- Yoo, J.; Chang, J.; Lim, J.-Y.; Cheon, J.-S.; Lee, T.-H.; Kim, S.K.; Lee, K.L.; Joo, H.-K. Overall system description and safety characteristics of Prototype Gen IV Sodium Cooled Fast Reactor in Korea. Nucl. Eng. Technol. 2016, 48, 1059–1070. [Google Scholar] [CrossRef]
- Kern, T.-U.; Staubi, M.; Scarlin, B. The european efforts in material development for 650 °C USC power plants-COST522. ISIJ Int. 2002, 42, 1515–1519. [Google Scholar] [CrossRef]
- Masuyama, F. History of power plants and progress in heat resistant steels. ISIJ Int. 2001, 41, 612–625. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, C.; Yu, L.; Liu, Y.; Li, H. Phase transformation behavior and microstructural control of high-Cr martensitic/ferritic heat-resistant steels for power and nuclear plants: A review. J. Mater. Sci. Technol. 2015, 31, 235–242. [Google Scholar] [CrossRef]
- Kleh, R.L.; Hashimoto, N.; Maziasz, P.J. Development of new nano-particle-strengthened martensitic steels. Scripta Mater. 2005, 53, 275–280. [Google Scholar] [CrossRef]
- Ukai, S.; Ohtsuka, S.; Kaito, T.; Carlan, Y.; Ribis, J.; Malaplate, J. Oxide dispersion-strengthen/ferrite-martensite steels as core materials for Generation IV nuclear reactors. In Structural Materials for Generation IV Nuclear Reactors, 1st ed.; Yvon, P., Ed.; Woodhead Publishing: Duxford, UK, 2017; pp. 357–414. [Google Scholar] [CrossRef]
- Kim, T.K.; Noh, S.; Kang, S.H.; Park, J.J.; Jin, H.J.; Lee, M.K.; Jang, J.; Rhee, C.K. Current status and future prospective of advanced radiation resistant oxide dispersion strengthened steel (ARROSS) development for nuclear reactor system applications. Nucl. Eng. Technol. 2016, 48, 572–594. [Google Scholar] [CrossRef]
- Quadakkers, W.J.; Holzbrecher, H.; Briefs, K.G.; Beske, H. Differences in growth mechanisms of oxide scales formed on ODS and conventional wrought alloys. Oxid. Met. 1989, 32, 67–88. [Google Scholar] [CrossRef]
- Chen, Y.; Srindharan, K.; Ukai, S.; Allen, T.R. Oxidation of 9Cr oxide dispersion strengthened steel exposed in supercritical water. J. Nucl. Mater. 2007, 371, 118–128. [Google Scholar] [CrossRef]
- Ehlers, J.; Young, D.J.; Smaardijk, E.J.; Tyagi, A.K.; Penkalla, H.J.; Singheiser, L.; Quadakkers, W.J. Enhanced oxidation of the 9%Cr steel P91 in water vapour containing environments. Corros. Sci. 2006, 48, 3428–3454. [Google Scholar] [CrossRef]
- Schmidt, D.; Galetz, M.C.; Schutze, M. Ferritic-martensitic steels: Improvement of the oxidation behavior in steam environments via diffusion coatings. Surf. Coat. Technol. 2013, 237, 23–29. [Google Scholar] [CrossRef]
- Wu, S.; Guo, B.; Li, T.; Gui, D. Oxidation of chromium carbide coated Q235 steel in wet and dry air at 750 °C. Constr. Build. Mater. 2015, 81, 11–14. [Google Scholar] [CrossRef]
- Kim, C.; Kim, S.H.; Cha, J.-H.; Jang, C.; Kim, T.K. Cr diffusion coating to improve the corrosion resistance of an ODS steel in super-critical carbon dioxide environment. Surf. Coat. Technol. 2019, 374, 666–673. [Google Scholar] [CrossRef]
- Kim, D.; Kim, D.; Lee, H.J.; Jang, C.; Yoon, D.J. Corrosion characteristics of Ni-base superalloys in high temperature steam with and without hydrogen. J. Nucl. Mater. 2013, 441, 612–622. [Google Scholar] [CrossRef]
- Ren, P.; Zhu, S.; Wang, F. Spontaneous reaction formation of Cr23C6 diffusion barrier layer between nanocrystalline MCrAlY coating and Ni-base superalloy at high temperature. Corros. Sci. 2015, 99, 219–226. [Google Scholar] [CrossRef]
- Kučera, J.; Stránský, K. Diffusion in iron, iron solid solutions and steels. Mater. Sci. Eng. 1982, 52, 1–38. [Google Scholar] [CrossRef]
- Sundararajan, T.; Kuroda, S.; Kawakita, J.; Seal, S. High temperature corrosion of nanoceria coated 9Cr-1Mo ferritic steel in air and steam. Surf. Coat. Technol. 2006, 201, 2124–2130. [Google Scholar] [CrossRef]
- Jian, L.; Jian, P.; Bing, H.; Xie, G. Oxidation kinetics of Haynes 230 alloy in air at temperature between 650 and 850 °C. J. Power Sources 2006, 159, 641–645. [Google Scholar] [CrossRef]
- Lobnig, R.E.; Schmidt, H.P.; Hennesen, K.; Grabke, H.J. Diffusion of cations in chromia layer grown on iron-base alloys. Oxid. Met. 1992, 37, 81–93. [Google Scholar] [CrossRef]
- Quadakkers, W.J.; Ennis, P.J.; Zurek, J.; Michalik, M. Steam oxidation of ferritic steels-laboratory test kinetic data. Mater. High. Temp. 2005, 22, 47–60. [Google Scholar] [CrossRef]
- Young, D.J. Effects of water vapour on the oxidation of chromia formers. Mater. Sci. Forum 2008, 595–598, 1189–1197. [Google Scholar] [CrossRef]
- Zurek, J.; Young, D.J.; Essuman, E.; Hänsel, M.; Penkalla, H.J.; Niewolak, L.; Quadakker, W.J. Growth and adherence of chromia based surface scales on Ni-base alloys in high- and low-pO2 gases. Mater. Sci. Eng. A 2008, 477, 259–270. [Google Scholar] [CrossRef]
- Othman, N.K.; Othman, N.; Zhang, J.; Young, D.J. Effects of water vapour on isothermal oxidation of chromia-forming alloys in Ar/O2 and Ar/H2 atmospheres. Corros. Sci. 2009, 51, 3039–3049. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Zhang, J.; Young, D.J. Effect of silicon and water vapour on corrosion of Fe-20Cr and Fe-20Cr-20Ni alloys in CO2 at 650 °C. Oxid. Met. 2017, 87, 541–573. [Google Scholar] [CrossRef]



| Fe | Cr | Mo | Mn | V | Ti | C | Y2O3 |
|---|---|---|---|---|---|---|---|
| Bal. | 9 | 1 | 0.5 | 0.1 | 0.2 | 0.15 | 0.25 |
| Target | Number of Guns | Mode (Power) | Time | Working Pressure (Ar) | Base Pressure | Coating Thickness |
|---|---|---|---|---|---|---|
| Cr (99.95%) | 2 guns | DC (250 W) | 1 h | 0.003 torr | ≤3.0 × 10−6 torr | ~4 μm |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, C.; Kim, S.H.; Cha, J.-H.; Jang, C.; Kim, T.K. Chromium Diffusion Coating on an ODS Ferritic-Martensitic Steel and Its Oxidation Behavior in Air and Steam Environments. Coatings 2020, 10, 492. https://doi.org/10.3390/coatings10050492
Kim C, Kim SH, Cha J-H, Jang C, Kim TK. Chromium Diffusion Coating on an ODS Ferritic-Martensitic Steel and Its Oxidation Behavior in Air and Steam Environments. Coatings. 2020; 10(5):492. https://doi.org/10.3390/coatings10050492
Chicago/Turabian StyleKim, Chaewon, Sung Hwan Kim, Ji-Hwan Cha, Changheui Jang, and Tae Kyu Kim. 2020. "Chromium Diffusion Coating on an ODS Ferritic-Martensitic Steel and Its Oxidation Behavior in Air and Steam Environments" Coatings 10, no. 5: 492. https://doi.org/10.3390/coatings10050492
APA StyleKim, C., Kim, S. H., Cha, J.-H., Jang, C., & Kim, T. K. (2020). Chromium Diffusion Coating on an ODS Ferritic-Martensitic Steel and Its Oxidation Behavior in Air and Steam Environments. Coatings, 10(5), 492. https://doi.org/10.3390/coatings10050492





