2.1. Materials
Microcrystalline cellulose (commercial grade Avicel™ PH 101, FMC, Cork, Ireland), lactose monohydrate 200 mesh (Meggle GmbH, Wasserburg am Inn, Germany), polyvinylpyrrolidone (commercial grade Kollidon® 30, BASF AG GmbH, Ludwigshafen, Germany) were used for the laboratory-scale manufacture of the inert pellets by extrusion-spheronization. Meloxicam (Cipla Ltd., Mumbai, India) was kindly provided as a gift sample by Labormed Pharma S.R.L., Bucharest, Romania.
A dispersion containing meloxicam, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (≥99.0%, Sigma-Aldrich Inc., Saint Louis, MO, USA), talc (Imerys Talc Luzenac, Luzenac, France), and distilled water was used for the drug-layering of the pellets. The film-coating of the meloxicam pellets was performed using Acryl-EZE® 93O (Colorcon, West Point, PA, USA), Eudragit® RS 30-D (Evonik Industries AG, Essen, Germany), Methocel™ E5 Premium LV (Dow Chemical Company, Midland, MI, USA). PEG 400 (BASF AG GmbH, Ludwigshafen, Germany) was associated as a plasticizer in the Eudragit® coating dispersions. The film-coated pellets were filled into hard gelatin capsules, size 00 (Capsugel, Bornem, Belgium).
Meloxicam sodium salt hydrate (≥98%, Sigma-Aldrich, St. Louis, MO, USA) was used for the construction of the calibration curve. Hydrochloric acid (37%, Honeywell Riedel-de Haën AG, Seelze, Germany) and trisodium phosphate dodecahydrate (≥98.0%, Honeywell Riedel-de Haën AG, Seelze, Germany) were used for the preparation of the dissolution media. CAPWHT-XL helix wire sinkers (QLA, Telford, PA, USA) were used in order to prevent the capsules from floating.
2.2. Methods
2.2.1. Obtaining and Characterization of the of Inert Pellets by Extrusion-Spheronization
The composition of the inert pellet cores included microcrystalline cellulose (40.0%, w/w of the moist mass) as spheronization aid and filler and lactose monohydrate (25.0%, w/w of the moist mass) as filler. The powder ingredients were dry mixed in a cubic mixer (Erweka KU1 drive with a cube mixer made of acrylic glass, 3.5 l volume, Erweka GmbH, Frankfurt am Main, Germany) for 20 min at 20 rpm. A binding solution (2.5%, w/w aqueous solution of povidone) was gradually added by spraying (in five sequences) to the homogenous powder blend, over a 20 min timespan. Each addition of a fraction of the binder solution was followed by a new mixing sequence (15 min, 20 rpm). The total amount of binder solution added was equivalent to 35.0%, w/w of the resulting moist mass.
This mass was further processed by extrusion and spheronization, using benchtop equipments: an extruder (Model 25, Caleva Process Solutions Ltd., Dorset, UK) and a spheronizer (Model 120, Caleva Process Solutions Ltd., Dorset, UK). The process parameters (spheronization speed and time) are presented in
Table 1.
Following manufacture, the resulting pellets were collected and dried in a dry air tray oven (Memmert GmbH + Co. KG, Schwabach, Germany), at 40 °C for 24 h, in order to remove the residual water.
Several characteristics of resulting pellets, deemed relevant for subsequent processing, were evaluated: particle size distribution, shape descriptors, and Hausner ratio.
Particle size distribution was determined by sieving a sample of pellets through a set of standard sieves using a CISA Sieve Shaker (CISA Cedaceria, Barcelona, Spain). The amplitude of the electromagnetic shaker was set at 1.5 mm and a sample of 100 g of inert pellets was sieved for 10 min. The fractions of pellets remaining on the sieves with 1.18 mm, 0.8 mm, 0.6 mm, 0.315 mm, and 0.250 mm eye openings were determined by weighting and expressed as percentages of the total weight of the sample. During the formulation and process development of the inert pellets, it was intended to obtain a large fraction of particles with sizes between 0.8 and 1.8 mm. Such a size distribution was considered to be optimal for ulterior coating and encapsulation of the pellets.
The pellets were also evaluated for several shape descriptors, such as circularity, aspect ratio, roundness, and sphericity. These parameters were considered relevant as a high degree of sphericity is desired. This characteristic offers certain advantages to the pellets, such as a minimal contact, improved flowing behavior and would promote the deposition of uniform polymeric films on the surface of the pellets.
According to scientific literature, circularity alone is incapable of differentiating between particles with very different aspect ratios, but may, however, be of value when it is used in conjunction with aspect ratio or with other factors sensitive to aspect ratio [
35]. Aspect ratio describes the ratio between the width and length of the pellet and is related to the elongation of the particle, whereas sphericity is described by the value of roundness [
36].
The shape of the pellets was assessed by the following procedure: Individual photographs were taken for 200 pellets laid out on a dark background using a Visioscan
® VC 98 (Courage + Khazakha GmbH, Köln, Germany) camera, with a magnification degree of 30×. Each image was analyzed using the ImageJ software (U.S. National Institute of Health, Bethesda, Maryland, USA). The software allows the calculation of certain shape descriptors of the photographed objects based on the following equations:
Also, given that the software reports the area and perimeter of the photographed objects, it was possible to calculate the sphericity of the pellets (a measure of their roundness) according to the equation:
where
PEQPC is the perimeter of a circle that has an area equivalent to that of the pellet image and
Preal represents the perimeter of the pellet image [
37].
According to the ImageJ software manual and to scientific literature [
35,
36,
37,
38], for either circularity or aspect ratio, values closer to 1 indicate that the photographed objects have a circular shape. Thus, it can be assumed that the closer the values of these two parameters are closer to 1, the more spherical the pellets are.
Hausner ratio is an expression of the friction forces between particles during particle flow, thus being considered an important parameter for the uniform filling of the pellets into hard gelatin capsules. In order to determine the values of this parameter, tapped and bulk densities of the pellets after 300 taps were determined using a Erweka SVM 101 tapped density tester (Erweka GmbH, Heusenstamm, Germany), fitted with a 25 mL cylinder. Based on these results, the Hausner ratio was determined according to the following equation:
where
is the tapped density and
is the bulk density of the solid material. A Hausner ratio value lower than 1.2 is an indicator of good flowing characteristics [
39].
The fraction of inert pellet cores with sizes between 800 and 1180 μm, separated by sieving, was used for further processing (drug loading).
2.2.2. Loading of the Inert Pellets with Meloxicam by Drug Layering
The inert pellets previously obtained were loaded with meloxicam by drug layering. This technique involved spraying of a meloxicam dispersion on the inert pellet cores in a fluidized bed coating equipment.
First, a meloxicam dispersion was prepared. The composition of this dispersion is shown in
Table 2.
Samples of 30 g inert pellets were preheated (at 40 °C) in a fluidized bed coating equipment (Caleva Mini Coater/Drier 2, Caleva Process Solutions Ltd., Dorset, UK).
The meloxicam dispersion, maintained under constant stirring at 800 rpm (Variomag Mono Direct magnetic stirrer, Thermo Fischer Scientific Inc., Waltham, MA, USA) during the coating process, was sprayed on the preheated pellet cores in a fluidized bed coating equipment (Caleva Mini Coater/Drier 2, Caleva Process Solutions Ltd., Dorset, UK). The process parameters used during fluid bed coating of the pellets are shown in
Table 3.
For the experimental batches (30 g of inert pellet cores), the amount of meloxicam dispersion used was of 55 g, and the duration of the drug loading stage was of 2 h. The duration of the drug-loading stage was established based of meloxicam assay results obtained for small samples of pellets that were periodically taken out of the coating chamber.
The assay of drug content was performed on the resulting drug-loaded pellets, by UV spectrophotometry, using a Jasco V-530 UV-Vis spectrophotometer (Jasco International Co., Ltd., Tokyo, Japan). Approximately 3 g of pellets were triturated and 80 mg of pellet powder were accurately weighted into a 25 mL volumetric flask, and methanol was added to make up the volume. The samples were dispersed by sonication at room temperature (sonication bath Elma S30H, Elma Schmidbauer GmbH, Singen, Germany) for 10 min. Subsequently, samples of 5 mL were filtered through 0.45 μm regenerated cellulose non-sterile filters (Phenomenex, Torrance, CA, USA). Each sample was diluted with methanol (1:9 v/v) and its absorbance was recorded at 361.4 nm.
A calibration curve generated for dilutions of meloxicam reference standard in methanol, in the concentration range 0.05–400 μg/mL was used for drug quantification.
2.2.3. Film Coating of Meloxicam Pellets
The meloxicam pellets were subsequently subjected to a film-coating process.
Three coating materials were selected, namely Acryl-EZE® 93O white (Colorcon, West Point, PA, USA), Eudragit® RS 30-D (Evonik Industries AG, Essen, Germany), and an experimental composite film made up from Eudragit® RS 30-D and Methocel™ E5 Premium LV (Dow Chemical Company, Midland, MI, USA).
Acryl-EZE
® 93O white (further referred to as ”Acryl-EZE
®”) is a fully formulated, ready-to-use product, used for enteric coating, in which the film-forming agent is represented by an anionic methacrylic acid—ethyl acrylate (1:1) copolymer, type C [
40]. The free carboxylic groups of the copolymer form salts in an alkaline environment, thus the polymer is soluble in solutions with a pH value greater than 5.5.
Eudragit
® RS 30-D (Eudragit
®) is a liquid dispersion containing a copolymer of ethyl acrylate, methyl metacrylate, and small amounts of methacrylic acid esters with quaternary ammonium groups, which are present as salts, thus making the polymer permeable to aqueous solutions. Film-coating with a dispersion of this product leads to insoluble films, but which are slightly permeable to water, the swelling of the film being independent of the environmental pH values. Drug release from oral preparations film-coated with this product is usually time-dependent. The amount of drug released can be controlled by the film thickness [
21,
41], this parameter influencing the duration of the drug diffusion stage. Another means of ensuring a prolonged release is to incorporate pore-forming agents in the polymeric film formula.
Methocel™ E5 Premium LV (Methocel™) is a commercial grade of hypromellose, a water-soluble film-forming agent made up of methyl and hydroxypropyl mixed ether of cellulose, with pH-independent solubility profile. In this study, taking into consideration the characteristics of the Eudragit
® films (insoluble and slightly permeable), Methocel™ was used as a pore-forming agent, with the aim of adjusting the permeability of the Eudragit
® film [
42].
The polymeric film coatings were applied using the same equipment that was used for the coating of the pellets with the meloxicam layer.
Three experimental formulations with different type of polymer coating were subsequently obtained.
The first formulation, (coded “MXA”), was obtained by spray coating of the meloxicam pellets with a 20% (w/w) dispersion of Acryl-EZE® in distilled water.
The pellets were preheated in the fluid bed equipment at 35 °C and the Acryl-EZE
® dispersion was sprayed onto the meloxicam pellets, using the settings of the technological parameters described in
Table 4.
The dispersion was sprayed onto the pellet bed until a weight gain of 8% of the pellets, taking into consideration that the product manufacturer (Colorcon, West Point, PA, USA) recommends a 7–12% weight gain of the substrate, in order to obtain enteric properties. After the spraying stage, the pellets were kept in the fluidized bed, with an air flow temperature of 40 °C, in order to allow the film to cure.
For the second formulation (coded “MXE”) meloxicam pellets were coated with a dispersion of Eudragit
® in water, prepared by the formula presented in
Table 5 and the technological parameters described in
Table 6.
It should be noted that the amount of Eudragit® product used in the formulation translates into a 10% solid content of the final coating dispersion.
The dispersion was sprayed until a 5% weight gain of the pellets, followed by curing of the film by keeping the pellets in fluidized bed, at 42 °C, for 20 min.
Finally, a third formulation (coded MX-EM) was coated with a film in which Eudragit
® (as a film-forming agent) and Methocel™ (as a pore-forming agent) were combined. The composition of this film was based on the formula of the Eudragit
® dispersion previously used, where 5% of Methocel™ was associated. No significant increase of the viscosity of the coating dispersion could be observed (usually, 2% Methocel™ aqueous solutions have a nominal viscosity of 4–6 mPa
s). The formula of the coating solution is depicted in
Table 7. The same process parameters as those presented in
Table 6 were used in this experiment.
The formula was sprayed onto the pellets until a weight increase of 5% was determined. The same film curing conditions were applied (the pellets were kept in the fluid bed air current, at 42 °C, for 20 min).
The three variants of film coatings would lead to an enteric release of the BCS class II drug (for the pellets coated with Acryl-EZE®) or to a controlled, extended release (for the Eudragit® coated pellets).
2.2.4. Evaluation of Shape and Surface Morphology of Coated and Uncoated Meloxicam Pellets by Scanning Electron Microscopy (SEM)
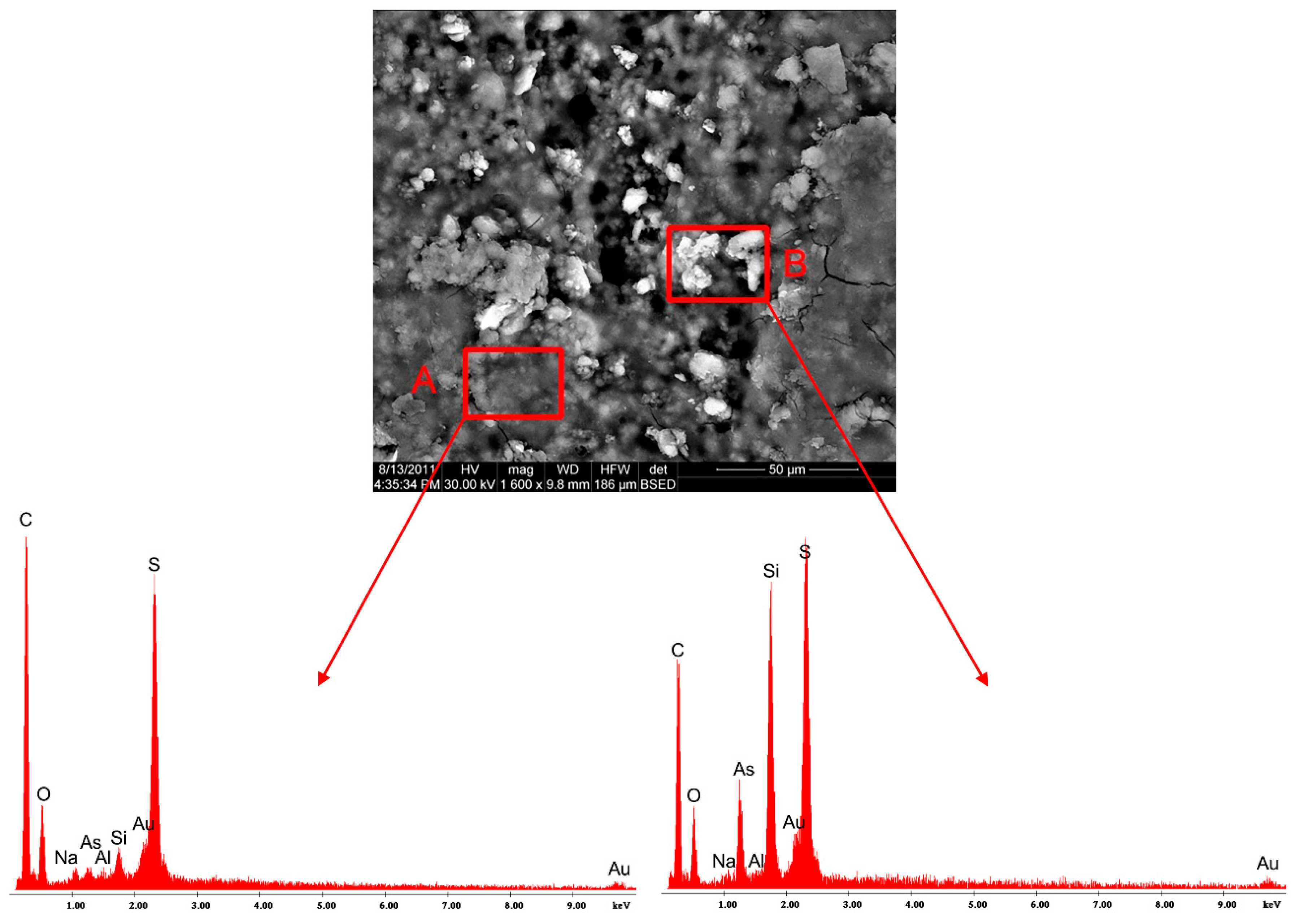
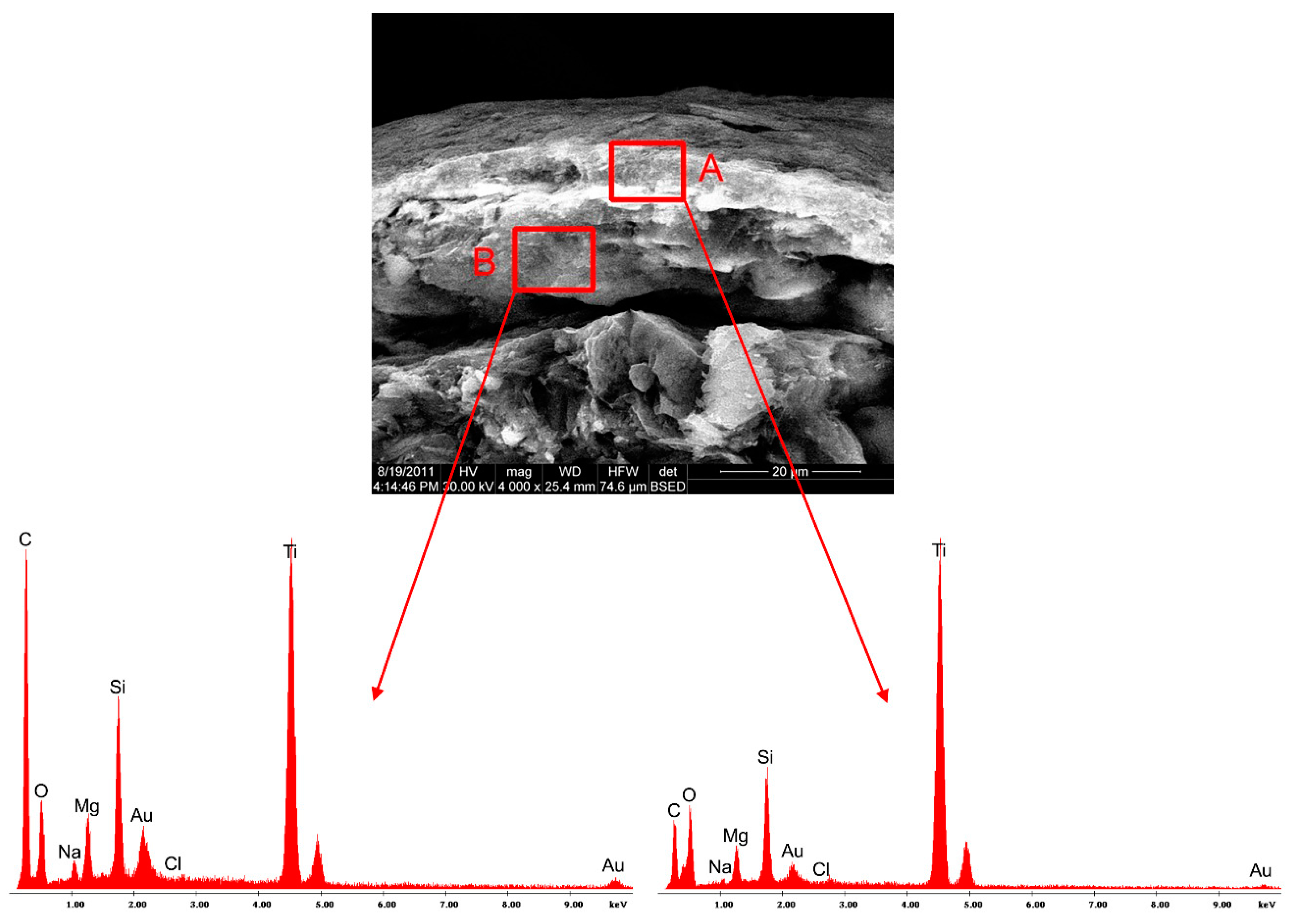
In order to evaluate the shape and surface morphology of the meloxicam pellets (both uncoated and film-coated) and to study and better understand the drug layering and film-coating processes, samples of inert pellets (“IP”), uncoated meloxicam pellets (“MXU”), and film-coated meloxicam pellets (film-coated with Acryl-EZE®-sample “MXA”, coated with Eudragit®-sample “MXE”, and coated with the Eudragit® and Methocel™ mixture sample “MX-EM”), respectively, were analyzed by using a Quanta Inspect F scanning electron microscope (FEI Co., Eindhoven, The Netherlands), operated in high vacuum mode (<6 Pa).
Cross-section images of the pellet samples were also taken. For this, pellet samples were lightly crushed with pestle in a mortar, resulting fractured pellet samples, which were subsequently processed for SEM analysis.
Before each SEM experiment, the surface of the samples was gold coated in argon atmosphere by means of an E5400 Sputter Coater (Bio-RAD Polaron, Kennett Square, PA, USA), thus avoiding the charging of the pellet samples during the analysis. The electron microscope has two detectors—a Everhart-Thornley detector (ETD) for secondary electrons and a back-scatter detector (BSED) and a magnification range of 50× to 1,000,000×. Also, the equipment has an energy dispersive X-ray analyzer (EDAX), which allows the local chemical composition and the distribution of significant elements on the pellet surface to be determined.
SEM images with 800×, 1000×, 2000×, and 4000× degree of magnification were used for the study of the surface morphology. Furthermore, X ray spectra were recorded for samples of meloxicam pellets, using the EDAX detector of the electron microscope, with the aim of elucidating the nature of the formations that could be observed on the surface of the pellets in the images with a high degree of magnification.
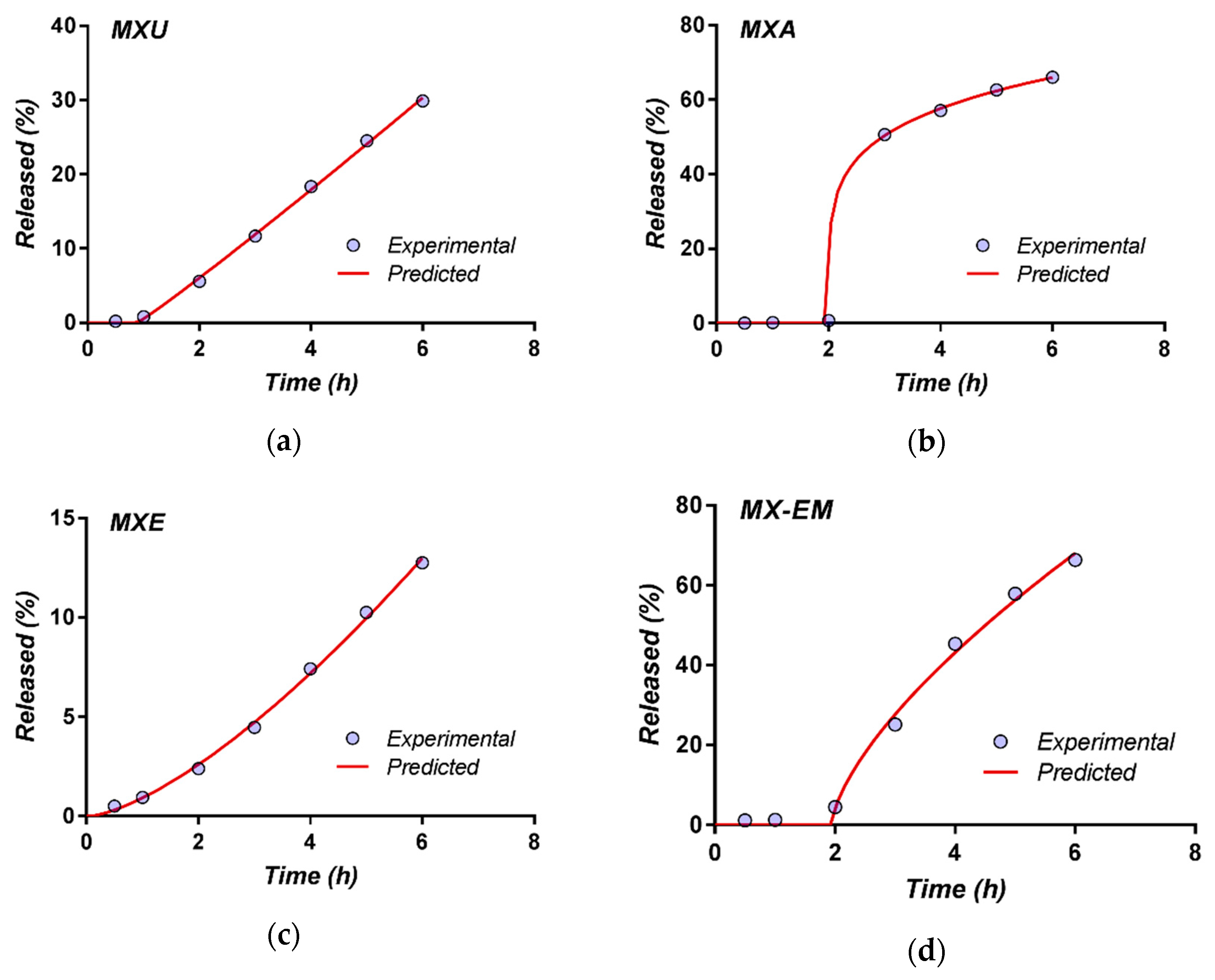
2.2.5. Study of the In Vitro Release Profile of Meloxicam from the Experimental Multi-Particulate Systems
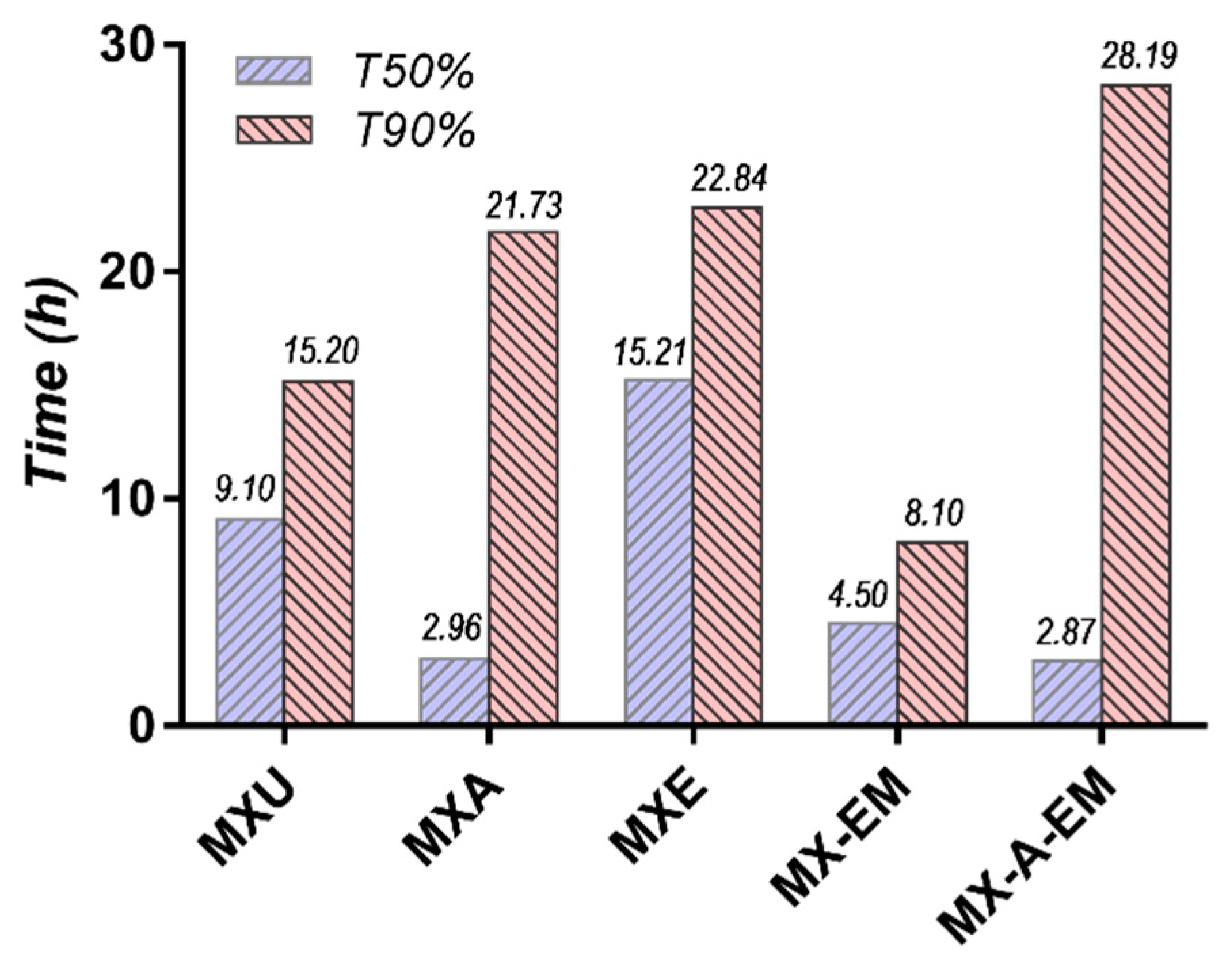
Evaluation of the in vitro release profiles of meloxicam from uncoated and film-coated pellets was performed in order to study the impact of the polymeric coating on the API release profile and subsequently the potential therapeutic performances of the experimental pellets.
In order to study meloxicam release kinetics, the experimental pellets were formulated as single-unit dosage forms (size 00 hard gelatin capsules, Capsugel, Bornem, Belgium).
Accurately weighed pellets (0.625 g), equivalent of 15 mg of meloxicam were manually loaded into the gelatin capsules, according to the method described by Manda A. et al. [
43].
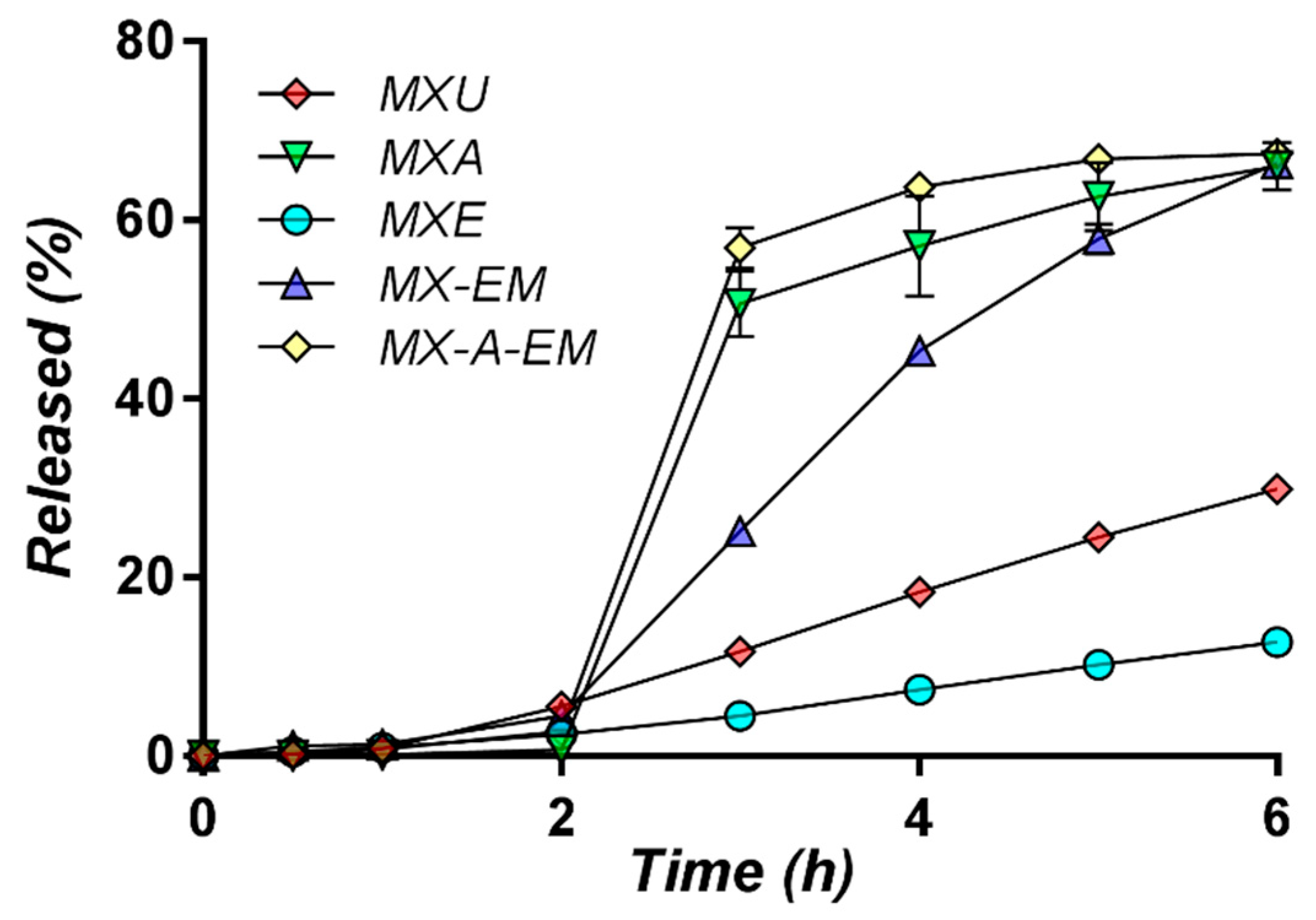
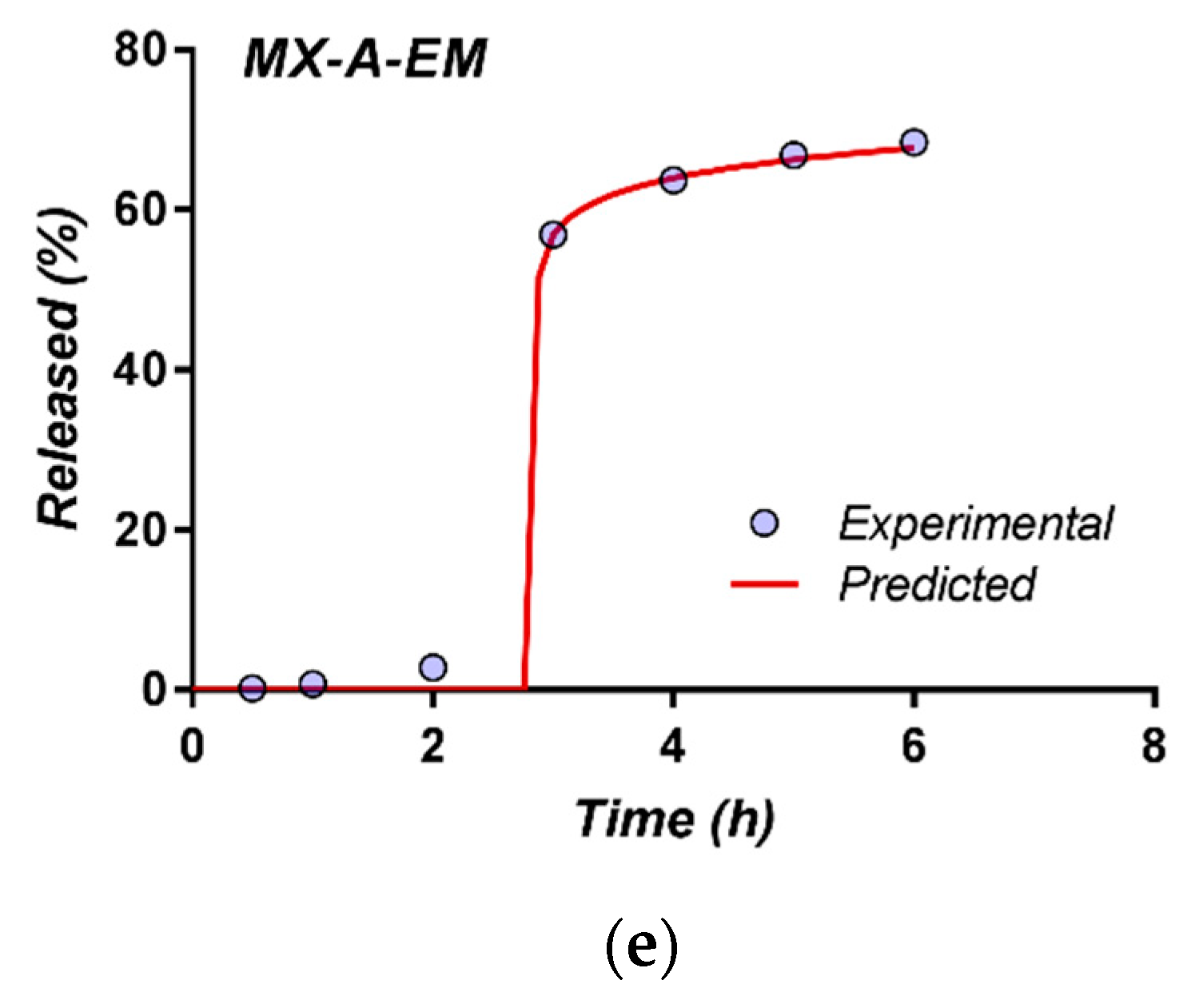
Capsules filled with the previously obtained meloxicam pellets (MXU, MXA, MXE, and MX-EM) were prepared and included in this study. Furthermore, in an additional experiment, the capsule content was represented by a combination of meloxicam pellets coated with Acryl-EZE® (MXA) and pellets coated with the composite film in which Eudragit® was associated with Methocel™ (MX-EM). By combining the two fractions of pellets into the same dosage unit, it was intended to achieve a reduced rate of release for the first two hours of the dissolution test (from the fraction of pellets coated with Eudragit® associated with Methocel™). The amount of drug released from these pellets in the pH 6.8 dissolution media was supplemented by the one yielded from the pellets coated with Acryl-EZE®, following the dissolution of this film at changing the pH of the medium. The two fractions of pellets were combined in a 1:1 ratio so that the dosage unit strength would be of 15 mg meloxicam per capsule. This sample was coded “MX-A-EM.”
The in vitro release of meloxicam from the experimental pellet formulations was performed in a Hanson SR8 Dissolution Test System (Teledyne Hanson Research, Chatsworth, CA, USA) using USP Apparatus 2 (paddle) at 75 rpm and 37 ± 0.5 °C. In vitro drug release testing was performed following the recommendations for delayed-release formulations (method A) from the United States Pharmacopoeia [
44] and comprised of two stages: An acid stage (2 h) in simulated gastric conditions (750 mL of 0.1 N hydrochloric acid) followed by a 4-h buffer stage (1000 mL of 50 mM pH = 6.8 phosphate buffer), for testing the drug release rate in intestinal conditions. After the completion of the acidic stage, a volume of 250 mL of preheated 0.20 M tribasic sodium phosphate was added, and the pH adjusted, if necessary, to a value of 6.8 ± 0.05.
Aliquots of 5 ± 0.1 mL were withdrawn at 0.5, 1, 2, 3, 4, 5, and 6 h, and replaced with blank medium at the same temperature. Six replicates were performed for each of the tested formulations. Dissolution profile of pure drug in both media as was used for reference.
The samples were filtered through a 0.45 mm regenerated cellulose non-sterile filters (Phenomenex, Torrance, CA, USA), diluted as needed, and analyzed by UV spectroscopy versus blank medium, using a Jasco V-530 spectrophotometer (Jasco International Co., Tokio, Japan) at 334 nm for pH 1.2 and 361.4 nm for pH 6.8.
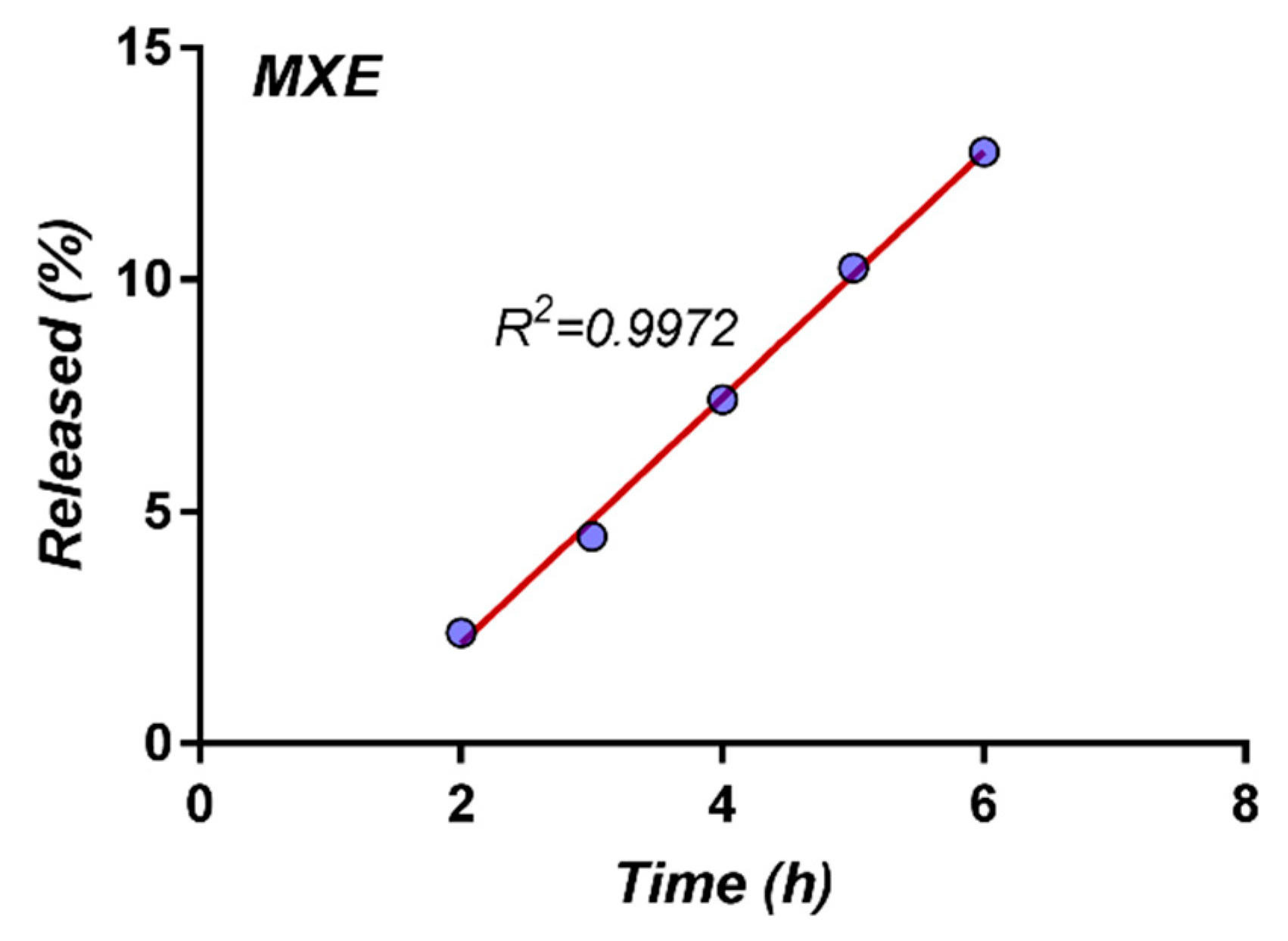
2.2.6. Mathematical Modeling of the Meloxicam In Vitro Release Kinetics
One method of elucidating the mechanism of API release from dosage forms is based on the best fit of experimental data on different mathematical models. [
45].
The modeling of meloxicam release profiles was performed by using the DDSolver Excel add-in software package [
46]. In order to elucidate the mechanism of meloxicam release from the experimental multi-particulate systems, first 60% drug release data were fitted in Korsmeyer-Peppas model [
47]:
where
F is the percentage of drug released at time
t,
is the release constant incorporating structural and geometric characteristics of the dosage form, and
n is the diffusional exponent indicating the drug-release mechanism.