The History of Photovoltaics with Emphasis on CdTe Solar Cells and Modules
Abstract
1. Introduction
2. Inorganic Photovoltaics: A Brief History
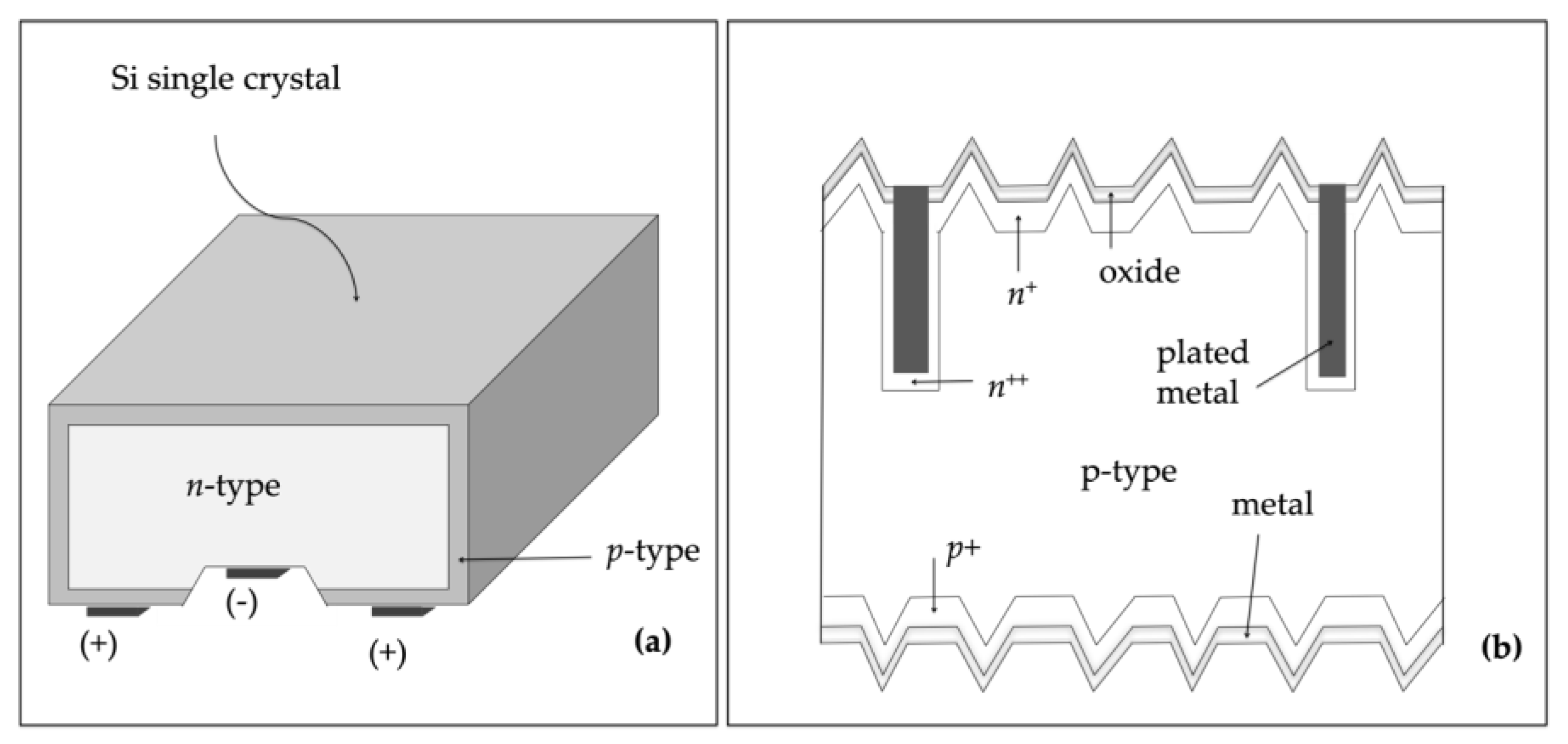
2.1. Silicon (Si)
2.2. Gallium Arsenide (GaAs)
- Monocrystalline Ge substrate that, in addition to allowing an epitaxial growth of GaAs, provides a p/n junction sensitive to infrared light;
- Epitaxial GaAs layer, thin enough to allow infrared light to reach the junction with Ge, grown by MOCVD process;
- Upper AlGaAs layer with the primary function of eliminating surface defects and acting as a window layer for the cell;
- Antireflecting bi-layer (SiO2 + Al2O3) coating with the double function of minimizing the sunlight reflection and preventing any damage to the underneath AlGaAs layer due to humidity;
- Interdigitated metal (Ti–Pd–Ag) external contacts. In order to avoid any possible diffusion of metal atoms into the active layers, a capping p+ GaAs coating was deposited between AlGaAs and the contacts.
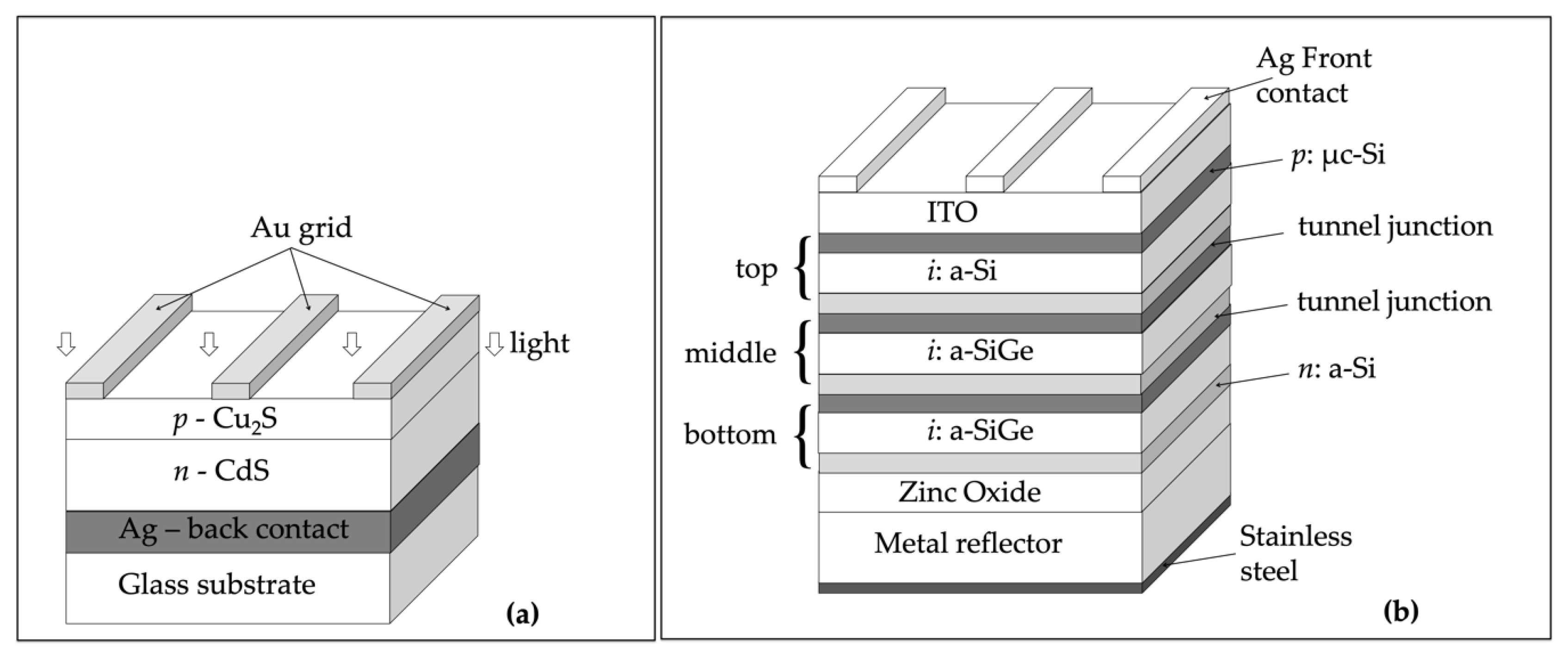
2.3. Copper Sulfide (Cu2S)
2.4. Hydrogenated Amorphous Silicon (a-Si:H)
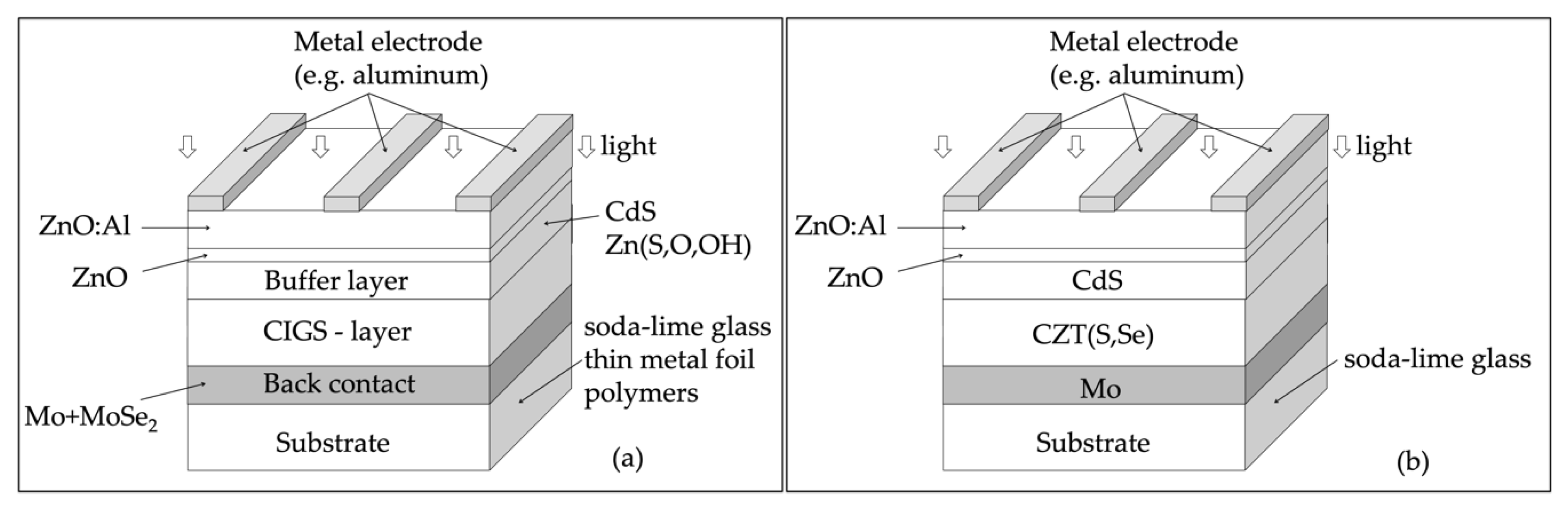
2.5. Copper Indium Gallium Diselenide (CuInGaSe2)
2.6. Cu2ZnSn(S, Se)4—(CZTSSe)
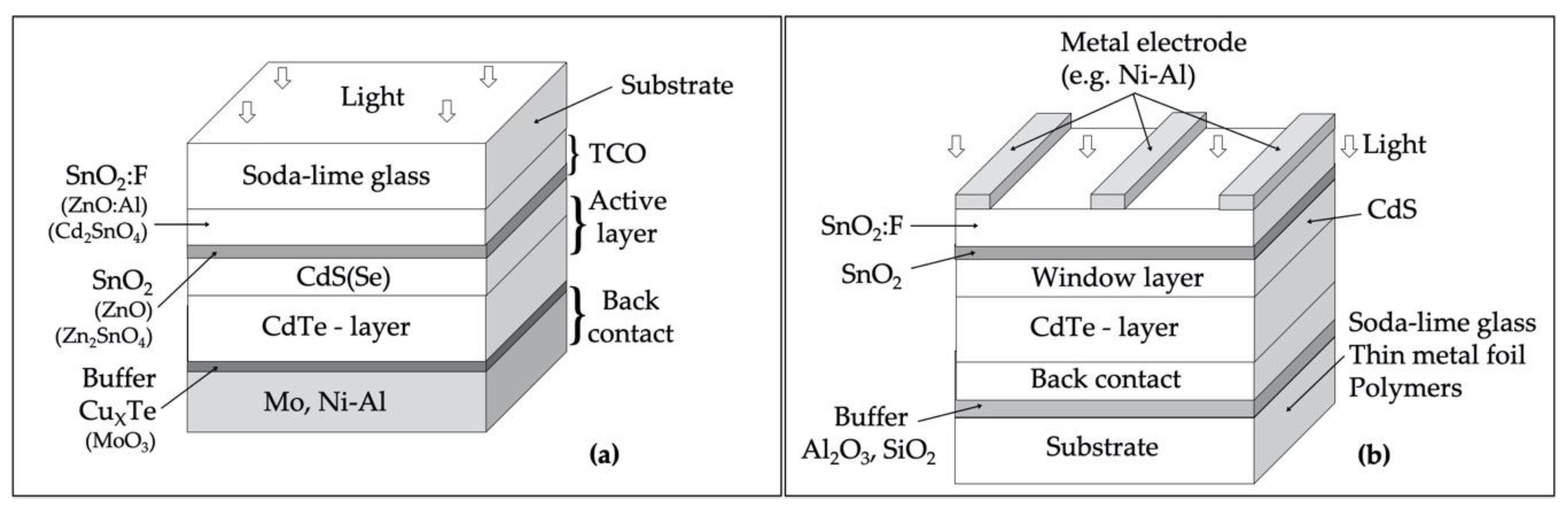
3. CdTe-Based Technology
3.1. The Solar Cell
3.1.1. The Substrate
3.1.2. The Transparent Electrode
- Large enough to avoid optical absorption;
- Small enough to allow the filling of the conduction band by a near-free electron at the working temperature.
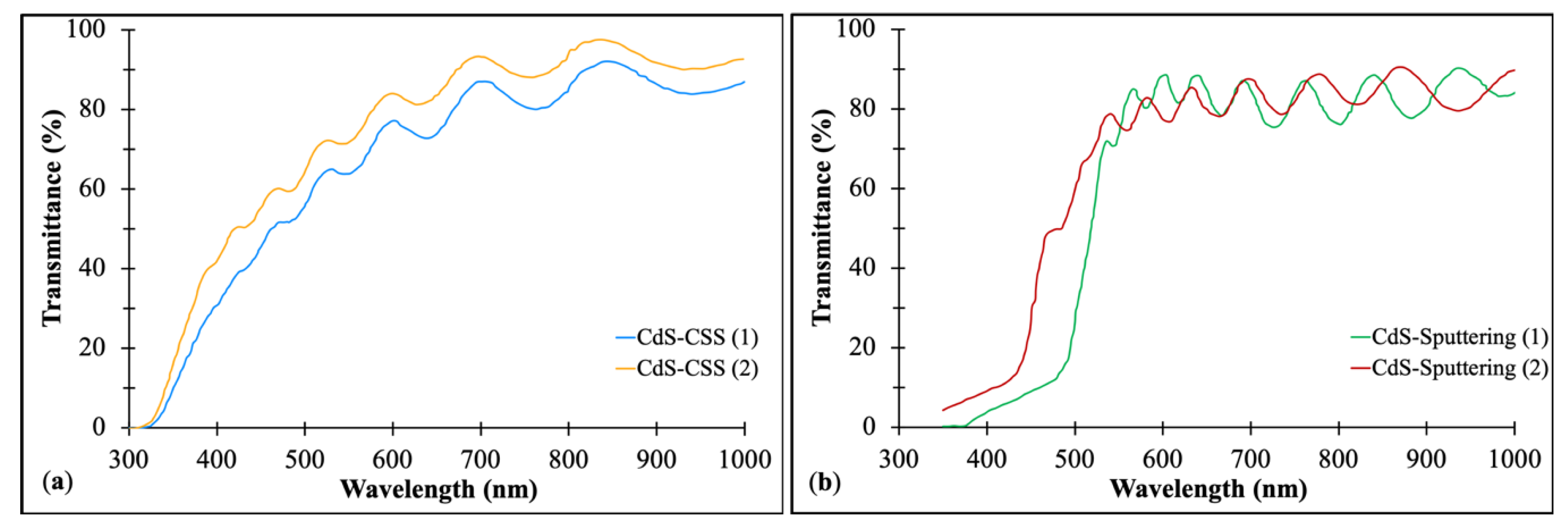
3.1.3. The Window Layer
3.1.4. The Absorber Layer
3.1.5. The Heat-Treatment
3.1.6. The Back-Contact
3.2. The Industrial Production
3.3. Recycling and Life Cycle Assessment
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gill, W.D.; Bube, R.H. Photovoltaic properties of Cu2S-CdS heterojunctions. J. Appl. Phys. 1970, 41, 3731. [Google Scholar] [CrossRef]
- Devaney, W.; Barnett, A.; Storti, G.; Meakin, J. The design and fabrication of CdS/Cu2S cells of 8.5-percent conversion efficiency. IEEE Trans. Electron Devices 1979. [Google Scholar] [CrossRef]
- Green, M.A.; Hishikawa, I.; Dunlop, E.D.; Levi, D.H.; Hohl-Ebinger, J.; Yoshit, M.; Ho-Baillie, A.W. Solar cell efficiency tables (Version 54). Prog. Photovolt. Res. 2019, 27, 565–575. [Google Scholar] [CrossRef]
- Khattak, C.P.; Schmid, F. Growth of silicon ingots by HEM for photovoltaic applications. In Silicon Processing for Photovoltaics II, Amsterdam; Khattak, C.P., Ravi, K.V., Eds.; Elsevier: Amsterdam, The Netherlands, 1987. [Google Scholar] [CrossRef]
- Hanak, J.J. Monolithic solar cell panel of amorphous silicon. Sol. Energy 1979, 23, 145–147. [Google Scholar] [CrossRef]
- Becquerel, A.E. Mémoire sur les effets électriques produits sous l’influence des rayons solaires. C. R. 1839, 9, 561–567. [Google Scholar]
- Willoughby, S. Selenium: Its Electrical Qualities and the Effect of Light Thereon; Hayman Bros. and Lilly Printers: London, UK, 1877; pp. 1–21. [Google Scholar]
- Fritts, C. On the Fritts selenium cell and batteries. Nostrands Eng. Mag. 1885, 32, 388–395. [Google Scholar]
- Ohl, R. Light Sensitive Electric Device. U.S. Patent 2402662, 27 May 1941. [Google Scholar]
- Ohl, R. Light-Sensitive Electric Device Including Silicon. U.S. Patent 2443542, 27 May 1941. [Google Scholar]
- Kingsbury, E.; Ohl, R. Photoelectric properties of ionically bombarded silicon. Bell Syst. Tech. J. 1952, 31, 802–815. [Google Scholar] [CrossRef]
- Chapin, D.; Fuller, C.; Pearson, G. A new silicon p-n junction photocell for converting solar radiation into electrical power. J. Appl. Phys. 1954, 25, 676–677. [Google Scholar] [CrossRef]
- Shockley, W. Electrons and Holes in Semiconductors; D. van Nostrand: New York, NY, USA, 1953. [Google Scholar]
- Easton, R.L.; Votaw, M.J. Vanguard I IGY Satellite (1958 Beta). Rev. Sci. Instrum. 1959, 30, 70–75. [Google Scholar] [CrossRef]
- Czochralski, J. Ein neues verfahren zur messung der kristallisationsgeschwindigkeit der metalle. Z. Phys. Chem. 1918, 92, 219. [Google Scholar] [CrossRef]
- Langhammer, W. A Method for Producing High-Purity Silicon. Germany Patent DE1222481B, 25 February 1958. [Google Scholar]
- Gremmelmaier, R. GaAs-photoelement. Z. Phys. Chem. 1955, 10, 501. [Google Scholar]
- Gobat, A.; Lamorte, M.; Mclver, G. Characteristics of high-conversion-efficiency gallium arsenide solar cells. IRE Trans. Mil. Electron. 1962, 20. [Google Scholar] [CrossRef]
- Shockley, W.; Queisser, H.J. Detailed balance limit of efficiency of p-n junction solar cells. J. Appl. Phys. 1961, 32, 510. [Google Scholar] [CrossRef]
- Lofersky, J. Recent research on photovoltaic solar energy converters. Proc. IEEE 1963, 51, 667–674. [Google Scholar] [CrossRef]
- Ellis, B.; Moss, T.S. Calculated efficiencies of practical gaas and si solar cells including the effect of built-in electric fields. Solid State Electron. 1970, 13, 1. [Google Scholar] [CrossRef]
- Woodall, J.; Hovel, H. High-efficiency Ga1−xAlxAs-GaAs solar cells. Appl. Phys. Lett. 1990, 21, 379. [Google Scholar] [CrossRef]
- Hovel, H.; Woodall, J. Ga1−xAlxAs p-p-n heterojunction solar cells. J. Electrochem. Soc. 1973, 120, 1246. [Google Scholar] [CrossRef]
- Anspaugh, B.E. GaAs Solar Cell Radiation Handbook; Technical Report JPL 96-9; Jet Propulsion Laboratory-California Institute of Technology: Pasadena, CA, USA, 1 July 1996.
- Iles, P.A.; Chang, K.I.; Leung, D.; Yeh, Y.C.M. The role of the AlGaAs Window layer in GaAs Heteroface Solar Cells. In Proceedings of the 18th IEEE Photovoltaic Specialist Conference, Las Vegas, NV, USA, 21–25 October 1985; p. 304. [Google Scholar]
- Brambilla, L.; Caon, A.; Contini, R.; D’Accolti, G.; Rossi, E.; Verzeni, G.; Flores, C.; Paletta, F.; Rapp, E.; Viola, F. GaAs Photovoltaic Technology Application: Arsene Solar Array. In Proceedings of the 23rd IEEE Photovoltaic Specialists Conference, Louisville, KY, USA, 10–14 May 1993; p. 1453. [Google Scholar] [CrossRef]
- Swanson, R.M. The promise of Concentrators. Prog. Photvolt. Res. Appl. 2000, 8, 93–111. [Google Scholar] [CrossRef]
- Bosi, M.; Pelosi, C. The potential of III-V semiconductors as terrestrial photovoltaic devices. Prog. Photovolt. Res. Appl. 2007, 15, 51–68. [Google Scholar] [CrossRef]
- Konagai, M.S.M.; Takahashi, K. High efficiency GaAs thin film solar cells by peeled film technology. J. Crys. Growth 1978, 45, 277–280. [Google Scholar] [CrossRef]
- Lin, Q.; Huang, H.; Jing, Y.; Fu, H.; Chang, P.; Li, D.; Yao, Y.; Fan, Z. Flexible photovoltaic technologies. J. Mater. Chem. 2014, 2, 1233–1247. [Google Scholar] [CrossRef]
- Adams, J.; Elarde, V.; Hains, A.; Stender, C.; Tuminello, F.; Youtsey, C.; Osowski, M. Demonstration of multiple substrate reuses for inverted metamorphic solar cells. In Proceedings of the 38th IEEE PV Specialists Conf. (PVSC), Austin, TX, USA, 3–8 June 2012. [Google Scholar] [CrossRef]
- Bauhuis, G.J.; Mulder, P.; Haverkamp, E.J.; Schermer, J.J.; Bongers, E.; Oomen, G.; Strobl, G. Wafer reuse for repeated growth of III-V solar cells. Prog. Photovolt. Res. Appl. 2010, 18, 155–159. [Google Scholar] [CrossRef]
- Lee, K.; Zimmerman, J.D.; Xiao, X.; Sun, K.; Forrest, S.R. Reuse of GaAs substrates for epitaxial lift-off by employing protection layers. J. Appl. Phys. 2012, 111, 033527–0033532. [Google Scholar] [CrossRef]
- Hall, R.B.; Birkmire, R.W.; Phillips, J.E.; Meakin, J.D. Thin-film polycrystalline Cu2S/Cd1−xZnx solar cells of 10% efficiency. Appl. Phys. Lett. 1981, 38, 925. [Google Scholar] [CrossRef]
- Brandhorst, H.W., Jr.; Bernatowicz, D.T. The Degradation of Cu2S-CdS Thin Film Solar Cells Under Simulated Orbital Conditions. In Proceedings of the International Colloquium on Solar Cells, Toulouse, France, 6–10 July 1970. [Google Scholar]
- Spear, W.E.; le Comber, P.G. Substitutional doping of amorphous silicon. Solid State Commun. 1975, 17, 1193–1196. [Google Scholar] [CrossRef]
- Carlson, D.E.; Wronski, C.R. Amorphous silicon solar cell. Appl. Phys. Lett. 1976, 28, 671. [Google Scholar] [CrossRef]
- Staebler, D.L.; Wronski, C.R. Reversible conductivity changes in discharge-produced amorphous Si. Appl. Phys. Lett. 1977, 31, 291–294. [Google Scholar] [CrossRef]
- Meier, J.; Dubail, S.; Fluckinger, R.; Fisher, D.; Keppner, H.; Shah, A. Intrinsic microcrystalline silicon (µc-Si:H)—A promising new thin film solar cell material. In Proceedings of the 1st World Conference on Photovoltaic energy conversion, Waikoloa, HI, USA, 5–9 December 1994. [Google Scholar] [CrossRef]
- Matsuda, A. Control of plasma and surface conditions for low defect density a-Si:H at high growth rates. In Proceedings of the Conference Record of the Twenty Fifth IEEE Photovoltaic Specialists Conference, Washington, DC, USA, 13–17 May 1996. [Google Scholar] [CrossRef]
- Yang, J.; Guha, S. Amorphous silicon alloy materials and solar cells near the threshold of microcrystallinity. MRS Online Proc. Libr. Arch. 1999, 557. [Google Scholar] [CrossRef]
- Yang, J.; Banerjee, A.; Guha, S. Triple-junction amorphous silicon alloy solar cell with 14.6% initial and 13.0% stable conversion efficiencies. Appl. Phys. Lett. 1997, 70, 2975. [Google Scholar] [CrossRef]
- Wagner, S.; Shay, J.L.; Migliorato, P.; Kasper, H.M. CuInSe2/CdS heterojunction photovoltaic detectors. Appl. Phys. Lett. 1974, 434, 25. [Google Scholar] [CrossRef]
- Kazmerski, L.L.; White, F.R.; Ayyagari, M.S.; Juang, Y.J.; Patterson, R.P. Growth and characterization of thin film compound semiconductor photovoltaic heterojunctions. J. Vac. Sci. Technol. 1977, 14, 65. [Google Scholar] [CrossRef]
- Devaney, W.E.; Chen, W.S.; Stewart, J.M.; Mickelsen, R. Structure and properties of high efficiency ZnO/CdZnS/CuInGaSe2 solar cells. Electron Devices IEEE Trans. 1990, 37, 428–433. [Google Scholar] [CrossRef]
- Tuttle, J.R.; Ward, J.S.; Duda, A.; Berens, T.A.; Contreras, M.A.; Ramanathan, K.R.; Tennant, A.; Keane, J.; Cole, E.D.; Emery, K.; et al. The performance of Cu (In, Ga) Se2-based solar cells in conventional and concentrator applications. MRS Proc. 1996. [Google Scholar] [CrossRef]
- Contreras, M.; Egaas, B.; Ramanathan, K.; Hiltner, J.; Swartzlander, A.; Hasoon, F.; Noufi, R. Progress toward 20% efficiency in Cu(In,Ga)Se2 polycrystalline thin-film solar cells. Prog. Photovolt. Res. Appl. 1999, 7, 311–316. [Google Scholar] [CrossRef]
- Ramanathan, K.; Contreras, M.; Perkins, C.; Asher, S.; Hasoon, F.; Keane, J.; Young, D.; Romero, M.; Metzger, W.; Noufi, R.; et al. Properties of 19.2% efficiency ZnO/CdS/CuInGaSe2 thin-film solar cells. Prog. Photovolt. Res. Appl. 2003, 11, 225–230. [Google Scholar] [CrossRef]
- Menossi, D.; Bosio, A.; Romeo, N. Key Developments in CuInGaSe2 Thin Film Solar Cell; LAP LAMBERT Academic Publishing: Saarbrüchen, Germany, 2014. [Google Scholar]
- Rosa, G.; Bosio, A.; Menossi, D.; Romeo, N. How the starting precursor influences the properties of polycrystalline cuingase2 thin films prepared by sputtering and selenization. Energies 2016, 9, 354. [Google Scholar] [CrossRef]
- Repins, I.; Contreras, M.A.; Egaas, B.; DeHart, C.; Craig, J.S.; Perkins, L.; To, B.; Noufi, R. 19.9%-efficient ZnO/CdS/CuInGaSe2 solar cell with 81·2% fill factor. Prog. Photovolt. Res. Appl. 1999, 7, 311–316. [Google Scholar] [CrossRef]
- Jackson, P.; Hariskos, D.; Lotter, E.; Paetel, S.; Wuerz, R.; Menner, R.; Wischmann, W.; Powalla, M. New world record efficiency for Cu (In, Ga) Se2 thin-film solar cells beyond 20%. Prog. Photovolt. Res. Appl. 2011, 19, 894–897. [Google Scholar] [CrossRef]
- Chirilă, A.; Reinhard, P.; Pianezzi, F.; Bloesch, P.; Uhl, A.R.; Fella, C.; Kranz, L.; Keller, D.; Gretener, C.; Hagendorfer, H.; et al. Potassium-induced surface modification of Cu(In, Ga)Se2 thin films for high-efficiency solar cells. Nat. Mater. 2013, 12, 1107–1111. [Google Scholar] [CrossRef]
- Jackson, P.; Hariskos, D.; Wuerz, R.; Wischmann, W.; Powalla, M. Compositional investigation of potassium doped Cu (In, Ga)Se2 solar cells with efficiencies up to 20.8%. Phys. Status Solidi RRL 2014, 8, 219–222. [Google Scholar] [CrossRef]
- Jackson, P.; Hariskos, D.; Wuerz, R.; Kiowski, O.; Bauer, A.; Friedlmeier, T.M.; Powalla, M. Properties of Cu(In, Ga)Se2 solar cells with new record efficiencies up to 21.7%. Phys. Status Solidi RRL 2014, 9, 28–31. [Google Scholar] [CrossRef]
- Solar Frontier Achieves World Record Thin-Film Solar Cell Efficiency of 23.35%. 17 January 2019. Available online: http://www.solar-frontier.com/eng/news/2019/0117_press.html (accessed on 25 November 2019).
- Bazilian, M.; Onyeji, I.; Liebreich, M.; MacGill, I.; Chase, J.; Shah, J.; Gielen, D.; Arent, D.; Landfear, D.; Zhengrong, S. Re-considering the economics of photovoltaic power. Renew. Energy 2013, 53, 329–338. [Google Scholar] [CrossRef]
- Hernández-Moro, J.; Martínez-Duart, J.M. Analytical model for solar PV and CSP electricity costs: Present LCOE values and their future evolution. Renew. Sustain. Energy Rev. 2013, 20, 119–132. [Google Scholar] [CrossRef]
- Ueckerdt, F.; Hirtha, L.; Luderer, G.; Edenhofer, O. System LCOE: What are the costs of variable renewables? Energy 2013, 63, 61–75. [Google Scholar] [CrossRef]
- Feltrin, A.; Freundlich, A. Material considerations for terawatt level deployment of photovoltaics. Renew. Energy 2008, 33, 180–185. [Google Scholar] [CrossRef]
- Andersson, B.A. Materials Availability for large-scale thin-film photovoltaics. Prog. Photovolt. Res. Appl. 2000, 8, 61–76. [Google Scholar] [CrossRef]
- Guo, Q.; Ford, G.M.; Yang, W.C.; Walker, B.C.; Stach, E.A.; Hillhouse, H.W.A.R. Fabrication of 7.2% efficient CZTSSe solar cells using CZTS nanocrystals. J. Am. Chem. Soc. 2010, 132, 17384–17386. [Google Scholar] [CrossRef]
- Repins, I.; Beall, C.; Vora, N.; DeHart, C.; Kuciauskas, D.; Dippo, P.; To, B.; Mann, J.; Hsu, W.C.; Goodrich, A.; et al. Co-evaporated Cu2ZnSnSe4 films and devices. Sol. Energy Mater. Sol. Cells 2012, 101, 154–159. [Google Scholar] [CrossRef]
- Bag, S.; Gunawan, O.; Gokmen, T.; Zhu, Y.; Todorov, T.K.; Mitzi, D.B. Low band gap liquid-processed CZTSe solar cell with 10.1% efficiency. Energy Environ. Sci. 2012, 5, 7060–7065. [Google Scholar] [CrossRef]
- Katagiri, H.; Jimbo, K.; Maw, W.S.; Oishi, K.; Yamazaki, M.; Araki, H.; Takeuchi, A. Development of CZTS-based thin film solar cells. Thin Solid Films 2009, 517, 2455–2460. [Google Scholar] [CrossRef]
- Wang, W.; Winkler, M.T.; Gunawan, O.; Gokmen, T.; Todorov, T.K.; Zhu, Y.; Mitzi, D.B. Device characteristics of CZTSSe thin-film solar cells with 12.6% efficiency. Adv. Energy Mater. 2014, 4, 1301465. [Google Scholar] [CrossRef]
- Kumar, M.S.; Madhusudanan, S.P.; Batabyal, S.K. Substitution of Zn in Earth-Abundant Cu2ZnSn(S,Se)4 based thin film solar cells—A status review. Sol. Energy Mater. Sol. Cells 2018, 185, 287–299. [Google Scholar] [CrossRef]
- Metzger, W.K.; Gloeckler, M. The impact of charged grain boundaries on thin-film solar cells and characterization. J. Appl. Phys. 2005, 98, 063701. [Google Scholar] [CrossRef]
- Rau, U.; Taretto, K.; Siebentritt, S. Grain boundaries in Cu(In, Ga)(Se, S)2 thin-film solar cells. Appl. Phys. A 2009, 96, 221. [Google Scholar] [CrossRef]
- Jiang, C.S.; Noufi, R.; Ramanathan, K.; AbuShama, J.; Moutinho, H.R.; Al-Jassim, M.M. Does the local built-in potential on grain boundaries of Cu (In, Ga)Se2 thin films benefit photovoltaic performance of the device? Appl. Phys. Lett. 2004, 85, 2625. [Google Scholar] [CrossRef]
- Nakazawa, T.; Takamizawa, K.; Ito, K. High efficiency indium oxide/cadmium telluride solar cells. Appl. Phys. Lett. 1987, 50, 279. [Google Scholar] [CrossRef]
- Aranovich, J.A.; Golmayo, D.; Fahrenbruch, A.L.; Bube, R.H. Photovoltaic properties of ZnO/CdTe heterojunctions prepared by spray pyrolysis. J. Appl. Phys. 1980, 51, 4260–4268. [Google Scholar] [CrossRef]
- Arroyo, J.M.; Marfaing, Y.; Cohen-Solal, G.; Triboulet, R. Electric and photovoltaic properties of CdTe pn homojunctions. Sol. Energy Mater. 1979, 171, 171–180. [Google Scholar] [CrossRef]
- Bonnet, D.; Rabenhorst, H. New results on the development of a thin film p-CdTe/n-CdS heterojunction solar cell. In Proceedings of the Conference Record of the 9th IEEE Photovoltaic Specialists Conference, Silver Springs, Atlantic City, NJ, USA, 2–4 May 1972. [Google Scholar]
- Tyan, Y.S.; Albuerne, E.A. Efficient thin film CdS/CdTe solar cells. In Proceedings of the 16th IEEE Photovoltaic Specialist Conference, New York, NY, USA, 27–30 September 1982. [Google Scholar]
- Britt, J.; Ferekides, C. Thin-film CdS/CdTe solar cell with 15.8% efficiency. Appl. Phys. Lett. 1993, 62, 2851–2852. [Google Scholar] [CrossRef]
- Wu, X.; Keane, J.C.; Dhere, R.G.; Dehert, C.; Albin, D.S.; Dude, A.; Gessert, T.A.; Asher, S.; Levi, D.; Sheldon, P. 16.5% Efficient CdS/CdTe Polycrystalline Thin-Film Solar cell. In Proceedings of the 17th European Photovoltaic Solar Energy Conference, Munich, Germany, 22–26 October 2001. [Google Scholar]
- First Solar Establishes New World Record for CdTe Efficiency. 23 February 2016. Available online: https://www.solarpowerworldonline.com/2016/02/24939/ (accessed on 20 January 2020).
- Cunningham, D.; Rubcich, M.; Skinner, D. Cadmium telluride PV module manufacturing at BP Solar. Prog. Photovolt. Res. Appl. 2002, 10, 159–168. [Google Scholar] [CrossRef]
- Bosio, A.; Menossi, D.; Mazzamuto, S.; Romeo, N. Manufacturing of CdTe thin film photovoltaic modules. Thin Solid Films 2011, 519, 7522–7525. [Google Scholar] [CrossRef]
- FirstSolar.com, Series 6 Datasheet. Available online: http://www.firstsolar.com/-/media/First-Solar/Technical-Documents/Series-6-Datasheets/Series-6-Datasheet.ashx (accessed on 20 January 2020).
- Gretener, C.; Perrenoud, J.; Kranz, L.; Kneer, L.; Schmitt, R.; Buecheler, S.; Tiwari, A. CdTe/CdS thin film solar cells grown in substrate configuration. Prog. Photovolt. Res. Appl. 2013, 21, 1580–1586. [Google Scholar] [CrossRef]
- Dhere, R.; Duenow, J.; DeHart, C.; Li, J.; Kuciauskas, D.; Gessert, T. Development of substrate structure CdTe photovoltaic devices with performance exceeding 10%. In Proceedings of the Photovoltaic Specialists Conference, 38th IEEE, Austin, TX, USA, 3–8 June 2012. [Google Scholar] [CrossRef]
- Bosio, A.; Rosa, G. Past present and future of the thin film CdTe/CdS solar cells. Sol. Energy 2018, 175, 31–43. [Google Scholar] [CrossRef]
- Salavei, A.; Artegiani, E.; Piccinelli, F.; di Mare, S.; Menossi, D.; Bosio, A.; Romeo, N.; Romeo, A. Flexible CdTe solar cells on polyimide and flexible glass substrates. In Proceedings of the 31st European Photovoltaic Solar Energy Conference, Berlin/Heidelberg, Germany, 14–18 September 2015. [Google Scholar]
- Moss, T. The interpretation of the properties of indium antimonide. In Proceedings of the Physical Society, Section B, London, UK, 2 June 1954. [Google Scholar] [CrossRef]
- Kumar, A.; Zhou, C. The race to replace tin-doped indium oxide: Which material will win? ACS Nano 2010, 4, 11–14. [Google Scholar] [CrossRef]
- Wu, X.; Ribelin, R.; Dhere, R.G.; Albin, D.S.; Gessert, T.A.; Asher, S.; Levi, D.H.; Mason, A.; Moutinho, H.R.; Sheldon, P. High-efficiency Cd2SnO4/Zn2SnO4/ZnxCd1−xS/CdS/CdTe polycrystalline thin-film solar cells. In Proceedings of the Conference Record of the Twenty-Eighth IEEE Photovoltaic Specialists Conference, Anchorage, AK, USA, 15–22 September 2000. [Google Scholar] [CrossRef]
- Kartopu, G.; Clayton, A.J.; Brooks, W.S.; Hodgson, S.D.; Barrioz, V.; Maertens, A.; Lamb, D.A.; Irvine, S.J. Effect of window layer composition in Cd1−xZnxS/CdTe solar cells. Prog. Photovolt. Res. Appl. 2014, 22, 18–23. [Google Scholar] [CrossRef]
- Ortega-Borges, R.; Lincot, D. Mechanism of chemical bath deposition of cadmium sulfide thin films in the ammonia-thiourea system in situ kinetic study and mod- elization. J. Electrochem. Soc. 1993, 140, 3464–3473. [Google Scholar] [CrossRef]
- Dona, J.M.; Herrero, J. Chemical bath deposition of CdS thin films: An approach to the chemical mechanism through study of the film microstructure. J. Electrochem. Soc. 1997, 144, 4081–4091. [Google Scholar] [CrossRef]
- Salavei, A.; Rimmaudo, I.; Piccinelli, F.; Menossi, D.; Romeo, N.; Bosio, A.; Dharmadasa, R.; Romeo, A. Flexible CdTe Solar Cells by a Low Temperature Process on ITO/ZnO Coated Polymers. In Proceedings of the 27th European Photovoltaic Solar Energy Conference and Exhibition, Frankfurt, Germany, 24–28 September 2012. [Google Scholar]
- Romeo, N.; Bosio, A.; Canevari, V. The role of CdS preparation method in the performance of CdTe/CdS thin film solar cell. In Proceedings of the 3rd World Conference on Photovoltaic Energy Conversion, WCPEC-3, Osaka, Japan, 11–18 May 2003. [Google Scholar] [CrossRef]
- Ferekides, C.; Ceekala, V.; Dugan, K.; Killian, L.; Oman, D.; Swaminathan, R.; Morel, D. Recent advances in thin film CdTe solar cells. In Proceedings of the AIP Conference, Lakewood, CO, USA, 16−19 May 1996. [Google Scholar] [CrossRef]
- Anthony, T.; Fahrenbruch, A.; Peters, M.; Bube, R. Electrical properties of CdTe films and junctions. J. Appl. Phys. 1985, 57, 400–410. [Google Scholar] [CrossRef]
- Chu, T.; Chu, S.; Ang, S. Electrical properties of CdS/CdTe heterojunctions. J. Appl. Phys. 1988, 64, 1233–1237. [Google Scholar] [CrossRef]
- Fahrenbruch, A.; Bube, R. Fundamentals of Solar Cells: Photovoltaic Solar Energy Conversion; Academic Press: New York, NY, USA, 1983. [Google Scholar] [CrossRef]
- Bosio, A.; Romeo, A.; Romeo, N. Polycrystalline CdTe thin films solar cells. In Alessio Bosio Alessandro Romeo. Thin Film Solar Cells: Current Status and Future Trends; Nova Science Publishers Inc.: New York, NY, USA, 2011. [Google Scholar]
- Bosio, A.; Rosa, G.; Menossi, D.; Romeo, N. How the chlorine treatment and the stoichiometry influences the grain boundary passivation in polycrystalline CdTe thin films. Energies 2016, 9, 254. [Google Scholar] [CrossRef]
- Rühle, S. Tabulated values of the Shockley–Queisser limit for single junction solar cells. Sol. Energy 2016, 130, 139–147. [Google Scholar] [CrossRef]
- Wei, S.; Zhang, S.; Zunger, A. First-principles calculation of band offsets, optical bowings, and defects in CdS, CdSe, CdTe, and their alloys. J. Appl. Phys. 2000, 87, 1304–1311. [Google Scholar] [CrossRef]
- Mia, M.; Swartz, C.; Paul, S.; Sohal, S.; Grice, C.; Yan, Y.; Holtz, M.; Li, J. Electrical and optical characterization of CdTe solar cells with CdS and CdSe buffers—A comparative study. J. Vac. Sci. Technol. B 2018, 36, 052904. [Google Scholar] [CrossRef]
- Kephart, J.; McCamy, J.; Ma, Z.; Ganjoo, A.; Alamgir, F.; Sampath, W. Band alignment of front contact layers for high-efficiency CdTe solar cells. Sol. Energy Mater. Sol. Cells 2016, 157, 266–275. [Google Scholar] [CrossRef]
- Munshi, A.; Kephart, J.; Abbas, A.; Shimpi, T.; Barth, K.; Walls, J.; Sampath, W. Polycrystalline CdTe photovoltaics with efficiency over 18% through improved absorber passivation and current collection. Sol. Energy Mater. Sol. Cells 2018, 176. [Google Scholar] [CrossRef]
- Fiducia, T.; Mendis, B.; Li, K.; Grovenor, C.; Munshi, A.; Barth, K.; Sampath, W.; Wright, L.; Abbas, A.; Bowers, J. Understanding the role of selenium in defect passivation for highly efficient selenium-alloyed cadmium telluride solar cells. Nat. Energy 2019, 504–511. [Google Scholar] [CrossRef]
- Romeo, A.; Bätzner, D.; Zogg, H.; Vignali, C.; Tiwari, A. Influence of CdS growth process on structural and photovoltaic properties of CdTe/CdS solar cells. Sol. Energy Mater. Sol. Cells 2001, 67, 311–321. [Google Scholar] [CrossRef]
- Moutinho, H.; Al-Jassim, M.; Abulfotuh, F.; Levi, D.; Dippo, P.; Dhere, R.; Kazmerski, L. Studies of Recrystallization of CdTe thin filmsafter CdCI, treatment. In Proceedings of the Conference Record of the Twenty Sixth IEEE Photovoltaic Specialists Conference, Anaheim, CA, USA, 29 September–3 October 1997. [Google Scholar]
- Perrenoud, J.; Buecheler, S.; Tiwari, A.N. Flexible CdTe solar cells and modules: Challenges and prospects. In Proceedings of the SPIE, San Diego, CA, USA, 2–4 August 2009. [Google Scholar] [CrossRef]
- Danaher, W.; Lyons, L. Photoelectrochemical cell with cadmium telluride film. Nature 1978, 271, 139. [Google Scholar] [CrossRef]
- Kröger, F. Cathodic deposition and characterization of metallic or semiconducting binary alloys or compounds. J. Electrochem. Soc. 1978, 125, 2028–2034. [Google Scholar] [CrossRef]
- Fulop, G.; Taylor, R. Electrodeposition of semiconductors. Ann. Rev. Mater. Sci. 1985, 15, 197–210. [Google Scholar] [CrossRef]
- Basol, B. High-efficiency electroplated heterojunction solar cell. J. Appl. Phys. 1984, 55, 601–603. [Google Scholar] [CrossRef]
- Ojo, A.; Dharmadasa, I. 15.3% efficient graded bandgap solar cells fabricated using electroplated CdS and CdTe thin films. Sol. Energy 2016, 136, 10–14. [Google Scholar] [CrossRef]
- Soliman, M.M.; Shabana, M.M.; Abulfotuh, F. CdS/CdTe solar cell using sputtering technique. Renew. Energy 1996, 8, 386–389. [Google Scholar] [CrossRef]
- Paudel, N.; Wieland, K.; Compaan, A. Ultrathin CdS/CdTe solar cells by sputtering. Sol. Energy Mater. Sol. Cells 2012, 105, 109–112. [Google Scholar] [CrossRef]
- Compaan, A.D.; Gupta, A.; Lee, S.; Wang, S.; Drayton, J. High efficiency, magnetron sputtered CdS/CdTe solar cells. Sol. Energy 2004, 77, 815–822. [Google Scholar] [CrossRef]
- Bosio, A.; Romeo, N.; Mazzamuto, S.; Canevari, V. Polycrystalline CdTe thin films for photovoltaic applications. Progress Cryst. Growth Charact. Mater. 2006, 52, 247–279. [Google Scholar] [CrossRef]
- Bayhan, H. Investigation of the effect of CdCl2 processing on vacuum deposited CdS/CdTe thin film solar cells by DLTS. J. Phys. Chem. Solids 2004, 65, 1817–1822. [Google Scholar] [CrossRef]
- Hiie, J. CdTe:CdCl2:O2 annealing process. Thin Solid Films 2003, 431, 90–93. [Google Scholar] [CrossRef]
- Niles, D.W.; Waters, D.; Rose, D. Chemical reactivity of CdCl2 wet-deposited on CdTe films studied by X-ray photoelectron spectroscopy. Appl. Surf. Sci. 1998, 136, 221–229. [Google Scholar] [CrossRef]
- Romeo, N.; Bosio, A.; Tedeschi, R.; Canevari, V. A highly efficient and stable CdTe/CdS thin film solar cell. Sol. Energy Mater. Sol. Cells 1999, 58, 209–218. [Google Scholar] [CrossRef]
- Major, J.; Treharne, R.; Phillips, L.; Durose, K. A low-cost non-toxic post-growth activation step for CdTe solar cells. Nature 2014, 511, 334. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Reiter, N.; Powell, R.; Sasala, R.; Meyers, P. Vapor chloride treatment of polycrystalline CdTe/CdS films. In Proceedings of the 1994 IEEE 1st World Conference on Photovoltaic Energy Conversion—WCPEC, Waikiloa, HI, USA, 5–9 December 1994. [Google Scholar] [CrossRef]
- Qu, Y.; Meyers, P.; McCandless, B. HCl vapor post-deposition heat treatment of CdTe/CdS films. In Proceedings of the Twenty Fifth IEEE Photovoltaic Specialists Conference, Washington, DC, USA, 13–17 May 1996. [Google Scholar] [CrossRef]
- Albright, S.; Jordan, J.F.; Akerman, B.; Chamberlain, R.C. Developments on CdS/CdTe photovoltaic panels at Photon Energy, Inc. Sol. Cells 1989, 27, 77. [Google Scholar] [CrossRef]
- Gessert, T.A.; Mason, A.R.; Sheldon, P.; Swartzlander, A.; Niles, D.; Coutts, T.J. Development of Cu-doped ZnTe as back-contact interface layer for thin-film CdS/CdTe solar cells. J. Vac. Sci. Technol. 1996, 14, 806. [Google Scholar] [CrossRef]
- Uda, H.; Ikegami, S.; Sonomura, H. Compositional change of the Au–Cu2Te contact for thin-film CdS/CdTe solar cells. Jpn. J. Appl. Phys. 1990, 29, 495. [Google Scholar] [CrossRef]
- McCandless, B.E.; Qu, Y.; Birkmire, R.W. A Treatment to Allow Contacting CdTe with Different Conductors. In Proceedings of the First World Conference on Photovoltaic Energy Conversion, Waikoloa, HI, USA, 5–9 December 1994. [Google Scholar]
- Du, M. First-principles study of back-contact effects on CdTe thin-film solar cells. Phys. Rev. B 2009, 80, 205322. [Google Scholar] [CrossRef]
- Durose, K.; Boyle, D.; Abken, A.; Ottley, C.; Nollet, P.; Degrave, S.; Burghelman, M.; Wendt, R.; Bonnet, D. Key aspects of CdTe/CdS solar cells. Phys. Status Solidi 2002, 229, 1055–1064. [Google Scholar] [CrossRef]
- Bosio, A.; Ciprian, R.; Lamperti, A.; Rago, I.; Ressel, B.; Rosa, G.; Stupard, M.; Weschke, E. Interface phenomena between CdTe and ZnTe:Cu back contact. Sol. Energy 2018, 176, 186–193. [Google Scholar] [CrossRef]
- Bosio, A.; Sozzi, M.; Menossi, D.; Selleri, S.; Cucinotta, A.; Romeo, N. Polycrystalline CdTe thin film mini-modules monolithically integrated by fiber laser. Thin Solid Films 2014, 562, 638–647. [Google Scholar] [CrossRef]
- Zayed, P.; Philippe, S. Acute oral and inhalation toxicities in rats with Cadmium Telluride. Int. J. Toxicol. 2009, 28, 259–265. [Google Scholar] [CrossRef]
- Fthenakis, V.M.; Fuhrmann, M.; Heiser, J.; Lanzirotti, A.; Fitts, J.; Wang, W. Emissions and encapsulation of cadmium in CdTe PV modules during fires. Prog. Photovolt. Res. Appl. 2005, 13, 713–723. [Google Scholar] [CrossRef]
- Kim, H.; Cha, K.; Fthenakis, V.M.; Sinha, P.; Hur, T. Life cycle assessment of cadmium telluride photovoltaic (CdTe PV) systems. Sol. Energy 2014, 103, 78–88. [Google Scholar] [CrossRef]
- Sinha, P. Life cycle materials and water management for CdTe photovoltaics. Sol. Energy Mater. Sol. Cells 2013, 119, 271–275. [Google Scholar] [CrossRef]







| Solar Cells | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameters/ Producers | Si Single-Crystal | Si Multi-Crystal | GaAs SJ | GaAs 3-J | GaAs 6-J | Cu2S | CIS | CIGS | CZTS | CdTe |
| PCE [%] | 26.7 | 23.2 | 32.8 | 37.9 | 47.1 § | 10.0 | 15.4 | 23.35 | 12.6 | 22.1 |
| Area [cm2] | 79 * | 247.79 *** | 1000 ** | 1047 ** | 0.099 | ≈1.0 | 100.0 *** | 1043 * | 0.4209 ** | 0.4798 * |
| Producer | Kaneka | Trina Solar | LG Electronics | Sharp | NREL | IEC | IPE | Solar Frontier | IBM | First Solar |
| Modules | ||||||||||
| PCE [%] | 24.4 | 20.4 | – | 31.2 | – | – | – | 18.6 | – | 19.0 |
| Area [cm2] | 13,177 * | 14,818 ** | – | 968 * | – | – | – | 10,858 ** | – | 23,573 * |
| Producer | Kaneka | Hanwa Q Cells | – | Sharp | – | – | – | Miasolé | – | First Solar |
| Oxide | SiO2 | Na2O | CaO | MgO | Al2O3 | K2O | TiO2 | SO3 | Fe2O3 | Others |
|---|---|---|---|---|---|---|---|---|---|---|
| Weight [%] | 72.85 | 12.42 | 8.15 | 4.09 | 1.27 | 0.47 | 0.37 | 0.18 | 0.10 | 0.10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bosio, A.; Pasini, S.; Romeo, N. The History of Photovoltaics with Emphasis on CdTe Solar Cells and Modules. Coatings 2020, 10, 344. https://doi.org/10.3390/coatings10040344
Bosio A, Pasini S, Romeo N. The History of Photovoltaics with Emphasis on CdTe Solar Cells and Modules. Coatings. 2020; 10(4):344. https://doi.org/10.3390/coatings10040344
Chicago/Turabian StyleBosio, Alessio, Stefano Pasini, and Nicola Romeo. 2020. "The History of Photovoltaics with Emphasis on CdTe Solar Cells and Modules" Coatings 10, no. 4: 344. https://doi.org/10.3390/coatings10040344
APA StyleBosio, A., Pasini, S., & Romeo, N. (2020). The History of Photovoltaics with Emphasis on CdTe Solar Cells and Modules. Coatings, 10(4), 344. https://doi.org/10.3390/coatings10040344





