Organometallic Coatings for Electroluminescence Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of IrQ(ppy)2 Organometallic
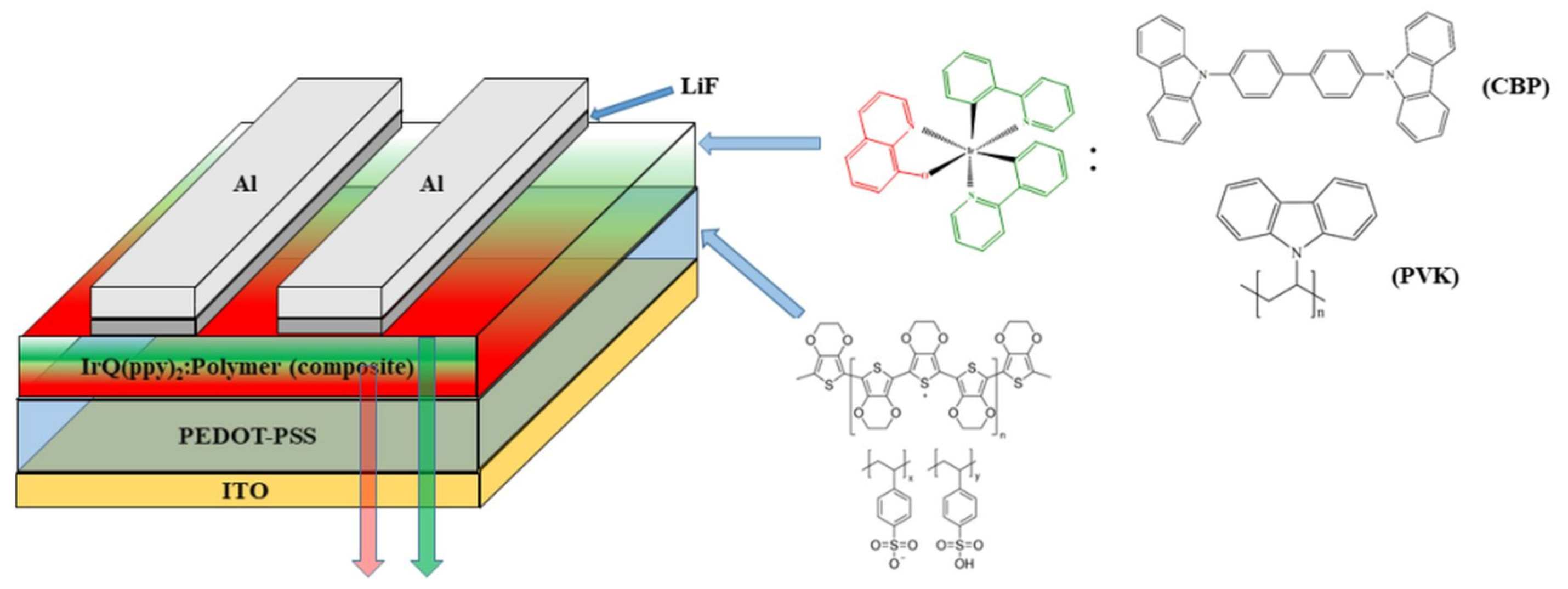
2.2. Devices Fabrication
3. Results
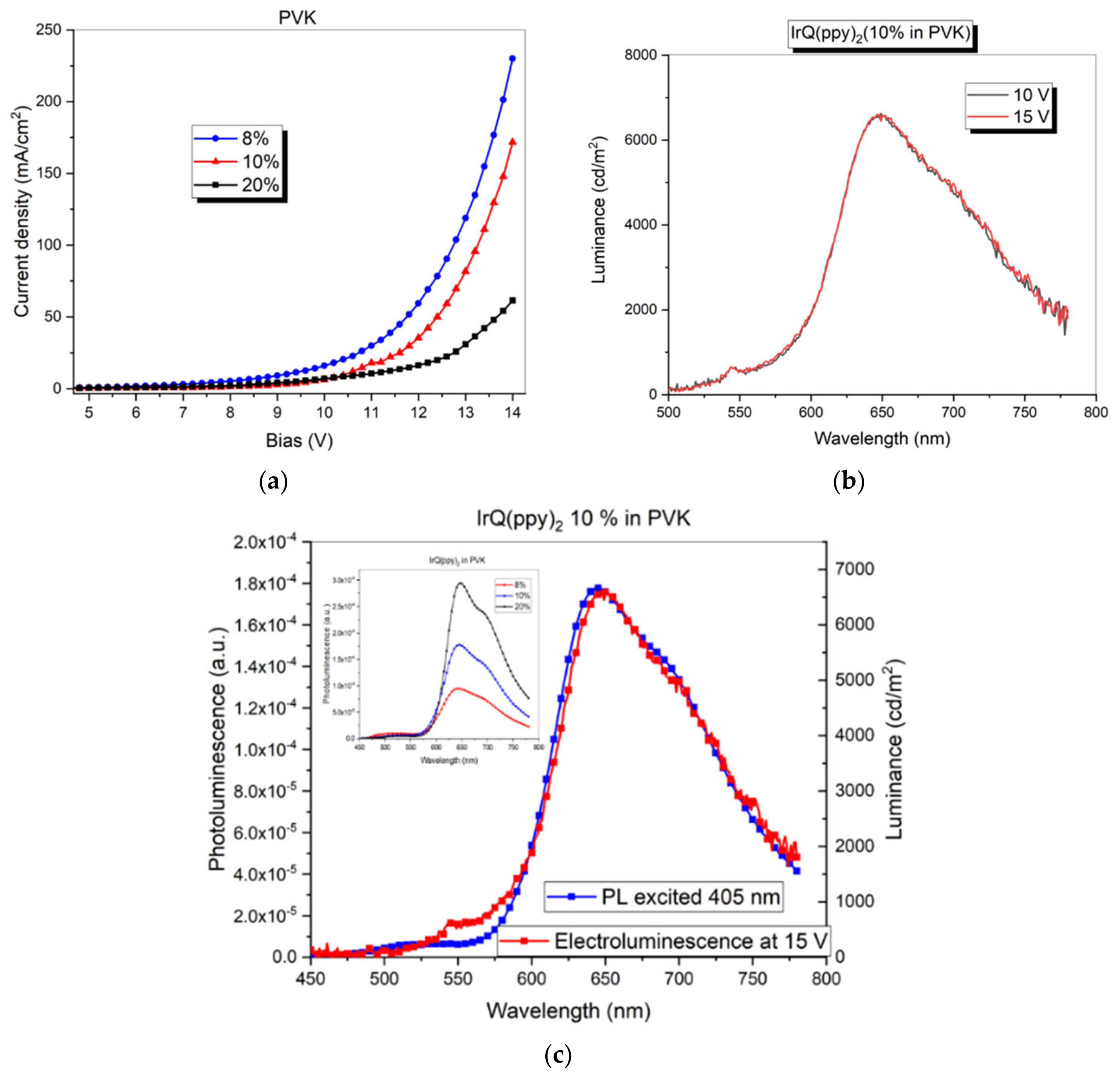
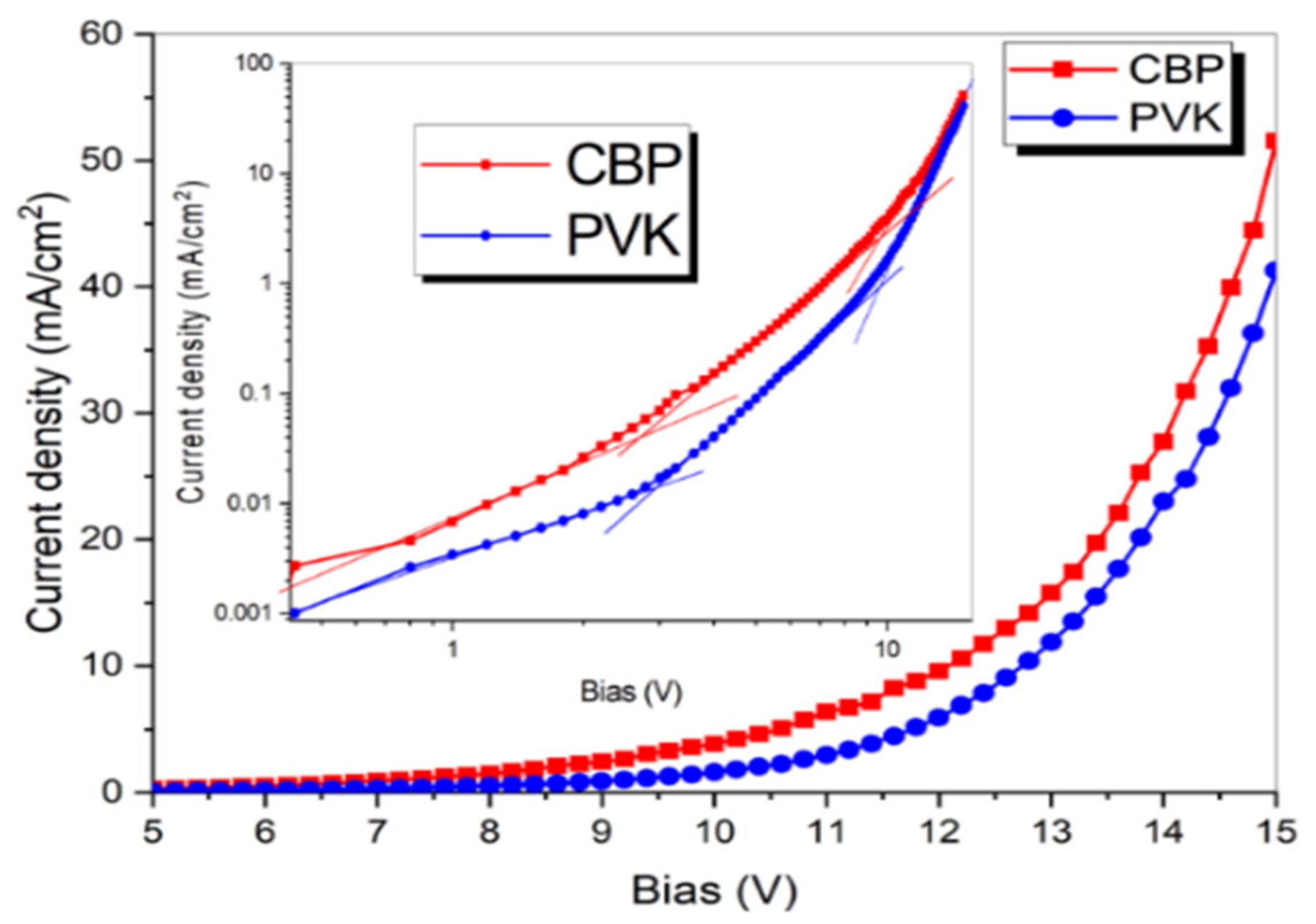
3.1. Current–Voltage Characteristics: Electroluminescence of IrQ(ppy)2 in PVK
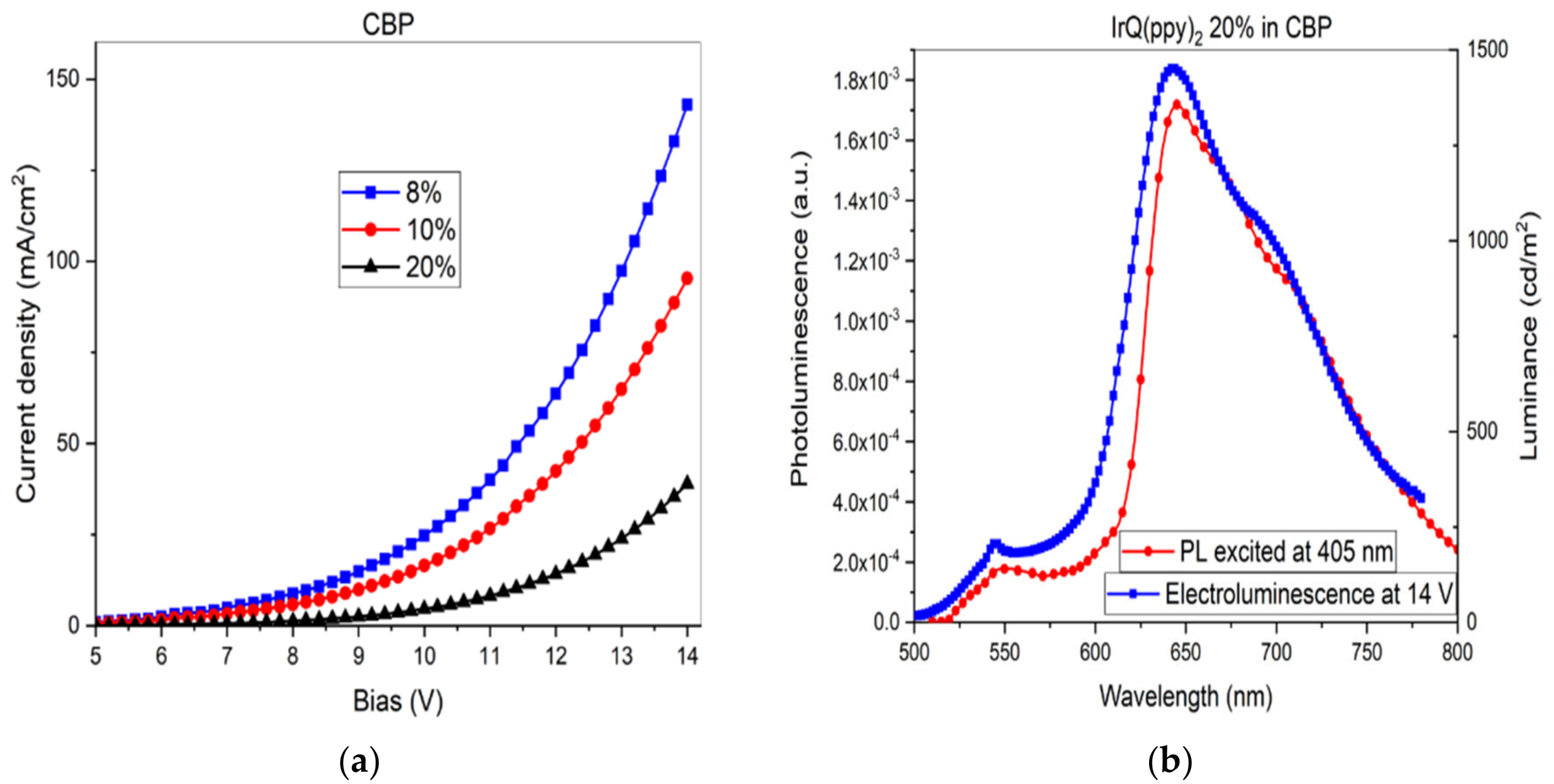
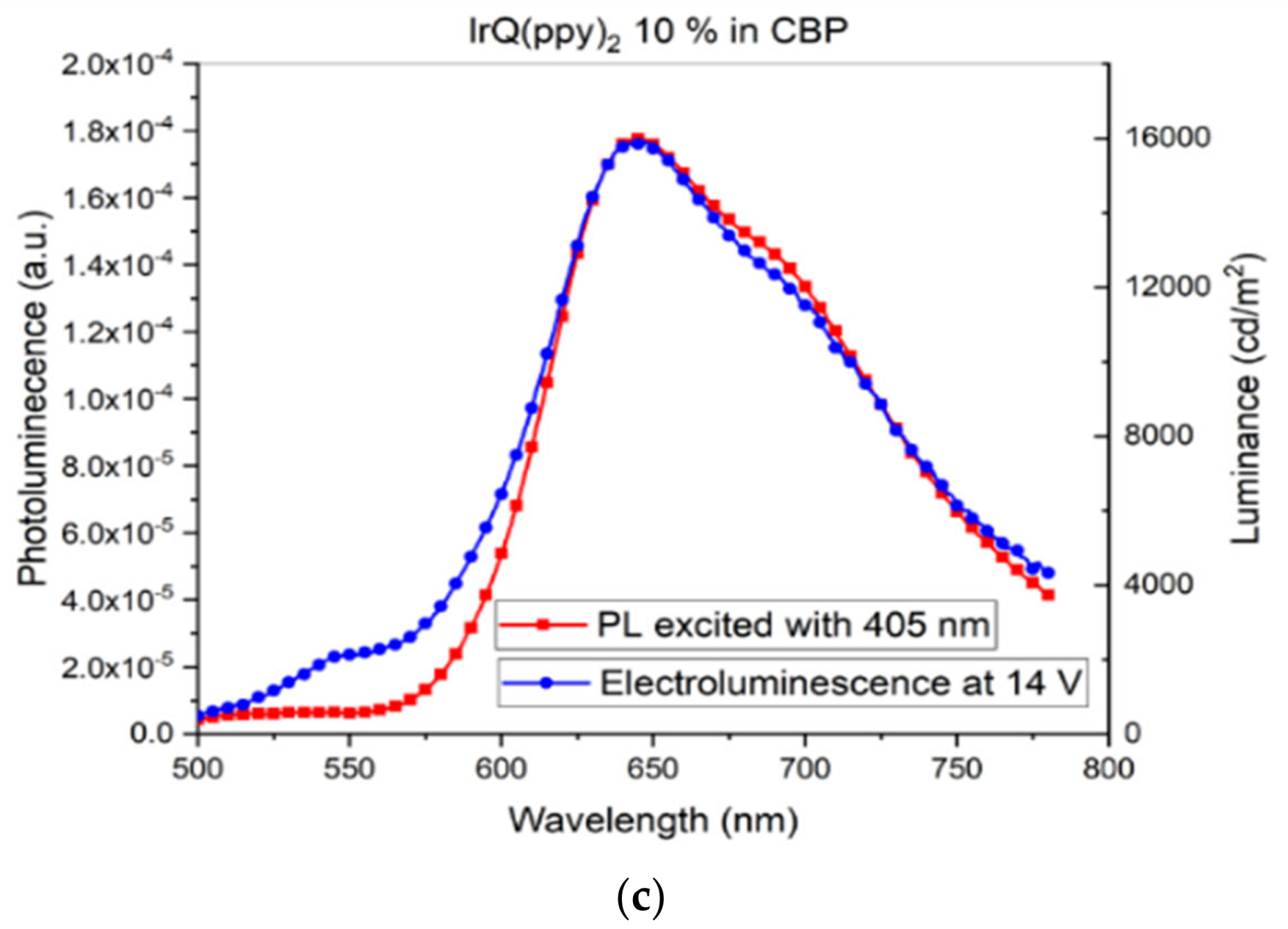
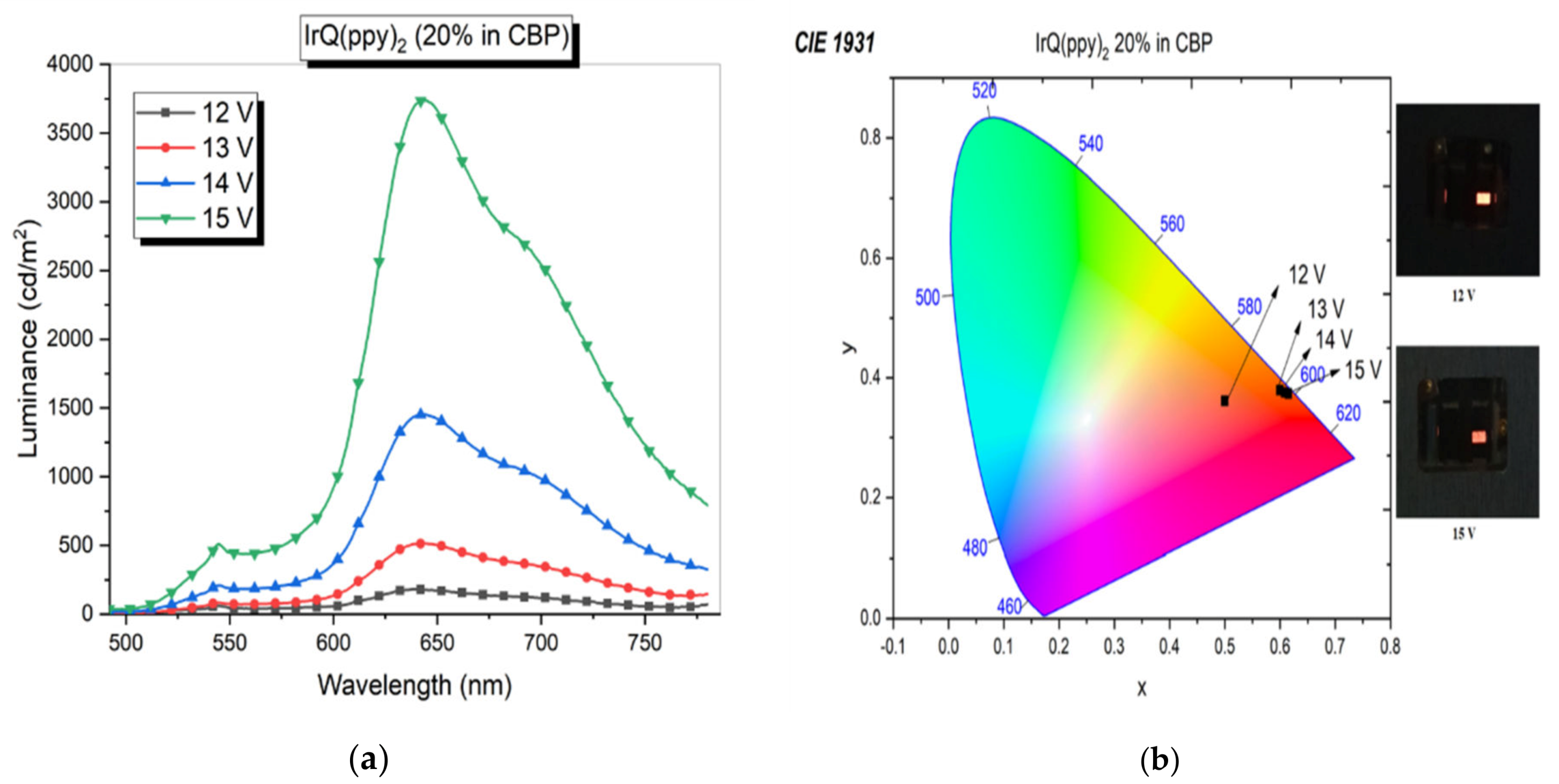
3.2. Current–Voltage Characteristics: Electroluminescence of IrQ(ppy)2 in CBP
4. Discussion
- Current efficiency in cd/A as the following equation:where the L (cd/m2) is the electroluminescence and j (mA/cm2) the current density.
- Power efficiency in lm/W as the following equation:
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tang, C.W.; Van Slyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- Della Rosa, G.; Vona, D.; Aloisi, A.; Ragni, R.; Di Corato, R.; Presti, M.L.; Cicco, S.R.; Altamura, E.; Taurino, A.; Catalano, M.; et al. Luminescent Silica-Based Nanostructures from in Vivo Iridium-Doped Diatoms Microalgae. ACS Sustain. Chem. Eng. 2019, 7, 2207–2215. [Google Scholar] [CrossRef]
- Binding, S.C.; Pernik, I.; Gonçales, V.R.; Wong, C.M.; Webster, R.F.; Cheong, S.; Tilley, R.D.; Garcia-Bennett, A.E.; Gooding, J.J.; Messerle, B.A. Simultaneous Functionalization of Carbon Surfaces with Rhodium and Iridium Organometallic Complexes: Hybrid Bimetallic Catalysts for Hydroamination. Organometallics 2019, 38, 780–787. [Google Scholar] [CrossRef]
- Jespersen, D.; Keen, B.; Day, J.I.; Singh, A.; Briles, J.; Mullins, D.; Weaver, J.D. Solubility of Iridium and Ruthenium Organometallic Photoredox Catalysts. Org. Process. Res. Dev. 2019, 23, 1087–1095. [Google Scholar] [CrossRef]
- Kong, D.; Tian, M.; Guo, L.; Liu, X.; Zhang, S.; Song, Y.; Meng, X.; Wu, S.; Zhang, L.; Liu, Z. Novel iridium(III) iminopyridine complexes: Synthetic, catalytic, and in vitro anticancer activity studies. J. Biol. Inorg. Chem. 2018, 23, 819–832. [Google Scholar] [CrossRef]
- Ritacco, I.; Russo, N.; Sicilia, E. DFT Investigation of the Mechanism of Action of Organoiridium (III) Complexes as Anticancer Agents. Inorg. Chem. 2015, 54, 10801–10810. [Google Scholar] [CrossRef]
- Lu, L.; Liu, L.J.; Chao, W.C.; Zhong, H.J.; Wang, M.; Chen, X.P.; Lu, J.J.; Li, R.; Ma, D.L.; Leung, C.H. Identification of an iridium(III) complex with anti-bacterial and anti-cancer activity. Sci. Rep. 2015, 5, 14544. [Google Scholar] [CrossRef]
- Dua, Q.; Yanga, Y.; Guoa, L.; Tiana, M.; Gea, X.; Tiana, Z.; Zhaoa, L.; Xua, Z.; Lia, J.; Liu, Z. Fluorescent half-sandwich phosphine-sulfonate iridium (III) and ruthenium (II) complexes as potential lysosome-targeted anticancer agents. Dyes Pigment. 2019, 162, 821–830. [Google Scholar] [CrossRef]
- Wang, J.; Liu, H.B.; Tong, Z.; Ha, C.S. Fluorescent/luminescent detection of natural amino acids by organometallic systems. Coord. Chem. Rev. 2015, 303, 139–184. [Google Scholar] [CrossRef]
- Caporale, C.; Massi, M. Cyclometalated iridium (III) complexes for life science. Coord. Chem. Rev. 2018, 363, 71–91. [Google Scholar] [CrossRef]
- Lowry, M.S.; Goldsmith, J.I.; Slinker, J.D.; Rohl, R.; Pascal, R.A.; Malliaras, G.G.; Bernhard, S. Single-Layer Electroluminescent Devices and Photoinduced Hydrogen Production from an Ionic Iridium (III) Complex. Chem. Mater. 2005, 17, 5712–5719. [Google Scholar] [CrossRef]
- McDaniel, N.D.; Coughlin, F.J.; Tinker, L.L.; Bernhard, S. Cyclometalated Iridium (III) Aquo Complexes: Efficient and Tunable Catalysts for the Homogeneous Oxidation of Water. J. Am. Chem. Soc. 2008, 130, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Farid, R.S.; Chang, I.J.; Winkler, J.R.; Gray, H.B. Photoinduced Electron Transfer in Iridium pyridinium Complexes. J. Phys. Chem. 1994, 98, 5176–5179. [Google Scholar] [CrossRef][Green Version]
- Koren, K.; Borisov, S.M.; Saf, R.; Klimant, I. Strongly Phosphorescent Iridium (III)–Porphyrins—New Oxygen Indicators with Tuneable Photophysical Properties and Functionalities. Eur. J. Inorg. Chem. 2011, 10, 1531–1534. [Google Scholar] [CrossRef]
- Dragonetti, C.; Valore, A.; Colombo, A.; Righetto, S.; Trifiletti, V. Simple novel cyclometallated iridium complexes for potential application in dye-sensitized solar cells. Inorg. Chim. Acta 2012, 388, 163–167. [Google Scholar] [CrossRef]
- Liu, Y.N.; Wang, S.F.; Tao, Y.T.; Huang, W. Heavy metal complex containing organic/polymer materials for bulk-heterojunction photovoltaic devices. Chin. Chem. Lett. 2016, 27, 1250–1258. [Google Scholar] [CrossRef]
- Minaev, B.; Li, X.; Ning, Z.; Tian, H.; Ågren, H. Organometallic Materials for Electroluminescent and Photovoltaic Devices. In Organic Light Emitting Diode—Material, Process and Devices; Ko, S.H., Ed.; InTech: Goyang, Korea, 2011; pp. 61–100. [Google Scholar]
- Wong, W.Y. Challenges in organometallic research—Great opportunity for solar cells and OLEDs. J. Organomet. Chem. 2009, 694, 2644–2647. [Google Scholar] [CrossRef]
- Baranoff, E.; Yum, J.H.; Graetzel, M.; Nazeeruddin, M.K. Cyclometallated iridium complexes for conversion of light into electricity and electricity into light. J. Organomet. Chem. 2009, 694, 2661–2670. [Google Scholar] [CrossRef]
- Tao, Y.; Wu, Q.; Cheng, Y.; Xue, Z.; Gao, X.; Wang, M.; Yuan, W.; Hüttner, S.; Wan, S.; Cao, X.; et al. Cyclometalating organic ligand with an Iridium center toward dramatically improved photovoltaic performance in organic solar cells. Chem. Commun. 2019, 55, 2640–2643. [Google Scholar]
- Kapturkiewicz, A. Cyclometalated iridium (III) chelates—A new exceptional class of the electrochemiluminescent luminophores. Anal. Bioanal. Chem. 2016, 408, 7013–7033. [Google Scholar] [CrossRef]
- Kajjam, A.B.; Vaidyanathan, S. Structural Mimics of Phenyl Pyridine (ppy)—Substituted, Phosphorescent Cyclometalated Homo and Heteroleptic Iridium (III) Complexes for Organic Light Emitting Diodes—An Overview. Chem. Rec. 2018, 18, 293–349. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Li, Y.; Wang, X.; Wang, L.; Zhang, J. The influence of different cyclometalated ligand substituents and ancillary ligand on the phosphorescent properties of iridium (III) complexes. Mater. Chem. Phys. 2016, 177, 179–189. [Google Scholar] [CrossRef]
- Sun, J.; Wang, H.; Xu, H.; Li, J.; Wu, Y.; Du, X.; Xu, B. Synthesis, structure, photophysical and electro-luminescent properties of a blue-green self-host phosphorescent iridium (III) complex. Mater. Chem. Phys. 2015, 162, 392–399. [Google Scholar] [CrossRef]
- Chen, L.; Ma, Z.; Ding, J.; Wang, L.; Jing, X.; Wang, F. Effect of dendron generation on properties of self-host heteroleptic green light-emitting iridium dendrimers. Org. Electron. 2012, 13, 2160–2166. [Google Scholar] [CrossRef]
- Lai, P.N.; Brysacz, C.H.; Alam, M.K.; Ayoub, N.A.; Gray, T.G.; Bao, J.; Teets, T.S. Highly Efficient Red-Emitting Bis-Cyclometalated Iridium Complexes. J. Am. Chem. Soc. 2018, 140, 10198–10207. [Google Scholar] [CrossRef]
- Lin, S.H. Rate of Interconversion of Electronic and Vibrational Energy. J. Chem. Phys. 1966, 44, 3759–3767. [Google Scholar] [CrossRef]
- Bixon, M.; Jortner, J. Intramolecular Radiationless Transitions. J. Chem. Phys. 1968, 48, 715–726. [Google Scholar] [CrossRef]
- Englman, R.; Jortner, J. The energy gap law for radiationless transitions in large molecules. J. Mol. Phys. 1970, 18, 145–164. [Google Scholar] [CrossRef]
- Phillips, K.A.; Stonelake, T.M.; Horton, P.N.; Coles, S.J.; Hallett, A.J.; O’Kell, S.P.; Beames, J.M.; Pope, S.J.A. Dual visible/NIR emission from organometallic iridium(III) complexes. J. Organomet. Chem. 2019, 893, 11–20. [Google Scholar] [CrossRef]
- Yang, H.S.; Guo, H.Y.; Wu, L.S. Green phosphorescent organic light-emitting devices based on different electron transport layers combining with fluorescent sub-monolayer. Optoelectron. Lett. 2017, 13, 116–119. [Google Scholar] [CrossRef]
- Polosan, S.; Matei, E.; Ciobotaru, I.C.; Ciobotaru, C.C. Templateless electrodeposition ZnO nanowires for charge transport optimization in OLED structures. Mater. Res. Express. 2016, 3, 105018. [Google Scholar] [CrossRef]
- Ciobotaru, C.C.; Ciobotaru, I.C.; Schinteie, G.; Negrea, R.; Polosan, S. Enhancement of the electroluminescence of organic light emitting devices based on Ir(ppy)3 by doping with metallic and magnetic nanoparticles. Mater. Sci. Semicond. Process. 2017, 72, 78–84. [Google Scholar] [CrossRef]
- Wang, H.Y.; Jing, L.X.; Wang, H.Q.; Ye, J.T.; Qiu, Y.Q. Improving the NLO response of bis-cyclometalated iridium (III) complexes by modifying ligands: A DFT study. J. Organomet. Chem. 2018, 869, 18–25. [Google Scholar] [CrossRef]
- Gonzalez-Vazquez, J.P.; Burn, P.L.; Powell, B.J. Interplay of Zero-Field Splitting and Excited State Geometry Relaxation in fac-Ir(ppy)3. Inorg. Chem. 2015, 54, 10457–10461. [Google Scholar] [CrossRef] [PubMed]
- Baldoni, M.; Lorenzoni, A.; Pecchia, A.; Mercuri, F. Spatial and orientational dependence of electron transfer parameters in aggregates of iridium containing host materials for OLEDs: Coupling constrained density functional theory with molecular dynamics. Phys. Chem. Chem. Phys. 2018, 20, 28393–28399. [Google Scholar] [CrossRef]
- Polosan, S.; Ciobotaru, C.C.; Ciobotaru, I.C.; Tsuboi, T. Crystallization properties of IrQ(ppy)2 organometallic complex films. J. Mater. Res. 2017, 32, 1735–1740. [Google Scholar] [CrossRef]
- Xie, L.S.; Alexandrov, E.V.; Skorupskii, G.; Proserpio, D.M.; Dinca, M. Diverse p–p stacking motifs modulate electrical conductivity in tetrathiafulvalene-based metal–organic frameworks. Chem. Sci. 2019, 10, 8558–8565. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Zhong, G.Y.; Cao, X.A. Concentration quenching of electroluminescence in neat Ir(ppy)3 organic light-emitting diodes. J. Appl. Phys. 2010, 108, 083107. [Google Scholar] [CrossRef]
- Polosan, S.; Ciobotaru, I.C.; Enculescu, I.; Ciobotaru, C.C. Structural characteristics of iridium dual-emitter organometallic compound. J. Mater. Res. 2014, 29, 2898–2904. [Google Scholar] [CrossRef]
- Polosan, S.; Ciobotaru, I.C.; Tsuboi, T. Absorption, phosphorescence and Raman spectra of IrQ(ppy)(2) organometallic compound. Mater. Chem. Phys. 2015, 162, 822–830. [Google Scholar] [CrossRef]
- Eslamian, M. Inorganic and Organic Solution-Processed Thin Film Devices. Nano-Micro Lett. 2017, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Polosan, S.; Ciobotaru, I.C. Electroluminescence of OLED based IrQ(ppy)2-5Cl organometallic compounds: Theoretical considerations. J. Optoelectron. Adv. Mater. 2014, 16, 87–92. [Google Scholar]
- Nonoyama, M.; Yamasaki, K. Rhodium (III) complexes of benzo[h]quinoline and 2-phenylpyridine. Inorg. Nucl. Chem. Lett. 1971, 7, 943–946. [Google Scholar] [CrossRef]
- Nonoyama, M. Benzo[h]quinolin-10-yl-N Iridium (III) Complexes. Bull. Chem. Soc. Jpn. 1974, 47, 767–768. [Google Scholar] [CrossRef]
- Nonoyama, M. Chelating C-metallation of some organic nitrogen compounds with hexachlorotetrakis (tri-n-butylphosphine)-dirhodium (II). J. Organomet. Chem. 1975, 92, 89–95. [Google Scholar] [CrossRef]
- Nonoyama, M. Synthesis of several bis (benzo[h]quinolin-10-yl-n) rhodium (III) complexes. J. Organomet. Chem. 1974, 82, 271–276. [Google Scholar] [CrossRef]
- King, K.A.; Spellane, P.J.; Watts, R.J. Excited-State Properties of a Triply Ortho-Metalated Iridium (III) Complex. J. Am. Chem. Soc. 1985, 107, 1431–1432. [Google Scholar] [CrossRef]
- Maity, A.; Sinha, D.; Rajak, K.K. Experimental and theoretical studies of structural and photophysical properties of a novel heteroleptic cyclometalated iridium (III) complex with 8-hydroxyquinoline-phenylazo ligand. J. Mol. Struct. 2018, 1158, 122–132. [Google Scholar] [CrossRef]
- Huang, D.F.; Chow, T.J.; Wu, C.Y.; Sun, S.S.; Tsai, S.H.; Wen, Y.S.; Polosan, S.; Tsuboi, T. The preparation of (8-hydroxyquinolinato)bis(2-phenylpyridil)iridium complexes and their photophysical properties. J. Chin. Chem. Soc. 2008, 55, 439–448. [Google Scholar] [CrossRef]
- Ma, D.; Hummelgen, I.A.; Jing, X.; Hong, Z.; Wang, L.; Zhao, X.; Wang, F.; Karasz, F.E. Charge transport in a blue-emitting alternating block copolymer with a small spacer to conjugated segment length ratio. J. Appl. Phys. 2000, 87, 312. [Google Scholar] [CrossRef]
- Ma, D.; Hummelgen, I.A.; Rosamaria, W.; Li, C.; Gruber, J. Charge injection and transport in poly (4, 4′-biphenylenevinylene). J. Phys. D Appl. Phys. 2000, 33, 1376–1379. [Google Scholar] [CrossRef]
- Juhasz, P.; Nevrela, J.; Micjan, M.; Novota, M.; Uhrik, J.; Stuchlikova, L.; Jakabovic, J.; Harmatha, L.; Weis, M. Charge injection and transport properties of an organic light-emitting diode. Beilstein J. Nanotechnol. 2016, 7, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lu, Z.; Peng, Y.; Liu, Y.; Yang, Y. Effect of energy transfer on electroluminescent performance in blend-layer organic light-emitting devices. J. Lum 2008, 128, 1523–1527. [Google Scholar] [CrossRef]
- Ohmori, Y.; Hirotake, K.; Sawatani, T.; Ueta, H.; Yoshino, K. Enhancement of electroluminescence utilizing confined energy transfer for red light emission. Thin Solid Films 2001, 393, 407–411. [Google Scholar] [CrossRef]
- Kasha, M. Characterization of electronic transitions in complex molecules. Discuss. Faraday Soc. 1950, 9, 14–19. [Google Scholar] [CrossRef]
- Turro, N.J.; Ramamurthy, V.; Cherry, W.; Farneth, W. The effect of wavelength on organic photoreactions in solution. Reactions from upper excited states. Chem. Rev. 1978, 78, 125–145. [Google Scholar] [CrossRef]
- Tsuboi, T.; Polosan, S.; Hang, D.F.; Chow, T.J. Optical characteristics of organic light emitting diode with IrQ(ppy)2-5Cl and its emitter. Thin Solid Films 2008, 516, 2788–2793. [Google Scholar] [CrossRef]
- Sun, Y.; Yang, X.; Liu, B.; Dang, J.; Li, Y.; Zhou, G.; Wu, Z.; Wong, W.Y. Towards high performance solution-processed orange organic light-emitting devices: Precisely adjusting properties of Ir (III) complexes by reasonably engineering the asymmetric configuration with second functionalized cyclometalating ligands. J. Mater. Chem. C 2019, 7, 8836–8846. [Google Scholar] [CrossRef]
- Wu, Y.; Li, X.; Zhao, H.; Li, J.; Miao, Y.; Wang, H.; Zhu, F.; Xu, B. Pyrene-based hyperbranched porous polymers with doped Ir(piq)2(acac) red emitter for highly efficient white polymer light-emitting diodes. Org. Electron. 2020, 76, 105487. [Google Scholar] [CrossRef]
- Park, Y.S.; Lee, S.; Kim, K.H.; Kim, S.Y.; Wan Lee, J.H.; Kim, J.J. Exciplex-Forming Co-host for Organic Light-Emitting Diodes with Ultimate Efficiency. Adv. Funct. Mater. 2013, 23, 4914–4920. [Google Scholar] [CrossRef]
- Ciobotaru, C.C.; Polosan, S.; Ciobotaru, C.I. Electroluminescence Properties of IrQ(ppy)2 Dual-Emitter. Organometallic Compound in Organic Light-Emitting Devices. J. Electron. Mater. 2018, 47, 1490–1496. [Google Scholar] [CrossRef]


| IrQ(ppy)2 | Electroluminescence at 14 V (cd/m2) | Current–Voltage at 14 V (mA/cm2) | PE (lm/W) | CE (cd/A) |
|---|---|---|---|---|
| 10% in CBP | 19,200 | 172 | 2.5 | 11.2 |
| 20% in CBP | 3680 | 62.7 | 1.3 | 5.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polosan, S.; Ciobotaru, I.C.; Ciobotaru, C.C. Organometallic Coatings for Electroluminescence Applications. Coatings 2020, 10, 277. https://doi.org/10.3390/coatings10030277
Polosan S, Ciobotaru IC, Ciobotaru CC. Organometallic Coatings for Electroluminescence Applications. Coatings. 2020; 10(3):277. https://doi.org/10.3390/coatings10030277
Chicago/Turabian StylePolosan, Silviu, Iulia Corina Ciobotaru, and Claudiu Constantin Ciobotaru. 2020. "Organometallic Coatings for Electroluminescence Applications" Coatings 10, no. 3: 277. https://doi.org/10.3390/coatings10030277
APA StylePolosan, S., Ciobotaru, I. C., & Ciobotaru, C. C. (2020). Organometallic Coatings for Electroluminescence Applications. Coatings, 10(3), 277. https://doi.org/10.3390/coatings10030277






