Comparative Study Between Ammonium Phosphate and Ethyl Silicate Towards Conservation of Prehistoric Paintings in the Magura Cave (Bulgaria)
Abstract
1. Introduction
- (1)
- the presence of the guano layer creating the paintings. The case of the Magura cave is different from the case of wall paintings [9] and also from the case of other prehistoric paintings, such as those in the Altamira [10] and Lascaux [11] caves. In the wall paintings, the pigments were dispersed in limewater and then applied directly onto the wet plaster (in the so-called “fresco” technique) and/or they were applied onto the dried plaster by using an organic binder, such as egg, milk, or vegetal extractives [9]. In the case of other prehistoric paintings, the pigments, usually red (Fe2O3), black (charcoal, soot, bone charcoal, or Mn oxides), and more rarely yellow (FeOOH) and brown (ochres), were mixed with an extender and/or a binder [10,11]. The case of the Magura cave, with paintings laid on the cave walls by bat guano having a plastic consistency similar to clay [5], is substantially different from the situations discussed above, with only a few similar cases, such as the Baylovo Cave in Bulgaria and the “Grotta dei Cervi” (Deer cave) in Italy [7]. To our knowledge, no case of consolidation of cave paintings made of bat guano has been reported in the scientific literature.
- (2)
- the presence of soluble salts inside the stone pores. This is an issue for two reasons: (i) salts already present inside the pores may lead to failure of the consolidation intervention, if the tip of the cracks among grains is filled with salts, so that no effective sealing of the crack tip can be achieved by the consolidant [12]. To prevent this, it is usually recommended that salts be extracted by poulticing before consolidants are applied, but this is useless in the present case, as salts are continuously supplied from the ground; (ii) salt crystallization cycles taking place after stone consolidation may lead to stress at the grain boundaries, with possible failure of the consolidation intervention if the stone tensile strength is exceeded. Experiences of application of various consolidants onto salt-laden stones, possibly later subjected to further salt crystallization cycles, have been reported in the literature, as detailed in the following.
- (3)
- the environmental conditions in the cave. Because of its hypogeous nature, the temperature in the cave ranges between 12 and 25 °C, while the relative humidity (RH) is constantly in the range 65–90%. Such environmental conditions may have an impact on a consolidant performance, as discussed below.
- (1)
- To evaluate the effects of the two consolidants not only on limestone but also on the cave paintings, two types of laboratory specimens were considered: (i) specimens made of a limestone similar to that in the cave and (ii) specimens made of the same limestone with a layer of sterilized bat guano deposited on top, to resemble the prehistoric paintings in the cave. The procedure for the deposition of the guano layer, described in detail in Section 2.2, was developed in such a way to likely resemble the process of creation of the paintings, even though, of course, freshly prepared samples could not be identical to cave paintings that have undergone about 5500 years of fossilization.
- (2)
- To evaluate the effects of the two consolidants onto salt-laden samples, to be further subjected to salt crystallization cycles after consolidation, would have been ideal. However, this would have made the evaluation of the consolidants on the two types of specimens (stone and stone+guano) even more difficult, with the risk that the influence of salts in the pore and the guano layer on the stone surface could be hard to distinguish. For this reason, at the present stage of the research, the consolidants were applied onto uncontaminated specimens, which were used to evaluate the effectiveness and the compatibility of the treatments in these simplified conditions. Application onto salt-laden samples, together with the evaluation of the durability of the consolidated substrates, was left to a future stage of the research, once the performance on this simplified system has been assessed.
- (3)
- At this stage of the research, the effects of the treatments were evaluated in laboratory conditions (T = 19–23 °C, RH = 45–55%), which were regarded as conservative with respect to the environmental conditions in the cave (T = 12–25 °C, RH = 65–90%). In fact, the higher relative humidity in the cave was expected to positively influence the rate of hydrolysis-condensation reactions of ethyl silicate (while no effect was expected on the reaction of the ammonium phosphate treatment). Nonetheless, to obtain some preliminary information on the performance of two consolidants in the real environmental conditions, specimens treated with the two consolidants were exposed in the Magura cave for 9 months, as described in detail in the following.
2. Materials and Methods
2.1. Stone Specimens
2.2. Stone+Guano Specimens
2.3. Consolidants
2.3.1. Ethyl Silicate-Based Consolidant
2.3.2. Phosphate Consolidant
2.4. Pre-treatment by Plasma
2.5. Sample Characterization
2.5.1. Morphology of the New Phases
2.5.2. Composition of the New Phases
2.5.3. Penetration Depth
2.5.4. Dynamic Elastic Modulus
2.5.5. Resistance to Abrasion
2.5.6. Color Change
2.5.7. Pore Size Distribution
2.5.8. Static Contact Angle
2.5.9. Water Sorptivity and Water Absorption
2.5.10. Water Vapor Permeability
2.5.11. Field Exposure
3. Results and Discussion
3.1. Morphology of the New Phases
3.2. Composition of the New Phases
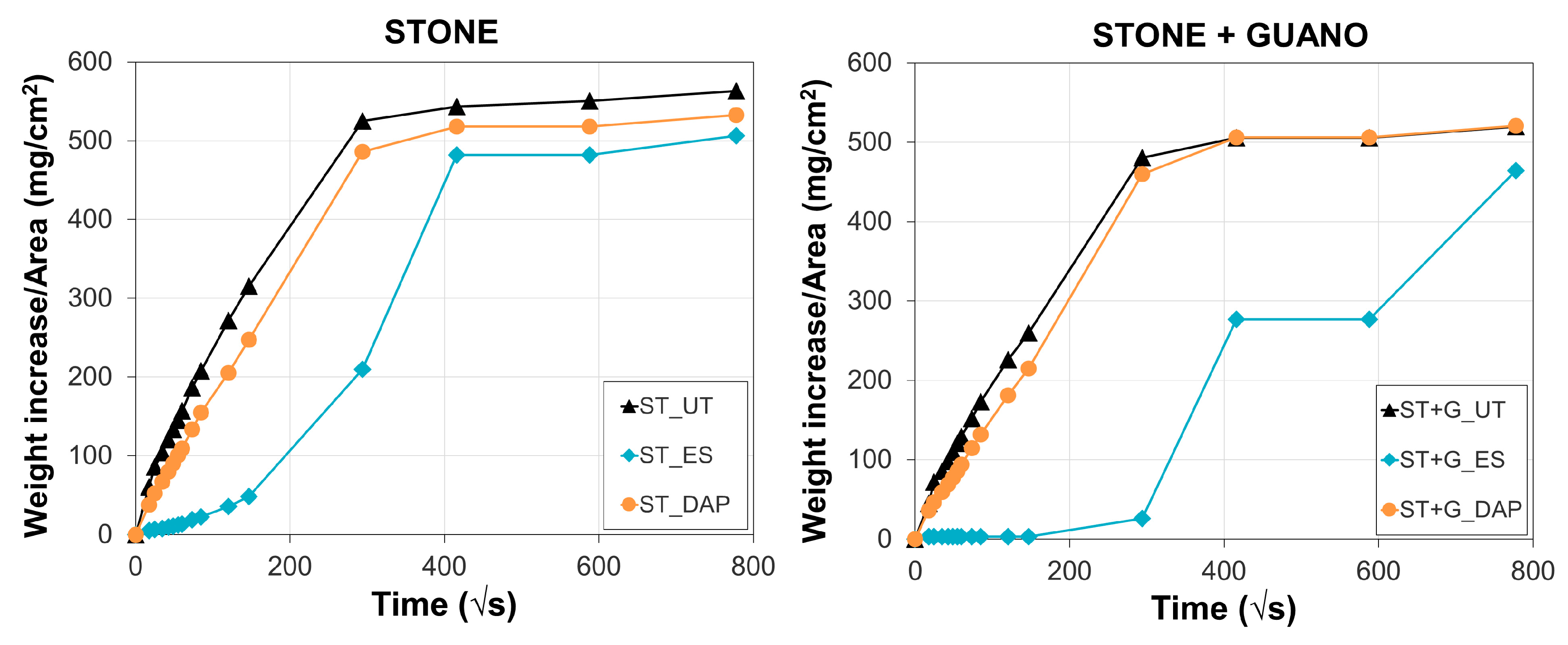
3.3. Penetration Depth
3.4. Dynamic Elastic Modulus
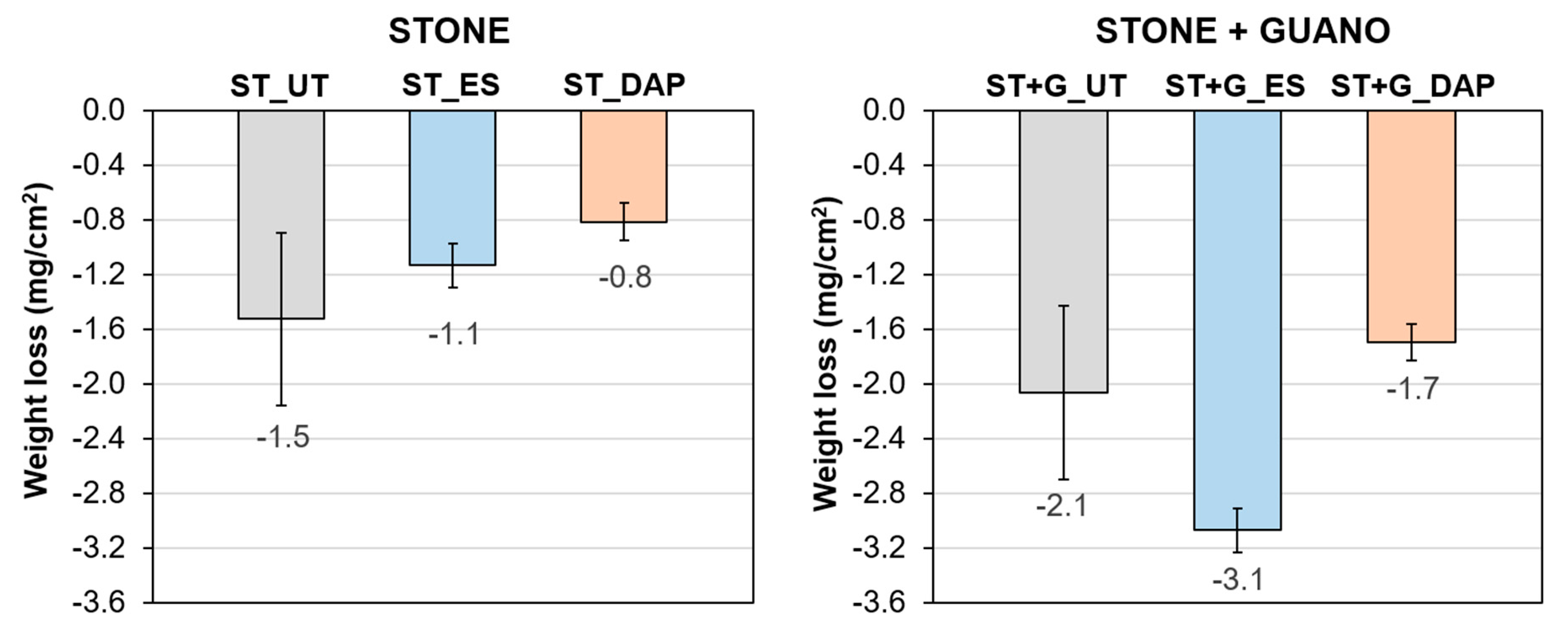
3.5. Resistance to Abrasion
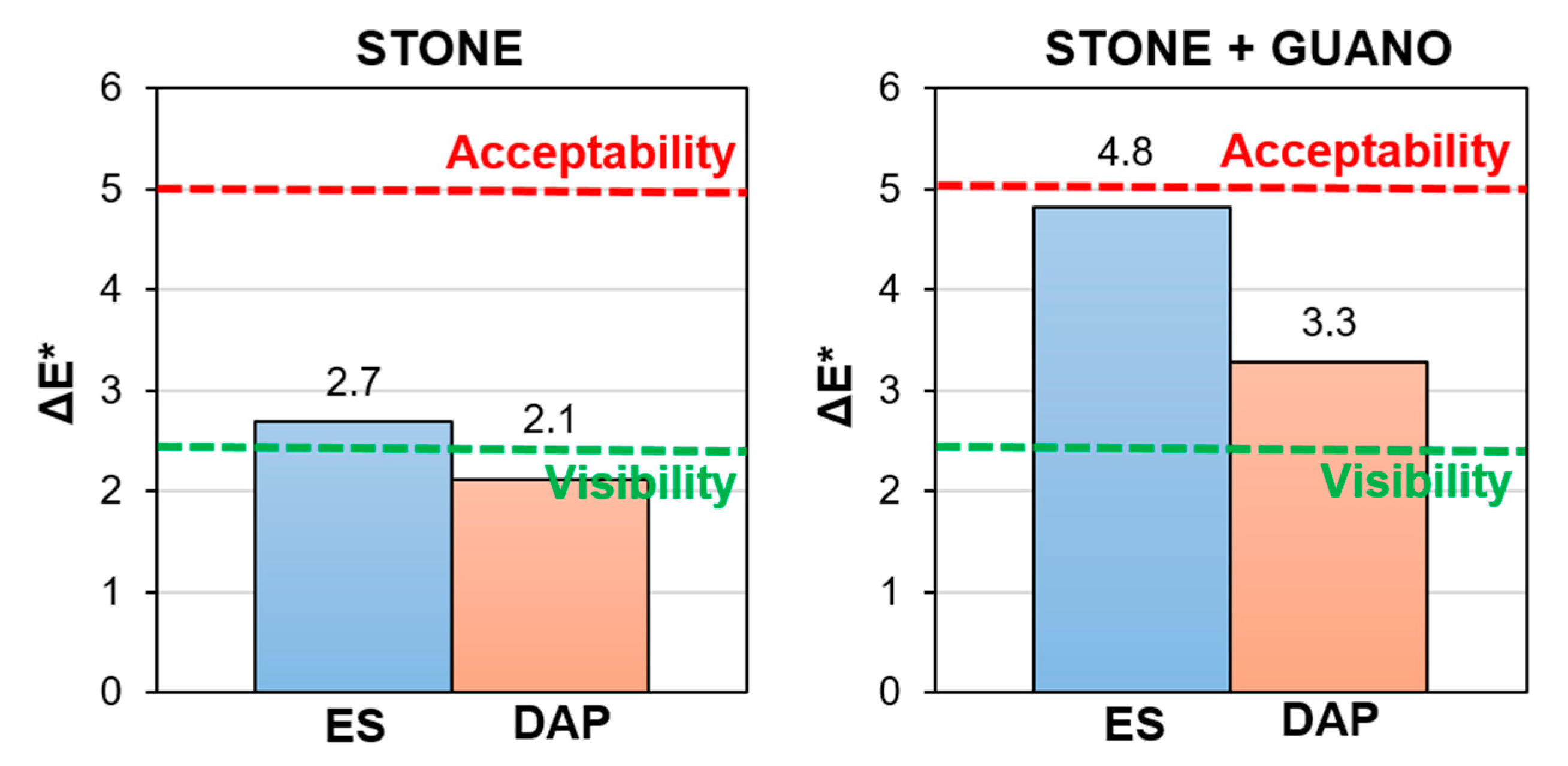
3.6. Color Change
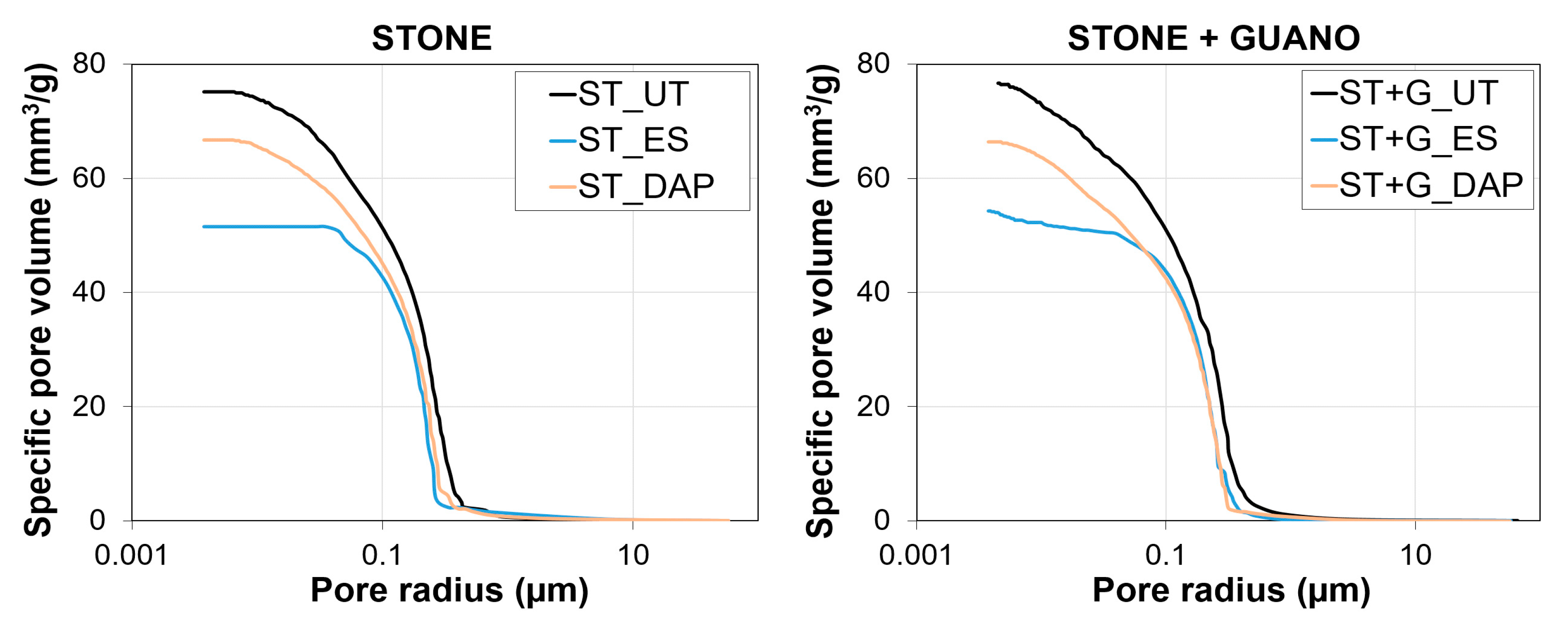
3.7. Pore Size Distribution
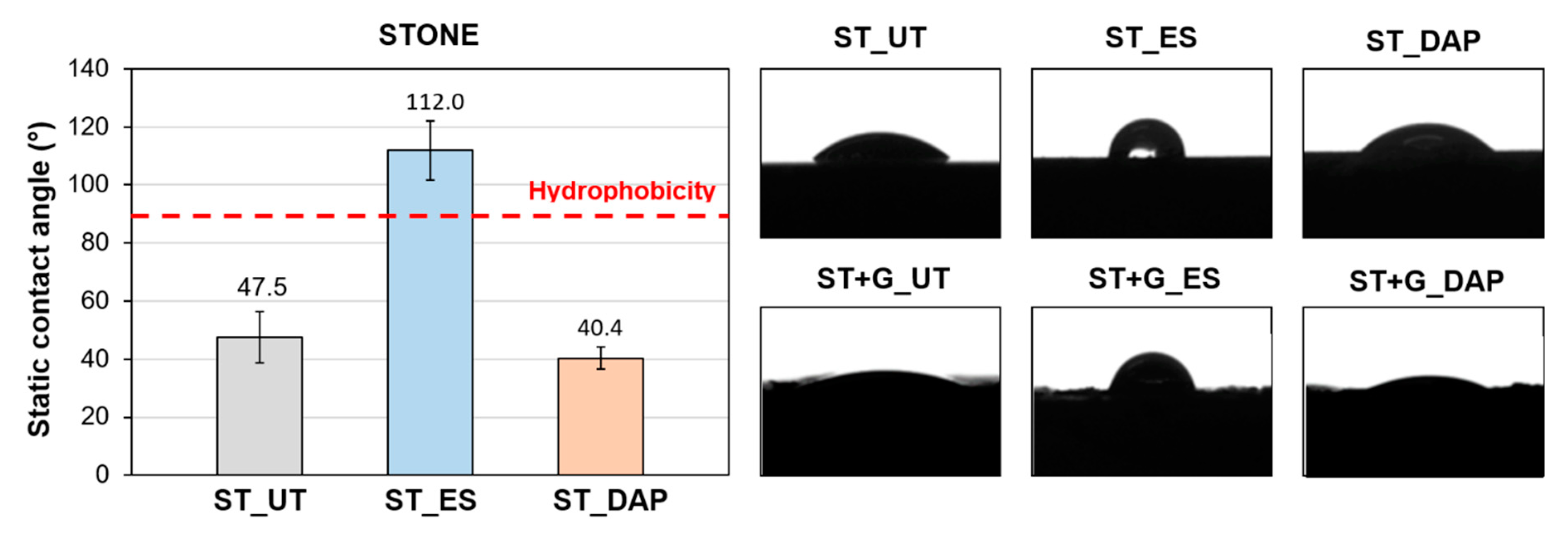
3.8. Static Contact Angle
3.9. Water Sorptivity and Water Absorption
3.10. Water Vapor Permeability
3.11. Field Exposure
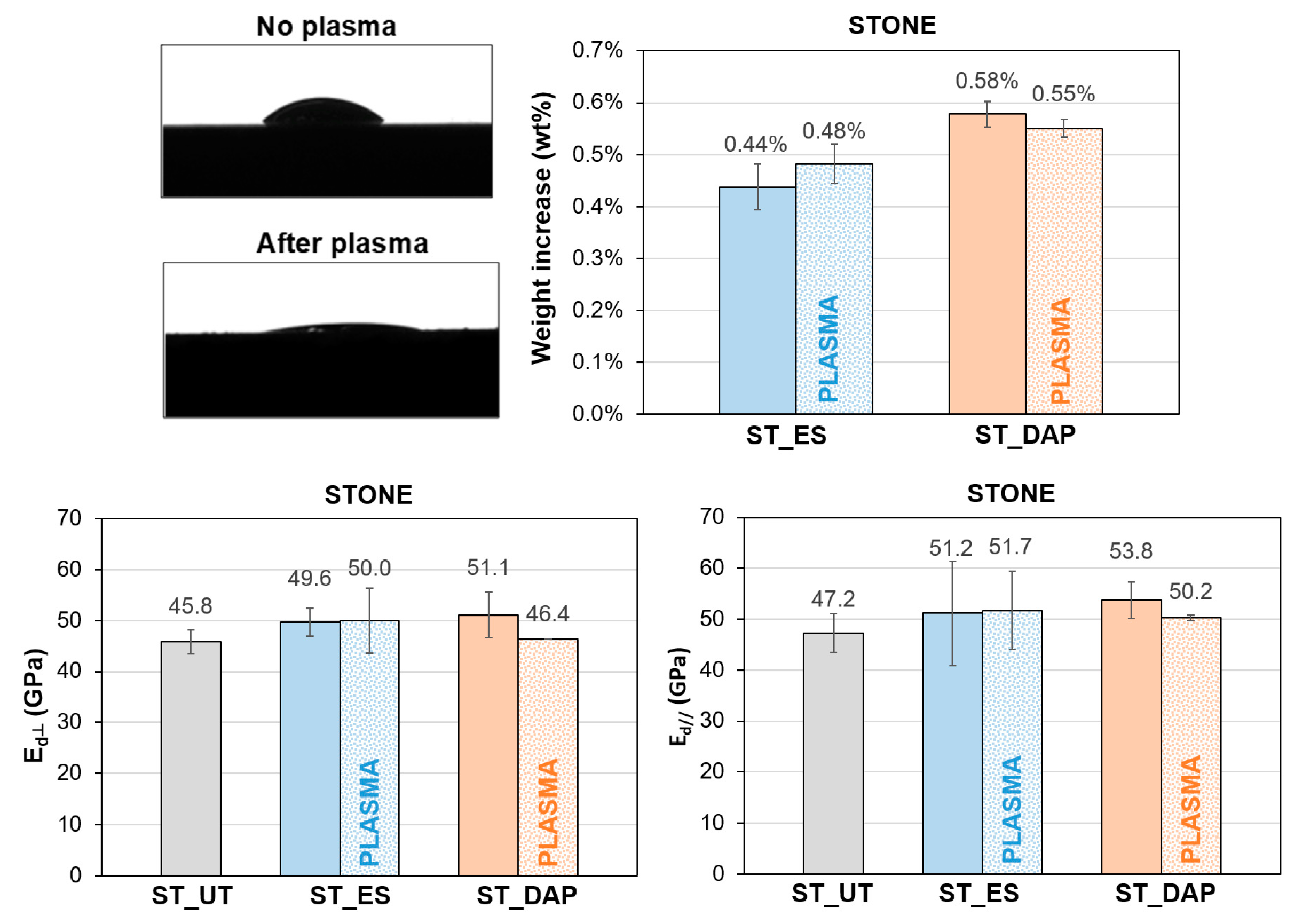
3.12. Pre-treatment by Plasma
4. Conclusions
- In the case of the stone specimens, both the treatment based on ethyl silicate (“ES”) and that based on ammonium phosphate (“DAP”) were able to increase stone mechanical properties, the increase being higher for “DAP”. Both treatments caused acceptable color changes, while a significant difference was registered in terms of alterations in water and water vapor transport properties. “DAP” left the stone hydrophilic, with negligible alterations in static contact angle and water sorptivity, while “ES” made the stone hydrophobic, even 8 weeks after the consolidant application. Moreover, “DAP” also left the water vapor permeability basically unaltered, while a reduction in permeability was caused by “ES”. After exposure in the cave for 9 months, none of the treated specimens had shown evident signs of biodeterioration or pulverization, which could be regarded as an encouraging result, even though long term data are necessary to conclusively ascertain the treatment durability.
- In the case of the stone+guano specimens, the presence of the guano layer affected the penetration of the two consolidants into the stone, thus partly reducing the consolidating effectiveness. A more intense color change was registered, compared to the stone samples, alongside cracking of the guano layer. Although more difficult to quantify, the general trend of modification in water transport properties was confirmed: “DAP” caused minor alterations, while “ES” made the stone+guano specimens hydrophobic. It was not possible to reliably determine the water vapor permeability of the stone+guano specimens by the adopted methodology, but results substantially similar to those of the stone specimens were expected (similarly to the case of the water transport properties).
- Regarding the possible benefit deriving from pre-treatment by non-thermal plasma before consolidant application, a significant decrease in the static contact angle of the stone surface was found after plasma treatment. This might be useful for the application of conservation treatments that are intended to remain mostly on the stone surface (e.g., protective treatments). Nonetheless, no significant increase in the amount of retained product or mechanical consolidation was found when pre-treatment by plasma was performed because the wettability of the stone surface was conveniently modified, but that of the pore walls remained unaltered.
- the presence of the surface guano layer: an improved methodology of guano application onto the limestone substrate needs to be developed. In fact, the methodology adopted in this study produced specimens very prone to pulverization and detachment of the guano layer, differently from the real guano paintings in the Magura cave. To reproduce the paintings in a manner that fully resembles the condition of the real ones is extremely difficult, as the prehistoric paintings have undergone millennials of fossilization. A possible strategy to improve the reliability of the stone+guano specimens produced in the laboratory may be to subject the stone+guano specimens to accelerated aging, aimed at mimicking the natural fossilization process.
- the presence of salts in the substrate: after evaluating the effects of the two consolidants onto uncontaminated specimens, their effects onto specimens containing salts in the pores need to be evaluated, as this would be the condition in the cave (complete salts removal before the consolidant application is not feasible). To do so, the amount and nature of the salts present in the limestone cave need to be specifically investigated and characterized, so that laboratory specimens can be prepared accordingly. Then, after preliminary salt contamination and consolidation, the durability of the treated specimens to further salt weathering should be evaluated by accelerated salt crystallization cycles. This should be carried out in the current environmental conditions of the cave or, in case it is possible to control them, in the adjusted environmental conditions that would be adopted for cave conservation.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ivanova, S.; Gurova, M.; Spassov, N.; Hristova, L.; Tzankov, N.; Popov, V.; Marinova, E.; Makedonska, J.; Smith, V.; Ottoni, C.; et al. Magura Cave, Bulgaria: A multidisciplinary study of Late Pleistocene human palaeoenvironment in the Balkans. Quatern Int. 2016, 415, 86–108. [Google Scholar] [CrossRef]
- Stoytchev, T. Eneolithic paintings from Magoura cave, Bulgaria. Annu. Dep. Archaeol. NBU 1994, 1, 307–320. (In Bulgarian) [Google Scholar]
- Stoytchev, T. Rock Art: General Classification; Agato Press: Sofia, Bulgaria, 2005; p. 24. (In Bulgarian) [Google Scholar]
- Nougier, R. Les “ballerines” de Magura. Préhistoire Ariégeoise 1977, XXXII, 123–131. [Google Scholar]
- Kunov, A.; Arnaudov, V.; Molar, M. First 14C dating of the bat guano used for the ancient drawings of Magura cave, NW Bulgaria. In Proceedings of the Bulgarian Geological Society, National Conference “GEOSCIENCES 2014”, Sofia, Bulgaria, 10–11 December 2014. [Google Scholar]
- Illustrated Glossary on Stone Deterioration Patterns = Glossaire Illustré sur les Forms D’altération de la Pierre, 1st ed.; Vergès-Belmin, V., Ed.; English-French ed.; Monuments & Sites no. 15; ICOMOS and (ISCS) International Scientific Committee for Stone: Paris, France, 2008. [Google Scholar]
- Mitova, M.M.; Iliev, M.; Nováková, A.; Gorbushina, A.A.; Groudeva, V.I.; Martin-Sanchez, P.M. Diversity and biocide susceptibility of fungal assemblages dwelling in the Art Gallery of Magura Cave. Bulg. Int. J. Speleol. 2017, 46, 67–80. [Google Scholar] [CrossRef]
- Stefanova, M.; Kamenarov, Z.; Sassoni, E.; Franzoni, E.; Ripà, M.; Patelli, A.; Scopece, P.; Verga Falzacappa, E.; Sakaj, M. Innovative solutions for prehistoric paintings -atmospheric pressure plasma and phosphate consolidant for the preservation of the Magura Cave (Bulgaria) 13–15 May 2020. Florence (IT) Proc. Heri-Tech 2020, in press. [Google Scholar]
- Chelazzi, D.; Poggi, P.; Jaidar, Y.; Toccafondi, N.; Giorgi, R.; Baglioni, P. Hydroxide nanoparticles for cultural heritage: Consolidation and protection of wall paintings and carbonate materials. J. Colloid Interface Sci. 2013, 392, 42–49. [Google Scholar] [CrossRef]
- Valladas, H.; Cachier, H.; Maurice, P.; Bernaldo de Quirost, F.; Clottes, J.; Cabrera Valdes, V.; Uzquiano, P.; Arnold, M. Direct radiocarbon dates for prehistoric paintings at the Altamira, EI Castillo and Niaux caves. Nature 1992, 357, 68–70. [Google Scholar] [CrossRef]
- Chalmin, E.; Vignaud, C.; Menu, M. Palaeolithic painting matter: Natural or heat-treated pigment? Appl. Phys. A 2004, 79, 187–191. [Google Scholar] [CrossRef]
- Scherer, G.W.; Wheeler, G.S. Silicate consolidants for stone. Key Eng. Mater. 2009, 391, 1–25. [Google Scholar] [CrossRef]
- Baglioni, P.; Carretti, E.; Chelazzi, D. Nanomaterials in art conservation. Nat. Nanotechnol. 2015, 10, 287–290. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, C.; Ruiz-Agudo, E. Nanolimes: From synthesis to application. Pure Appl. Chem. 2018, 90, 523–550. [Google Scholar] [CrossRef]
- Ambrosi, M.; Dei, L.; Giorgi, R.; Neto, C.; Baglioni, P. Colloidal Particles of Ca(OH)2: Properties and Applications to Restoration of Frescoes. Langmuir 2001, 17, 4251–4255. [Google Scholar] [CrossRef]
- Giorgi, R.; Ambrosi, M.; Toccafondi, T.; Baglioni, P. Nanoparticles for Cultural Heritage Conservation: Calcium and Barium Hydroxide Nanoparticles for Wall Painting Consolidation. Chem. Eur. J. 2010, 16, 9374–9382. [Google Scholar] [CrossRef] [PubMed]
- Ruffolo, S.A.; La Russa, M.F.; Aloise, P.; Belfiore, C.M.; Macchia, A.; Pezzino, A.; Crisci, G.M. Efficacy of nanolime in restoration procedures of salt weathered limestone rock. Appl. Phys. A 2014, 114, 753–758. [Google Scholar] [CrossRef]
- Ruffolo, S.A.; La Russa, M.F.; Ricca, M.; Belfiore, C.M.; Macchia, A.; Comite, V.; Pezzino, A.; Crisci, G.M. New insights on the consolidation of salt weathered limestone: The case study of Modica stone. Bull. Eng. Geol. Environ. 2017, 76, 11–20. [Google Scholar] [CrossRef]
- Matteini, M. Inorganic treatments for the consolidation and protection of stone artefacts. Conserv. Sci. Cult. Herit. 2008, 8, 13–27. [Google Scholar]
- Pinna, D.; Salvadori, B.; Porcinai, S. Evaluation of the application conditions of artificial protection treatments on salt-laden limestones and marble. Construct. Build. Mater. 2011, 25, 2723–2732. [Google Scholar] [CrossRef]
- Dreyfuss, T. Interactions on site between powdering porous limestone, natural salt mixtures and applied ammonium oxalate. Herit. Sci. 2019, 7, 5. [Google Scholar] [CrossRef]
- Sassoni, E.; Naidu, S.; Scherer, G.W. The use of hydroxyapatite as a new inorganic consolidant for damaged carbonate stones. J. Cult. Herit. 2011, 12, 346–355. [Google Scholar] [CrossRef]
- Naidu, S.; Sassoni, E.; Scherer, G.W. New treatment for corrosion-resistant coatings for marble and consolidation of limestone. In Jardins de Pierres—Conservation of Stone in Parks, Gardens and Cemeteries; Stefanaggi, M., Vergès-Belmin, V., Eds.; XL Print: Paris, France, 2011; pp. 289–294. [Google Scholar]
- Dorozhkin, S.V. Calcium orthophosphates: Occurrence, properties, biomineralization, pathological calcification and biomimetic applications. Biomatter 2011, 1, 121–164. [Google Scholar] [CrossRef]
- Sassoni, E. Hydroxyapatite and Other Calcium Phosphates for the Conservation of Cultural Heritage: A Review. Materials 2018, 11, 557. [Google Scholar] [CrossRef] [PubMed]
- Matteini, M.; Rescic, S.; Fratini, F.; Botticelli, G. Ammonium phosphates as consolidating agents for carbonatic stone materials used in architecture and cultural heritage: Preliminary research. Int. J. Archit. Herit. 2011, 5, 717–736. [Google Scholar] [CrossRef]
- Yang, F.W.; Liu, Y.; Zhu, Y.C.; Long, S.J.; Zuo, G.F.; Wang, C.Q.; Guo, F.; Zhang, B.J.; Jiang, S.W. Conservation of weathered historic sandstone with biomimetic apatite. Chin. Sci. Bull. 2012, 57, 2171–2176. [Google Scholar] [CrossRef]
- Sassoni, E.; Franzoni, E.; Pigino, B.; Scherer, G.W.; Naidu, S. Consolidation of calcareous and siliceous sandstones by hydroxyapatite: Comparison with a TEOS-based consolidant. J. Cult. Herit. 2013, 14, e103–e108. [Google Scholar] [CrossRef]
- Graziani, G.; Sassoni, E.; Franzoni, E. Consolidation of porous carbonate stones by an innovative phosphate treatment: Mechanical strengthening and physical-microstructural compatibility in comparison with TEOS-based treatments. Herit. Sci. 2015, 3, 1–6. [Google Scholar] [CrossRef]
- Molina, E.; Rueda-Quero, L.; Benavente, D.; Burgos-Cara, A.; Ruiz-Agudo, E.; Cultrone, G. Gypsum crust as a source of calcium for the consolidation of carbonate stones using a calcium phosphate-based consolidant. Construct. Build. Mater. 2017, 143, 298–311. [Google Scholar] [CrossRef]
- Ma, X.; Balonis, M.; Pasco, H.; Toumazou, M.; Counts, D.; Kakoulli, I. Evaluation of hydroxyapatite effects for the consolidation of a Hellenistic-Roman rock-cut chamber tomb at Athienou-Malloura in Cyprus. Construct. Build. Mater. 2017, 150, 333–344. [Google Scholar] [CrossRef]
- Possenti, E.; Conti, C.; Gatta, G.D.; Realini, M.; Colombo, C. Diammonium hydrogen phosphate treatment on dolostone: The role of Mg in the crystallization process. Coatings 2019, 9, 169. [Google Scholar] [CrossRef]
- Balonis-Sant, M.; Ma, X.; Kakoulli, I. Preliminary results on biomimetic methods based on soluble ammonium phosphate precursors for the consolidation of archaeological wall paintings. In Archaeological Chemistry VIII, ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2013; Volume 1147, pp. 419–447. [Google Scholar] [CrossRef]
- Sassoni, E.F. Lime and cement mortar consolidation by ammonium phosphate. Construct. Build. Mater. 2020, 245, 118409. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Franzoni, E.; Scherer, G.W. Conversion of calcium sulfate dihydrate into calcium phosphates as a route for conservation of gypsum stuccoes and sulfated marble. Construct. Build. Mater. 2018, 170, 290–301. [Google Scholar] [CrossRef]
- Ma, X.; Pasco, H.; Balonis, M.; Kakoulli, I. Investigation of the optical, physical, and chemical interactions between diammonium hydrogen phosphate (DAP) and pigments. Sustainability 2019, 11, 3803. [Google Scholar] [CrossRef]
- Graziani, G.; Sassoni, E.; Scherer, G.W.; Franzoni, E. Phosphate-based treatments for consolidation of salt-bearing Globigerina limestone. IOP Conf. Ser. Mater. Sci. Eng. 2018, 364, 012082. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Franzoni, E. An innovative phosphate-based consolidant for limestone. Part 2: Durability in comparison with ethyl silicate. Construct. Build. Mater. 2016, 102, 931–942. [Google Scholar] [CrossRef]
- Molina, E.; Fiol, C.; Cultrone, G. Assessment of the efficacy of ethyl silicate and dibasic ammonium phosphate consolidants in improving the durability of two building sandstones from Andalusia (Spain). Environ. Earth Sci. 2018, 77, 302. [Google Scholar] [CrossRef]
- Shekofteh, A.; Molina, M.; Rueda-Quero, L.; Arizzi, A.; Cultrone, G. The efficiency of nanolime and dibasic ammonium phosphate in the consolidation of beige limestone from the Pasargadae World Heritage Site. Archaeol. Anthropol. Sci. 2019. [Google Scholar] [CrossRef]
- Wheeler, G. Alkoxysilanes and the Consolidation of Stone. In Research in Conservation; The Getty Conservation Institute: Los Angeles, CA, USA, 2005. [Google Scholar]
- Sassoni, E.; Graziani, G.; Franzoni, E. An innovative phosphate-based consolidant for limestone. Part 1: Effectiveness and compatibility in comparison with ethyl silicate. Construct. Build. Mater. 2016, 102, 918–930. [Google Scholar] [CrossRef]
- Maravelaki-Kalaitzaki, P.; Kallithrakas-Kontos, N.; Korakaki, D.; Agioutantis, Z.; Maurigiannakis, S. Evaluation of silicon-based strengthening agents on porous limestones. Prog. Org. Coat. 2006, 57, 140–148. [Google Scholar] [CrossRef]
- da Fonseca, B.S.; Piçarra, S.; Pinto, A.P.F.; Montemor, M.d. Polyethylene glycol oligomers as siloxane modificators in consolidation of carbonate stones. Pure Appl. Chem. 2016, 88, 1117–1128. [Google Scholar] [CrossRef]
- Burgos-Cara, A.; Rodríguez-Navarro, C.; Ortega-Huertas, M.; Ruiz-Agudo, E. Bioinspired Alkoxysilane Conservation Treatments for Building Materials Based on Amorphous Calcium Carbonate and Oxalate Nanoparticles. ACS Appl. Nano Mater. 2019, 2, 4954–4967. [Google Scholar] [CrossRef]
- Moropoulou, A.; Haralampopoulos, G.; Tsiourva, T.; Auger, F.; Birginie, M. Artificial weathering and non-destructive tests for the performance evaluation of consolidation materials applied on porous stones. Mater. Struct. 2003, 36, 210–217. [Google Scholar] [CrossRef]
- Franzoni, E.; Graziani, G.; Sassoni, E. TEOS-based treatments for stone consolidation: Acceleration of hydrolysis-condensation reactions by poulticing. J. Sol-Gel Sci. Tech. 2015, 74, 398–405. [Google Scholar] [CrossRef]
- Naidu, S.; Liu, C.; Scherer, G.W. Hydroxyapatite-based consolidant and the acceleration of hydrolysis of silicate-based consolidants. J. Cult. Herit. 2015, 16, 94–101. [Google Scholar] [CrossRef]
- Wurster, C.M.; Munksgaard, N.; Zwart, C.; Bird, M. The biogeochemistry of insectivorous cave guano: A case study from insular Southeast Asia. Biogeochemisty 2015, 124, 163–175. [Google Scholar] [CrossRef]
- Shahack-Gross, R.; Berna, F.; Karkanas, P.; Weiner, S. Bat guano and preservation of archaeological remains in cave sites. J. Archaeol. Sci. 2004, 31, 1259–1272. [Google Scholar] [CrossRef]
- Naidu, S.; Scherer, G.W. Nucleation, growth and evolution of calcium phosphate films on calcite. J. Colloid Interface Sci. 2014, 435, 128–137. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Franzoni, E.; Scherer, G.W. Calcium phosphate coatings for marble conservation: Influence of ethanol and isopropanol addition to the precipitation medium on the coating microstructure and performance. Corros. Sci. 2018, 136, 255–267. [Google Scholar] [CrossRef]
- Tendero, C.; Tixier, C.; Tristant, P.; Desmaison, J.; Leprince, P. Atmospheric pressure plasmas: A review. Spectrochim. Acta B 2006, 61, 2–30. [Google Scholar] [CrossRef]
- Patelli, A.; Verga, E.; Nodari, L.; Petrillo, S.M.; Delva, A.; Ugo, P.; Scopece, P. A customised atmospheric pressure plasma jet for conservation requirements. IOP Conf. Ser. Mater. Sci. Eng. 2018, 364, 012079. [Google Scholar] [CrossRef]
- Sandrolini, F.; Franzoni, E.; Cuppini, G. Predictive diagnostics for decayed ashlars substitution in architectural restoration in Malta. Mater. Eng. 2000, 11, 323–337. [Google Scholar]
- Conservation of Cultural Property -Test Methods -Determination of Water Absorption by Capillarity; European Standard EN 15801; European Standard EN: Brussels, Belgium, 2010.
- Conservation of Cultural Property -Test Methods -Determination of Water Vapor Permeability (δp); European Standard EN 15803; European Standard EN: Brussels, Belgium, 2010.
- Sandrolini, F.; Franzoni, E.; Sassoni, E.; Diotallevi, P.P. The contribution of urban-scale environmental monitoring to materials diagnostics: A study on the Cathedral of Modena (Italy). J. Cult. Herit. 2011, 12, 441–450. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Ridolfi, G.; Bignozzi, M.C.; Franzoni, E. Thermal behavior of Carrara marble after consolidation by ammonium phosphate, ammonium oxalate and ethyl silicate. Mater. Des. 2017, 120, 345–353. [Google Scholar] [CrossRef]
- Sassoni, E.; Andreotti, S.; Scherer, G.W.; Franzoni, E.; Siegesmund, S. Bowing of marble slabs: Can the phenomenon be arrested and prevented by inorganic treatments? Environ. Earth Sci. 2018, 77, 387. [Google Scholar] [CrossRef]
- Karampas, I.A.; Kontoyannis, C.G. Characterization of calcium phosphates mixtures. Vib. Spectrosc. 2013, 64, 126–133. [Google Scholar] [CrossRef]
- Franzoni, E.; Sassoni, E.; Graziani, G. Brushing, poultice or immersion? The role of the application technique on the performance of a novel hydroxyapatite-based consolidating treatment for limestone. J. Cult. Herit. 2015, 16, 173–184. [Google Scholar] [CrossRef]
- Graziani, G.; Sassoni, E.; Scherer, G.W.; Franzoni, E. Penetration depth and redistribution of an aqueous ammonium phosphate solution used for porous limestone consolidation by brushing and immersion. Construct. Build. Mater. 2017, 148, 571–578. [Google Scholar] [CrossRef]
- Sassoni, E.; Graziani, G.; Franzoni, E. Repair of sugaring marble by ammonium phosphate: Comparison with ethyl silicate and ammonium oxalate and pilot application to historic artifact. Mater. Des. 2015, 88, 1145–1157. [Google Scholar] [CrossRef]
- Sassoni, E.; D’Amen, E.; Roveri, N.; Scherer, G.W.; Franzoni, E. Durable self-cleaning coatings for architectural surfaces by incorporation of TiO2 nanoparticles into hydroxyapatite films. Materials 2018, 11, 177. [Google Scholar] [CrossRef]
- Delgado Rodrigues, J.; Grossi, A. Indicators and ratings for the compatibility assessment of conservation actions. J. Cult. Herit. 2007, 8, 32–43. [Google Scholar] [CrossRef]
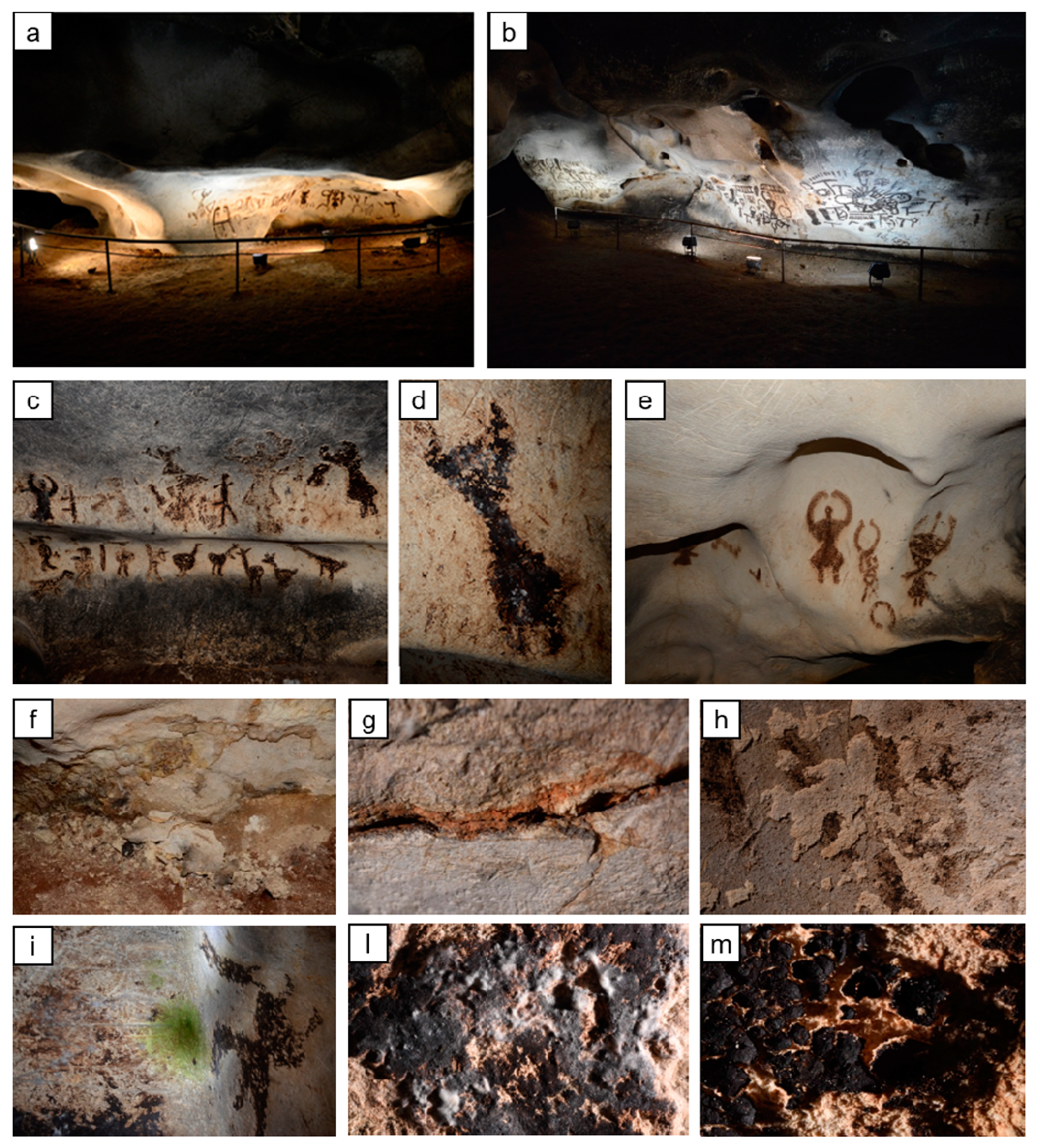

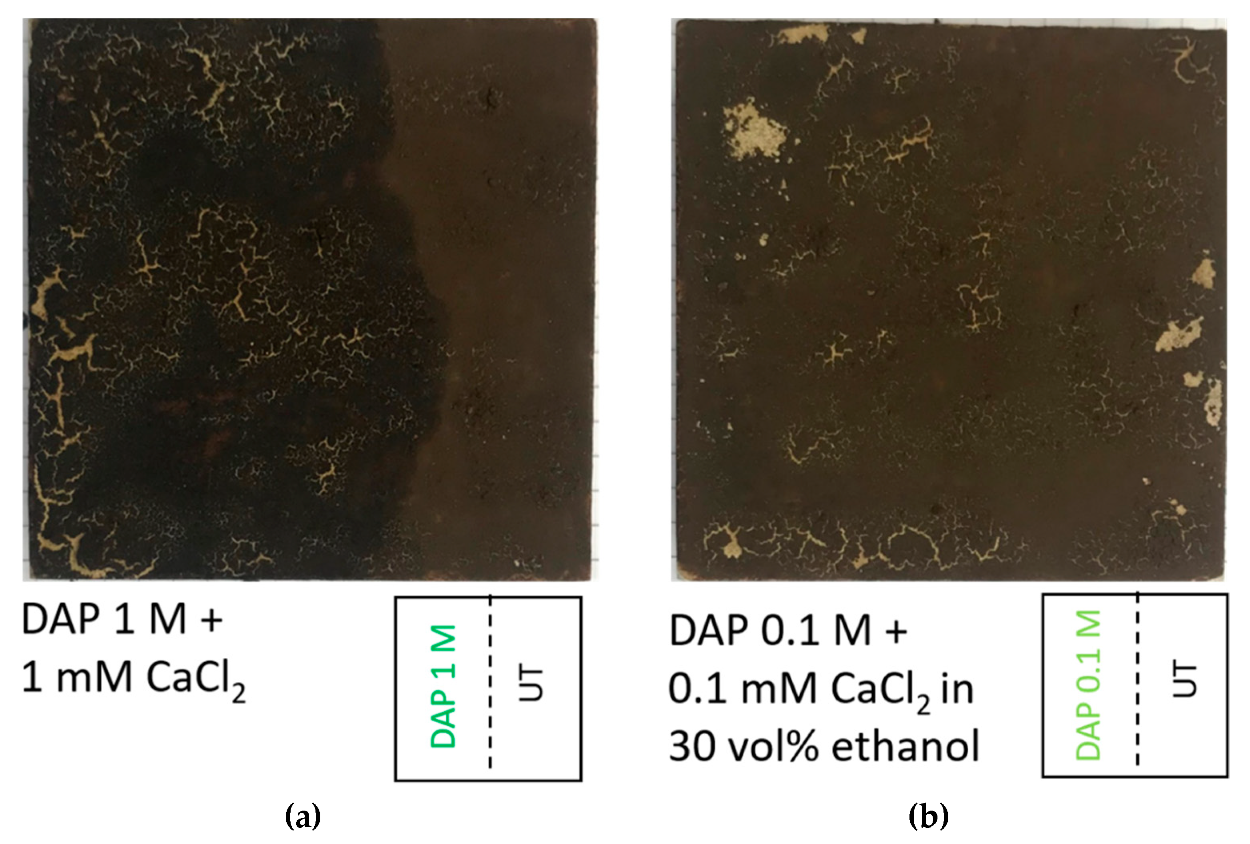

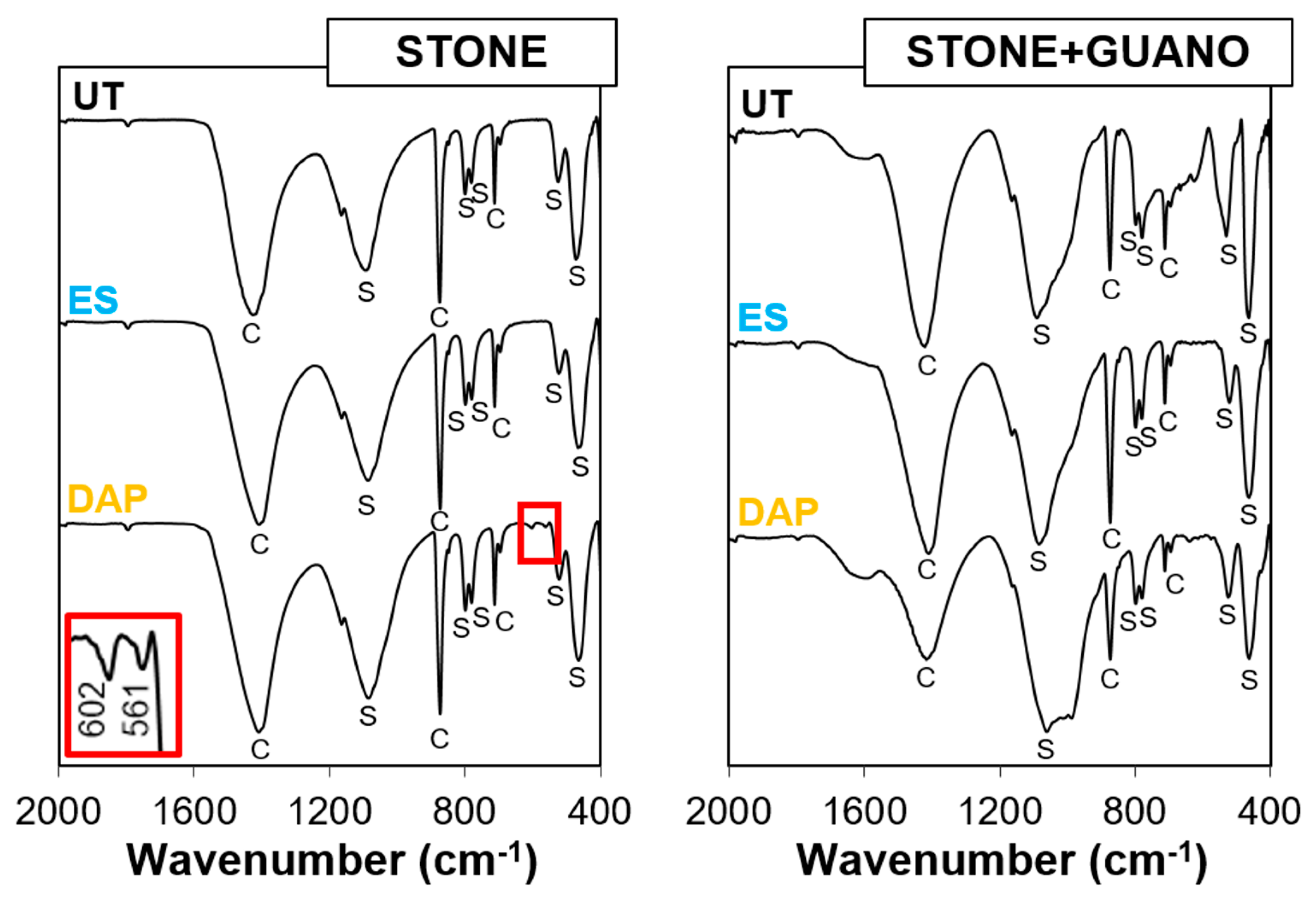

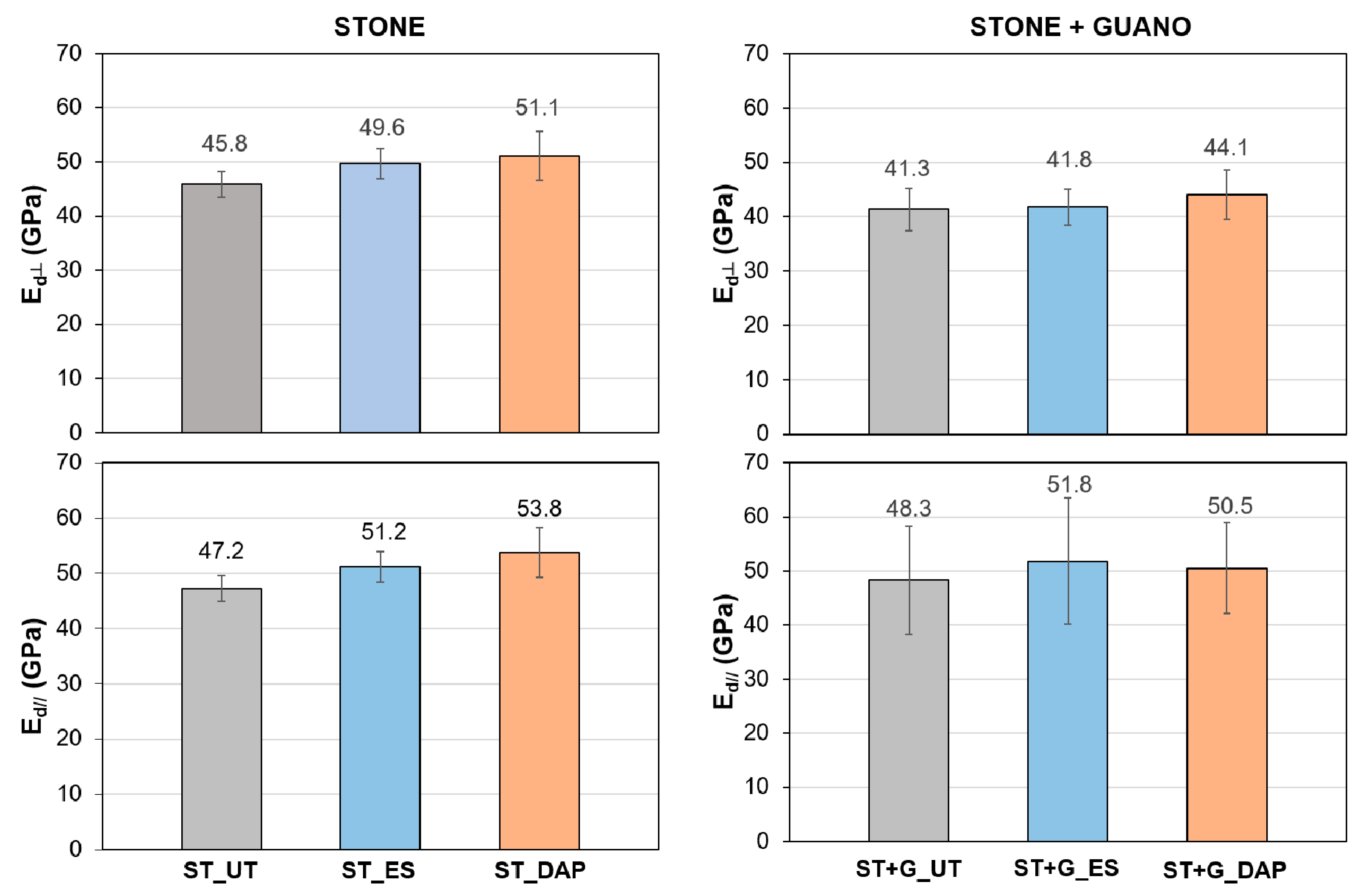

| Specimens | Condition | L* | a* | b* |
|---|---|---|---|---|
| ST | UT | 79.55 ± 0.76 | 2.86 ± 0.18 | 10.36 ± 0.58 |
| ES | 77.20 ± 2.16 | 4.07 ± 0.90 | 10.90 ± 1.90 | |
| DAP | 81.10 ± 1.22 | 2.53 ± 0.23 | 8.98 ± 0.28 | |
| ST+G | UT | 29.13 ± 1.17 | 6.31 ± 0.85 | 7.02 ± 0.29 |
| ES | 24.91 ± 0.88 | 6.46 ± 1.20 | 4.70 ± 0.43 | |
| DAP | 32.25 ± 1.39 | 5.29 ± 0.55 | 7.24 ± 0.31 |
| Specimens | Condition | S (mg/cm2√s) | WA24h (wt.%) | WA7d (wt.%) |
|---|---|---|---|---|
| ST | UT | 1.79 ± 0.14 | 4.9 ± 0.4 | 5.2 ± 0.4 |
| ES | n.d. | 1.9 ± 0.8 | 4.6 ± 0.1 | |
| DAP | 1.65 ± 0.06 | 4.5 ± 0.2 | 4.9 ± 0.3 | |
| ST+G | UT | 1.64 ± 0.01 | 4.0 ± 0 | 4.7 ± 0 |
| ES | n.d. | 0.2 ± 0 | 4.1 ± 0.1 | |
| DAP | 1.57 ± 0.05 | 4.2 ± 0.1 | 4.8 ± 0 |
| Specimens | Condition | μ |
|---|---|---|
| ST | UT | 47.8 ± 0.7 |
| ES | 82.4 ± 11.6 | |
| DAP | 45.8 ± 0.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sassoni, E.; Franzoni, E.; Stefanova, M.; Kamenarov, Z.; Scopece, P.; Verga Falzacappa, E. Comparative Study Between Ammonium Phosphate and Ethyl Silicate Towards Conservation of Prehistoric Paintings in the Magura Cave (Bulgaria). Coatings 2020, 10, 250. https://doi.org/10.3390/coatings10030250
Sassoni E, Franzoni E, Stefanova M, Kamenarov Z, Scopece P, Verga Falzacappa E. Comparative Study Between Ammonium Phosphate and Ethyl Silicate Towards Conservation of Prehistoric Paintings in the Magura Cave (Bulgaria). Coatings. 2020; 10(3):250. https://doi.org/10.3390/coatings10030250
Chicago/Turabian StyleSassoni, Enrico, Elisa Franzoni, Milyana Stefanova, Zdravko Kamenarov, Paolo Scopece, and Emanuele Verga Falzacappa. 2020. "Comparative Study Between Ammonium Phosphate and Ethyl Silicate Towards Conservation of Prehistoric Paintings in the Magura Cave (Bulgaria)" Coatings 10, no. 3: 250. https://doi.org/10.3390/coatings10030250
APA StyleSassoni, E., Franzoni, E., Stefanova, M., Kamenarov, Z., Scopece, P., & Verga Falzacappa, E. (2020). Comparative Study Between Ammonium Phosphate and Ethyl Silicate Towards Conservation of Prehistoric Paintings in the Magura Cave (Bulgaria). Coatings, 10(3), 250. https://doi.org/10.3390/coatings10030250







