Influence of Process Conditions on the Growth and Texture of CVD Alpha-Alumina
Abstract
1. Introduction
2. Experimental
2.1. CVD Process
2.2. α-Al2O3 Coatings
2.2.1. Multilayer Coatings
2.2.2. Coatings for Texture Analysis
2.2.3. Manipulation of α-Al2O3 Microstructures
2.3. Analysis
3. Results and Discussion
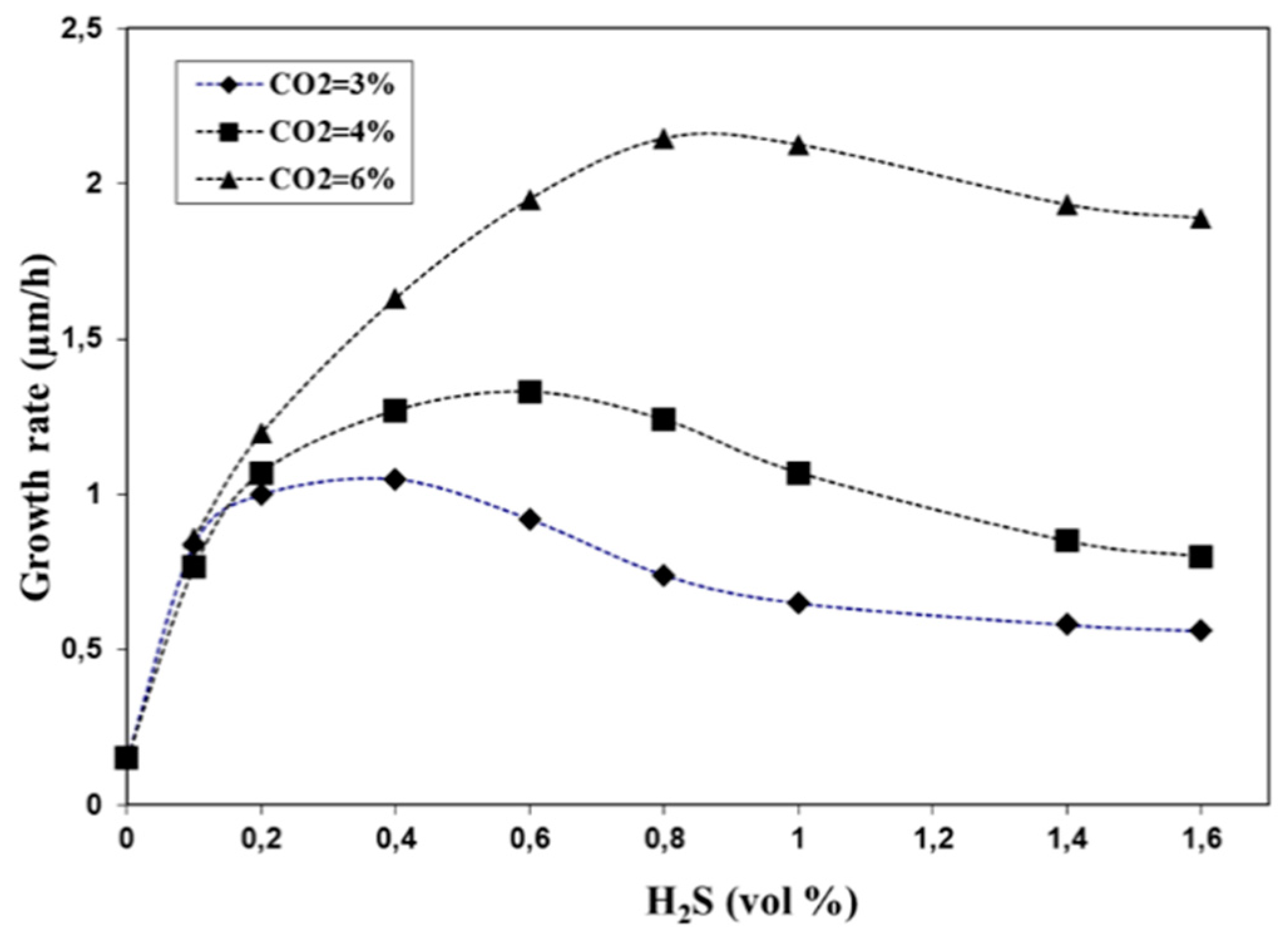
3.1. Influence of H2S on Growth Rate of α-Al2O3
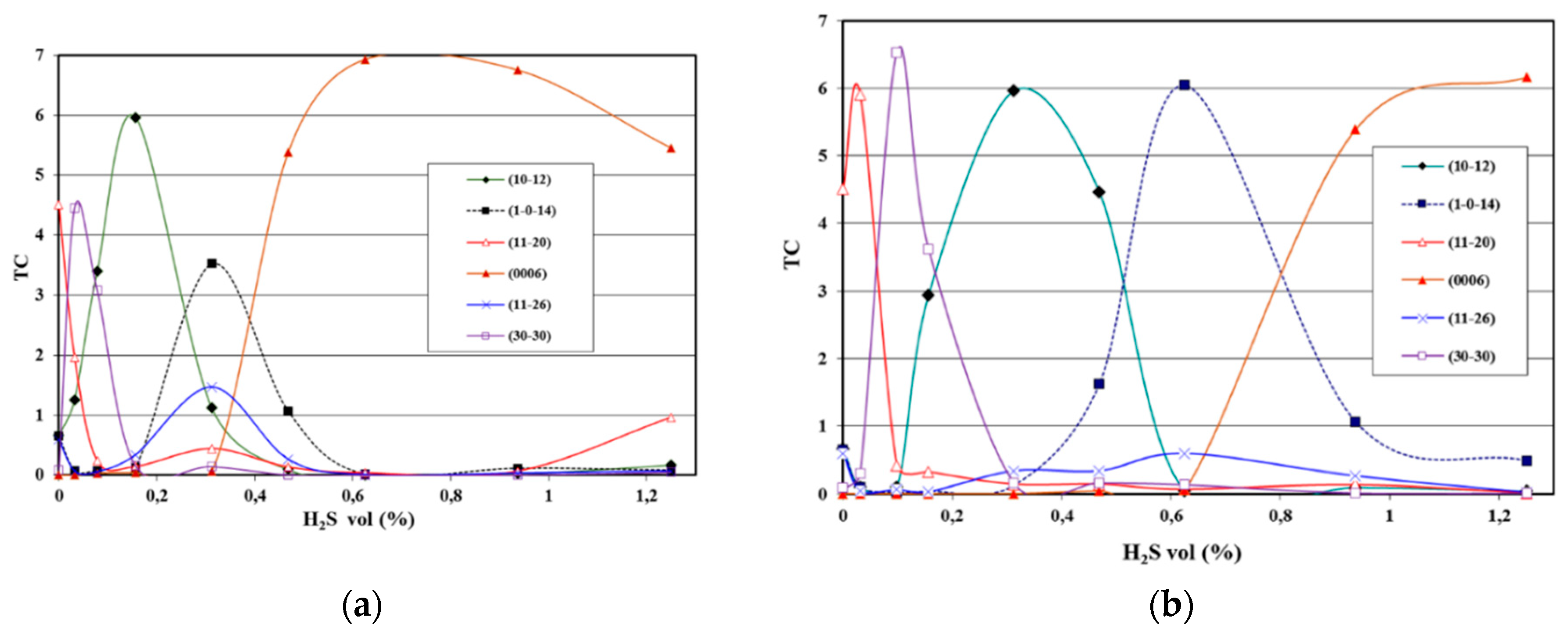
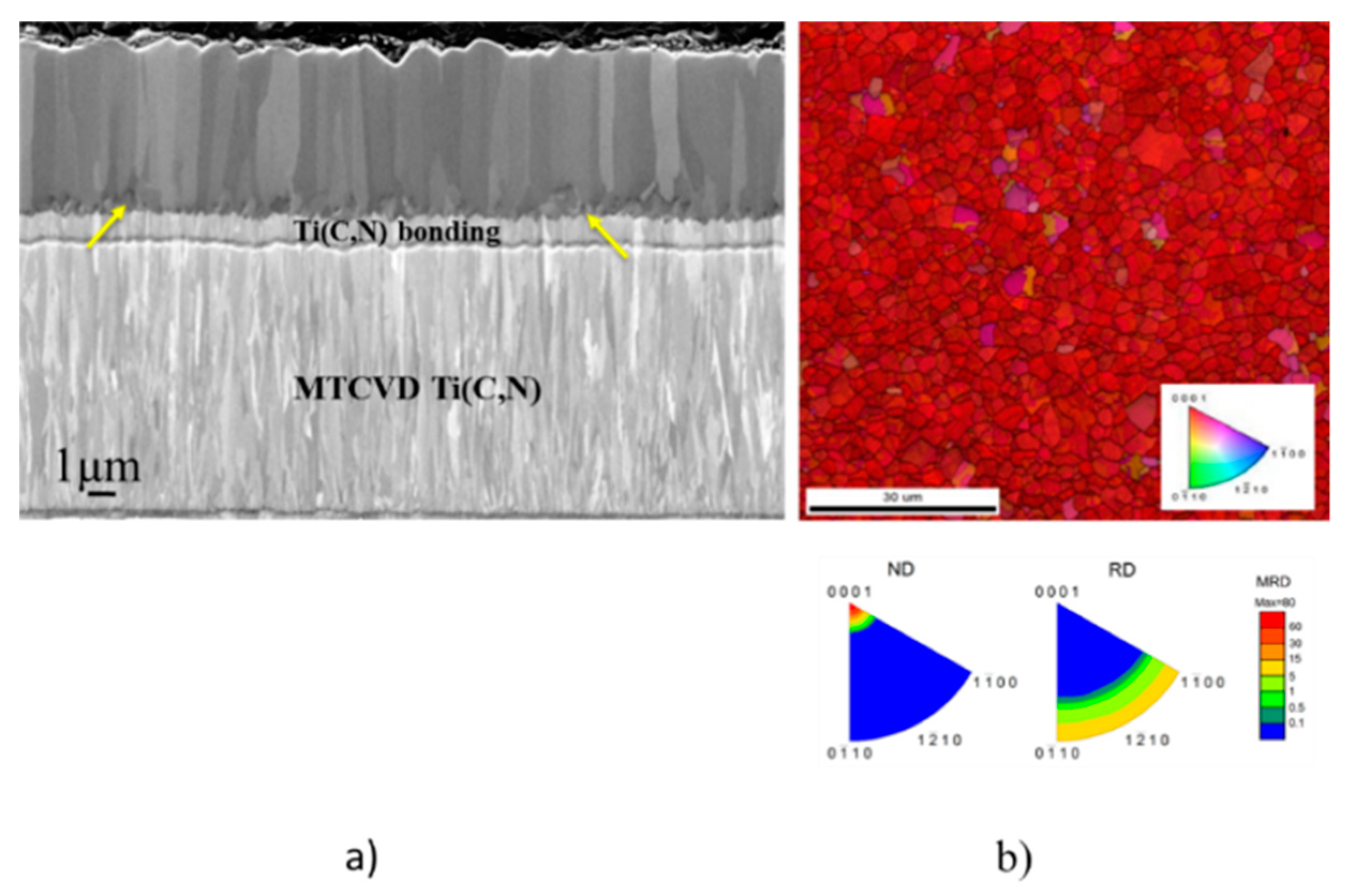
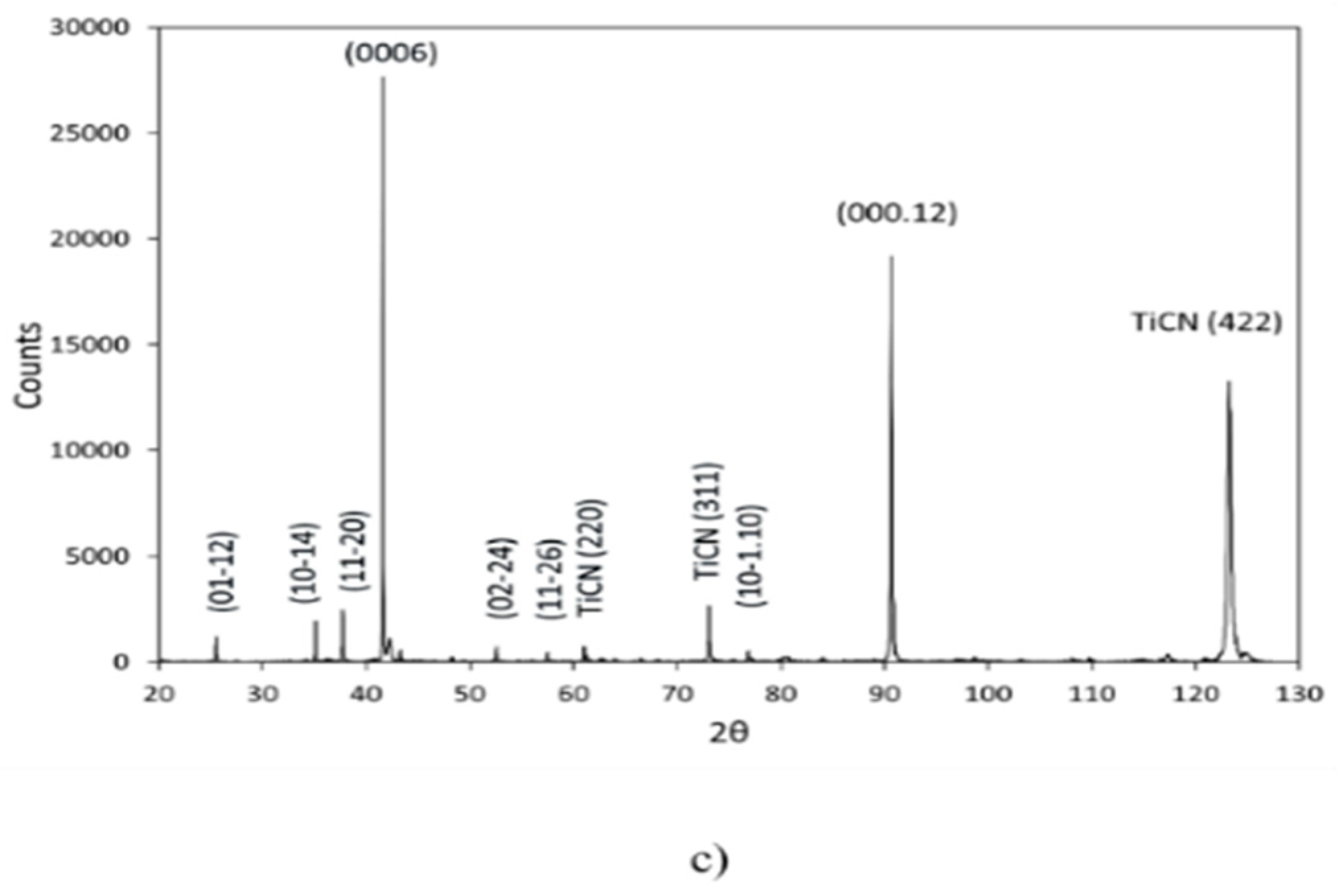
3.2. Texture and Microstructure of α-Al2O3
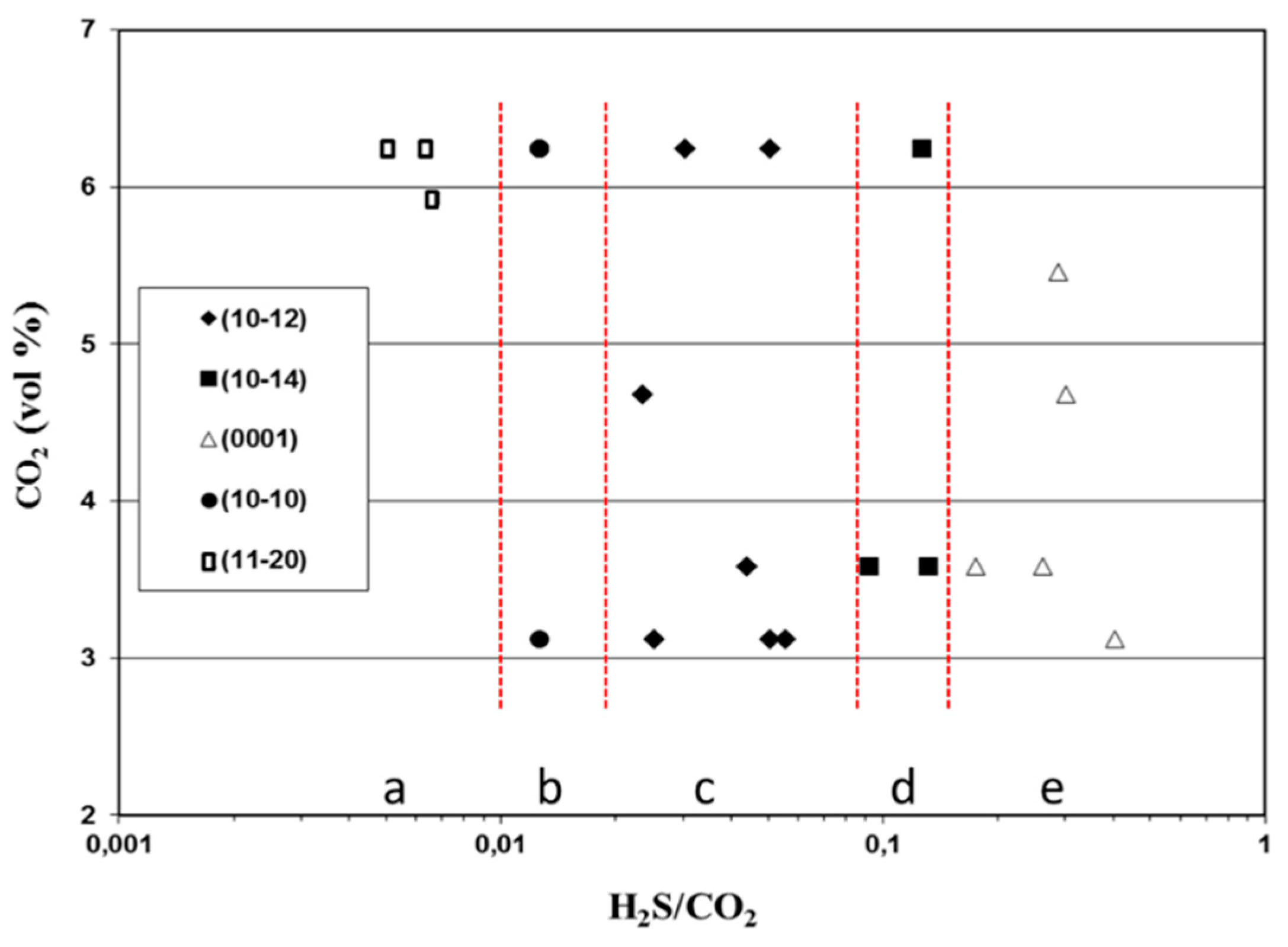
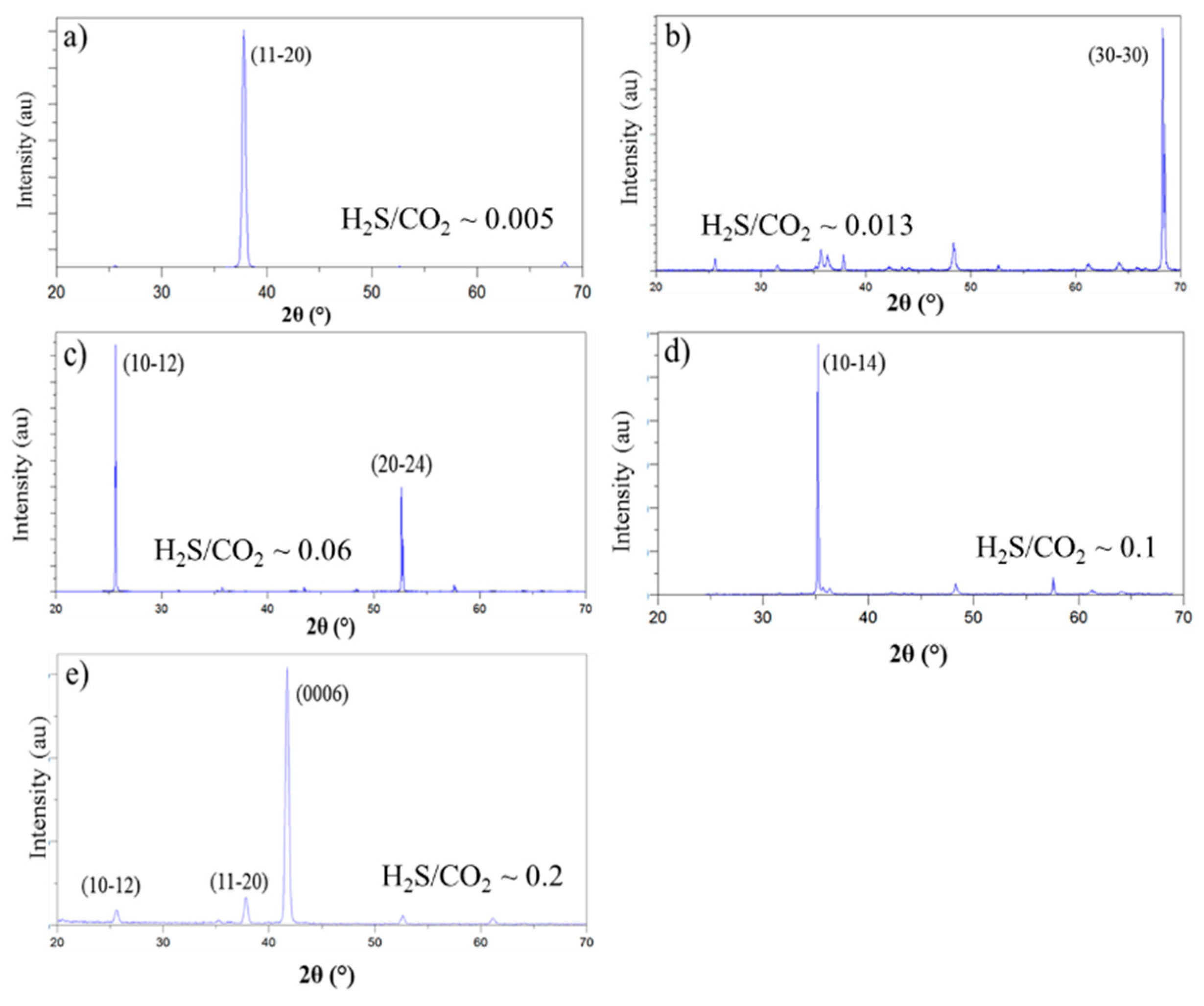
3.2.1. Texture Development
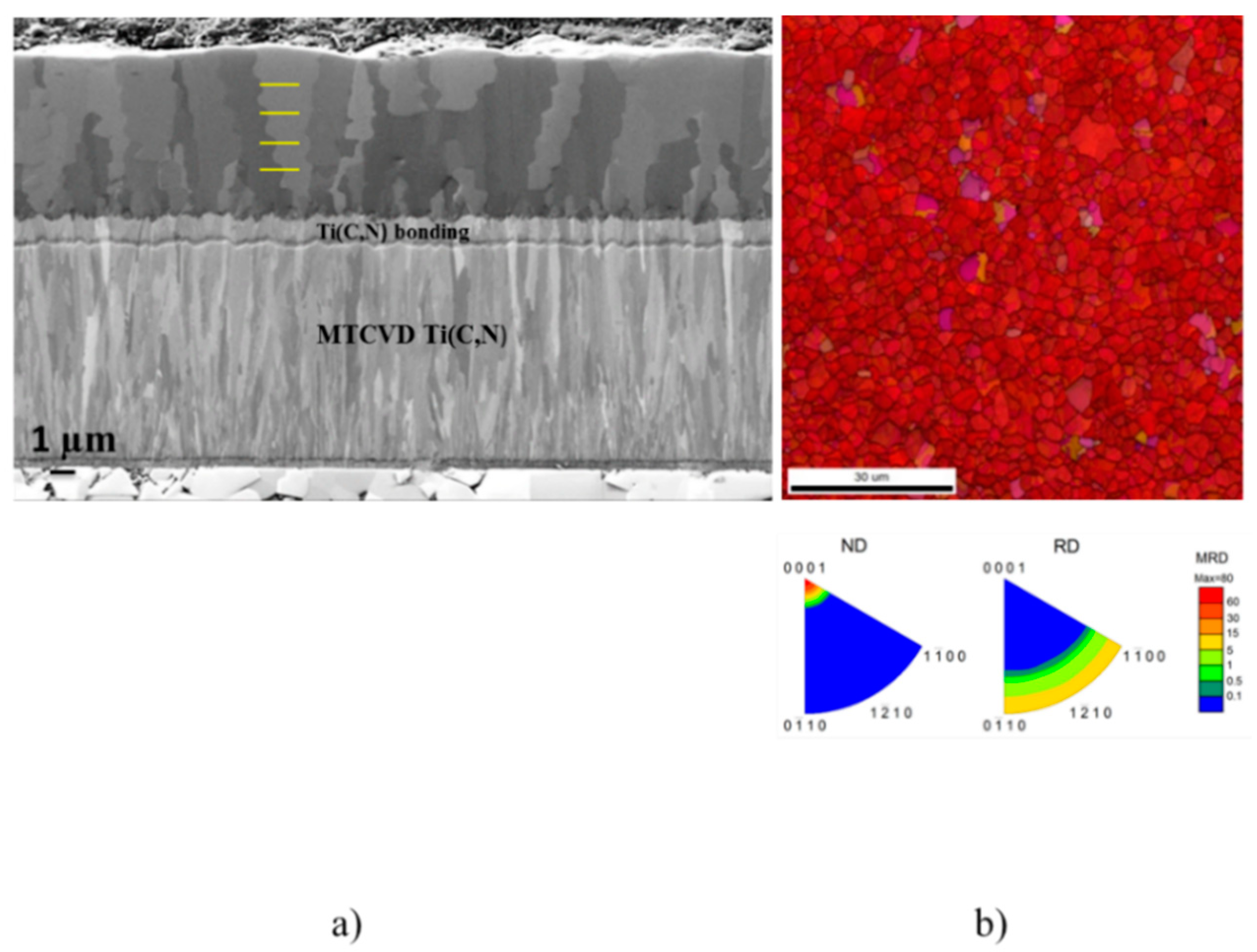
3.2.2. Microstructure
3.2.3. Texture-Manipulated Structures
3.2.4. Discussion
4. Conclusions
- The experiments confirmed that the growth rate of α-Al2O3 could be increased considerably by applying high CO2 and H2S partial pressures.
- When deposited uncatalyzed (H2S = 0) and using very low H2S concentrations the resulting α-Al2O3 layer exhibited (110) texture.
- As a result of the increment of H2S at constant CO2 concentration the texture of α-Al2O3 changed from (110), which was obtained at very low H2S concentrations, through (100), (102) and (104) textures to (0001) texture. Consequently, the fiber axis of α-Al2O3 was rotated by 90° as a result of H2S increment.
- The H2S/CO2 ratio was identified as an important parameter to control the texture of α-Al2O3. Growth regimes for (110), (100), (102), (104) and (0001) textures were depicted as a function of the H2S/CO2 ratio. With the increment of H2S/CO2 ratio the texture of α-Al2O3 changed from (110), which was obtained at H2S/CO2 < 0.01 through the (100), (102) and (104) textures to the (0001) texture, which was obtained at the H2S/CO2 ratios exceeding ~0.2.
- Even though this work has focused on the effect of H2S concentration at constant CO2 levels, it should be noted that based on the present results is straightforward to conclude that the H2S/CO2 ratio can also be varied by varying the CO2 concentration at a constant H2S level.
- This work also unveiled new possibilities to manipulate the surface and grain boundary structures of the CVD α-Al2O3 coating by utilizing the texture-control.
- The work demonstrated that the properties of the MTCVD Ti(C,N) layer and especially the bonding, nucleation and oxidation procedures are of crucial importance for full utilization of the process windows according to this work.
5. Suggestions for the Further Work
Funding
Conflicts of Interest
References
- Lux, B.; Colombier, C.; Altena, H.; Stjernberg, K. Preparation of alumina coatings by chemical vapour deposition. Thin Solid Films 1986, 138, 49–64. [Google Scholar] [CrossRef]
- Kramer, B.M.; Suh, N.P. Tool wear by solution: A quantitative understanding. J. Eng. Ind. 1980, 102, 303–309. [Google Scholar] [CrossRef]
- Kramer, B.M.; Judd, P.K. Computational design of wear coatings. J. Vac. Sci. Technol. A 1985, 3, 2439–2444. [Google Scholar] [CrossRef]
- Ruppi, S. Advances in chemically vapour deposited wear resistant coatings. J. Phys. IV 2001, 11, Pr3-847–Pr3-859. [Google Scholar] [CrossRef]
- Lindström, J.; Schachner, H. Non-equilibrium conditions for CVD of alumina. In Proceedings of the 3rd European Conference on CVD, Neuchatel, Switzerland, 16–18 April 1980; Hinterman, H.E., Ed.; LSRH: Neuchatel, Switzerland, 1980; pp. 208–217. [Google Scholar]
- Lindström, J.; Stjernberg, K. Rate determining steps at CVD of Al2O3, TiC and TiN. In Proceedings of the 5th European Conference on CVD, Uppsala, Sweden, 17–20 June 1985; Carlsson, J.-O., Lindström, J.J., Eds.; Uppsala University: Uppsala, Sweden, 1985; pp. 169–182. [Google Scholar]
- Kim, J.G.; Park, C.S.; Chun, J.S. Effect of partial pressure of the reactant gas on the chemical vapour deposition of Al2O3. Thin Solid Films 1982, 97, 97–106. [Google Scholar] [CrossRef]
- Lhermitte-Sebire, I.; Colmet, R.; Naslain, R.; Bernard, C. The Chemical Vapour Deposition of alumina from AlCl3-H2-CO2 on Stoichiometric TiC substarte: A thermodynamic approach. J. Less Common Met. 1968, 118, 83–102. [Google Scholar] [CrossRef]
- Colmet, R.; Naslain, R. Chemical vapour deposition of alumina on cutting tool inserts from AlCl3-H2-CO2 mixtures: Influence of the chemical vapour deposition parameters and the nature of the inserts on the morphology and wear resistance of the coatings. Wear 1982, 80, 221–231. [Google Scholar] [CrossRef]
- Ruppi, S.; Larsson, A. Chemical vapour deposition of κ-Al2O3. Thin Solid Films 2001, 388, 50–61. [Google Scholar] [CrossRef]
- Fredriksson, E.; Carlsson, J.-O. Chemical vapour deposition of Al2O3 on TiO. Thin Solid Films 1995, 263, 28–36. [Google Scholar] [CrossRef]
- Park, C.S.; Kim, J.G.; Chun, J.S. Crystallographic Orientation and Surface Morphology of Chemical Vapor Deposited Al2O3. J. Electrochem. Soc. 1983, 130, 1607–1611. [Google Scholar] [CrossRef]
- Catoire, L.; Swihart, M.T. High-temperature kinetics of AlCl3 decomposition in the presence of additives for chemical vapor deposition. J. Electrochem. Soc. 2002, 149, C261–C267. [Google Scholar] [CrossRef]
- Swihart, M.T.; Catoire, L. Reactions in the Al-H-Cl system by ab initio molecular orbital and density functional methods. J. Phys. Chem. A 2001, 105, 264–273. [Google Scholar] [CrossRef]
- Tan, P.; Müller, J. Gas-phase kinetic modelling of the AlCl3 decomposition in the AlCl3-CO2 H2-HCl system for a hot-wall CVD reactor, D. Neuschütz. J. Electrochem. Soc. 2005, 152, C288–C293. [Google Scholar] [CrossRef]
- Blomqvist, A.; Arhammar, C.; Pedersen, H.; Silvearv, F.; Norgren, S.; Ahuja, R. Understanding the catalytic effects of H2S on CVD-growth of α-alumina: Thermodynamic gas-phase simulations and density functional theory. Surf. Coat. Technol. 2011, 206, 1771–1779. [Google Scholar] [CrossRef]
- Smith, U.K.; Lindstrom, J.N. Method of Making a Coated Cemented Carbide Body and Resulting Body. U.S. Patent 4,619,866 A, 2 April 1985. [Google Scholar]
- Vuorinen, S.; Karlsson, L. Phase transformation in chemically vapour deposited, κ-Al2O3. Thin Solid Films 1992, 214, 132–143. [Google Scholar] [CrossRef]
- Vuorinen, S.; Skogsmo, J. Characterization of α-Al2O3, κ-Al2O3 and α-κ multioxide coatings on cemented carbides. Thin Solid Films 1990, 536, 193–194. [Google Scholar] [CrossRef]
- Ruppi, S. Deposition, Microstructure and properties of texture-controlled CVD α-Al2O3 coatings. Int. J. Refract. Met. Hard Mater. 2005, 23, 306–316. [Google Scholar] [CrossRef]
- Ruppi, S. Enhanced performance of α-Al2O3 coatings by control of crystal orientation. Surf. Coat. Technol. 2008, 202, 4257–4269. [Google Scholar] [CrossRef]
- Ruppi, S. Enhanced Alpha Alumina Layer Produced by CVD. U.S. Patent 7,396,581 B2, 8 July 2008. [Google Scholar]
- Ruppi, S. Alumina Layer with Enhanced Texture. U.S. Patent 7,993,742 B2, 9 August 2011. [Google Scholar]
- Ruppi, S. Texture Hardened Alpha-Alumina Coated Tool. U.S. Patent 7,923,101 B2, 12 April 2011. [Google Scholar]
- Stylianou, R.; Tkadletz, M.; Schalk, N.; Penoy, M.; Czettl, C.; Mitterer, C. Effects of reference materials on texture coefficients determined for a CVD α-Al2O3coating. Surf. Coat. Technol. 2019, 359, 314–322. [Google Scholar] [CrossRef]
- Oshika, T.; Nishiyama, A.; Nakaso, K.; Shimada, M.; Okuyama, K. Unveiling the magic of H2S on CVD-Al2O3 coating. J. Phys. IV 1999, 9, 877–883. [Google Scholar] [CrossRef]
- Oshika, T.; Sato, M.; Nishiyama, A. Unveiling the magic of H2S on the CVD-Al2O3coating. Effect of H2S on the water gas concentration. J. Phys. IV 2002, 12, 113–120. [Google Scholar] [CrossRef]
- Slager, T.; Amberg, C. Infrared investigation of H2S adsorption and decomposition on alumina and alumina supported molybdenum sulfide. Can. J. Chem. 1972, 50, 3416–3423. [Google Scholar] [CrossRef]
- Arhammar, C.; Silvearv, F.; Bergman, A.; Norgren, S.; Pedersen, H.; Ahuja, R. A theoretical study of possible point defects incorporated into α-alumina deposited by chemical vapor deposition. Theor. Chem. Acc. 2014, 133, 1–11. [Google Scholar] [CrossRef]
- Shoja, S.; Mortazavi, N.; Lindahl, E.; Norgren, S.; Bäcke, O.; Halvarsson, M. Microstructural investigation of textured CVD alumina coatings. Int. J. Refract. Met. Hard Mater. 2020, 87, 105125. [Google Scholar] [CrossRef]
- Harris, G.B. Quantitative measurement of preferred orientation in rolled uranium bars. Data Commun. Natl. Phys. Lab. 1952, 43, 113–123. [Google Scholar] [CrossRef]
- Grimmer, H. Coincidence orientations of grains in rhombohedral materials. Acta Crytallogr. A 1989, 45, 505–523. [Google Scholar] [CrossRef]
- Brandon, D.G. Structure of high-angle grain boundaries. Acta Metall. 1966, 14, 1479–1484. [Google Scholar] [CrossRef]
- Engler, O.; Randle, V. Introduction to Texture Analysis: Macrotexture, Microtexture and Orientation Mapping, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Hishahi, H.; Osada, A.; Nakamura, E.; Takuya, H. Surface-Coated Cermet Cutting Tool with Hard Coating Layer Exhibiting Excellent Chipping Resistance in High-Speed Intermittent Cutting. U.S. Patent 7,442,433 B2, 28 October 2008. [Google Scholar]
- Stiens, D.; Ruppi, S. Grain Boundary Engineered Alpha-Alumina Coated Cutting Tool. U.S. Patent 9,206,510 B2, 8 December 2015. [Google Scholar]
- Ruppi, S.; Stiens, D.; Manns, T. Grain Boundary Engineered α-Al2O3 Coatings. ICMCTF, San Diego, CA. 2014. Available online: http://www.researchgate.net/publication/274192418 (accessed on 7 February 2020).
- Vonlanthen, P.; Grobety, B. CSL grain boundary distribution in alumina and zirconia ceramics. Ceram. Int. 2008, 34, 1459–1472. [Google Scholar] [CrossRef]
- Stiens, D.; Ruppi, S. Alumina Coated Cutting Tool with Zigzag Alumina Grain Boundaries. European Patent 2,902,528 B1, 29 June 2016. [Google Scholar]
- Ruppi, S.; Engström, H.; Lauridsen, J.; Alm, O.; Matsson, P.; Larsson, T.; Lindahl, E.; Engqvist, J.; Stiens, D. Alumina Coated Cutting Tool. WO Patent 2,015,114,049 A1, 6 August 2015. [Google Scholar]
- Mårtensson, P. Influence of the concentration of ZrCl4 on texture, morphology and growth rate of CVD grown α-Al2O3 coatings deposited by the AlCl3/ZrCl4/H2/CO2/H2S process. Surf. Coat. Technol. 2006, 200, 3626. [Google Scholar] [CrossRef]
- Kathrein, M.; Schintlmeister, W.; Wallgram, W.; Schleinkofer, U. Doped CVD Al2O3 coating for high performance cutting tools. Surf. Coat. Technol. 2003, 163, 181–188. [Google Scholar] [CrossRef]
- Kronberg, M.L. Plastic deformation of single crystals of sapphire: Basal slip and twinning. Acta Met. 1957, 5, 507–524. [Google Scholar] [CrossRef]
- Hartman, P. The attachment energy as a habit controlling factor III. Application to corundum. J. Cryst. Growth 1980, 29, 166–170. [Google Scholar] [CrossRef]
- Hinneman, B.; Carter, E. Adsorption of Al, O, Hf, Y, Pt, and S atoms on α-Al2O3 (0001). Phys. Chem. C 2007, 111, 7105–7126. [Google Scholar] [CrossRef]
- Wang, X.-G.; Chaga, A.; Scheffler, M. Effect of the environment on (0001) α-Al2O3 surface structures. Phys. Rev. Lett. 2000, 16, 3650–3653. [Google Scholar]
- Marmier, A.; Parker, S.C. Ab initio morphology and surface thermodynamics of α-Al2O3. Phys. Rev. B 2004, 69, 115409. [Google Scholar] [CrossRef]
- Liu, Y.; Ning, X.-S. Termination stability and electronic structures of α-Al2O3 (0114) surface: An ab initio study. Appl. Surf. Sci. 2014, 303, 210–216. [Google Scholar] [CrossRef]
- Ranea, V.A.; Schneider, W.F.; Carmichael, I. DFT characterization of coverage dependent molecular water adsorption modes on α-Al2O3 (0001). Surf. Sci. 2008, 602, 268–275. [Google Scholar] [CrossRef]



| H2S (vol %) | CO2 (vol %) | AlCl3 (vol %) | HCl (vol %) | H2 (vol %) | P (mbar) |
|---|---|---|---|---|---|
| 0.1–1.6 | 3 | 2.5 | 1 | Balance | 75 |
| 0.1–1.6 | 4 | 2.5 | 1 | Balance | 75 |
| 0.1–1.6 | 6 | 2.5 | 1 | Balance | 75 |
| Texture Experiments | Reference Coating | |
|---|---|---|
| H2S (vol %) | 0.02–1.6 | 0 |
| CO2 (vol %) | 3.0–6.5 | 3.25 |
| H2S/CO2 ratio | 0.004-0.40 | 0 |
| AlCl3 (vol %) | 2.5 | 2.5 |
| HCl (vol %) | 1.0 | 1.0 |
| H2 (vol %) | Balance | Balance |
| P (mbar) | 75 | 75 |
| Time (min) | 480 | 1200 |
| Coating A | |
|---|---|
| H2S/CO2 ratio | 0.40 |
| AlCl3 (vol %) | 2.65 |
| HCl (vol %) | 1.1 |
| H2 (vol %) | Balance |
| P (mbar) | 75 |
| Time (min) | 480 |
| First Part | Second Part | |
|---|---|---|
| H2S/CO2 ratio | 0.40 | <0.01 |
| AlCl3 (vol %) | 2.65 | 2.65 |
| HCl (vol %) | 1.5 | 1.5 |
| H2 (vol %) | Balance | Balance |
| P (mbar) | 85 | 55 |
| Time (min) | 420 | 60 |
| (hkil) | TC |
|---|---|
| (102) | 0.67 |
| (104) | 0.65 |
| (110) | 4.50 |
| (0006) | 0 |
| (113) | 0.50 |
| (116) | 0.59 |
| (300) | 0.09 |
| Texture (hkil) | TC (hkil) | Fraction of Σ3 CSL GBs (%) |
|---|---|---|
| (110) | 5.9 | ~5 |
| (100) | 6.5 | ~5 |
| (102) | 5.9 | ~12 |
| (0001) | 6.2 | ~50 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruppi, S. Influence of Process Conditions on the Growth and Texture of CVD Alpha-Alumina. Coatings 2020, 10, 158. https://doi.org/10.3390/coatings10020158
Ruppi S. Influence of Process Conditions on the Growth and Texture of CVD Alpha-Alumina. Coatings. 2020; 10(2):158. https://doi.org/10.3390/coatings10020158
Chicago/Turabian StyleRuppi, S. 2020. "Influence of Process Conditions on the Growth and Texture of CVD Alpha-Alumina" Coatings 10, no. 2: 158. https://doi.org/10.3390/coatings10020158
APA StyleRuppi, S. (2020). Influence of Process Conditions on the Growth and Texture of CVD Alpha-Alumina. Coatings, 10(2), 158. https://doi.org/10.3390/coatings10020158





