Antibacterial Coatings for Improving the Performance of Biomaterials
Abstract
1. Introduction
1.1. Common Materials Used in Biomedical Devices
1.1.1. Ceramic
1.1.2. Metal and Metal Alloys
1.1.3. Polymers
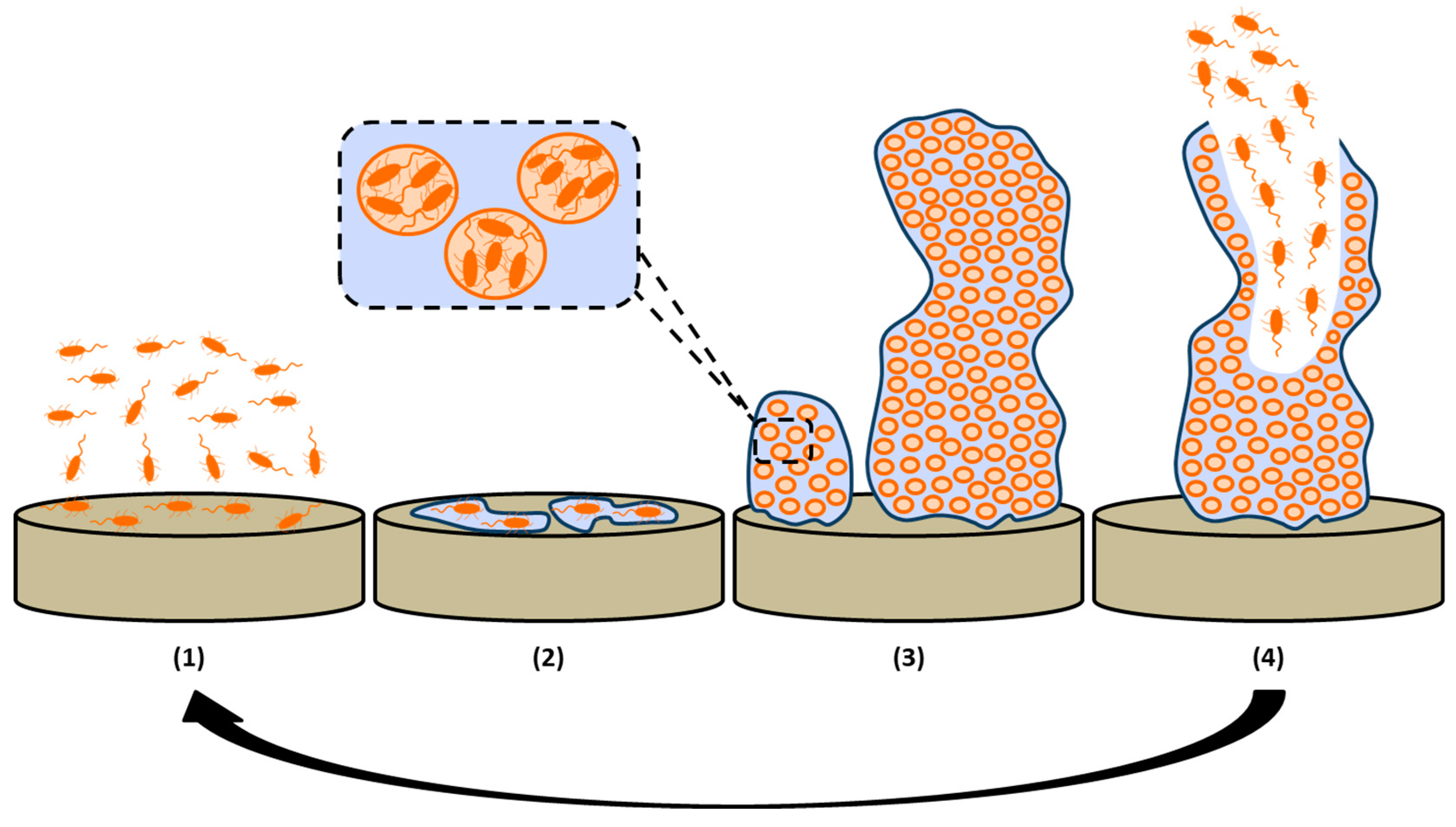
2. Biofilm Formation
2.1. Reversible Attachment of Bacteria Cells
2.2. Irreversible Attachment due to Extracellular Polymeric Matrix (EPM) Secretion
2.3. Development of Biofilm Structure
2.4. Maturation and Dispersion of Biofilm
3. Antibacterial Mechanisms
3.1. Bacteria Repelling Surfaces
3.1.1. Hydrophylic Surfaces
3.1.2. Charged Surfaces
3.1.3. Superhydrophobic Surfaces
3.2. Active Action (Killing)
3.2.1. Contact-Based Antibacterial Surfaces
3.2.2. Release-Based Antibacterial Surfaces
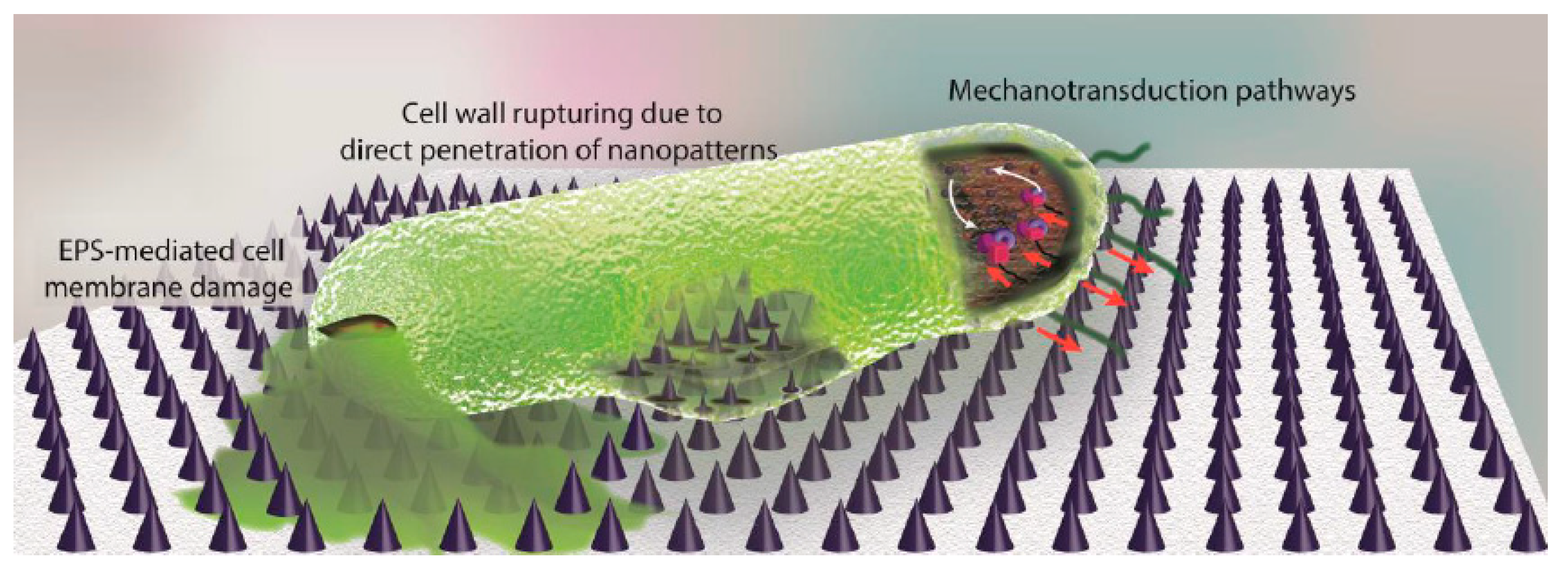
3.2.3. Nanopatterned Surfaces with Antibacterial Behaviour
4. Conclusions and Future Work
Author Contributions
Funding
Conflicts of Interest
References
- Yin, J.; Luan, S. Opportunities and challenges for the development of polymer-based biomaterials and medical devices. Regen. Biomater. 2016, 3, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Narayana, S.V.V.S.P.; Srihari, S.V.V.P. A review on surface modifications and coatings on implants to prevent biofilm. Regen. Eng. Transl. Med. 2019. [Google Scholar] [CrossRef]
- Ben, M.; Mavri, T.; Kralj-igli, V. The importance of antibacterial surfaces in biomedical applications. Adv. Biomembr. Lipid Self Assem. 2018, 28, 115–165. [Google Scholar]
- Langer, R.S.; Lendlein, A. Biodegradable, elastic shape-memory polymers for potential biomedical applications. Science. 2002, 1673–1676. [Google Scholar] [CrossRef]
- Sotiropoulos, F.; Borazjani, I. A review of state-of-the-art numerical methods for simulating flow through mechanical heart valves. Med. Biol. Eng. Comput. 2009, 47, 245–256. [Google Scholar] [CrossRef]
- Singha, P.; Locklin, J.; Handa, H. A review of the recent advances in antimicrobial coatings for urinary catheters. Acta Biomater. 2017, 50, 20–40. [Google Scholar] [CrossRef]
- Gallieni, M.; Giordano, A.; Pinerolo, C.; Cariati, M. Type of peritoneal dialysis catheter and outcomes. J. Vasc. Access 2015, 16, S68–S72. [Google Scholar] [CrossRef]
- Mani, G.; Feldman, M.D.; Patel, D.; Agrawal, C.M. Coronary stents: A materials perspective. Biomaterials 2007, 28, 1689–1710. [Google Scholar] [CrossRef]
- Jayaswal, G.P.; Dange, S.P.; Khalikar, A.N. Bioceramic in dental implants: A review. J. Indian Prosthodont. Soc. 2010, 10, 8–12. [Google Scholar] [CrossRef]
- Xinming, L.; Yingde, C.; Lloyd, A.W.; Mikhalovsky, S.V.; Sandeman, S.R.; Howel, C.A.; Liewen, L. Polymeric hydrogels for novel contact lens-based ophthalmic drug delivery systems: A review. Contact Lens Anterior Eye 2008, 31, 57–64. [Google Scholar] [CrossRef]
- Chehade, M.; Elder, M.J. Intraocular lens materials and styles: A review. Aust. N. Z. J. Ophthalmol. 1997, 25, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Dormer, K.J.; Gan, R.Z. Biomaterials for implantable middle ear hearing devices. Otolaryngol. Clin. N. Am. 2001, 34, 289–297. [Google Scholar] [CrossRef]
- Granchi, D.; Cenni, E.; Trisolino, G.; Giunti, A.; Baldini, N. Sensitivity to implant materials in patients undergoing total hip replacement. J. Biomed. Mater. Res. Part B Appl. Biomater. 2006, 77B, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Price, A.J.; Alvand, A.; Troelsen, A.; Katz, J.N.; Hooper, G.; Gray, A.; Carr, A.; Beard, D. Knee replacement. Lancet 2018, 392, 1672–1682. [Google Scholar] [CrossRef]
- Barrows, T. Degradable implant materials: A review of synthetic absorbable polymers and their applications. Clin. Mater. 1986, 1, 233–257. [Google Scholar] [CrossRef]
- Thamaraiselvi, T.; Rajeswari, S. Biological evaluation of bioceramic materials-a review. Trends Biomater. Artif. Organs 2004, 18, 9–17. [Google Scholar]
- Thompson, J.Y.; Stoner, B.R.; Piascik, J.R.; Smith, R. Adhesion/cementation to zirconia and other non-silicate ceramics: Where are we now? Dent. Mater. 2011, 27, 71–82. [Google Scholar] [CrossRef]
- Hisbergues, M.; Vendeville, S.; Vendeville, P. Zirconia: Established facts and perspectives for a biomaterial in dental implantology. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 88B, 519–529. [Google Scholar] [CrossRef]
- Savvides, N.; Bell, T.J. Hardness and elastic modulus of diamond and diamond-like carbon films. Thin Solid Films 1993, 228, 289–292. [Google Scholar] [CrossRef]
- Love, C.A.; Cook, R.B.; Harvey, T.J.; Dearnley, P.A.; Wood, R.J.K. Diamond like carbon coatings for potential application in biological implants—A review. Tribol. Int. 2013, 63, 141–150. [Google Scholar] [CrossRef]
- Kohn, D.H. Metals in medical applications. Curr. Opin. Solid State Mater. Sci. 1998, 3, 309–316. [Google Scholar] [CrossRef]
- Zhao, L.; Chu, P.K.; Zhang, Y.; Wu, Z. Antibacterial coatings on titanium implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 91B, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Teo, A.J.T.; Mishra, A.; Park, I.; Kim, Y.-J.; Park, W.-T.; Yoon, Y.-J. Polymeric Biomaterials for Medical Implants and Devices. ACS Biomater. Sci. Eng. 2016, 2, 454–472. [Google Scholar] [CrossRef]
- Biomedical Composites; Elsevier: Amsterdsm, Holland, 2017; ISBN 9780081007525.
- Saad, M.; Akhtar, S.; Srivastava, S. Composite polymer in orthopedic implants: A review. Mater. Today Proc. 2018, 5, 20224–20231. [Google Scholar] [CrossRef]
- Schulman, A.; Piliero, S.J.; Pentel, L.; Ott, W.R.; Moskowitz, H.D. Composite materials for dental implants. Oral Surg. Oral Med. Oral Pathol. 1975, 40, 183–193. [Google Scholar] [CrossRef]
- Zobell, C.E. The effect of solid surfaces upon bacterial activity. J. Bacteriol. 1943, 46, 39–56. [Google Scholar] [CrossRef]
- Vasilev, K. Nanoengineered antibacterial coatings and materials: A perspective. Coatings. 2019, 9, 654. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Speziale, P.; Montanaro, L.; Costerton, J.W. Biofilm formation in Staphylococcus implant infections. A review of molecular mechanisms and implications for biofilm-resistant materials. Biomaterials 2012, 33, 5967–5982. [Google Scholar] [CrossRef]
- Bazaka, K.; Jacob, M.V.; Crawford, R.J.; Ivanova, E.P. Efficient surface modification of biomaterial to prevent biofilm formation and the attachment of microorganisms. Appl. Microbiol. Biotechnol. 2012, 95, 299–311. [Google Scholar] [CrossRef]
- Veerachamy, S.; Yarlagadda, T.; Manivasagam, G.; Yarlagadda, P.K. Bacterial adherence and biofilm formation on medical implants: A review. Proc. Inst. Mech. Eng. Part. H J. Eng. Med. 2014, 228, 1083–1099. [Google Scholar] [CrossRef]
- Armbruster, C.R.; Parsek, M.R. New insight into the early stages of biofilm formation. Proc. Natl. Acad. Sci. USA 2018, 115, 4317–4319. [Google Scholar] [CrossRef] [PubMed]
- Stanley, P.M. Factors affecting the irreversible attachment of Pseudomonas aeruginosa to stainless steel. Can. J. Microbiol. 1983, 29, 1493–1499. [Google Scholar] [CrossRef] [PubMed]
- Danese, P.N.; Pratt, L.A.; Kolter, R. Exopolysaccharide production is required for development of Escherichia coli K-12 biofilm architecture. J. Bacteriol. 2000, 182, 3593–3596. [Google Scholar] [CrossRef] [PubMed]
- Otto, M. Staphylococcal infections: Mechanisms of biofilm maturation and detachment as critical determinants of pathogenicity. Annu. Rev. Med. 2013, 64, 175–188. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, L.; Levänen, E. Superhydrophobic surfaces for the reduction of bacterial adhesion. RSC Adv. 2013, 3, 12003–12020. [Google Scholar] [CrossRef]
- Mi, G.; Shi, D.; Wang, M.; Webster, T.J. Reducing bacterial infections and biofilm formation using nanoparticles and nanostructured antibacterial surfaces. Adv. Healthc. Mater. 2018, 7, 1800103. [Google Scholar] [CrossRef]
- Watson, G.S.; Green, D.W.; Schwarzkopf, L.; Li, X.; Cribb, B.W.; Myhra, S.; Watson, J.A. A gecko skin micro/nano structure—A low adhesion. Acta Biomater. 2015, 21, 109–122. [Google Scholar] [CrossRef]
- Hasan, J.; Crawford, R.J.; Ivanova, E.P. Antibacterial surfaces: The quest for a new generation of biomaterials. Trends Biotechnol. 2013, 31, 295–304. [Google Scholar] [CrossRef]
- Chen, S.; Li, L.; Zhao, C.; Zheng, J. Surface hydration: Principles and applications toward low-fouling/nonfouling biomaterials. Polym. Guildf. 2010, 51, 5283–5293. [Google Scholar] [CrossRef]
- Song, L.; Sun, L.; Zhao, J.; Wang, X.; Yin, J.; Luan, S.; Ming, W. Synergistic superhydrophobic and photodynamic cotton textiles with remarkable antibacterial activities. ACS Appl. Bio. Mater. 2019, 2, 2756–2765. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Z.; Guo, S.; Zhang, J.; Song, Y.; Dong, X.; Wang, X.; Yu, J. Antibacterial and anti-adhesive zeolite coatings on titanium alloy surface. Microporous Mesoporous Mater. 2011, 146, 216–222. [Google Scholar] [CrossRef]
- Lin, X.; Yang, M.; Jeong, H.; Chang, M.; Hong, J. Durable superhydrophilic coatings formed for anti-biofouling and oil-water separation. J. Memb. Sci. 2016, 506, 22–30. [Google Scholar] [CrossRef]
- Pinter, B.; Fievez, T.; Bickelhaupt, F.M.; Geerlings, P.; De Proft, F. On the origin of the steric effect. Phys. Chem. Chem. Phys. 2012, 14, 9846–9854. [Google Scholar] [CrossRef] [PubMed]
- Park, K.D. Bacterial adhesion on PEG modified surfaces. Biomaterials 1998, 19, 851–859. [Google Scholar] [CrossRef]
- Peng, L.; Chang, L.; Liu, X.; Lin, J.; Liu, H.; Han, B.; Wang, S. Antibacterial property of a polyethylene glycol-grafted dental material. ACS Appl. Mater. Interfaces 2017, 9, 17688–17692. [Google Scholar] [CrossRef]
- Perrin, F.X.; Nguyen, T.D.H.; Nguyen, D.L. Formation, structure and antibacterial activities of silazane networks grafted with poly (ethylene glycol) branches. Prog. Org. Coatings 2015, 88, 92–105. [Google Scholar] [CrossRef]
- Vasilev, K.; Griesser, S.S.; Griesser, H.J. Antibacterial surfaces and coatings produced by plasma techniques. Plasma Process. Polym. 2011, 8, 1010–1023. [Google Scholar] [CrossRef]
- Kingshott, P.; Wei, J.; Bagge-Ravn, D.; Gadegaard, N.; Gram, L. Covalent attachment of poly(ethylene glycol) to surfaces, critical for reducing bacterial adhesion. Langmuir 2003, 19, 6912–6921. [Google Scholar] [CrossRef]
- Minko, S. Grafting on Solid Surfaces: “Grafting to” and “Grafting from” Methods; Polymer Surfaces and Interfaces; Stamm, M., Ed.; Springer: Berlin, Germany, 2008; ISBN 9783540738640. [Google Scholar]
- Zdyrko, B.; Klep, V.; Li, X.; Kang, Q.; Minko, S.; Wen, X.; Luzinov, I. Polymer brushes as active nanolayers for tunable bacteria adhesion. Mater. Sci. Eng. C 2009, 29, 680–684. [Google Scholar] [CrossRef]
- Hadjesfandiari, N.; Yu, K.; Mei, Y.; Kizhakkedathu, J.N. Polymer brush-based approaches for the development of infection-resistant surfaces. J. Mater. Chem. B 2014, 2, 4968. [Google Scholar] [CrossRef]
- Roosjen, A.; Busscher, H.J.; Norde, W.; Van der Mei, H.C. Bacterial factors influencing adhesion of Pseudomonas aeruginosa strains to a poly (ethylene oxide) brush. Microbiology 2006, 152, 2673–2682. [Google Scholar] [CrossRef]
- He, T.; Jańczewski, D.; Jana, S.; Parthiban, A.; Guo, S.; Zhu, X.; Lee, S.S.C.; Parra-Velandia, F.J.; Teo, S.L.M.; Vancso, G.J. Efficient and robust coatings using poly(2-methyl-2-oxazoline) and its copolymers for marine and bacterial fouling prevention. J. Polym. Sci. Part. A Polym. Chem. 2016, 54, 275–283. [Google Scholar] [CrossRef]
- Herrwerth, S.; Eck, W.; Reinhardt, S.; Grunze, M. Factors that determine the protein resistance of oligoether self-assembled monolayers—Internal hydrophilicity, terminal hydrophilicity, and lateral packing density. J. Am. Chem. Soc. 2003, 125, 9359–9366. [Google Scholar] [CrossRef]
- Li, X.; Wu, B.; Chen, H.; Nan, K.; Jin, Y.; Wang, B. Recent developments in smart antibacterial surfaces to inhibit biofilm formation and bacterial infections. J. Mater. Chem. B 2018, 6, 4274–4292. [Google Scholar] [CrossRef]
- Weng, X.D.; Ji, Y.L.; Ma, R.; Zhao, F.Y.; An, Q.F.; Gao, C.J. Superhydrophilic and antibacterial zwitterionic polyamide nanofiltration membranes for antibiotics separation. J. Memb. Sci. 2016, 510, 122–130. [Google Scholar] [CrossRef]
- Lalani, R.; Liu, L. Electrospun zwitterionic poly (sulfobetaine methacrylate) for nonadherent, superabsorbent, and antimicrobial wound dressing applications. Biomacromolecules 2012, 13, 1853–1863. [Google Scholar] [CrossRef]
- Chen, S.; Yuan, L.; Li, Q.; Li, J.; Zhu, X.; Jiang, Y.; Sha, O.; Yang, X.; Xin, J.H.; Wang, J.; et al. Durable antibacterial and nonfouling cotton textiles with enhanced comfort via zwitterionic sulfopropylbetaine coating. Small 2016, 12, 3516–3521. [Google Scholar] [CrossRef]
- Timma, L.M.; Lewald, L.; Gier, F.; Homey, L.; Neyer, C.; Nickisch-Hartfiel, A.; Gutmann, J.S.; Oberthür, M. Nonfouling textiles with tunable antimicrobial activity based on a zwitterionic polyamine finish. RSC Adv. 2019, 9, 9783–9791. [Google Scholar] [CrossRef]
- He, H.; Xiao, Z.; Zhou, Y.; Chen, A.; Xuan, X.; Li, Y.; Guo, X.; Zheng, J.; Xiao, J.; Wu, J. Zwitterionic poly (sulfobetaine methacrylate) hydrogels with optimal mechanical properties for improving wound healing: In vivo. J. Mater. Chem. B 2019, 7, 1697–1707. [Google Scholar] [CrossRef]
- Cheng, G.; Li, G.; Xue, H.; Chen, S.; Bryers, J.D.; Jiang, S. Zwitterionic carboxybetaine polymer surfaces and their resistance to long-term biofilm formation. Biomaterials 2009, 30, 5234–5240. [Google Scholar] [CrossRef]
- Kang, S.M.; You, I.; Cho, W.K.; Shon, H.K.; Lee, T.G.; Choi, I.S.; Karp, J.M.; Lee, H. One-step modification of superhydrophobic surfaces by a mussel-inspired polymer coating. Angew. Chem. Int. Ed. 2010, 49, 9401–9404. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, B.; Liu, Y.; Suo, X.; Li, H.; Wu, S.; Zhang, B.; Liu, Y.; Suo, X.; Li, H. In fluence of surface topography on bacterial adhesion: A review. Biointerphases 2018, 13, 060801. [Google Scholar] [CrossRef]
- Morán, G.; Ramos-Chagas, G.; Hugelier, S.; Xie, X.; Boudjemaa, R.; Ruckebusch, C.; Sliwa, M.; Darmanin, T.; Gaucher, A.; Prim, D.; et al. Superhydrophobic polypyrene films to prevent: Staphylococcus aureus and Pseudomonas aeruginosa biofilm adhesion on surfaces: High efficiency deciphered by fluorescence microscopy. Photochem. Photobiol. Sci. 2018, 17, 1023–1035. [Google Scholar] [CrossRef]
- Boinovich, L.; Emelyanenko, A.M.; Pashinin, A.S. Analysis of long-term durability of superhydrophobic properties under continuous contact with water. ACS Appl. Mater. Interfaces 2010, 2, 1754–1758. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jung, U.T.; Kim, S.K.; Lee, J.H.; Choi, H.S.; Kim, C.S.; Jeong, M.Y. Nanostructured multifunctional surface with antireflective and antimicrobial characteristics. ACS Appl. Mater. Interfaces 2015, 7, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Privett, B.J.; Youn, J.; Hong, S.A.; Lee, J.; Han, J.; Shin, J.H.; Schoenfisch, M.H. Antibacterial fluorinated silica colloid superhydrophobic surfaces. Langmuir 2011, 27, 9597–9601. [Google Scholar] [CrossRef] [PubMed]
- Tiller, J.C.; Liao, C.J.; Lewis, K.; Klibanov, A.M. Designing surfaces that kill bacteria on contact. Proc. Natl. Acad. Sci. USA 2001, 98, 5981–5985. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Liu, S. Antibacterial surface design—Contact kill. Prog. Surf. Sci. 2016, 91, 136–153. [Google Scholar] [CrossRef]
- Jennings, M.C.; Minbiole, K.P.C.; Wuest, W.M. Quaternary ammonium compounds: An antimicrobial mainstay and platform for innovation to address bacterial resistance. ACS Infect. Dis. 2016, 1, 288–303. [Google Scholar] [CrossRef]
- Sugii, M.M.; de Souza Ferreira, F.A.; Müller, K.C.; Rodrigues Filho, U.P.; Baggio Aguiar, F.H. Quaternary ammonium compound derivatives for biomedical applications. Mater. Biomed. Eng. 2019, 153–175. [Google Scholar] [CrossRef]
- Makvandi, P.; Jamaledin, R.; Jabbari, M.; Nikfarjam, N.; Borzacchiello, A. Antibacterial quaternary ammonium compounds in dental materials: A systematic review. Dent. Mater. 2018, 34, 851–867. [Google Scholar] [CrossRef] [PubMed]
- Tiller, J.C.; Lee, S.B.; Lewis, K.; Klibanov, A.M. Polymer surfaces derivatized with poly (vinyl-N-hexylpyridinium) kill airborne and waterborne bacteria. Biotechnol. Bioeng. 2002, 79, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Nikawa, H.; Ishida, K.; Hamada, T.; Satoda, T.; Murayama, T.; Takemoto, T.; Tamamoto, M.; Tajima, H.; Shimoe, S.; Fujimoto, H.; et al. Immobilization of octadecyl ammonium chloride on the surface of titanium and its effect on microbial colonization in vitro. Dent. Mater. J. 2005, 24, 570–582. [Google Scholar] [CrossRef] [PubMed]
- Murata, H.; Koepsel, R.R.; Matyjaszewski, K.; Russell, A.J. Permanent, non-leaching antibacterial surfaces-2: How high density cationic surfaces kill bacterial cells. Biomaterials 2007, 28, 4870–4879. [Google Scholar] [CrossRef]
- Chiono, V.; Pulieri, E.; Vozzi, G.; Ciardelli, G.; Ahluwalia, A.; Giusti, P. Genipin-crosslinked chitosan/gelatin blends for biomedical applications. J. Mater. Sci. Mater. Med. 2008, 19, 889–898. [Google Scholar] [CrossRef]
- Tan, Y.; Leonhard, M.; Ma, S.; Moser, D.; Schneider-Stickler, B. Efficacy of carboxymethyl chitosan against Candida tropicalis and Staphylococcus epidermidis monomicrobial and polymicrobial biofilms. Int. J. Biol. Macromol. 2018, 110, 150–156. [Google Scholar] [CrossRef]
- Onaizi, S.A.; Leong, S.S.J. Tethering antimicrobial peptides: Current status and potential challenges. Biotechnol. Adv. 2011, 29, 67–74. [Google Scholar] [CrossRef]
- Thallinger, B.; Prasetyo, E.N.; Nyanhongo, G.S.; Guebitz, G.M. Antimicrobial enzymes: An emerging strategy to fight microbes and microbial biofilms. Biotechnol. J. 2013, 8, 97–109. [Google Scholar] [CrossRef]
- Meléndez-Ortiz, H.I.; Alvarez-Lorenzo, C.; Burillo, G.; Magariños, B.; Concheiro, A.; Bucio, E. Radiation-grafting of N-vinylimidazole onto silicone rubber for antimicrobial properties. Radiat. Phys. Chem. 2015, 110, 59–66. [Google Scholar] [CrossRef]
- Lim, K.; Chua, R.R.Y.; Saravanan, R.; Basu, A.; Mishra, B.; Tambyah, P.A.; Ho, B.; Leong, S.S.J. Immobilization studies of an engineered arginine-tryptophan-rich peptide on a silicone surface with antimicrobial and antibiofilm activity. ACS Appl. Mater. Interfaces 2013, 5, 6412–6422. [Google Scholar] [CrossRef]
- Thoma, L.M.; Boles, B.R.; Kuroda, K. Cationic methacrylate polymers as topical antimicrobial agents against staphylococcus aureus nasal colonization. Biomacromolecules 2014, 15, 2933–2943. [Google Scholar] [CrossRef] [PubMed]
- Microban Antimicrobial Medical Device Coatings. Available online: https://www.microban.com/blog/antimicrobial-medical-device-coatings (accessed on 3 February 2020).
- DESSAN Enzymatic. Available online: http://www.ab-laboratorios.com/en/cleaning-and-disinfection/1393-desinfectante-sanitario.html (accessed on 3 February 2020).
- Vasilev, K.; Cook, J.; Griesser, H.J. Antimicrobial surfaces for biomedical devices. Expert Rev. Med. Devices 2009, 6, 553–567. [Google Scholar] [CrossRef] [PubMed]
- Kufner, S.; Cassese, S.; Valeskini, M.; Neumann, F.J.; Schulz-Schüpke, S.; Hoppmann, P.; Fusaro, M.; Schunkert, H.; Laugwitz, K.L.; Kastrati, A.; et al. Long-term efficacy and safety of paclitaxel-eluting balloon for the treatment of drug-eluting stent restenosis: 3-year results of a randomized controlled trial. JACC Cardiovasc. Interv. 2015, 8, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Bucolo, C.; Gozzo, L.; Longo, L.; Mansueto, S.; Vitale, D.C.; Drago, F. Long-term efficacy and safety profile of multiple injections of intravitreal dexamethasone implant to manage diabetic macular edema: A systematic review of real-world studies. J. Pharmacol. Sci. 2018, 138, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Zander, Z.K.; Becker, M.L. Antimicrobial and antifouling strategies for polymeric medical devices. ACS Macro Lett. 2018, 7, 16–25. [Google Scholar] [CrossRef]
- Valverde, A.; Pérez-Álvarez, L.; Ruiz-Rubio, L.; Pacha Olivenza, M.A.; García Blanco, M.B.; Díaz-Fuentes, M.; Vilas-Vilela, J.L. Antibacterial hyaluronic acid/chitosan multilayers onto smooth and micropatterned titanium surfaces. Carbohydr. Polym. 2019, 207, 824–833. [Google Scholar] [CrossRef]
- Lv, H.; Chen, Z.; Yang, X.; Cen, L.; Zhang, X.; Gao, P. Layer-by-layer self-assembly of minocycline-loaded chitosan/alginate multilayer on titanium substrates to inhibit biofilm formation. J. Dent. 2014, 42, 1464–1472. [Google Scholar] [CrossRef]
- Choi, D.; Heo, J.; Park, J.H.; Jo, Y.; Jeong, H.; Chang, M.; Choi, J.; Hong, J. Nano-film coatings onto collagen hydrogels with desired drug release. J. Ind. Eng. Chem. 2016, 36, 326–333. [Google Scholar] [CrossRef]
- Schwartz, V.B.; Thétiot, F.; Ritz, S.; Pütz, S.; Choritz, L.; Lappas, A.; Förch, R.; Landfester, K.; Jonas, U. Antibacterial surface coatings from zinc oxide nanoparticles embedded in poly (N-isopropylacrylamide) hydrogel surface layers. Adv. Funct. Mater. 2012, 22, 2376–2386. [Google Scholar] [CrossRef]
- Noimark, S.; Weiner, J.; Noor, N.; Allan, E.; Williams, C.K.; Shaffer, M.S.P.; Parkin, I.P. Dual-mechanism antimicrobial polymer-ZnO nanoparticle and crystal violet-encapsulated silicone. Adv. Funct. Mater. 2015, 25, 1367–1373. [Google Scholar] [CrossRef]
- Lyutakov, O.; Hejna, O.; Solovyev, A.; Kalachyova, Y.; Svorcik, V. Polymethylmethacrylate doped with porphyrin and silver nanoparticles as light-activated antimicrobial material. RSC Adv. 2014, 4, 50624–50630. [Google Scholar] [CrossRef]
- Ahmad, Z.; Vargas-Reus, M.A.; Bakhshi, R.; Ryan, F.; Ren, G.G.; Oktar, F.; Allaker, R.P. Antimicrobial Properties of Electrically Formed Elastomeric Polyurethane-Copper Oxide Nanocomposites for Medical and Dental Applications, 1st ed.; Elsevier Inc.: Amsterdam, Holland, 2012; Volume 509, ISBN 9780123918581. [Google Scholar]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.A.; Hocking, D.M.; O’Connor, A.J. In situ formation of antimicrobial silver nanoparticles and the impregnation of hydrophobic polycaprolactone matrix for antimicrobial medical device applications. Mater. Sci. Eng. C 2015, 47, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Lyutakov, O.; Goncharova, I.; Rimpelova, S.; Kolarova, K.; Svanda, J.; Svorcik, V. Silver release and antimicrobial properties of PMMA films doped with silver ions, nano-particles and complexes. Mater. Sci. Eng. C 2015, 49, 534–540. [Google Scholar] [CrossRef]
- Tran, P.A.; Webster, T.J. Antimicrobial selenium nanoparticle coatings on polymeric medical devices. Nanotechnology 2013, 24. [Google Scholar] [CrossRef]
- Kelson, A.B.; Carnevali, M.; Truong-Le, V. Gallium-based anti-infectives: Targeting microbial iron-uptake mechanisms. Curr. Opin. Pharmacol. 2013, 13, 707–716. [Google Scholar] [CrossRef]
- Aquino, R.P.; Auriemma, G.; Mencherini, T.; Russo, P.; Porta, A.; Adami, R.; Liparoti, S.; Porta, G.D.; Reverchon, E.; Del Gaudio, P. Design and production of gentamicin/dextrans microparticles by supercritical assisted atomisation for the treatment of wound bacterial infections. Int. J. Pharm. 2013, 440, 188–194. [Google Scholar] [CrossRef]
- Ramos, A.I.; Braga, T.M.; Fernandes, J.A.; Silva, P.; Ribeiro-Claro, P.J.; Almeida Paz, F.A.; de Fátima Silva Lopes, M.; Braga, S.S. Analysis of the microcrystalline inclusion compounds of triclosan with β-cyclodextrin and its tris-O-methylated derivative. J. Pharm. Biomed. Anal. 2013, 80, 34–43. [Google Scholar] [CrossRef]
- Pérez-Álvarez, L.; Ruiz-Rubio, L.; Lizundia, E.; Hernáez, E.; León, L.M.; Vilas-Vilela, J.L. Active release coating of multilayer assembled branched and ionic β-cyclodextrins onto poly(ethylene terephthalate). Carbohydr. Polym. 2017, 174, 65–71. [Google Scholar] [CrossRef]
- Pérez-Álvarez, L.; Matas, J.; Gómez-Galván, F.; Ruiz-Rubio, L.; León, L.M.; Vilas-Vilela, J.L. Branched and ionic β-Cyclodextrins multilayer assembling onto polyacrylonitrile membranes for removal and controlled release of triclosan. Carbohydr. Polym. 2017, 156, 143–151. [Google Scholar] [CrossRef]
- Rodriguez, L.C.; Palmer, K.; Montagner, F.; Rodrigues, D.C. A novel chlorhexidine-releasing composite bone cement: Characterization of antimicrobial effectiveness and cement strength. J. Bioact. Compat. Polym. 2015, 30, 34–47. [Google Scholar] [CrossRef]
- Baveja, J.K.; Willcox, M.D.P.; Hume, E.B.H.; Kumar, N.; Odell, R.; Poole-Warren, L.A. Furanones as potential anti-bacterial coatings on biomaterials. Biomaterials 2004, 25, 5003–5012. [Google Scholar] [CrossRef] [PubMed]
- Slomberg, D.L.; Lu, Y.; Broadnax, A.D.; Hunter, R.A.; Carpenter, A.W.; Schoenfisch, M.H. Role of size and shape on biofilm eradication for nitric oxide-releasing silica nanoparticles. ACS Appl. Mater. Interfaces 2013, 5, 9322–9329. [Google Scholar] [CrossRef] [PubMed]
- Lawson, M.C.; Shoemaker, R.; Hoth, K.B.; Bowman, C.N.; Anseth, K.S. Polymerizable vancomycin derivatives for bactericidal biomaterial surface modification: Structure-function evaluation. Biomacromolecules 2009, 10, 2221–2234. [Google Scholar] [CrossRef]
- Stigter, M.; De Groot, K.; Layrolle, P. Incorporation of tobramycin into biomimetic hydroxyapatite coating on titanium. Biomaterials 2002, 23, 4143–4153. [Google Scholar] [CrossRef]
- Kim, H.W.; Knowles, J.C.; Kim, H.E. Hydroxyapatite/poly (ε-caprolactone) composite coatings on hydroxyapatite porous bone scaffold for drug delivery. Biomaterials 2004, 25, 1279–1287. [Google Scholar] [CrossRef]
- Baier, G.; Cavallaro, A.; Friedemann, K.; Müller, B.; Glasser, G.; Vasilev, K.; Landfester, K. Enzymatic degradation of poly (l-lactide) nanoparticles followed by the release of octenidine and their bactericidal effects. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 131–139. [Google Scholar] [CrossRef]
- Carmona-Ribeiro, A.M.; de Melo Carrasco, L.D. Cationic antimicrobial polymers and their assemblies. Int. J. Mol. Sci. 2013, 14, 9906–9946. [Google Scholar] [CrossRef]
- Ivanova, K.; Fernandes, M.M.; Mendoza, E.; Tzanov, T. Enzyme multilayer coatings inhibit Pseudomonas aeruginosa biofilm formation on urinary catheters. Appl. Microbiol. Biotechnol. 2015, 99, 4373–4385. [Google Scholar] [CrossRef]
- Zhou, B.; Li, Y.; Deng, H.; Hu, Y.; Li, B. Antibacterial multilayer films fabricated by layer-by-layer immobilizing lysozyme and gold nanoparticles on nanofibers. Colloids Surfaces B Biointerfaces 2014, 116, 432–438. [Google Scholar] [CrossRef]
- Shirai, T.; Shimizu, T.; Ohtani, K.; Zen, Y.; Takaya, M.; Tsuchiya, H. Antibacterial iodine-supported titanium implants. Acta Biomater. 2011, 7, 1928–1933. [Google Scholar] [CrossRef] [PubMed]
- Campoccia, D.; Montanaro, L.; Arciola, C.R. A review of the biomaterials technologies for infection-resistant surfaces. Biomaterials 2013, 34, 8533–8554. [Google Scholar] [CrossRef] [PubMed]
- InnovoSILTM Antimicrobial Products. Available online: https://www.innovotech.ca/products/innovosil-antimicrobial-products/ (accessed on 3 February 2020).
- Surmodics Drug Delivery Coatings. Available online: http://www.surmodics.com/medical-device/coating-technologies/drug-delivery-coatings-overview/ (accessed on 3 February 2020).
- Medtronic TYRX Absorbable Antibacterial Envelope. Available online: https://www.medtronic.com/us-en/healthcare-professionals/products/cardiac-rhythm/infection-control/tyrx-antibacterial-envelope.html (accessed on 3 February 2020).
- Ostrikov, K.; MacGregor-Ramiasa, M.; Cavallaro, A.; Vasilev, K. Bactericidal effects of plasma-modified surface chemistry of silicon nanograss. J. Phys. D Appl. Phys. 2016, 49. [Google Scholar] [CrossRef]
- Dickson, M.N.; Liang, E.I.; Rodriguez, L.A.; Vollereaux, N.; Yee, A.F. Nanopatterned polymer surfaces with bactericidal properties. Biointerphases 2015, 10, 021010. [Google Scholar] [CrossRef]
- Rodríguez-Hernández, J. Polymers against microorganisms: On the race to efficient antimicrobial materials. Polym. Against Microorg. Race Effic. Antimicrob. Mater. 2016, 1–278. [Google Scholar] [CrossRef]
- Modaresifar, K.; Azizian, S.; Ganjian, M.; Fratila-Apachitei, L.E.; Zadpoor, A.A. Bactericidal effects of nanopatterns: A systematic review. Acta Biomater. 2019, 83, 29–36. [Google Scholar] [CrossRef]
- Bhadra, C.M.; Werner, M.; Baulin, V.A.; Truong, V.K.; Kobaisi, M.A.; Nguyen, S.H.; Balcytis, A.; Juodkazis, S.; Wang, J.Y.; Mainwaring, D.E.; et al. Subtle variations in surface properties of black silicon surfaces influence the degree of bactericidal efficiency. Nano Micro Lett. 2018, 10, 36. [Google Scholar] [CrossRef]
- Hazell, G.; Fisher, L.E.; Murray, W.A.; Nobbs, A.H.; Su, B. Bioinspired bactericidal surfaces with polymer nanocone arrays. J. Colloid Interface Sci. 2018, 528, 389–399. [Google Scholar] [CrossRef]
- Nowlin, K.; Boseman, A.; Covell, A.; LaJeunesse, D. Adhesion-dependent rupturing of Saccharomyces cerevisiae on biological antimicrobial nanostructured surfaces. J. R. Soc. Interface 2014, 12. [Google Scholar] [CrossRef]
- Ivanova, E.P.; Hasan, J.; Webb, H.K.; Truong, V.K.; Watson, G.S.; Watson, J.A.; Baulin, V.A.; Pogodin, S.; Wang, J.Y.; Tobin, M.J.; et al. Natural bactericidal surfaces: Mechanical rupture of pseudomonas aeruginosa cells by cicada wings. Small 2012, 8, 2489–2494. [Google Scholar] [CrossRef]

| Implant | Material | Reference |
|---|---|---|
| Heart valve | Titanium and its alloys, graphite, polyester, cobalt-chromium alloys | [5] |
| Urinary catheter | Latex, silicone, nylon, polyethylene terephthalate | [6] |
| Peritoneal catheter | Silicone, polyurethane | [7] |
| Stents | Cobalt-chromium, titanium and magnesium alloys, diamondlike carbon | [8] |
| Dental | Titanium, zirconia and cobalt-chromium alloys, alumina, bioglass | [9] |
| Contact and intraocular lenses | Polymethylmethacrylate, polyhydroethylmethacrylate, silicone, acrylic | [10,11] |
| Cochlear | Titanium, platinum, silicone, polytetrafluoroethylene | [12] |
| Knee and hip | Stainless steel, titanium and cobalt-chromium alloys, polyethylene, ceramics | [13,14] |
| Released Antimicrobial Substance | Release Systems | References |
|---|---|---|
| Zinc oxide nanoparticles (ZnO NP) | Poly(N-isopropylacrylamide) | [93] |
| Silicone | [94] | |
| Silver nanoparticles (AgNP) | Poly(methyl methacrylate) | [95] |
| Copper oxide nanoparticles (CuO NP) | Polyurethane | [96] |
| Zinc | Polysaccharides | [97] |
| Silver | Polycaprolactone | [98] |
| Poly(methyl methacrylate) | [99] | |
| Copper | Polyssacharides | [97] |
| Selenium | Polyvinyl chloride (PVC), silicone, polyurethane | [100] |
| Galium | - | [101] |
| Gentamicin | Dextrans | [102] |
| Triclosan | Cyclodextrin | [103] |
| Hyaluronic acid and chitosan | [90] | |
| Poly(ethylene terephthalate) | [104] | |
| Polyacrilonitrile | [105] | |
| Chlorhexidrine | Acrylic bone cements | [106] |
| Furanones | Polyethylene, polypropylene | [107] |
| Nitrogen oxide | Silica nanoparticles | [108] |
| Vancomyzin | Poly(ethylene glycol) (PEG)-acrylate | [109] |
| Tobramycin | Hydroxyapatite | [110] |
| Tetracycline hydrochloride | Hydroxyapatite and polycaprolactone | [111] |
| Octenidine | Poly(L-lactide) | [112] |
| Cationic surfactants | Poly(methyl methacrylate) | [113] |
| Acylase enzyme | Polydimethyl/vinylmethyl-siloxane | [114] |
| Lysozyme enzyme | Cellulose | [115] |
| Halogens | Titanium | [116] |
| Antibiotics | Penicillins Quinilones Aminoglycosides | [117] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olmo, J.A.-D.; Ruiz-Rubio, L.; Pérez-Alvarez, L.; Sáez-Martínez, V.; Vilas-Vilela, J.L. Antibacterial Coatings for Improving the Performance of Biomaterials. Coatings 2020, 10, 139. https://doi.org/10.3390/coatings10020139
Olmo JA-D, Ruiz-Rubio L, Pérez-Alvarez L, Sáez-Martínez V, Vilas-Vilela JL. Antibacterial Coatings for Improving the Performance of Biomaterials. Coatings. 2020; 10(2):139. https://doi.org/10.3390/coatings10020139
Chicago/Turabian StyleOlmo, Jon Andrade-Del, Leire Ruiz-Rubio, Leyre Pérez-Alvarez, Virginia Sáez-Martínez, and José Luis Vilas-Vilela. 2020. "Antibacterial Coatings for Improving the Performance of Biomaterials" Coatings 10, no. 2: 139. https://doi.org/10.3390/coatings10020139
APA StyleOlmo, J. A.-D., Ruiz-Rubio, L., Pérez-Alvarez, L., Sáez-Martínez, V., & Vilas-Vilela, J. L. (2020). Antibacterial Coatings for Improving the Performance of Biomaterials. Coatings, 10(2), 139. https://doi.org/10.3390/coatings10020139








