Evaluation of Intravenous Fosfomycin Disodium Dosing Regimens in Critically Ill Patients for Treatment of Carbapenem-Resistant Enterobacterales Infections Using Monte Carlo Simulation
Abstract
1. Introduction
2. Results
2.1. MIC Distributions
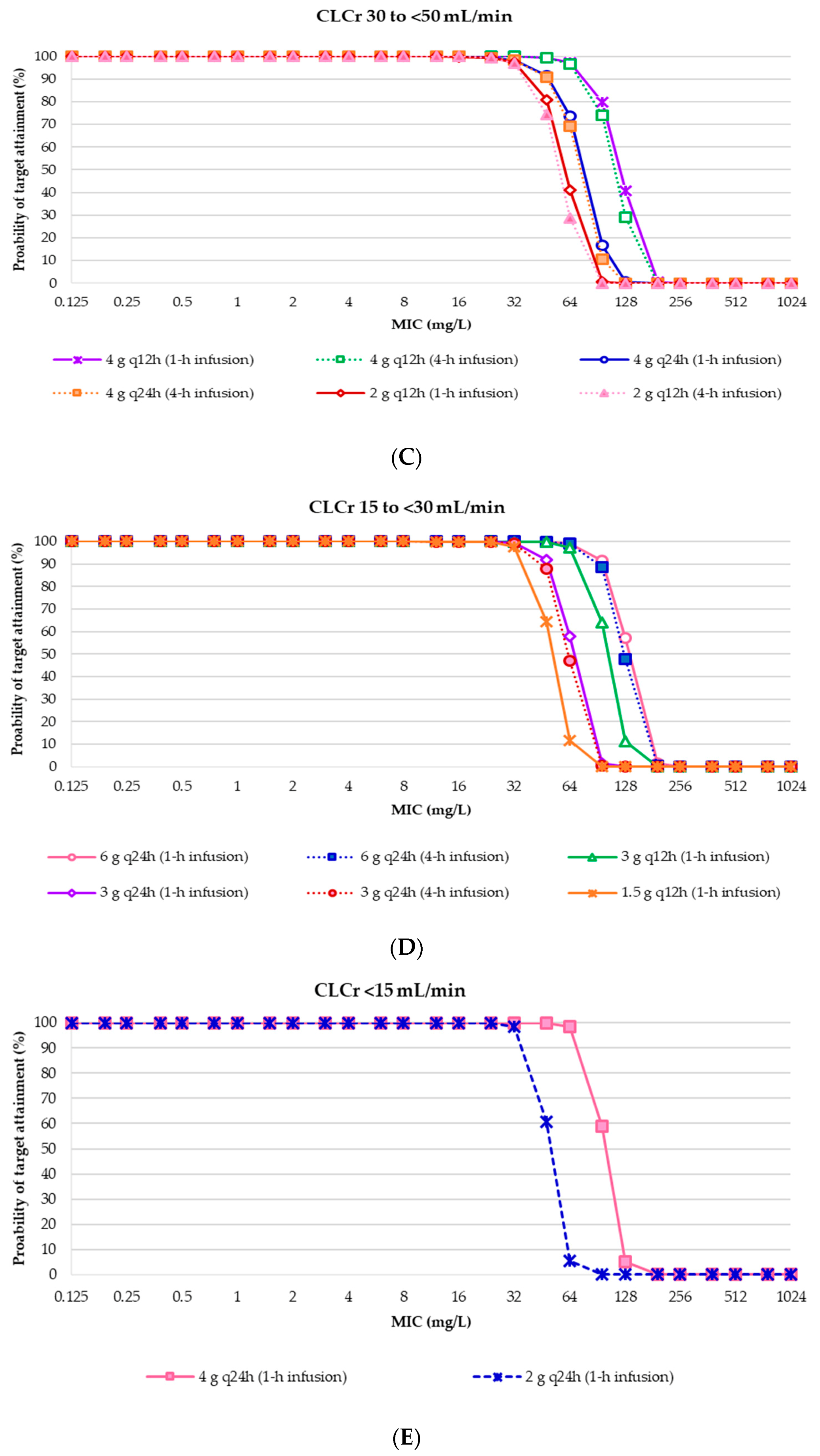
2.2. Pharmacokinetic/Pharmacodynamic Simulations
3. Discussion
4. Materials and Methods
4.1. Pharmacokinetic Parameters
4.2. Microbiology
4.3. Pharmacodynamic Parameters
4.4. Intravenous Fosfomycin Disodium Dosing Regimens
4.5. Monte Carlo Simulation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodríguez-Baño, J.; Gutiérrez-Gutiérrez, B.; Machuca, I.; Pascual, Á. Treatment of Infections Caused by Extended-Spectrum-Beta-Lactamase-, AmpC-, and Carbapenemase-ProducingEnterobacteriaceae. Clin. Microbiol. Rev. 2018, 31, e00079-17. [Google Scholar] [CrossRef]
- Sheu, C.-C.; Chang, Y.-T.; Lin, S.-Y.; Chen, Y.-H.; Hsueh, P.-R. Infections Caused by Carbapenem-Resistant Enterobacteriaceae: An Update on Therapeutic Options. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Dimopoulos, G.; Koulenti, D.; Parker, S.L.; A Roberts, J.; Arvaniti, K.; Poulakou, G. Intravenous fosfomycin for the treatment of multidrug-resistant pathogens: What is the evidence on dosing regimens? Expert Rev. Anti Infect. Ther. 2019, 17, 201–210. [Google Scholar] [CrossRef]
- Candel, F.J.; David, M.M.; Barberán, J. New perspctives for reassessing fosfomycin: Applicability in current clinical practice. Rev. Esp. Quimioter. 2019, 32, 1–7. [Google Scholar]
- European Committee on Antimicrobial Susceptibility Testing. Fosfomycin: Rationale for the Clinical Breakpoints, Version 1.0. 2013. Available online: http://www.eucast.org/documents/rd (accessed on 24 July 2019).
- Fomicyt 40 mg/ml Powder for Solution for Infusion. Available online: https://www.medicines.org.uk/emc/medicine/28971 (accessed on 20 October 2019).
- Burgos, R.M.; A Rodvold, K. ZTI-01 (fosfomycin for injection) in the treatment of hospitalized patients with complicated urinary tract infections. Futur. Microbiol. 2019, 14, 461–475. [Google Scholar] [CrossRef]
- Trinh, T.D.; Smith, J.R.; Rybak, M.J. Parenteral Fosfomycin for the Treatment of Multidrug Resistant Bacterial Infections: The Rise of the Epoxide. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2019, 39, 1077–1094. [Google Scholar] [CrossRef]
- Merino-Bohorquez, V.; Pérez, F.D.; Sojo, J.; Morales, I.; Lupión, C.; Martín, D.; Camean, M.; Hope, W.; Álvaro, P.; Rodríguez-Baño, J. Population pharmacokinetics and pharmacodynamics of fosfomycin in non–critically ill patients with bacteremic urinary infection caused by multidrug-resistant Escherichia coli. Clin. Microbiol. Infect. 2018, 24, 1177–1183. [Google Scholar] [CrossRef]
- Trang, M.; Ellis-Grosse, E.J.; Eckburg, P.B.; Skarinsky, D.; Bhavnani, S.M.; Rubino, C.M. Population Pharmacokinetic (PPK) Analysis of ZTI-01 (Fosfomycin for Injection) Using Data from Healthy Subjects and Patients with Complicated Urinary Tract Infections (cUTI). Open Forum. Infect. Dis. 2017, 4. [Google Scholar] [CrossRef][Green Version]
- Parker, S.L.; Frantzeskaki, F.; Wallis, S.C.; Diakaki, C.; Giamarellou, H.; Koulenti, D.; Karaiskos, I.; Lipman, J.; Dimopoulos, G.; A Roberts, J. Population Pharmacokinetics of Fosfomycin in Critically Ill Patients. Antimicrob. Agents Chemother. 2015, 59, 6471–6476. [Google Scholar] [CrossRef]
- Florent, A.; Chichmanian, R.-M.; Cua, E.; Pulcini, C. Adverse events associated with intravenous fosfomycin. Int. J. Antimicrob. Agents 2011, 37, 82–83. [Google Scholar] [CrossRef]
- Albur, M.S.; Noel, A.; Bowker, K.; MacGowan, A. The combination of colistin and fosfomycin is synergistic against NDM-1-producing Enterobacteriaceae in in vitro pharmacokinetic/pharmacodynamic model experiments. Int. J. Antimicrob. Agents 2015, 46, 560–567. [Google Scholar] [CrossRef]
- Lepak, A.J.; Zhao, M.; Vanscoy, B.; Taylor, D.S.; Ellis-Grosse, E.; Ambrose, P.G.; Andes, D.R. In Vivo Pharmacokinetics and Pharmacodynamics of ZTI-01 (Fosfomycin for Injection) in the Neutropenic Murine Thigh Infection Model against Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef]
- Wang, J.; He, J.-T.; Bai, Y.; Wang, R.; Cai, Y. Synergistic Activity of Colistin/Fosfomycin Combination against Carbapenemase-Producing Klebsiella pneumoniae in an In Vitro Pharmacokinetic/Pharmacodynamic Model. Biol. Med. Res. Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Asin-Prieto, E.; Rodríguez-Gascón, A.; Isla, A. Applications of the pharmacokinetic/pharmacodynamic (PK/PD) analysis of antimicrobial agents. J. Infect. Chemother. 2015, 21, 319–329. [Google Scholar] [CrossRef]
- Trang, M.; Dudley, M.N.; Bhavnani, S.M. Use of Monte Carlo simulation and considerations for PK-PD targets to support antibacterial dose selection. Curr. Opin. Pharmacol. 2017, 36, 107–113. [Google Scholar] [CrossRef]
- EUCAST. Breakpoint Tables for Interpretation of MIC’s and Zone Diameters. Version 10.0. January 2020. Available online: https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_10.0_Breakpoint_Tables.pdf (accessed on 9 January 2020).
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing: 30th Information Supplement; M100Ed30E; Clinical Laboratory Standards Institute: Wayne, PA, USA, 2020; Available online: http://em100.edaptivedocs.net/GetDoc.aspx?doc=CLSI%20M100%20ED30:2020&format=SPDF (accessed on 8 January 2020).
- Logan, L.K.; Weinstein, R.A. The Epidemiology of Carbapenem-Resistant Enterobacteriaceae: The Impact and Evolution of a Global Menace. J. Infect. Dis. 2017, 215, S28–S36. [Google Scholar] [CrossRef]
- Díez-Aguilar, M.; Canton, R. New microbiological aspects of fosfomycin. Rev. Esp. Quim. Publ. Soc. Esp. Quim. 2019, 32, 8–18. [Google Scholar]
- Yang, T.-Y.; Lu, P.-L.; Tseng, S.-P. Update on fosfomycin-modified genes in Enterobacteriaceae. J. Microbiol. Immunol. Infect. 2019, 52, 9–21. [Google Scholar] [CrossRef]
- Falagas, M.E.; Vouloumanou, E.K.; Samonis, G.; Vardakas, K.Z. Fosfomycin. Clin. Microbiol. Rev. 2016, 29, 321–347. [Google Scholar] [CrossRef]
- National Antimicrobial Resistance Surveillance Center, Thailand. Antibiograms. Available online: http://narst.dmsc.moph.go.th/antibiograms.html (accessed on 21 August 2019).
- Camarlinghi, G.; Parisio, E.M.; Antonelli, A.; Nardone, M.; Coppi, M.; Giani, T.; Mattei, R.; Rossolini, G.M. Discrepancies in fosfomycin susceptibility testing of KPC-producing Klebsiella pneumoniae with various commercial methods. Diagn. Microbiol. Infect. Dis. 2019, 93, 74–76. [Google Scholar] [CrossRef]
- Chueansuwan, W.; Chatsuwan, T.; Vanichanan, J.; Jutivorakool, K. 1546. Incidence of Carbapenemase-Producing Klebsiella pneumoniae Colonization in Hematopoietic Stem Cell Transplant Recipients in King Chulalongkorn Memorial Hospital (KCMH), Thailand. Open Forum. Infect. Dis. 2018, 5, S480. [Google Scholar] [CrossRef][Green Version]
- Noopetch, P.; Plongla, R.; Suwanpimolkul, G.; Chatsuwan, T. 2064. Rapid Detection of Carbapenmase-Producing Klebsiella pneumoniae (CPK) Directly from Respiratory Secretion and Clinical Characteristics of Patients with CPK from a 1,200-Bed Tertiary Care Hospital in Thailand. Open Forum. Infect. Dis. 2018, 5, S602–S603. [Google Scholar] [CrossRef]
- Tangkosku, T.; Seenama, C.; Pathi, N.; Chayakulkeeree, M.; Thamlikitkul, V. Antibiotic susceptibility and carbapenemase genes in carbapenem resistant Enterobacteriaceae (CRE) isolated from hospitalized patients at Siriraj Hospital. J. Infect. Dis. Antimicrob. Agents 2015, 32, 85. [Google Scholar]
- Laolerd, W.; Akeda, Y.; Preeyanon, L.; Ratthawongjirakul, P.; Santanirand, P. Carbapenemase-Producing Carbapenem-Resistant Enterobacteriaceae from Bangkok, Thailand, and Their Detection by the Carba NP and Modified Carbapenem Inactivation Method Tests. Microb. Drug Resist. 2018, 24, 1006–1011. [Google Scholar] [CrossRef]
- Kaewnirat, K.; Chukamnerd, A.; Chusri, S.; Pomwised, R.; Phoo, M.T.P.; Terbtothakun, P.; Hortiwakul, T.; Charoenmak, B.B. The occurrence of carbapenem-resistant Klebsiella pneumoniae (CRKP) isolated from patients in the medicine ward, Songklanagarind Hospital. J. Infect. Dis Antimicrob. Agents 2019, 36, 153. [Google Scholar]
- Albiero, J.; Sy, S.K.B.; Mazucheli, J.; Caparroz-Assef, S.M.; Costa, B.B.; Alves, J.L.B.; Gales, A.C.; Tognim, M.C.B. Pharmacodynamic Evaluation of the Potential Clinical Utility of Fosfomycin and Meropenem in Combination Therapy against KPC-2-Producing Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2016, 60, 4128–4139. [Google Scholar] [CrossRef]
- Rodríguez-Gascón, A.; Canut-Blasco, A. Deciphering pharmacokinetics and pharmacodynamics of fosfomycin. Rev. Esp. Quim. Publ. Soc. Esp. Quim. 2019, 32, 19–24. [Google Scholar]
- Bhavnani, S.M.; Trang, M.; Rubino, C.M.; Lepak, A.J.; Andes, D.R.; Flamm, R.K.; Sweeney, K.; Ellis-Grosse, E.J.; Ambrose, P.G. Pharmacokinetics-Pharmacodynamics (PK-PD) Target Attainment Analyses to Support ZTI-01 (Fosfomycin for Injection) Dose Selection for Patients with Complicated Urinary Tract Infections (cUTI); ASM Microbe: New Orleans, LA, USA, 2017. [Google Scholar]
- Michalopoulos, A.; Virtzili, S.; Rafailidis, P.; Chalevelakis, G.; Damala, M.; Falagas, M.E. Intravenous fosfomycin for the treatment of nosocomial infections caused by carbapenem-resistant Klebsiella pneumoniae in critically ill patients: A prospective evaluation. Clin. Microbiol. Infect. 2010, 16, 184–186. [Google Scholar] [CrossRef]
- Pontikis, K.; Karaiskos, I.; Bastani, S.; Dimopoulos, G.; Kalogirou, M.; Katsiari, M.; Oikonomou, A.; Poulakou, G.; Roilides, E.; Giamarellou, H. Outcomes of critically ill intensive care unit patients treated with fosfomycin for infections due to pandrug-resistant and extensively drug-resistant carbapenemase-producing Gram-negative bacteria. Int. J. Antimicrob. Agents 2014, 43, 52–59. [Google Scholar] [CrossRef]
- Kaye, K.S.; Rice, L.B.; Dane, A.L.; Stus, V.; Sagan, O.; Fedosiuk, E.; Das, A.F.; Skarinsky, D.; Eckburg, P.B.; Ellis-Grosse, E.J. Fosfomycin for Injection (ZTI-01) Versus Piperacillin-tazobactam for the Treatment of Complicated Urinary Tract Infection Including Acute Pyelonephritis: ZEUS, A Phase 2/3 Randomized Trial. Clin. Infect. Dis. 2019, 69, 2045–2056. [Google Scholar] [CrossRef]
- Eckburg, P.B.; Skarinsky, D.; Das, A.; Ellis-Grosse, E.J. Phenotypic Antibiotic Resistance in ZEUS: A Multi-center, Randomized, Double-Blind Phase 2/3 Study of ZTI-01 vs. Piperacillin-Tazobactam (P-T) in the Treatment of Patients with Complicated Urinary Tract Infections (cUTI) Including Acute Pyelonephritis (AP). Open Forum. Infect. Dis. 2017, 4. [Google Scholar] [CrossRef][Green Version]
- United States Centers for Disease Control and Prevention. Facility Guidance for Control of Carbapenem-Resistant Enterobacteriaceae (CRE): November 2015 Update—CRE Toolkit. Available online: https://www.cdc.gov/hai/organisms/cre/cre-toolkit/index.html (accessed on 7 August 2019).



| Carbapenem-Resistant Enterobacterales Isolates | Number of Isolates | Fosfomycin MICs (mg/L) | % of Isolates | |||
|---|---|---|---|---|---|---|
| CLSI a | EUCAST b | |||||
| MIC50 | MIC90 | MICs Range | S/I/R | S/R | ||
| Klebsiella pneumoniae | 116 | 64 | >1024 | 0.38 to >1024 | - | 40.52/59.48 |
| Escherichia coli | 12 | NA c | NA c | 1 to 16 | 100/0/0 | 100/0 |
| Enterobacter cloacae | 1 | NA c | NA c | 12 | - | 100/0 |
| All isolates | 129 | 48 | >1024 | 0.38 to >1024 | - | 46.51/53.49 |
| CLCr (mL/min) a | Daily Doses Suggestion | Dosage Adjustments | |
|---|---|---|---|
| CLSI Breakpoints b | EUCAST Breakpoints c | ||
| ≥80 | 16 g per day (in 2 or 4 divided doses) (1 h or 4 h infusion) | 8 g per day (in 2 divided doses) (1 h or 4 h infusion) | Initial dosage |
| 50 to <80 | 12 g per day (in 2 to 3 divided doses) (1 h or 4 h infusion) | 6 g per day (in 1 to 3 divided doses) (1 h or 4 h infusion) | Reduce maintenance dosage by 25% |
| 30 to <50 | 8 g per day (in 2 divided doses) (1 h or 4 h infusion) | 4 g per day (in 1 to 2 divided doses) (1 h or 4 h infusion) | Reduce maintenance dosage by 50% |
| 15 to <30 | 6 g per day (in 1 to 2 divided doses) (1 h or 4 h infusion) | 3 g per day (in 1 to 2 divided doses) (1 h or 4 h infusion) | Reduce maintenance dosage by 62.5% |
| <15 | 4 g per day (in 1 divided doses) (1 h infusion) | 2 g per day (in 1 divided doses) (1 h infusion) | Reduce maintenance dosage by 75% |
| Dosing Regimens for Monte Carlo Simulation | CLCr (mL/min) a | |||||
|---|---|---|---|---|---|---|
| Daily Doses | Dosing Regimens | ≥80 | 50 to <80 | 30 to <50 | 15 to <30 | <15 |
| 24 g/day | 8 g q8h (1 h infusion) | / b | / c | |||
| 8 g q8h (4 h infusion) | / c | / c | ||||
| 18 g/day | 6 g q8h (1 h infusion) | / b | / c | |||
| 6 g q8h (4 h infusion) | / c | |||||
| 16 g/day | 8 g q12h (1 h infusion) | / b | / b | |||
| 8 g q12h (4 h infusion) | / c | / c | ||||
| 4 g q6h (1 h infusion) | / b | |||||
| 4 g q6h (4 h infusion) | / c | |||||
| 12 g/day | 6 g q12h (1 h infusion) | / b | / c | / b | ||
| 6 g q12h (4 h infusion) | / c | / c | ||||
| 4 g q8h (1 h infusion) | / b | / c | / b | / b | ||
| 4 g q8h (4 h infusion) | / c | / c | / c | |||
| 8 g/day | 4 g q12h (1 h infusion) | / c | / c | / b | / b | |
| 4 g q12h (4 h infusion) | / c | / c | ||||
| 6 g/day | 6 g q24h (1 h infusion) | / c | /c | / c | ||
| 6 g q24h (4 h infusion) | / c | / c | / c | / c | ||
| 3 g q12h (1 h infusion) | / c | / b | / c | |||
| 3 g q12h (4 h infusion) | / c | |||||
| 2 g q8h (1 h infusion) | / c | |||||
| 2 g q8h (4 h infusion) | / c | |||||
| 4 g/day | 4 g q24h (1 h infusion) | / c | / b | |||
| 4 g q24h (4 h infusion) | / c | / c | ||||
| 2 g q12h (1 h infusion) | / c | |||||
| 2 g q12h (4 h infusion) | / c | |||||
| 3 g/day | 3 g q24h (1 h infusion) | / c | / b | |||
| 3 g q24h (4 h infusion) | / c | |||||
| 1.5 g q12h (1 h infusion) | / c | |||||
| 2 g/day | 2 g q24h (1 h infusion) | / c | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leelawattanachai, P.; Wattanavijitkul, T.; Paiboonvong, T.; Plongla, R.; Chatsuwan, T.; Usayaporn, S.; Nosoongnoen, W.; Montakantikul, P. Evaluation of Intravenous Fosfomycin Disodium Dosing Regimens in Critically Ill Patients for Treatment of Carbapenem-Resistant Enterobacterales Infections Using Monte Carlo Simulation. Antibiotics 2020, 9, 615. https://doi.org/10.3390/antibiotics9090615
Leelawattanachai P, Wattanavijitkul T, Paiboonvong T, Plongla R, Chatsuwan T, Usayaporn S, Nosoongnoen W, Montakantikul P. Evaluation of Intravenous Fosfomycin Disodium Dosing Regimens in Critically Ill Patients for Treatment of Carbapenem-Resistant Enterobacterales Infections Using Monte Carlo Simulation. Antibiotics. 2020; 9(9):615. https://doi.org/10.3390/antibiotics9090615
Chicago/Turabian StyleLeelawattanachai, Pannee, Thitima Wattanavijitkul, Taniya Paiboonvong, Rongpong Plongla, Tanittha Chatsuwan, Sang Usayaporn, Wichit Nosoongnoen, and Preecha Montakantikul. 2020. "Evaluation of Intravenous Fosfomycin Disodium Dosing Regimens in Critically Ill Patients for Treatment of Carbapenem-Resistant Enterobacterales Infections Using Monte Carlo Simulation" Antibiotics 9, no. 9: 615. https://doi.org/10.3390/antibiotics9090615
APA StyleLeelawattanachai, P., Wattanavijitkul, T., Paiboonvong, T., Plongla, R., Chatsuwan, T., Usayaporn, S., Nosoongnoen, W., & Montakantikul, P. (2020). Evaluation of Intravenous Fosfomycin Disodium Dosing Regimens in Critically Ill Patients for Treatment of Carbapenem-Resistant Enterobacterales Infections Using Monte Carlo Simulation. Antibiotics, 9(9), 615. https://doi.org/10.3390/antibiotics9090615





