Sonochemical Synthesis of 2’-Hydroxy-Chalcone Derivatives with Potential Anti-Oomycete Activity
Abstract
:1. Introduction
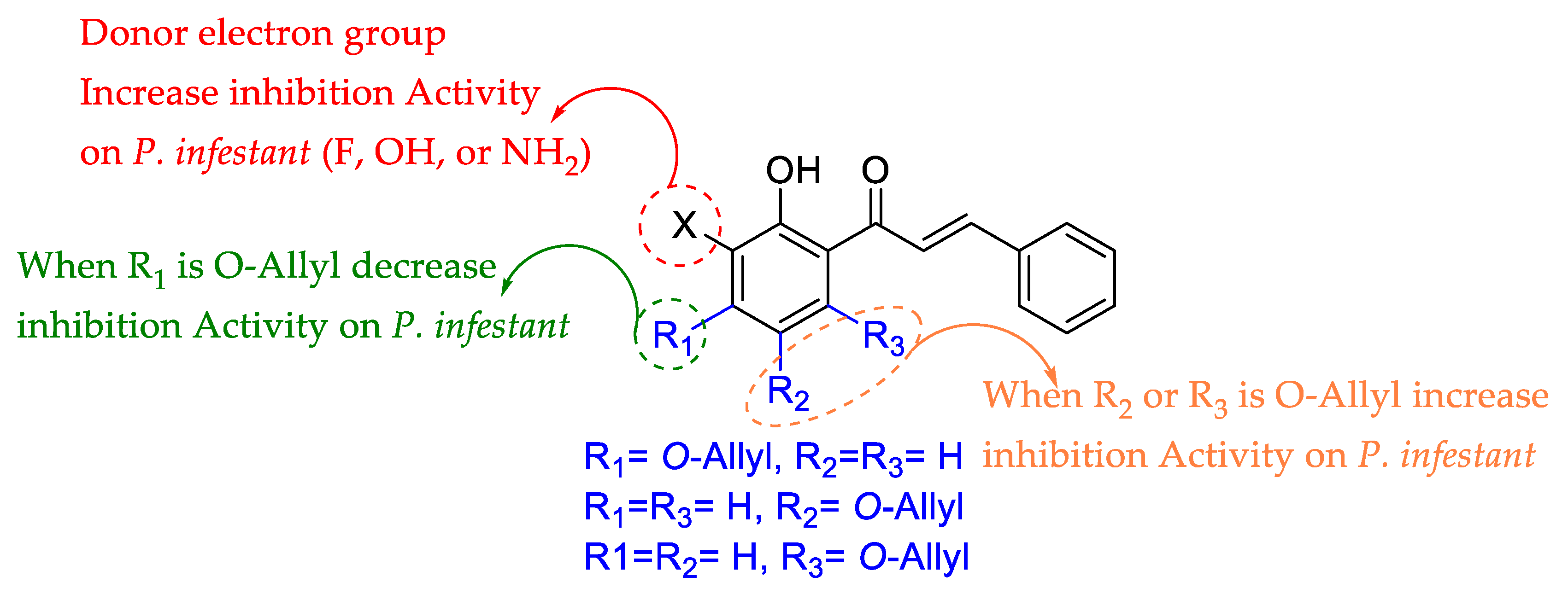
2. Results and Discussion
N = 12; r = 0.925; r2 = 0.856; SD = 0.032; F = 26.8; q2 = 0.832
N = 12; r = 0.934; r2 = 0.871; SD = 0.029; F = 30.5; q2 = 0.852
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. General Procedure: Synthesis
3.2.1. Alkoxy Acetophenones
3.2.2. Oxyalkyl Chalcones
3.3. Anti-Oomycete Activity
3.4. Membrane Damage
3.5. Computational Details
3.6. Quantitative Structure–Activity Relationship (QSAR)
3.7. Statistical
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fuglie, K.O. Is agricultural productivity slowing? Glob. Food Sec. 2018, 17, 73–83. [Google Scholar] [CrossRef]
- Okonya, J.S.; Ocimati, W.; Nduwayezu, A.; Kantungeko, D.; Niko, N.; Blomme, G.; Legg, J.P.; Kroschel, J. Farmer Reported Pest and Disease Impacts on Root, Tuber, and Banana Crops and Livelihoods in Rwanda and Burundi. Sustainability 2019, 11, 1592. [Google Scholar] [CrossRef] [Green Version]
- Picard, C.; Alonso, T.; Benko-Beloglavec, A.; Karadjova, O.; Matthews-Berry, S.; Paunovic, S.A.; Pietsch, M.; Reed, P.; van der Gaag, D.J.; Ward, M. Recommended regulated non-quarantine pests (RNQP s), associated thresholds and risk management measures in the European and Mediterranean region. Bull. OEPP 2018, 48, 552–568. [Google Scholar] [CrossRef] [Green Version]
- Pandey, S. International and Regional Institutions and Instruments for Agricultural Policy, Research, and Development. In Encyclopedia of Agriculture and Food Systems, 1st ed.; Van Alfen, N.K., Ed.; Academic Press: London, UK, 2014; pp. 44–48. [Google Scholar]
- Judelson, H.S. Sexual Reproduction in Oomycetes: Biology, Diversity, and Contributions to Fitness. In Oomycete Genetics and Genomics: Diversity, Interactions, and Research Tools, 1st ed.; Lamour, K., Kamoun, S., Eds.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2009; pp. 121–138. [Google Scholar]
- Singh, D.; Sharma, R.R. Postharvest Disinfection of Fruits and Vegetables. In Postharvest Diseases of Fruits and Vegetables and Their Management, 1st ed.; Siddiqui, M.W., Ed.; Academic Press: New York, NY, USA, 2018; pp. 1–52. [Google Scholar]
- Meszka, B.; Michalecka, M. Identification of Phytophthora spp. isolated from plants and soil samples on strawberry plantations in Poland. J. Plant. Dis Prot. 2016, 123, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Scott, P.; Bader, M.; Burgess, T.; Hardy, G.; Williams, N. Global biogeography and invasion risk of the plant pathogen genus Phytophthora. Environ. Sci. Policy 2019, 101, 175–182. [Google Scholar] [CrossRef]
- Fry, W.E.; Grunwald, N.J.; Cooke, D.; McLeod, A.; Forbes, G.A.; Cao, K. Population Genetics and Population Diversity of Phytophthora infestans. In Oomycete Genetics and Genomics: Diversity, Interactions, and Research Tools, 1st ed.; Lamour, K., Kamoun, S., Eds.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2009; pp. 139–164. [Google Scholar]
- Leadbeater, A.J. Plant Health Management: Fungicides and Antibiotics. In Encyclopedia of Agriculture and Food Systems, 2nd ed.; Van Alfen, N.K., Ed.; Academic Press: New York, NY, USA, 2018; pp. 408–424. [Google Scholar]
- Bena, Y.; Fua, C.; Hua, M.; Liua, L.; Wonga, M.H.; Zheng, C. Human health risk assessment of antibiotic resistance associated with antibiotic residues in the environment: A review. Environ. Res. 2019, 169, 483–493. [Google Scholar] [CrossRef]
- Joshi, R.K. Role of Natural Products against Microorganisms. Am. J. Clin. Microbiol. Antimicrob. 2018, 1, 1005. [Google Scholar]
- Chen, X.; Mukwaya, E.; Wong, M.S.; Zhang, Y. A systematic review on biological activities of prenylated flavonoids. Pharm. Biol. 2014, 52, 655–660. [Google Scholar] [CrossRef]
- Cheenpracha, S.; Karalai, C.; Ponglimanont, C.; Subhadhirasakul, S.; Tewtrakul, S. Anti-HIV-1 protease activity of compounds from Boesenbergia pandurata. Bioorg. Med. Chem. 2006, 14, 1710–1714. [Google Scholar] [CrossRef]
- Hans, R.; Guantai, E.M.; Lategan, C.; Smith, P.J.; Wan, B.; Franzblau, S.G.; Gut, J.; Rosenthal, P.J.; Chibale, K. Synthesis, antimalarial and antitubercular activity of acetylenic chalcones. Bioorg. Med. Chem. Lett. 2010, 20, 942–944. [Google Scholar] [CrossRef]
- Tadigoppula, N.; Korthikunta, V.; Gupta, S.; Kancharla, P.; Khaliq, T.; Soni, A.; Srivastava, R.K.; Srivastava, K.; Puri, S.K.; Raju, K.S.R. Synthesis and insight into the structure-activity relationships of chalcones as antimalarial agents. J. Med. Chem. 2012, 56, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Muškinja, J.; Burmudžija, A.; Ratkovic, Z.; Rankovic, B.; Kosanic, M.; Bogdanovic, G.A.; Novakovic, S.B. Ferrocenyl chalcones with O-alkylated vanillins: Synthesis, spectral characterization, microbiological evaluation, and single-crystal X-ray analysis. Med. Chem. Res. 2016, 25, 1744–1753. [Google Scholar] [CrossRef]
- Escobar, B.; Montenegro, I.; Villena, J.; Werner, E.; Godoy, P.; Olguín, Y.; Madrid, A. Hemi-Synthesis and Anti-Oomycete Activity of Analogues of Isocordoin. Molecules 2017, 22, 968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miles, C.O.; Main, L.; Nicholson, B.K. Synthesis of 2′,6′-Dihydroxychalcones by Using Tetrahydropyran-2-yl and Trialkylsilyl Protective Groups: The Crystal Structure Determination of 2′,6′-Dihydroxy-2,4,6-trimethoxychalcone. Aust. J. Chem. 1989, 42, 1103–1113. [Google Scholar] [CrossRef]
- Li, J.T.; Yang, W.Z.; Wang, S.X.; Li, S.H.; Li, T.S. Improved synthesis of chalcones under ultrasound irradiation. Ultrason. Sonochem. 2002, 9, 237–239. [Google Scholar] [CrossRef]
- Marquina, S.; Maldonado-Santiagoa, M.; Sánchez-Carranza, J.N.; Antúnez-Mojica, M.; González-Maya, L.; Razo-Hernández, R.S.; Alvarez, L. Design, synthesis and QSAR study of 2′-hydroxy-4′-alkoxy chalcone derivatives that exert cytotoxic activity by the mitochondrial apoptotic pathway. Bioorg. Med. Chem. 2019, 27, 43–54. [Google Scholar] [CrossRef]
- Ngaini, Z.; Fadzillah, S.M.H.; Hussain, H. Synthesis and antimicrobial studies of hydroxylated chalcone derivatives with variable chain length. Nat. Prod. Res. 2012, 26, 892–902. [Google Scholar] [CrossRef]
- Prusky, D.; Keen, N.T. Involvement of preformed antifungal compounds in the resistance of subtropical fruits to fungal decay. Plant. Dis. 1993, 77, 114–119. [Google Scholar] [CrossRef] [Green Version]
- Medina-Alarcón, K.P.; Singulani, J.; Dutra, L.; Pitangui, N.S.; Pereira-da-Silva, M.A.; dos Santos, M.B.; Ayusso, G.M.; Regasini, L.O.; Soares, C.P.; Chorilli, M.; et al. Antifungal activity of 2-hydroxychalcone loaded in nanoemulsion against Paracoccidioides spp. Future Microbiol. 2020, 15, 1. [Google Scholar] [CrossRef]
- Barron, D.; Ibrahim, R. Isoprenylated flavonoids—A survey. Phytochemistry 1996, 43, 921–982. [Google Scholar] [CrossRef]
- Marcos, I.S.; Escola, M.A.; Moro, R.F.; Basabe, P.; Díez, D.; Sanz, F.; Mollinedo, F.; de la Iglesia-Vicente, J.; Sierra, B.G.; Urones, J.G. Synthesis of novel antitumoural analogues of dysidiolide from ent-halimic acid. Bioorg. Med. Chem. 2007, 15, 5719–5737. [Google Scholar] [CrossRef] [PubMed]
- Basabe, P.; de Román, M.; Díez, D.; Marcos, I.S.; Blanco, A.; Bodero, O.; Mollinedo, F.; Sierra, B.G.; Urones, J.G. Prenylflavonoids and prenyl/alkyl-phloroacetophenones: Synthesis and antitumour biological evaluation. Eur. J. Med. Chem. 2010, 45, 4258–4269. [Google Scholar] [CrossRef]
- Mellado, M.; Espinoza, L.; Madrid, A.; Mella, J.; Chávez-Weisser, E.; Diaz, K.; Cuellar, M. Design, synthesis, antifungal activity, and structure–activity relationship studies of chalcones and hybrid dihydrochromane–chalcones. Mol. Divers. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mellado, M.; Madrid, A.; Reyna, M.; Weinstein-Oppenheimer, C.; Mella, J.; Salas, C.O.; Sánchez, E.; Cuellar, M. Synthesis of chalcones with antiproliferative activity on the SH-SY5Y neuroblastoma cell line: Quantitative Structure–Activity Relationship Models. Med. Chem. Res. 2018, 27, 2414–2425. [Google Scholar] [CrossRef]
- Montenegro, I.; Muñoz, O.; Villena, J.; Werner, E.; Mellado, M.; Ramírez, I.; Caro, N.; Flores, S.; Madrid, A. Structure-Activity Relationship of Dialkoxychalcones to Combat Fish Pathogen Saprolegnia australis. Molecules 2018, 23, 1377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montenegro, I.; Mellado, M.; Russo, A.; Said, B.; Besoain, X.; Godoy, P.; Werner, E.; Caro, N.; Madrid, A. Carveoylphenols and Their Antifungal Potential against Pathogenic Yeasts. Antibiotics 2019, 8, 185. [Google Scholar] [CrossRef] [Green Version]
- Olea, A.F.; Espinoza, L.; Sedan, C.; Thomas, M.; Martínez, R.; Mellado, M.; Carrasco, H.; Díaz, K. Synthesis and In Vitro Growth Inhibition of 2-Allylphenol Derivatives Against Phythopthora cinnamomi Rands. Molecules 2019, 24, 4196. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, G.D.; Da Silva, M.G.; Souza, E.; Barison, A.; Simões, S.C.; Varotti, F.P.; Barbosa, L.A.; Viana, G.; Villar, J. Design and Synthesis of New Chacones Substituted with Azide/Triazole Groups and Analysis of Their Cytotoxicity Towards HeLa Cells. Molecules 2012, 17, 10331–10343. [Google Scholar] [CrossRef] [Green Version]
- Martin, R. Handbook of Hydroxyacetophenones, 1st ed.; Springer: Dordrecht, The Netherlands, 1997; pp. 200–296. [Google Scholar]
- Venkateswararao, E.; Sharma, V.K.; Yun, J.; Kim, Y.; Jung, S. Anti-proliferative effect of chalcone derivatives through inactivation of NF-jB in human cancer cells. Bioorg. Med. Chem 2014, 22, 3386–3392. [Google Scholar] [CrossRef]
- Nguyen, H.D.; Okada, T.; Kitamura, S.; Yamaoka, S.; Horaguchi, Y.; Kasanami, Y.; Sekiguchi, F.; Tsubota, M.; Yoshida, S.; Nishikawa, H.; et al. Design and synthesis of novel anti-hyperalgesic agents based on 6-prenylnaringenin as the T-type calcium channel blockers. Bioorg. Med. Chem 2018, 26, 4410–4427. [Google Scholar] [CrossRef]
- Flores, S.; Montenegro, I.; Villena, J.; Cuellar, M.; Werner, E.; Godoy, P.; Madrid, A. Synthesis and Evaluation of novel oxyalkylated derivatives of 2′,4′-dihydroxychalcone as anti-oomycete agents against bronopol resistant strains of Saprolegnia sp. Int. J. Mol. Sci. 2016, 17, 1366. [Google Scholar] [CrossRef] [Green Version]
- Mitani, S.; Araki, S.; Yamaguchi, T.; Takii, Y.; Ohshima, T.; Matsuo, N. Antifungal Activity of the Novel Fungicide Cyazofamid against Phytophthora infestans and Other Plant Pathogenic Fungi in Vitro. Pestic. Biochem. Physiol. 2001, 70, 92–99. [Google Scholar] [CrossRef]
- Madrid-Villegas, A.; Díaz, K.; González, C.; Catalán, K.; Espinoza, L. Antiphytopathogenic Activity of Psoralea glandulosa (Fabaceae) Against Botrytis cinerea and Phytophthora cinnamomi. Nat. Prod. Res. 2015, 29, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Mellado, M.; Madrid, A.; Martinez, U.; Mella, J.; Salas, C.O.; Cuellar, M. Hansch’s analysis application to chalcone synthesis by Claisen-Schmidt reaction based in DFT methodology. Chem. Pap. 2018, 72, 703–709. [Google Scholar] [CrossRef]
- Golbraikh, A.; Tropsha, A. Beware of q2! J. Mol. Graph. Model. 2002, 20, 269–276. [Google Scholar] [CrossRef]

| Compound | a EC50 | b MIC | c MOC |
|---|---|---|---|
| 7a | 44.0 ± 0.3 | 25 | 50 |
| 7b | 67.0 ± 0.4 | 50 | 75 |
| 7c | 67.4 ± 0.2 | 50 | 75 |
| 7d | 75.9 ± 0.3 | 50 | 75 |
| 8a | 32.9 ± 0.6 | 12.5 | 25 |
| 8b | 53.5 ± 1.0 | 25 | 50 |
| 8c | 53.3 ± 0.2 | 25 | 50 |
| 8d | 67.0 ± 2.3 | 50 | 75 |
| 9a | 44.0 ± 1.7 | 25 | 50 |
| 9b | 57.8 ± 0.2 | 50 | 75 |
| 9c | 55.6 ± 0.3 | 50 | 75 |
| 9d | 65.9 ± 0.2 | 50 | 75 |
| Difenoconazole | 113.8 ± 0.4 | 100 | 125 |
| Metalaxyl | 28.6 ± 0.7 | 12.5 | 12.5 |
| Compounds | % Membrane Lysis a |
|---|---|
| 7a | 50.1 ± 0.3 |
| 7b | 31.1 ± 0.6 |
| 7c | 29.0 ± 0.5 |
| 7d | 27.3 ± 0.3 |
| 8a | 100 ± 0.0 |
| 8b | 47.0 ± 0.1 |
| 8c | 45.0 ± 0.3 |
| 8d | 30.0 ± 0.2 |
| 9a | 55.0 ± 0.2 |
| 9b | 42.7 ± 0.4 |
| 9c | 41.2 ± 0.3 |
| 9d | 33.0 ± 0.1 |
| SDS | 100 ± 0.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López, G.; Mellado, M.; Werner, E.; Said, B.; Godoy, P.; Caro, N.; Besoain, X.; Montenegro, I.; Madrid, A. Sonochemical Synthesis of 2’-Hydroxy-Chalcone Derivatives with Potential Anti-Oomycete Activity. Antibiotics 2020, 9, 576. https://doi.org/10.3390/antibiotics9090576
López G, Mellado M, Werner E, Said B, Godoy P, Caro N, Besoain X, Montenegro I, Madrid A. Sonochemical Synthesis of 2’-Hydroxy-Chalcone Derivatives with Potential Anti-Oomycete Activity. Antibiotics. 2020; 9(9):576. https://doi.org/10.3390/antibiotics9090576
Chicago/Turabian StyleLópez, Génesis, Marco Mellado, Enrique Werner, Bastián Said, Patricio Godoy, Nelson Caro, Ximena Besoain, Iván Montenegro, and Alejandro Madrid. 2020. "Sonochemical Synthesis of 2’-Hydroxy-Chalcone Derivatives with Potential Anti-Oomycete Activity" Antibiotics 9, no. 9: 576. https://doi.org/10.3390/antibiotics9090576





