Candida auris Urinary Tract Infections and Possible Treatment
Abstract
1. Introduction
2. Epidemiology/Pathophysiology/Risk Factors
3. Identification/Susceptibilities
4. Review of Published Reports of Candia auris Isolates in Urine
5. Management of C. auris UTIs
5.1. Infection Control
5.2. Infection Surveillance
5.3. Source Control
5.4. Antifungal Therapy
5.4.1. Triazole Antifungals
5.4.2. Echinocandins
5.4.3. Amphotericin B
5.4.4. Flucytosine
5.4.5. Combination Therapy
5.4.6. Amphotericin B Bladder Irrigations
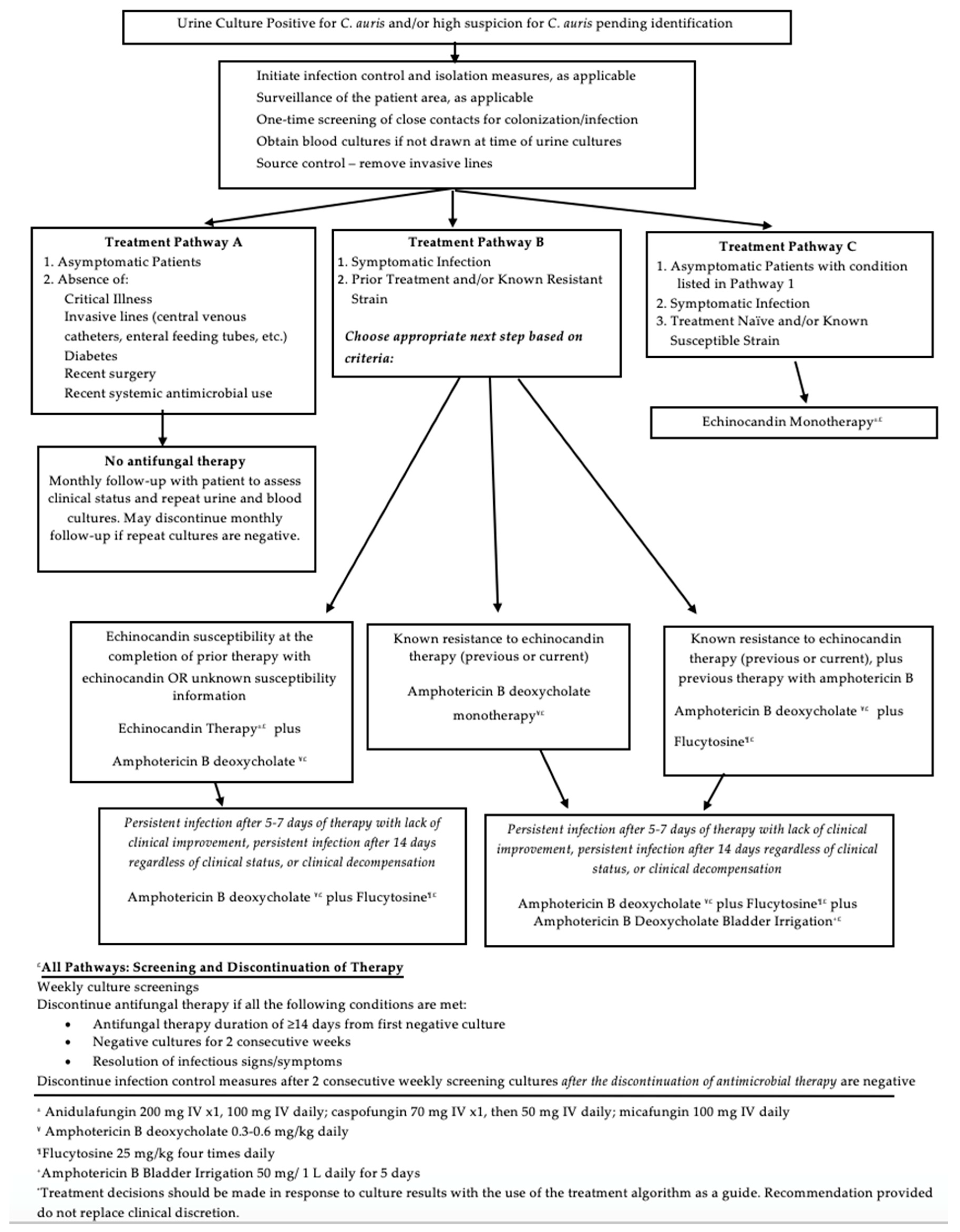
5.5. Treatment Algorithm
5.6. Eradication Screening
6. Discussion
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fisher, J.F.; Sobel, J.D.; Kauffman, C.A.; Newman, C.A. Candida urinary tract Infections—Treatment. Clin. Infect. Dis. 2011, 52, S457–S466. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, P.; Behzadi, E.; Ranjbar, R. Urinary tract infections and candida albicans. Cent. Eur. J. Urol. 2015, 68, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Bongomin, F.; Gago, S.; Oladele, R.; Denning, D. Global and multi-national prevalence of fungal Diseases—Estimate precision. J. Fungi (Basel) 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Gharanfoli, A.; Mahmoudi, E.; Torabizadeh, R.; Katiraee, F.; Faraji, S. Isolation, characterization, and molecular identification of candida species from urinary tract infections. Curr. Med. Mycol. 2019, 5, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M.; Dóczi, I.; Ábrók, M.; Lázár, A.; Burián, K. Epidemiology of candiduria and candida urinary tract infections in inpatients and outpatients: Results from a 10-year retrospective survey. Cent. Eur. J. Urol. 2019, 72, 209–214. [Google Scholar] [CrossRef]
- Fazeli, A.; Kordbacheh, P.; Nazari, A.; GHAZVINI, R.D.; Mirhendi, H.; Safara, M.; Bakhshi, H.; Yaghoubi, R. Candiduria in hospitalized patients and identification of isolated candida species by morphological and molecular methods in Ilam, Iran. Iran. J. Public Health 2019, 48, 156–161. [Google Scholar] [PubMed]
- Candida Auris. Available online: https://www.cdc.gov/fungal/candida-auris/index.html (accessed on 1 August 2020).
- Biagi, M.J.; Wiederhold, N.P.; Gibas, C.; Wickes, B.L.; Lozano, V.; Bleasdale, S.C.; Danziger, L. Development of high-level echinocandin resistance in a patient with recurrent candida auris candidemia secondary to chronic candiduria. Open Forum Infect. Dis. 2019, 6, ofz262. [Google Scholar] [CrossRef]
- Bradley, S.F. What is known about candida auris. JAMA 2019, 322, 1510. [Google Scholar] [CrossRef]
- Satoh, K.; Makimura, K.; Hasumi, Y.; Nishiyama, Y.; Uchida, K.; Yamaguchi, H. Candida auris sp. nov., a novel ascomycetous yeast isolated from the external ear canal of an inpatient in a Japanese hospital. Microbiol. Immunol. 2009, 53, 41–42. [Google Scholar] [CrossRef]
- Lee, W.G.; Shin, J.H.; Uh, Y.; Kang, M.G.; Kim, S.H.; Park, K.H.; He, J. First three reported cases of nosocomial fungemia caused by candida auris. J. Clin. Microbiol. 2011, 49, 3139–3142. [Google Scholar] [CrossRef]
- Kathuria, S.; Singh, P.K.; Sharma, C.; Prakash, A.; Masih, A.; Kumar, A.; Meis, J.F.; Chowdhary, A. Multidrug-resistant candida auris misidentified as candida haemulonii: Characterization by matrix-assisted laser desorption Ionization-Time of flight mass spectrometry and DNA sequencing and its antifungal susceptibility profile variability by vitek 2, CLSI broth microdilution, and etest method. J. Clin. Microbiol. 2015, 53, 1823–1830. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.N.; Shin, J.H.; Sung, H.; Lee, K.; Kim, E.C.; Ryoo, N.; Lee, J.S.; Jung, S.I.; Park, K.H.; Kee, S.J.; et al. Candida haemulonii and closely related species at 5 university hospitals in korea: Identification, antifungal susceptibility, and clinical features. Clin. Infect. Dis. 2009, 48, e57–e61. [Google Scholar] [CrossRef] [PubMed]
- Welsh, R.M.; Bentz, M.L.; Shams, A.; Houston, H.; Lyons, A.; Rose, L.J.; Litvintseva, A.P. Survival, persistence, and isolation of the emerging multidrug-resistant pathogenic yeast candida auris on a plastic health care surface. J. Clin. Microbiol. 2017, 55, 2996–3005. [Google Scholar] [CrossRef] [PubMed]
- Kean, R.; Delaney, C.; Sherry, L.; Borman, A.; Johnson, E.M.; Richardson, M.D.; Rautemaa-Richardson, R.; Williams, C.; Ramage, G. Transcriptome assembly and profiling of candida auris reveals novel insights into biofilm-mediated resistance. mSphere 2018, 3, 334. [Google Scholar] [CrossRef] [PubMed]
- Short, B.; Brown, J.; Delaney, C.; Sherry, L.; Williams, C.; Ramage, G.; Kean, R. Candida auris exhibits resilient biofilm characteristics in vitro: Implications for environmental persistence. J. Hosp. Infect. 2019, 103, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.; Ahmad, S.; Al-Sweih, N.; Joseph, L.; Alfouzan, W.; Asadzadeh, M. Increasing prevalence, molecular characterization and antifungal drug susceptibility of serial candida auris isolates in kuwait. PLoS ONE 2018, 13, e0195743. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.R.; Etienne, K.A.; Vallabhaneni, S.; Farooqi, J.; Chowdhary, A.; Govender, N.P.; Colombo, A.L.; Calvo, B.; Cuomo, C.A.; Desjardins, C.A.; et al. Simultaneous emergence of multidrug-resistant candida auris on 3 continents confirmed by whole-genome sequencing and epidemiological analyses. Clin. Infect. Dis. 2017, 64, 134–140. [Google Scholar] [CrossRef]
- Healey, K.R.; Kordalewska, M.; Jiménez Ortigosa, C.; Singh, A.; Berrío, I.; Chowdhary, A.; Perlin, D.S. Limited ERG11 mutations identified in isolates of candida auris directly contribute to reduced azole susceptibility. Antimicrob. Agents Chemother. 2018, 62, e01427-18. [Google Scholar] [CrossRef]
- Escandón, P.; Chow, N.A.; Caceres, D.H.; Gade, L.; Berkow, E.L.; Armstrong, P.; Rivera, S.; Misas, E.; Duarte, C.; Moulton-Meissner, H.; et al. Molecular epidemiology of candida auris in colombia reveals a highly related, countrywide colonization with regional patterns in amphotericin B resistance. Clin. Infect. Dis. 2018, 68, 15–21. [Google Scholar] [CrossRef]
- Chowdhary, A.; Prakash, A.; Sharma, C.; Kordalewska, M.; Kumar, A.; Sarma, S.; Tarai, B.; Singh, A.; Upadhyaya, G.; Upadhyay, S.; et al. A multicentre study of antifungal susceptibility patterns among 350 candida auris isolates (2009–17) in india: Role of the ERG11 and FKS1 genes in azole and echinocandin resistance. J. Antimicrob. Chemother. 2018, 73, 891–899. [Google Scholar] [CrossRef]
- Charlier, C.; El Sissy, C.; Bachelier-Bassi, S.; Scemla, A.; Quesne, G.; Sitterlé, E.; Legendre, C.; Lortholary, O.; Bougnoux, M.-E. Acquired flucytosine resistance during combination therapy with caspofungin and flucytosine for candida glabrata cystitis. Antimicrob. Agents Chemother. 2015, 60, 662–665. [Google Scholar] [CrossRef] [PubMed]
- Chastain, D.B.; King, S.T.; Stover, K.R. Rethinking urinary antibiotic breakpoints: Analysis of urinary antibiotic concentrations to treat multidrug resistant organisms. BMC Res. Notes 2018, 11, 497. [Google Scholar] [CrossRef] [PubMed]
- Sayeed, M.A.; Farooqi, J.; Jabeen, K.; Awan, S.; Mahmood, S.F. Clinical spectrum and factors impacting outcome of candida auris: A single center study from Pakistan. BMC Infect. Dis. 2019, 19, 384. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Gaitán, A.; Moret, A.M.; Tasias-Pitarch, M.; Aleixandre-Lopez, A.I.; Martínez-Morel, H.; Calabuig, E.; Salavert-Lletí, M.; Ramírez, P.; Hontangas, J.L.L.; Hagen, F.; et al. An outbreak due to candida auris with prolonged colonisation and candidaemia in a tertiary care european hospital. Mycoses 2018, 61, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Schelenz, S.; Hagen, F.; Rhodes, J.L.; Abdolrasouli, A.; Chowdhary, A.; Hall, A.; Ryan, L.; Shackleton, J.; Trimlett, R.; Meis, J.F.; et al. First hospital outbreak of the globally emerging candida auris in a european hospital. Antimicrob. Resist. Infect. Control. 2016, 5, 35–37. [Google Scholar] [CrossRef]
- Ahmad, S.; Khan, Z.; Al-Sweih, N.; Alfouzan, W.; Joseph, L. Candida auris in various hospitals across kuwait and their susceptibility and molecular basis of resistance to antifungal drugs. Mycoses 2019, 63, 104–112. [Google Scholar] [CrossRef]
- Barantsevich, N.E.; Orlova, O.E.; Shlyakhto, E.V.; Johnson, E.; Woodford, N.; Lass-Floerl, C.; Churkina, I.; Mitrokhin, S.; Shkoda, A.; Barantsevich, E.P. Emergence of candida auris in Russia. J. Hosp. Infect. 2019, 102, 445–448. [Google Scholar] [CrossRef]
- Adams, E.; Quinn, M.; Tsay, S.; Poirot, E.; Chaturvedi, S.; Southwick, K.; Greenko, J.; Fernandez, R.; Al, E.A.E.; Vallabhaneni, S.; et al. Candida auris in healthcare facilities, new york, USA, 2013–2017. Emerg. Infect. Dis. 2018, 24, 1816–1824. [Google Scholar] [CrossRef]
- Al-Siyabi, T.; Busaidi, I.A.; Balkhair, A.; Al-Muharrmi, Z.; Al-Salti, M.; Al’Adawi, B. First report of candida auris in Oman: Clinical and microbiological description of five candidemia cases. J. Infect. 2017, 75, 373–376. [Google Scholar] [CrossRef]
- Hamprecht, A.; Barber, A.E.; Mellinghoff, S.C.; Thelen, P.; Walther, G.; Yu, Y.; Neurgaonkar, P.; Dandekar, T.; Cornely, O.A.; Martin, R.; et al. Candida auris in Germany and previous exposure to foreign healthcare. Emerg. Infect. Dis. 2019, 25, 1763–1765. [Google Scholar] [CrossRef]
- Ostrowsky, B.; Greenko, J.; Adams, E.; Quinn, M.; O’Brien, B.; Chaturvedi, V.; Berkow, E.; Vallabhaneni, S.; Forsberg, K.; Chaturvedi, S.; et al. Candida auris isolates resistant to three classes of antifungal medications—New York, 2019. MMWR Morb. Mortal Wkly Rep. 2020, 69, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Vallabhaneni, S.; Kallen, A.; Tsay, S.; Chow, N.; Welsh, R.; Kerins, J.; Kemble, S.K.; Pacilli, M.; Black, S.R.; Landon, E.; et al. Investigation of the First Seven Reported Cases of Candida auris, a Globally Emerging Invasive, Multidrug-Resistant Fungus—United States, May 2013–August 2016. MMWR Morb. Mortal Wkly Rep. 2016, 65, 1234–1237. [Google Scholar] [CrossRef] [PubMed]
- Mohsin, J.; Hagen, F.; Al-Balushi, Z.A.M.; De Hoog, S.; Chowdhary, A.; Meis, J.F.; Al-Hatmi, A.M. The first cases of candida auris candidaemia in Oman. Mycoses 2017, 60, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Candida Auris in Healthcare Settings. Available online: https://www.ecdc.europa.eu/en/publications-data/candida-auris-healthcare-settings (accessed on 1 August 2020).
- Candida Auris. Available online: https://www.gov.uk/government/collections/candida-auris#guidance-for-laboratories,-healthcare-providers-and-healthcare-professionals (accessed on 1 August 2020).
- Cadnum, J.L.; Shaikh, A.A.; Piedrahita, C.T.; Sankar, T.; Jencson, A.L.; Larkin, E.L.; Ghannoum, M.A.; Donskey, C.J. Effectiveness of disinfectants against candida auris and other candida species. Infect. Control. Hosp. Epidemiol. 2017, 38, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Abdolrasouli, A.; Armstrong-James, D.; Ryan, L.; Schelenz, S. In vitro efficacy of disinfectants utilised for skin decolonisation and environmental decontamination during a hospital outbreak with candida auris. Mycoses 2017, 60, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.; Schelenz, S.; Borman, A.M.; Johnson, E.M.; Brown, C.S. Yeasticidal activity of chemical disinfectants and antiseptics against candida auris. J. Hosp Infect. 2017, 97, 371–375. [Google Scholar] [CrossRef]
- Selected EPA-Registered Disinfectants. Available online: https://www.epa.gov/pesticide-registration/selected-epa-registered-disinfectants#candida-auris (accessed on 1 August 2020).
- Fluconazole Tablet. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=5590e3fc-b9a1-4863-9420-900bf437a3cc (accessed on 1 August 2020).
- Eraxis-Anidulafungin Injection, Powder, Lyophilized, for Solution. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a88d9010-55fb-4a02-baff-042cd27688ea (accessed on 1 August 2020).
- Amphotec-Amphotericin B Injection, Lipid Complex. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=9b8ea543-1de8-472f-9666-34f99ca2f183 (accessed on 1 August 2020).
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical practice guideline for the management of candidiasis: 2016 update by the infectious diseases society of America. Clin. Infect. Dis. 2016, 62, e1–e50. [Google Scholar] [CrossRef]
- Flucytosine Capsule. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=8b55832c-6771-4f5b-9aef-f88a1d22795e (accessed on 1 August 2020).
- Barchiesi, F.; Arzeni, D.; Caselli, F.; Scalise, G. Primary resistance to flucytosine among clinical isolates of Candida spp. J. Antimicrob. Chemother. 2000, 45, 408–409. [Google Scholar] [CrossRef]
- Kneale, M.; Bartholomew, J.S.; Davies, E.; Denning, D.W. Global access to antifungal therapy and its variable cost. J. Antimicrob. Chemother. 2016, 71, 3599–3606. [Google Scholar] [CrossRef]
- Bidaud, A.L.; Botterel, F.; Chowdhary, A.; Dannaoui, E. In vitro antifungal combination of flucytosine with amphotericin B, voriconazole, or micafungin against candida auris shows no antagonism. Antimicrob. Agents Chemother 2019, 63, e01393-19. [Google Scholar] [CrossRef]
- Drew, R.H.; Arthur, R.R.; Perfect, J.R. Is it time to abandon the use of amphotericin B bladder irrigation? Clin. Infect. Dis. 2005, 40, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.A.; Caylor, M.M.; Lin, F.C.; Campbell-Bright, S. Comparison of amphotericin B bladder irrigations versus fluconazole for the treatment of candiduria in intensive care unit patients. J. Pharm. Pract. 2017, 30, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Tuon, F.F.; Amato, V.S.; Filho, S.R.P. Bladder irrigation with amphotericin B and fungal urinary tract infection—Systematic review with meta-analysis. Int. J. Infect. Dis. 2009, 13, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Iyer, K.R.; Whitesell, L.; Porco, J.A.; Henkel, T.; Brown, L.E.; Robbins, N.; Cowen, L.E. Translation Inhibition by Rocaglates Activates a Species-Specific Cell Death Program in the Emerging Fungal Pathogen Candida auris. mBio 2020, 11. [Google Scholar] [CrossRef]
- Novel Antifungal Shows Early Promise against Candida Auris. Available online: https://www.cidrap.umn.edu/news-perspective/2019/04/novel-antifungal-shows-early-promise-against-candida-auris (accessed on 2 December 2020).
- Naber, K.G.; Niggemann, H.; Stein, G.; Stein, G. Review of the literature and individual patients’ data meta-analysis on efficacy and tolerance of nitroxoline in the treatment of uncomplicated urinary tract infections. BMC Infect. Dis. 2014, 14, 628. [Google Scholar] [CrossRef]
| Patient | Year | Age (y), Sex | Medical History 2 | Site | Prior Antifungal Treatment | Treatment 3 | Infection Diagnosis | Clinical Outcome |
|---|---|---|---|---|---|---|---|---|
| 1 [30] | 2016–2017 | 62, M | Current: central venous catheter, ICU admission PMH: cerebrovascular accident, obstructive uropathy | Blood Urine | Yes | AFG | Candidemia | Survived to day 30 |
| 2 [30] | 2016–2017 | 31, F | Current: central venous catheter, ICU admission PMH: kidney transplant, systemic lupus erythematosus, Pneumocystis jirovecii pneumonia, Cytomegalovirus infection, pneumonitis | Blood Urine | Yes | AFG | Candidemia | Deceased |
| 3 [31] | 2017 | 67, F | PMH: intracranial hemorrhage | Urinary catheter Groin Trach | N/A | None | None | Transferred to rehabilitation facility |
| 4 [31] | 2017 | 48, F | Current: neurologic disorder | Urinary catheter | N/A | None | None | Discharged alive |
| 5 [31] | 2017 | 15, M | Not available | Urine Blood | N/A | LipAmpB VRC | Candidemia | Cultures–6 months to clear |
| 6 [31] | 2017 | 60, M | Not available | Urine | N/A | N/A | None | N/A |
| 7 [32] | 2018 | >50, N/A | Current: chronic mechanical ventilation, alcohol dependence | Urine Tracheal aspirate | No | Echinocandin | Noted to have infection | Deceased |
| 8 [32] | 2017 | >50, N/A | Current: chronic ventilator dependence | CVC tip Blood Urine | No | Echinocandin AmpB | Candidemia | Deceased |
| 9 [33] | 2016 | N/A | Current: paraplegia with long-term, indwelling Foley catheter | N/A | N/A | Fluconazole | Not stated | Survived |
| 10 [34] | 2017 | 70, F | Current: congestive cardiac failure, cellulitis, acute kidney injury on hemodialysis, brain infarcts, duodenal ulcer, H. pylori infection PMH: heart failure, ischemic heart disease, chronic kidney disease, diabetes, hypertension | Urine Blood | No | AFG | Candidemia Urosepsis | Deceased |
| 11 [8] | 2018 | 54, M | Current: sepsis, DVT, tracheostomy, enteric feeding tube, colostomy PMH: quadriplegia, chronic wounds, osteomyelitis with abscess | Urine Blood | Yes | MFG POS FLU | Candidemia | Discharged on long-term antifungal and antimicrobial therapy |
| Patient | Susceptibility Testing Method | Minimum Inhibitory Concentrations 1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| FLU 2 | ITR 3 | ISA 4 | POS 5 | VRC 6 | CAS 7 | MFG 8 | AFG 9 | AmpB 10 | Flucy 11 | ||
| 1 | YestOne Sensititre | 128 | 0.12 | N/A | 0.06 | 0.5 | 0.12 | 0.12 | 0.12 | 1 | 8 |
| 2 | YestOne Sensititre | 256 | 0.25 | N/A | 0.12 | 2 | 0.12 | 0.12 | 0.12 | 2 | 0.12 |
| 3 | Broth Microdilution | ≥64 | 2 | 1 | 0.5 | 2 | N/A | N/A | 0.25 | N/A | N/A |
| 4 | Broth Microdilution | ≥64 | 0.25 | ≤0.016 | ≤0.016 | 0.25 | N/A | N/A | 0.125 | N/A | N/A |
| 5 | Broth Microdilution | ≥64 | 0.5 | 0.25 | ≤0.016 | 0.5 | N/A | N/A | 16 | N/A | N/A |
| 6 | Broth Microdilution | ≥64 | 0.25 | ≤0.016 | 0.06 | 2 | N/A | N/A | 0.5 | N/A | N/A |
| 7 | Broth Microdilution/E-Test | >256 | N/A | N/A | 0.5 | 2 | 2 | 4 | 4 | 2 | N/A |
| 8 | Broth Microdilution/E-Test | >256 | N/A | N/A | 0.25 | 2 | 16 | 4 | 4 | 2 | N/A |
| 9 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 10 | Broth Microdilution | 64 | 0.031 | <0.016 | <0.016 | 0.125 | N/A | 0.063 | 0.031 | 2 | N/A |
| 11 | YestOne Sensititre/Broth Microdilution | 2 | N/A | ≤0.03 | N/A | 0.015 | 0.06 | 0.12 | 0.12 | 1 | N/A |
| 2 | N/A | ≤0.03 | N/A | 0.06 | >8 | >8 | 4 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Griffith, N.; Danziger, L. Candida auris Urinary Tract Infections and Possible Treatment. Antibiotics 2020, 9, 898. https://doi.org/10.3390/antibiotics9120898
Griffith N, Danziger L. Candida auris Urinary Tract Infections and Possible Treatment. Antibiotics. 2020; 9(12):898. https://doi.org/10.3390/antibiotics9120898
Chicago/Turabian StyleGriffith, Nicole, and Larry Danziger. 2020. "Candida auris Urinary Tract Infections and Possible Treatment" Antibiotics 9, no. 12: 898. https://doi.org/10.3390/antibiotics9120898
APA StyleGriffith, N., & Danziger, L. (2020). Candida auris Urinary Tract Infections and Possible Treatment. Antibiotics, 9(12), 898. https://doi.org/10.3390/antibiotics9120898





