Antibiogram Profiles and Risk Factors for Multidrug Resistance of Salmonella enterica Recovered from Village Chickens (Gallus gallus domesticus Linnaeus) and Other Environmental Sources in the Central and Southern Peninsular Malaysia
Abstract
1. Introduction
2. Results
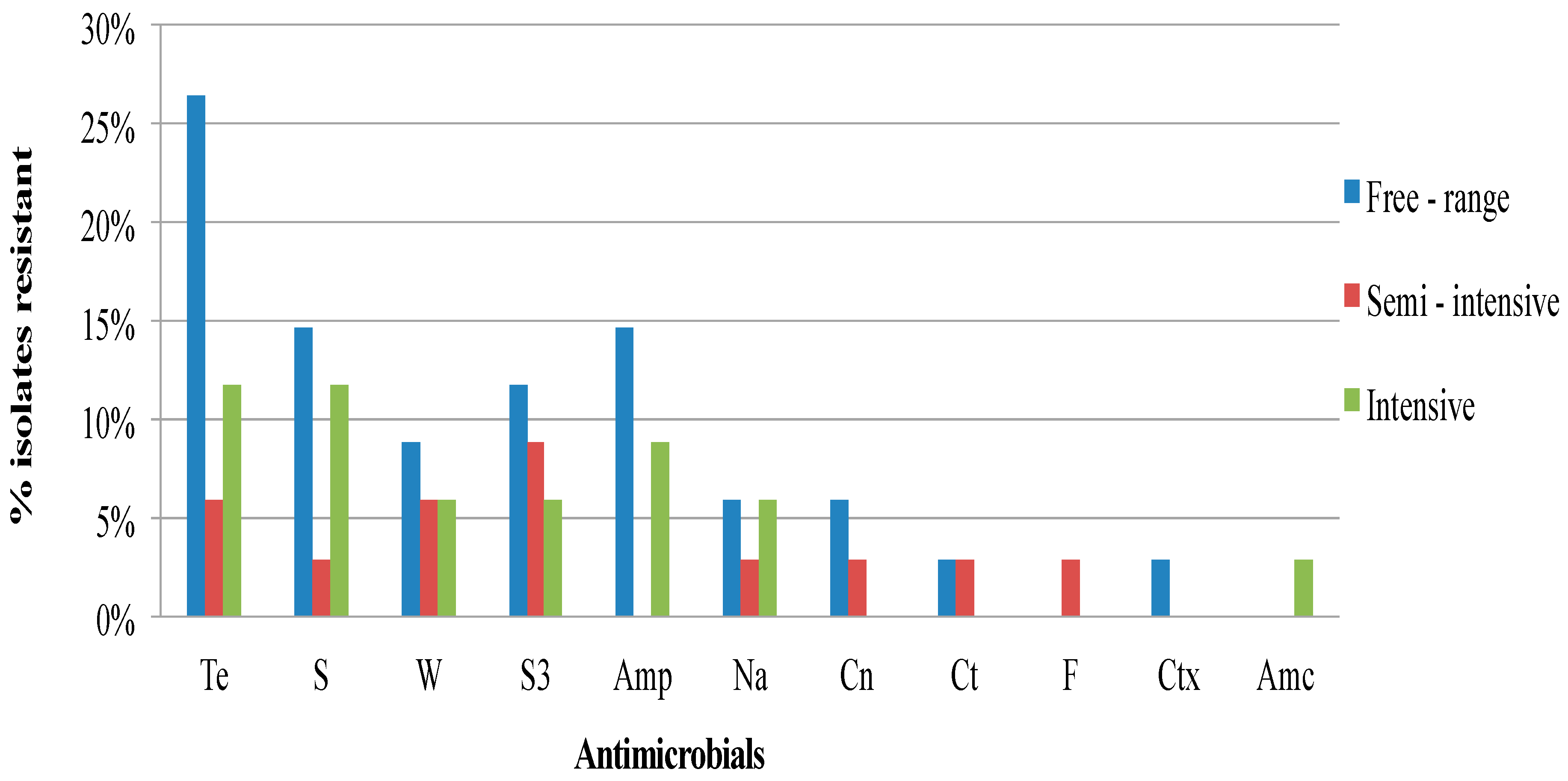
2.1. Antimicrobial Resistance and Antibiogram Profiles of the Salmonella Isolates
2.2. Analysis of Risk Factors for MDR Salmonella
3. Discussion
4. Materials and Methods
4.1. Source of the Isolates
4.2. Antimicrobial Susceptibility Testing
4.3. Data Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brown, A.C.; Grass, J.E.; Richardson, L.C.; Nisler, A.L.; Bicknese, A.S.; Gould, L.H. Antimicrobial resistance in Salmonella that caused foodborne disease outbreaks: United States, 2003–2012. Epidemiol. Infect. 2016, 145, 766–774. [Google Scholar] [CrossRef]
- Collignon, P.C.; Conly, J.M.; Andremont, A.; McEwen, S.A.; Aidara-Kane, A. World Health Organization Ranking of Antimicrobials According to Their Importance in Human Medicine: A Critical Step for Developing Risk Management Strategies to Control Antimicrobial Resistance from Food Animal Production. Clin. Infect. Dis. 2016, 63, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Nation, R.L.; Turnidge, J.D.; Milne, R.W.; Coulthard, K.; Rayner, C.R.; Paterson, D.L. Colistin: The re-emerging antibiotic for multidrug-resistant Gram-negative bacterial infections. Lancet Infect. Dis. 2006, 6, 589–601. [Google Scholar] [CrossRef]
- Kempf, I.; Jouy, E.; Chauvin, C. Colistin use and colistin resistance in bacteria from animals. Int. J. Antimicrob. Agents 2016, 48, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Poirel, L.; Jayol, A.; Nordmann, P. Polymyxins: Antibacterial Activity, Susceptibility Testing, and Resistance Mechanisms Encoded by Plasmids or Chromosomes. Clin. Microbiol. Rev. 2017, 30, 557–596. [Google Scholar] [CrossRef]
- Hassan, L.; Zaleha Suhaimi, S.; Saleha, A.A. The detection and comparison of antimicrobial resistance pattern of vancomycin-resistant enterococci and Salmonella isolated from eggs of commercial layers and free-range chickens. J. Vet. Malays. 2005, 17, 7–11. [Google Scholar]
- Miao, Z.H.; Glatz, P.C.; Ru, Y.J. Free-range Poultry Production—A Review. Asian Australas. J. Anim. Sci. 2005, 18, 113–132. [Google Scholar] [CrossRef]
- Majowicz, S.E.; Musto, J.; Scallan, E.; Angulo, F.J.; Kirk, M.; O’Brien, S.J.; Jones, T.F.; Fazil, A.; Hoekstra, R.M.; The International Collaboration on Enteric Disease “Burden of Illness” Studies. The Global Burden of Nontyphoidal Salmonella Gastroenteritis. Clin. Infect. Dis. 2010, 50, 882–889. [Google Scholar] [CrossRef]
- Eng, S.-K.; Pusparajah, P.; Ab Mutalib, N.-S.; Ser, H.-L.; Chan, K.-G.; Lee, L.-H. Salmonella: A review on pathogenesis, epidemiology and antibiotic resistance. Front. Life Sci. 2015, 8, 284–293. [Google Scholar] [CrossRef]
- Shah, D.H.; Paul, N.C.; Sischo, W.C.; Crespo, R.; Guard-Petter, J. Population dynamics and antimicrobial resistance of the most prevalent poultry-associated Salmonella serotypes. Poult. Sci. 2017, 96, 687–702. [Google Scholar] [CrossRef]
- Mezal, E.H.; Sabol, A.; Khan, M.A.; Ali, N.; Stefanova, R.; Khan, A.A. Isolation and molecular characterization of Salmonella enterica serovar Enteritidis from poultry house and clinical samples during 2010. Food Microbiol. 2014, 38, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Heal. 2015, 109, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Mouttotou, N.; Ahmad, S.; Kamran, Z.; Koutoulis, K. Prevalence, Risks and Antibiotic Resistance of Salmonella in Poultry Production Chain. In Current Topics in Salmonella and Salmonellosis; IntechOpen: London, UK, 2017; pp. 216–234. ISBN 9789537619343. [Google Scholar]
- Foley, S.L.; Lynne, A.M.; Nayak, R. Salmonella challenges: Prevalence in swine and poultry and potential pathogenicity of such isolates1,2. J. Anim. Sci. 2008, 86, E149–E162. [Google Scholar] [CrossRef] [PubMed]
- New, C.; Ubong, A.; Premarathne, J.; Thung, T.; Lee, E.; Chang, W.; Loo, Y.; Kwan, S.; Tan, C.; Kuan, C.; et al. Microbiological food safety in Malaysia from the academician’s perspective. Food Res. 2017, 1, 183–202. [Google Scholar] [CrossRef]
- Rusul, G.; Khair, J.; Radu, S.; Cheah, Y.K.; Yassin, R. Prevalence of Salmonella in broilers at retail outlets, processing plants and farms in Malaysia. Int. J. Food Microbiol. 1996, 33, 183–194. [Google Scholar] [CrossRef]
- Adzitey, F.; Rusul, G.; Huda, N. Prevalence and antibiotic resistance of Salmonella serovars in ducks, duck rearing and processing environments in Penang, Malaysia. Food Res. Int. 2012, 45, 947–952. [Google Scholar] [CrossRef]
- Arumugaswamy, R.; Rusul, G.; Hamid, S.A.; Cheah, C. Prevalence of Salmonella in raw and cooked foods in Malaysia. Food Microbiol. 1995, 12, 3–8. [Google Scholar] [CrossRef]
- Ong, L.P.; Muniandy, K.; How, S.P.; Yip, L.S.; Lim, B.K. Salmonella Isolation from Poultry farms in Malaysia from 2011 to 2013. Malays. J. Vet. Res. 2014, 5, 1–7. [Google Scholar]
- Somasundram, C.; Razali, Z.; Santhirasegaram, V. A Review on Organic Food Production in Malaysia. Horticulturae 2016, 2, 12. [Google Scholar] [CrossRef]
- Ishida, A.; Law, S.-H.; Aita, Y. Changes in food consumption expenditure in Malaysia. Agribusiness 2003, 19, 61–76. [Google Scholar] [CrossRef]
- Rahman, W.A.; Haziqah, F. Ectoparasitic fauna of scavenging chickens (Gallus domesticus) from Penang Island, Peninsular Malaysia. Malays. J. Vet. Res. 2015, 6, 33–42. [Google Scholar]
- Cardoso, M.O.; Ribeiro, A.R.; Dos Santos, L.R.; Pilotto, F.; De Moraes, H.L.; Salle, C.T.P.; Rocha, S.L.D.S.; Nascimento, V.P.D. Antibiotic resistance in Salmonella Enteritidis isolated from broiler carcasses. Braz. J. Microbiol. 2006, 37, 368–371. [Google Scholar] [CrossRef]
- Cheah, Y.-K.; Learn-Han, L.; Noorzaleha, A.; Son, R.; Sabrina, S.; Jiun-Horng, S.; Chai-Hoon, K. Characterization of multiple-antimicrobial-resistant Salmonella enterica Subsp. enterica isolated from indigenous vegetables and poultry in Malaysia. Lett. Appl. Microbiol. 2008, 46, 318–324. [Google Scholar] [CrossRef]
- Thong, K.L. Characterization of drug resistant Salmonella enterica Serotype Typhimurium by Antibiograms, Plasmids, Integrons, Resistance Genes and PFGE. J. Microbiol. Biotechnol. 2010, 20, 1042–1052. [Google Scholar] [CrossRef]
- Khoo, E.; Roseliza, R.; Khoo, L.; Nafizah, M.; Saifu Nazri, R.; Hasnah, Y.; Norazariyah, M.; Rosnah, Y.; Rosna, D.; Siti Nor Hanani, R.; et al. Antimicrobial resistance of Salmonella enterica serovar Typhimurium from various meats received in VRI. Malays. J. Vet. Res. 2015, 6, 61–65. [Google Scholar]
- Lamas, A.; Fernandez-No, I.C.; Miranda, J.M.; Vázquez, B.I.; Cepeda, A.; Franco, C.M. Prevalence, molecular characterization and antimicrobial resistance of Salmonella serovars isolated from northwestern Spanish broiler flocks (2011–2015). Poult. Sci. 2016, 95, 2097–2105. [Google Scholar] [CrossRef]
- Abdel-Maksoud, M.; Abdel-Khalek, R.; El-Gendy, A.; Gamal, R.F.; Abdelhady, H.M.; House, B.L. Genetic characterisation of multidrug-resistant Salmonella enterica serotypes isolated from poultry in Cairo, Egypt. Afr. J. Lab. Med. 2015, 4, 7. [Google Scholar] [CrossRef]
- Zhang, L.; Fu, Y.; Xiong, Z.; Ma, Y.; Wei, Y.; Qu, X.; Zhang, H.; Zhang, J.; Liao, M. Highly Prevalent Multidrug-Resistant Salmonella From Chicken and Pork Meat at Retail Markets in Guangdong, China. Front. Microbiol. 2018, 9, 2104. [Google Scholar] [CrossRef]
- Eguale, T. Non-typhoidal Salmonella serovars in poultry farms in central Ethiopia: Prevalence and antimicrobial resistance. BMC Vet. Res. 2018, 14, 217. [Google Scholar] [CrossRef]
- Shang, K.; Wei, B.; Kang, M. Distribution and dissemination of antimicrobial-resistant Salmonella in broiler farms with or without enrofloxacin use. BMC Vet. Res. 2018, 14, 257. [Google Scholar] [CrossRef]
- Chashni, E.; Hassanzadeh, S.H.; Fard, B. Characterization of the Salmonella Isolates from Backyard Chickens in North of Iran, by Serotyping, Multiplex PCR and Antibiotic Resistance Analysis. Arch. Razi Inst. 2009, 64, 77–83. [Google Scholar]
- Ghoddusi, A.; Fasaei, B.N.; Karimi, V.; Tamai, I.A.; Moulana, Z.; Salehi, T.Z. Molecular identification of Salmonella Infantis isolated from backyard chickens and detection of their resistance genesby PCR. Iran. J. Vet. Res. 2015, 16, 293–297. [Google Scholar]
- Manning, J.; Gole, V.; Chousalkar, K.K. Screening for Salmonella in backyard chickens. Prev. Vet. Med. 2015, 120, 241–245. [Google Scholar] [CrossRef]
- Zhao, X.; Gao, Y.; Ye, C.; Yang, L.; Wang, T.; Chang, W. Prevalence and Characteristics of Salmonella Isolated from Free-Range Chickens in Shandong Province, China. BioMed Res. Int. 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Jafari, R.; Ghorbanpour, M.; Jaideri, A. An Investigation into Salmonella Infection Status in Backyard Chickens in Iran. Int. J. Poult. Sci. 2007, 6, 227–229. [Google Scholar] [CrossRef]
- Geidam, Y.A.; Zakaria, Z.; Aziz, S.A.; Bejo, S.K.; Abu, J.; Omar, S. High Prevalence of Multi-drug Resistant Bacteria in Selected Poultry Farms in Selangor, Malaysia. Asian J. Anim. Vet. Adv. 2012, 7, 891–897. [Google Scholar] [CrossRef]
- Borges, K.A.; Furian, T.; Souza, S.; Salle, C.; Moraes, H.; Nascimento, V.P.D. Antimicrobial Resistance and Molecular Characterization of Salmonella Enterica Serotypes Isolated from Poultry Sources in Brazil. Rev. Bras. Ciência Avícola 2019, 21, 21. [Google Scholar] [CrossRef]
- Rech, D.V.; Vaz, C.S.L.; Alves, L.; Coldebella, A.; Leão, J.A.; Rodrigues, D.P.; Back, A. A temporal study of Salmonella enterica serotypes from broiler farms in Brazil. Poult. Sci. 2015, 94, 433–441. [Google Scholar] [CrossRef]
- Esengupta, S.; Chattopadhyay, M.K.; Grossart, H.-P. The multifaceted roles of antibiotics and antibiotic resistance in nature. Front. Microbiol. 2013, 4, 47. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, K.S.; Jeon, J.H.; Lee, H.S. Antibiotic resistance in soil. Lancet Infect. Dis. 2018, 18, 1306–1307. [Google Scholar] [CrossRef]
- Tyrrell, C.; Burgess, C.M.; Brennan, F.P.; Walsh, F. Antibiotic resistance in grass and soil. Biochem. Soc. Trans. 2019, 47, 477–486. [Google Scholar] [CrossRef]
- Almakki, A.; Jumas-Bilak, E.; Marchandin, H.; Licznar-Fajardo, P. Antibiotic resistance in urban runoff. Sci. Total Environ. 2019, 667, 64–76. [Google Scholar] [CrossRef]
- Sanganyado, E.; Gwenzi, W. Antibiotic resistance in drinking water systems: Occurrence, removal, and human health risks. Sci. Total Environ. 2019, 669, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Shobrak, M.Y.; Abo-Amer, A.E. Role of wild birds as carriers of multi-drug resistant Escherichia coli and Escherichia vulneris. Braz. J. Microbiol. 2015, 45, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Atterby, C.; Ramey, A.M.; Hall, G.G.; Järhult, J.; Börjesson, S.; Bonnedahl, J. Increased prevalence of antibiotic-resistantE. coliin gulls sampled in Southcentral Alaska is associated with urban environments. Infect. Ecol. Epidemiol. 2016, 6, 32334. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ramey, A.M.; Hernandez, J.; Tyrlöv, V.; Uher-Koch, B.D.; Schmutz, J.A.; Atterby, C.; Järhult, J.D.; Bonnedahl, J. Antibiotic-Resistant Escherichia coli in Migratory Birds Inhabiting Remote Alaska. EcoHealth 2017, 15, 72–81. [Google Scholar] [CrossRef]
- Ahlstrom, C.A.; Bonnedahl, J.; Woksepp, H.; Hernandez, J.; Olsen, B.; Ramey, A.M. Acquisition and dissemination of cephalosporin-resistant E. coli in migratory birds sampled at an Alaska landfill as inferred through genomic analysis. Sci. Rep. 2018, 8, 7361. [Google Scholar] [CrossRef]
- Singh, R.; Yadav, A.S.; Tripathi, V.; Singh, R.P. Antimicrobial resistance profile of Salmonella present in poultry andpoultry environment in north India. Food Control 2013, 33, 545–548. [Google Scholar] [CrossRef]
- Musa, I.W.; Mansur, M.S.; Sa’Idu, L.; Mohammed, B.; Aliyu, H.B. Poultry environment and farm practices influencing the isolation rate of multi-drug resistant Salmonella from water and poultry feed in Zaria, Nigeria. J. Appl. Biol. Biotechnol. 2014, 2, 13–16. [Google Scholar]
- Rossi, F.; Girardello, R.; Cury, A.P.; Di Gioia, T.S.R.; De Almeida, J.N.; Duarte, A.J.D.S. Emergence of colistin resistance in the largest university hospital complex of São Paulo, Brazil, over five years. Braz. J. Infect. Dis. 2017, 21, 98–101. [Google Scholar] [CrossRef]
- Rolain, J.-M.; Olaitan, A.O.; Information, P.E.K.F.C. Plasmid-mediated colistin resistance: The final blow to colistin? Int. J. Antimicrob. Agents 2016, 47, 4–5. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Baron, S.; Hadjadj, L.; Rolain, J.-M.; Olaitan, A.O. Molecular mechanisms of polymyxin resistance: Knowns and unknowns. Int. J. Antimicrob. Agents 2016, 48, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Al-Tawfiq, J.A.; Laxminarayan, R.; Mendelson, M. How should we respond to the emergence of plasmid-mediated colistin resistance in humans and animals? Int. J. Infect. Dis. 2017, 54, 77–84. [Google Scholar] [CrossRef]
- Huang, X.; Yu, L.; Chen, X.; Zhi, C.; Yao, X.; Liu, Y.; Wu, S.; Guo, Z.; Yi, L.; Zeng, Z.; et al. High Prevalence of Colistin Resistance and mcr-1 Gene in Escherichia coli Isolated from Food Animals in China. Front. Microbiol. 2017, 8, 562. [Google Scholar] [CrossRef]
- Hembach, N.; Schmid, F.; Alexander, J.; Hiller, C.; Rogall, E.T.; Schwartz, T. Occurrence of the mcr-1 Colistin Resistance Gene and other Clinically Relevant Antibiotic Resistance Genes in Microbial Populations at Different Municipal Wastewater Treatment Plants in Germany. Front. Microbiol. 2017, 8, 1282. [Google Scholar] [CrossRef]
- Partridge, S.R.; Di Pilato, V.; Doi, Y.; Feldgarden, M.; Haft, D.H.; Klimke, W.; Kumar-Singh, S.; Liu, J.-H.; Malhotra-Kumar, S.; Prasad, A.; et al. Proposal for assignment of allele numbers for mobile colistin resistance (mcr) genes. J. Antimicrob. Chemother. 2018, 73, 2625–2630. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Zhou, Y.; Li, J.; Yin, W.; Wang, S.; Zhang, S.; Shen, J.; Shen, Z.; Wang, Y. Emergence of a novel mobile colistin resistance gene, mcr-8, in NDM-producing Klebsiella pneumoniae. Emerg. Microbes Infect. 2018, 7, 1–9. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Zhou, Y.; Wang, Z.; Wang, Y.; Zhang, S.; Shen, Z. Emergence of Colistin Resistance Gene mcr-8 and Its Variant in Raoultella ornithinolytica. Front. Microbiol. 2019, 10, 228. [Google Scholar] [CrossRef]
- Stoesser, N.; Mathers, A.J.; E Moore, C.; Day, N.P.J.; Crook, D.W. Colistin resistance gene mcr-1 and pHNSHP45 plasmid in human isolates of Escherichia coli and Klebsiella pneumoniae. Lancet Infect. Dis. 2016, 16, 285–286. [Google Scholar] [CrossRef]
- Ström, G.; Börjesson, S.; Sokerya, S.; Sothyra, T.; Magnusson, U. Detection of mcr-Mediated Colistin Resistance in Escherichia coli Isolates from Pigs in Small-Scale Farms in Cambodia. Antimicrob. Agents Chemother. 2019, 63, 63. [Google Scholar] [CrossRef]
- Yassin, A.K.; Zhang, J.; Wang, J.; Chen, L.; Kelly, P.; Butaye, P.; Lu, G.; Gong, J.; Li, M.; Wei, L.; et al. Identification and characterization of mcr mediated colistin resistance in extraintestinal Escherichia coli from poultry and livestock in China. FEMS Microbiol. Lett. 2017, 364, 364. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hulth, A.; E Nilsson, L.; Börjesson, S.; Chen, B.; Bi, Z.; Wang, Y.; Schwarz, S.; Wu, C. Occurrence of the mobile colistin resistance gene mcr-3 in Escherichia coli from household pigs in rural areas. J. Antimicrob. Chemother. 2018, 73, 1721–1723. [Google Scholar] [CrossRef]
- Kusumoto, M.; Ogura, Y.; Gotoh, Y.; Iwata, T.; Hayashi, T.; Akiba, M. Colistin-resistant mcr-1 –positive pathogenic Escherichia coli in swine, Japan, 2007−2014. Emerg. Infect. Dis. 2016, 22, 1315–1317. [Google Scholar] [CrossRef]
- Ohsaki, Y.; Hayashi, W.; Saito, S.; Osaka, S.; Taniguchi, Y.; Koide, S.; Kawamura, K.; Nagano, Y.; Arakawa, Y.; Nagano, N. First Detection of an Escherichia coli Strain Harboring the mcr-1 Gene in Retail Domestic Chicken Meat in Japan. Jpn. J. Infect. Dis. 2017, 70, 590–592. [Google Scholar] [CrossRef]
- Olaitan, A.O.; Thongmalayvong, B.; Akkhavong, K.; Somphavong, S.; Paboriboune, P.; Khounsy, S.; Morand, S.; Rolain, J.-M. Clonal transmission of a colistin-resistant Escherichia coli from a domesticated pig to a human in Laos: Table 1. J. Antimicrob. Chemother. 2015, 70, 3402–3404. [Google Scholar] [CrossRef]
- Joshi, P.R.; Thummeepak, R.; Paudel, S.; Acharya, M.; Pradhan, S.; Banjara, M.R.; Leungtongkam, U.; Sitthisak, S. Molecular Characterization of Colistin-Resistant Escherichia coli Isolated from Chickens: First Report from Nepal. Microb. Drug Resist. 2019, 25, 846–854. [Google Scholar] [CrossRef]
- Mohsin, M.; Raza, S.; Roschanski, N.; Schaufler, K.; Guenther, S. First description of plasmid-mediated colistin-resistant extended-spectrum β-lactamase-producing Escherichia coli in a wild migratory bird from Asia. Int. J. Antimicrob. Agents 2016, 48, 463–464. [Google Scholar] [CrossRef]
- Azam, M.; Ehsan, I.; Rahman, S.-U.; Saleemi, M.K.; Javed, M.R.; Mohsin, M. Detection of the colistin resistance gene mcr-1 in avian pathogenic Escherichia coli in Pakistan. J. Glob. Antimicrob. Resist. 2017, 11, 152–153. [Google Scholar] [CrossRef]
- Ghafur, A.; Shankar, C.; Soundari, P.G.; Venkatesan, M.; A Thirunarayan, M.; Mani, D.; Veeraraghavan, B. Detection of chromosomal and plasmid-mediated mechanisms of colistin resistance in Escherichia coli and Klebsiella pneumoniae from Indian food samples. J. Glob. Antimicrob. Resist. 2019, 16, 48–52. [Google Scholar] [CrossRef]
- Eiamphungporn, W.; Yainoy, S.; Jumderm, C.; Tan-Arsuwongkul, R.; Tiengrim, S.; Thamlikitkul, V. Prevalence of the colistin resistance gene mcr-1 in colistin-resistant Escherichia coli and Klebsiella pneumoniae isolated from humans in Thailand. J. Glob. Antimicrob. Resist. 2018, 15, 32–35. [Google Scholar] [CrossRef]
- Malhotra-Kumar, S.; Xavier, B.B.; Das, A.J.; Lammens, C.; Hoang, H.T.T.; Pham, N.T.; Goossens, H. Colistin-resistant Escherichia coli harbouring mcr-1 isolated from food animals in Hanoi, Vietnam. Lancet Infect. Dis. 2016, 16, 286–287. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Kawahara, R.; Harada, K.; Teruya, S.; Nakayama, T.; Motooka, D.; Nakamura, S.; Nguyen, P.D.; Kumeda, Y.; Van Dang, C.; et al. The presence of colistin resistance gene mcr-1 and -3 in ESBL producing Escherichia coli isolated from food in Ho Chi Minh City, Vietnam. FEMS Microbiol. Lett. 2018, 365, 365. [Google Scholar] [CrossRef] [PubMed]
- Jajere, S.M.; Hassan, L.; Aziz, S.A.; Zakaria, Z.; Abu, J.; Nordin, F.; Faiz, N.M. Salmonella in native “village” chickens (Gallus domesticus): Prevalence and risk factors from farms in South-Central Peninsular Malaysia. Poult. Sci. 2019, 98, 5961–5970. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals, 4th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018. [Google Scholar]
- Matuschek, E.; Åhman, J.; Webster, C.; Kahlmeter, G. Antimicrobial susceptibility testing of colistin—evaluation of seven commercial MIC products against standard broth microdilution for Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter spp. Clin. Microbiol. Infect. 2018, 24, 865–870. [Google Scholar] [CrossRef]
- World Health Organization. Critically Important Antimicrobials for Human Medicine; WHO: Geneva, Switzerland, 2011; Available online: http://apps.who.int/iris/bitstream/10665/77376/1/9789241504485_eng.pdf (accessed on 12 June 2019).
- Magiorakos, A.-P.; Srinivasan, A.; Carey, R.; Carmeli, Y.; Falagas, M.; Giske, C.; Harbarth, S.; Hindler, J.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef]
- Amoako, D.G.; Somboro, A.M.; Abia, A.L.K.; Molechan, C.; Perrett, K.; Bester, L.A.; Essack, S.Y. Antibiotic Resistance in Staphylococcus aureus from Poultry and Poultry Products in uMgungundlovu District, South Africa, Using the “Farm to Fork” Approach. Microb. Drug Resist. 2020, 26, 402–411. [Google Scholar] [CrossRef]
- Krumperman, P.H. Multiple antibiotic resistance indexing of Escherichia coli to identify high-risk sources of fecal contamination of foods. Appl. Environ. Microbiol. 1983, 46, 165–170. [Google Scholar] [CrossRef]


| Antimicrobial Agents | No. Tested | Antibiogram of Salmonella Isolates | ||
|---|---|---|---|---|
| Resistant (%) | Intermediate (%) | Sensitive (%) | ||
| Gentamicin | 34 | 00 | 1 (2.9) | 33 (97.1) |
| Amoxicillin-Clavulanate | 34 | 2 (5.9) | 2 (5.9) | 30 (88.2) |
| Nitrofurantoin | 34 | 4 (11.8) | 00 | 30 (83.8) |
| Ciprofloxacin | 34 | 00 | 00 | 34 (100) |
| Kanamycin | 34 | 1 (2.9) | 2 (5.9) | 31 (91.2) |
| Trimethoprim | 34 | 7 (20.6) | 00 | 27 (79.4) |
| Norfloxacin | 34 | 00 | 1 (2.9) | 33 (97.1) |
| Tetracycline | 34 | 12 (35.3) | 1 (2.9) | 21 (61.8) |
| Nalidixic acid | 34 | 5 (14.7) | 00 | 29 (85.3) |
| Chloramphenicol | 34 | 4 (11.8) | 00 | 30 (88.2) |
| Ampicillin | 34 | 6 (17.6) | 00 | 28 (82.4) |
| Cefotaxime | 34 | 1 (2.9) | 00 | 33 (97.1) |
| Streptomycin | 34 | 12 (35.3) | 11 (32.4) | 11 (32.4) |
| Sulfonamides | 34 | 10 (29.4) | 00 | 24 (70.6) |
| Ceftiofur | 34 | 0 | 1 (2.9) | 33 (97.1) |
| Colistin 1 | 34 | 5 (14.7) | 0 | 29 (85.3) |
| Sources | Resistance Profiles | Salmonella Serovars | Colistin MIC 1 | MAR Index 2 | |
|---|---|---|---|---|---|
| Conc. (mg/L) | R/I/S 3 | ||||
| Cloacal swab | AmpTeWS3SFNaCt | Salmonella spp. | 4 | R | ≥0.2 |
| Flies | TeWS3SFCtxAmc | S. Molade | 2 | S | ≥0.2 |
| Cloacal swab | AmpS3WNaCn | S. Weltevreden | 8 | R | ≥0.2 |
| Cloacal swab | AmpS3WNaCn | S. Weltevreden | 8 | R | ≥0.2 |
| Drinking water | AmpTeS3SCn | S. Albany | 16 | R | ≥0.2 |
| Cloacal swab | TeWS3 | S. Corvallis | 0.25 | S | ≥0.2 |
| Cloacal swab | TeS3S | Salmonella spp. | 2 | S | ≥0.2 |
| Drinking water | TeS3S | S. Albany | 2 | S | ≥0.2 |
| Feed | TeNa | S. Weltevreden | 2 | S | <0.2 |
| Drinking water | TeNa | Salmonella spp. | 2 | S | <0.2 |
| Drinking water | AmpTe | S. Weltevreden | 2 | S | <0.2 |
| Drinking water | AmpS3 | S. Weltevreden | 0.5 | S | <0.2 |
| Cloacal swab | TeS | S. Molade | 0.5 | S | <0.2 |
| Cloacal swab | TeW | S. Typhimurium | 4 | R | <0.2 |
| Cloacal swab | TeW | S. Agona | 2 | S | <0.2 |
| Cloacal swab | Ct | S. Enteritidis | 2 | S | <0.2 |
| Cloacal swab | Te | Salmonella spp. | 0.25 | S | <0.2 |
| Cloacal swab | Te | S. Typhimurium | 0.25 | S | <0.2 |
| Cloacal swab | Te | S. Typhimurium | 0.25 | S | <0.2 |
| Feed | Amp | S. Typhimurium | 0.5 | S | <0.2 |
| Feed | Amp | S. Albany | 0.25 | S | <0.2 |
| Cloacal swab | S | S. Agona | 0.25 | S | <0.2 |
| Cloacal swab | S | S. Agona | 0.25 | S | <0.2 |
| Cloacal swab | S | S. Agona | 0.25 | S | <0.2 |
| Cloacal swab | S | S. Agona | 0.25 | S | <0.2 |
| Sources | No. of MDR Salmonella | % Positive 1 |
|---|---|---|
| Cloacal swabs (n = 17) | 5 | 29.4% |
| Drinking water (n = 9) | 2 | 22.2% |
| Flies (n = 3) | 1 | 33.3% |
| Variables | Frequency | MDR (%) | OR (95% CI) 1 | p-Value |
|---|---|---|---|---|
| Flock size (number of birds) | ||||
| <500 | 22 | 6 (27.3) | 1.84 (0.32–15.52) | 0.533 |
| ≥500 | 12 | 2 (16.7) | Reference | - |
| Poultry production system | ||||
| Free-range | 22 | 5 (22.7) | 0.78 (0.14, 4.90) | 0.779 |
| Semi-intensive/Intensive | 12 | 3 (25.0) | Reference | - |
| Use of antibiotics in the farm | ||||
| Yes | 20 | 6 (30.0) | 1.96 (0.34–16.20) | 0.478 |
| No | 14 | 2 (14.3) | Reference |
| Serovars Isolated | Sources | Number (%) Total n = 34 |
|---|---|---|
| Salmonella Weltevreden | Cloacal swabs, feed, water | 7 (20.6) |
| Salmonella Typhimurium | Cloacal swabs, feeds | 6 (17.6) |
| Salmonella Agona | Cloacal swabs | 6 (17.6) |
| Salmonella Enteritidis | Cloacal swabs, water, feeds | 3 (8.8) |
| Salmonella Albany | Water, feeds | 3 (8.8) |
| Salmonella Molade | Cloacal swabs, flies | 2 (5.9) |
| Salmonella Corvallis | Cloacal swabs | 2 (5.9) |
| Salmonella Schleissheim | Flies | 1 (2.9) |
| Salmonella spp. | Cloacal swabs, water | 4 (11.8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohammed Jajere, S.; Hassan, L.; Zakaria, Z.; Abu, J.; Abdul Aziz, S. Antibiogram Profiles and Risk Factors for Multidrug Resistance of Salmonella enterica Recovered from Village Chickens (Gallus gallus domesticus Linnaeus) and Other Environmental Sources in the Central and Southern Peninsular Malaysia. Antibiotics 2020, 9, 701. https://doi.org/10.3390/antibiotics9100701
Mohammed Jajere S, Hassan L, Zakaria Z, Abu J, Abdul Aziz S. Antibiogram Profiles and Risk Factors for Multidrug Resistance of Salmonella enterica Recovered from Village Chickens (Gallus gallus domesticus Linnaeus) and Other Environmental Sources in the Central and Southern Peninsular Malaysia. Antibiotics. 2020; 9(10):701. https://doi.org/10.3390/antibiotics9100701
Chicago/Turabian StyleMohammed Jajere, Saleh, Latiffah Hassan, Zunita Zakaria, Jalila Abu, and Saleha Abdul Aziz. 2020. "Antibiogram Profiles and Risk Factors for Multidrug Resistance of Salmonella enterica Recovered from Village Chickens (Gallus gallus domesticus Linnaeus) and Other Environmental Sources in the Central and Southern Peninsular Malaysia" Antibiotics 9, no. 10: 701. https://doi.org/10.3390/antibiotics9100701
APA StyleMohammed Jajere, S., Hassan, L., Zakaria, Z., Abu, J., & Abdul Aziz, S. (2020). Antibiogram Profiles and Risk Factors for Multidrug Resistance of Salmonella enterica Recovered from Village Chickens (Gallus gallus domesticus Linnaeus) and Other Environmental Sources in the Central and Southern Peninsular Malaysia. Antibiotics, 9(10), 701. https://doi.org/10.3390/antibiotics9100701





