Exclusive Production of Gentamicin C1a from Micromonospora purpurea by Metabolic Engineering
Abstract
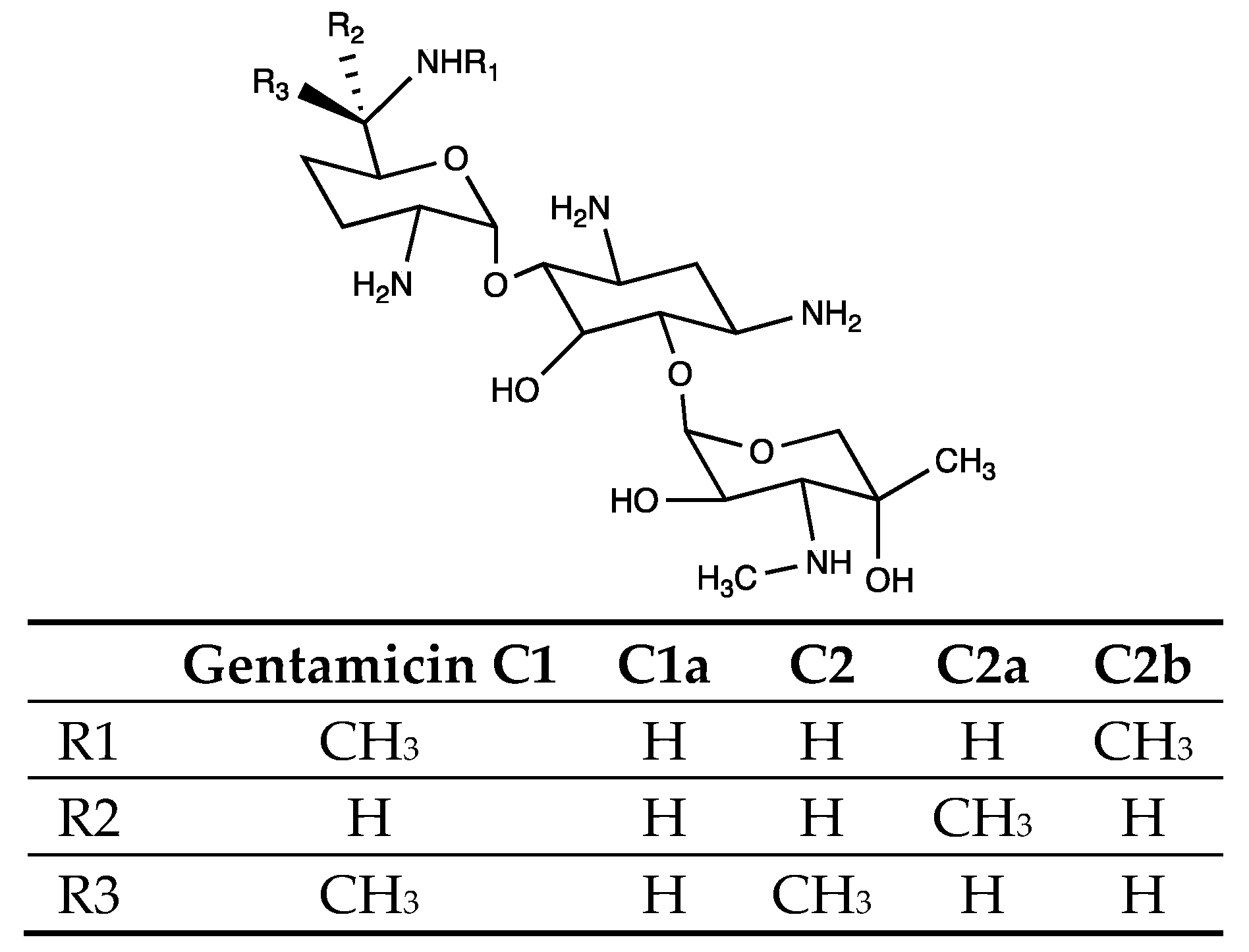
1. Introduction
2. Material and Methods
2.1. Strains and Plasmids
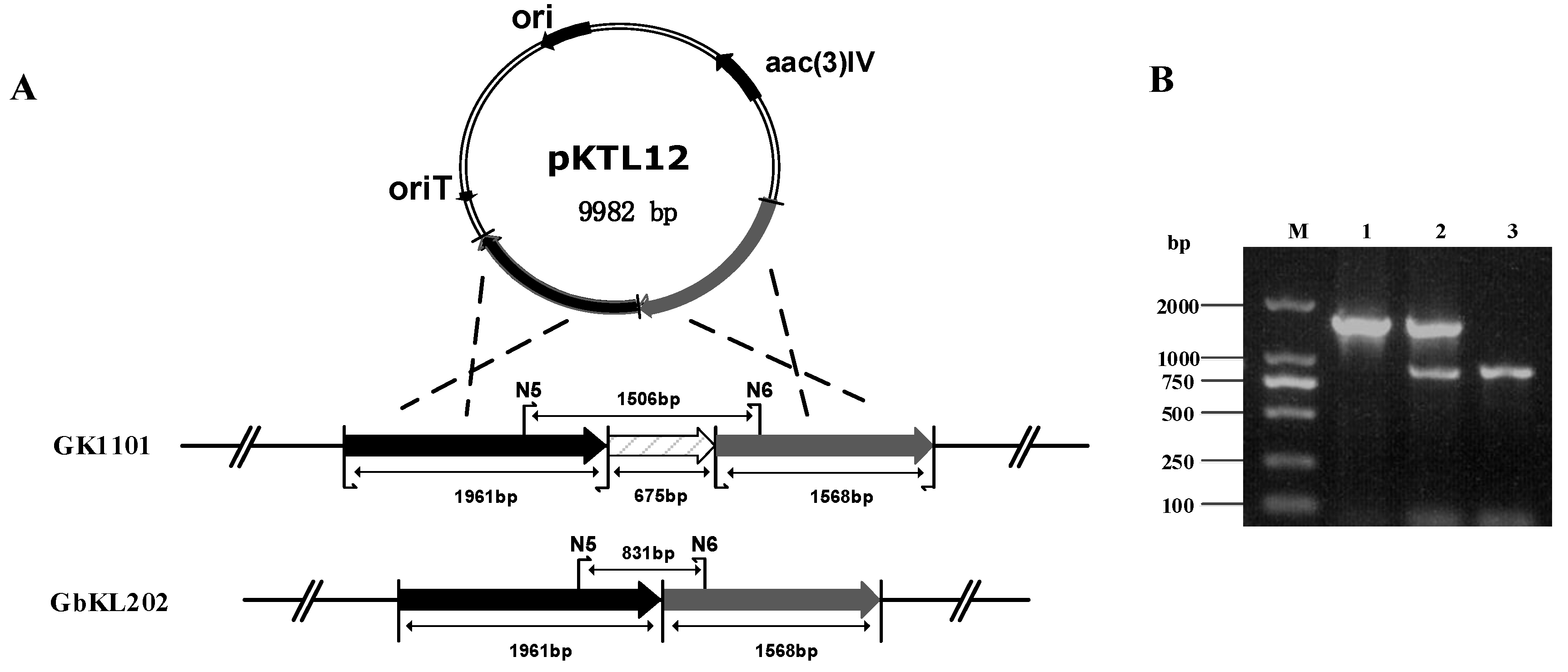
2.2. Construction of the Disruption Plasmid
2.3. Construction of the Disruption Strain
2.4. Antibiotic Separation and Analysis
2.5. Measurement of Titers of Gentamicin
- θ: Ratio of actual titer over estimated titer;
- S1: Diameter of clear zones with 5 u/mL gentamicin;
- S2: Diameter of clear zones with 10 u/mL gentamicin;
- T1: Diameter of clear zones with estimated 5 u/mL supernatant;
- T2: Diameter of clear zones with estimated 10 u/mL supernatant;
- I: I = 0.301
3. Results
3.1. Construction of the Disruption Strain
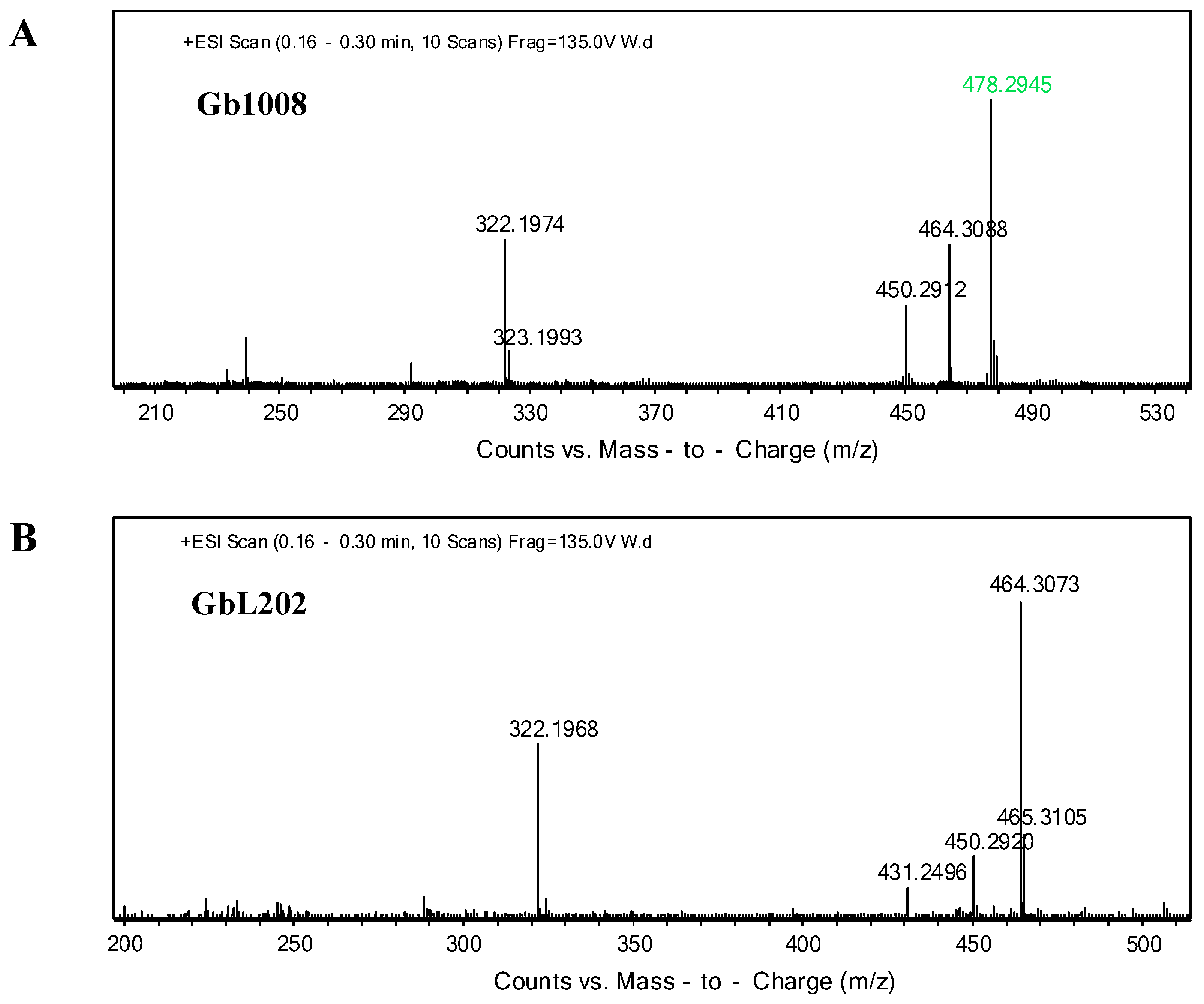
3.2. Analysis of the Metabolites
3.3. genL is Involved in Converting C2 to C1 in M. Purpurea
4. Discussions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weinstein, M.J.; Luedemann, G.M.; Oden, E.M.; Wagman, G.H.; Rosselet, J.P.; Marquez, J.A.; Coniglio, C.T.; Charney, W.; Herzog, H.L.; Black, J. Gentamicin, a new antibiotic complex from micromonospora. J. Med. Chem. 1963, 6, 463–464. [Google Scholar] [CrossRef] [PubMed]
- Rodriquez, M.; Cretoso, D.S.; Euterpio, M.A.; Russo, P.; Crescenzi, C.; Aquino, R.P. Fast determination of underivatized gentamicin C components and impurities by LC-MS using a porous graphitic carbon stationary phase. Anal. Bioanal. Chem. 2015, 407, 7691–7701. [Google Scholar] [CrossRef] [PubMed]
- Bezdjian, A.; Mujica-Mota, M.A.; Devic, S.; Daniel, S.J. The effect of radiotherapy on gentamicin ototoxicity: An animal model. Otolaryngol. Head Neck Surg. 2015, 152, 1094–1101. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Sone, M.; Umemura, M.; Nabeshima, T.; Nakashima, T.; Hellstrom, S. Comparisons of cochleotoxicity among three gentamicin compounds following intratympanic application. Acta Otolaryngol. 2008, 128, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Boehr, D.D.; Jenkins, S.I.; Wright, G.D. The molecular basis of the expansive substrate specificity of the antibiotic resistance enzyme aminoglycoside acetyltransferase-6′-aminoglycoside phosphotransferase-2″. The role of ASP-99 as an active site base important for acetyl transfer. J. Biol. Chem. 2003, 278, 12873–12880. [Google Scholar] [CrossRef] [PubMed]
- Boehr, D.D.; Daigle, D.M.; Wright, G.D. Domain-domain interactions in the aminoglycoside antibiotic resistance enzyme AAC (6′)-APH (2″). Biochemistry 2004, 43, 9846–9855. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Zhao, M.; Liu, J. Aminoglycoside antibiotics 89-07: Semisythetic and structure measurement. Chin. J. Antibiot. 1995, 20, 401–406. [Google Scholar]
- Chaudhary, M.; Kesava, N.G.; Kumar, S.; Payasi, A. Comparative antibacterial activity of a novel semisynthetic antibiotic: Etimicin sulphate and other aminoglycosides. World. J. Microbiol. Biotechnol. 2012, 28, 3365–3371. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Z.; Wu, L.; Zhang, X.; Wang, H.; Ye, W.; Li, P. Isolation and structure characterization of related impurities in etimicin intermediate P1 by LC/ESI-MSn and NMR. J. Pharm. Biomed. Anal. 2014, 97, 97–102. [Google Scholar] [CrossRef]
- Yoshizawa, S.; Fourmy, D.; Puglisi, J.D. Structural origins of gentamicin antibiotic action. EMBO J. 1998, 17, 6437–6448. [Google Scholar] [CrossRef]
- Hong, W.; Yan, L. Identification of gntK, a gene required for the methylation of purpurosamine C-6′ in gentamicin biosynthesis. J. Gen. Appl. Microbiol. 2012, 58, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; McCarty, R.M.; Ogasawara, Y.; Liu, Y.N.; Mansoorabadi, S.O.; LeVieux, J.; Liu, H.W. GenK-catalyzed C-6′ methylation in the biosynthesis of gentamicin: Isolation and characterization of a cobalamin-dependent radical SAM enzyme. J. Am. Chem. Soc. 2013, 135, 8093–8096. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Chen, J.; Wang, C.; Li, J.A.; Tang, Y.; Chen, D.; Liu, W. Characterization of a key aminoglycoside phosphotransferase in gentamicin biosynthesis. Bioorg. Med. Chem. Lett. 2013, 23, 1438–1441. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Ni, X.; Ren, J.; Gao, H.; Wang, D.; Xia, H. Biosynthesis of Epimers C2 and C2a in the Gentamicin C Complex. Chembiochem 2015, 16, 1933–1942. [Google Scholar] [CrossRef] [PubMed]
- Ni, X.P.; Sun, Z.P.; Zhang, H.Y.; He, H.; Ji, Z.X.; Xia, H.Z. Genetic engineering combined with random mutagenesis to enhance G418 production in Micromonospora echinospora. J. Ind. Microbiol. Biot. 2014, 41, 1383–1390. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Huang, F.; Huang, C.; Duan, X.; Jian, X.; Leeper, F.; Deng, Z.; Leadlay, P.F.; Sun, Y. Specificity and promiscuity at the branch point in gentamicin biosynthesis. Chem. Biol. 2014, 21, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Guo, J.; Reva, A.; Huang, F.; Xiong, B.; Liu, Y.; Deng, Z.; Leadlay, P.F.; Sun, Y. Methyltransferases of gentamicin biosynthesis. Proc. Natl. Acad. Sci. USA 2018, 115, 1340–1345. [Google Scholar] [CrossRef]
- Hobbs, G.; Frazer, C.M.; Gardner, D.C.J.; Cullum, J.A.; Oliver, S.G. Dispersed growth of Streptomyces in liquid culture. Appl. Microbiol. Biotechnol. 1989, 31, 272–277. [Google Scholar] [CrossRef]
- Russell, D.W.; Sambrook, J. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2001. [Google Scholar]
- Hong, W.; Yan, S. Engineering Streptomyces tenebrarius to synthesize single component of carbamoyl tobramycin. Lett. Appl. Microbiol. 2012, 55, 33–39. [Google Scholar] [CrossRef]
- Wan, Y.; Hong, W.; Shi, X. Study on the function of sisI, a biosynthetic gene of Sisomicin. J. Yanbian Univ. 2016, 42, 130–135. [Google Scholar]
- Pharmacopoeia Commission of the Ministry of Health of the People’s Republic of China. Pharmacopoeia of the People’s Republic of China; China Medical Science and Technology Press: Beijing, China, 2010. [Google Scholar]
- Park, J.W.; Hong, J.S.; Parajuli, N.; Koh, H.S.; Park, S.R.; Lee, M.O.; Lim, S.K.; Yoon, Y.J. Analytical profiling of biosynthetic intermediates involved in the gentamicin pathway of Micromonospora echinospora by high-performance liquid chromatography using electrospray ionization mass spectrometric detection. Anal. Chem. 2007, 79, 4860–4869. [Google Scholar] [CrossRef] [PubMed]


| Strains or Plasmids | Relevant Characteristic | Reference or Source | |
|---|---|---|---|
| Strains | M. purpurea Gb1008 | Wild-Type Strain | Our lab |
| GK1101 | M. purpurea Gb1008 with ΔgenK | Our Lab | |
| E. coli Top10 | F-, mcrAΔ(mrr-hsd RMS-mcrBC), Δ(ara-leu)769, recA1, φ80, araΔ139 lacZΔM15 △lacX74 galK galU, rps, nupG, (Strr) endA1 | Thermo Fisher | |
| E. coli ET12567/pUZ8002 | Nonmethylating E. coli, CmL R; Tra+, OriTRK2 derivative, Kan R | Our lab | |
| Bacillus pumilus | Used as detecting bacteria | Our lab | |
| GbL201 | Single crossover mutant of M. purpurea Gb1008, with plasmid pKTL12 | This study | |
| GbL202 | M. purpurea Gb1008 with genL (orf6255) disrupted | This study | |
| GbKL201 | Single crossover mutant of GK1101, with plasmid pKTP12 | This study | |
| GbKL202 | GK1101 with genL(orf6255) disrupted | This study | |
| Plasmids | pMD19-T | Amp R, Cloning vector for E. coli | TaKaRa |
| pKC1139 | 6.5 kb, E. coli–Streptomyces shutter vector, Streptomyces replicon pSG5 ori E. coli replicon pUC oriT lacZa MCS Rep ts Ap R | Our lab | |
| pTL1 | pMD19-T containing upstream fragments, Amp R | This study | |
| pTL2 | pMD19-T containing downstream fragments, Amp R | This study | |
| pKTL12 | pKC1139 containing upstream and downstream fragments, used for genL (orf6255) disruption, AP R | This study | |
| Name | Sequence a |
|---|---|
| L1 | 5′-TTAGAATTCAGCAGGCGGGCCTCGTCGAGAAAGCGTT-3’ |
| L2 | 5’-GGTCTAGAGATCGGAGATGCTCAAGATGG-3’ |
| L3 | 5’-TTTCTAGATCTACTCCGTCGGCGAGTCG-3’ |
| L4 | 5′-GGGAAGCTTAAAGTGGGCGACCACCAAGCACAAGAAG-3’ |
| L5 | 5′-TTCGAGATCGTCAAGTACCGGGTC-3’ |
| L6 | 5’-GGATGATGATGGAGATGGGCTTCG-3’ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, Z.; Shi, X.; Lian, R.; Wang, W.; Hong, W.; Guo, S. Exclusive Production of Gentamicin C1a from Micromonospora purpurea by Metabolic Engineering. Antibiotics 2019, 8, 267. https://doi.org/10.3390/antibiotics8040267
Wei Z, Shi X, Lian R, Wang W, Hong W, Guo S. Exclusive Production of Gentamicin C1a from Micromonospora purpurea by Metabolic Engineering. Antibiotics. 2019; 8(4):267. https://doi.org/10.3390/antibiotics8040267
Chicago/Turabian StyleWei, Zeng, Xianai Shi, Rong Lian, Weibin Wang, Wenrong Hong, and Shaobin Guo. 2019. "Exclusive Production of Gentamicin C1a from Micromonospora purpurea by Metabolic Engineering" Antibiotics 8, no. 4: 267. https://doi.org/10.3390/antibiotics8040267
APA StyleWei, Z., Shi, X., Lian, R., Wang, W., Hong, W., & Guo, S. (2019). Exclusive Production of Gentamicin C1a from Micromonospora purpurea by Metabolic Engineering. Antibiotics, 8(4), 267. https://doi.org/10.3390/antibiotics8040267





