The Use of Selected Bacteria and Yeasts to Control Vibrio spp. in Live Food
Abstract
1. Introduction
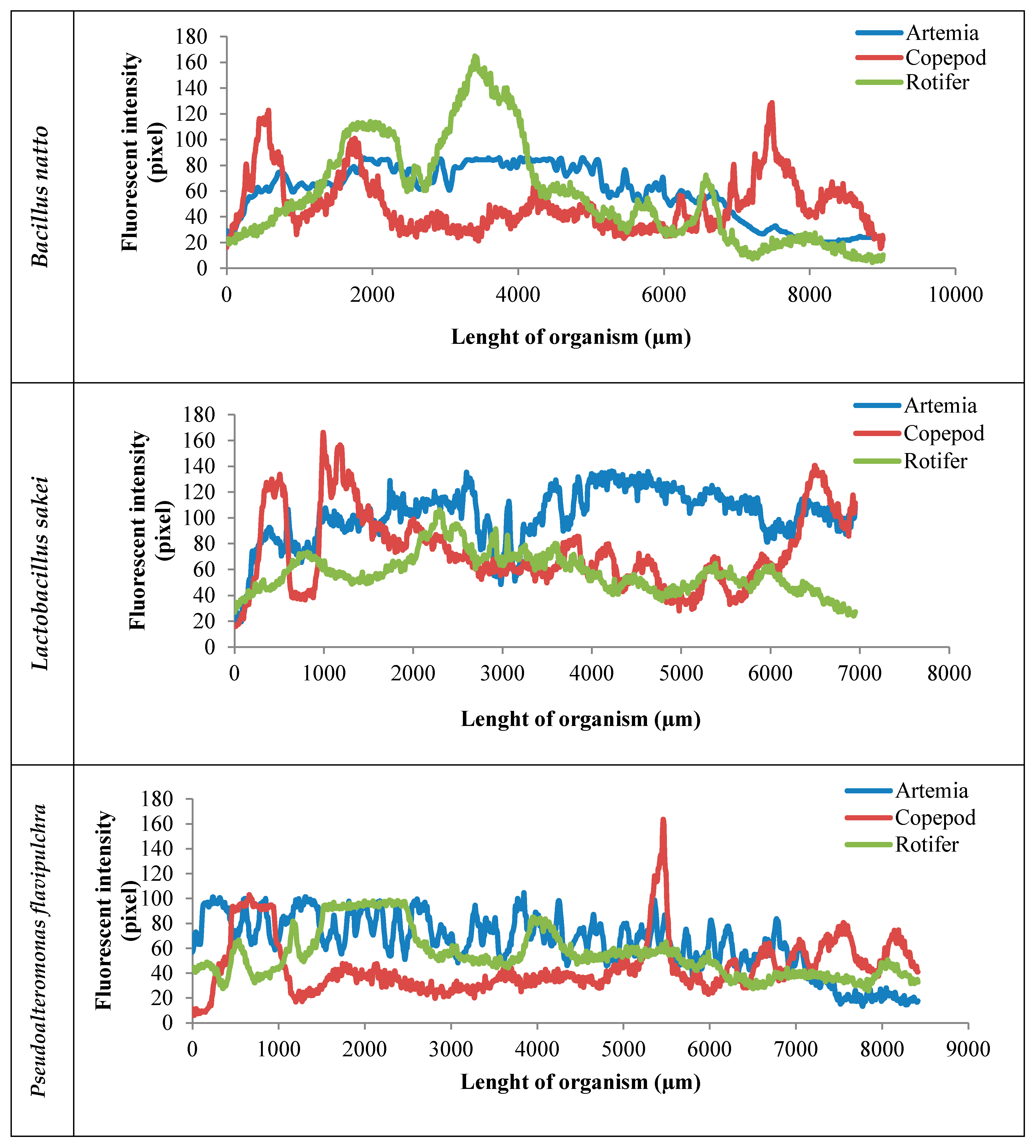
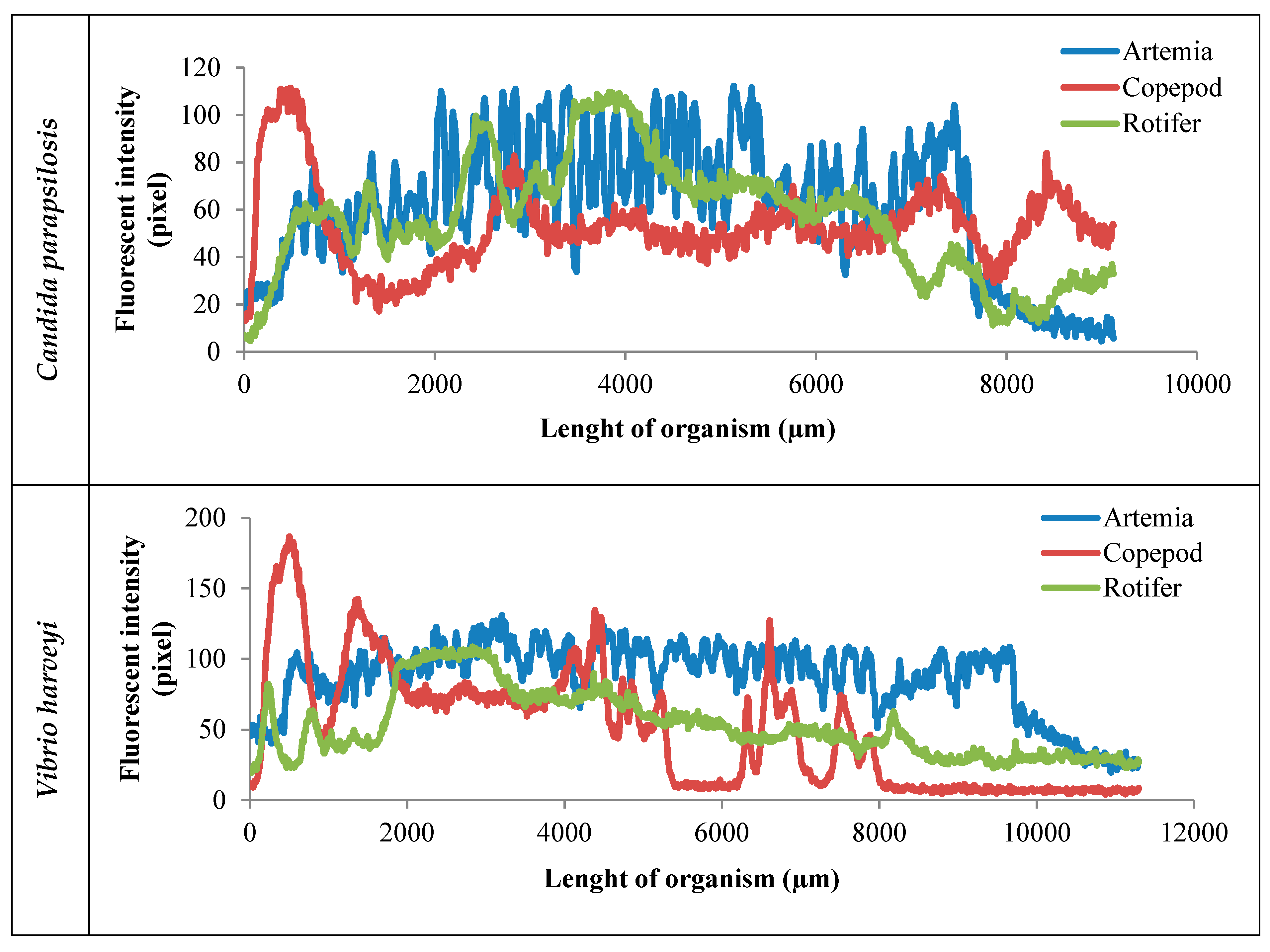
2. Results
3. Discussion
4. Materials and Methods
4.1. Organisms and Culture Conditions
- Tryptic soy broth (TSB) for Bacillus strains. Composition (g/L): trypton—17, soya peptone —3, NaCl—5, K2HPO4—2.5, glucose—2.5. pH ~ 7.3; Sterilization—15 min at 121 °C.
- Man, Rogosa, and Sharpe broth (MRS) for Lactobacillus strains. Composition (g/L): protose peptone—10, beef extract—10, yeast extract—5, dextrose—3, ammonium citrate—2, sodium acetate—5, magnesium sulfate—0.1, manganese sulfate—0.05, dipotassium phosphate—2. pH ~ 6.2 and Tween 80 1 mL−1; Sterilization—15 min at 121 °C.
- Yeast extracts peptone dextrose broth (YPD) for yeast strains. Composition (g/L): yeast extract—10, peptone—20, dextrose—20. pH ~ 5.5; Sterilization—30 min at 115 °C.
- E2216 broth. Composition (g/L) for Marine Bacterium strains and Vibrio species: Peptone—5, yeast extract—1, ferric phosphate—0.1. pH ~ 7.5–7.7; Sterilization—15 min at 121 °C.
4.2. Microbial Susceptibility Assay
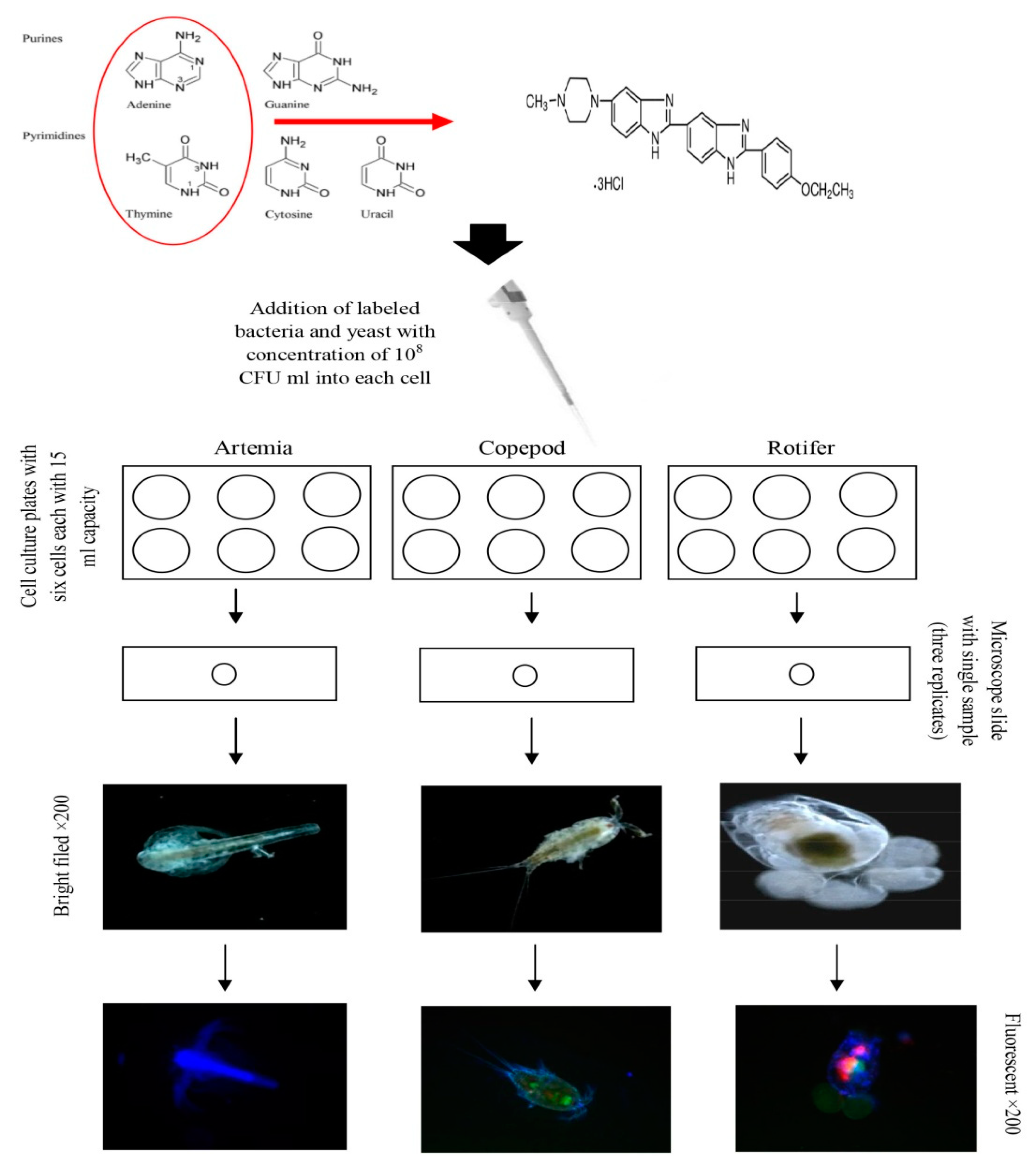
4.3. Microbial Fluorescent Labeling
4.4. Live Food Culture
4.5. Loading of Vibrio Speciesin Live Food
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dhont, K.; Roets, A.; Van-Hiel, A. The intergenerational transmission of need for closure underlies the transmission of authoritarianism and anti-immigrant prejudice. Personal. Individ. Differ. 2013, 54, 779–784. [Google Scholar] [CrossRef]
- Sorgeloos, P.; Dhert, P.; Candreva, P. Use of the brine shrimp, Artemia spp., in marine fish larviculture. Aquaculture 2001, 200, 147–159. [Google Scholar] [CrossRef]
- Yu, L.; Zhai, Q.; Zhu, J.; Zhang, C.; Li, T.; Liu, X.; Zhao, J.; Zhang, H.; Tian, F.; Chen, W. Dietary Lactobacillus plantarum supplementation enhances growth performance and alleviates aluminum toxicity in tilapia. Ecotoxicol. Environ. Saf. 2017, 143, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Lavilla-Pitogo, C.R.; Leano, E.M.; Paner, M.G. Mortalities of pond-cultured juvenile shrimp, Penaeus monodon, associated with dominance of luminescent vibrios in the rearing environment. Aquaculture 1998, 164, 337–349. [Google Scholar] [CrossRef]
- Tang, K.W. Copepods as microbial hotspots in the ocean: Effects of host feeding activities on attached bacteria. Aquat. Microb. Ecol. 2005, 38, 31–40. [Google Scholar] [CrossRef]
- Soto-Rodriguez, S.A.; Roque, A.; Lizarraga-Partida, M.L.; Guerra-Flores, A.L.; Gomez-Gil, B. Virulence of luminous vibrios to Artemia franciscana nauplii. Dis. Aquat. Org. 2003, 53, 231–240. [Google Scholar] [CrossRef]
- Saifedden, G.; Farinazleen, G.; Nor-khaizura, A.; Kayali, A.Y.; Nakaguchi, Y.; Nishibuchi, M.; Son, R. Antibiotic Susceptibility profile of Vibrio parahaemolyticus isolated from shrimp in Selangor, Malaysia. Int. Food Res. J. 2016, 23, 2732–2736. [Google Scholar]
- Cabello, F.C. Heavy use of prophylactic antibiotics in aquaculture: A growing problem for human and animal health and for the environment. Environ. Microbiol. 2006, 8, 1137–1144. [Google Scholar] [CrossRef]
- Karunasagar, I.; Pai, R.; Malathi, G.R.; Karunasagar, I. Mass mortality of Penaeus monodon larvae due to antibiotic resistant Vibrio harveyi infection. Aquaculture 1994, 128, 203–209. [Google Scholar] [CrossRef]
- Okoh, A.I.; Igbinosa, E.O. Antibiotic susceptibility profiles of some Vibrio strains isolated from wastewater final effluents in a rural community of the Eastern Cape Province of South Africa. BMC Microbiol. 2010, 10, 143. [Google Scholar] [CrossRef]
- Silvester, R.; Alexander, D.; Ammanamveetil, M.H.A. Prevalence, antibiotic resistance, virulence and plasmid profiles of Vibrio parahaemolyticus from a tropical estuary and adjoining traditional prawn farm along the southwest coast of India. Ann. Microbiol. 2015, 65, 2141–2149. [Google Scholar] [CrossRef]
- Sahandi, J.; Jafariyan, H.; Roozbehfar, R.; Babaei, S.; Dehestani, M. The use of two enrichment forms (Brachionus plicatilis enrichment and rearing water enrichment) with probiotic bacilli spore on growth and survival of silver carp (Hypophthalmichthys molitrix). Iran. J. Vet. Res. 2012, 13, 289–295. [Google Scholar]
- Luna-Gonzalez, A.; Quinonez-Zuniga, D.; Fierro-Coronado, J.A.; Gonzalez-Ocampo, H.A.; Campa-Cordova, A.I.; Flores-Miranda, M.D.C.; Peraza-Gomez, V. Effect of Pediococcus parvulus and Candida parapsilosis on growth and survival of tilapia, Oreochromis niloticus and Oreochromis sp. Afr. J. Microbiol. Res. 2013, 7, 2976–2982. [Google Scholar]
- Ouwehand, A.C.; Isolauri, E.; Kirjavainen, P.V.; Tölkkö, S.; Salminen, S.J. The mucus binding of Bifidobacterium lactis Bb12 is enhanced in the presence of Lactobacillus GG and Lacto delbrueckii sub sp bulgaricus. Lett. Appl. Microbiol. 2000, 30, 10–13. [Google Scholar] [CrossRef]
- Xu, H.M.; Rong, Y.J.; Zhao, M.X.; Song, B.; Chi, Z.M. Antibacterial activity of the lipopetides produced by Bacillus amyloliquefaciens M1 against multidrug-resistant Vibrio spp. isolated from diseased marine animals. Appl. Microbiol. Biotechnol. 2013, 97, 1711–1723. [Google Scholar] [CrossRef]
- Younis, G.; Awad, A.; Dawod, R.E.; Yousef, N.E. Antimicrobial activity of yeasts against some pathogenic bacteria. Vet. World. 2017, 10, 979–983. [Google Scholar] [CrossRef]
- Olsson, J.C.; Westerdahl, A.; Conway, P.L.; Kjelleberg, S. Intestinal colonization potential of Turbot (Scophthalmus maximus) and Dab (Limanda limanda)—Associated bacteria with inhibitory effects against Vibrio anguillarum. Appl. Environ. Microbiol. 1992, 58, 551–556. [Google Scholar]
- Makridis, P.; Fjellheim, A.J.; Skjermo, J.; Vadstein, O. Control of the bacterial flora of Brachionus plicatilis and Artemia franciscana by incubation in bacterial suspensions. Aquaculture 2000, 185, 207–216. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.; Xiao, P.; Zhu, W.; Mo, Z. The ability of marine Bacillus spp. isolated from fish gastrointestinal tract and culture pond sediment to inhibit growth of aquatic pathogenic bacteria. Iran. J. Fish. Sci. 2016, 15, 701–714. [Google Scholar]
- Georgieva, R.; Yocheva, L.; Tserovska, L.; Zhelezova, G.; Stefanova, N.; Atanasova, A.; Danguleva, A.; Ivanova, G.; Karapetkov, N.; Rumyan, N.; et al. Antimicrobial activity and antibiotic susceptibility of Lactobacillus and Bifidobacterium spp. intended for use as starter and probiotic cultures. Biotechnol. Biotechnol. Equip. 2015, 29, 84–91. [Google Scholar] [CrossRef]
- Chaurasia, B.; Pandey, A.; Palni, L.M.S.; Trivedi, P.; Kumar, B.; Colvin, N. Diffusible and volatile compounds produced by an antagonistic Bacillus subtilis strain cause structural deformations in pathogenic fungi in vitro. Microbiol. Res. 2005, 160, 75–81. [Google Scholar] [CrossRef]
- Ghadiri, M.; Kazemi, S.; Heidari, B.; Rassa, M. Bioactivity of aqueous and organic extracts of sea cucumber Holothuria leucospilota (Brandt 1835) on pathogenic Candida and Streptococci. Int. Aquat. Res. 2018, 10, 31–34. [Google Scholar] [CrossRef]
- Gatesoupe, F.J. Live yeasts in the gut: Natural occurrence, dietary introduction, and their effects on fish health and development. Aquaculture 2007, 267, 20–30. [Google Scholar] [CrossRef]
- Gedek, B.R. Adherence of Escherichia coli serogroup O 157 and the Salmonella typhimurium mutant DT 104 to the surface of Saccharomyces boulardii. Mycoses 1999, 42, 261–264. [Google Scholar] [CrossRef]
- Soccol, C.R.; Vandenberghe, L.P.; Spier, M.R.; Medeiros, A.B.; Yamaguishi, C.T.; Lindner, J.D.; Ashok, P.; Vanete, T.S. The potential of probiotics: A review. Food Technol. Biotechnol. 2010, 48, 413–434. [Google Scholar]
- Aras-Hisar, S.; Kaban, G.; Hisar, O.; Yanik, T.; Kaya, M. Effect of Lactobacillus sakei Lb706 on behavior of Listeria monocytogenes in vacuum packed rainbow trout fillets. Turk. J. Vet. Anim. Sci. 2005, 29, 1039–1044. [Google Scholar]
- Koga, T.; Mizobe, T.; Takumi, K. Antibacterial activity of Lactobacillus species against Vibrio species. Microbiol. Res. 1998, 153, 271–275. [Google Scholar] [CrossRef]
- Yu, M.; Wang, J.; Tang, K.; Shi, X.; Wang, S.; Zhu, W.M.; Zhang, X.H. Purification and characterization of antibacterial compounds of Pseudoalteromonas flavipulchra JG1. Microbiology 2012, 158, 835–842. [Google Scholar] [CrossRef]
- Jin, G.; Wang, S.; Yu, M.; Yan, S.; Zhang, X. Identification of a marine antagonistic strain JG1 and establishment of a polymerase chain reaction detection technique based on the gyrB gene. Aquac. Res. 2010, 41, 1867–1874. [Google Scholar] [CrossRef]
- Isnansetyo, A.; Istiqomah, I.; Muhtadi-Sinansari, S.; Hernawan, R.; Triyanto, K.; Widada, J. A potential bacterial bio-control agent, strain S2V2 against pathogenic marine Vibrio in aquaculture. World J. Microbiol. Biotechnol. 2009, 25, 1103–1113. [Google Scholar] [CrossRef]
- Richards, G.P.; Watson, M.A.; Needleman, D.S.; Uknalis, J.; Boyd, E.F.; Fay, J.P. Mechanisms for Pseudoalteromonas piscicida induced killing of Vibrios and other bacterial pathogens. J. Appl. Environ. Microbiol. 2017, 83, e00175-17. [Google Scholar] [CrossRef]
- Parta, S.K.; Mohamed, K.S. Enrichment of Artemia nauplii with the probiotic yeast Sacharomyces boulardii and its resistance against a pathogenic Vibrio. Aquac. Int. 2003, 11, 505–514. [Google Scholar]
- Finaly, B.B.; Falkow, S. Common themes in microbial Pathogenicity revisited. Microbiol. Mol. Biol. Rev. 1997, 61, 136–169. [Google Scholar]
- Li, F.; Shi, J.; Cheung, S.G.; Shin, P.K.S.; Liu, X.; Sun, Y.; Mu, F. The combined effects of elevated pCO2 and food availability on Tigriopus japonicus Mori larval development, reproduction, and superoxide dismutase activity. Mar. Pollut. Bull. 2017, 126, 623–628. [Google Scholar] [CrossRef]
- Grossart, H.P.; Kiørboe, T.; Tang, K.W.; Allgaier, M.; Yam, E.M.; Ploug, H. Interactions between marine snow and heterotrophic bacteria: Aggregate formation and microbial dynamics. Aquat. Microb. Ecol. 2006, 42, 19–26. [Google Scholar] [CrossRef]
- Moller, E.F.; Riemann, L.; Søndergaard, M. Bacteria associated with copepods: Abundance, activity and community composition. Aquat. Microb. Ecol. 2007, 47, 99–106. [Google Scholar] [CrossRef]
- Tang, K.W.; Turk, V.; Grossart, H.P. Linkage between crustacean zooplankton and aquatic bacteria. Aquat. Microb. Ecol. 2010, 61, 261–277. [Google Scholar] [CrossRef]
- Liu, C.H.; Chiu, C.S.; Ho, P.L.; Wang, S.W. Improvement in the growth performance of white shrimp, Litopenaeus vannamei, by a protease-producing probiotic, Bacillus subtilis E20, from natto. J. Appl. Microbiol. 2009, 107, 1031–1041. [Google Scholar] [CrossRef]
- Queiroz, F.; Boyd, C. Effects of bacterial inoculums in channel catfish ponds. J. World. Aquac. Soc. 1998, 29, 67–73. [Google Scholar] [CrossRef]
- Srisapoome, P.; Areechon, N. Efficacy of viable Bacillus pumilus isolated from farmed fish on immune responses and increased disease resistance in Nile tilapia (Oreochromis niloticus): Laboratory and on-farm trials. J. Fish. Shellfish. Immunol. 2017, 67, 199–210. [Google Scholar] [CrossRef]
- Truong, T.H.T.; Tri, N.N.; Quy, O.M.; Fotedar, R.; Kannika, K.; Unajak, S.; Areechon, N. Effects of the dietary supplementation of mixed probiotic spores of Bacillus amyloliquefaciens54A, and Bacillus pumilus47B on growth, innate immunity and stress responses of striped catfish (Pangasianodon hypophthalmus). J. Fish. Shellfish. Immunol. 2017, 60, 391–399. [Google Scholar] [CrossRef]
- James, C.M.; Dias, P.; Salman, A.E. The use of marine yeast (Candida sp.) and bakers’ yeast (Saccharomyces cerevisiae) in combination with Chlorella sp. for mass culture of the rotiferBrachionusplicatilis. Hydrobiologia 1987, 147, 263–268. [Google Scholar] [CrossRef]
- Wang, Y. Effect of probiotics on growth performance and digestive enzyme activity of the shrimp Penaeus vannamei. Aquaculture 2007, 269, 259–264. [Google Scholar] [CrossRef]
- Reyes-Becerril, M.; Ascencio-Valle, F.; Meseguer, J.; Tapia-Paniagua, S.T.; Morinigo, M.A.; Esteban, M.A. Debaryomyces hansenii L2 enriched diet enhances the immunity status, gene expression and intestine functionality in gilthead sea bream (Sparus aurata L.). Aquac. Res. 2011, 43, 1107–1118. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Kim, M.C.; Kim, J.S.; Balasundaramb, C.; Heo, M.S. Protective effect of herbal and probiotics enriched diet on haematological and immunity status of Oplegnathus fasciatus (Temminck & Schlegel) against Edwardsiella tarda. Fish. Shellfish. Immunol. 2011, 30, 886–893. [Google Scholar]
- García-de-la-Banda, I.; Chereguini, O.; Rasines, I. Influencia de la adición de bacteria lácticasen el cultivolarvario del rodaballo (Scophthalmus maximus L.). Bol. Inst. Esp. Oceanogr. 1992, 8, 247–254. (In Spanish) [Google Scholar]
- Gomez-Gil, B.; Soto-Rodriguez, S.; Garcia-Gasca, A.; Roque, A.; Vazquez-Juarez, R.; Thompson, F.L.; Swings, J. Molecular identification of Vibrio harveyi related isolates associated with diseased aquatic organisms. Microbiology 2004, 150, 1769–1777. [Google Scholar] [CrossRef]
- Coyle, M.B. Manual of Antimicrobial Susceptibility Testing; American Society for Microbiology: Washington, DC, USA, 2005. [Google Scholar]
- Sorgeloos, P.; Bossuyt, E.; Lavina, E.; Baeza-Mesa, M.; Persoone, G. Decapsulation of Artemia cysts: A simple technique for the improvement of brine shrimp in aquaculture. Aquaculture 1977, 12, 311–315. [Google Scholar] [CrossRef]
- Mahios, A.S.; Gatesoupe, F.J.; Hervi, M.; Metailler, R.; Ollevier, F. Effect of dietary inulin and oligosaccharides and other prebiotics for weaning turbot Psetts maxima (Linnaeus C 1758). Aquac. Int. 2006, 14, 219–229. [Google Scholar] [CrossRef]
| Yeast Strains | V. anguillarum | V. harveyi | V. campbellii | V. parahaemolyticus |
|---|---|---|---|---|
| Candida pseudolambica1401 | ++ | + | − | + |
| Candida parapsilosis1019 | + | ++ | ++ | + |
| Pichia sydowiorum3901 | +++++ | − | − | + |
| Pichia sydowiorum4901 | +++ | − | − | + |
| Saturnisporasilvae1501 | ++ | − | − | + |
| Torulaspora spp. 82 | ++ | − | − | + |
| Pichia anomala YF07b | ++ | − | − | ++ |
| Pichia philogaea K176 | +++ | − | − | + |
| Candida spp. 4201 | +++ | − | − | − |
| Debaryomyces hanseii K226 | ++++ | − | − | − |
| Marine Bacterium Strains | V. anguillarum | V. harveyi | V. campbellii | V. parahaemolyticus |
| Ruegeria mobilis YJ3 | ++ | − | − | − |
| Pseudoalteromonas flaripulchra JG1 | +++ | +++++ | +++ | ++++ |
| Lactobacillus Strains | V. anguillarum | V. harveyi | V. campbellii | V. parahaemolyticus |
| Lactobacillus plantarum JS201 | − | − | − | − |
| Lactobacillus plantarum JS202 | − | − | − | − |
| Pediococcus acidlactici JS203 | − | − | − | − |
| Lactobacillus sakei JS204 | ++ | + | ++ | ++ |
| Lactobacillus bulgaricus JS205 | − | − | + | + |
| Streptococcus thermophilus JS206 | − | − | − | − |
| Bacillus Strains | V. anguillarum | V. harveyi | V. campbellii | V. parahaemolyticus |
| Bacillus laterosporus JS01 | ++ | − | − | + |
| Bacillus coagulans JS207 | − | − | − | + |
| Bacillus megaterium JS02 | − | + | + | − |
| Bacillus natto JS03 | +++++ | ++ | ++++ | + |
| Bacillus mucilaginosus JS04 | − | ++ | ++ | − |
| Bacillus amyloliquefaciens JS05 | +++ | +++ | ++++ | + |
| Bacillus pumilis JS06 | − | + | + | − |
| Bacillus subtilis JS07 | − | − | − | − |
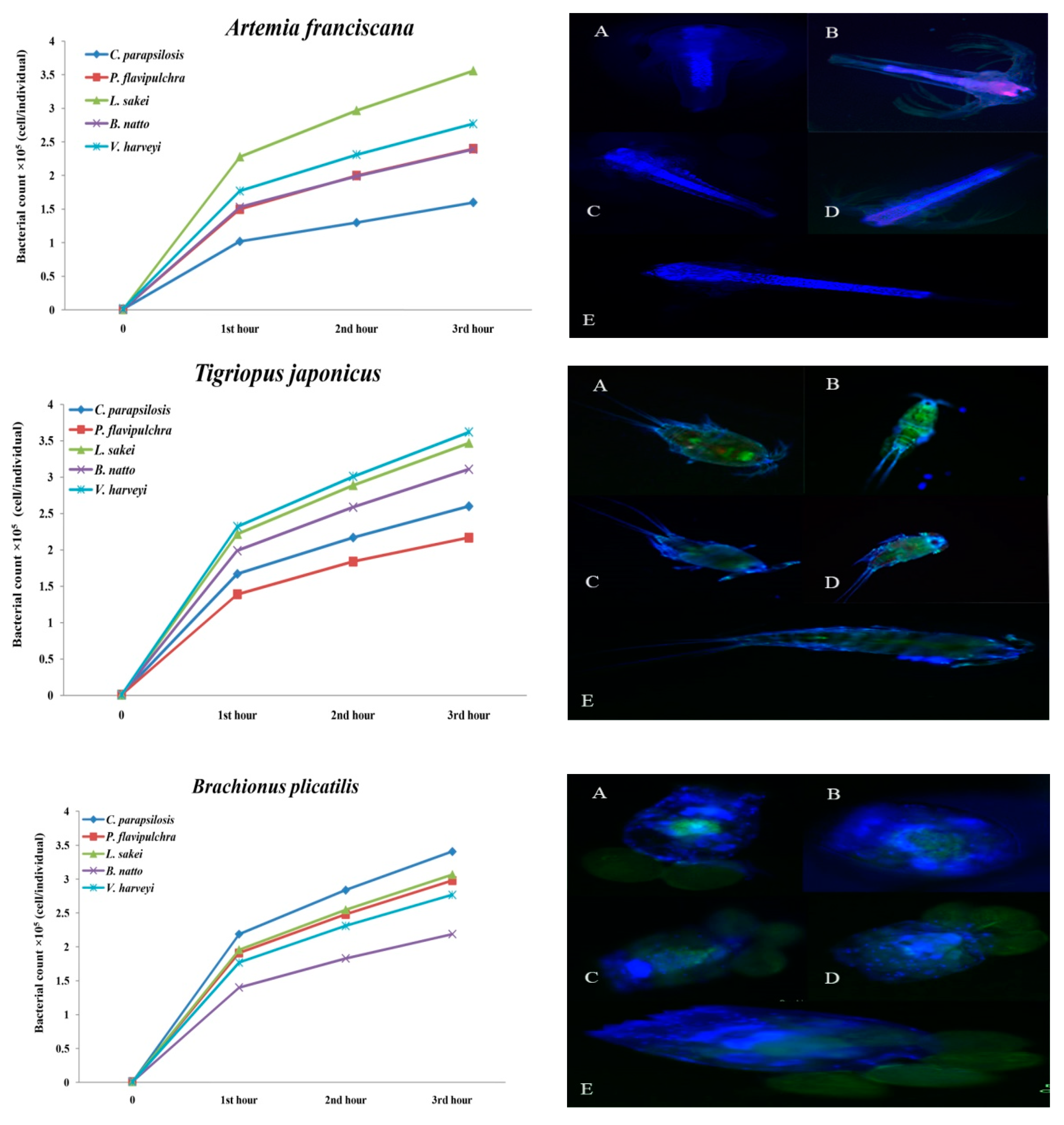
| Live Food | Microbial Strains (CTCF × 105 cell/individual) | ||||
|---|---|---|---|---|---|
| C. parapsilosis | P. flavipulchra | L. sakei | B. natto | V. harveyi | |
| A. franciscana | 1.6 ± 0.18 c | 2.40 ± 035 b,c | 3.56 ± 0.55 a | 2.39 ± 0.04 b,c | 2.77 ± 0.5 a,b |
| B. plicatilis | 3.41 ± 0.21 a | 2.98 ± 0.85 a,b | 3.07 ± 0.2 a,b | 2.19 ± 0.53 b | 277 ± 0.38 a,b |
| T. japonicus | 2.60 ± 0.36 a,b | 2.17 ± 0.11c | 3.47 ± 0.44 b,c | 3.11 ± 0.24 a,b,c | 3.62 ± 4.66 a |
| Treatment | 107 CFU/g Artemia nauplii | |||
|---|---|---|---|---|
| 5 h | 10 h | 15 h | 20 h | |
| Control | 7 ± 0.1 c | 8.3 ± 0.2 c | 8.3 ± 0.4 c | 10.1 ± 0.1 d |
| C. parapsilosis | 2.9 ± 0.1 b | 3.7 ± 0.1 b | 2.7 ± 0.1 b,c | 4.9 ± 0.2 c |
| P. flavipulchra | 0.9 ± 0.2 a | 1.1 ± 0.2 a | 0.7 ± 0.3 a | 1.7 ± 0.2 b |
| L. sakei | 0.5 ± 0.1 a | 0.9 a | 1.4 ± 0.2 b | 1.1 ± 0.3 a |
| B. natto | 0.7 ± 0.1 a | 0.4 ± 0.1 a | 1.4 ± 0.2 b | 1.2 ± 0.2 a |
| 107 CFU/g rotifer | ||||
| Control | 12.1 ± 0.3 d | 12.6 ± 0.3 d | 12.2 ± 0.5 d | 14 ± 0.3 d |
| C. parapsilosis | 8.0 c | 7.5 ± 0.2 c | 6.5 ± 0.2 c | 6.5 ± 0.3 c |
| P. flavipulchra | 1.6 ± 0.2 a | 1.1 ± 0.2 a | 0.4 ± 0.2 a | 0.2 a |
| L. sakei | 0.9 ± 0.1 a | 0.7 a | 0.4 a | 0.3 a |
| B. natto | 6.0 ± 0.2 b | 4.4 ± 0.5 b | 2.3 ± 0.4 b | 1.7 ± 0.1 b |
| 106 CFU/g copepod | ||||
| Control | 10.6 ± 0.2 d | 10.8 ± 0.1 d | 11.3 ± 0.2 d | 13.1 ± 0.3 d |
| C. parapsilosis | 8.3 ± 0.2 c | 9.1 ± 0.1 c | 8.1 ± 0.2 c | 8.1 ± 0.2 c |
| P. flavipulchra | 3.3 ± 0.1 a | 1.1 ± 0.2 a | 0.7 ± 0.3 a | 2.1 ± 0.3 a |
| L. sakei | 5.3 ± 0.2 a,b | 3.7 ± 0.1 b | 2.5 ± 0.3 b | 1.1 ± 0.3 a |
| B. natto | 6.3 ± 0.5 b | 3.9 ± 0.2 b | 3.2 ± 0.1 b | 2.5 ± 0.1 b |
| Strain Code | Bacterial Strains | Reference | Strain Code | Yeast Strains | Reference |
|---|---|---|---|---|---|
| S4 | aBacillus subtilis | [38] | 82 | aTorulasporaspp | - |
| - | bBacillus laterosporus | [12] | 1401 | aCandida pseudolambica | - |
| - | aBacillus megaterium | [39] | 1501 | aSaturnisporasilvae | - |
| SRB-3 | aBacillus pumilus | [40] | 3901 | aPichia sydowiorum | - |
| S18 | aBacillus amyloliquefaciens | [41] | 4201 | aCandida spp. | [42] |
| - | bBacillus natto | - | 4901 | aPichia sydowiorum | - |
| - 1.19 | bBacillus mucilaginosus cLactobacillus plantarum | - [3] | 6101 1019 | aRhodotorula mucilaginosa | - - |
| aCandida parapsilosis | |||||
| - | bBacillus coagulans | [43] | K226 | aDebaryomyces hansenii | [44] |
| 1.2696 | cLactobacillus acidlactici | - | YF07b | aPichia anomala | - |
| 1.6 | cLactobacillus sakei | [45] | K176 | aPichia philogaea | - |
| - | dLactobacillus bulgaricus | [46] | 82 | ||
| - YJ3 | dStreptococcus thermophilus aRuegeria mobilis | - - | |||
| JG1 | aPseudoalteromonas flavipulchra | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahandi, J.; Sorgeloos, P.; Xiao, H.; Wang, X.; Qi, Z.; Zheng, Y.; Tang, X. The Use of Selected Bacteria and Yeasts to Control Vibrio spp. in Live Food. Antibiotics 2019, 8, 95. https://doi.org/10.3390/antibiotics8030095
Sahandi J, Sorgeloos P, Xiao H, Wang X, Qi Z, Zheng Y, Tang X. The Use of Selected Bacteria and Yeasts to Control Vibrio spp. in Live Food. Antibiotics. 2019; 8(3):95. https://doi.org/10.3390/antibiotics8030095
Chicago/Turabian StyleSahandi, Javad, Patrick Sorgeloos, Hui Xiao, Xianghong Wang, Zizhong Qi, Yanfen Zheng, and Xuexi Tang. 2019. "The Use of Selected Bacteria and Yeasts to Control Vibrio spp. in Live Food" Antibiotics 8, no. 3: 95. https://doi.org/10.3390/antibiotics8030095
APA StyleSahandi, J., Sorgeloos, P., Xiao, H., Wang, X., Qi, Z., Zheng, Y., & Tang, X. (2019). The Use of Selected Bacteria and Yeasts to Control Vibrio spp. in Live Food. Antibiotics, 8(3), 95. https://doi.org/10.3390/antibiotics8030095





