Degradation Kinetics of Clavulanic Acid in Fermentation Broths at Low Temperatures
Abstract
1. Introduction
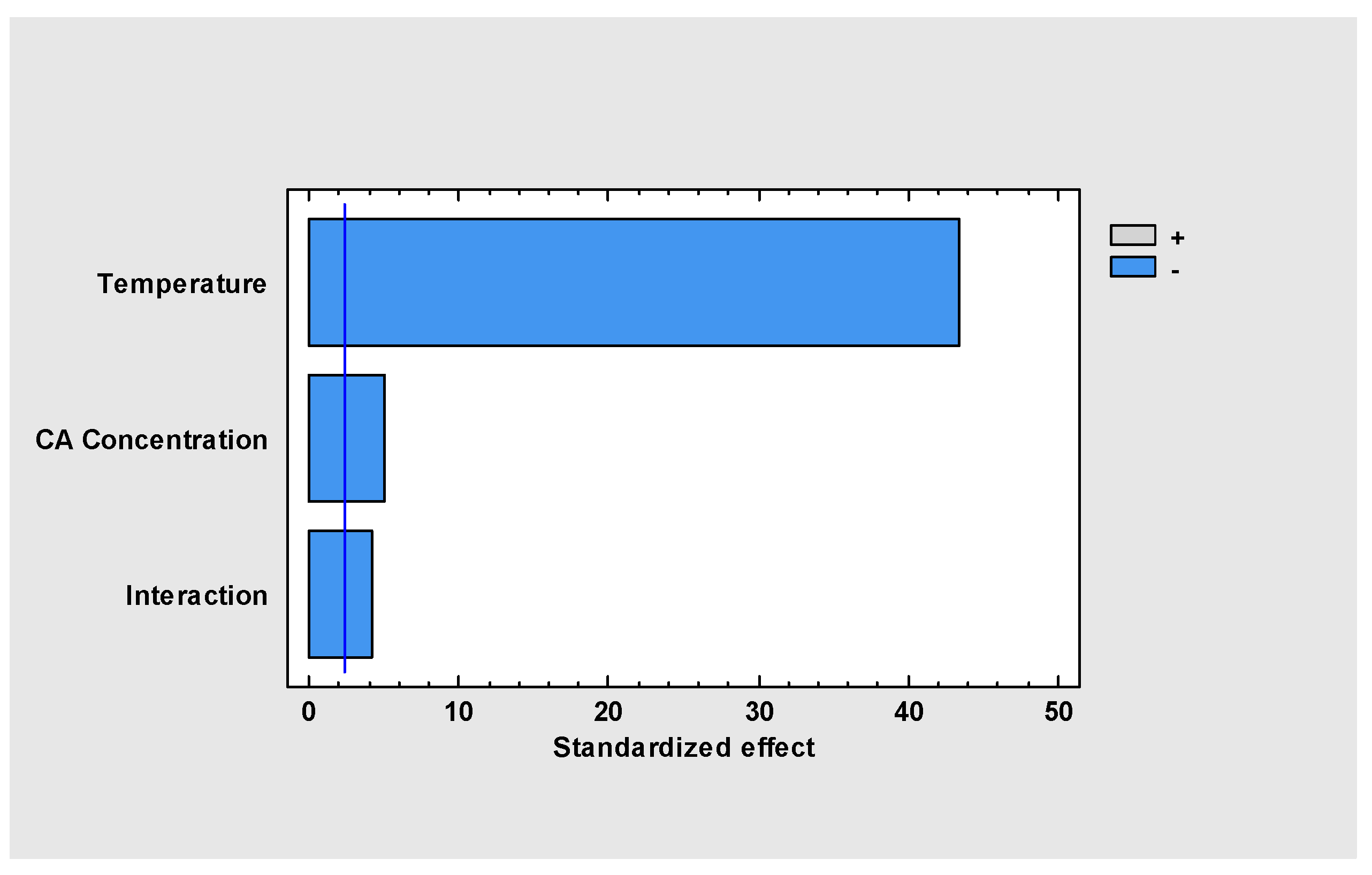
2. Results and Discussion
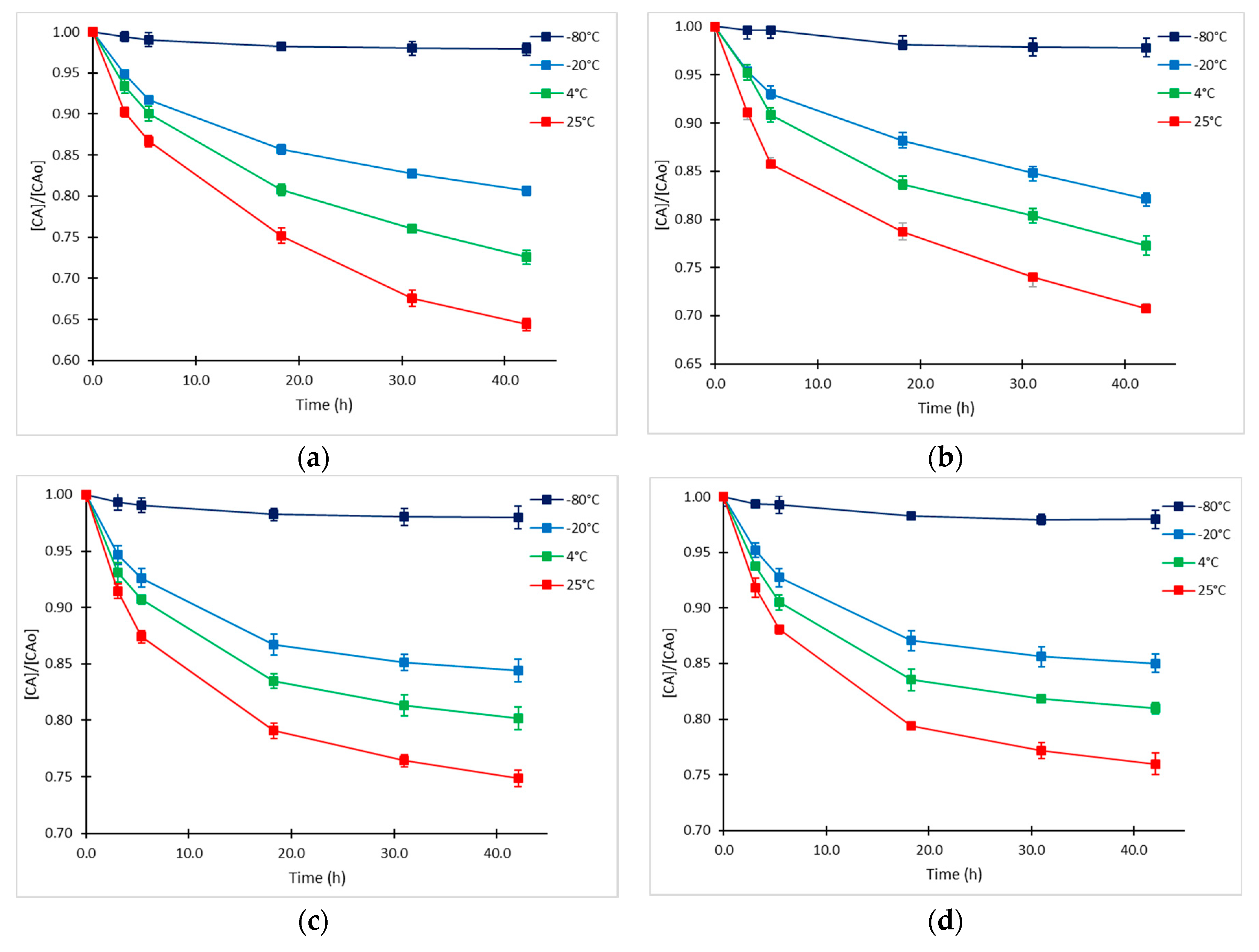
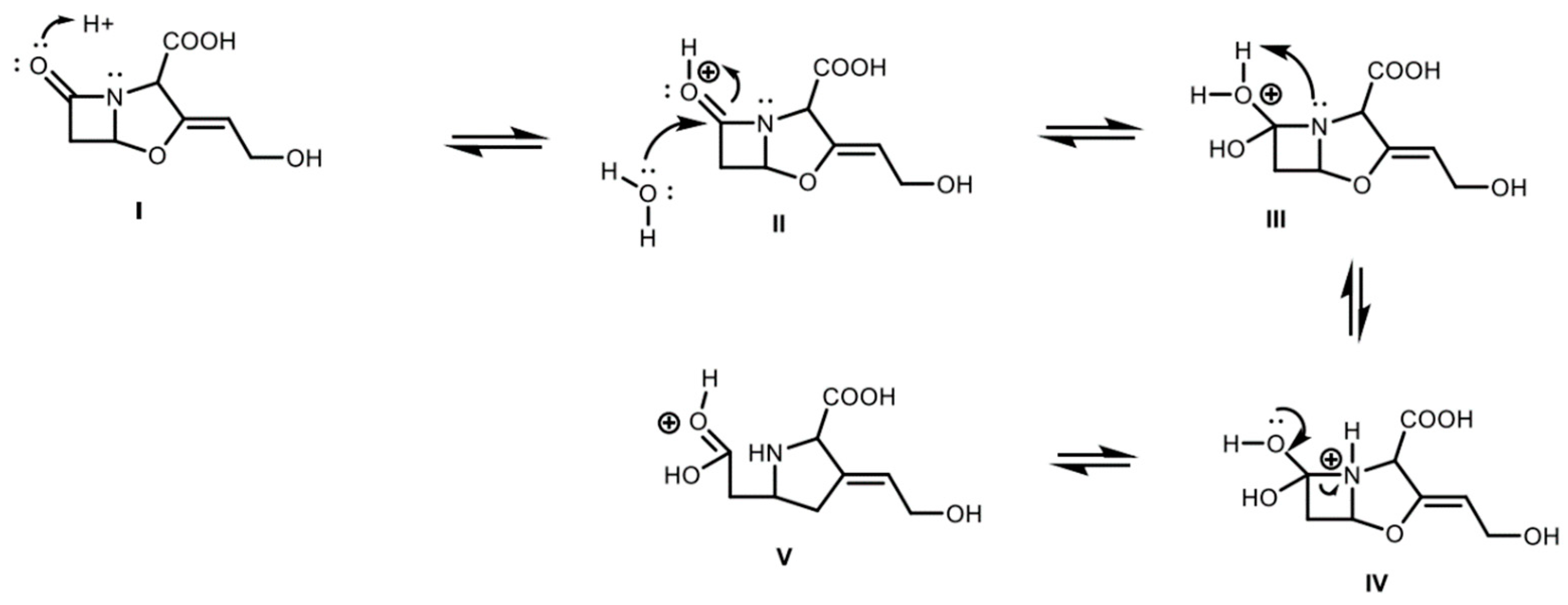
2.1. Clavulanic Acid Degradation
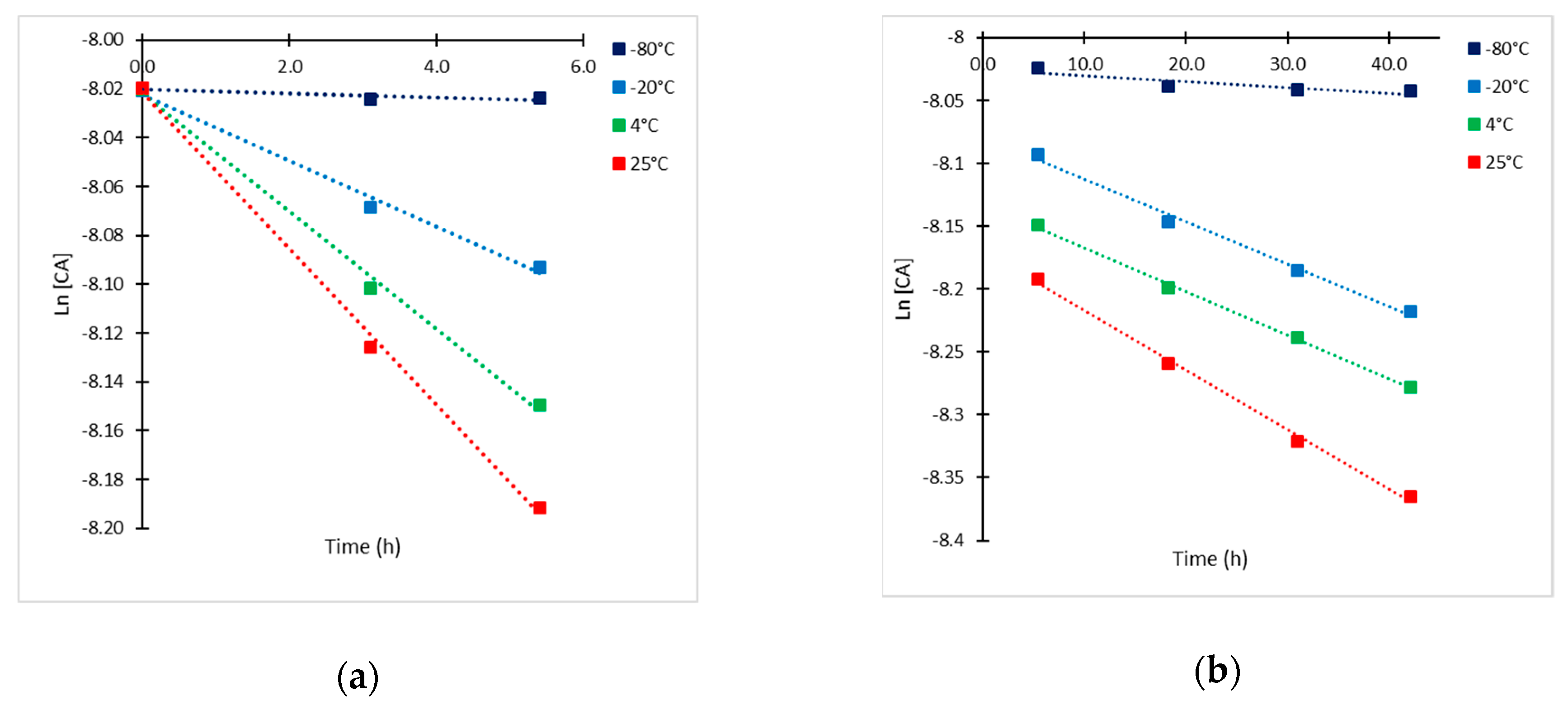
2.2. Kinetic Approach of CA Degradation
2.3. Kinetic Model Validation
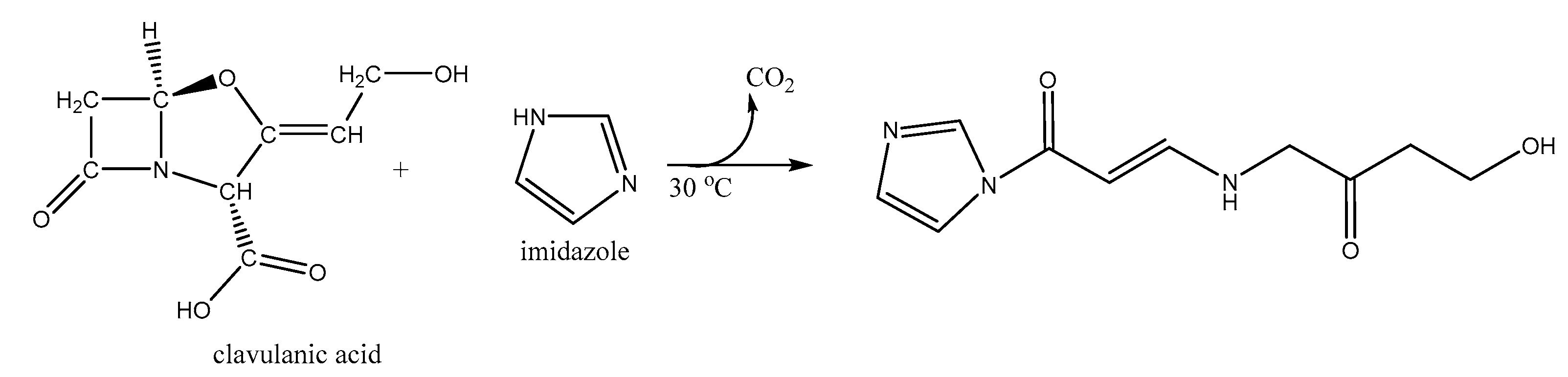
2.4. Derivatized Clavulanate-Imidazole Degradation Kinetics
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ramirez-Malule, H. Bibliometric analysis of global research on clavulanic acid. Antibiotics 2018, 7, 102. [Google Scholar] [CrossRef] [PubMed]
- De Araujo Viana Marques, D.; Feitosa Machado, S.E.; Santos Ebinuma, V.C.; De Albuquerque Lima Duarte, C.; Converti, A.; Porto, A.L.F. Production of β-lactamase inhibitors by Streptomyces species. Antibiotics 2018, 7, 61. [Google Scholar]
- Takahashi, Y.; Nakashima, T. Actinomycetes, an inexhaustible source of naturally occurring antibiotics. Antibiotics 2018, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Manteca, Á.; Yagüe, P. Streptomyces differentiation in liquid cultures as a trigger of Secondary metabolism. Antibiotics 2018, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Ser, H.-L.; Law, J.W.-F.; Chaiyakunapruk, N.; Jacob, S.A.; Palanisamy, U.D.; Chan, K.-G.; Goh, B.-H.; Lee, L.-H. Fermentation conditions that affect clavulanic acid production in Streptomyces clavuligerus: A systematic review. Front. Microbiol. 2016, 7, 522. [Google Scholar] [CrossRef] [PubMed]
- Saudagar, P.S.; Singhal, R.S. Optimization of nutritional requirements and feeding strategies for clavulanic acid production by Streptomyces clavuligerus. Bioresour. Technol. 2007, 98, 2010–2017. [Google Scholar] [CrossRef]
- Bellão, C.; Antonio, T.; Araujo, M.L.G.C.; Badino, A.C. Production of clavulanic acid and cephamycin c by Streptomyces clavuligerus under different fed-batch conditions. Braz. J. Chem. Eng. 2013, 30, 257–266. [Google Scholar] [CrossRef]
- Teodoro, J.C.; Baptista-Neto, A.; Araujo, M.L.G.C.; Hokka, C.O.; Badino, A.C. Influence of glycerol and ornithine feeding on clavulanic acid production by Streptomyces clavuligerus. Braz. J. Chem. Eng. 2010, 27, 499–506. [Google Scholar] [CrossRef]
- Neto, A.B.; Hirata, D.B.; Cassiano Filho, L.C.M.; Bellão, C.; Badino, A.C.; Hokka, C.O. A study on clavulanic acid production by Streptomyces clavuligerus in batch, FED-batch and continuous processes. Braz. J. Chem. Eng. 2005, 22, 557–563. [Google Scholar] [CrossRef]
- Bersanetti, P.A.; Almeida, R.M.R.G.; Barboza, M.; Araújo, M.L.G.C.; Hokka, C.O. Kinetic studies on clavulanic acid degradation. Biochem. Eng. J. 2005, 23, 31–36. [Google Scholar] [CrossRef]
- Marques, D.A.V.; Oliveira, R.P.S.; Perego, P.; Porto, A.L.F.; Pessoa, A.; Converti, A. Kinetic and thermodynamic investigation on clavulanic acid formation and degradation during glycerol fermentation by Streptomyces DAUFPE 3060. Enzyme Microb. Technol. 2009, 45, 169–173. [Google Scholar] [CrossRef]
- Rosa, J.C.; Baptista Neto, A.; Hokka, C.O.; Badino, A.C. Influence of dissolved oxygen and shear conditions on clavulanic acid production by Streptomyces clavuligerus. Bioprocess Biosyst. Eng. 2005, 27, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Teodoro, J.C.; Baptista-Neto, A.; Cruz-Hernández, I.L.; Hokka, C.O.; Badino, A.C. Influence of feeding conditions on clavulanic acid production in fed-batch cultivation with medium containing glycerol. Appl. Microbiol. Biotechnol. 2006, 72, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Bushell, M.E.; Kirk, S.; Zhao, H.; Avignone-rossa, C.A. Manipulation of the physiology of clavulanic acid biosynthesis with the aid of metabolic flux analysis. Enzyme Microb. Technol. 2006, 39, 149–157. [Google Scholar] [CrossRef]
- Cerri, M.O.; Badino, A.C. Shear conditions in clavulanic acid production by Streptomyces clavuligerus in stirred tank and airlift bioreactors. Bioprocess Biosyst. Eng. 2012, 35, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.L.L.; Badino, A.C. Production of clavulanic acid by Streptomyces clavuligerus in batch cultures without and with glycerol pulses under different temperature conditions. Biochem. Eng. J. 2012, 69, 1–7. [Google Scholar] [CrossRef]
- Carvalho, V.; Brandão, J.F.; Brandão, R.; Rangel-yagui, C.O.; Couto, J.A.; Converti, A.; Pessoa, A. Stability of clavulanic acid under variable pH, ionic strength and temperature conditions. A new kinetic approach. Biochem. Eng. J. 2009, 45, 89–93. [Google Scholar]
- Ishida, K.; Hung, T.V.; Lee, H.C.; Liou, K.K.; Shin, C.H.; Yoon, Y.J.; Sohng, J.K. Degradation of clavulanic Acid During the cultivation of Streptomyces clavuligerus; Instability of clavulanic Acid by Metabolites and Proteins from the Strain. J. Microbiol. Biotechnol. 2006, 16, 590–596. [Google Scholar]
- Brethauer, S.; Held, M.; Panke, S. Clavulanic acid decomposition is catalyzed by the compound itself and by its decomposition products. J. Pharm. Sci. 2008, 97, 3451–3455. [Google Scholar] [CrossRef]
- Roubos, J.A.; Krabben, P.; De Laat, W.; Heijnen, J.J. Clavulanic acid degradation in Streptomyces clavuligerus fed-batch cultivations. Biotechnol. Prog. 2002, 18, 451–457. [Google Scholar] [CrossRef]
- Nickolai, D.J.; Lammel, C.J.; Byford, B.A.; Morris, J.H.; Kaplan, E.B.; Hadley, W.K.; Brooks, G.F. Effects of storage temperature and pH on the stability of eleven b-lactam antibiotics in MIC trays. J. Clin. Microbiol. 1985, 21, 366–370. [Google Scholar] [PubMed]
- Okerman, L.; Van Hende, J.; De Zutter, L. Stability of frozen stock solutions of beta-lactam antibiotics, cephalosporins, tetracyclines and quinolones used in antibiotic residue screening and antibiotic susceptibility testing. Anal. Chim. Acta 2007, 586, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Vahdat, L.; Sunderland, V.B. Kinetics of amoxicillin and clavulanate degradation alone and in combination in aqueous solution under frozen conditions. Int. J. Pharm. 2007, 342, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Vetráková, L. Study of Processes Associated with Freezing of Aqueous Solutions. Ph.D. Thesis, Masaryk University, Brno, Czech Republic, 2017. [Google Scholar]
- Vahdat, L.; Sunderland, B. The influence of potassium clavulanate on the rate of amoxicillin sodium degradation in phosphate and acetate buffers in the liquid state. Drug Dev. Ind. Pharm. 2009, 35, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Jerzsele, Á.; Nagy, G. The stability of amoxicillin trihydrate and potassium clavulanate combination in aqueous solutions. Acta Vet. Hung. 2009, 57, 485–493. [Google Scholar] [CrossRef]
- Nur, A.O.; Hassan, A.A.; Gadkariem, E.A.; Osman, Z. Stability of co-amoxiclav reconstituted injectable solution. Eur. J. Pharm. Med. Res. 2015, 2, 109–123. [Google Scholar]
- Haginaka, J.; Yasuda, H.; Uno, T.; Nakagawa, T. Degradation of clavulanic acid in aqueous alkaline solution: isolation and structural investigation of degradation products. Chem. Pharm. Bull. 1985, 33, 218–224. [Google Scholar] [CrossRef]
- Finn, M.J.; Harris, M.A.; Hunt, E. Studies on the hydrolysis of clavulanic. J. Chem. Soc. Perkin Trans. 1984, 10, 1345–1349. [Google Scholar] [CrossRef]
- Zhong, C.; Cao, G.; Jin, X.; Wang, F. Studies on the formation and forming mechanism of the related substance E in potassium clavulanate production by HPLC-MS/MS. Braz. J. Pharm. Sci. 2014, 50, 391–399. [Google Scholar] [CrossRef]
- Soroka, D.; De La Sierra-Gallay, I.L.; Dubée, V.; Triboulet, S.; Van Tilbeurgh, H.; Compain, F.; Ballell, L.; Barros, D.; Mainardi, J.L.; Hugonnet, J.E.; et al. Hydrolysis of clavulanate by Mycobacterium tuberculosis β-lactamase BlaC harboring a canonical SDN motif. Antimicrob. Agents Chemother. 2015, 59, AAC-00598. [Google Scholar] [CrossRef] [PubMed]
- Peace, N.; Olubukola, O.; Moshood, A. Stability of reconstituted amoxicillin clavulanate potassium under simulated in-home storage conditions. J. Appl. Pharm. Sci. 2012, 2, 28–31. [Google Scholar]
- Gour-Salin, B.J. Hydrolysis rates of some acetylimidazole derivatives. Can. J. Chem. 1983, 61, 2059–2061. [Google Scholar] [CrossRef]
- Joule, J.A.; Mills, K. 1,3-Azoles: Imidazoles, thiazoles and oxazoles: Reactions and synthesis. In Heterocyclic Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 461–467. ISBN 978-1-4051-3300-5. [Google Scholar]
- Ramirez-malule, H.; Junne, S.; Cruz-bournazou, M.N.; Neubauer, P. Streptomyces clavuligerus shows a strong association between TCA cycle intermediate accumulation and clavulanic acid biosynthesis. Appl. Microbiol. Biotechnol. 2018, 102, 4009–4023. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Malule, H.; Junne, S.; López, C.; Zapata, J.; Sáez, A.; Neubauer, P.; Rios-Estepa, R. An improved HPLC-DAD method for clavulanic acid quantification in fermentation broths of Streptomyces clavuligerus. J. Pharm. Biomed. Anal. 2016, 120, 241–247. [Google Scholar] [CrossRef] [PubMed]
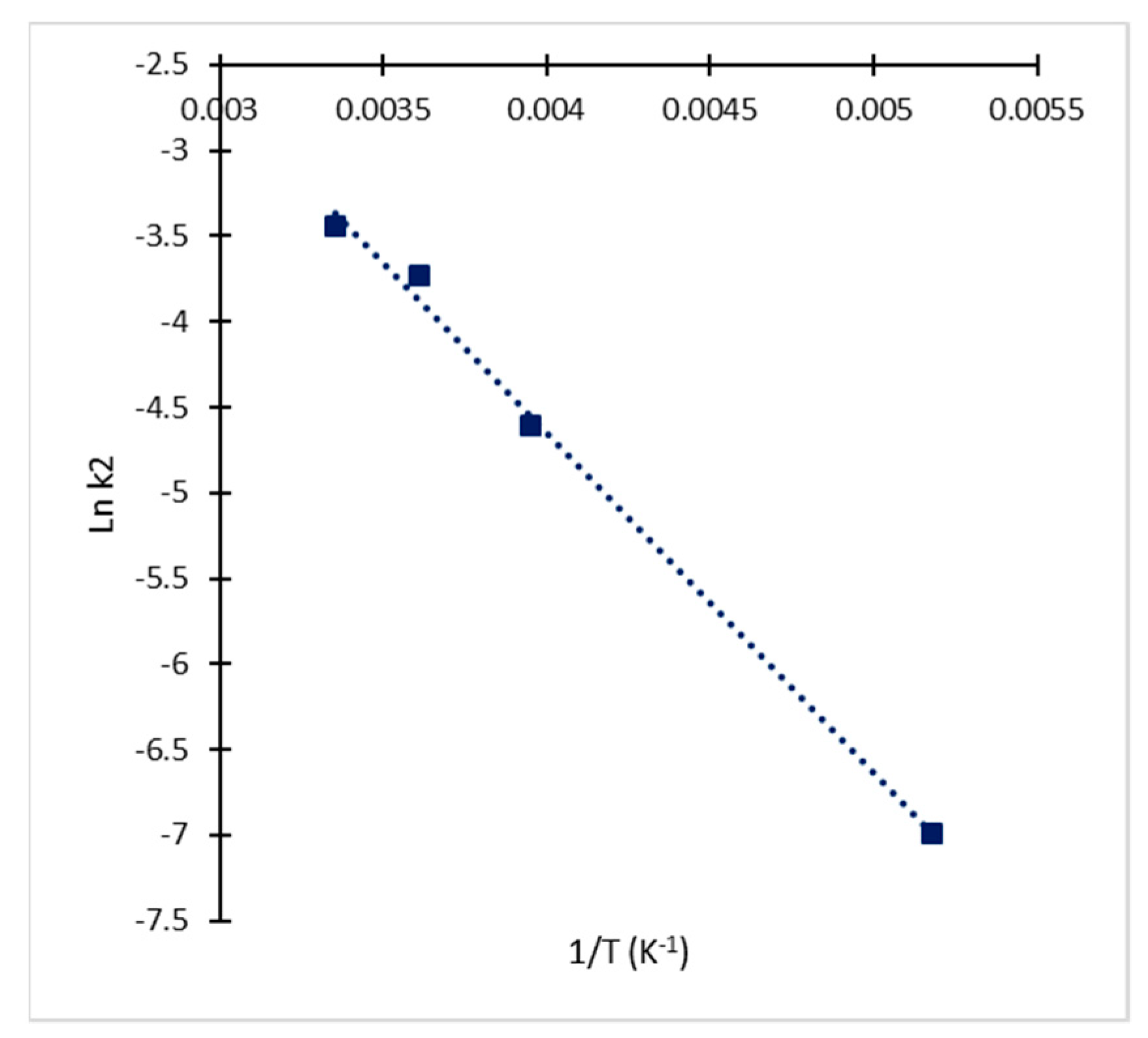
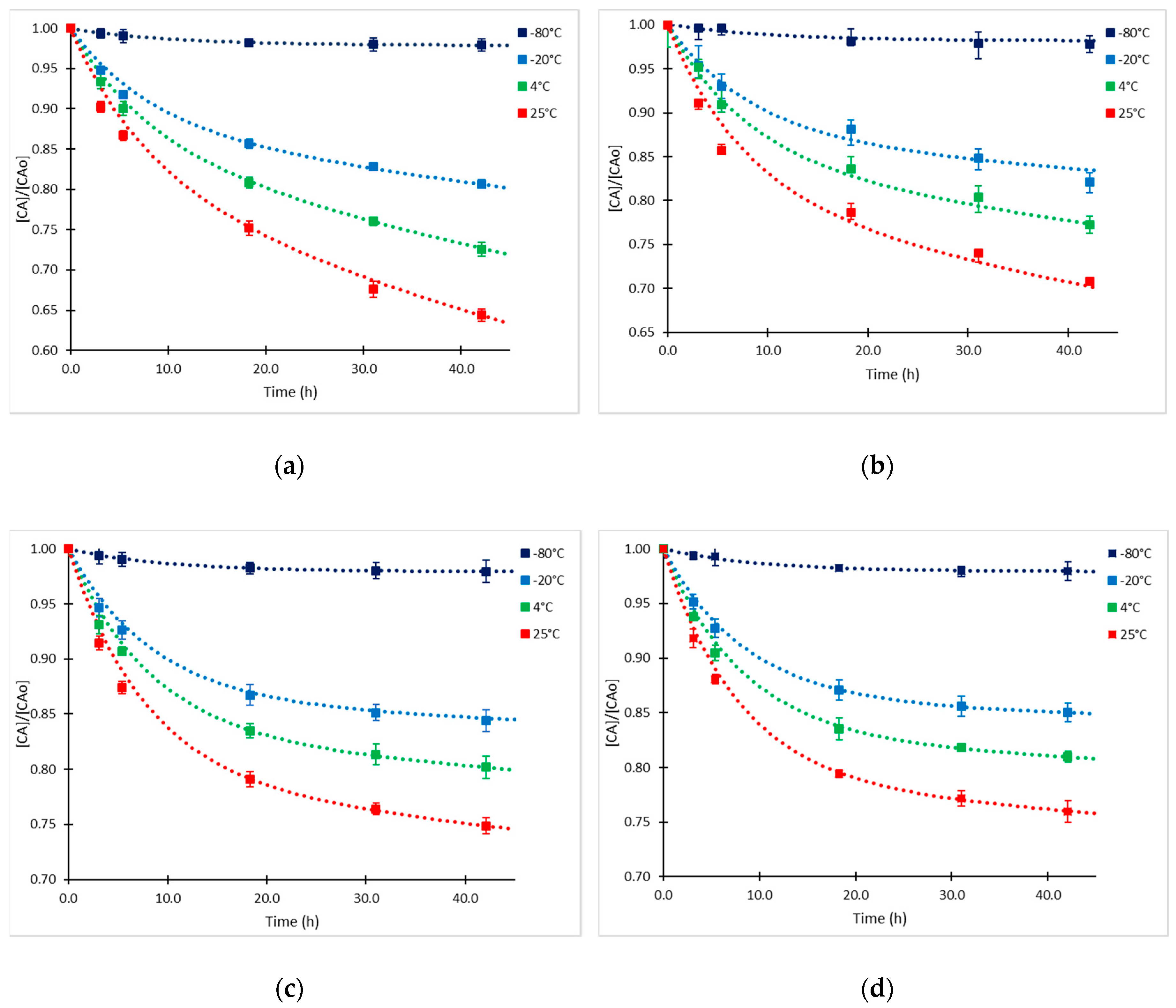
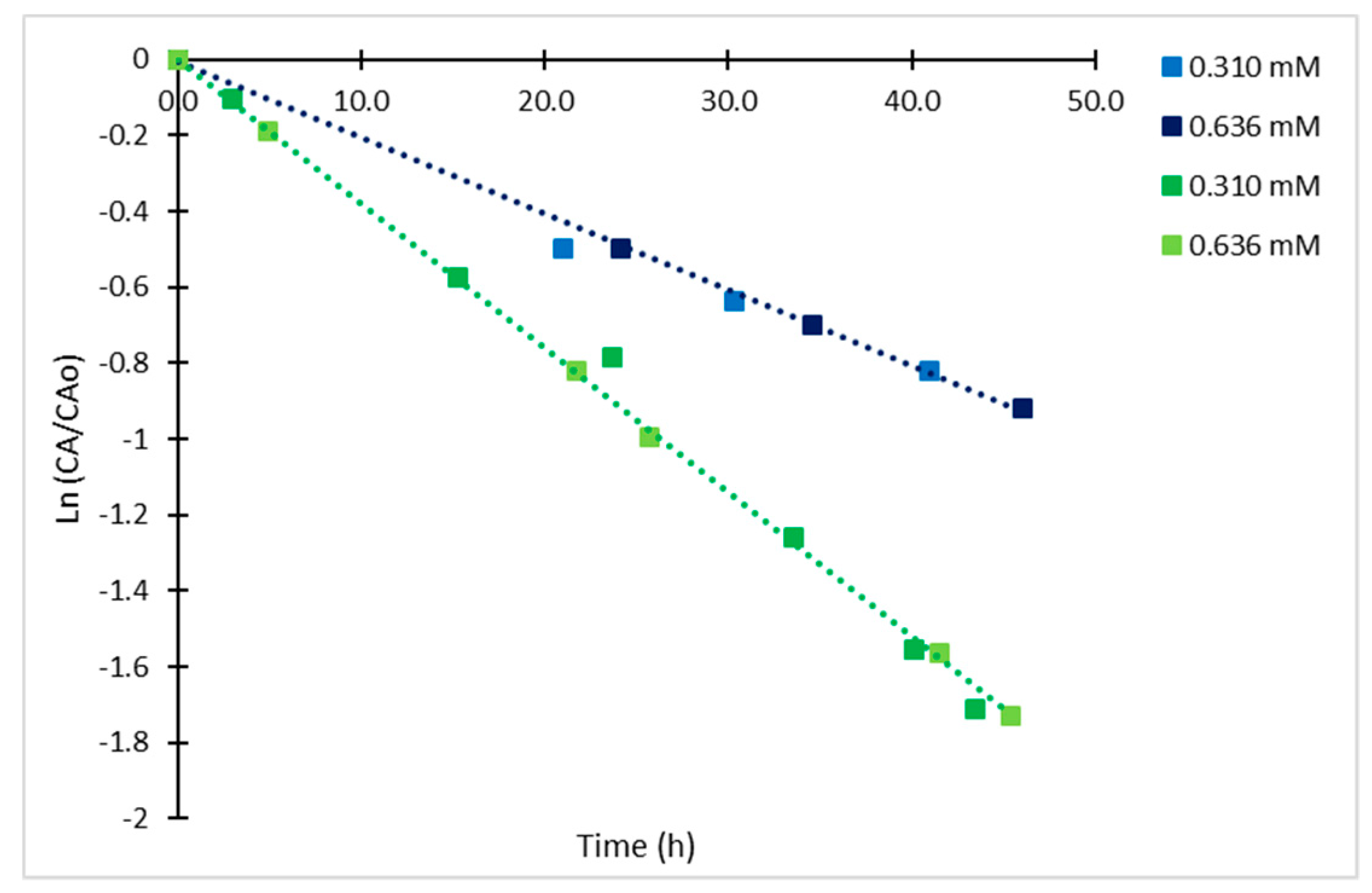
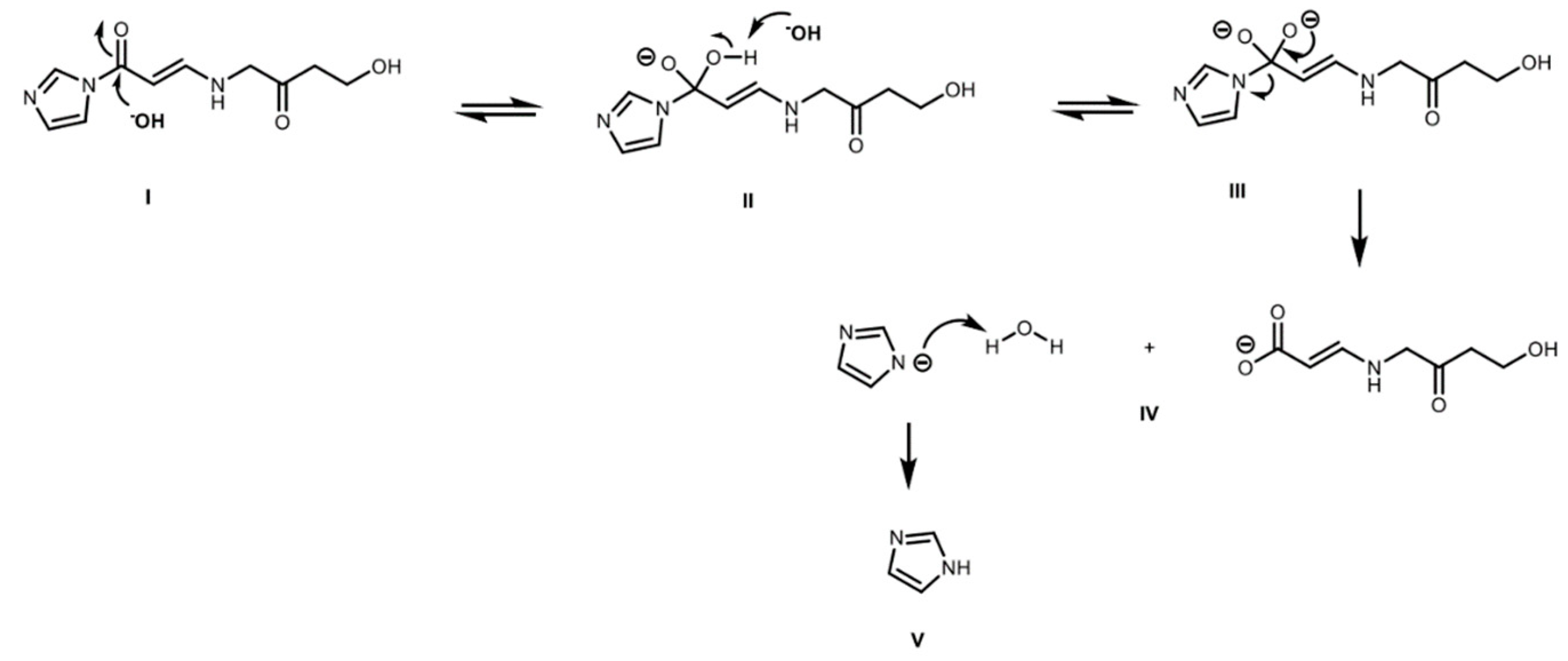
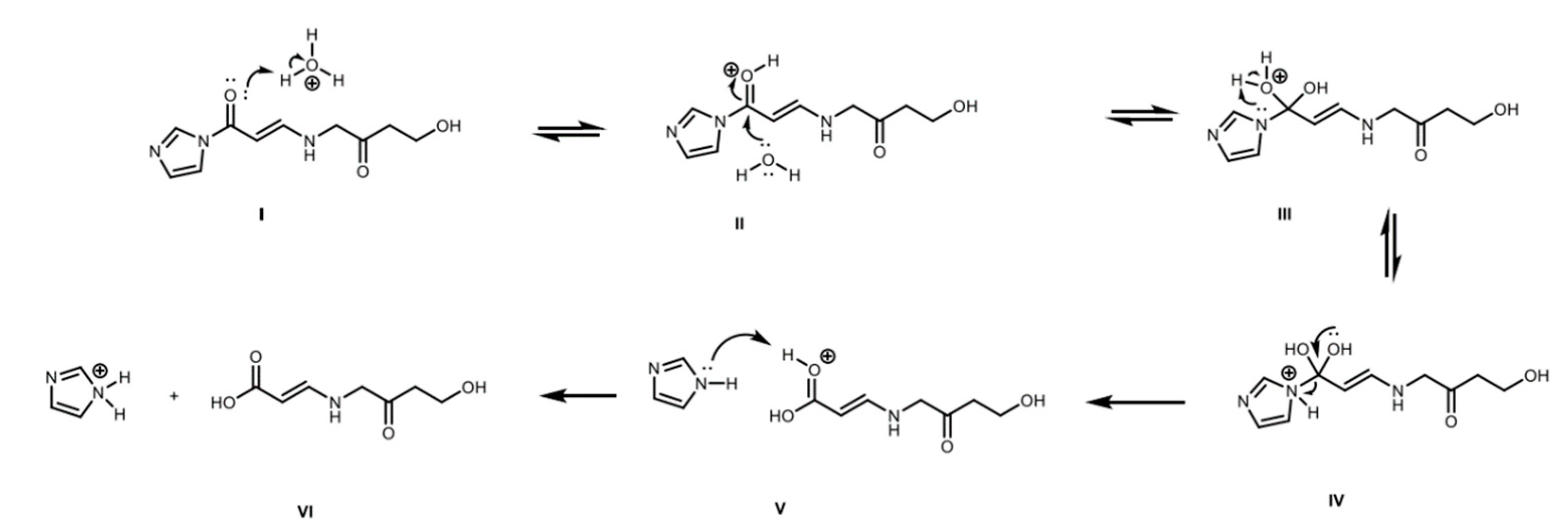
| Temperature | t < 5.5 h | t > 5.5 h | ||
|---|---|---|---|---|
| kobs,1 (h−1) | r2 | kobs,2 (h−1) | r2 | |
| −80 °C | 0.0009 | 0.969 | 0.0005 | 0.924 |
| −20 °C | 0.0135 | 0.991 | 0.0034 | 0.992 |
| 4 °C | 0.0241 | 0.996 | 0.0035 | 0.998 |
| 25 °C | 0.0320 | 0.998 | 0.0047 | 0.997 |
| Temperature | Equilibrium Constant Keq | Irreversible Rate Constant k2 (h−1) |
|---|---|---|
| −80 °C | 0.018 | 0.0009 |
| −20 °C | 0.162 | 0.0135 |
| 4 °C | 0.210 | 0.0241 |
| 25 °C | 0.280 | 0.0320 |
| Concentration (mg/L) | Temperature (°C) | NRMSE (%) | Shelf Life (t90) |
|---|---|---|---|
| 126.7 | −80 | 1.87 | 5.1 months |
| 126.7 | −20 | 3.22 | 9.0 h |
| 126.7 | 4 | 2.18 | 6.5 h |
| 126.7 | 25 | 3.78 | 4.5 h |
| 65.5 | −80 | 1.95 | 9.8 months |
| 65.5 | −20 | 4.98 | 9.7 h |
| 65.5 | 4 | 4.93 | 6.6 h |
| 65.5 | 25 | 4.45 | 4.6 h |
| 25.3 | −80 | 1.92 | 2.1 years |
| 25.3 | −20 | 3.36 | 9.9 h |
| 25.3 | 4 | 3.27 | 6.7 h |
| 25.3 | 25 | 3.40 | 4.7 h |
| 16.3 | −80 | 3.89 | 3.1 years |
| 16.3 | −20 | 1.98 | 10 h |
| 16.3 | 4 | 2.53 | 6.8 h |
| 16.3 | 25 | 2.42 | 4.8 h |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Ríos, D.; Ramírez-Malule, H.; Neubauer, P.; Junne, S.; Ríos-Estepa, R. Degradation Kinetics of Clavulanic Acid in Fermentation Broths at Low Temperatures. Antibiotics 2019, 8, 6. https://doi.org/10.3390/antibiotics8010006
Gómez-Ríos D, Ramírez-Malule H, Neubauer P, Junne S, Ríos-Estepa R. Degradation Kinetics of Clavulanic Acid in Fermentation Broths at Low Temperatures. Antibiotics. 2019; 8(1):6. https://doi.org/10.3390/antibiotics8010006
Chicago/Turabian StyleGómez-Ríos, David, Howard Ramírez-Malule, Peter Neubauer, Stefan Junne, and Rigoberto Ríos-Estepa. 2019. "Degradation Kinetics of Clavulanic Acid in Fermentation Broths at Low Temperatures" Antibiotics 8, no. 1: 6. https://doi.org/10.3390/antibiotics8010006
APA StyleGómez-Ríos, D., Ramírez-Malule, H., Neubauer, P., Junne, S., & Ríos-Estepa, R. (2019). Degradation Kinetics of Clavulanic Acid in Fermentation Broths at Low Temperatures. Antibiotics, 8(1), 6. https://doi.org/10.3390/antibiotics8010006







