Participation of S. Typhimurium cysJIH Operon in the H2S-mediated Ciprofloxacin Resistance in Presence of Sulfate as Sulfur Source
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Determination of the Minimal Inhibitory Concentration (MIC) of CIP
2.3. Determination of Intracellular ROS Levels
2.4. Determination of Superoxide Dismutase (SOD) Activity
2.5. Determination of Reduced Thiols
2.6. H2S Production
2.7. Statistics
3. Results and Discussion
3.1. Deletion of S. Typhimurium cysJIH Results in Increased Intracellular Levels of ROS and Decreased SOD Activity in Presence of Ciprofloxacin When Bacteria Were Cultured with Sulfate as the Sole Sulfur Source
| Strain * | MIC CIP (μM) sulfate | MIC CIP (μM) cysteine |
|---|---|---|
| Salmonella Typhimurium ATCC 14028s | 3.64 | 3.64 |
| Salmonella Typhimurium ATCC 14028s ΔcysJIH | 1.82 | 3.64 |
| Salmonella Typhimurium ATCC 14028s ΔcysJIH/pBADcysJIH | 3.64 | 3.64 |
| Salmonella Typhimurium ATCC 14028s WT/pBADcysJIH | 5.46 | 3.64 |
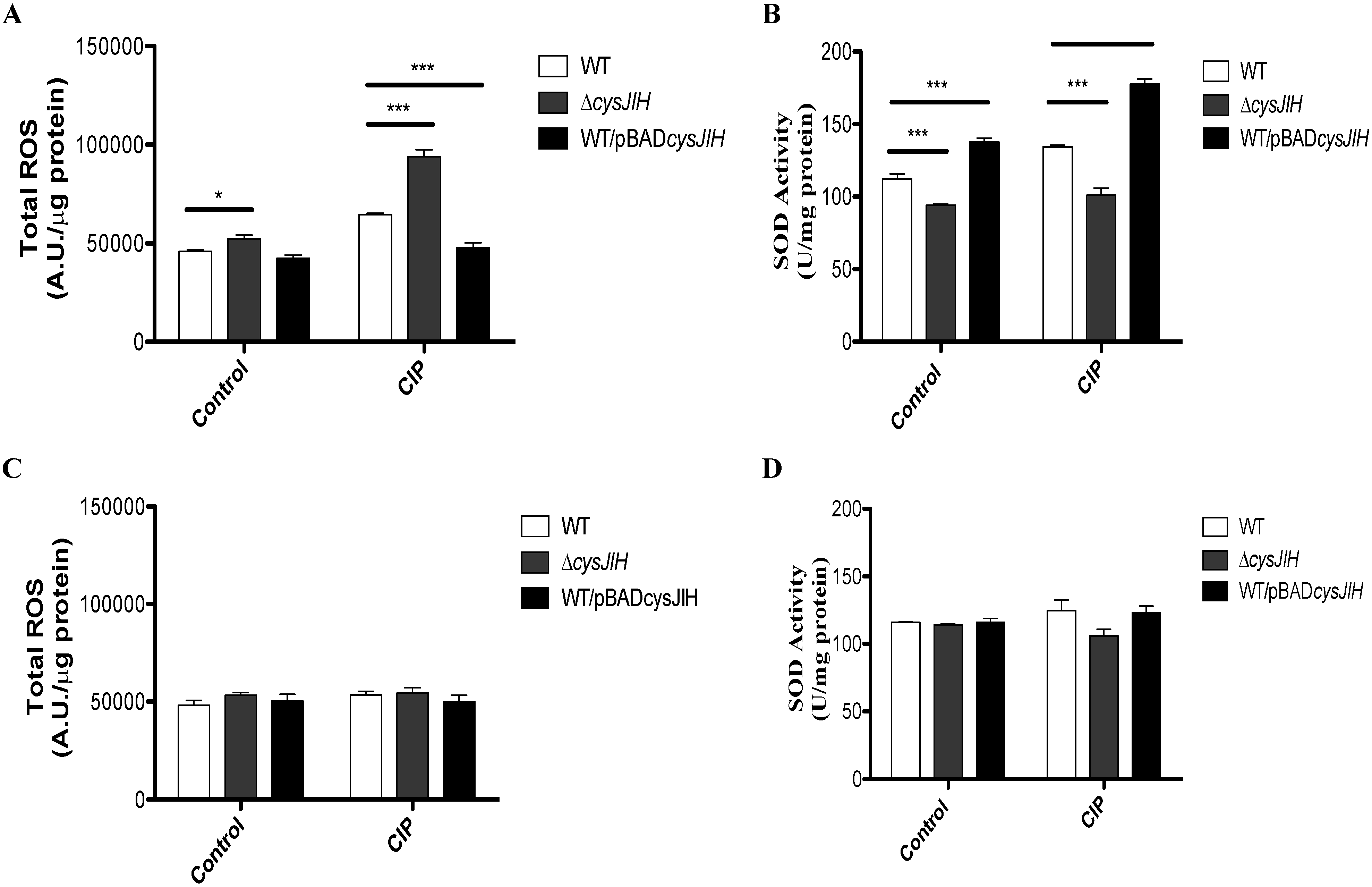
3.2. CIP Induces the Accumulation of Reduced Thiols and H2S in a cysJIH-Dependent Manner with Sulfate as the Sole Sulfur Source

4. Conclusions
- cysJIH operon are involved in CIP-resistance by increasing both the levels of H2S and reduced thiols
- The protective effect of cysJIH operon was observed only when bacteria were cultured in sulfate as the sole sulfur source.
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Sekowska, A.; Kung, H.F.; Danchin, A. Sulfur metabolism in Escherichia coli and related bacteria: Facts and fiction. J. Mol. Microbiol. Biotechnol. 2000, 2, 145–177. [Google Scholar] [PubMed]
- Jones, R.T.; Thai, L.P.; Silver, R.P. Genetic and molecular characterization of an Escherichia coli plasmid coding for hydrogen sulfide production and drug resistance. Antimicrob. Agents Chemother. 1978, 14, 765–770. [Google Scholar] [CrossRef]
- Wang, R. Physiological implications of hydrogen sulfide: A whiff exploration that blossomed. Physiol. Rev. 2012, 92, 791–896. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Yoshimura, M.; Ohara, N.; Yoshimura, S.; Nagashima, S.; Takehara, T.; Nakayama, K. Hydrogen sulfide production from cysteine and homocysteine by periodontal and oral bacteria. J. Periodontol. 2009, 80, 1845–1851. [Google Scholar] [CrossRef] [PubMed]
- Gadalla, M.M.; Snyder, S.H. Hydrogen sulfide as a gasotransmitter. J. Neurochem. 2010, 113, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H. Hydrogen sulfide: From brain to gut. Antioxid. Redox Signaling 2010, 12, 1111–1123. [Google Scholar] [CrossRef] [PubMed]
- Wang, R. Hydrogen sulfide: The third gasotransmitter in biology and medicine. Antioxid. Redox Signaling 2010, 12, 1061–1064. [Google Scholar] [CrossRef] [PubMed]
- Lund, M.E.; Matsen, J.M.; Blazevic, D.J. Biochemical and antibiotic susceptibility studies of H2S-negative Citrobacter. Appl. Microbiol. 1974, 28, 22–25. [Google Scholar] [PubMed]
- Shatalin, K.; Shatalina, E.; Mironov, A.; Nudler, E. H2S: A universal defense against antibiotics in bacteria. Science 2011, 18, 986–990. [Google Scholar] [CrossRef] [PubMed]
- Piras, C.; Soggiu, A.; Bonizzi, L.; Gaviraghi, A.; Deriu, F.; de Martino, L.; Iovane, G.; Amoresano, A.; Roncada, P. Comparative proteomics to evaluate multi drug resistance in Escherichia coli. Mol. Biosyst. 2012, 8, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Haverkamp, T.; Schwenn, J.D. Structure and function of a cysBJIH gene cluster in the purple sulphur bacterium Thiocapsa roseopersicina. Microbiology 1999, 145, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, R.; Neumann, G.; Frávega, J.; Díaz, F.; Tejías, C.; Collao, B; Fuentes, J.A.; Paredes-Sabja, D.; Calderón, I.L.; Gil, F. CysB-dependent upregulation of the Salmonella Typhimurium cysJIH operon in response to antimicrobial compounds that induce oxidative stress. Biochem. Biophys. Res. Commun. 2015, 458, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Datsenko, K.A.; Wanner, B.L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 2000, 97, 6640–6645. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, P.; Collao, B.; Álvarez, R.; Salinas, H.; Morales, E.H.; Calderón, I.L.; Saavedra, C.P.; Gil, F. Salmonella enterica serovar Typhimurium BaeSR two-component system positively regulates sodA in response to ciprofloxacin. Microbiology 2013, 159, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- Kohanski, M.A.; Dwyer, D.J.; Hayete, B.; Lawrence, C.A.; Collins, J.J. A common mechanism of cellular death induced by bactericidal antibiotics. Cell 2007, 130, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, A.L.; Surette, M.G. L-Cysteine is required for induced antibiotic resistance in actively swarming Salmonella enterica serovar Typhimurium. Microbiology 2008, 154, 3410–3419. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, A.L.; Surette, M.G. Cysteine biosynthesis, oxidative stress and antibiotic resistance in Salmonella Typhimurium. Res. Microbiol. 2010, 161, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Imlay, J.A. High levels of intracellular cysteine promote oxidative DNA damage by driving the Fenton reaction. J. Bacteriol. 2003, 185, 1942–1950. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Tandon, V. Contribution of mutations in DNA gyrase and topoisomerase IV genes to ciprofloxacin resistance in Escherichia coli clinical isolates. Int. J. Antimicrob. Agents 2011, 37, 253–255. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Dahiya, S.; Jangra, P.; Das, B.K.; Kumar, R.; Sood, S.; Kapil, A. Study of the role of efflux pump in ciprofloxacin resistance in Salmonella enterica serotype Typhi. Indian J. Med. Microbiol. 2013, 31, 374–378. [Google Scholar] [PubMed]
- Piras, C.; Soggiu, A.; Greco, V.; Martino, P.A.; del Chierico, F.; Putignani, L.; Urbani, A.; Nally, J.E.; Bonizzi, L.; Roncada, P. Mechanisms of antibiotic resistance to enrofloxacin in uropathogenic Escherichia coli in dog. J. Proteomics 2015. [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Álvarez, R.; Frávega, J.; Rodas, P.I.; Fuentes, J.A.; Paredes-Sabja, D.; Calderón, I.L.; Gil, F. Participation of S. Typhimurium cysJIH Operon in the H2S-mediated Ciprofloxacin Resistance in Presence of Sulfate as Sulfur Source. Antibiotics 2015, 4, 321-328. https://doi.org/10.3390/antibiotics4030321
Álvarez R, Frávega J, Rodas PI, Fuentes JA, Paredes-Sabja D, Calderón IL, Gil F. Participation of S. Typhimurium cysJIH Operon in the H2S-mediated Ciprofloxacin Resistance in Presence of Sulfate as Sulfur Source. Antibiotics. 2015; 4(3):321-328. https://doi.org/10.3390/antibiotics4030321
Chicago/Turabian StyleÁlvarez, Ricardo, Jorge Frávega, Paula I. Rodas, Juan A. Fuentes, Daniel Paredes-Sabja, Iván L. Calderón, and Fernando Gil. 2015. "Participation of S. Typhimurium cysJIH Operon in the H2S-mediated Ciprofloxacin Resistance in Presence of Sulfate as Sulfur Source" Antibiotics 4, no. 3: 321-328. https://doi.org/10.3390/antibiotics4030321
APA StyleÁlvarez, R., Frávega, J., Rodas, P. I., Fuentes, J. A., Paredes-Sabja, D., Calderón, I. L., & Gil, F. (2015). Participation of S. Typhimurium cysJIH Operon in the H2S-mediated Ciprofloxacin Resistance in Presence of Sulfate as Sulfur Source. Antibiotics, 4(3), 321-328. https://doi.org/10.3390/antibiotics4030321







