1. Introduction
H. pylori is still one of the most common human bacterial infections worldwide. Its eradication remains a critical public health goal due to its negative effects, such as its role in causing infection-associated cancer.
Current consensus guidelines (the American College of Gastroenterology (ACG) 2024 Guideline and Maastricht VI/Florence consensus) recommends susceptibility-guided or local antibiotic resistance-guided treatments instead of empirical eradication regimens. Our short guideline overview is focused only on the two most popular clarithromycin-based triple and tetracycline-based quadruple regimens. In regions, where primary clarithromycin resistance exceeds approximately 15%, and in cases where no previous antibiotic susceptibility testing is available, a 14-day tetracycline-based quadruple therapy (consisting of a PPI, bismuth, metronidazole, and tetracycline) is advised generally as first line treatment. In our area, the empirical use of the classical clarithromycin-based triple therapy (PPI + clarithromycin + amoxicillin or metronidazole) is not recommended as first line therapy. In regions, where estimated clarithromycin resistance is under 15%, both the amoxicillin–clarithromycin-based triple therapy (amoxicillin + clarithromycin + PPI) and the above-mentioned tetracycline-based quadruple therapies are recommended as possible first line treatments. Furthermore, in countries where commercial preparations containing antibiotics and bismuth are available, these can be given with PPI to achieve better patient compliance, with success rates exceeding 90%. Eradication rates using PPI–clarithromycin–amoxicillin/metronidazole triple therapies decreased over time, due to the increasing prevalence of clarithromycin resistance related to the frequent use of macrolide antibiotics in clinical practice [
1,
2].
A wide range—from 5.5% (macrolide-naïve patients) to 17.3%of estimated
H. pylori clarithromycin resistance is reported in Hungary, based on the literature [
3,
4]. In routine clinical practice, therefore, no equivocal data are available to guide treatment decisions. Hetero-resistance—where resistant and susceptible strains coexist however, may occur in ~7% of cases, which contributes to treatment failure even below classical thresholds [
5]. Based on the literature, and the continuous increase in the use of macrolide antibiotics, Hungary is considered as a clarithromycin-resistant area. Empirical eradication therapy in Hungary has typically used the amoxicillin–clarithromycin-based triple therapy in treatment-naïve patients. With rising macrolide use over time, however, treatment failures have become more common [
6]. Recent trends in European practice, using the Hp-EuReg registry, emphasize a shift toward a single capsule bismuth quadruple therapy, achieving eradication rates of 90–95% both as first line and rescue therapy in resistant settings. These results can only be achieved by a minimum of 10 days of eradication treatments [
7]. Although there is limited Hungary-specific data, the overall Hungarian antibiotic resistance pattern likely mirrors the broader Central European pattern, resulting in a general treatment success rate of around 80% [
8]. Based on published data,
H. pylori treatment success rate in Hungary is comparable with the international rate when tetracycline-based quadruple therapies were used (93.6%). Conventional clarithromycin-based regimens, however, only had a much lower, 75%, eradication rate [
9].
Regarding pharmaceutical access, the availability of tetracycline and bismuth salts (as individual agents and in fixed dose combinations) are limited in Hungary. Tetracycline is only available by individualized drug import; and bismuth drugs are only available in the form of bismuth oxide. Bismuth citrate, which is pharmacologically similar to the commercially available bismuth oxide, is available as a magistral preparation. A combination drug approved in Europe since 2009 that contains bismuth sub-citrate, metronidazole and tetracycline is not yet available in Hungary. Tetracycline and bismuth compounds are available as prescription medications in Hungary, but they are historically under-used due to entrenched triple therapy prescription habits. Furthermore, the Hungarian National Formulary gives the opportunity to prepare magistral bismuth–metronidazole, but it has a very limited availability (it is prepared only in one pharmacy) for use in routine clinical settings.
Hungary can therefore generally be considered a tetracycline and bismuth naïve area, so a possible shift towards using tetracycline containing eradication regimens could be a promising direction to make H. pylori eradication rates comparable to international statistics. As previously mentioned, despite the limitation of its availability, the classic bismuth-containing quadruple therapy (PPI, bismuth, tetracycline, and metronidazole) has been recommended as a first-line therapy in areas with high and uncleared clarithromycin resistance.
Tetracycline-based regimens typically contain bismuth as a supplement. It is well known that addition of bismuth to a 14-day standard triple therapy with clarithromycin and amoxicillin eradicates
H. pylori infection in more than 90% of patients, resulting in a potential therapeutic gain (10–20%) in populations with moderate to high clarithromycin resistance, with an acceptable safety profile and adherence rate [
8]. Although a range of data support bismuth addition to any eradication protocols [
10,
11,
12], comparison of the different currently available bismuth compounds (commercial bismuth oxide, magistral bismuth–citrate and bismuth–metronidazole) have not yet been compared for eradication efficiency. Meta-analyses and clinical studies also support the addition of probiotics to standard treatment protocols, showing that these adjuvant probiotic supplements modestly increase eradication rates while reducing antibiotic related side effects, especially when combined with tetracycline-based quadruple therapy [
13]. Data from the European registry also indicate that more frequent probiotic co-prescription is associated with improved tolerability and possibly better treatment adherence [
14,
15].
Limited data are available on the effect of different probiotic strains in this setting, with no data on the direct comparison of their effect on treatment efficacy. In many original articles, reviews, and meta-analyses, assessment of clinical symptoms was the trial outcome, with a presumed positive eradication result. In contrast, in this study, we reviewed treatment success rates without collecting patient-reported outcomes. A large meta-analysis of 25 randomized controlled trials (RCTs-3769 patients) evaluating single strains as adjuncts to standard eradication therapy found that only
Saccharomyces boulardii CNCM I-745 significantly improved eradication rates.
Lactobacillus rhamnosus GG,
Lactobacillus acidophilus,
Lactobacillus casei,
Lactobacillus reuteri,
Clostridium butyricum showed no clear benefit on eradication success [
13]. Another high-quality systematic review (2025; 5036 cases, 19 RCTs) confirmed that the addition of
Saccharomyces boulardii significantly increased eradication rates [
16]. The efficacy of Lactobacillus strains was investigated by a comprehensive RCT involving
Lactobacillus reuteri DSM17648 as adjunct to standard therapy, achieving eradication rates exceeding 90% [
17]. However, pooled RCTs in meta-analyses failed to show a statistically significant improvement in eradication rates for
Lactobacillus reuteri. As a head-to-head or combined probiotic comparison, a smaller RCT combined
Saccharomyces boulardii and
Lactobacillus reuteri with quadruple therapy. While data on direct comparison was limited, the combined regimen suggested a higher eradication rate and better tolerability. The sample size, however, was small, and no definitive strain efficacy separation was provided [
18].
Besides the lack of data for direct comparison of probiotic strains, their unique additional effect on different treatment protocols is still an open question. Combination use of both supplements (bismuth and probiotics) has not yet been examined regarding efficacy; however, promising potential can be foreseen in further enhancement of eradication success.
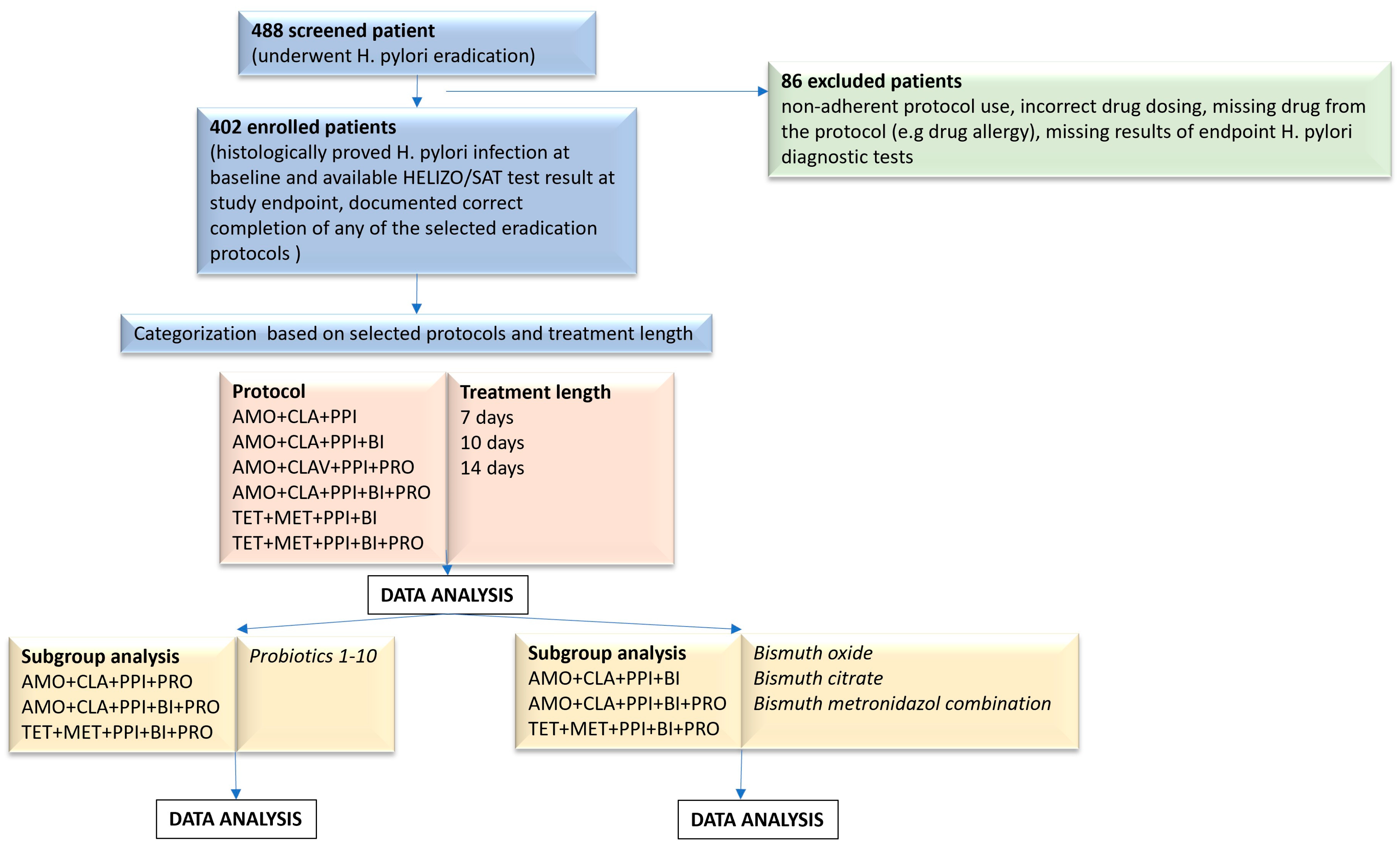
The aim of our retrospective real world data analysis of 402 cases was to give a comprehensive view and an optimalization of the routine use of clinical guidelines in H. pylori eradication in a clarithromycin-resistant and tetracycline-bismuth-naive area by covering the following topics: (1) description of trends regarding the number of eradication cases along with the protocols used, their treatment lengths and success rates, and the frequency of supplements used throughout the years in our clinic; (2) investigation of overall treatment protocol success rates; (3) determining the optimal duration of treatment; (4) finding the treatment protocol(s) that achieve the highest eradication rates; (5) clarifying whether the addition of probiotics improves treatment outcomes in different settings; (6) clarifying the effect of adding bismuth to clarithromycin-based protocols, and investigating the possible difference between bismuth compounds regarding treatment success; (7) finding the most effective probiotic strain and determining future efforts to improve patient care.
3. Discussion
Our retrospective study is an internationally unique and comprehensive assessment of the success rate of different
H. pylori eradication protocols that conducted in Hungary: 1. the amoxicillin–clarithromycin-based, and 2. the tetracycline-metronidazole-based protocols. Furthermore, the combination of three different treatment lengths, along with the use of two possible supplements (bismuth and probiotics), were compared in our study. Although previous studies are available describing gold standard one of these gold standard therapy protocols [
19,
20,
21], or a modified version, along with other treatment protocols [
22,
23,
24], comparison of the two current gold standard treatments along with the assessment of the usefulness of two promising supplements (bismuth and probiotics) was missing.
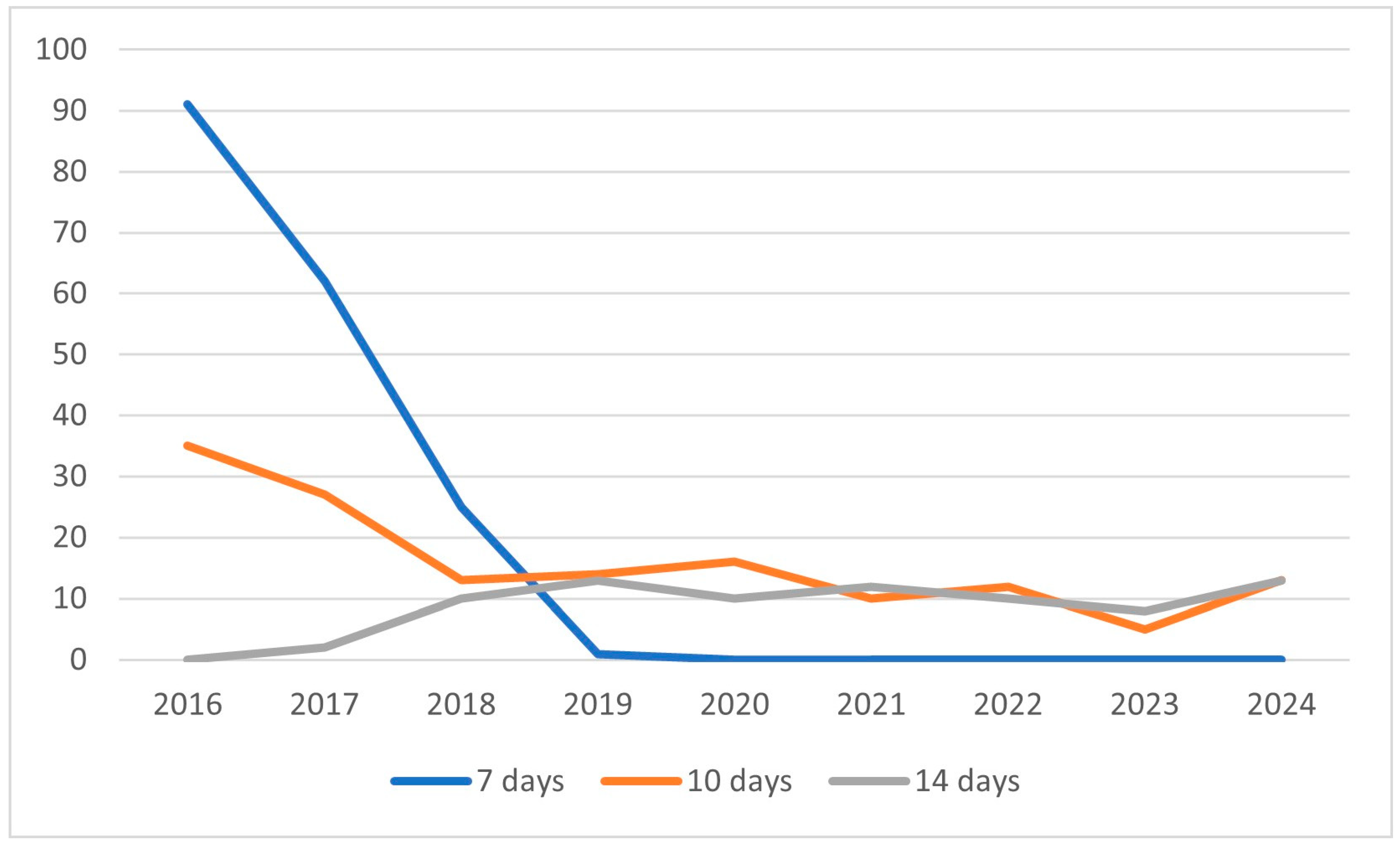
The first main result of this study is in line with previous publications and with the current treatment guidelines [
7,
8,
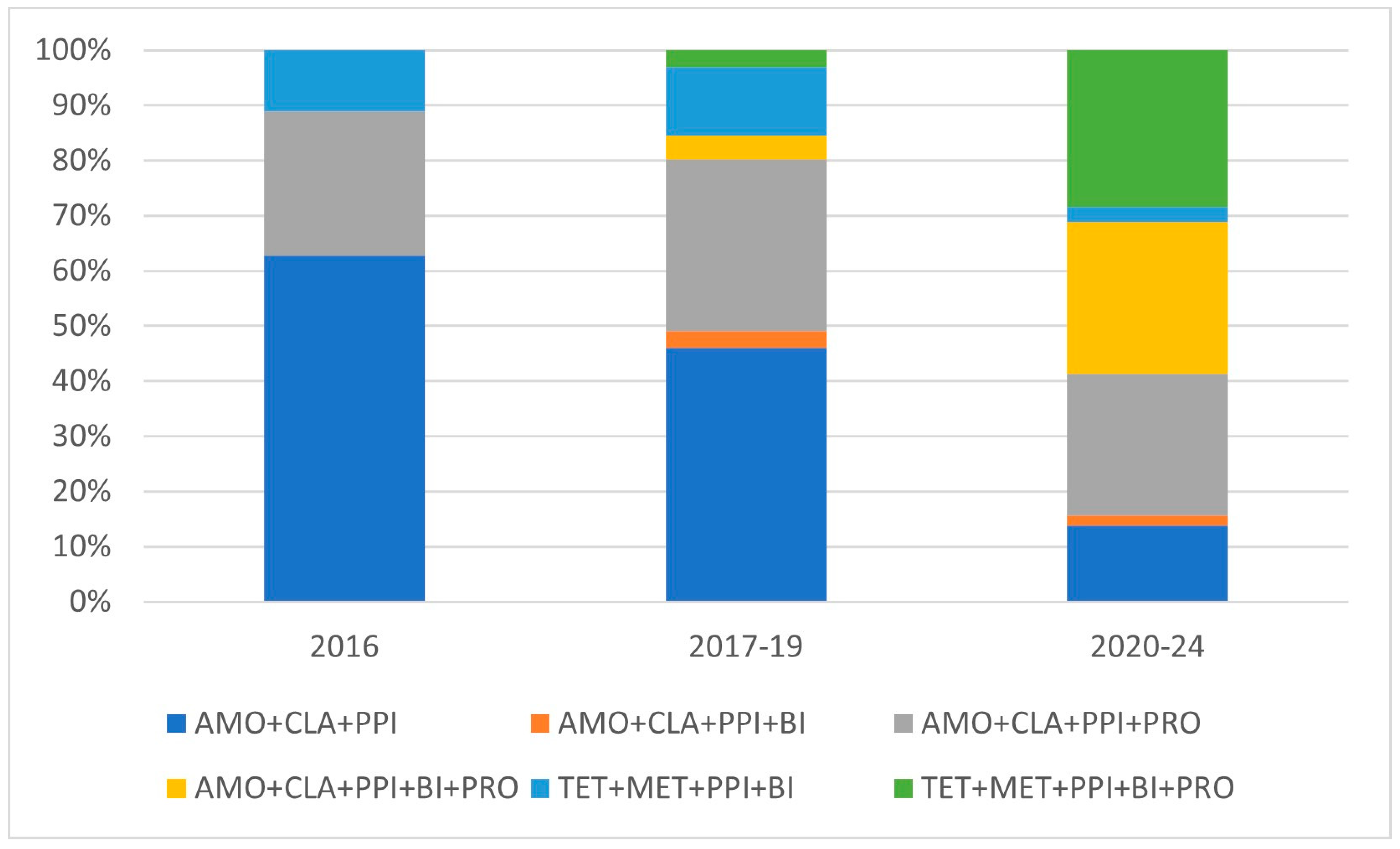
25], showing the superiority of the 14-day treatment duration compared to the 7- and 10-day treatments. Our analysis showed that treatment success rates were significantly higher in the three probiotic-containing regimens investigated (AMO + CLA + PPI + PRO, AMO + CLA + PPI + BI + PRO, TET + MET + PPI + BI + PRO) when compared to no-probiotic-containing treatment protocols. Furthermore, our results proved that, despite high local clarithromycin resistance, comparable success rates could be achieved by supplementation of the amoxicillin–clarithromycin-based protocols with bismuth and probiotics. Another relevant result of our study is that the most prominent effect of probiotics on treatment success rates with both treatment protocols were observed when they were also supplemented with bismuth. Hungary being a tetracycline-bismuth naive area, it is not unexpected that the best treatment success rates were seen with tetracycline-based protocols. Although in international clinical guidelines this treatment combination is suggested as first line-treatment, due to issues around tetracycline`s availability in Hungary, amoxicillin–clarithromycin-based protocols were often used in our clinic, showing non-inferior treatment success rates with supplementation.
As previously mentioned in the introduction, earlier data are available on the effect of different bismuth compounds; however, results of their comparison on treatment success are missing. In this study, direct comparison of different bismuth compounds (e.g., bismuth oxide, bismuth citrate and bismuth–metronidazole) were analyzed, showing an equivalent treatment success rate amongst the different types of product. Based on our data, we can say that the addition of any type of bismuth supplementation to any 14-day treatment protocols that include probiotics showed better overall treatment success rates in our cohort.
In our study, further analysis was performed to compare the effect of these four probiotics where sample size allowed statistical analysis. Our results, however, showed no statistical difference in success rates amongst these groups. The analysis included Probiotic 1 (Lactobacillus acidophilus, Lactobacillus bulgaricus, Bacillus coagulans, Bifidobacterium animalis lactis, Streptococcus thermophilus), Probiotic 2 (Enterococcus faecium L3), Probiotic 3 (Lactobacillus acidophilus (LA-5), Bifidobacterium (BB-12), Lactobacillus paracasei (L. CASEI 431)) and Probiotic 5 (Saccharomyces boulardii CNCM I-745). In previous literature (see introduction section), only Saccharomyces boulardii was found to increase eradication success rates in RCTs, which also showed a beneficial effect in our study. Our data, however, also showed non-inferiority of the other species listed. Future randomized controlled clinical trials and retrospective studies should aim to confirm the usefulness of Lactobacillus and other strains in a bigger cohort.
An important finding of our study is that Probiotic 8 (Lactobacillus reuteri) supplementation, i.e., the “PRO” part of AMO + CLA + PPI + BI + PRO and TET + MET + PPI + BI + PRO treatment protocols, had a very high 85.2% and 100% treatment success rate, respectively. This finding also requires further studies with larger case numbers.
In conclusion, when following clinical recommendations is not possible due to limitations in tetracycline availability, comparable success rates can be achieved using clarithromycin-based protocols with probiotics and bismuth in a 14-day regimen. The cost of the two basic protocols is approximately the same, so cost considerations should not influence treatment choices in a real-world setting.
As this was a retrospective trial in which we collected real-world data on local clinical routines, a limitation existed in the statistical analysis and interpretation of results in that physician choice might have influenced the treatment protocol or the added probiotic strains. Therefore, as can be seen in
Table 12, each treatment protocol was mainly supplemented by the same probiotics, as physicians used their preferred treatments for patient care, which caused a shift in case numbers, making statistical analysis impossible.
Besides clinical trials, several data sources from microbiological and other preclinical studies support the beneficial effects of probiotics on
H. pylori eradication. As this study is a database search, only a brief explanation of the mechanism of action is provided here. Probiotics affect the colonization of
H. pylori in several ways: they interfere with bacterial adhesion to epithelial cells [
26]; inhibit bacterial enzyme activity (urease, catalase, and carbonic anhydrase); and interfere with the secretion of antibacterial substances (e.g., bacteriocins such as reuterin, lacticin, and bulgaricin) [
27]. Probiotics also have a negative effect on biofilm formation and modulate the host cell immune response to infection by influencing the balance of pro- and anti-inflammatory cytokines (e.g., IL-8, IL-10, TNFα, IL-1β, and IL-4) [
28,
29,
30]. They also regulate gastric microecology. Probiotic supplementation can regulate gastric flora structure and promote gastric microecological recovery, which is considered to be related to
H. pylori eradication [
31]. Especially, quadruple antibiotic therapy for
H. pylori infection can exacerbate gastrointestinal microecological disorders. Probiotics, on the other hand, restore the balance of intestinal flora through different mechanisms, improve eradication rates, and reduce adverse reactions [
32]. In conclusion, probiotics play an important role in the prevention and treatment of gastrointestinal diseases by antagonizing
H. pylori colonization, regulating gastric pH, secreting antibacterial substances, stimulating immune responses, and regulating gastrointestinal flora [
15,
33,
34].
Based on our data, a promising future direction would be to carry out a randomized clinical trial, inorder to directly compare the treatment success of the addition of probiotics to amoxicillin- vs. tetracycline-based bismuth regimens. A possible shift to longer (14-day) treatments raises safety and adherence issues that require further investigation. In addition, the optimal dosage and the choice of the best probiotic strain, along with treatment duration and timing of supplementation relative to antibiotics treatment, would also require further study and standardization before an official recommendation can be made (study results are summarized at
Table 13).
4. Materials and Methods
4.1. Patients
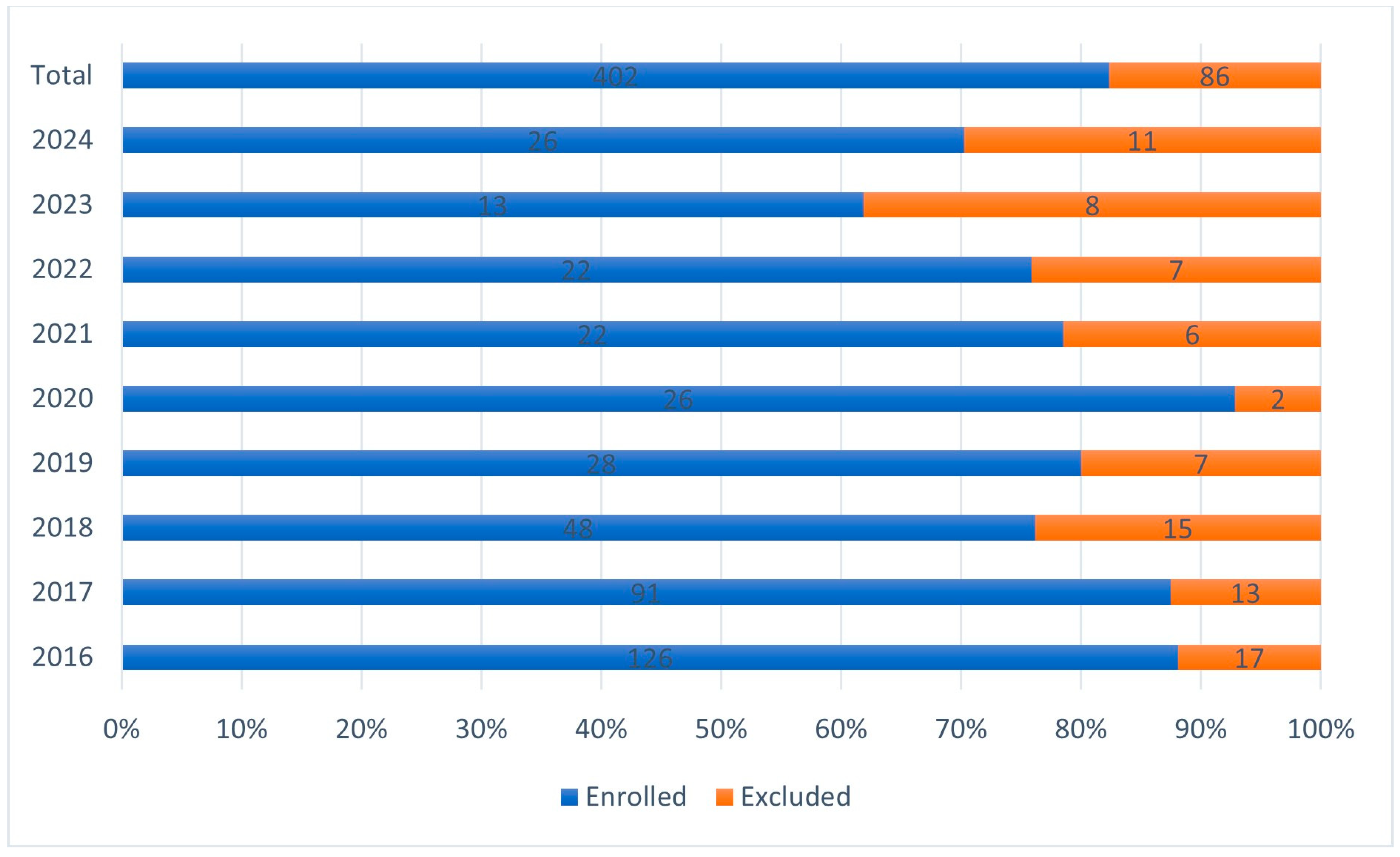
In this real-world retrospective trial, the medical records of patients who underwent H. pylori eradication in our outpatient clinic were investigated in a nine-year period (2016–2024). A total number of 486 eradication cases were screened and 402 cases were selected who met the inclusion criteria and enrolled to our trial after exclusion of non-suitable cases (see exclusion criteria below) and underwent further data collection.
4.2. Diagnostic Tools
The initial diagnosis of H. pylori infection was performed on gastric antrum biopsies, using histological examination, which used Giemsa staining or immunohistochemistry (H. pylori rabbit monoclonal antibody manufactured by Sigma-Aldrich, St. Louis, MO, USA), the latter in cases where Giemsa staining showed questionable results. Envision Target Retrieval System was used for detection purposes. For the determination of treatment outcome, results of HELIZO™ exhalation tests (performed at the Nuclear Medicine Department of Buda Hospital of the Hospitaller Order of Saint John of God), or stool Helicobacter antigen (SAT) tests (provided by the Central Laboratory of Buda Hospital of the Hospitaller Order of Saint John of God) were used. The HELIZO™ exhalation tests are based on the 14C content of exhaled air, containing radioactively labelled carbamide. The HELIZO™ exhalation test and HELIPROBE™ equipment, used for detection, were manufactured by Isotope Institute Ltd, Budapest, Hungary. For stool antibody testing, DiaQuick H. pylori stool cassette (sensitivity 98.8%, specificity 98.4%, manufactured by DiaLab Ltd., Neudorf, Austria) were used.
4.3. Database Search and Case Screening
Data collection was performed trough institutional database search (EMMA™). The basis of patient screening in our study was a histologically proven H. pylori infection, along with the availability of either the HELIZO™ exhalation tests or stool Helicobacter antigen (SAT) tests at study endpoint.
4.4. Inclusion Criteria
We selected only patient data that met all the inclusion criteria: (1) the initial diagnosis of H. pylori infection, (2) the presence of the final result on eradication by the diagnostic tests (HELIZO™ or SAT) 6 to 8 weeks after completing eradication treatment, (3) and the availability of prescription details of antibiotics used for eradication (agent name or drug trade name and dose).
4.5. Exclusion Criteria
Case documentations containing incomplete data (e.g., missing treatment length, exact type of medications), as well as treatments deviating from the protocol (e.g., alternative combinations due to the patient’s drug allergy, the patient’s intolerance to one of the components, or any conditions resulting in earlier termination of the prescribed treatment) were not included in the final statistical calculation. All eradication cases were recorded regardless of which therapy protocol (in first line, or after a failure as second or later attempt) the patient received.
4.6. Determination of Investigated Outcomes in Database
“Baseline- H. pylori infection” meant that the initial histology proved H. pylori infection. Cases where the result of HELIZO™ or SAT test were negative after completing eradication treatment were labelled “Success”. In contrast, positive HELIZO™ or SAT tests after treatment were labelled as “Failure”.
4.7. Antibiotics Combinations, PPI and Supplementations
Commercially available amoxicillin, clarithromycin and PPIs are most often used in our clinical practice. The tetracycline used was a commercially available medication that was available only via an individual import process through one pharmacy in Hungary. Several efforts were made to improve success rates in clinical routine by using bismuth compounds as a part of tetracycline-based regimes and as a supplement in clarithromycin-based protocols. Although the commercially available bismuth oxide is pharmacologically similar to the bismuth–citrate magistral preparation (in pharmacological nomenclature they are considered synonyms), due to their different preparation methods, they were treated as two separate entities in our study. In some cases, magistral bismuth–metronidazole capsules were also prescribed by a physician in our clinic. Regarding the probiotics, commercially available probiotic strains were used at the choice of the gastroenterologist.
4.8. Treatment Protocol
In line with the international recommendations, two standard types of eradication protocol, a clarithromycin-based (AMO + CLA + PPI), and an original tetracycline-based (TET + MET + PPI) were collected from the database. As Hungary is considered to be a clarithromycin-resistant area, and because the tetracycline and bismuth compound availability is limited, physicians altered the clarithromycin-based protocol by adding supplements, mainly probiotics, and in some cases bismuth compounds (AMO + CLA + PPI ± BI ± PRO) in order to improve treatment success rates. The tetracycline-based protocols normally contain bismuth, and in many cases they are supplemented with probiotics (TET + MET + BI ± PRO). Therefore, the following eradication therapies were seen in our clinic: (1) standard triple clarithromycin-based protocol containing amoxicillin, clarithromycin, proton pump inhibitor, (AMO + CLAV + PPI); (2) a quadruple clarithromycin-based combination containing amoxicillin, clarithromycin, proton pump inhibitor and bismuth (AMO + CLA + PPI + BIZ), (3) a quadruple clarithromycin-based protocol containing amoxicillin, clarithromycin, proton pump inhibitor with probiotics (AMO + CLA + PPI + PRO), (4) a quintuple protocol of amoxicillin, clarithromycin, proton pump inhibitor, supplemented both bismuth and probiotics (AMO + CLA + PPI + BIZ + PRO). The other types of investigated regimens were the standard (5) tetracycline + metronidazole + PPI + bismuth regimen (TET + MET + PPI + BIZ), (6) supplemented with probiotics (TET+ MET + PPI + BIZ + PRO) (
Table 14 and
Table 15). The treatment lengths of all the above combinations were 7, 10 or 14 days. The type of proton pump inhibitor was not examined in this study, therefore no details are provided. In all cases, however, PPIs were given in their standard prescription doses. Any other prescription or non-prescription medications were excluded from our data collection.
4.9. Investigation of Supplements
As a separate part of our study, we examined the additional value of ten different probiotic strains in those three protocols where they were used (AMO + CLA + PPI + PRO, AMO + CLA + PPI + BIZ + PRO, TET + MET + PPI + BIZ + PRO) (
Table 15). Additionally, bismuth was given in three different formulations for these treatment protocols (AMO + CLA + PPI +BIZ, AMO + CLA + PPI + BIZ + PRO, TET + MET + PPI + BIZ, TET+ MET + PPI + BIZ + PRO), using appropriate dosing: (1) commercially available bismuth oxide, (2) magistral preparation of bismuth–citrate compound, (3) magistral preparation of bismuth–metronidazole combination (
Table 16).
4.10. Database Setup
The data collected were saved in a Microsoft Excel table. Physicians were anonymized and marked with randomly assigned numbers. Collected data were as follows: date of birth and gender of the patient, length of the treatment, type of eradication protocol and medications used (antibiotics, bismuth and proton pump inhibitor and doses), additional probiotic or bismuth use (where applicable), and outcome of the therapy (success or failure) based on HELIZO™ or SAT test results. No data on patient reported outcomes were collected (e.g., symptoms, compliance) due to the retrospective nature of our study, and because the main goal of the study was to optimize social guidelines for our daily clinical practice.
4.11. Statistical Analysis
Statistical analysis was performed using TIBCO Statistica Software (version 14.1.0). The calculation was based on 2 × 2 or in some cases r × c contingence tables using the chi-square test. The significance level was 5%.