Evaluation of Ceftaroline Use in Pediatric Patients: A Retrospective Case Series
Abstract
1. Introduction
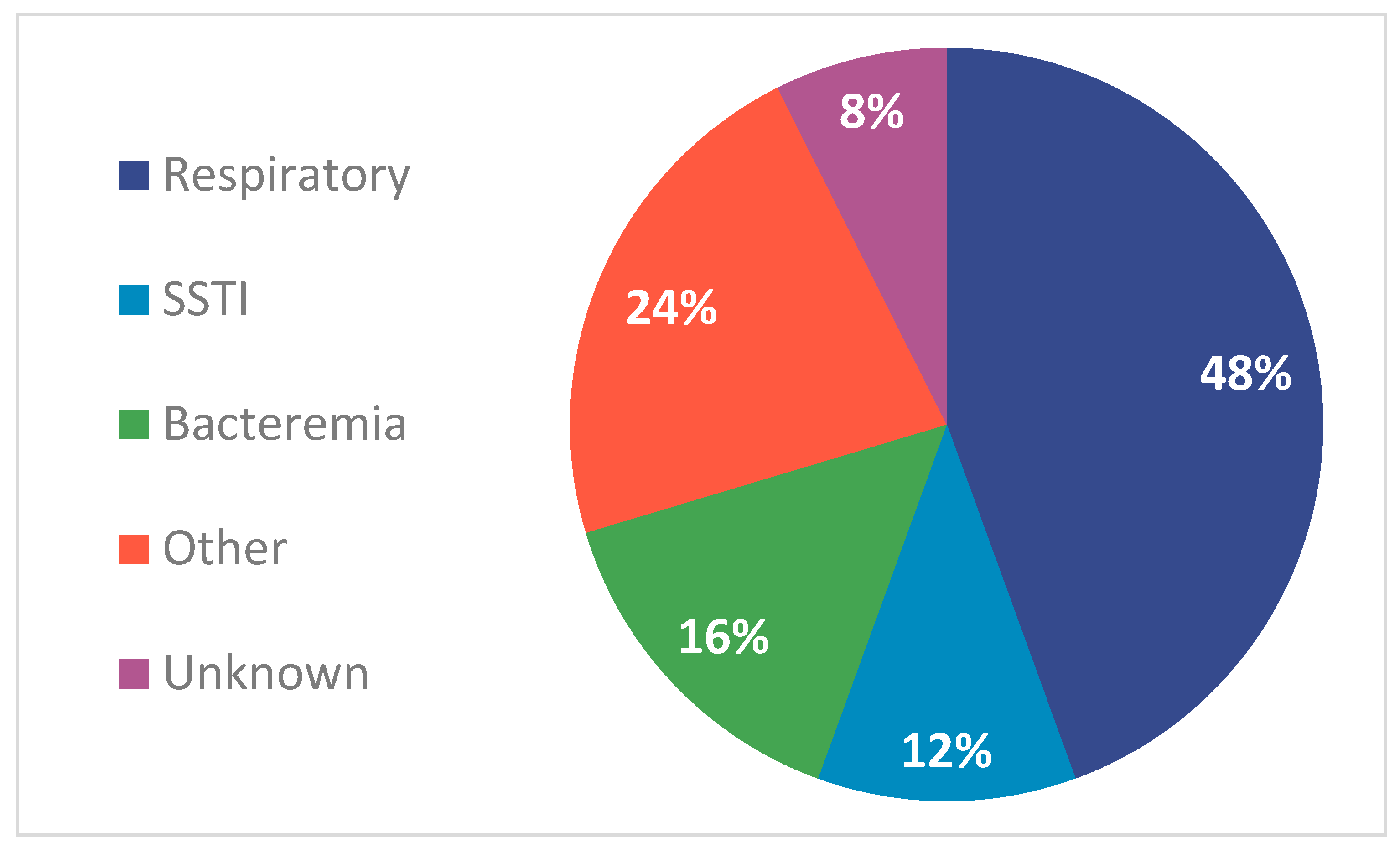
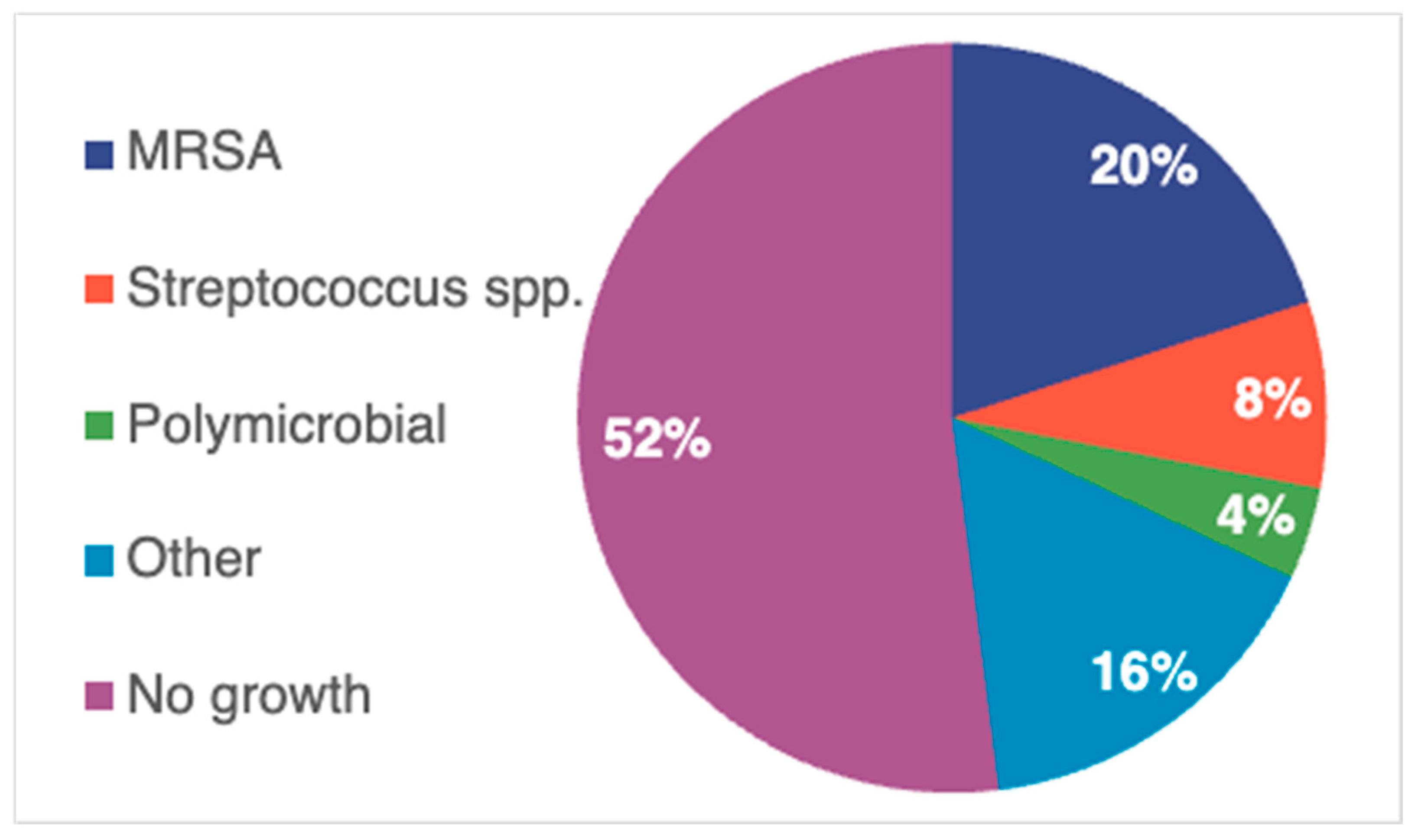
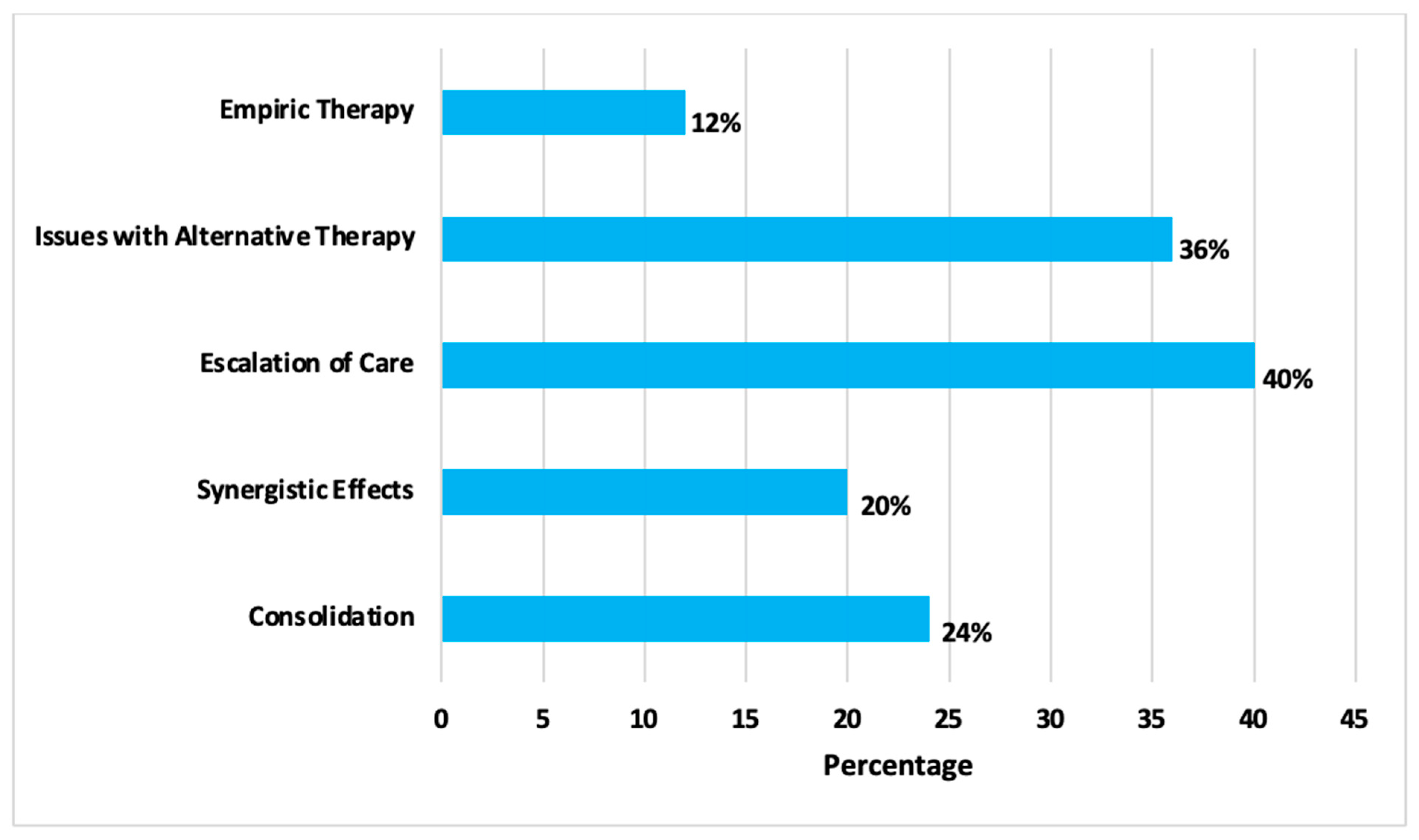
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Romandini, A.; Pani, A.; Schenardi, P.A.; Pattarino, G.A.C.; De Giacomo, C.; Scaglione, F. Antibiotic resistance in pediatric infections: Global emerging threats, predicting the near future. Antibiotics 2021, 10, 393. [Google Scholar] [CrossRef]
- World Health Organization. Shaping the Global Innovation and Access Landscape for Better Paediatric Medicines; World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2019.
- Prochaska, E.C.; Xiao, S.; Tamma, P.D.; Sick-Samuels, A.; Schumacher, C.; Gadala, A.; Carroll, K.C.; Milstone, A.M. Trends in pediatric community-onset Staphylococcus aureus antibiotic susceptibilities over a five-year period in a multihospital health system. Antimicrob. Steward. Healthc. Epidemiol. 2023, 3, e12. [Google Scholar] [CrossRef] [PubMed]
- Gerber, J.S.; Coffin, S.E.; Smathers, S.A.; Zaoutis, T.E. Trends in the incidence of methicillin-resistant Staphylococcus aureus infection in children’s hospitals in the United States. Clin. Infect. Dis. 2009, 49, 65–71. [Google Scholar] [CrossRef]
- Chiusaroli, L.; Liberati, C.; Rulli, L.; Barbieri, E.; De Pieri, M.; Di Chiara, C.; Mengato, D.; Giaquinto, C.; Donà, D. Therapeutic options and outcomes for the treatment of children with gram-positive bacteria with resistances of concern: A systematic review. Antibiotics 2023, 12, 261. [Google Scholar] [CrossRef] [PubMed]
- Rybak, M.J.; Le, J.; Lodise, T.P.; Levine, D.P.; Bradley, J.S.; Liu, C.; Mueller, B.A.; Pai, M.P.; Wong-Beringer, A.; Rotschafer, J.C.; et al. Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: A revised consensus guideline and review by the American Society of Health-System Pharmacists, the Infectious Disease Society of America, The Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Am. J. Health System Pharm. 2020, 77, 835–864. [Google Scholar]
- Daptomycin [Package Insert]; Xellia Pharmaceuticals USA, LLC.: Buffalo Grove, IL, USA, 2023.
- Linezolid [Package Insert]; Pfizer: New York, NY, USA, 2013.
- Clemett, D.; Markham, A. Linezolid. Drugs 2000, 59, 815–827. [Google Scholar] [CrossRef]
- Woytowish, M.R.; Maynor, L.M. Clinical relevance of linezolid-associated serotonin toxicity. Ann. Pharmacother. 2013, 47, 388–397. [Google Scholar] [CrossRef]
- Duplessis, C.; Crum-Cianflone, N.F. Ceftaroline: A new cephalosporin with activity against methicillin-resistant Staphylococcus aureus (MRSA). Clin. Med. Rev. Ther. 2011, 3, a2466. [Google Scholar] [CrossRef]
- Croisier-Bertin, D.; Piroth, L.; Charles, P.E.; Larribeau, A.; Biek, D.; Ge, Y.; Chavanet, P. Ceftaroline versus ceftriaxone in a highly penicillin-resistant pneumococcal pneumonia rabbit model using simulated human dosing. Antimicrob. Agents Chemother. 2011, 55, 3557–3563. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Biedenbach, D.J.; Iaconis, J.P.; Sahm, D.F. Comparative in vitro activities of ceftaroline and ceftriaxone against bacterial pathogens associated with respiratory tract infections: Results from the AWARE surveillance study. J. Antimicrob. Chemother. 2016, 71, 3459–3464. [Google Scholar] [CrossRef]
- Kempf, M.; Arhin, F.F.; Kuraieva, A.; Utt, E. In vitro activity of ceftaroline against isolates of gram-positive bacteria from patients with bloodstream infections collected as a part of ATLAS between 2017 and 2020. Infect. Drug Resist. 2024, 17, 343–354. [Google Scholar] [CrossRef]
- Utt, E.; Kantecki, M.; Cabezas-Camarero, G.; Esposito, S. Evaluation of in vitro activity of ceftaroline against pathogens associated with community-acquired pneumonia: ATLAS program 2017–2019. J. Glob. Antimicrob. Resist. 2023, 33, 360–367. [Google Scholar] [CrossRef]
- Ceftaroline [Package Insert]; AbbVie Inc.: North Chicago, IL, USA, 2024.
- Valentino, M.S.; Borgia, P.; Deut, V.; Lorenzi, I.; Barabino, P.; Ugolotti, E.; Mariani, M.; Bagnasco, F.; Castagnola, E. Changes in the Use of Antibiotics for Methicillin-Resistant Staphylococcus aureus Bloodstream Infections in Children: A 5-Year Retrospective, Single Center Study. Antibiotics 2023, 12, 216. [Google Scholar] [CrossRef]
- Branstetter, J.; Searcy, H.; Benner, K.; Yarbrough, A.; Crowder, C.; Troxler, B. Ceftaroline vs vancomycin for the treatment of acute pulmonary exacerbations in pediatric patients with cystic fibrosis. Pediatr. Pulmonol. 2020, 55, 3337–3342. [Google Scholar] [CrossRef]
- Callies, A.; Martin-Perceval, L.; Crémet, L.; Gély, L.; Ruellan, A.-L.; Verdier, M.-C.; Gregoire, M.; Flamant, C.; Guillouzouic, A.; Prot-Labarthe, S.; et al. Safety and efficacy of ceftaroline in neonates with staphylococcal late-onset sepsis: A case series analysis. Pediatr. Infect. Dis. J. 2023, 42, 888–892. [Google Scholar] [CrossRef]
- Cies, J.J.; Moore, W.S., 2nd; Enache, A.; Chopra, A. Ceftaroline for suspected or confirmed invasive methicillin-resistant Staphylococcus aureus: A pharmacokinetic case series. Pediatr. Crit. Care Med. 2018, 19, e292–e299. [Google Scholar] [CrossRef]
- Williams, A.W.; Newman, P.M.; Ocheltree, S.; Beaty, R.; Hassoun, A. Ceftaroline fosamil use in 2 pediatric patients with invasive methicillin-resistant Staphylococcus aureus infections. J. Pediatric Pharmacol. Ther. 2015, 20, 476–480. [Google Scholar] [CrossRef]
- Soriano, A.; Bassetti, M.; Gogos, C.; Ferry, T.; de Pablo, R.; Ansari, W.; Kantecki, M.; Schweikert, B.; Luna, G.; Blasi, F. Ceftaroline fosamil treatment patterns and outcomes in adults with community-acquired pneumonia: A real-world multinational, retrospective study. JAC Antimicrob. Resist. 2024, 6, dlae078. [Google Scholar] [CrossRef]
- Ferry, T.; Gogos, C.; Soriano, A.; Blasi, F.; Ansari, W.; Kantecki, M.; Schweikert, B.; Luna, G.; Bassetti, M. Real-world use and treatment outcomes of ceftaroline fosamil in patients with complicated skin and soft tissue infection: A multinational retrospective study. Infect. Drug Resist. 2024, 17, 2773–2783. [Google Scholar] [CrossRef]
- de la Villa, S.; Escrihuela-Vidal, F.; Fernández-Hidalgo, N.; Escudero-Sánchez, R.; Cabezón, I.; Boix-Palop, L.; Díaz-Pollán, B.; Goikoetxea, A.J.; García-País, M.J.; Pérez-Rodríguez, M.T.; et al. Ceftaroline for bloodstream infections caused by methicillin-resistant Staphylococcus aureus: A multicentre retrospective cohort study. Clin. Microbiol. Infect. 2025, 31, 793–801. [Google Scholar] [CrossRef]
- Hammond, J.; Benigno, M.; Bleibdrey, N.; Ansari, W.; Nguyen, J.L. Ceftaroline fosamil for the treatment of methicillin-resistant Staphylococcus aureus bacteremia: A real-world comparative clinical outcomes study. Drugs Real. World Outcomes. 2024, 11, 273–283. [Google Scholar] [CrossRef]
- Liu, C.; Bayer, A.; Cosgrove, S.E.; Daum, R.S.; Fridkin, S.K.; Gorwitz, R.J.; Kaplan, S.L.; Karchmer, A.W.; Levine, D.P.; Murray, B.E.; et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin. Infect. Dis. 2011, 52, e18–e55. [Google Scholar] [CrossRef]
- Haynes, A.S.; Maples, H.; Parker, S. Time for a change: Considering vancomycin alternatives for pediatric methicillin-resistant Staphylococcus aureus bacteremia. J. Pediatric Infect. Dis. Soc. 2023, 12, 308–318. [Google Scholar] [CrossRef]
- Rodvold, K.A.; McConeghy, K.W. Methicillin-resistant Staphylococcus aureus therapy: Past, present, and future. Clin. Infect. Dis. 2014, 58 (Suppl. S1), S20–S27. [Google Scholar] [CrossRef]
- Riccobene, T.A.; Khariton, T.; Knebel, W.; Das, S.; Li, J.; Jandourek, A.; Carrothers, T.J.; Bradley, J.S. Population PK modeling and target attainment simulations to support dosing of ceftaroline fosamil in pediatric patients with acute bacterial skin and skin structure infections and community-acquired pneumonia. J. Clin. Pharmacol. 2017, 57, 345–355. [Google Scholar] [CrossRef]
- Chan, P.L.S.; McFadyen, L.; Quaye, A.; Leister-Tebbe, H.; Hendrick, V.M.; Hammond, J.; Raber, S. The use of extrapolation based on modeling and simulation to support high-dose regimens of ceftaroline fosamil in pediatric patients with complicated skin and soft-tissue infections. CPT Pharmacometrics Syst. Pharmacol. 2021, 10, 551–563. [Google Scholar] [CrossRef]
- Korczowski, B.; Antadze, T.; Giorgobiani, M.; Stryjewski, M.E.; Jandourek, A.; Smith, A.; O’nEal, T.; Bradley, J.S. A multicenter, randomized, observer-blinded, active-controlled study to evaluate the safety and efficacy of ceftaroline versus comparator in pediatric patients with acute bacterial skin and skin structure infection. Pediatr. Infect. Dis. J. 2016, 35, e239–e247. [Google Scholar] [CrossRef]
- Blumer, J.L.; Ghonghadze, T.; Cannavino, C.; O’nEal, T.; Jandourek, A.; Friedland, H.D.; Bradley, J.S. A multicenter, randomized, observer-blinded, active-controlled study evaluating the safety and effectiveness of ceftaroline compared with ceftriaxone plus vancomycin in pediatric patients with complicated community-acquired bacterial pneumonia. Pediatr. Infect. Dis. J. 2016, 35, 760–766. [Google Scholar] [CrossRef]
- Cannavino, C.R.; Nemeth, A.; Korczowski, B.; Bradley, J.S.; O’nEal, T.; Jandourek, A.; Friedland, H.D.; Kaplan, S.L. A randomized, prospective study of pediatric patients with community-acquired pneumonia treated with ceftaroline versus ceftriaxone. Pediatr. Infect. Dis. J. 2016, 35, 752–759. [Google Scholar] [CrossRef]
- Coella, R.; Glynn, J.R.; Gaspar, C.; Picazo, J.J.; Fereres, J. Risk factors for developing clinical infection with methicillin-resistant Staphylococcus aureus (MRSA) amongst hospital patients initially only colonized with MRSA. J. Hosp. Infect. 1997, 37, 39–46. [Google Scholar] [CrossRef]
- Sadoyama, G.; Gontijo Filho, P.P. Risk factors for methicillin-resistant and sensitive Staphylococcus aureus infection in a Brazilian university hospital. Braz. J. Infect. Dis. 2000, 4, 135–143. [Google Scholar]
- Epstein, L.; Me, Y.; Belflower, R.; Scott, J.; Ray, S.; Dumyati, G.; Felsen, C.; Petit, S.; Yousey-Hindes, K.; Nadle, J.; et al. Risk factors for invasive methicillin-resistant Staphylococcus aureus infection after recent discharge from an acute care hospitalization, 2011–2013. Clin. Infect. Dis. 2015, 62, 45–52. [Google Scholar] [CrossRef]
- Tsai, C.-E.; Yang, C.-J.; Chuang, Y.-C.; Wang, J.-T.; Sheng, W.-H.; Chen, Y.-C.; Chang, S.-C. Evaluation of the synergistic effect of ceftaroline against methicillin-resistant Staphylococcus aureus. Int. J. Infect. Dis. 2022, 122, 230–236. [Google Scholar] [CrossRef]
- Barber, K.E.; Werth, B.J.; Rybak, M.J. The combination of ceftaroline plus daptomycin allows for therapeutic de-escalation and daptomycin sparing against MRSA. J. Antimicrob. Chemother. 2015, 70, 505–509. [Google Scholar] [CrossRef]
| Parameters | n = 25 |
|---|---|
| Male | 17 (68) |
| Age, years, median (IQR) | 3.4 (1.4–14.3) |
| Age < 2 years | 8 (32) |
| Age 2–11 years | 10 (40) |
| Age 12–17 years | 7 (28) |
| Weight, kg, median (IQR) | 15.7 (10.7–54.4) |
| Race | |
| White | 11 (44) |
| Black | 9 (36) |
| ICU Admission | |
| PICU | 14 (56) |
| PCICU | 1 (4) |
| NICU | 1 (4) |
| MRSA Risk Factors | |
| Hospitalization within 12 months | 9 (36) |
| Antibiotic exposure within 12 months | 11 (44) |
| MRSA colonization or infection within 12 months | 0 (0) |
| Invasive procedures within 12 months | 6 (24) |
| Long-term central venous access within 12 months | 11 (44) |
| Prior ABSSSI within 12 months | 0 (0) |
| Vital Signs and Laboratory Values *, median (IQR) | |
| Temperature, °Celsius | 37.2 (36.7–37.9) |
| HR, beats per minute | 126.0 (115.0–155.0) |
| RR, breaths per minute | 26.0 (24.0–36.0) |
| WBC, ×109/L | 8.2 (6.2–17.0) |
| CRP, mg/L ^ | 12.8 (11.8–17.2) |
| Procalcitonin, ng/mL ^^ | 0.4 (0.3–3.9) |
| SCr, mg/dL ^^^ | 0.5 (0.3–0.7) |
| Parameters | n = 25 |
|---|---|
| Hospital length of stay, days, median (IQR) | 10.0 (7.8–34.8) |
| Pediatric ID consult | 25 (100) |
| CPT recommended from pediatric ID | 22 (88) |
| Antibiotics received prior to CPT initiation | 22 (88) |
| Total duration of antibiotics prior to CPT initiation (hours), median (IQR) | 73.0 (20.9–139.8) |
| CPT 12 mg/kg q8h | 9 (36) |
| CPT 8 mg/kg q8h | 7 (28) |
| Duration of CPT, days, median (IQR) | 4.6 (1.7–10.0) |
| Concomitant antibiotics | 16 (64) |
| CPT at discharge | 4 (16) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miller, A.; Grizzle, M.; Van Poppel, H.; Alvira-Arill, G.R.; Lueking, R.; Thacker, S.A.; Hornback, K.M.; Morrisette, T. Evaluation of Ceftaroline Use in Pediatric Patients: A Retrospective Case Series. Antibiotics 2025, 14, 864. https://doi.org/10.3390/antibiotics14090864
Miller A, Grizzle M, Van Poppel H, Alvira-Arill GR, Lueking R, Thacker SA, Hornback KM, Morrisette T. Evaluation of Ceftaroline Use in Pediatric Patients: A Retrospective Case Series. Antibiotics. 2025; 14(9):864. https://doi.org/10.3390/antibiotics14090864
Chicago/Turabian StyleMiller, Amy, Madison Grizzle, Halee Van Poppel, Gustavo R. Alvira-Arill, Richard Lueking, Stephen A. Thacker, Krutika Mediwala Hornback, and Taylor Morrisette. 2025. "Evaluation of Ceftaroline Use in Pediatric Patients: A Retrospective Case Series" Antibiotics 14, no. 9: 864. https://doi.org/10.3390/antibiotics14090864
APA StyleMiller, A., Grizzle, M., Van Poppel, H., Alvira-Arill, G. R., Lueking, R., Thacker, S. A., Hornback, K. M., & Morrisette, T. (2025). Evaluation of Ceftaroline Use in Pediatric Patients: A Retrospective Case Series. Antibiotics, 14(9), 864. https://doi.org/10.3390/antibiotics14090864






