Whole-Genome Sequencing and Molecular Analysis of Ceftazidime–Avibactam-Resistant KPC-Producing Klebsiella pneumoniae from Intestinal Colonization in Elderly Patients
Abstract
1. Introduction
2. Results
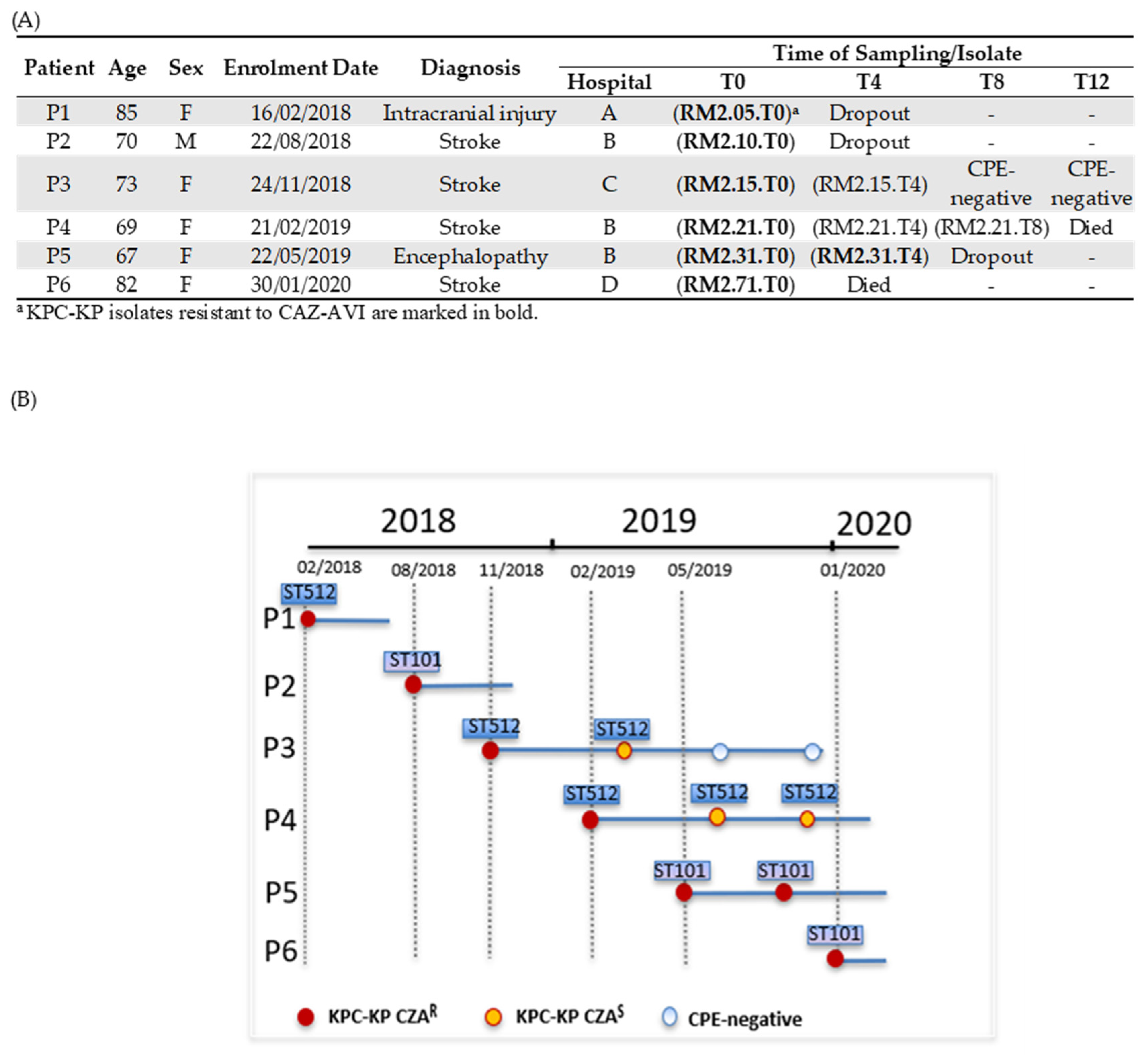
2.1. Patients Colonized by KPC-KP Resistant to CAZ-AVI and KPC-KP Isolates
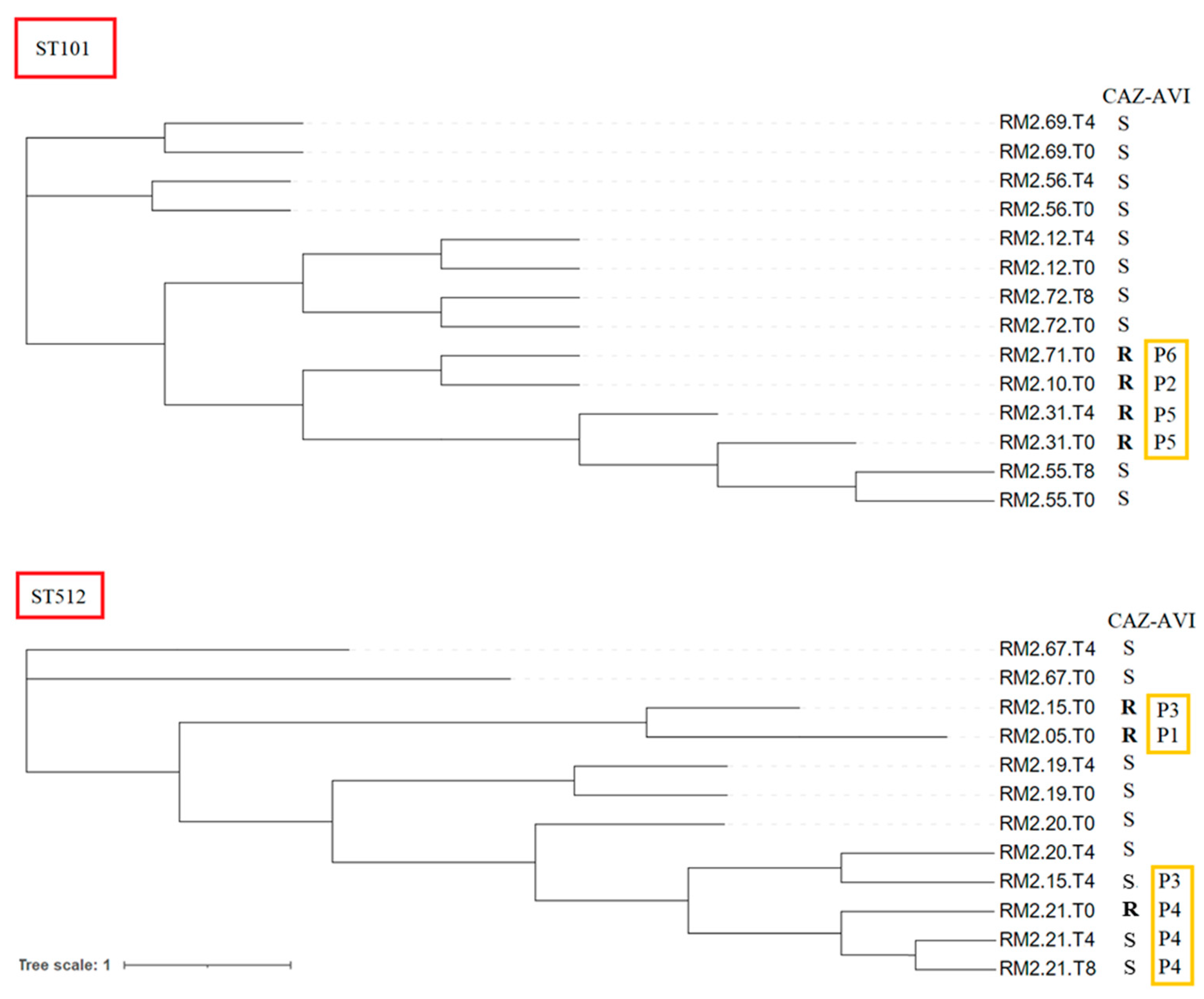
2.2. Whole-Genome Sequencing and In Silico Analysis
3. Discussion
4. Materials and Methods
4.1. Bacterial Strain Identification and Antimicrobial Susceptibility Testing (AST)
4.2. Whole-Genome Sequencing and In Silico Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- David, S.; Reuter, S.; Harris, S.R.; Glasner, C.; Feltwell, T.; Argimon, S.; Abudahab, K.; Goater, R.; Giani, T.; Errico, G.; et al. Epidemic of carbapenem-resistant Klebsiella pneumoniae in Europe is driven by nosocomial spread. Nat. Microbiol. 2019, 4, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.; Adler, A.; Abu-Hanna, J.; Percia, S.C.; Matalon, M.K.; Carmeli, Y. Spread of KPC-producing carbapenem-resistant Enterobacteriaceae: The importance of super-spreaders and rectal KPC concentration. Clin. Microbiol. Infect. 2015, 21, 470.e1–470.e7. [Google Scholar] [CrossRef]
- Tumbarello, M.; Viale, P.; Viscoli, C.; Trecarichi, E.M.; Tumietto, F.; Marchese, A.; Spanu, T.; Ambretti, S.; Ginocchio, F.; Cristini, F.; et al. Predictors of Mortality in Bloodstream Infections Caused by Klebsiella pneumoniae Carbapenemase-Producing K. pneumoniae: Importance of Combination Therapy. Clin. Infect. Dis. 2012, 55, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Di Pilato, V.; Errico, G.; Monaco, M.; Giani, T.; Del Grosso, M.; Antonelli, A.; David, S.; Lindh, E.; Camilli, R.; Aanensen, D.M.; et al. The changing epidemiology of car-bapenemase-producing Klebsiella pneumoniae in Italy: Toward polyclonal evolution with emergence of high-risk lineages. J. Antimicrob. Chemother. 2021, 76, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Porreca, A.M.; Sullivan, K.V.; Gallagher, J.C. The Epidemiology, Evolution, and Treatment of KPC-Producing Organisms. Curr. Infect. Dis. Rep. 2018, 20, 13. [Google Scholar] [CrossRef]
- Poulakou, G.; Bassetti, M.; Righi, E.; Dimopoulos, G. Current and future treatment options for infections caused by multidrug-resistant Gram-negative pathogens. Future Microbiol. 2014, 9, 1053–1069. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Pogue, J.; Bonomo, R.A.; Kaye, K.S. Ceftazidime/Avibactam, Meropenem/Vaborbactam, or Both? Clinical and Formulary Considerations. Clin. Infect. Dis. 2019, 68, 519–524. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. Interplay between β-lactamases and new β-lactamase inhibitors. Nat. Rev. Microbiol. 2019, 17, 295–306. [Google Scholar] [CrossRef]
- Thaden, J.T.; Pogue, J.M.; Kaye, K.S. Role of newer and re-emerging older agents in the treatment of infections caused by car-bapenem-resistant Enterobacteriaceae. Virulence 2017, 8, 403–416. [Google Scholar] [CrossRef]
- Shields, R.K.; Chen, L.; Cheng, S.; Chavda, K.D.; Press, E.G.; Snyder, A.; Pandey, R.; Doi, Y.; Kreiswirth, B.N.; Nguyen, M.H.; et al. Emergence of Ceftazidime-Avibactam Resistance Due to Plasmid-Borne bla KPC-3 Mutations during Treatment of Carbapenem-Resistant Klebsiella pneumoniae Infections. Antimicrob. Agents Chemother. 2016, 61, e02097-16. [Google Scholar] [CrossRef] [PubMed]
- David, S.; Wong, J.L.C.; Sanchez-Garrido, J.; Kwong, H.-S.; Low, W.W.; Morecchiato, F.; Giani, T.; Rossolini, G.M.; Brett, S.J.; Clements, A.; et al. Widespread emergence of OmpK36 loop 3 insertions among multidrug-resistant clones of Klebsiella pneumoniae. PLoS Pathog. 2022, 18, e1010334. [Google Scholar] [CrossRef]
- Sun, D.; Rubio-Aparicio, D.; Nelson, K.; Dudley, M.N.; Lomovskaya, O. Meropenem-Vaborbactam Resistance Selection, Resistance Prevention, and Molecular Mechanisms in Mutants of KPC-Producing Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2017, 61, e01694-17. [Google Scholar] [CrossRef] [PubMed]
- Gaibani, P.; Giani, T.; Bovo, F.; Lombardo, D.; Amadesi, S.; Lazzarotto, T.; Coppi, M.; Rossolini, G.M.; Ambretti, S. Resistance to Ceftazidime/Avibactam, Meropenem/Vaborbactam and Imipenem/Relebactam in Gram-Negative MDR Bacilli: Molecular Mechanisms and Susceptibility Testing. Antibiotics 2022, 11, 628. [Google Scholar] [CrossRef]
- Schechner, V.; Kotlovsky, T.; Kazma, M.; Mishali, H.; Schwartz, D.; Navon-Venezia, S.; Schwaber, M.; Carmeli, Y. Asymptomatic rectal carriage of blaKPC producing carbapenem-resistant Enterobacteriaceae: Who is prone to become clinically infected? Clin. Microbiol. Infect. 2013, 19, 451–456. [Google Scholar] [CrossRef]
- Tinelli, M.; Rossini, A.; Scudeller, L.; Zabzuni, D.; Errico, G.; Fogato, E.; D’Angelo, R.; Silverj, F.G.; Cesana, E.; Bergamaschini, L.C.; et al. Dynamics of carbapenemase-producing Enterobacterales intestinal colonisation in the elderly population after hospital discharge, Italy, 2018–2020. Int. J. Antimicrob. Agents 2022, 59, 106594. [Google Scholar] [CrossRef]
- Lim, C.J.; Cheng, A.C.; Kennon, J.; Spelman, D.; Hale, D.; Melican, G.; Sidjabat, H.E.; Paterson, D.L.; Kong, D.C.M.; Peleg, A.Y. Prevalence of multidrug-resistant organisms and risk factors for carriage in long-term care facilities: A nested case-control study. J. Antimicrob. Chemother. 2014, 69, 1972–1980. [Google Scholar] [CrossRef]
- Tinelli, M.; Tiseo, G.; Falcone, M. ESCMID Study Group for Infections in the Elderly. Prevention of the spread of multi-drug-resistant organisms in nursing homes. Aging Clin. Exp. Res. 2021, 33, 679–687. [Google Scholar] [CrossRef]
- WHO Regional Office for Europe/European Centre for Disease Prevention and Control. Antimicrobial Resistance Surveillance in Europe 2022–2020 Data; WHO Regional Office for Europe: Copenhagen, Denmark, 2022. [Google Scholar]
- Roe, C.C.; Vazquez, A.J.; Esposito, E.P.; Zarrilli, R.; Sahl, J.W. Diversity, Virulence, and Antimicrobial Resistance in Isolates From the Newly Emerging Klebsiella pneumoniae ST101 Lineage. Front Microbiol. 2019, 10, 542. [Google Scholar] [CrossRef] [PubMed]
- Oteo, J.; Pérez-Vázquez, M.; Bautista, V.; Ortega, A.; Zamarrón, P.; Saez, D.; Fernández-Romero, S.; Lara, N.; Ramiro, R.; Aracil, B.; et al. The spread of KPC-producing Enterobacteriaceae in Spain: WGS analysis of the emerging high-risk clones of Klebsiella pneumoniae ST11/KPC-2, ST101/KPC-2 and ST512/KPC-3. J. Antimicrob. Chemother. 2016, 71, 3392–3399. [Google Scholar] [CrossRef]
- Kiratisin, P.; Kazmierczak, K.; Stone, G.G. In vitro activity of ceftazidime/avibactam and comparators against carbapenemase-producing Enterobacterales and Pseudomonas aeruginosa isolates collected globally between 2016 and 2018. J. Glob. Antimicrob. Resist. 2021, 27, 132–141. [Google Scholar] [CrossRef]
- Winkler, M.L.; Papp-Wallace, K.M.; Bonomo, R.A. Activity of ceftazidime/avibactam against isogenic strains of Escherichia coli containing KPC and SHV b-lactamases with single amino acid substitutions in the X-loop. J. Antimicrob. Chemother. 2015, 70, 2279–2286. [Google Scholar] [CrossRef]
- Moreira, N.K.; Caierão, J. Ceftazidime-avibactam: Are we safe from class A carbapenemase producers’ infections? Folia Microbiol. 2021, 66, 879–896. [Google Scholar] [CrossRef] [PubMed]
- Nicola, F.; Cejas, D.; González-Espinosa, F.; Relloso, S.; Herrera, F.; Bonvehí, P.; Smayevsky, J.; Figueroa-Espinosa, R.; Gutkind, G.; Radice, M. Outbreak of Klebsiella pneumoniae ST11 Resistant to Ceftazidime-Avibactam Producing KPC-31 and the Novel Variant KPC-115 during COVID-19 Pandemic in Argentina. Microbiol. Spectr. 2022, 10, e0373322. [Google Scholar] [CrossRef] [PubMed]
- Hernández-García, M.; Castillo-Polo, J.A.; Cordero, D.G.; Pérez-Viso, B.; García-Castillo, M.; de la Fuente, J.S.; Morosini, M.I.; Cantón, R.; Ruiz-Garbajosa, P. Impact of Ceftazidime-Avibactam Treatment in the Emergence of Novel KPC Variants in the ST307-Klebsiella pneumoniae High-Risk Clone and Consequences for Their Routine Detection. J. Clin. Microbiol. 2022, 60, e0224521. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Ding, B.; Ye, M.; Wang, P.; Bi, Y.; Wu, S.; Xu, X.; Guo, Q.; Wang, M. High ceftazidime hydrolysis activity and porin OmpK35 deficiency contribute to the decreased susceptibility to ceftazidime/avibactam in KPC-producing Klebsiella pneu-moniae. J. Antimicrob. Chemother. 2017, 72, 1930–1936. [Google Scholar] [CrossRef]
- Castanheira, M.; Doyle, T.B.; Hubler, C.; Sader, H.S.; Mendes, R.E. Ceftazidime-avibactam activity against a challenge set of carbapenem-resistant Enterobacterales: Ompk36 L3 alterations and β-lactamases with ceftazidime hydrolytic activity lead to elevated MIC values. Int. J. Antimicrob. Agents 2020, 56, 106011. [Google Scholar] [CrossRef] [PubMed]
- Findlay, J.; Poirel, L.; Juhas, M.; Nordmann, P. KPC-Mediated Resistance to Ceftazidime-Avibactam and Collateral Effects in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2021, 65, e0089021. [Google Scholar] [CrossRef]
- Di Pilato, V.; Principe, L.; Andriani, L.; Aiezza, N.; Coppi, M.; Ricci, S.; Giani, T.; Luzzaro, F.; Rossolini, G.M. Deciphering variable resistance to novel carbapenem-based β-lactamase inhibitor combinations in a multi-clonal outbreak caused by Klebsiella pneumoniae carbapenemase (KPC)-producing Klebsiella pneumoniae resistant to ceftazidime/avibactam. Clin. Microbiol. Infect. 2023, 29, 537.e1–537.e8. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of MICs and Zone Diameters. Version 13.0. 2023. Available online: http://www.eucast.org (accessed on 18 February 2023).
- Kaas, R.S.; Leekitcharoenphon, P.; Aarestrup, F.M.; Lund, O. Solving the Problem of Comparing Whole Bacterial Genomes across Different Sequencing Platforms. PLoS ONE 2014, 9, e104984. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows—Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
| Porins | MIC (mg/L) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient | Isolate | ST | Carba | OmpK35 * | OmpK36 | CAZ-AVI | MEM | MVB | IMI | IMR | |||||
| P1 | RM2.05.T0 | 512 | KPC-3 | AA89 stop | GD ins | 16/4 | R | >64 | R | 1/8 | S | >8 | R | 0.5/4 | S |
| P2 | RM2.10.T0 | 101 | KPC-31 | AA63 stop | TD ins | >64/4 | R | 2 | S | 0.25/8 | S | ≤1 | S | 0.12/4 | S |
| P3 | RM2.15.T0 | 512 | KPC-31 | AA89 stop | GD ins | 64/4 | R | 2 | S | 1/8 | S | ≤1 | S | 0.12/4 | S |
| RM2.15.T4 | 512 | KPC-3 | AA89 stop | GD ins | 8/4 | S | >64 | R | 0.25/8 | S | >8 | R | 0.25/4 | S | |
| P4 | RM2.21.T0 | 512 | KPC-3 | AA89 stop | GD ins | 16/4 | R | >64 | R | 8/8 | S | >8 | R | 0.25/4 | S |
| RM2.21.T4 | 512 | KPC-3 | AA89 stop | GD ins | 4/4 | S | 32 | R | ≤0.06/8 | S | 4 | I | 0.12/4 | S | |
| RM2.21.T8 | 512 | KPC-3 | AA89 stop | GD ins | 2/4 | S | >64 | R | 0.5/8 | S | >8 | R | 0.12/4 | S | |
| P5 | RM2.31.T0 | 101 | KPC-46 | AA63 stop | TD ins | 32/4 | R | 1 | S | 0.25/8 | S | ≤1 | S | 0.12/4 | S |
| RM2.31.T4 | 101 | KPC-46 | AA63 stop | TD ins | 64/4 | R | 1 | S | 0.25/8 | S | ≤1 | S | 0.12/4 | S | |
| P6 | RM2.71.T0 | 101 | KPC-46 | AA63 stop | TD ins | 32/4 | R | 1 | S | 0.25/8 | S | ≤1 | S | 0.12/4 | S |
| Patient | Isolate | ST | β-Lactamases | Aminoglycoside Modifying Enzyme | Fluoroquinolones | MLS | Phenicols | RIF | SUL | TET | TMT |
|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | RM2.05.T0 | 512 | KPC-3, TEM-1D, SHV-11 | aac(6′)-Ib, aadA2, aph(3′)-Ia | GyrA-S83I, ParC-S80I | mphA | catA1 | - | sul1 | - | dfrA12 |
| P2 | RM2.10.T0 | 101 | KPC-31, SHV-1 | armA | GyrA-S83Y/D87N ParC-S80I | mphE, msrE | - | - | - | - | - |
| P3 | RM2.15.T0 | 512 | KPC-31, SHV-11 | aac(6′)-Ib’,aadA2, aph(3′)-Ia | GyrA-S83I, ParC-S80I | mphA | catA1 | - | sul1 | - | dfrA12 |
| RM2.15.T4 | 512 | KPC-3, SHV-11 | aac(6′)-Ib’ | GyrA-S83I, ParC-S80I | - | - | - | - | - | - | |
| P4 | RM2.21.T0 | 512 | KPC-3, CMY-16, OXA-10, SHV11 | aadA1, aadA2, aac(6′)-Ib3, strA | GyrA-S83I, ParC-S80I | mphA | catA1, cmlA5, floR | arr2 | sul1, sul2 | tetA | dfrA12, dfrA14 |
| RM2.21.T4 | 512 | KPC-3, CMY-16, OXA-10, SHV-11, TEM-1D | aadA1, aadA2, aac(6′)-Ib3,strA | GyrA-83I, ParC-80I | - | cmlA5, floR | arr2 | sul1, sul2 | tetA | dfrA14 | |
| RM2.21.T8 | 512 | KPC-3, CMY-16, OXA-10, SHV-11, TEM-1D | aadA1, aadA2, aac(6′)-Ib3, strA | GyrA-83I, ParC-80I | - | cmlA5, floR | arr2 | sul1, sul2 | tetA | dfrA14 | |
| P5 | RM2.31.T0 | 101 | KPC-46, SHV-1 | armA | GyrA-S83Y/D87N ParC-S80I | mphE, msrE | - | - | - | - | - |
| RM2.31.T4 | 101 | KPC-46, SHV-1 | armA | GyrA-S83Y/D87N ParC-S80I | mphE, msrE | - | - | - | - | - | |
| P6 | RM2.71.T0 | 101 | KPC-46, SHV-1 | - | GyrA-S83Y/D87NParC-S80I | mphE, msrE | - | - | - | - | - |
| Patient | Isolate | ST | KLocus | Virulence Factor | Plasmid | pMLST |
|---|---|---|---|---|---|---|
| P1 | RM2.05.T0 | 512 | KL107 (wzi-154) | - | IncFIB, IncFII, IncX3,ColRNAI | IncF [K1:A-:B-] |
| P2 | RM2.10.T0 | 101 | KL17 (wzi-137) | ybt9/ ICEKp3 | IncFIA, IncFIB, IncFII, IncR,Col440,ColRNAI | IncF [K1:A13:B-] |
| P3 | RM2.15.T0 | 512 | KL107 (wzi-154) | - | IncFIB, IncFII, IncX3 | IncF [K2:A-:B-] |
| RM2.15.T4 | 512 | KL107 (wzi-154) | - | IncFIB, IncFII, IncX3, ColRNAI | IncF [K2:A-:B-] | |
| P4 | RM2.21.T0 | 512 | KL107 (wzi-154) | - | IncFIB, IncFII, IncC IncX3 | IncF [K2:A-:B-] IncC [C3] |
| RM2.21.T4 | 512 | KL107 (wzi-154) | - | IncFIB, IncFII, IncC IncX3 | IncF [K2:A-:B-] IncC [C3] | |
| RM2.21.T8 | 512 | KL107 (wzi-154) | - | IncFIB, IncFII, IncC IncX3 | IncF [K2:A-:B-] IncC [C3] | |
| P5 | RM2.31.T0 | 101 | KL17 (wzi-137) | ybt9/ ICEKp3 | IncFIA, IncFIB, IncFII,IncR,Col440,ColRNAI | IncF [K2:A13:B-] |
| RM2.31.T4 | 101 | KL17 (wzi-137) | ybt9/ ICEKp3 | IncFIA, IncFIB, IncFII,IncR,Col440,ColRNAI | IncF [K2:A13:B-] | |
| P6 | RM2.71.T0 | 101 | KL17 (wzi-137) | ybt9/ ICEKp3 | IncFIA, IncFIB, IncFII,IncR,Col440,ColRNAI | IncF [K2:A13:B-] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Errico, G.; Del Grosso, M.; Pagnotta, M.; Marra, M.; Carollo, M.; Cerquetti, M.; Fogato, E.; Cesana, E.; Gentiloni Silverj, F.; Zabzuni, D.; et al. Whole-Genome Sequencing and Molecular Analysis of Ceftazidime–Avibactam-Resistant KPC-Producing Klebsiella pneumoniae from Intestinal Colonization in Elderly Patients. Antibiotics 2023, 12, 1282. https://doi.org/10.3390/antibiotics12081282
Errico G, Del Grosso M, Pagnotta M, Marra M, Carollo M, Cerquetti M, Fogato E, Cesana E, Gentiloni Silverj F, Zabzuni D, et al. Whole-Genome Sequencing and Molecular Analysis of Ceftazidime–Avibactam-Resistant KPC-Producing Klebsiella pneumoniae from Intestinal Colonization in Elderly Patients. Antibiotics. 2023; 12(8):1282. https://doi.org/10.3390/antibiotics12081282
Chicago/Turabian StyleErrico, Giulia, Maria Del Grosso, Michela Pagnotta, Manuela Marra, Maria Carollo, Marina Cerquetti, Elena Fogato, Elisabetta Cesana, Flaminia Gentiloni Silverj, Dorjan Zabzuni, and et al. 2023. "Whole-Genome Sequencing and Molecular Analysis of Ceftazidime–Avibactam-Resistant KPC-Producing Klebsiella pneumoniae from Intestinal Colonization in Elderly Patients" Antibiotics 12, no. 8: 1282. https://doi.org/10.3390/antibiotics12081282
APA StyleErrico, G., Del Grosso, M., Pagnotta, M., Marra, M., Carollo, M., Cerquetti, M., Fogato, E., Cesana, E., Gentiloni Silverj, F., Zabzuni, D., Rossini, A., Pantosti, A., Tinelli, M., Monaco, M., & Giufrè, M. (2023). Whole-Genome Sequencing and Molecular Analysis of Ceftazidime–Avibactam-Resistant KPC-Producing Klebsiella pneumoniae from Intestinal Colonization in Elderly Patients. Antibiotics, 12(8), 1282. https://doi.org/10.3390/antibiotics12081282







