Genomic Characterization of Aeromonas veronii Provides Insights into Taxonomic Assignment and Reveals Widespread Virulence and Resistance Genes throughout the World
Abstract
1. Introduction
2. Results
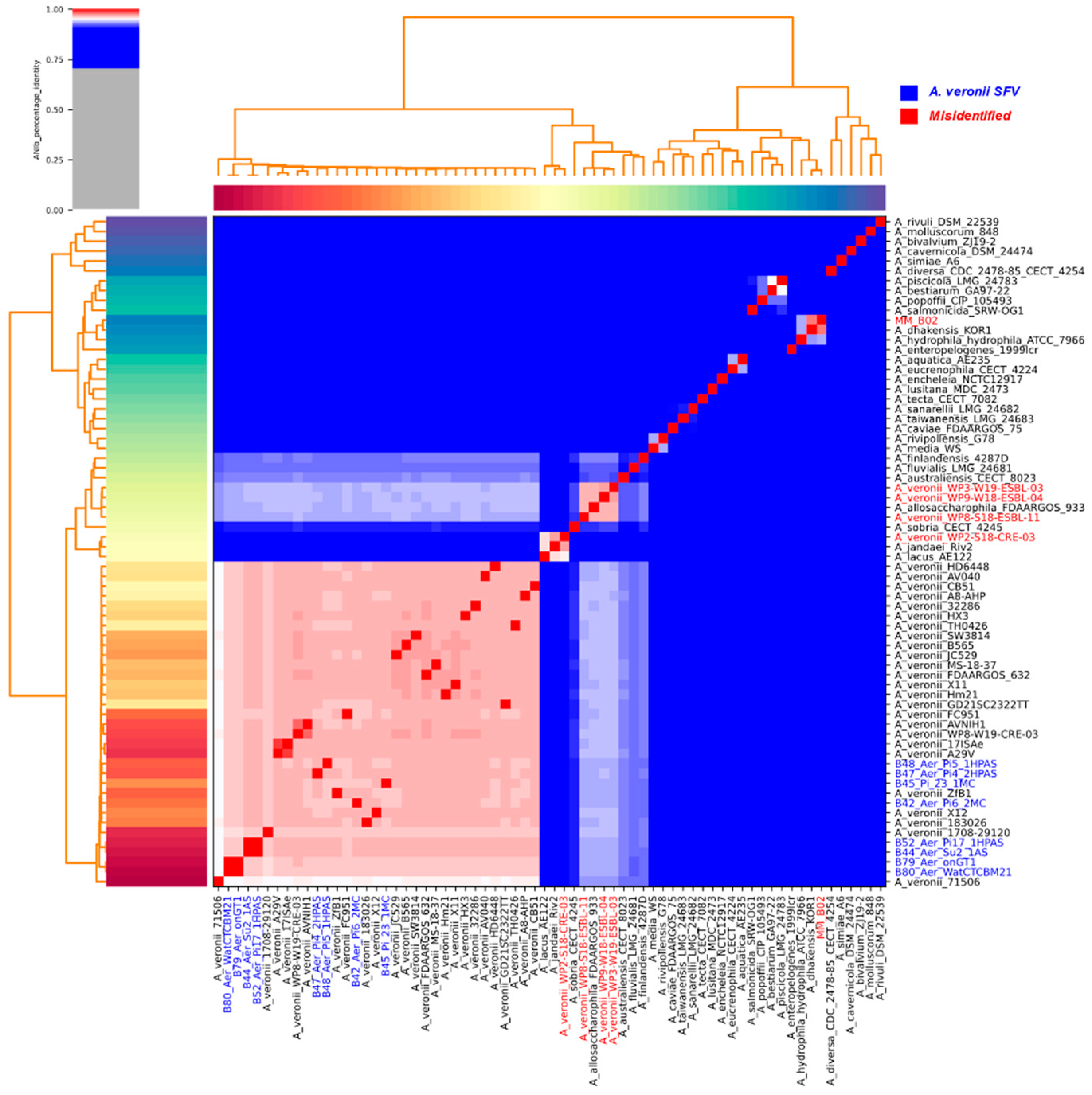
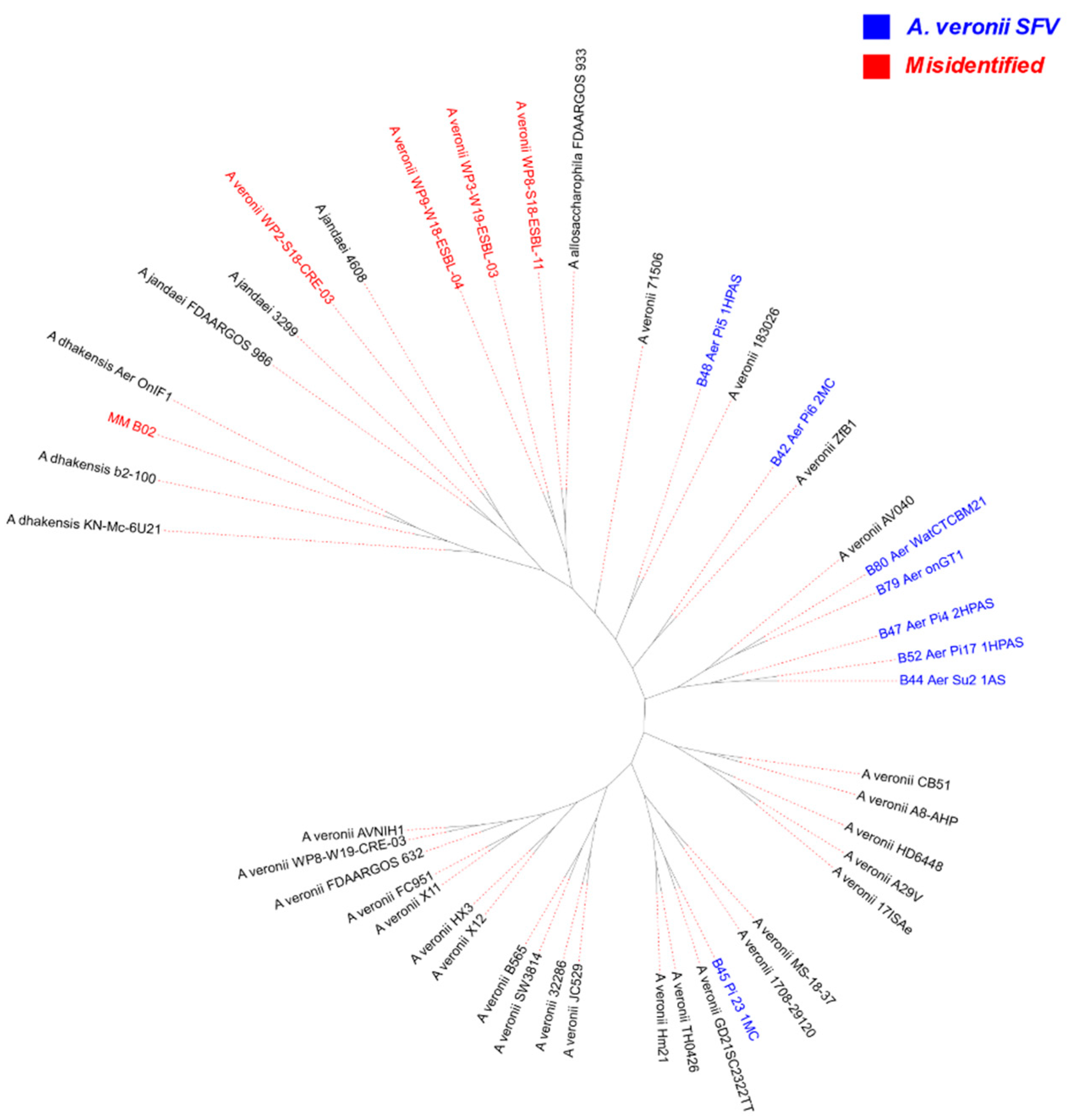
2.1. MLST, TETRA, ANI, and Phylogeny
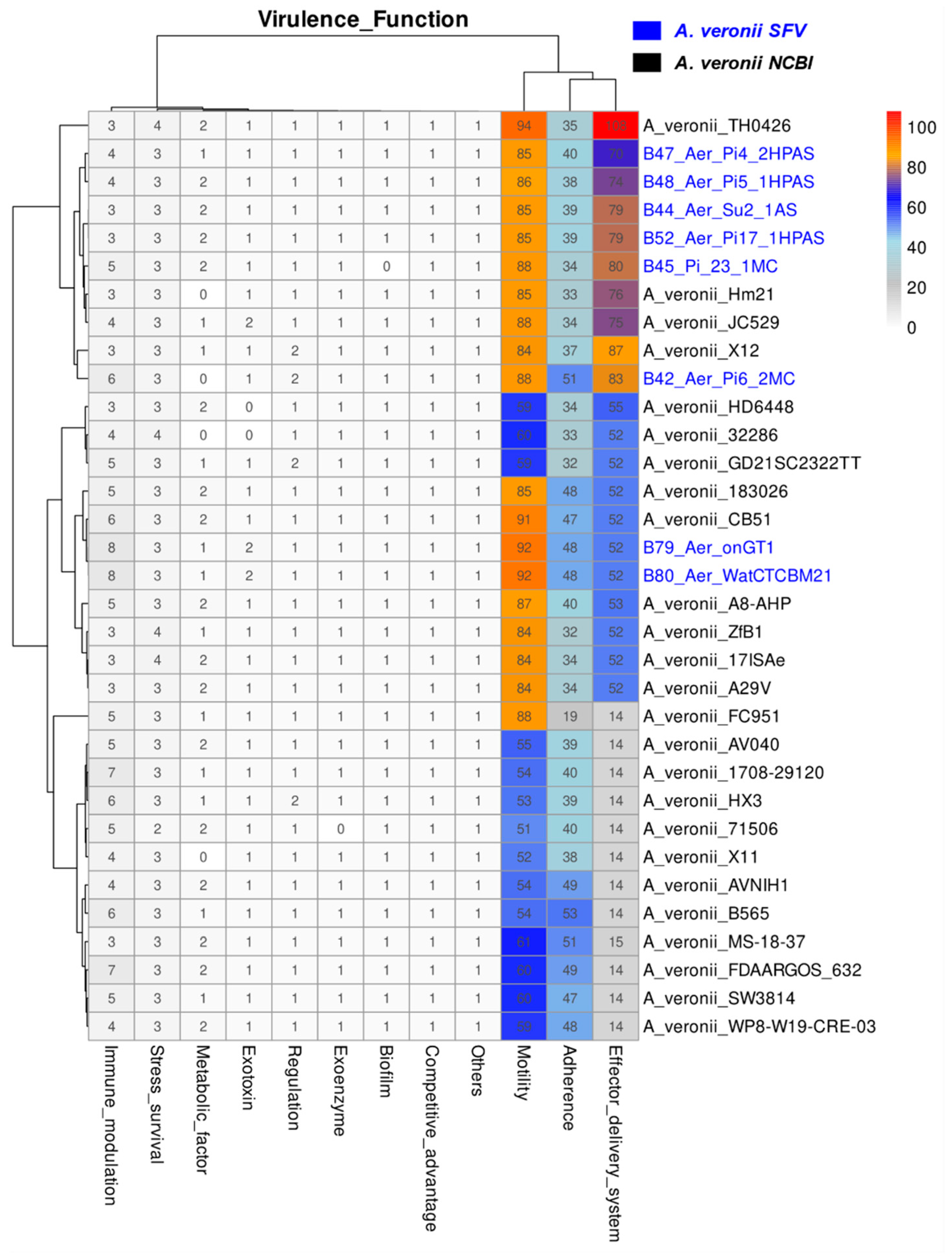
2.2. Virulence
2.3. Resistance
3. Discussion
4. Materials and Methods
4.1. Genomes from the São Francisco Valley (SFV)
4.2. NCBI Genomes
4.3. São Francisco Valley Genome Assembly
4.4. Plasmids
4.5. Annotation
4.6. Typing and Genomes Classification
4.7. Phylogeny
4.8. Virulence and Resistance Genes Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martin-Carnahan, A.; Joseph, S.W. Aeromonas Stanier 1943, 213. In Bergey’s Manual of Systematic Bacteriology; Brenner, D.J., Krieg, N.R., Staley, J.T., Garrity, G.M., Eds.; John Wiley & Sons, Inc.: Philadelphia, PA, USA, 2015; pp. 1–44. ISBN 9781118960608. [Google Scholar]
- Fernández-Bravo, A.; Figueras, M.J. An Update on the Genus Aeromonas: Taxonomy, Epidemiology, and Pathogenicity. Microorganisms 2020, 8, 129. [Google Scholar] [CrossRef]
- Dong, H.T.; Techatanakitarnan, C.; Jindakittikul, P.; Thaiprayoon, A.; Taengphu, S.; Charoensapsri, W.; Khunrae, P.; Rattanarojpong, T.; Senapin, S. Aeromonas jandaei and Aeromonas veronii Caused Disease and Mortality in Nile Tilapia, Oreochromis niloticus (L.). J. Fish Dis. 2017, 40, 1395–1403. [Google Scholar] [CrossRef] [PubMed]
- Ran, C.; Qin, C.; Xie, M.; Zhang, J.; Li, J.; Xie, Y.; Wang, Y.; Li, S.; Liu, L.; Fu, X.; et al. Aeromonas veronii and Aerolysin are Important for the Pathogenesis of Motile Aeromonad Septicemia in Cyprinid Fish. Environ. Microbiol. 2018, 20, 3442–3456. [Google Scholar] [CrossRef]
- Yuwono, C.; Wehrhahn, M.C.; Liu, F.; Riordan, S.M.; Zhang, L. The Isolation of Aeromonas Species and Other Common Enteric Bacterial Pathogens from Patients with Gastroenteritis in an Australian Population. Microorganisms 2021, 9, 1440. [Google Scholar] [CrossRef]
- Liu, F.; Yuwono, C.; Tay, A.C.Y.; Wehrhahn, M.C.; Riordan, S.M.; Zhang, L. Analysis of Global Aeromonas veronii Genomes Provides Novel Information on Source of Infection and Virulence in Human Gastrointestinal Diseases. BMC Genom. 2022, 23, 166. [Google Scholar] [CrossRef]
- Prediger, K.D.C.; Dallagassa, C.B.; Moriel, B.; Vizzotto, B.S.; Volanski, W.; Souza, E.M.; Pedrosa, F.O.; Weiss, V.; Alberton, D.; Guizelini, D.; et al. Virulence Characteristics and Antimicrobial Resistance of Aeromonas veronii Biovar Sobria 312M, a Clinical Isolate. Braz. J. Microbiol. 2020, 51, 511–518. [Google Scholar] [CrossRef]
- Tekedar, H.C.; Kumru, S.; Blom, J.; Perkins, A.D.; Griffin, M.J.; Abdelhamed, H.; Karsi, A.; Lawrence, M.L. Comparative Genomics of Aeromonas veronii: Identification of a Pathotype Impacting Aquaculture Globally. PLoS ONE 2019, 14, e0221018. [Google Scholar] [CrossRef]
- Yang, B.; Chen, C.; Sun, Y.; Cao, L.; Zhang, D.; Sun, W.; Zhang, L.; Wang, G.; Shan, X.; Kang, Y.; et al. Comparative Genomic Analysis of Different Virulence Strains Reveals Reasons for the Increased Virulence of Aeromonas veronii. J. Fish Dis. 2021, 44, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Sheng, T.; Song, G.; Yue, T.; Zhang, J.; Wang, W.; Yang, Z.; Lu, Q. Whole-Genome Sequencing and Antimicrobial Resistance Analysis of Multidrug-Resistant Aeromonas veronii strain JC529 from a Common Carp. J. Glob. Antimicrob. Resist. 2021, 27, 118–122. [Google Scholar] [CrossRef]
- Yang, Q.; Zhao, M.; Wang, K.Y.; Wang, J.; He, Y.; Wang, E.L.; Liu, T.; Chen, D.F.; Lai, W. Multidrug-Resistant Aeromonas veronii Recovered from Channel Catfish (Ictalurus punctatus) in China: Prevalence and Mechanisms of Fluoroquinolone Resistance. Microb. Drug Resist. 2017, 23, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Bello-López, J.M.; Cabrero-Martínez, O.A.; Ibáñez-Cervantes, G.; Hernández-Cortez, C.; Pelcastre-Rodríguez, L.I.; Gonzalez-Avila, L.U.; Castro-Escarpulli, G. Horizontal Gene Transfer and Its Association with Antibiotic Resistance in the Genus Aeromonas spp. Microorganisms 2019, 7, 363. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Li, H.; Shen, Y.; Liu, Z.; Wang, S.; Shen, Z.; Zhang, R.; Walsh, T.R.; Shen, J.; Wang, Y. Beijing Novel Plasmid-Mediated Colistin Resistance Gene mcr-3 in Escherichia coli Wenjuan. MBio 2017, 8, e00543-17. [Google Scholar] [PubMed]
- Yang, Y.Q.; Li, Y.X.; Lei, C.W.; Zhang, A.Y.; Wang, H.N. Novel Plasmid-Mediated Colistin Resistance Gene mcr-7.1 in Klebsiella pneumoniae. J. Antimicrob. Chemother. 2018, 73, 1791–1795. [Google Scholar] [CrossRef] [PubMed]
- Cherak, Z.; Loucif, L.; Moussi, A.; Rolain, J.M. Epidemiology of Mobile Colistin Resistance (mcr) Genes in Aquatic Environments. J. Glob. Antimicrob. Resist. 2021, 27, 51–62. [Google Scholar] [CrossRef]
- Pessoa, R.B.G.; de Oliveira, W.F.; dos Santos Correia, M.T.; Fontes, A.; Coelho, L.C.B.B. Aeromonas and Human Health Disorders: Clinical Approaches. Front. Microbiol. 2022, 13, 1266. [Google Scholar] [CrossRef]
- Beaz-Hidalgo, R.; Hossain, M.J.; Liles, M.R.; Figueras, M.J. Strategies to Avoid Wrongly Labelled Genomes Using as Example the Detected Wrong Taxonomic Affiliation for Aeromonas Genomes in the Genbank Database. PLoS ONE 2015, 10, e0115813. [Google Scholar] [CrossRef]
- Awan, F.; Dong, Y.; Liu, J.; Wang, N.; Mushtaq, M.H.; Lu, C.; Liu, Y. Comparative Genome Analysis Provides Deep Insights into Aeromonas hydrophila Taxonomy and Virulence-Related Factors. BMC Genomics 2018, 19, 712. [Google Scholar] [CrossRef]
- Lamy, B.; Laurent, F.; Verdier, I.; Decousser, J.W.; Lecaillon, E.; Marchandin, H.; Roger, F.; Tigaud, S.; de Montclos, H.; Kodjo, A. Accuracy of 6 Commercial Systems for Identifying Clinical Aeromonas Isolates. Diagn. Microbiol. Infect. Dis. 2010, 67, 9–14. [Google Scholar] [CrossRef]
- Ceric, O.; Tyson, G.H.; Goodman, L.B.; Mitchell, P.K.; Zhang, Y.; Prarat, M.; Cui, J.; Peak, L.; Scaria, J.; Antony, L.; et al. Enhancing the One Health Initiative by Using Whole Genome Sequencing to Monitor Antimicrobial Resistance of Animal Pathogens: Vet-LIRN Collaborative Project with Veterinary Diagnostic Laboratories in United States and Canada. BMC Vet. Res. 2019, 15, 130. [Google Scholar] [CrossRef]
- Richter, M.; Rosselló-Móra, R. Shifting the Genomic Gold Standard for the Prokaryotic Species Definition. Proc. Natl. Acad. Sci. USA 2009, 106, 19126–19131. [Google Scholar] [CrossRef]
- Colston, S.M.; Fullmer, M.S.; Beka, L.; Lamy, B.; Gogarten, J.P. Bioinformatic Genome Comparisons for Taxonomic and Phylogenetic Assignments Using Aeromonas as a Test Case. MBio 2014, 5, e02136-14. [Google Scholar] [CrossRef] [PubMed]
- Beaz-Hidalgo, R.; Latif-Eugenín, F.; Hossain, M.J.; Berg, K.; Niemi, R.M.; Rapala, J.; Lyra, C.; Liles, M.R.; Figueras, M.J. Aeromonas aquatica sp. nov., Aeromonas finlandiensis sp. nov. and Aeromonas lacus sp. nov. Isolated from Finnish Waters Associated with Cyanobacterial Blooms. Syst. Appl. Microbiol. 2015, 38, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Hadi, N.; Yang, Q.; Barnett, T.C.; Tabei, S.M.B.; Kirov, S.M.; Shaw, J.G. Bundle-Forming Pilus Locus of Aeromonas veronii bv. sobria. Infect. Immun. 2012, 80, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- Werneburg, G.T.; Thanassi, D.G. Pili Assembled by the Chaperone/Usher Pathway in Escherichia coli and Salmonella. EcoSal Plus 2018, 8, 631–632. [Google Scholar] [CrossRef]
- Dacanay, A.; Boyd, J.M.; Fast, M.D.; Knickle, L.C.; Reith, M.E. Aeromonas salmonicida Type I Pilus System Contributes to Host Colonization but Not Invasion. Dis. Aquat. Organ. 2010, 88, 199–206. [Google Scholar] [CrossRef]
- Kirov, S.M.; Tassell, B.C.; Semmler, A.B.T.; O’Donovan, L.A.; Rabaan, A.A.; Shaw, J.G. Lateral Flagella and Swarming Motility in Aeromonas Species. J. Bacteriol. 2002, 184, 547–555. [Google Scholar] [CrossRef]
- Gavín, R.; Merino, S.; Altarriba, M.; Canals, R.; Shaw, J.G.; Tomás, J.M. Lateral Flagella Are Required for Increased Cell Adherence, Invasion and Biofilm Formation by Aeromonas spp. FEMS Microbiol. Lett. 2003, 224, 77–83. [Google Scholar] [CrossRef]
- Green, E.R.; Mecsas, J. Bacterial Secretion Systems: An Overview. Microbiol. Spectr. 2016, 4, 213–239. [Google Scholar] [CrossRef]
- Mekasha, S.; Linke, D. Secretion Systems in Gram-Negative Bacterial Fish Pathogens. Front. Microbiol. 2021, 12, 782673. [Google Scholar] [CrossRef]
- Naskar, S.; Hohl, M.; Tassinari, M.; Low, H.H. The Structure and Mechanism of the Bacterial Type II Secretion System. Mol. Microbiol. 2021, 115, 412–424. [Google Scholar] [CrossRef]
- Korotkov, K.V.; Sandkvist, M. Architecture, Function, and Substrates of the Type II Secretion System. EcoSal Plus. 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, T.; Schmid, A.; Notermans, S.; Benz, R. Aerolysin of Aeromonas sobria: Evidence for Formation of Ion-Permeable Channels and Comparison with Alpha-Toxin of Staphylococcus aureus. Infect. Immun. 1990, 58, 2127–2132. [Google Scholar] [CrossRef] [PubMed]
- Hews, C.L.; Tran, S.L.; Wegmann, U.; Brett, B.; Walsham, A.D.S.; Kavanaugh, D.; Ward, N.J.; Juge, N.; Schüller, S. The StcE Metalloprotease of Enterohaemorrhagic Escherichia coli Reduces the Inner Mucus Layer and Promotes Adherence to Human Colonic Epithelium ex vivo. Cell. Microbiol. 2017, 19, e12717. [Google Scholar] [CrossRef]
- Dey, S.; Chakravarty, A.; Guha Biswas, P.; De Guzman, R.N. The Type III Secretion System Needle, Tip, and Translocon. Protein Sci. 2019, 28, 1582–1593. [Google Scholar] [CrossRef] [PubMed]
- Pinaud, L.; Sansonetti, P.J.; Phalipon, A. Host Cell Targeting by Enteropathogenic Bacteria T3SS Effectors. Trends Microbiol. 2018, 26, 266–283. [Google Scholar] [CrossRef] [PubMed]
- Silver, A.C.; Graf, J. Prevalence of Genes Encoding the Type Three Secretion System and the Effectors AexT and AexU in the Aeromonas veronii Group. DNA Cell Biol. 2009, 28, 383–388. [Google Scholar] [CrossRef]
- Yu, K.W.; Xue, P.; Fu, Y.; Yang, L. T6SS Mediated Stress Responses for Bacterial Environmental Survival and Host Adaptation. Int. J. Mol. Sci. 2021, 22, 478. [Google Scholar] [CrossRef]
- Si, M.; Wang, Y.; Zhang, B.; Zhao, C.; Kang, Y.; Bai, H.; Wei, D.; Zhu, L.; Zhang, L.; Dong, T.G.; et al. The Type VI Secretion System Engages a Redox-Regulated Dual-Functional Heme Transporter for Zinc Acquisition. Cell Rep. 2017, 20, 949–959. [Google Scholar] [CrossRef]
- Schauder, S.; Shokat, K.; Surette, M.G.; Bassler, B.L. The LuxS Family of Bacterial Autoinducers: Biosynthesis of a Novel Quorum-Sensing Signal Molecule. Mol. Microbiol. 2001, 41, 463–476. [Google Scholar] [CrossRef]
- Liu, L.; Wu, R.; Zhang, J.; Li, P. Overexpression of LuxS Promotes Stress Resistance and Biofilm Formation of Lactobacillus paraplantarumL-ZS9 by Regulating the Expression of Multiple Genes. Front. Microbiol. 2018, 9, 2628. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, B.; Grenier, D.; Yi, L. Regulatory Mechanisms of the LuxS/AI-2 System and Bacterial Resistance. Am. Soc. Microbiol. 2019, 63, e01186-19. [Google Scholar] [CrossRef]
- Ma, Y.; Hao, L.; Ke, H.; Liang, Z.; Ma, J.; Liu, Z.; Li, Y. LuxS/AI-2 in Streptococcus agalactiae Reveals a Key Role in Acid Tolerance and Virulence. Res. Vet. Sci. 2017, 115, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Castanheira, M.; Simner, P.J.; Bradford, P.A. Extended-Spectrum β-Lactamases: An Update on Their Characteristics, Epidemiology and Detection. JAC Antimicrob. Resist. 2021, 3, dlab092. [Google Scholar] [CrossRef]
- Bahr, G.; González, L.J.; Vila, A.J. Metallo-β-Lactamases in the Age of Multidrug Resistance: From Structure and Mechanism to Evolution, Dissemination, and Inhibitor Design. Chem. Rev. 2021, 121, 7957–8094. [Google Scholar] [CrossRef] [PubMed]
- Du, D.; Wang-Kan, X.; Neuberger, A.; van Veen, H.W.; Pos, K.M.; Piddock, L.J.V.; Luisi, B.F. Multidrug Efflux Pumps: Structure, Function and Regulation. Nat. Rev. Microbiol. 2018, 16, 523–539. [Google Scholar] [CrossRef] [PubMed]
- Blair, J.M.A.; Richmond, G.E.; Piddock, L.J.V. Multidrug Efflux Pumps in Gram-Negative Bacteria and Their Role in Antibiotic Resistance. Future Microbiol. 2014, 9, 1165–1177. [Google Scholar] [CrossRef] [PubMed]
- Cloeckaert, A.; Baucheron, S.; Chaslus-Dancla, E. Nonenzymatic Chloramphenicol Resistance Mediated by IncC Plasmid R55 Is Encoded by a floR Gene Variant. Antimicrob. Agents Chemother. 2001, 45, 2381–2382. [Google Scholar] [CrossRef]
- Shi, Y.; Shi, Y.; Tian, Z.; Tian, Z.; Gillings, M.R.; Zhang, Y.; Zhang, Y.; Zhang, H.; Huyan, J.; Huyan, J.; et al. Novel Transposon Tn 6433 Variants Accelerate the Dissemination of tet(E) in Aeromonas in an Aerobic Biofilm Reactor under Oxytetracycline Stresses. Environ. Sci. Technol. 2020, 54, 6781–6791. [Google Scholar] [CrossRef]
- McArthur, A.G.; Waglechner, N.; Nizam, F.; Yan, A.; Azad, M.A.; Baylay, A.J.; Bhullar, K.; Canova, M.J.; De Pascale, G.; Ejim, L.; et al. The Comprehensive Antibiotic Resistance Database. Antimicrob. Agents Chemother. 2013, 57, 3348–3357. [Google Scholar] [CrossRef]
- Von Wintersdorff, C.J.H.; Penders, J.; Van Niekerk, J.M.; Mills, N.D.; Majumder, S.; Van Alphen, L.B.; Savelkoul, P.H.M.; Wolffs, P.F.G. Dissemination of Antimicrobial Resistance in Microbial Ecosystems through Horizontal Gene Transfer. Front. Microbiol. 2016, 7, 173. [Google Scholar] [CrossRef]
- Shintani, M.; Sanchez, Z.K.; Kimbara, K. Genomics of Microbial Plasmids: Classification and Identification Based on Replication and Transfer Systems and Host Taxonomy. Front. Microbiol. 2015, 6, 242. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Han, M.; Yang, P.; Chen, C.; Yu, H.; Wang, L.; Ning, K. Comprehensive Analysis Reveals the Evolution and Pathogenicity of Aeromonas, Viewed from Both Single Isolated Species and Microbial Communities. mSystems 2019, 4, e00252-19. [Google Scholar] [CrossRef]
- Talagrand-Reboul, E.; Colston, S.M.; Graf, J.; Lamy, B.; Jumas-Bilak, E. Comparative and Evolutionary Genomics of Isolates Provide Insight into the Pathoadaptation of Aeromonas. Genome Biol. Evol. 2020, 12, 535–552. [Google Scholar] [CrossRef]
- Pessoa, R.B.G.; Marques, D.S.C.; Lima, R.O.H.A.; Oliveira, M.B.M.; Lima, G.M.S.; Maciel de Carvalho, E.V.M.; Coelho, L.C.B.B. Molecular Characterization and Evaluation of Virulence Traits of Aeromonas spp. Isolated from the Tambaqui Fish (Colossoma macropomum). Microb. Pathog. 2020, 147, 104273. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Raza, S.H.A.; Yang, B.; Sun, Y.; Wang, G.; Sun, W.; Qian, A.; Wang, C.; Kang, Y.; Shan, X. Aeromonas veronii Infection in Commercial Freshwater Fish: A Potential Threat to Public Health. Animals 2020, 10, 608. [Google Scholar] [CrossRef] [PubMed]
- Didugu, H.; Thirtham, M.; Nelapati, K.; Reddy, K.K.; Kumbhar, B.S.; Poluru, A.; Pothanaboyina, G. A Study on the Prevalence of Aeromonas spp. and Its Enterotoxin Genes in Samples of Well Water, Tap Water, and Bottled Water. Vet. World 2015, 8, 1237–1242. [Google Scholar] [CrossRef] [PubMed]
- Snyman, Y.; Whitelaw, A.C.; Barnes, J.M.; Maloba, M.R.B.; Newton-Foot, M. Characterisation of Mobile Colistin Resistance Genes (mcr-3 and mcr-5) in River and Storm Water in Regions of the Western Cape of South Africa. Antimicrob. Resist. Infect. Control 2021, 10, 1–9. [Google Scholar] [CrossRef]
- Shao, S.; Hu, Y.; Cheng, J.; Chen, Y. Research Progress on Distribution, Migration, Transformation of Antibiotics and Antibiotic Resistance Genes (ARGs) in Aquatic Environment. Crit. Rev. Biotechnol. 2018, 38, 1195–1208. [Google Scholar] [CrossRef]
- Gouveia, J.J.S.; Regitano, L.C.A. Extração de DNA. In Protocolos em Biologia Molecular Aplicada à Produção Animal; Regitano, L.C.A., Niciura, S.C.M., Ibelli, A.M.G., Gouveia, J.J., Eds.; Embrapa: São Carlos, Brazil; pp. 3–8. ISBN 9788586763127.
- Trivedi, U.H.; Cézard, T.; Bridgett, S.; Montazam, A.; Nichols, J.; Blaxter, M.; Gharbi, K. Quality Control of Next-Generation Sequencing Data without a Reference. Front. Genet. 2014, 5, 111. [Google Scholar] [CrossRef]
- Lindgreen, S. AdapterRemoval: Easy Cleaning of next-Generation Sequencing Reads. BMC Res. Notes 2012, 5, 337. [Google Scholar] [CrossRef]
- Melsted, P.; Halldórsson, B.V. KmerStream: Streaming Algorithms for k-Mer Abundance Estimation. Bioinformatics 2014, 30, 3541–3547. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A New Genome Assembly Algorithm and Its Applications to Single-Cell Sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, D.; François, P.; Farinelli, L.; Østerås, M.; Schrenzel, J. De Novo Bacterial Genome Sequencing: Millions of Very Short Reads Assembled on a Desktop Computer. Genome Res. 2008, 18, 802–809. [Google Scholar] [CrossRef]
- Wick, R.R.; Judd, L.M.; Gorrie, C.L.; Holt, K.E. Unicycler: Resolving Bacterial Genome Assemblies from Short and Long Sequencing Reads. PLoS Comput. Biol. 2017, 13, e1005595. [Google Scholar] [CrossRef]
- Li, W.; Godzik, A. Cd-Hit: A Fast Program for Clustering and Comparing Large Sets of Protein or Nucleotide Sequences. Bioinformatics 2006, 22, 1658–1659. [Google Scholar] [CrossRef] [PubMed]
- Gurevich, A.; Saveliev, V.; Vyahhi, N.; Tesler, G. QUAST: Quality Assessment Tool for Genome Assemblies. Bioinformatics 2013, 29, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.; Nash, J.H.E. MOB-Suite: Software Tools for Clustering, Reconstruction and Typing of Plasmids from Draft Assemblies. Microb. Genom. 2018, 4, e000206. [Google Scholar] [CrossRef]
- Seemann, T. Prokka: Rapid Prokaryotic Genome Annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Pritchard, L.; Glover, R.H.; Humphris, S.; Elphinstone, J.G.; Toth, I.K. Genomics and Taxonomy in Diagnostics for Food Security: Soft-Rotting Enterobacterial Plant Pathogens. Anal. Methods 2016, 8, 12–24. [Google Scholar] [CrossRef]
- Lee, I.; Kim, Y.O.; Park, S.C.; Chun, J. OrthoANI: An Improved Algorithm and Software for Calculating Average Nucleotide Identity. Int. J. Syst. Evol. Microbiol. 2016, 66, 1100–1103. [Google Scholar] [CrossRef]
- Page, A.J.; Cummins, C.A.; Hunt, M.; Wong, V.K.; Reuter, S.; Holden, M.T.G.; Fookes, M.; Falush, D.; Keane, J.A.; Parkhill, J. Roary: Rapid Large-Scale Prokaryote Pan Genome Analysis. Bioinformatics 2015, 31, 3691–3693. [Google Scholar] [CrossRef]
- Katoh, K.; Misawa, K.; Kuma, K.I.; Miyata, T. MAFFT: A Novel Method for Rapid Multiple Sequence Alignment Based on Fast Fourier Transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; Von Haeseler, A.; Lanfear, R.; Teeling, E. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, J.; Yu, J.; Yao, Z.; Sun, L.; Shen, Y.; Jin, Q. VFDB: A Reference Database for Bacterial Virulence Factors. Nucleic Acids Res. 2005, 33, 325–328. [Google Scholar] [CrossRef]
- Jaccard, P. Étude Comparative de La Distribution Florale Dans Une Portion Des Alpes et Du Jura. Bull. Soc. Vaud. Sci. Nat. 1901, 37, 547–579. [Google Scholar] [CrossRef]
- Gower, J.C.; Legendre, P. Metric and Euclidean Properties of Dissimilarity Coefficients. J. Classif. 1986, 3, 5–48. [Google Scholar] [CrossRef]
- Gower, J.C. Some Distance Properties of Latent Root and Vector Methods Used in Multivariate Analysis. Biometrika 1966, 53, 325. [Google Scholar] [CrossRef]

| ID | Isolates | Isolates | Host | Location | Patient | Plasmids |
|---|---|---|---|---|---|---|
| B42 | PRJNA590960 | B42_Aer_Pi6_2MC | Phractocephalus hemioliopterus | CODEVASF * | kidney | - |
| B44 | PRJNA594379 | B44_Aer_Su2_1AS | Pseudoplatystoma corruscans | CODEVASF * | kidney | Yes |
| B45 | PRJNA590964 | B45_Pi_23_1MC | Phractocephalus hemioliopterus | CODEVASF * | kidney | Yes |
| B47 | PRJNA590505 | B47_Aer_Pi4_2HPAS | Phractocephalus hemioliopterus | CODEVASF * | kidney | - |
| B48 | PRJNA594423 | B48_Aer_Pi5_1HPAS | Phractocephalus hemioliopterus | CODEVASF * | kidney | - |
| B52 | PRJNA595563 | B52_Aer_Pi17_ | Phractocephalus hemioliopterus | CODEVASF * | kidney | Yes |
| 1HPAS | ||||||
| B79 | PRJNA590958 | B79_Aer_onGT1 | Oreochromis niloticus | Instituto Federal | kidney | - |
| B80 | PRJNA594352 | B80_Aer_ | Environmental (water) | CODEVASF * | Not applicable | - |
| WatCTCBM21 | ||||||
| MM_B02 | - | MM_B02 | Homo sapiens | Teaching Hospital | Soft tissues infection | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maia, J.C.d.S.; Silva, G.A.d.A.; Cunha, L.S.d.B.; Gouveia, G.V.; Góes-Neto, A.; Brenig, B.; Araújo, F.A.; Aburjaile, F.; Ramos, R.T.J.; Soares, S.C.; et al. Genomic Characterization of Aeromonas veronii Provides Insights into Taxonomic Assignment and Reveals Widespread Virulence and Resistance Genes throughout the World. Antibiotics 2023, 12, 1039. https://doi.org/10.3390/antibiotics12061039
Maia JCdS, Silva GAdA, Cunha LSdB, Gouveia GV, Góes-Neto A, Brenig B, Araújo FA, Aburjaile F, Ramos RTJ, Soares SC, et al. Genomic Characterization of Aeromonas veronii Provides Insights into Taxonomic Assignment and Reveals Widespread Virulence and Resistance Genes throughout the World. Antibiotics. 2023; 12(6):1039. https://doi.org/10.3390/antibiotics12061039
Chicago/Turabian StyleMaia, José Cleves da Silva, Gabriel Amorim de Albuquerque Silva, Letícia Stheffany de Barros Cunha, Gisele Veneroni Gouveia, Aristóteles Góes-Neto, Bertram Brenig, Fabrício Almeida Araújo, Flávia Aburjaile, Rommel Thiago Jucá Ramos, Siomar Castro Soares, and et al. 2023. "Genomic Characterization of Aeromonas veronii Provides Insights into Taxonomic Assignment and Reveals Widespread Virulence and Resistance Genes throughout the World" Antibiotics 12, no. 6: 1039. https://doi.org/10.3390/antibiotics12061039
APA StyleMaia, J. C. d. S., Silva, G. A. d. A., Cunha, L. S. d. B., Gouveia, G. V., Góes-Neto, A., Brenig, B., Araújo, F. A., Aburjaile, F., Ramos, R. T. J., Soares, S. C., Azevedo, V. A. d. C., Costa, M. M. d., & Gouveia, J. J. d. S. (2023). Genomic Characterization of Aeromonas veronii Provides Insights into Taxonomic Assignment and Reveals Widespread Virulence and Resistance Genes throughout the World. Antibiotics, 12(6), 1039. https://doi.org/10.3390/antibiotics12061039











