Antibiotic Resistance of Bacterial Isolates from Smallholder Poultry Droppings in the Guinea Savanna Zone of Nigeria
Abstract
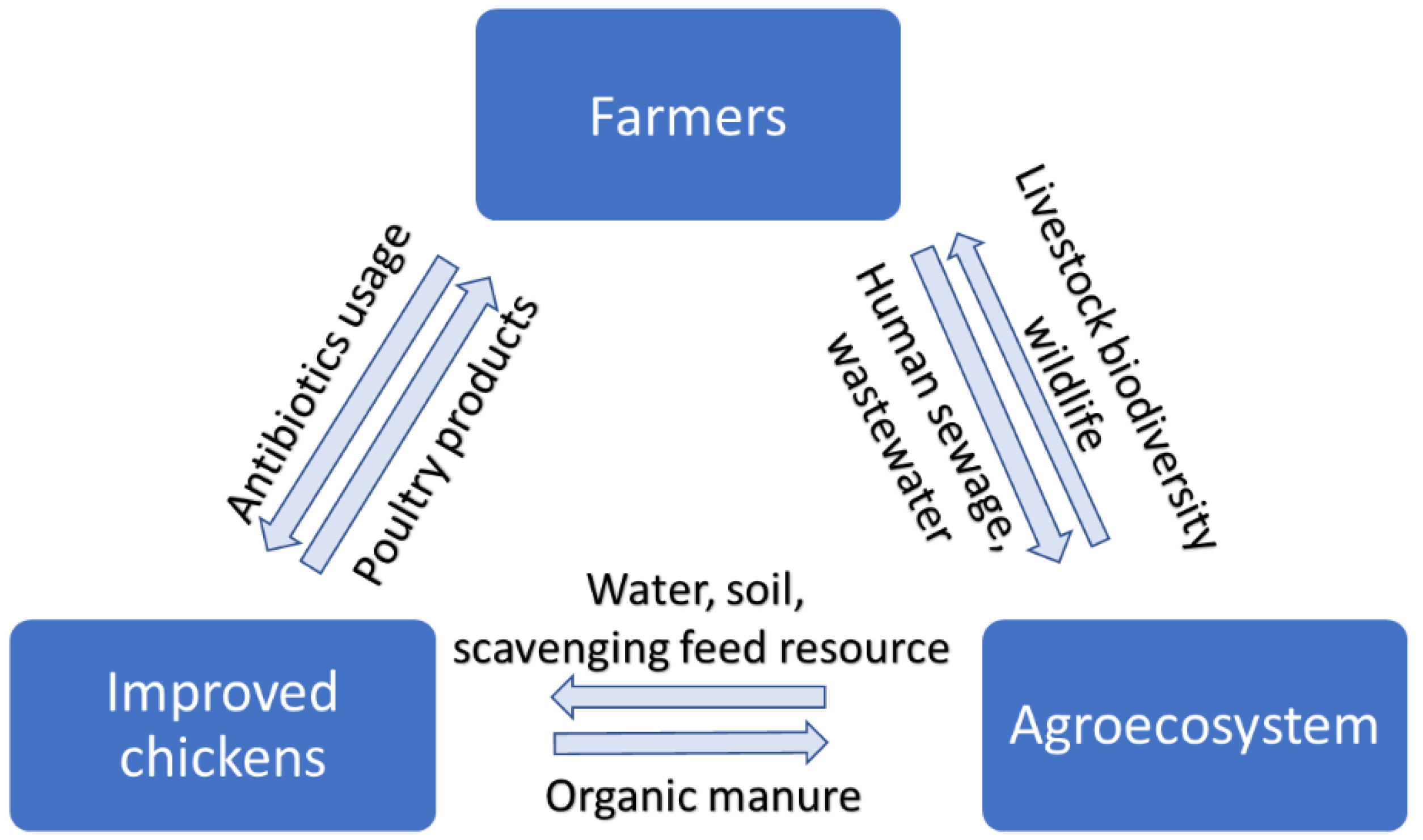
1. Introduction
2. Results
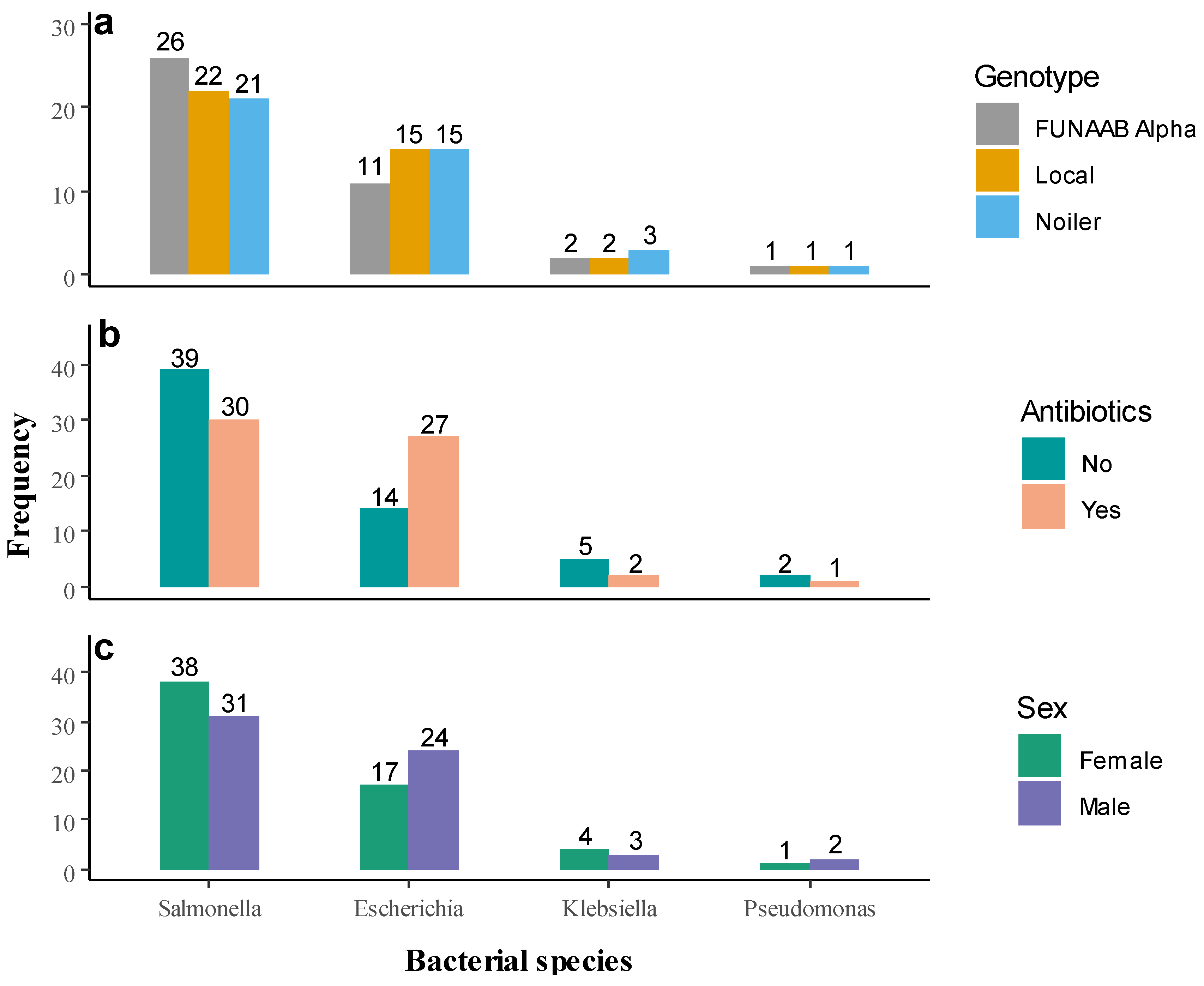
2.1. Microbial Count and Prevalence of Bacterial Pathogen
2.2. Antibiotic Resistance Pattern
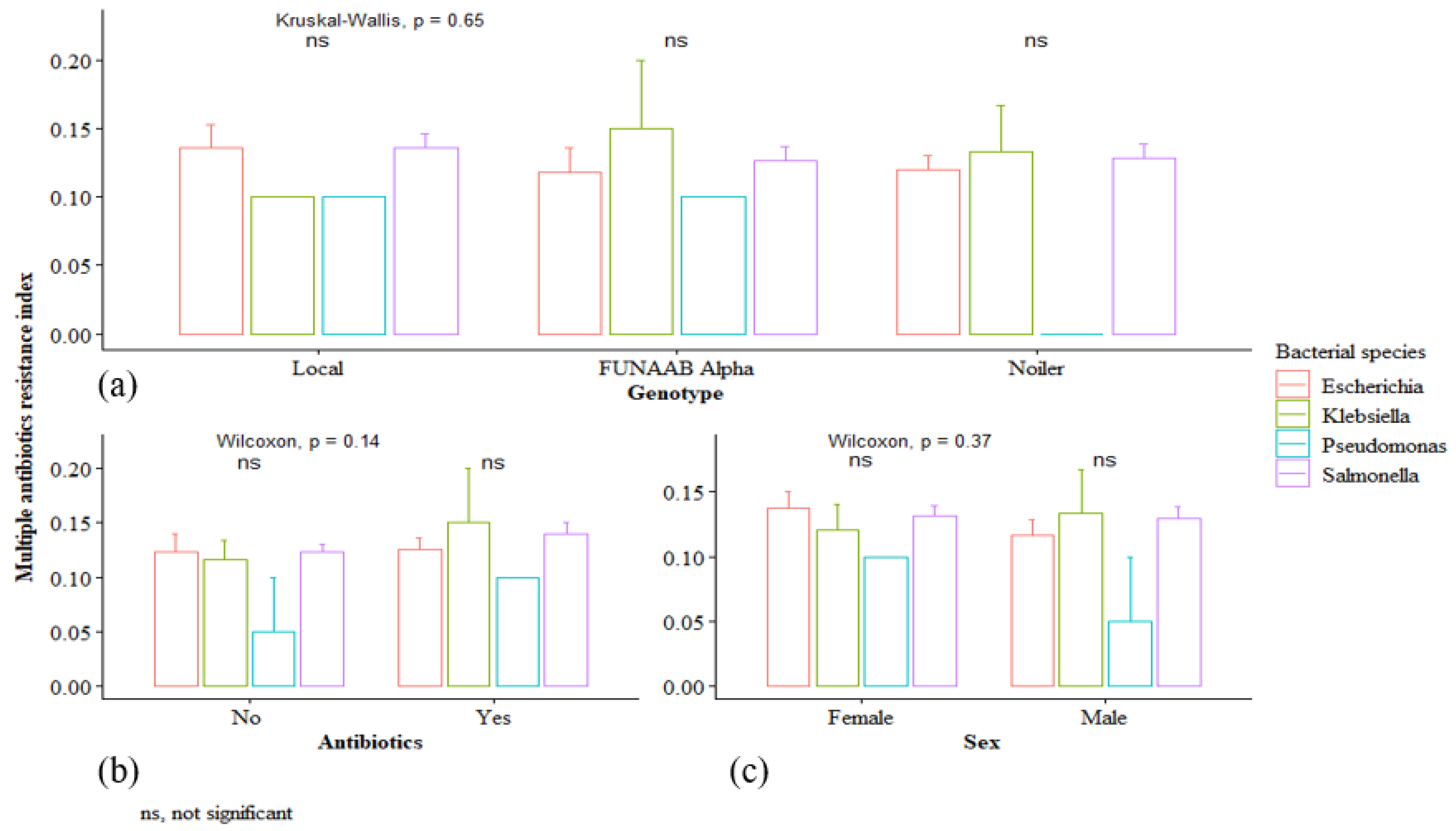
2.3. Multiple Antibiotic Resistance Index
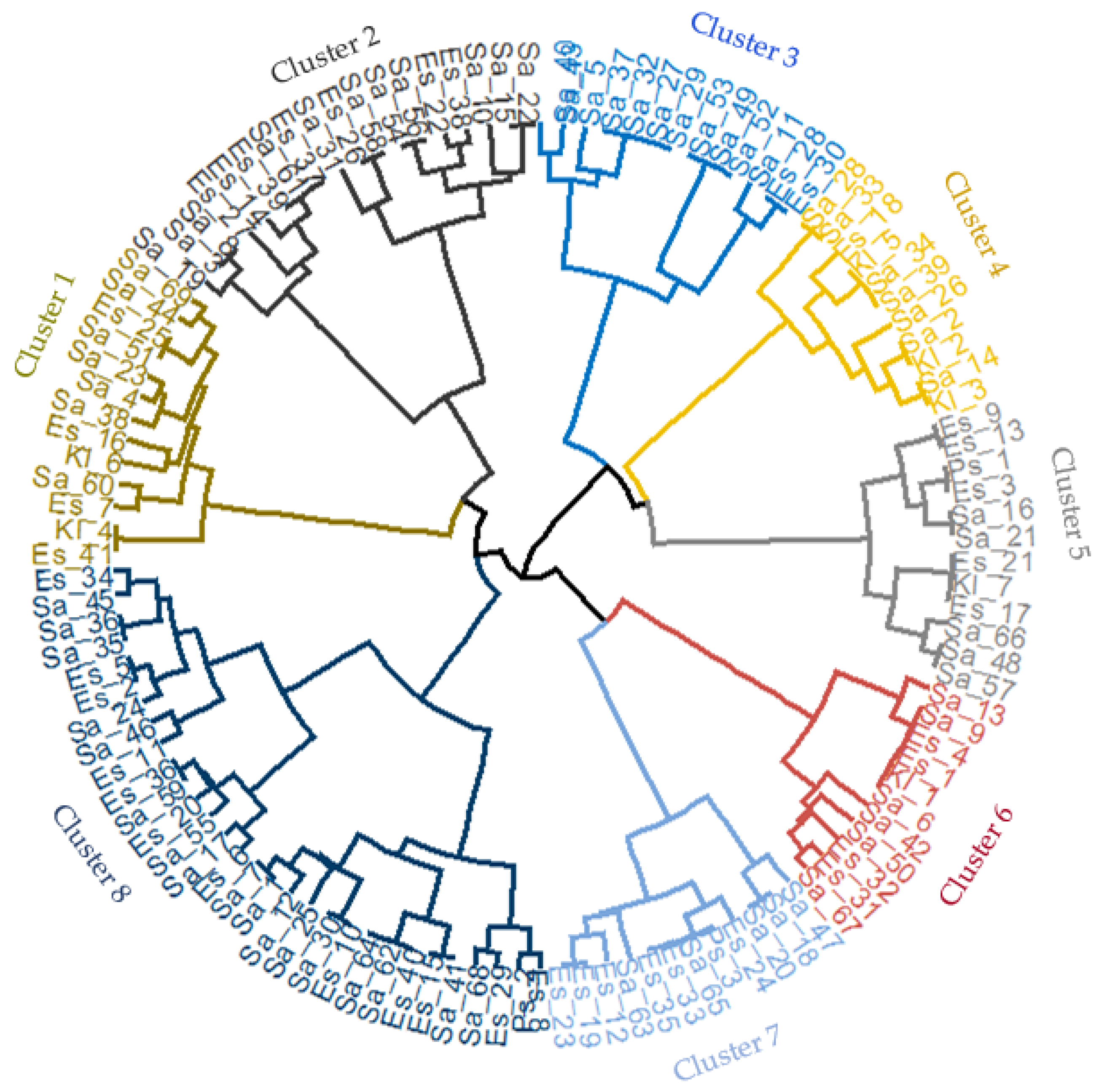
2.4. Hierarchical Clustering of Bacterial Isolates
3. Discussion
4. Materials and Methods
4.1. Sampling Location and Farmer Selection
4.2. Collection of Samples
4.3. Isolation and Identification of Bacteria Isolates
4.4. Antimicrobial Susceptibility Testing
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 9 December 2021).
- FAO. Small Scale Poultry Production: A Technical Guide; Food and Agriculture Organization of the United Nations: Rome, Italy, 2004. [Google Scholar]
- FAO CountrySTAT. Available online: http://nigeria.countrystat.org/search-and-visualize-data/en/ (accessed on 8 December 2021).
- Van Boeckel, T.P.; Brower, C.; Gilbert, M.; Grenfell, B.T.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global trends in antimicrobial use in food animals. Proc. Natl. Acad. Sci. USA 2015, 112, 5649–5654. [Google Scholar] [CrossRef]
- Roth, N.; Käsbohrer, A.; Mayrhofer, S.; Zitz, U.; Hofacre, C.; Domig, K.J. The application of antibiotics in broiler production and the resulting antibiotic resistance in Escherichia coli: A global overview. Poult. Sci. 2019, 98, 1791–1804. [Google Scholar] [CrossRef] [PubMed]
- Gupta, C.L.; Blum, S.E.; Kattusamy, K.; Druyan, D.T.; Shapira, S.; Krifucks, R.; Zhu, O.; Yong-Guan, Z.; Zhou, X.-Y.; Su, J.-Q.; et al. Longitudinal study on the effects of growth-promoting and therapeutic antibiotics on the dynamics of chicken cloacal and litter microbiomes and resistomes. Microbiome 2021, 9, 178. [Google Scholar] [CrossRef] [PubMed]
- Zalewska, M.; Błazejewska, A.; Czapko, A.; Popowska, M. Antibiotics and antibiotic resistance genes in animal manure—Consequences of its application in agriculture. Front. Microbiol. 2021, 12, 610656. [Google Scholar] [CrossRef] [PubMed]
- Oluwasile, B.B.; Agbaje, M.; Ojo, O.E.; Dipeolu, M.A. Antibiotic usage pattern in selected poultry farms in Ogun state. Sokoto J. Vet. Sci. 2014, 12, 45–50. [Google Scholar] [CrossRef]
- Sanderson, H.; Fricker, C.; Brown, R.S.; Majury, A.; Liss, S.N. Antibiotic resistance genes as an emerging environmental contaminant. Environ. Rev. 2016, 24, 205–218. [Google Scholar] [CrossRef]
- Mehdi, Y.; Létourneau-Montminy, M.-P.; Gaucher, M.-L.; Chorfi, Y.; Suresh, G.; Rouissi, T.; Brar, S.T.; Cote, C.; Ramirez, A.A.; Godbout, S. Use of antibiotics in broiler production: Global impacts and alternatives. Anim. Nutr. 2018, 4, 170–178. [Google Scholar] [CrossRef]
- Hedman, H.D.; Vasco, K.A.; Zhang, L. A Review of Antimicrobial Resistance in Poultry Farming within Low-Resource Settings. Animals 2020, 10, 264. [Google Scholar] [CrossRef]
- Tilman, D.; Balzer, C.; Hill, J.; Befort, B.L. Global food demand and the sustainable intensification of agriculture. Proc. Natl. Acad. Sci. USA 2011, 108, 20260–20264. [Google Scholar] [CrossRef]
- Hafez, H.M.; Attia, Y.A. Challenges to the Poultry Industry: Current Perspectives and Strategic Future After the COVID-19 Outbreak. Front. Vet. Sci. 2020, 7, 516. [Google Scholar] [CrossRef]
- Selaledi, A.L.; Mohammed, H.Z.; Manyelo, T.G.; Mabelebele, M. The Current Status of the Alternative Use to Antibiotics in Poultry Production: An African Perspective. Antibiotics 2020, 9, 594. [Google Scholar] [CrossRef]
- Yang, Y.; Ashworth, A.J.; Willett, C.; Cook, K.; Upadhyay, A.; Owens, P.R.; Ricke, S.C.; DeBruyn, J.M.; Moore, P.A., Jr. Review of Antibiotic Resistance, Ecology, Dissemination, and Mitigation in U.S. Broiler Poultry Systems. Front. Microbiol. 2019, 10, 2639. [Google Scholar] [CrossRef] [PubMed]
- UK-VARSS. Veterinary Antibiotic Resistance and Sales Surveillance Report (UK-VARSS 2019). Available online: www.gov.uk/government/collections/veterinary-antimicrobialresistance-and-sales-surveillance (accessed on 5 December 2021).
- Bean-Hodgins, L.; Kiarie, E.G. Mandated restrictions on the use of medically important antibiotics in broiler chicken production in Canada: Implications, emerging challenges, and opportunities for bolstering gastrointestinal function and health—A review. Can. J. Anim. Sci. 2021, 101, 602–629. [Google Scholar] [CrossRef]
- Bushen, A.; Tekalign, E.; Abayneh, M. Drug- and Multidrug-Resistance Pattern of Enterobacteriaceae Isolated from Droppings of Healthy Chickens on a Poultry Farm in Southwest Ethiopia. Infect. Drug Resist. 2021, 14, 2051–2058. [Google Scholar] [CrossRef] [PubMed]
- Mkize, N.; Zishiri, O.T.; Mukaratirwa, S. Genetic characterisation of antimicrobial resistance and virulence genes in Staphylococcus aureus isolated from commercial broiler chickens in the Durban metropolitan area, South Africa. J. S. Afr. Vet. Assoc. 2017, 88, a1416. [Google Scholar] [CrossRef]
- Ahlers, C.; Alders, R.G.; Bagnol, B.; Cambaza, A.B.; Harun, M.; Mgomezulu, R.; Msami, H.; Pym, B.; Wegener, P.; Wethli, E.; et al. Improving Village Chicken Production: A Manual for Field Workers and Trainers; no. 139; Australian Centre for International Agricultural Research: Canberra, Australia, 2009. Available online: https://www.aciar.gov.au/publication/books-and-manuals/improving-village-chicken-production-manual-field-workers-and-trainers (accessed on 9 December 2021).
- Hegde, N.V.; Kariyawasam, S.; DebRoy, C. Comparison of antimicrobial resistant genes in chicken gut microbiome grown on organic and conventional diet. Vet. Anim. Sci. 2016, 1–2, 9–14. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guèye, E.F. Ethnoveterinary medicine against poultry diseases in African villages. World’s Poult. Sci. J. 1999, 55, 187–198. [Google Scholar] [CrossRef]
- Abo-EL-Sooud, K. Ethnoveterinary perspectives and promising future. Int. J. Vet. Sci. Med. 2018, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yakubu, A.; Bamidele, O.; Hassan, W.A.; Ajayi, F.O.; Ogundu, U.E.; Alabi, O.; Sonaiya, E.B.; Adebambo, O.A. Farmers’ choice of genotypes and trait preferences in tropically-adapted chickens in five agro-ecological zones in Nigeria. Trop. Anim. Health Prod. 2020, 52, 95–107. [Google Scholar] [CrossRef]
- Bamidele, O.; Sonaiya, E.B.; Adebambo, O.A.; Dessie, T. On-station performance evaluation of improved tropically adapted chicken breeds for smallholder poultry production systems in Nigeria. Trop. Anim. Health Prod. 2020, 52, 1541–1548. [Google Scholar] [CrossRef]
- Bamidele, O.; Amole, T.A.; Oyewale, O.A.; Bamidele, O.O.; Yakubu, A.; Ogundu, U.E.; Ajayi, F.O.; Hassan, W.A. Antimicrobial Usage in Smallholder Poultry Production in Nigeria. Vet. Med. Int. 2022, 2022, 7746144. [Google Scholar] [CrossRef] [PubMed]
- Alabi, O.O.; Ajayi, F.O.; Bamidele, O.; Yakubu, A.; Ogundu, E.U.; Sonaiya, E.B.; Ojo, M.A.; Hassan, W.A.; Adebambo, O.A. Impact assessment of improved chicken genetics on livelihoods and food security of smallholder poultry farmers in Nigeria. Livest. Res. Rural. Dev. 2020, 32, 77. [Google Scholar]
- Hoffmann, I. Livestock biodiversity. Rev. Sci. Tech. 2010, 29, 73–86. [Google Scholar] [CrossRef] [PubMed]
- FAO. Smallholder Poultry Production—Livelihoods, Food Security and Sociocultural Significance; FAO Smallholder Poultry Production Paper No. 4; Kryger, K.N., Thomsen, K.A., Whyte, M.A., Dissing, M., Eds.; FAO: Rome, Italy, 2010. [Google Scholar]
- VKM. Antimicrobial Resistance Due to the Use of Biocides and Heavy Metals: A Literature Review; Scientific Opinion on the Panel on Microbial Ecology of the Norwegian Scientific Committee for Food Safety; VKM: Oslo, Norway, 2016; ISBN 978-82-8259-253-6. Available online: http://hdl.handle.net/11250/2471729 (accessed on 11 December 2021).
- Westphal-Settele, K.; Konradi, S.; Balzer, F.; Schönfeld, J.; Schmithausen, R. Die Umwelt als Reservoir für Antibiotikaresistenzen: Ein wachsendes Problem für die öffentliche Gesundheit? [The environment as a reservoir for antimicrobial resistance: A growing problem for public health?]. Bundesgesundheitsblatt Gesundh. Gesundh. 2018, 61, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Robles-Jimenez, L.E.; Aranda-Aguirre, E.; Castelan-Ortega, O.A.; Shettino-Bermudez, B.S.; Ortiz-Salinas, R.; Miranda, M.; Li, X. Worldwide Traceability of Antibiotic Residues from Livestock in Wastewater and Soil: A Systematic Review. Animals 2021, 12, 60. [Google Scholar] [CrossRef]
- Teale, C.J. Antimicrobial resistance and the food chain. J. Appl. Microbiol. Symp. Suppl. 2002, 92, 85S–89S. [Google Scholar] [CrossRef]
- FAO. The Economic Lives of Smallholder Farmers: An Analysis Based on Household Data from Nine Countries; Food and Agriculture Organization of the United Nations: Rome, Italy, 2015. [Google Scholar]
- Swift, B.M.C.; Bennett, M.; Waller, K.; Dodd, C.; Murray, A.; Gomes, R.L.; Humphreys, B.; Hobman, J.L.; Jones, M.A.; Whitlock, S.E.; et al. Anthropogenic environmental drivers of antimicrobial resistance in wildlife. Sci. Total Environ. 2019, 649, 12–20. [Google Scholar] [CrossRef]
- Franklin, A.M.; Williams, C.F.; Andrews, D.M.; Woodward, E.W.; Watson, J.E. Uptake of three antibiotics and an antiepileptic drug by wheat crops spray irrigated with wastewater treatment plant effluent. J. Environ. Qual. 2016, 45, 546–554. [Google Scholar] [CrossRef]
- Franklin, A.M.; Williams, C.F.; Watson, J.E. Assessment of Soil to Mitigate Antibiotics in the Environment Due to Release of Wastewater Treatment Plant Effluent. J. Environ. Qual. 2018, 47, 1347–1355. [Google Scholar] [CrossRef]
- Tripathi, V.; Cytryn, E. Impact of anthropogenic activities on the dissemination of antibiotic resistance across ecological boundaries. Essays Biochem. 2017, 61, 11–21. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fekede, G.; Tadesse, Y.; Esatu, W.; Dessie, T. On-farm comparative production and reproduction performance evaluation of Sasso, Sasso-RIR, Koekoek and Improved Local chicken breeds in Bako Tibe and Dano districts of western Oromia, Ethiopia. Livest. Res. Rural. Dev. 2021, 33, 15. [Google Scholar]
- Guni, F.S.; Mbaga, S.H.; Katule, A.M. Performance Evaluation of Kuroiler and Sasso Chicken Breeds Reared under On-farm and On-station Management Conditions in Tanzania. Eur. J. Agric. Food Sci. 2021, 3, 53–59. [Google Scholar] [CrossRef]
- Awogbemi, J.; Adeyeye, M.; Akinkunmi, E.O. A Survey of Antimicrobial Agents Usage in Poultry Farms and Antibiotic Resistance in Escherichia coli and Staphylococci Isolates from the Poultry in Ile-Ife, Nigeria. J. Infect. Dis. Epidemiol. 2018, 4, 47. [Google Scholar] [CrossRef]
- Sule, I.O.; Olorunfemi, A.A.; Otori, A.O. Mycological and bacteriological assessment of poultry droppings from poultry pens within Ilroin, Kwara, Nigeria. Sci. World J. 2019, 14, 1–6. [Google Scholar]
- King, D.E.; Malone, R.; Lilley, S.H. New classification and update on the quinolone antibiotics. Am. Fam. Physician. 2000, 61, 2741–2748. [Google Scholar]
- Osman, A.Y.; Elmi, S.A.; Simons, D.; Elton, L.; Haider, N.; Khan, M.A.; Othman, I.; Zumla, A.; McCoy, D.; Kock, R. Antimicrobial Resistance Patterns and Risk Factors Associated with Salmonella spp. Isolates from Poultry Farms in the East Coast of Peninsular Malaysia: A Cross-Sectional Study. Pathogens 2021, 10, 1160. [Google Scholar] [CrossRef] [PubMed]
- Ngogang, M.P.; Ernest, T.; Kariuki, J.; Mouliom Mouiche, M.M.; Ngogang, J.; Wade, A.; van der Sande, M.A.B. Microbial Contamination of Chicken Litter Manure and Antimicrobial Resistance Threat in an Urban Area Setting in Cameroon. Antibiotics 2021, 10, 20. [Google Scholar] [CrossRef]
- Kakooza, S.; Muwonge, A.; Nabatta, E.; Eneku, W.; Ndoboli, D.; Wampande, E.; Munyiirwa, D.; Kayaga, E.; Tumwebaze, M.A.; Afayoa, M.; et al. Retrospective analysis of antimicrobial resistance in pathogenic Escherichia coli and Salmonella spp. isolates from poultry in Uganda. Int. J. Vet. Sci. Med. 2021, 9, 11–21. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antimicrobial Resistance. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 6 January 2022).
- Martínez, J.L. Effect of antibiotics on bacterial populations: A multi-hierachical selection process. F1000Research 2017, 6, 51. [Google Scholar] [CrossRef]
- Trawińska, B.; Chmielowiec-Korzeniowska, A.; Nowakowicz-Dębek, B.; Tymczyna, L.; Bombik, T.; Pyrz, M.; Tymczyna-Sobotka, M. Evaluation of microbial contamination of feces and soil on a laying-hen farm depending on sampling site and season. Rev. Bras. Zootec. 2016, 45, 190–194. [Google Scholar] [CrossRef]
- do Amaral, L.A. Drinking Water as a Risk Factor to Poultry Health. Braz. J. Poult. Sci. 2004, 6, 191–199. [Google Scholar] [CrossRef]
- Rose, N.; Besuderau, F.; Drouin, P.; Toux, J.Y.; Rose, V.; Colin, P. Risk factors of Salmonella persistence after cleansing and disinfection in French broiler—Chicken house. Prevent. Vet. Med. 2000, 44, 9–20. [Google Scholar] [CrossRef]
- Kolář, M.; Pantůček, R.; Bardoň, J.; Vágnerová, I.; Typovská, H.; Válka, I.; Doškař, J. Occurrence of antibiotic-resistant bacterial strains isolated in poultry. Vet. Med. Czech. 2002, 47, 52–59. [Google Scholar] [CrossRef]
- Ajayi, K.O.; Omoya, F.O. Antibiotic Usage Pattern in Poultry and Resistance Pattern of Human Pathogenic Bacteria Isolated from Poultry Droppings in Akure, Nigeria. Int. J. Biomed. Sci. Eng. 2017, 5, 35–40. [Google Scholar] [CrossRef]
- Nhung, N.T.; Chansiripornchai, N.; Carrique-Mas, J.J. Antimicrobial Resistance in Bacterial Poultry Pathogens: A Review. Front. Vet. Sci. 2017, 4, 126. [Google Scholar] [CrossRef]
- Xu, J.; Sangthong, R.; McNeil, E.; Tang, R.; Chongsuvivatwong, V. Antibiotic use in chicken farms in northwestern China. Antimicrob. Resist. Infect. Control 2020, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, P.; Dai, Y.; Liu, Y.; Song, Y.; Yu, L.; Feng, C.; Liu, M.; Xie, Z.; Shang, Y.; et al. Longitudinal monitoring of multidrug resistance in Escherichia coli on broiler chicken fattening farms in Shandong, China. Poult. Sci. 2021, 100, 100887. [Google Scholar] [CrossRef]
- Afunwa, R.A.; Ezeanyinka, J.; Afunwa, E.C.; Udeh, A.S.; Oli, N.A.; Unachukwu, M. Multiple antibiotic resistant index of gram-negative bacteria from bird droppings in two commercial poultries in Enugu, Nigeria. Open J. Med. Microbiol. 2020, 10, 171–181. [Google Scholar] [CrossRef]
- Mpenda, F.N.; Schilling, M.A.; Campbell, Z.; Mngumi, E.B.; Buza, J. The genetic diversity of local african chickens: A potential for selection of chickens resistant to viral infections. J. Appl. Poult. Res. 2019, 28, 1–12. [Google Scholar] [CrossRef]
- Rotchell, D.; Paul, D. Multiple Antibiotic Resistance Index. Fitness and Virulence Potential in Respiratory Pseudomonas aeruginosa from Jamaica. J. Med. Microbiol. 2016, 65, 251–271. [Google Scholar]
- Gheyas, A.A.; Vallejo-Trujillo, A.; Kebede, A.; Lozano-Jaramillo, M.; Dessie, T.; Smith, J.; Hanotte, O. Integrated Environmental and Genomic Analysis Reveals the Drivers of Local Adaptation in African Indigenous Chickens. Mol. Biol. Evol. 2021, 38, 4268–4285. [Google Scholar] [CrossRef] [PubMed]
- Riaz, S.; Faisal, M.; Hasnain, S. Antibiotic Susceptibility Pattern and Multiple Antibiotic Resistance (MAR) Calculation of Extended Spectrum β-Lactamase (ESBL) Producing Escherichia coli and Klebsiella Species in Pakistan. Afr. J. Biotechnol. 2011, 10, 6325–6331. [Google Scholar]
- WHO. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery and Development of New Antibiotics. 2017. Available online: https://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf (accessed on 14 February 2021).
- WHO. The Selection and Use of Essential Medicines: Report of the WHO Expert Committee on Selection and Use of Essential Medicines; Including the 22nd WHO Model List of Essential Medicines and the 8th WHO Model List of Essential Medicines for Children; WHO Technical Report Series, No. 1035; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Murray, C.J.L.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; antimicrobial resistance collaborators. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Aworh, M.K.; Kwaga, J.K.P.; Hendriksen, R.S.; Okolocha, E.C.; Thakur, S. Genetic relatedness of multidrug resistant Escherichia coli isolated from humans, chickens and poultry environments. Antimicrob. Resist. Infect. Control 2021, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lyu, N.; Liu, F.; Liu, W.J.; Bi, Y.; Zhang, Z.; Ma, S.; Cao, J.; Song, X.; Wang, A.; et al. More diversified antibiotic resistance genes in chickens and workers of the live poultry markets. Environ. Int. 2021, 153, 106534. [Google Scholar] [CrossRef] [PubMed]
- Hull, D.M.; Harrell, E.; van Vliet, A.H.M.; Correa, M.; Thakur, S. Antimicrobial resistance and interspecies gene transfer in Campylobacter coli and Campylobacter jejuni isolated from food animals, poultry processing, and retail meat in North Carolina, 2018–2019. PLoS ONE 2021, 16, e0246571. [Google Scholar] [CrossRef]
- Suwono, B.; Eckmanns, T.; Kaspar, H.; Merle, R.; Zacher, B.; Kollas, C. Cluster analysis of resistance combinations in Escherichia coli from different human and animal populations in Germany 2014–2017. PLoS ONE 2021, 16, e0244413. [Google Scholar] [CrossRef] [PubMed]
- Alhaji, N.B.; Isola, T.O. Antimicrobial usage by pastoralists in food animals in North-central Nigeria: The associated socio-cultural drivers for antimicrobials misuse and public health implications. One Health 2018, 6, 41–47. [Google Scholar] [CrossRef]
- Bamidele, O.; Amole, T.A. Impact of COVID-19 on Smallholder Poultry Farmers in Nigeria. Sustainability 2021, 13, 11475. [Google Scholar] [CrossRef]
- Cheesbrough, M. District Laboratory Practice in Tropical Countries, 2nd ed.; Cambridge University Press: New York, NY, USA, 2006; 434p. [Google Scholar]
- Ochei, J.; Kolhatkar, A. Medical Laboratory Science: Theory and Practice, 10th ed.; Tata McGraw-Hill Publishing Company Ltd.: New Delhi, India, 2008; 1310p. [Google Scholar]
- WHO. AWaRe Classification. World Health Organization. Available online: https://www.who.int/publications/i/item/2021-aware-classification (accessed on 23 September 2021).
- Krumpermann, P.H. Multiple antibiotic resistance indexing of Escherihia coli to identify high-risk sources of fecal contamination in foods. Appl. Environ. Microbiol. 1983, 46, 165–170. [Google Scholar] [CrossRef]
- Nattino, G.; Pennell, M.L.; Lemeshow, S. Assessing the goodness of fit of logistic regression models in large samples: A modification of the Hosmer-Lemeshow test. Biometrics 2020, 76, 549–560. [Google Scholar] [CrossRef] [PubMed]
- IBM SPSS. Statistics for Windows, Version 27.0; IBM Corp.: Armonk, NY, USA, 2020. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 2 December 2021).
- Kassambara, A.; Mundt, F. Factoextra: Extract and Visualize the Results of Multivariate Data Analyses, R Package Version 1.0.7. 2020. Available online: https://CRAN.R-project.org/package=factoextra (accessed on 21 January 2022).

| Variables | Number | Mean ± S.E. (×105) | Minimum | Maximum |
|---|---|---|---|---|
| Bacterial species | ||||
| Salmonella spp. | 69 | 4.64 ± 0.20 | 2.0 | 7.4 |
| Pseudomonas spp. | 3 | 5.10 ± 0.47 | 4.4 | 6.0 |
| Klebsiella spp. | 7 | 5.19 ± 0.55 | 3.9 | 7.6 |
| E. coli | 41 | 4.70 ± 0.19 | 2.1 | 7.2 |
| Chicken genotype | ||||
| Local | 40 | 4.81 ± 0.26 | 2.0 | 7.6 |
| FUNAAB Alpha | 40 | 4.71 ± 0.22 | 2.3 | 7.3 |
| Noiler | 40 | 4.60 ± 0.23 | 2.1 | 7.4 |
| Sex | ||||
| Male | 60 | 4.69 ± 0.18 | 2.1 | 7.4 |
| Female | 60 | 4.72 ± 0.20 | 2.0 | 7.6 |
| Antibiotics usage | ||||
| No | 60 | 4.76 ± 0.22 | 2.0 | 7.6 |
| Yes | 60 | 4.65 ± 0.17 | 2.1 | 7.4 |
| Parameters | β | S.E. | Wald | df | p-Value | Odds Ratio | 95% C.I. |
|---|---|---|---|---|---|---|---|
| Chicken genotype (ref: Local) | 1.276 | 2 | 0.528 | ||||
| FUNAAB Alpha | −0.442 | 0.507 | 0.760 | 1 | 0.383 | 0.643 | 0.238 − 1.736 |
| Noiler | 0.106 | 0.493 | 0.046 | 1 | 0.830 | 1.112 | 0.423 − 2.921 |
| Sex (ref: male) | |||||||
| Female | −0.541 | 0.412 | 1.731 | 1 | 0.188 | 0.582 | 0.260 − 1.304 |
| Antibiotics usage (ref: No) | |||||||
| Yes | 0.937 | 0.416 | 5.071 | 1 | 0.024 * | 2.552 | 1.129 − 5.767 |
| Constant | −0.667 | 0.456 | 2.138 | 1 | 0.144 | 0.513 |
| Antibiotic Agents | Bacterial Species/Number (n) of Isolates | Total n = 120 | Kruskal–Wallis Test | p-Value | |||
|---|---|---|---|---|---|---|---|
| Salmonella spp. n = 69 | Pseudomonas spp. n= 3 | Klebsiella spp. n = 7 | E. coli n = 41 | ||||
| Ciprofloxacin | |||||||
| Susceptibility | 63 (91.3) | 2 (66.7) | 6 (85.7) | 35 (85.4) | 106 (88.3) | 2.335 | 0.506 |
| Resistance | 6 (8.7) | 1 (33.3) | 1 (14.3) | 6 (14.6) | 14 (11.7) | ||
| Ofloxacin | |||||||
| Susceptibility | 62 (89.9) | 3 (100.0) | 5 (71.4) | 37 (90.2) | 107 (89.2) | 2.705 | 0.439 |
| Resistance | 7 (10.1) | 0 (0.0) | 2 (28.6) | 4 (9.8) | 13 (10.8) | ||
| Nalidixic acid | |||||||
| Susceptibility | 55 (79.7) | 3 (100.0) | 7 (100.0) | 35 (85.4) | 100 (83.3) | 2.751 | 0.432 |
| Resistance | 14 (20.3) | 0 (0.0) | 0 (0.0) | 6 (14.6) | 20 (16.7) | ||
| Perfloxacin | |||||||
| Susceptibility | 59 (85.5) | 3 (100.0) | 7 (100.0) | 33 (80.5) | 102 (85.0) | 2.413 | 0.491 |
| Resistance | 10 (14.5) | 0 (0.0) | 0 (0.0) | 8 (19.5) | 18 (15.0) | ||
| Gentamicin | |||||||
| Susceptibility | 62 (89.9) | 3 (100.0) | 7 (100.0) | 38 (92.7) | 110 (91.7) | 1.250 | 0.741 |
| Resistance | 7 (10.1) | 0 (0.0) | 0 (0.0) | 3 (7.3) | 10 (8.3) | ||
| Amoxycillin-Clavulanic acid | |||||||
| Susceptibility | 61 (88.4) | 3 (100.0) | 6 (85.7) | 34 (82.9) | 104 (86.7) | 1.134 | 0.769 |
| Resistance | 8 (11.6) | 0 (0.0) | 1 (14.3) | 7 (17.1) | 16 (13.3) | ||
| Sulfamethoxazole-Trimethoprim | |||||||
| Susceptibility | 58 (84.1) | 2 (66.7) | 6 (85.7) | 34 (82.9) | 100 (83.3) | 0.654 | 0.884 |
| Resistance | 11 (15.9) | 1 (33.3) | 1 (14.3) | 7 (17.1) | 20 (16.7) | ||
| Streptomycin | |||||||
| Susceptibility | 58 (84.1) | 3 (100.0) | 7 (100.0) | 39 (95.1) | 107 (89.2) | 4.546 | 0.208 |
| Resistance | 11 (15.9) | 0 (0.0) | 0 (0.0) | 2 (4.9) | 13 (10.8) | ||
| Penicillin | |||||||
| Susceptibility | 61 (88.4) | 3 (100.0) | 4 (57.1) | 37 (90.2) | 105 (87.5) | 6.605 | 0.086 |
| Resistance | 8 (11.6) | 0 (0.0) | 3 (42.9) | 4 (9.8) | 15 (12.5) | ||
| Cephalexin | |||||||
| Susceptibility | 60 (87.0) | 3 (100.0) | 6 (85.7) | 37 (90.2) | 106 (88.3) | 0.709 | 0.871 |
| Resistance | 9 (13.0) | 0 (0.0) | 1 (14.3) | 4 (9.8) | 14 (11.7) | ||
| Antibiotic Agents | Chicken Genotype | Kruskal–Wallis Test | p-Value | ||
|---|---|---|---|---|---|
| Local n = 40 | FUNAAB Alpha n = 40 | Noiler n = 40 | |||
| Ciprofloxacin | 2 (5.0) | 7 (17.5) | 5 (12.5) | 3.047 | 0.218 |
| Ofloxacin | 3 (7.5) | 6 (15.0) | 4 (10.0) | 1.198 | 0.549 |
| Nalidixic acid | 4 (10.0) | 9 (22.4) | 7 (17.5) | 2.261 | 0.323 |
| Perfloxacin | 5 (12.5) | 7 (17.5) | 6 (15.0) | 0.389 | 0.823 |
| Gentamicin | 5 (12.5) | 3 (7.5) | 2 (5.0) | 1.515 | 0.469 |
| Amoxycillin-Clavulanic acid | 5 (12.5) | 7 (17.5) | 4 (10.0) | 1.001 | 0.606 |
| Sulfamethoxazole-Trimethoprim | 8 (20.0) | 4 (10.0) | 8 (20.0) | 1.904 | 0.386 |
| Streptomycin | 7 (17.5) | 0 (0.0) | 6 (15.0) | 7.357 | 0.025 * |
| Penicillin | 6 (15.0) | 3 (7.5) | 6 (15.0) | 1.360 | 0.507 |
| Ceporex | 8 (20.0) | 4 (10.0) | 2 (5.0) | 4.491 | 4.491 |
| Antibiotic Agents | Sex | Kruskal–Wallis Test | p-Value | |
|---|---|---|---|---|
| Male n = 60 | Female n = 60 | |||
| Ciprofloxacin | 7 (11.7) | 7 (11.7) | 0.000 | 1.000 |
| Ofloxacin | 4 (6.7) | 9 (15.0) | 2.502 | 0.114 |
| Nalidixic acid | 7 (11.7) | 13 (21.7) | 2.180 | 0.140 |
| Perfloxacin | 10 (16.7) | 8 (13.3) | 0.263 | 0.608 |
| Gentamicin | 6 (10.0) | 4 (6.7) | 0.436 | 0.509 |
| Amoxycillin-Clavulanic Acid | 7 (11.7) | 9 (15.0) | 0.688 | 0.407 |
| Sulfamethoxazole-Trimethoprim | 10 (16.7) | 10 (16.7) | 0.263 | 0.608 |
| Streptomycin | 4 (6.7) | 9 (15.0) | 2.161 | 0.142 |
| Penicillin | 11 (18.3) | 4 (6.7) | 3.337 | 0.068 |
| Cephalexin | 7 (11.7) | 7 (11.7) | 0.086 | 0.769 |
| Antibiotic Agents | Antibiotics Usage | |||
|---|---|---|---|---|
| No n = 60 | Yes n = 60 | Kruskal–Wallis Test | p-Value | |
| Ciprofloxacin | 4 (6.7) | 10 (16.7) | 1.179 | 0.278 |
| Ofloxacin | 6 (10.0) | 7 (11.7) | 0.036 | 0.849 |
| Nalidixic acid | 9 (15.0) | 11 (18.3) | 0.098 | 0.754 |
| Perfloxacin | 9 (15.0) | 9 (15.0) | 0.028 | 0.867 |
| Gentamicin | 4 (6.7) | 6 (10.0) | 0.292 | 0.589 |
| Amoxycillin-Clavulanic acid | 7 (11.7) | 9 (15.0) | 0.461 | 0.497 |
| Sulfamethoxazole-Trimethoprim | 9 (15.0) | 11 (18.3) | 0.119 | 0.730 |
| Streptomycin | 6 (10.0) | 7 (11.7) | 0.024 | 0.877 |
| Penicillin | 11 (18.3) | 4 (6.7) | 1.827 | 0.177 |
| Cephalexin | 8 (13.3) | 6 (10.0) | 0.188 | 0.665 |
| Parameters | Factors | Kruskal–Wallis Test | p-Value | |||
|---|---|---|---|---|---|---|
| Bacterial Species | ||||||
| Antibiotic Class | Salmonella spp. n = 91 | Pseudomonas spp. n = 2 | Klebsiella spp. n = 9 | E. coli n = 51 | ||
| Quinolones | 37 (40.7) | 1 (50.0) | 3 (33.3) | 24 (47.1) | 0.886 | 0.829 |
| β-lactams | 8 (8.8) | 0 (0.0) | 1 (11.1) | 7 (13.7) | ||
| Penicillins | 8 (8.8) | 0 (0.0) | 3 (33.3) | 4 (7.8) | ||
| Aminoglycosides | 18 (19.8) | 0 (0.0) | 0 (0.0) | 5 (9.8) | ||
| Sulfonamides | 11 (12.1) | 1 (50.0) | 1 (11.1) | 7 (13.7) | ||
| Cephalosporins | 9 (9.9) | 0 (0.0) | 1(11.1) | 4 (7.8) | ||
| Chicken genotype | ||||||
| Antibiotic Class | Local n = 53 | FUNAAB Alpha n = 50 | Noiler n = 50 | |||
| Quinolones | 14 (26.4) | 29 (58.0) | 22 (44.0) | 11.817 | 0.003 | |
| β-lactams | 5 (9.4) | 7 (14.0) | 4 (8.0) | |||
| Penicillins | 6 (11.3) | 3 (6.0) | 6 (12.0) | |||
| Aminoglycosides | 12 (22.6) | 3 (6.0) | 8 (16.0) | |||
| Sulfonamides | 8 (15.1) | 4 (8.0) | 8 (16.0) | |||
| Cephalosporins | 8 (15.1) | 4 (8.0) | 2 (4.0) | |||
| Sex | ||||||
| Antibiotic Class | Male n = 73 | Female n = 80 | ||||
| Quinolones | 28 (38.4) | 37 (46.3) | 0.566 | 0.452 | ||
| β-lactams | 7 (9.6) | 9 (11.3) | ||||
| Penicillins | 11 (15.1) | 4 (5.0) | ||||
| Aminoglycosides | 10 (13.7) | 13 (16.3) | ||||
| Sulfonamides | 10 (13.7) | 10 (12.5) | ||||
| Cephalosporins | 7 (9.6) | 7 (8.8) | ||||
| Antibiotics usage | ||||||
| Antibiotic Class | No n = 73 | Yes n = 80 | ||||
| Quinolones | 28 (38.4) | 37 (46.3) | 0.668 | 0.414 | ||
| β-lactams | 7 (9.6) | 9 (11.3) | ||||
| Penicillins | 11 (15.1) | 4 (5.0) | ||||
| Aminoglycosides | 10 (13.7) | 13 (16.3) | ||||
| Sulfonamides | 9 (12.3) | 11 (13.8) | ||||
| Cephalosporins | 8 (11.0) | 6 (7.5) | ||||
| Parameters | Cluster | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 n = 13 | 2 n = 18 | 3 n = 13 | 4 n = 11 | 5 n = 12 | 6 n = 11 | 7 n = 12 | 8 n = 30 | |
| Antibiotics usage | ||||||||
| No | 6 (46.2) | 7(38.9) | 7 (53.9) | 10 (90.9) | 6 (50.0) | 6 (54.6) | 3(25.0) | 16(53.3) |
| Yes | 7 (53.8) | 11 (61.1) | 6 (46.1) | 1 (9.1) | 6 (50.0) | 5 (45.4) | 9(75.0) | 14(46.7) |
| Genotype | ||||||||
| Local | 3 (23.1) | 8 (44.4) | 6 (46.2) | 2(18.2) | 4 (33.3) | 8 (72.7) | 1(8.3) | 7(23.3) |
| FUNAAB Alpha | 6 (46.2) | 7 (38.9) | 0 (0.0) | 3(27.3) | 3 (25.0) | 2 (18.2) | 7(58.3) | 12(40.0) |
| Noiler | 4 (30.7) | 3 (16.7) | 7 (53.8) | 6(54.5) | 5 (41.7) | 1 (9.0) | 4(33.7) | 11(36.7) |
| Antibiogram (%) | ||||||||
| AMC | 84.6 | 38.8 | 100 | 100 | 100 | 100 | 91.7 | 93.3 |
| CEX | 100 | 100 | 100 | 100 | 100 | 90.9 | 75.0 | 100 |
| CPX | 92.3 | 94.4 | 100 | 100 | 100 | 100 | 0.0 | 100 |
| GEN | 100 | 44.4 | 100 | 100 | 100 | 100 | 100 | 100 |
| PEF | 84.6 | 100 | 100 | 100 | 100 | 90.9 | 100 | 50.0 |
| PEN | 100 | 94.4 | 76.9 | 0.0 | 100 | 100 | 100 | 100 |
| STR | 100 | 100 | 7.7 | 100 | 100 | 100 | 100 | 100 |
| SXT | 92.6 | 100 | 69.2 | 81.8 | 0.0 | 90.9 | 100 | 96.7 |
| NA | 100 | 100 | 92.3 | 90.9 | 100 | 90.9 | 100 | 43.3 |
| OFX | 0.0 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Drug Class | ||||||||
| Quinolones | 69.2 | 98.6 | 98.1 | 97.7 | 100 | 95.5 | 75.0 | 73.3 |
| β-lactams | 84.6 | 38.8 | 100 | 100 | 100 | 100 | 91.7 | 93.3 |
| Penicillins | 100 | 94.4 | 76.9 | 0.0 | 100 | 100 | 100 | 100 |
| Aminoglycosides | 100 | 72.2 | 53.9 | 100 | 100 | 100 | 100 | 71.7 |
| Sulfonamides | 92.6 | 100 | 69.2 | 81.8 | 0.0 | 90.9 | 100 | 100 |
| Cephalosporins | 100 | 100 | 100 | 100 | 100 | 90.9 | 75.0 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bamidele, O.; Yakubu, A.; Joseph, E.B.; Amole, T.A. Antibiotic Resistance of Bacterial Isolates from Smallholder Poultry Droppings in the Guinea Savanna Zone of Nigeria. Antibiotics 2022, 11, 973. https://doi.org/10.3390/antibiotics11070973
Bamidele O, Yakubu A, Joseph EB, Amole TA. Antibiotic Resistance of Bacterial Isolates from Smallholder Poultry Droppings in the Guinea Savanna Zone of Nigeria. Antibiotics. 2022; 11(7):973. https://doi.org/10.3390/antibiotics11070973
Chicago/Turabian StyleBamidele, Oladeji, Abdulmojeed Yakubu, Ehase Buba Joseph, and Tunde Adegoke Amole. 2022. "Antibiotic Resistance of Bacterial Isolates from Smallholder Poultry Droppings in the Guinea Savanna Zone of Nigeria" Antibiotics 11, no. 7: 973. https://doi.org/10.3390/antibiotics11070973
APA StyleBamidele, O., Yakubu, A., Joseph, E. B., & Amole, T. A. (2022). Antibiotic Resistance of Bacterial Isolates from Smallholder Poultry Droppings in the Guinea Savanna Zone of Nigeria. Antibiotics, 11(7), 973. https://doi.org/10.3390/antibiotics11070973






