The Array of Antibacterial Action of Protocatechuic Acid Ethyl Ester and Erythromycin on Staphylococcal Strains
Abstract
:1. Introduction
2. Results
2.1. Antibiotic Resistance Profile of Tested Strains
2.2. The Fractional Inhibitory Concentration (FIC) Values for Protocatechuic Acid Ethyl Ester and Erythromycin against Staphylococcal Strains
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains
4.2. Antibiotic Resistance Profile
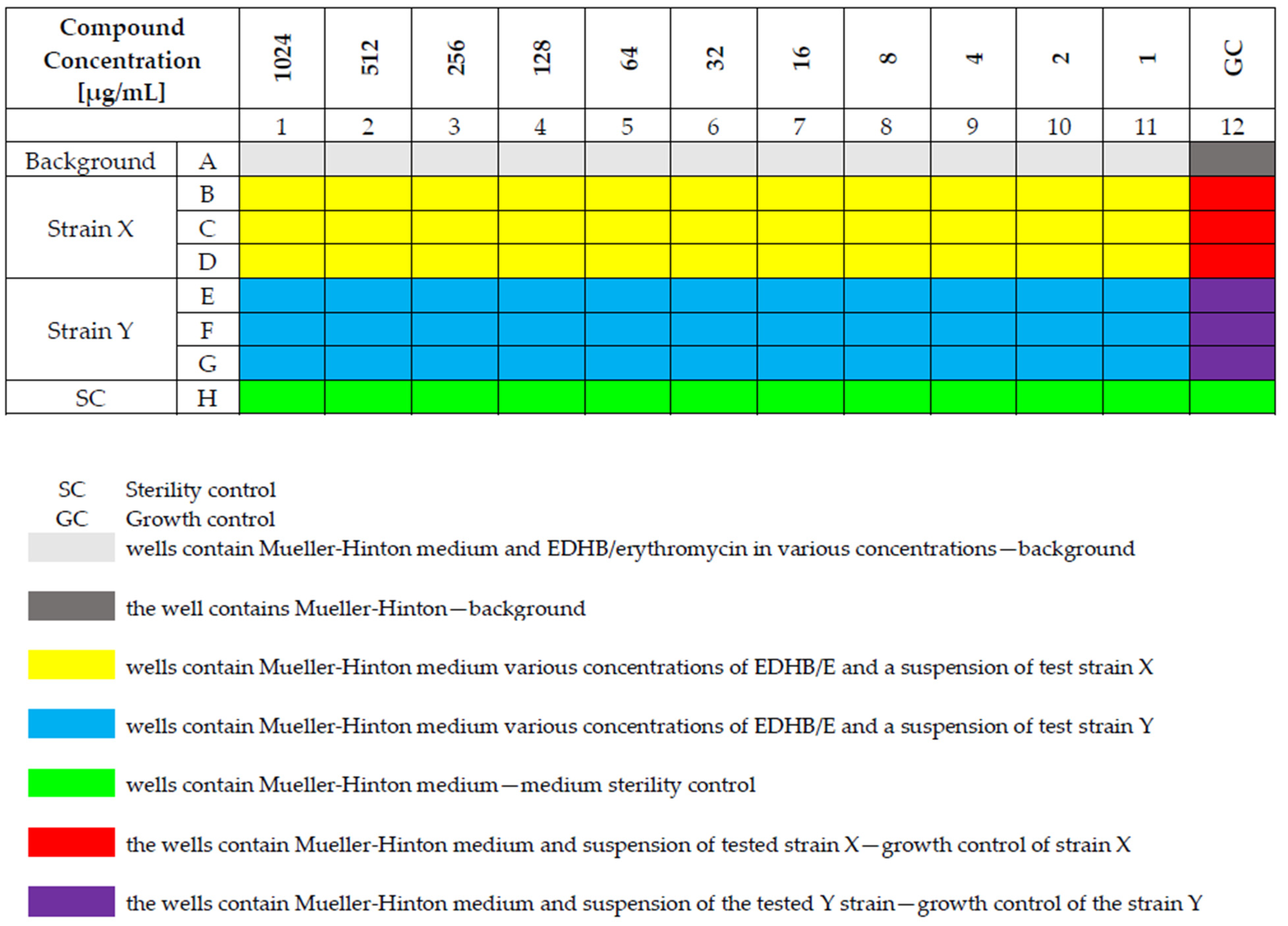
4.3. Susceptibility Testing of Staphylococcal Strains to Erythromycin and EDHB Using the Microdilution Method
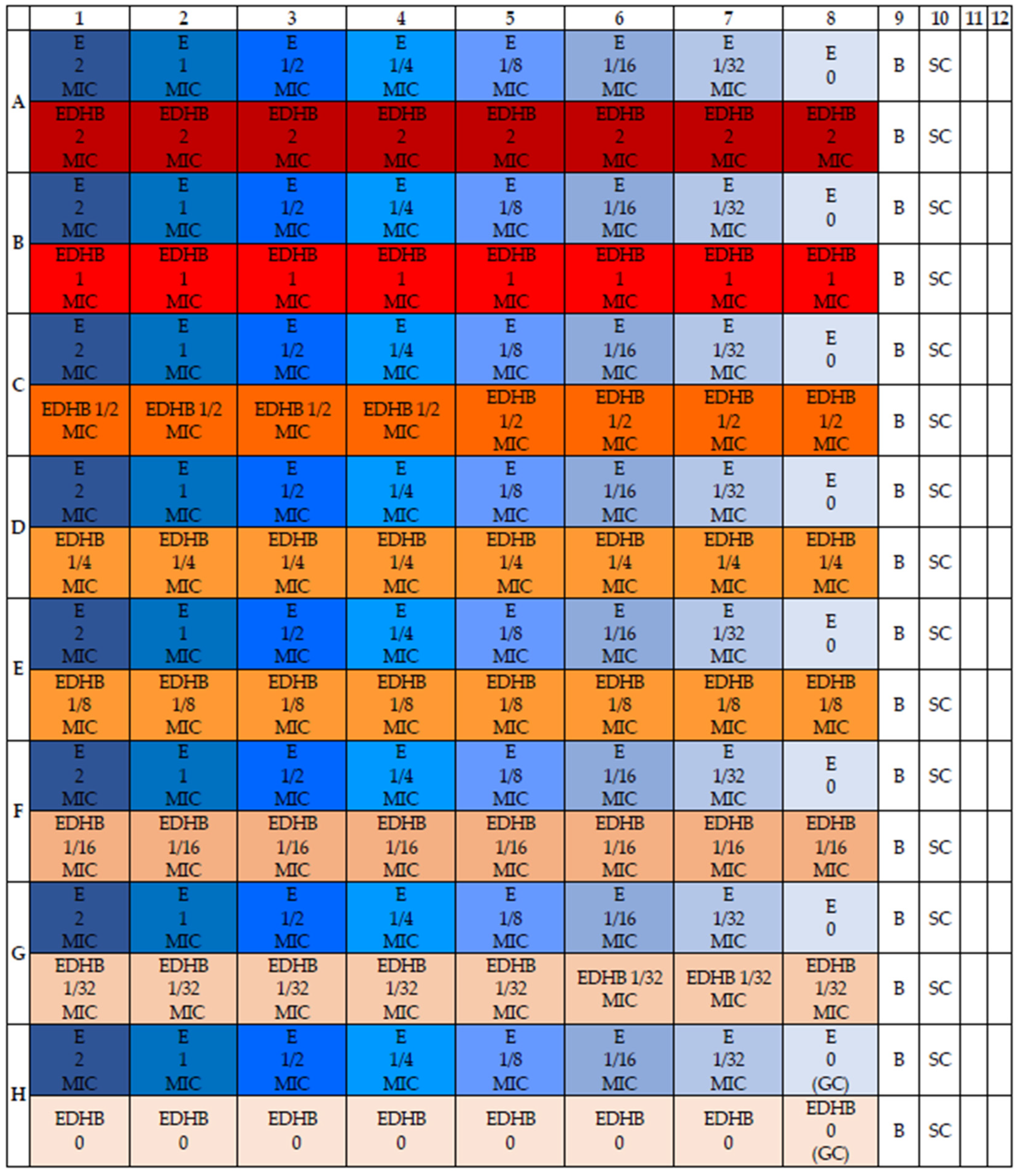
4.4. Determination of the Susceptibility of Staphylococcal Strains to the Erythromycin and EDHB Combination
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mazur, P.; Skiba-Kurek, I.; Mrowiec, P.; Karczewska, E.; Drożdż, R. Synergistic Ros-Associated Antimicrobial Activity of Silver Nanoparticles and Gentamicin against Staphylococcus epidermidis. Int. J. Nanomed. 2020, 15, 3551–3562. [Google Scholar] [CrossRef] [PubMed]
- Miklasińska, M.; Kepa, M.; Wojtyczka, R.D.; Idzik, D.; Zdebik, A.; Orlewska, K.; Wasik, T.J. Antibacterial Activity of Protocatechuic Acid Ethyl Ester on Staphylococcus aureus Clinical Strains Alone and in Combination with Antistaphylococcal Drugs. Molecules 2015, 20, 13536–13549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kȩpa, M.; Miklasińska-Majdanik, M.; Wojtyczka, R.D.; Idzik, D.; Korzeniowski, K.; Smoleń-Dzirba, J.; Wasik, T.J. Antimicrobial Potential of Caffeic Acid against Staphylococcus aureus Clinical Strains. Biomed Res. Int. 2018, 2018, 7413504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Atki, Y.; Aouam, I.; El Kamari, F.; Taroq, A.; Nayme, K.; Timinouni, M.; Lyoussi, B.; Abdellaoui, A. Antibacterial Activity of Cinnamon Essential Oils and Their Synergistic Potential with Antibiotics. J. Adv. Pharm. Technol. Res. 2019, 10, 63–67. [Google Scholar] [CrossRef]
- Ghassemi, A.; Farhangi, H.; Badiee, Z.; Banihashem, A.; Mosaddegh, M.R. Evaluation of Nosocomial Infection in Patients at Hematology-Oncology Ward of Dr. Sheikh Children’s Hospital. Iran. J. Pediatr. Hematol. Oncol. 2015, 5, 179–185. [Google Scholar]
- Vuong, C.; Otto, M. Staphylococcus epidermidis infections. Microbes Infect. 2002, 4, 481–489. [Google Scholar] [CrossRef]
- Wojtyczka, R.D.; Kȩpa, M.; Idzik, D.; Kubina, R.; Kabała-Dzik, A.; Dziedzic, A.; Wąsik, T.J. In Vitro Antimicrobial Activity of Ethanolic Extract of Polish Propolis against Biofilm Forming Staphylococcus epidermidis Strains. Evid. Based Complement. Altern. Med. 2013, 2013, 590703. [Google Scholar] [CrossRef] [Green Version]
- European Centre for Disease Prevention and Control. Multidrug-Resistant Staphylococcus Epidermidis; ECDC: Stockholm, Sweden, 2018; pp. 1–6.
- European Centre for Disease Prevention and Control. Healthcare-Associated Infections: Surgical Site Infections—Annual Epidemiological Report for 2016; ECDC: Stockholm, Sweden, 2018; pp. 1–15.
- Gadisa, E.; Weldearegay, G.; Desta, K.; Tsegaye, G.; Hailu, S.; Jote, K.; Takele, A. Combined Antibacterial Effect of Essential Oils from Three Most Commonly Used Ethiopian Traditional Medicinal Plants on Multidrug Resistant Bacteria. BMC Complement. Altern. Med. 2019, 19, 24. [Google Scholar] [CrossRef]
- Wojtyczka, R.D.; Dziedzic, A.; Kȩpa, M.; Kubina, R.; Kabała-Dzik, A.; Mularz, T.; Idzik, D. Berberine enhances the antibacterial activity of selected antibiotics against coagulase-negative staphylococcus strains in vitro. Molecules 2014, 19, 6583–6596. [Google Scholar] [CrossRef]
- Kyaw, B.M.; Arora, S.; Lim, C.S. Bactericidal Antibiotic-Phytochemical Combinations against Methicillin Resistant Staphylococcus aureus. Braz. J. Microbiol. 2012, 43, 938–945. [Google Scholar] [CrossRef] [Green Version]
- Institute of Medicine (US) Forum on Microbial Threats. Antibiotic Resistance: Implications for Global Health and Novel Intervention Strategies: Workshop Summary; National Academies Press: Washington, DC, USA, 2010; pp. 3–17. [Google Scholar]
- Semaming, Y.; Pannengpetch, P.; Chattipakorn, S.C.; Chattipakorn, N. Pharmacological Properties of Protocatechuic Acid and Its Potential Roles as Complementary Medicine. Evid. Based Complement. Altern. Med. 2015, 2015, 593902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kakkar, S.; Bais, S. A Review on Protocatechuic Acid and Its Pharmacological Potential. ISRN Pharmacol. 2014, 2014, 952943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, A.K.; Rashid, R.; Fatima, N.; Mahmood, S.; Mir, S.; Khan, S.; Jabeen, N.; Murtaza, G. Pharmacological Activities of Protocatechuic Acid. Acta Pol. Pharm. 2015, 72, 643–650. [Google Scholar] [PubMed]
- Abouaitah, K.; Piotrowska, U.; Wojnarowicz, J.; Swiderska-Sroda, A.; El-Desoky, A.H.H.; Lojkowski, W. Enhanced Activity and Sustained Release of Protocatechuic Acid, a Natural Antibacterial Agent, from Hybrid Nanoformulations with Zinc Oxide Nanoparticles. Int. J. Mol. Sci. 2021, 22, 5287. [Google Scholar] [CrossRef]
- Torrisi, C.; Malfa, G.A.; Acquaviva, R.; Castelli, F.; Sarpietro, M.G. Effect of Protocatechuic Acid Ethyl Ester on Biomembrane Models: Multilamellar Vesicles and Monolayers. Membranes 2022, 12, 283. [Google Scholar] [CrossRef]
- Reis, B.; Martins, M.; Barreto, B.; Milhazes, N.; Garrido, E.M.; Silva, P.; Garrido, J.; Borges, F. Structure—Property—Activity Relationship of Phenolic Acids and Derivatives. Protocatechuic Acid Alkyl Esters. J. Agric. Food Chem. 2010, 58, 6986–6993. [Google Scholar] [CrossRef]
- Li, W.; Hou, G.; Zhou, D.; Lou, X.; Xu, Y.; Liu, S.; Zhao, X. The Roles of AKR1C1 and AKR1C2 in Ethyl-3,4-Dihydroxybenzoateinduced Esophageal Squamous Cell Carcinoma Cell Death. Oncotarget 2016, 7, 21542–21555. [Google Scholar] [CrossRef]
- Han, B.; Li, W.; Sun, Y.; Zhou, L.; Xu, Y.; Zhao, X. A Prolyl-Hydroxylase Inhibitor, Ethyl-3,4-Dihydroxybenzoate, Induces Cell Autophagy and Apoptosis in Esophageal Squamous Cell Carcinoma Cellsvia up-Regulation of BNIP3 and N-Myc Downstream-Regulated Gene-1. PLoS ONE 2014, 9, e107204. [Google Scholar] [CrossRef]
- Nimker, C.; Kaur, G.; Revo, A.; Chaudhary, P.; Bansal, A. Ethyl 3,4-Dihydroxy Benzoate, a Unique Preconditioning Agent for Alleviating Hypoxia-Mediated Oxidative Damage in L6 Myoblasts Cells. J. Physiol. Sci. 2015, 65, 77–87. [Google Scholar] [CrossRef]
- Merkl, R.; Hrádková, I.; Filip, V.; Šmidrkal, J. Antimicrobial and Antioxidant Properties of Phenolic Acids Alkyl Esters. Czech. J. Food Sci. 2010, 28, 275–279. [Google Scholar] [CrossRef] [Green Version]
- Philipp, S.; Cui, L.; Ludolph, B.; Kelm, M.; Schulz, R.; Cohen, M.V.; Downey, J.M. Desferoxamine and Ethyl-3,4-Dihydroxybenzoate Protect Myocardium by Activating NOS and Generating Mitochondrial ROS. Am. J. Physiol. Hear. Circ. Physiol. 2006, 290, 450–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nandan, D.; Clarke, E.P.; Ball, E.H.; Sanwal, B.D. Ethyl-3,4-Dihydroxybenzoate Inhibits Myoblast Differentiation: Evidence for an Essential Role of Collagen. J. Cell Biol. 1990, 110, 1673–1679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Huo, X.; Chen, H.; Li, B.; Liu, J.; Ma, W.; Wang, X.; Xie, K.; Yu, Y.; Shi, K. Hydrogen-Rich Saline Activated Autophagy via HIF-1 α Pathways in Neuropathic Pain Model. Biomed. Res. Int. 2018, 2018, 4670834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, C.Y.; Yin, M.C. Antibacterial effects of roselle calyx extracts and protocatechuic acid in ground beef and apple juice. Foodborne Pathog. Dis. 2009, 6, 201–206. [Google Scholar] [CrossRef] [Green Version]
- Stojković, D.S.; Zivković, J.; Soković, M.; Glamočlija, J.; Ferreira, I.C.; Janković, T.; Maksimović, Z. Antibacterial activity of Veronica montana L. extract and of protocatechuic acid incorporated in a food system. Food Chem. Toxicol. 2013, 55, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Bassolé, I.H.N.; Juliani, H.R. Essential oils in combination and their antimicrobial properties. Molecules 2012, 17, 3989–4006. [Google Scholar] [CrossRef] [Green Version]
- Fratini, F.; Mancini, S.; Turchi, B.; Friscia, E.; Pistelli, L.; Giusti, G.; Cerri, D. A novel interpretation of the Fractional Inhibitory Concentration Index: The case Origanum vulgare L. and Leptospermum scoparium JR et G. Forst essential oils against Staphylococcus aureus strains. Microbiol. Res. 2017, 195, 11–17. [Google Scholar] [CrossRef]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Terminology relating to methods for the determination of susceptibility of bacteria to antimicrobial agents. Clin. Microbiol. Infect. 2000, 6, 503–508. [Google Scholar] [CrossRef] [Green Version]
- Mostafa, I.; Abbas, H.A.; Ashour, M.L.; Yasri, A.; El-Shazly, A.M.; Wink, M.; Sobeh, M. Polyphenols from Salix Tetrasperma Impair Virulence and Inhibit Quorum sensing of Pseudomonas Aeruginosa. Molecules 2020, 25, 1341. [Google Scholar] [CrossRef] [Green Version]
- Bernal-Mercado, A.T.; Vazquez-Armenta, F.J.; Tapia-Rodriguez, M.R.; Islas-Osuna, M.A.; Mata-Haro, V.; Gonzalez-Aguilar, G.A.; Lopez-Zavala, A.A.; Ayala-Zavala, J.F. Comparison of Single and Combined Use of Catechin, Protocatechuic, and Vanillic Acids as Antioxidant and Antibacterial Agents against Uropathogenic Escherichia Coli at Planktonic and Biofilm Levels. Molecules 2018, 23, 2813. [Google Scholar] [CrossRef] [Green Version]
- Bernal-Mercado, A.T.; Gutierrez-Pacheco, M.M.; Encinas-Basurto, D.; Mata-Haro, V.; Lopez-Zavala, A.A.; Islas-Osuna, M.A.; Gonzalez-Aguilar, G.A.; Ayala-Zavala, J.F. Synergistic mode of action of catechin, vanillic and protocatechuic acids to inhibit the adhesion of uropathogenic Escherichia coli on silicone surfaces. J. Appl. Microbiol. 2019, 128, 387–400. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, N.; Tiwari, S.; Bhandari, K.; Biswal, A.K.; Rawat, A.K.S. Novel derivatives of plant monomeric phenolics: Act as inhibitors of bacterial cell-to-cell communication. Microb. Pathog. 2020, 141, 103856. [Google Scholar] [CrossRef] [PubMed]
- Chai, B.; Jiang, W.; Hu, M.; Wu, Y.; Si, H. In vitro synergistic interactions of Protocatechuic acid and Chlorogenic acid in combination with antibiotics against animal pathogens. Synergy 2019, 9, 6–11. [Google Scholar] [CrossRef]
- Sanhueza, L.; Melo, R.; Montero, R.; Maisey, K.; Mendoza, L.; Wilkens, M. Synergistic interactions between phenolic compounds identified in grape pomace extract with antibiotics of different classes against Staphylococcus aureus and Escherichia coli. PLoS ONE 2017, 12, e0172273. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Bisignano, C.; D’Arrigo, M.; Ginestra, G.; Arena, A.; Tomaino, A.; Wickham, M.S. Antimicrobial potential of polyphenols extracted from almond skins. Lett. Appl. Microbiol. 2010, 51, 83–89. [Google Scholar] [CrossRef]
- Wang, L.H.; Zeng, X.A.; Wang, M.S.; Brennan, C.S.; Gong, D. Modification of Membrane Properties and Fatty Acids Biosynthesis-Related Genes in Escherichia Coli and Staphylococcus Aureus: Implications for the Antibacterial Mechanism of Naringenin. Biochim. Biophys. Acta Biomembr. 2018, 1860, 481–490. [Google Scholar] [CrossRef]
- Murakami, K.; Minamide, W.; Wada, K.; Nakamura, E.; Teraoka, H.; Watanabe, S. Identification of methicillin-resistant strains of Staphylococci by polymerase chain reaction. J. Clin. Microbiol. 1991, 29, 2240–2244. [Google Scholar] [CrossRef] [Green Version]
- Amsterdam, D. Susceptibility Testing of Antimicrobials in Liquid Media. In Antibiotics in Laboratory Medicine, 5th ed.; Loman, V., Ed.; Williams and Wilkins: Philadelphia, PA, USA, 2005; pp. 61–143. [Google Scholar]
- Devienne, K.F.; Raddi, M.S.G. Screening for antimicrobial activity of natural Products using a microplate photometer. Braz. J. Microbiol. 2002, 33, 166–168. [Google Scholar] [CrossRef] [Green Version]
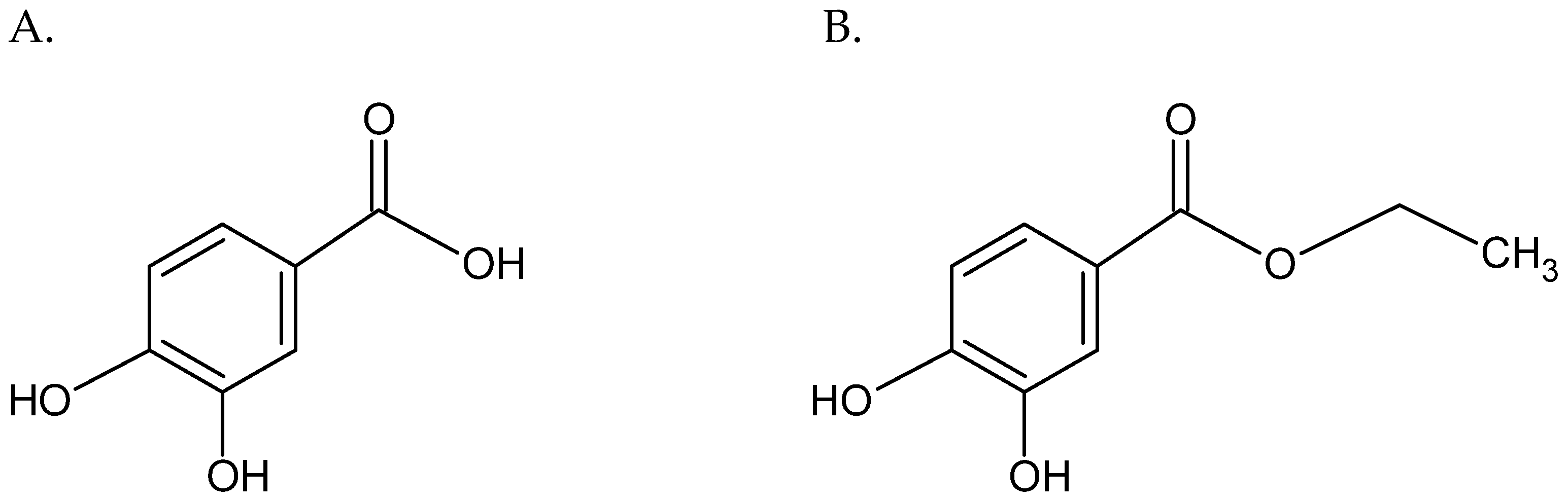
| Strain | Methicillin Resistance Profile | MLSB Resistance Profile |
|---|---|---|
| S. aureus ATCC 25923 | MSSA | - |
| S. aureus ATCC 43300 | MRSA | cMLSB |
| S. epidermidis ATCC 12228 | MSSE | - |
| S. epidermidis ATCC 35984 | MRSE | cMLSB |
| S. aureus 1 | MSSA | - |
| S. aureus 2 | MSSA | - |
| S. aureus 3 | MRSA | cMLSB |
| S. aureus 4 | MRSA | cMLSB |
| S. epidermidis 1 | MRSE | cMLSB |
| S. epidermidis 2 | MRSE | cMLSB |
| S. epidermidis 3 | MSSE | - |
| S. epidermidis 4 | MSSE | - |
| Staphylococcal Strain | MIC Values | |
|---|---|---|
| EDHB | Erythromycin | |
| S. aureus ATCC 25923 | 512 | 0.25 |
| S. aureus ATCC 43300 | 512 | 2048 |
| S. aureus 1 | 16 | 0.25 |
| S. aureus 2 | 16 | 0.25 |
| S. aureus 3 | 1024 | 2048 |
| S. aureus 4 | 1024 | 2048 |
| S. epidermidis ATCC 12228 | 1024 | 0.125 |
| S. epidermidis ATCC 35984 | 512 | 2048 |
| S. epidermidis 1 | 512 | 2048 |
| S. epidermidis 2 | 512 | 2048 |
| S. epidermidis 3 | 512 | 0.125 |
| S. epidermidis 4 | 512 | 2 |
| Strain | FIC Index | Interacion |
|---|---|---|
| S. aureus ATCC 25923 | 1.031 | indifference |
| S. aureus ATCC 43300 | 1.016 | indifference |
| S. epidermidis ATCC 12228 | 0.628 | additive |
| S. epidermidis ATCC 35984 | 1.063 | indifference |
| S. aureus 1 | 2 | indifference |
| S. aureus 2 | 1.125 | indifference |
| S. aureus 3 | 0.078 | synergism |
| S. aureus 4 | 1.016 | indifference |
| S. epidermidis 1 | 1.015 | indifference |
| S. epidermidis 2 | 1.015 | indifference |
| S. epidermidis 3 | 0.750 | additive |
| S. epidermidis 4 | 0.281 | synergism |
| Strain | MIC of EDHB | EDHB Concentration in Well with FIC Index | MIC of Erythromycin | MIC of Erythromycin with EDHB | Decrease of the MIC Value after the EDHB Addition [%] |
|---|---|---|---|---|---|
| S. aureus ATCC 25923 | 512 | 16 | 0.25 | 0.25 | 0 |
| S. aureus ATCC 43300 | 512 | 512 | 2048 | 32 | 98.44 |
| S. epidermidis ATCC 12228 | 1024 | 512 | 0.125 | 0.016 | 12.8 |
| S. epidermidis ATCC 35984 | 512 | 32 | 2048 | 1 | 99.95 |
| S. aureus 1 | 16 | 16 | 0.25 | 0.25 | 0 |
| S. aureus 2 | 16 | 16 | 0.25 | 0.03125 | 87.5 |
| S. aureus 3 | 1024 | 64 | 2048 | 32 | 98.44 |
| S. aureus 4 | 1024 | 1024 | 2048 | 32 | 98.44 |
| S. epidermidis 1 | 512 | 512 | 2048 | 1 | 99.95 |
| S. epidermidis 2 | 512 | 512 | 2048 | 32 | 99.95 |
| S. epidermidis 3 | 512 | 128 | 0.125 | 0.063 | 50.4 |
| S. epidermidis 4 | 512 | 32 | 2 | 0.5 | 75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miklasińska-Majdanik, M.; Kępa, M.; Kulczak, M.; Ochwat, M.; Wąsik, T.J. The Array of Antibacterial Action of Protocatechuic Acid Ethyl Ester and Erythromycin on Staphylococcal Strains. Antibiotics 2022, 11, 848. https://doi.org/10.3390/antibiotics11070848
Miklasińska-Majdanik M, Kępa M, Kulczak M, Ochwat M, Wąsik TJ. The Array of Antibacterial Action of Protocatechuic Acid Ethyl Ester and Erythromycin on Staphylococcal Strains. Antibiotics. 2022; 11(7):848. https://doi.org/10.3390/antibiotics11070848
Chicago/Turabian StyleMiklasińska-Majdanik, Maria, Małgorzata Kępa, Monika Kulczak, Maciej Ochwat, and Tomasz J. Wąsik. 2022. "The Array of Antibacterial Action of Protocatechuic Acid Ethyl Ester and Erythromycin on Staphylococcal Strains" Antibiotics 11, no. 7: 848. https://doi.org/10.3390/antibiotics11070848
APA StyleMiklasińska-Majdanik, M., Kępa, M., Kulczak, M., Ochwat, M., & Wąsik, T. J. (2022). The Array of Antibacterial Action of Protocatechuic Acid Ethyl Ester and Erythromycin on Staphylococcal Strains. Antibiotics, 11(7), 848. https://doi.org/10.3390/antibiotics11070848







