The Efficacy of Using Combination Therapy against Multi-Drug and Extensively Drug-Resistant Pseudomonas aeruginosa in Clinical Settings
Abstract
:1. Introduction
2. Results and Discussion
| Study No. | Treatment | Number of Patients Treated [n (%)] | Number of Patients with P. aeruginosa | Resistance | Microbiological Cure [n (%)] | Mortality [n (%)] | References | |
|---|---|---|---|---|---|---|---|---|
| Sensitive Strains [n (%)] | Resistant Strains [n (%)] | |||||||
| 8 | Colistin monotherapy | 12 (17.6) | 135 (52.3) ** | 123 (47.7) ** | MDR | - | 3 (25.0) * | [35] |
| Colistin–meropenem | 28 (41.2) | - | 4 (14.3) * | |||||
| Colistin–piperacillin–tazobactam | 10 (14.7) | - | 4 (40) * | |||||
| Colistin–ampicillin–sulbactam | 1 (1.5) | - | 0 (0) * | |||||
| Colistin with other agents I | 17 (25) | - | 6 (25.3) * | |||||
| 11 | β-lactam with aminoglycoside–fluoroquinolone | 28 (31.5) | 75 (77.3) 2 | 22 (23) 3 | XDR | - | 7 (25) | [38] |
| Colistin with other agents | 7 (7.9) | - | 4 (57.1) | |||||
| β-lactam with β-lactam | 2 (2.2) | - | 1 (50) | |||||
| β-lactam | 26 | - | 10 (38.5) | |||||
| Colistin | 8 | - | 3 (37.5) | |||||
| Fluoroquinolone | 11 | - | 1 (9.1) | |||||
| 12 | Colistin | 4 (14.7) | 14 (44.8) | 18 (56.2) 1 | MDR/XDR | 6 (35.3) | - | [39] |
| β-lactam | 15 (44.1) | - | ||||||
| Colistin with β-lactam | 13 (38.2) | 11 (64.7) | - | |||||
| Amikacin with β-lactam | 2 (5.9) | - | ||||||
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.-J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Subedi, D.; Vijay, A.K.; Willcox, M. Overview of mechanisms of antibiotic resistance in Pseudomonas aeruginosa: An ocular perspective. Clin. Exp. Optom. 2018, 101, 162–171. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, M.; Anju, C.P.; Biswas, L.; Anil Kumar, V.; Gopi Mohan, C.; Biswas, R. Antibiotic resistance in Pseudomonas aeruginosa and alternative therapeutic options. Int. J. Med. Microbiol. 2016, 306, 48–58. [Google Scholar] [CrossRef]
- Terahara, F.; Nishiura, H. Carbapenem-resistant Pseudomonas aeruginosa and carbapenem use in Japan: An ecological study. J. Int. Med. Res. 2019, 47, 4711–4722. [Google Scholar] [CrossRef] [Green Version]
- Papp-Wallace, K.M.; Endimiani, A.; Taracila, M.A.; Bonomo, R.A. Carbapenems: Past, Present, and Future. Antimicrob. Agents Chemother. 2011, 55, 4943–4960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Martínez, J.-M.; Poirel, L.; Nordmann, P. Molecular Epidemiology and Mechanisms of Carbapenem Resistance in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2009, 53, 4783–4788. [Google Scholar] [CrossRef] [Green Version]
- Tran, H.A.; Vu, T.N.B.; Trinh, S.T.; Tran, D.L.; Pham, H.M.; Ngo, T.H.H.; Nguyen, M.T.; Tran, N.D.; Pham, D.T.; Dang, D.A.; et al. Resistance mechanisms and genetic relatedness among carbapenem-resistant Pseudomonas aeruginosa isolates from three major hospitals in Hanoi, Vietnam (2011–2015). JAC-Antimicrob. Resist. 2021, 3, dlab103. [Google Scholar] [CrossRef]
- Gutiérrez, O.; Juan, C.; Cercenado, E.; Navarro, F.; Bouza, E.; Coll, P.; Pérez, J.L.; Oliver, A. Molecular Epidemiology and Mechanisms of Carbapenem Resistance in Pseudomonas aeruginosa Isolates from Spanish Hospitals. Antimicrob. Agents Chemother. 2007, 51, 4329–4335. [Google Scholar] [CrossRef] [Green Version]
- Poole, K. Pseudomonas aeruginosa: Resistance to the max. Front. Microbiol. 2011, 2, 65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rehman, A.; Patrick, W.M.; Lamont, I.L. Mechanisms of ciprofloxacin resistance in Pseudomonas aeruginosa: New approaches to an old problem. J. Med. Microbiol. 2019, 68, 1–10. [Google Scholar] [CrossRef]
- Dean, C.R.; Visalli, M.A.; Projan, S.J.; Sum, P.-E.; Bradford, P.A. Efflux-Mediated Resistance to Tigecycline (GAR-936) in Pseudomonas aeruginosa PAO1. Antimicrob. Agents Chemother. 2003, 47, 972–978. [Google Scholar] [CrossRef] [Green Version]
- Tai, A.S.; Bell, S.C.; Kidd, T.J.; Trembizki, E.; Buckley, C.; Ramsay, K.A.; David, M.; Wainwright, C.E.; Grimwood, K.; Whiley, D.M. Genotypic Diversity within a Single Pseudomonas aeruginosa Strain Commonly Shared by Australian Patients with Cystic Fibrosis. PLoS ONE 2015, 10, e0144022. [Google Scholar] [CrossRef] [PubMed]
- El-Gamal, M.I.; Brahim, I.; Hisham, N.; Aladdin, R.; Mohammed, H.; Bahaaeldin, A. Recent updates of carbapenem antibiotics. Eur. J. Med. Chem. 2017, 131, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Honoré, N.; Nicolas, M.H.; Cole, S.T. Regulation of enterobacterial cephalosporinase production: The role of a membrane-bound sensory transducer. Mol. Microbiol. 1989, 3, 1121–1130. [Google Scholar] [CrossRef]
- Kung, V.L.; Ozer, E.A.; Hauser, A.R. The Accessory Genome of Pseudomonas aeruginosa. Microbiol. Mol. Biol. Rev. 2010, 74, 621–641. [Google Scholar] [CrossRef] [Green Version]
- Botelho, J.; Grosso, F.; Peixe, L. Antibiotic resistance in Pseudomonas aeruginosa—Mechanisms, epidemiology and evolution. Drug Resist. Updates 2019, 44, 100640. [Google Scholar] [CrossRef]
- Ranjan, N.; Chaudhary, U.; Chaudhry, D.; Ranjan, K.P. Ventilator-associated pneumonia in a tertiary care intensive care unit: Analysis of incidence, risk factors and mortality. Indian J. Crit. Care Med. 2014, 18, 200–204. [Google Scholar] [CrossRef]
- Esposito, S.; Leone, S. Antimicrobial treatment for Intensive Care Unit (ICU) infections including the role of the infectious disease specialist. Int. J. Antimicrob. Agents 2007, 29, 494–500. [Google Scholar] [CrossRef]
- Asokan, G.V.; Ramadhan, T.; Ahmed, E.; Sanad, H. WHO Global Priority Pathogens List: A Bibliometric Analysis of Medline-PubMed for Knowledge Mobilization to Infection Prevention and Control Practices in Bahrain. Oman Med. J. 2019, 34, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Breidenstein, E.B.M.; de la Fuente-Núñez, C.; Hancock, R.E.W. Pseudomonas aeruginosa: All roads lead to resistance. Trends Microbiol. 2011, 19, 419–426. [Google Scholar] [CrossRef]
- Pachori, P.; Gothalwal, R.; Gandhi, P. Emergence of antibiotic resistance Pseudomonas aeruginosa in intensive care unit; a critical review. Genes Dis. 2019, 6, 109–119. [Google Scholar] [CrossRef]
- Breilh, D.; Texier-Maugein, J.; Allaouchiche, B.; Saux, M.-C.; Boselli, E. Carbapenems. J. Chemother. 2013, 25, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.; Garcia, J.; Gruenberg, K.; MacDougall, C. Multidrug-Resistant Pseudomonas Infections: Hard to Treat, But Hope on the Horizon? Curr. Infect. Dis. Rep. 2018, 20, 23. [Google Scholar] [CrossRef]
- Barnes, M.D.; Taracila, M.A.; Rutter, J.D.; Bethel, C.R.; Galdadas, I.; Hujer, A.M.; Caselli, E.; Prati, F.; Dekker, J.P.; Papp-Wallace, K.M.; et al. Deciphering the Evolution of Cephalosporin Resistance to Ceftolozane-Tazobactam in Pseudomonas aeruginosa. mBio 2018, 9, e02085-18. [Google Scholar] [CrossRef] [Green Version]
- Tamma, P.D.; Cosgrove, S.E.; Maragakis, L.L. Combination Therapy for Treatment of Infections with Gram-Negative Bacteria. Clin. Microbiol. Rev. 2012, 25, 450–470. [Google Scholar] [CrossRef] [Green Version]
- Tsuji, B.T.; Pogue, J.M.; Zavascki, A.P.; Paul, M.; Daikos, G.L.; Forrest, A.; Giacobbe, D.R.; Viscoli, C.; Giamarellou, H.; Karaiskos, I.; et al. International Consensus Guidelines for the Optimal Use of the Polymyxins: Endorsed by the American College of Clinical Pharmacy (ACCP), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Infectious Diseases Society of America (IDSA), International Society for Anti-infective Pharmacology (ISAP), Society of Critical Care Medicine (SCCM), and Society of Infectious Diseases Pharmacists (SIDP). Pharmacotherapy 2019, 39, 10–39. [Google Scholar] [CrossRef]
- Zhanel, G.G.; Lawson, C.D.; Adam, H.; Schweizer, F.; Zelenitsky, S.; Lagacé-Wiens, P.R.S.; Denisuik, A.; Rubinstein, E.; Gin, A.S.; Hoban, D.J.; et al. Ceftazidime-Avibactam: A Novel Cephalosporin/β-lactamase Inhibitor Combination. Drugs 2013, 73, 159–177. [Google Scholar] [CrossRef] [Green Version]
- Crusio, R.; Rao, S.; Changawala, N.; Paul, V.; Tiu, C.; van Ginkel, J.; Chapnick, E.; Kupfer, Y. Epidemiology and outcome of infections with carbapenem-resistant Gram-negative bacteria treated with polymyxin B-based combination therapy. Scand. J. Infect. Dis. 2014, 46, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rigatto, M.H.; Vieira, F.J.; Antochevis, L.C.; Behle, T.F.; Lopes, N.T.; Zavascki, A.P. Polymyxin B in Combination with Antimicrobials Lacking In Vitro Activity versus Polymyxin B in Monotherapy in Critically Ill Patients with Acinetobacter baumannii or Pseudomonas aeruginosa Infections. Antimicrob. Agents Chemother. 2015, 59, 6575–6580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deconinck, L.; Meybeck, A.; Patoz, P.; Grunderbeeck, N.V.; Boussekey, N.; Chiche, A.; Delannoy, P.-Y.; Georges, H.; Leroy, O. Impact of combination therapy and early de-escalation on outcome of ventilator-associated pneumonia caused by Pseudomonas aeruginosa. Infect. Dis. 2017, 49, 396–404. [Google Scholar] [CrossRef]
- Khawcharoenporn, T.; Chuncharunee, A.; Maluangnon, C.; Taweesakulvashra, T.; Tiamsak, P. Active monotherapy and combination therapy for extensively drug-resistant Pseudomonas aeruginosa pneumonia. Int. J. Antimicrob. Agents 2018, 52, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Park, B.K.; Kim, S.K.; Han, S.B.; Lee, J.W.; Lee, D.-G.; Chung, N.-G.; Cho, B.; Jeong, D.C.; Kang, J.H. Clinical characteristics and outcomes of Pseudomonas aeruginosa bacteremia in febrile neutropenic children and adolescents with the impact of antibiotic resistance: A retrospective study. BMC Infect. Dis. 2017, 17, 500. [Google Scholar] [CrossRef] [Green Version]
- Bassetti, M.; Echols, R.; Matsunaga, Y.; Ariyasu, M.; Doi, Y.; Ferrer, R.; Lodise, T.P.; Naas, T.; Niki, Y.; Paterson, D.L.; et al. Efficacy and safety of cefiderocol or best available therapy for the treatment of serious infections caused by carbapenem-resistant Gram-negative bacteria (CREDIBLE-CR): A randomised, open-label, multicentre, pathogen-focused, descriptive, phase 3 trial. Lancet Infect. Dis. 2021, 21, 226–240. [Google Scholar] [CrossRef]
- Bosaeed, M.; Ahmad, A.; Alali, A.; Mahmoud, E.; Alswidan, L.; Alsaedy, A.; Aljuhani, S.; Alalwan, B.; Alshamrani, M.; Alothman, A. Experience With Ceftolozane-Tazobactam for the Treatment of Serious Pseudomonas aeruginosa Infections in Saudi Tertiary Care Center. Infect. Dis. Res. Treat. 2020, 13, 1178633720905977. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Rafailidis, P.I.; Ioannidou, E.; Alexiou, V.G.; Matthaiou, D.K.; Karageorgopoulos, D.E.; Kapaskelis, A.; Nikita, D.; Michalopoulos, A. Colistin therapy for microbiologically documented multidrug-resistant Gram-negative bacterial infections: A retrospective cohort study of 258 patients. Int. J. Antimicrob. Agents 2010, 35, 194–199. [Google Scholar] [CrossRef] [Green Version]
- Paul, M.; Daikos, G.L.; Durante-Mangoni, E.; Yahav, D.; Carmeli, Y.; Benattar, Y.D.; Skiada, A.; Andini, R.; Eliakim-Raz, N.; Nutman, A.; et al. Colistin alone versus colistin plus meropenem for treatment of severe infections caused by carbapenem-resistant Gram-negative bacteria: An open-label, randomised controlled trial. Lancet Infect. Dis. 2018, 18, 391–400. [Google Scholar] [CrossRef]
- Furtado, G.H.C.; d’Azevedo, P.A.; Santos, A.F.; Gales, A.C.; Pignatari, A.C.C.; Medeiros, E.A.S. Intravenous polymyxin B for the treatment of nosocomial pneumonia caused by multidrug-resistant Pseudomonas aeruginosa. Int. J. Antimicrob. Agents 2007, 30, 315–319. [Google Scholar] [CrossRef]
- Samonis, G.; Vardakas, K.Z.; Kofteridis, D.P.; Dimopoulou, D.; Andrianaki, A.M.; Chatzinikolaou, I.; Katsanevaki, E.; Maraki, S.; Falagas, M.E. Characteristics, risk factors and outcomes of adult cancer patients with extensively drug-resistant Pseudomonas aeruginosa infections. Infection 2014, 42, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Ribera, A.; Benavent, E.; Lora-Tamayo, J.; Tubau, F.; Pedrero, S.; Cabo, X.; Ariza, J.; Murillo, O. Osteoarticular infection caused by MDR Pseudomonas aeruginosa: The benefits of combination therapy with colistin plus β-lactams. J. Antimicrob. Chemother. 2015, 70, 3357–3365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zusman, O.; Avni, T.; Leibovici, L.; Adler, A.; Friberg, L.; Stergiopoulou, T.; Carmeli, Y.; Paul, M. Systematic Review and Meta-Analysis of In Vitro Synergy of Polymyxins and Carbapenems. Antimicrob. Agents Chemother. 2013, 57, 5104–5111. [Google Scholar] [CrossRef] [Green Version]
- Montero, M.M.; Domene Ochoa, S.; López-Causapé, C.; VanScoy, B.; Luque, S.; Sorlí, L.; Campillo, N.; Padilla, E.; Prim, N.; Segura, C.; et al. Colistin plus meropenem combination is synergistic in vitro against extensively drug-resistant Pseudomonas aeruginosa, including high-risk clones. J. Glob. Antimicrob. Resist. 2019, 18, 37–44. [Google Scholar] [CrossRef]
- Martis, N.; Leroy, S.; Blanc, V. Colistin in multi-drug resistant Pseudomonas aeruginosa blood-stream infections: A narrative review for the clinician. J. Infect. 2014, 69, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Portsmouth, S.; van Veenhuyzen, D.; Echols, R.; Machida, M.; Ferreira, J.C.A.; Ariyasu, M.; Tenke, P.; Nagata, T.D. Cefiderocol versus imipenem-cilastatin for the treatment of complicated urinary tract infections caused by Gram-negative uropathogens: A phase 2, randomised, double-blind, non-inferiority trial. Lancet Infect. Dis. 2018, 18, 1319–1328. [Google Scholar] [CrossRef]
- Vazquez, J.A.; Patzán, L.D.G.; Stricklin, D.; Duttaroy, D.D.; Kreidly, Z.; Lipka, J.; Sable, C. Efficacy and safety of ceftazidime–avibactam versus imipenem–cilastatin in the treatment of complicated urinary tract infections, including acute pyelonephritis, in hospitalized adults: Results of a prospective, investigator-blinded, randomized study. Curr. Med. Res. Opin. 2012, 28, 1921–1931. [Google Scholar] [CrossRef]
- Qin, X.; Tran, B.G.; Kim, M.J.; Wang, L.; Nguyen, D.A.; Chen, Q.; Song, J.; Laud, P.J.; Stone, G.G.; Chow, J.W. A randomised, double-blind, phase 3 study comparing the efficacy and safety of ceftazidime/avibactam plus metronidazole versus meropenem for complicated intra-abdominal infections in hospitalised adults in Asia. Int. J. Antimicrob. Agents 2017, 49, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Wagenlehner, F.M.; Sobel, J.D.; Newell, P.; Armstrong, J.; Huang, X.; Stone, G.G.; Yates, K.; Gasink, L.B. Ceftazidime-avibactam Versus Doripenem for the Treatment of Complicated Urinary Tract Infections, Including Acute Pyelonephritis: RECAPTURE, a Phase 3 Randomized Trial Program. Clin. Infect. Dis. 2016, 63, 754–762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazuski, J.E.; Gasink, L.B.; Armstrong, J.; Broadhurst, H.; Stone, G.G.; Rank, D.; Llorens, L.; Newell, P.; Pachl, J. Efficacy and Safety of Ceftazidime-Avibactam Plus Metronidazole Versus Meropenem in the Treatment of Complicated Intra-abdominal Infection: Results From a Randomized, Controlled, Double-Blind, Phase 3 Program. Clin. Infect. Dis. 2016, 62, 1380–1389. [Google Scholar] [CrossRef]
- Mendes, R.E.; Castanheira, M.; Woosley, L.N.; Stone, G.G.; Bradford, P.A.; Flamm, R.K. Characterization of β-Lactamase Content of Ceftazidime-Resistant Pathogens Recovered during the Pathogen-Directed Phase 3 REPRISE Trial for Ceftazidime-Avibactam: Correlation of Efficacy against β-Lactamase Producers. Antimicrob. Agents Chemother. 2019, 63, e02655-18. [Google Scholar] [CrossRef] [Green Version]
- Stone, G.G.; Newell, P.; Gasink, L.B.; Broadhurst, H.; Wardman, A.; Yates, K.; Chen, Z.; Song, J.; Chow, J.W. Clinical activity of ceftazidime/avibactam against MDR Enterobacteriaceae and Pseudomonas aeruginosa: Pooled data from the ceftazidime/avibactam Phase III clinical trial programme. J. Antimicrob. Chemother. 2018, 73, 2519–2523. [Google Scholar] [CrossRef]
- Torres, A.; Zhong, N.; Pachl, J.; Timsit, J.-F.; Kollef, M.; Chen, Z.; Song, J.; Taylor, D.; Laud, P.J.; Stone, G.G.; et al. Ceftazidime-avibactam versus meropenem in nosocomial pneumonia, including ventilator-associated pneumonia (REPROVE): A randomised, double-blind, phase 3 non-inferiority trial. Lancet Infect. Dis. 2018, 18, 285–295. [Google Scholar] [CrossRef] [Green Version]
- Van Duin, D.; Bonomo, R.A. Ceftazidime/Avibactam and Ceftolozane/Tazobactam: Second-generation β-Lactam/β-Lactamase Inhibitor Combinations. Clin. Infect. Dis. 2016, 63, 234–241. [Google Scholar] [CrossRef] [Green Version]
- Falcone, M.; Paterson, D. Spotlight on ceftazidime/avibactam: A new option for MDR Gram-negative infections. J. Antimicrob. Chemother. 2016, 71, 2713–2722. [Google Scholar] [CrossRef] [Green Version]
- Goodlet, K.J.; Nicolau, D.P.; Nailor, M.D. Ceftolozane/tazobactam and ceftazidime/avibactam for the treatment of complicated intra-abdominal infections. Ther. Clin. Risk Manag. 2016, 12, 1811–1826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stone, G.G.; Bradford, P.A.; Newell, P.; Wardman, A. In Vitro Activity of Ceftazidime-Avibactam against Isolates in a Phase 3 Open-Label Clinical Trial for Complicated Intra-Abdominal and Urinary Tract Infections Caused by Ceftazidime-Nonsusceptible Gram-Negative Pathogens. Antimicrob. Agents Chemother. 2017, 61, e01820-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montero, M.M.; Domene Ochoa, S.; López-Causapé, C.; Luque, S.; Sorlí, L.; Campillo, N.; López Montesinos, I.; Padilla, E.; Prim, N.; Angulo-Brunet, A.; et al. Time-Kill Evaluation of Antibiotic Combinations Containing Ceftazidime-Avibactam against Extensively Drug-Resistant Pseudomonas aeruginosa and Their Potential Role against Ceftazidime-Avibactam-Resistant Isolates. Microbiol. Spectr. 2021, 9, e00585-21. [Google Scholar] [CrossRef] [PubMed]
- Onorato, L.; Di Caprio, G.; Signoriello, S.; Coppola, N. Efficacy of ceftazidime/avibactam in monotherapy or combination therapy against carbapenem-resistant Gram-negative bacteria: A meta-analysis. Int. J. Antimicrob. Agents 2019, 54, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Park, T.E.; Moy, S. Ceftazidime-Avibactam: A Novel Cephalosporin/β-Lactamase Inhibitor Combination for the Treatment of Resistant Gram-negative Organisms. Clin. Ther. 2016, 38, 431–444. [Google Scholar] [CrossRef]
- Buehrle, D.J.; Shields, R.K.; Chen, L.; Hao, B.; Press, E.G.; Alkrouk, A.; Potoski, B.A.; Kreiswirth, B.N.; Clancy, C.J.; Nguyen, M.H. Evaluation of the In Vitro Activity of Ceftazidime-Avibactam and Ceftolozane-Tazobactam against Meropenem-Resistant Pseudomonas aeruginosa Isolates. Antimicrob. Agents Chemother. 2016, 60, 3227–3231. [Google Scholar] [CrossRef] [Green Version]
- Wright, H.; Bonomo, R.A.; Paterson, D.L. New agents for the treatment of infections with Gram-negative bacteria: Restoring the miracle or false dawn? Clin. Microbiol. Infect. 2017, 23, 704–712. [Google Scholar] [CrossRef] [Green Version]
- Mikamo, H.; Monden, K.; Miyasaka, Y.; Horiuchi, T.; Fujimoto, G.; Fukuhara, T.; Yoshinari, T.; Rhee, E.G.; Shizuya, T. The efficacy and safety of tazobactam/ceftolozane in combination with metronidazole in Japanese patients with complicated intra-abdominal infections. J. Infect. Chemother. 2019, 25, 111–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mei, Q.; Geng, S.; Fang, X.; He, Y.; Liu, L.; Xu, M.; Zhu, C.; Pan, A. In vitro activity of ceftazidime-avibactam combined with colistin against extensively drug-resistant Pseudomonas aeruginosa. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2019, 31, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.-P.; Lee, W.; Tan, T.-Y.; Sasikala, S.; Teo, J.; Hsu, L.-Y.; Tan, T.-T.; Syahidah, N.; Kwa, A.L. Effective Antibiotics in Combination against Extreme Drug-Resistant Pseudomonas aeruginosa with Decreased Susceptibility to Polymyxin B. PLoS ONE 2011, 6, e28177. [Google Scholar] [CrossRef]
- Morata, L.; Cobos-Trigueros, N.; Martínez, J.A.; Soriano, Á.; Almela, M.; Marco, F.; Sterzik, H.; Núñez, R.; Hernández, C.; Mensa, J. Influence of Multidrug Resistance and Appropriate Empirical Therapy on the 30-Day Mortality Rate of Pseudomonas aeruginosa Bacteremia. Antimicrob. Agents Chemother. 2012, 56, 4833–4837. [Google Scholar] [CrossRef] [Green Version]
- Herrmann, G.; Yang, L.; Wu, H.; Song, Z.; Wang, H.; Høiby, N.; Ulrich, M.; Molin, S.; Riethmüller, J.; Döring, G. Colistin-Tobramycin Combinations Are Superior to Monotherapy Concerning the Killing of Biofilm Pseudomonas aeruginosa. J. Infect. Dis. 2010, 202, 1585–1592. [Google Scholar] [CrossRef] [Green Version]
- David, M.D.; Gill, M.J. Potential for underdosing and emergence of resistance in Acinetobacter baumannii during treatment with colistin. J. Antimicrob. Chemother. 2008, 61, 962–964. [Google Scholar] [CrossRef] [PubMed]
- Asempa, T.E.; Nicolau, D.P.; Kuti, J.L. In Vitro Activity of Imipenem-Relebactam Alone or in Combination with Amikacin or Colistin against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2019, 63, e00997-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ly, N.S.; Bulitta, J.B.; Rao, G.G.; Landersdorfer, C.B.; Holden, P.N.; Forrest, A.; Bergen, P.J.; Nation, R.L.; Li, J.; Tsuji, B.T. Colistin and doripenem combinations against Pseudomonas aeruginosa: Profiling the time course of synergistic killing and prevention of resistance. J. Antimicrob. Chemother. 2015, 70, 1434–1442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yaita, K.; Sameshima, I.; Takeyama, H.; Matsuyama, S.; Nagahara, C.; Hashiguchi, R.; Moronaga, Y.; Tottori, N.; Komatsu, M.; Oshiro, Y.; et al. Liver Abscess Caused by Multidrug-resistant Pseudomonas aeruginosa Treated with Colistin; A Case Report and Review of the Literature. Intern. Med. 2013, 52, 1407–1412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valour, F.; Dutronc, H.; Dinh, A.; Cazorla, C.; Pavèse, P.; Lesens, O.; Uçkay, I.; Chidiac, C.; Ferry, T. Difficult-to-treat Gram-negative bone and joint infections: Efficacy and safety of prolonged intravenous colistin. Int. J. Antimicrob. Agents 2013, 41, 197–199. [Google Scholar] [CrossRef]
- Tascini, C.; Menichetti, F.; Gemignani, G.; Palumbo, F.; Leonildi, A.; Tedeschi, A.; Piaggesi, A. Clinical and Microbiological Efficacy of Colistin Therapy in Combination With Rifampin and Imipenem in Multidrug-Resistant Pseudomonas aeruginosa Diabetic Foot Infection With Osteomyelitis. Int. J. Low. Extrem. Wounds 2006, 5, 213–216. [Google Scholar] [CrossRef]
- Harris, A.; Torres-Viera, C.; Venkataraman, L.; DeGirolami, P.; Samore, M.; Carmeli, Y. Epidemiology and Clinical Outcomes of Patients with Multiresistant Pseudomonas aeruginosa. Clin. Infect. Dis. 1999, 28, 1128–1133. [Google Scholar] [CrossRef] [Green Version]
- Woodford, N.; Ellington, M.J. The emergence of antibiotic resistance by mutation. Clin. Microbiol. Infect. 2007, 13, 5–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dettman, J.R.; Sztepanacz, J.L.; Kassen, R. The properties of spontaneous mutations in the opportunistic pathogen Pseudomonas aeruginosa. BMC Genom. 2016, 17, 27. [Google Scholar] [CrossRef] [Green Version]
- Rees, V.E.; Deveson Lucas, D.S.; López-Causapé, C.; Huang, Y.; Kotsimbos, T.; Bulitta, J.B.; Rees, M.C.; Barugahare, A.; Peleg, A.Y.; Nation, R.L.; et al. Characterization of Hypermutator Pseudomonas aeruginosa Isolates from Patients with Cystic Fibrosis in Australia. Antimicrob. Agents Chemother. 2019, 63, e02538-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samal, S.; Samir, S.B.; Patra, S.K.; Rath, A.; Dash, A.; Nayak, B.; Mohanty, D. Polymyxin Monotherapy vs. Combination Therapy for the Treatment of Multidrug-resistant Infections: A Systematic Review and Meta-analysis. Indian J. Crit. Care Med. 2021, 25, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Hu, Y.; Coates, A. Sudden cardiac death and tuberculosis-how much do we know? Tuberculosis 2012, 92, 307–313. [Google Scholar] [CrossRef]
| Mechanism of Resistance | Current Treatments | Emerging Treatments |
|---|---|---|
| β-lactamase production | Colistin | Ceftolozane–Tazobactam |
| Porin loss/mutation | Colistin | Ceftazidime–Avibactam |
| Efflux pump expression | Colistin | Aztreonam–Avibactam |
| Fosfomycin | ||
| Cefiderocol |
| Study No. | Study Title | Study Design | Number of Patients | Clinical Indication | Age | Male Sex n (%) | References | ||
|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Median (IQR) | ||||||||
| 1 | Epidemiology and outcome of infections with carbapenem-resistant Gram-negative bacteria treated with polymyxin B-based combination therapy | Single | OS | 104 | Various | 77 (±12.9) | - | 62 (59.6) | [28] |
| 2 | Polymyxin B in Combination with Antimicrobials Lacking In Vitro Activity versus Polymyxin B in Monotherapy in Critically Ill Patients with Acinetobacter baumannii or Pseudomonas aeruginosa Infections | Multi | RS | 101 | Various | 65.2 (±15.7) | - | 56 (55.4) | [29] |
| 3 | Impact of combination therapy and early de-escalation on outcome of ventilator-associated pneumonia caused by Pseudomonas aeruginosa | Single | RS | 100 | VAP | - | 64 (54–72) | 76 (76.0) | [30] |
| 4 | Active monotherapy and combination therapy for extensively drug-resistant Pseudomonas aeruginosa pneumonia | Single | RS | 136 | Various | - | 78 (70–83) | 74 (54) | [31] |
| 5 | Clinical characteristics and outcomes of Pseudomonas aeruginosa bacteremia in febrile neutropenic children and adolescents with the impact of antibiotic resistance: a retrospective study | Single | ROS | 31 | Febrile neutropenia | 9.5 (±5.4) | - | 26 (72.2) | [32] |
| 6 | Efficacy and safety of cefiderocol or best available therapy for the treatment of serious infections caused by carbapenem-resistant Gram-negative bacteria (CREDIBLE-CR): a randomised, open-label, multicentre, pathogen-focused, descriptive, phase 3 trial | Multi | RCT | 118 | Various | 63.0 (±16.7) | - | 101 (66.4) | [33] |
| 7 | Experience with Ceftolozane–Tazobactam for the Treatment of Serious Pseudomonas aeruginosa Infections in Saudi Tertiary Care Center | Single | ROS | 19 | Various | - | 57 (35–71) | 9 (47) | [34] |
| 8 | Colistin therapy for microbiologically documented multidrug-resistant Gram-negative bacterial infections: a retrospective cohort study of 258 patients | Single | RS | 258 | Various | 61.1 (±18.1) | - | 174 (67.4) | [35] |
| 9 | Colistin alone versus colistin plus meropenem for treatment of severe infections caused by carbapenem-resistant Gram-negative bacteria: an open-label, randomised controlled trial | Multi | RCT | 406 | Various | 66 (±16) | - | 151 (37.2) | [36] |
| 10 | Intravenous polymyxin B for the treatment of nosocomial pneumonia caused by multidrug-resistant Pseudomonas aeruginosa | Single | OS | 74 | Nosocomial pneumonia | - | 55 (17–89) | 50 (67.6) | [37] |
| 11 | Characteristics, risk factors and outcomes of adult cancer patients with extensively drug-resistant Pseudomonas aeruginosa infections | Single | RS | 89 | Cancer | - | 73 (21–87) | 18 (81.8) | [38] |
| 12 | Osteoarticular infection caused by MDR Pseudomonas aeruginosa: the benefits of combination therapy with colistin plus β-lactams | Single | RS | 34 | Osteoarticular infection | - | 69 (60–78) | 20 (58.8) | [39] |
| Study No. | Treatment | Number of Patients Treated [n (%)] | Number of Patients with P. aeruginosa | Resistance | Microbiological Cure [n (%)] | Mortality [n (%)] | References | |
|---|---|---|---|---|---|---|---|---|
| Sensitive Strains [n (%)] | Resistant Strains [n (%)] | |||||||
| 1 | Polymyxin B (combination) a | 104 | 0 (0) | 11 (10.5) | Carbapenem resistant | 20 | 50 | [28] |
| 2 | Polymyxin B (combination) b | 33 (34.7) | 0 (0) | 3 (9.1) 1,** | XDR | - | 0 (0) | [29] |
| Polymyxin B (monotherapy) c | 68 (65.3) | 15 (22) ** | 0 (0) | XDR | ||||
| 3 | Empirical combination therapy c | 85 (85.0) | 0 (0) | 26 (31) 2 | MDR | - | 32 (37.6) | [30] |
| Empirical monotherapy d | 15 (15) | 0 (0) | 5 (33) 2 | MDR | - | 7 (46.7) | ||
| 4 | Susceptible combination e | 40 (29.4) | 40 (29.4) 2 | 0 (0) | XDR | 36 (90) | 4 (10) | [31] |
| Susceptible monotherapy f | 74 (54.4) | 74 (54.4) 2 | 0 (0) | XDR | 40 (54) | 38 (51) | ||
| Resistant therapy g | 22 (16.2) | 0 (0) | 22 (16.2) 3 | XDR | 0 (0) | 22 (100) | ||
| 5 | Piperacillin–tazobactam with aminoglycoside | 16 (44.4) | 0 (0) | 16 (100) 1 | MDR | - | 3 (21.4) | [32] |
| Meropenem | 14 (38.9) | 14 (100) | 0 (0) | MDR | - | 10 (71.4) | ||
| Cefepime | 3 (7.4) | 3 (100) | 0 (0) | MDR | - | 0 (0) | ||
| Cefepime with aminoglycoside | 2 (5.6) | 2 (100) | 0 (0) | MDR | - | 0 (0) | ||
| Meropenem with aminoglycoside | 1 (2.8) | 1 (100) | 0 (0) | MDR | - | 1 (100) | ||
| 6 | Cefiderocol | 80 (67.8) | 12 (15) | 0 (0) | Carbapenem resistant | - | 2 (17) | [33] |
| Cefiderocol combination h | 38 (32.2) | 10 (26) | 0 (0) | Carbapenem resistant | - | 2 (20) | ||
| 7 | Ceftolozane–tazobactam | 19 (100) | 19 (100) | 0 (0) | Carbapenem resistant | 14 (74) | 4 (21) | [34] |
| 9 | Colistin | 198 (48.8) | 13 (4) | 0 (0) | Carbapenem resistant | - | 4 (31) | [36] |
| Colistin–meropenem | 208 (51.2) | 0 (0) | 8 (3.8) 1 | Carbapenem resistant | - | 2 (25) | ||
| 10 | Polymyxin B | 46 (62.2) | 46 (62.2) | 0 (0) ** | MDR | - | 25 (53) * | [37] |
| Polymyxin B combination i | 28 (37.8) | 0 (0) | 28 (37.8) 2,** | MDR | - | 14 (50) * | ||
| Study No. | Study Title | Study Design | Number of Patients | Clinical Indication | Age | Male Sex n (%) | Reference | ||
|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Median (IQR) | ||||||||
| 13 | A randomised, double-blind, phase 3 study comparing the efficacy and safety of ceftazidime–avibactam plus metronidazole versus meropenem for complicated intra-abdominal infections in hospitalised adults in Asia | Multi | RCT | 431 | cIAI | 48.5 ± 16.8 | - | 294 (68.2) | [45] |
| 14 | Ceftazidime–avibactam Versus Doripenem for the Treatment of Complicated Urinary Tract Infections, Including Acute Pyelonephritis: RECAPTURE, a Phase 3 Randomized Trial Program | Multi | RCT | 810 | cUTI | 51.4 ± 20.2 | - | 245 (30.2) | [46] |
| 15 | Efficacy and Safety of Ceftazidime–Avibactam Plus Metronidazole Versus Meropenem in the Treatment of Complicated Intra-abdominal Infection: Results from a Randomized, Controlled, Double-Blind, Phase 3 Program | Multi | RCT | 1043 | cIAI | 49.8 ± 17.5 | - | 658 (63.1) | [47] |
| 16 | Characterization of β-Lactamase Content of Ceftazidime-Resistant Pathogens Recovered during the Pathogen-Directed Phase 3 REPRISE Trial for Ceftazidime–Avibactam: Correlation of Efficacy against β-Lactamase Producers | Multi | RCT | 295 | Various | - | - | - | [48] |
| 17 | Clinical activity of ceftazidime–avibactam against MDR Enterobacteriaceae and Pseudomonas aeruginosa: pooled data from the ceftazidime–avibactam Phase III clinical trial programme | Multi | RCT | 1051 | Various | - | - | - | [49] |
| 18 | Ceftazidime–avibactam versus meropenem in nosocomial pneumonia, including ventilator-associated pneumonia (REPROVE): a randomised, double-blind, phase 3 non-inferiority trial | Multi | CT | 726 | VAP | - | 62·1 (16·6) | 542 (74.7) | [50] |
| Study No. | Treatment | Number of Patients with P. aeruginosa [n (%)] | Ceftazidime Resistance Profile | Number of Patients Treated [n (%)] | Microbiological Cure [n (%)] | Clinical Cure [n (%)] | References |
|---|---|---|---|---|---|---|---|
| 13 | Ceftazidime–avibactam | 11 (2.6) | Res | 1 (9.1) | - | 1 (100) | [45] |
| Sus | 10 (90.9) | - | 10 (100) | ||||
| 14 | Doripenem | 37 (4.6) | Res | 6 (16.2) | 5 (83.3) | - | [46] |
| Sus | 14 (37.8) | 10 (71.4) | - | ||||
| Ceftazidime–avibactam | Res | 7 (18.9) | 5 (71.4) | - | |||
| Sus | 10 (27.0) | 7 (70.0) | - | ||||
| 15 | Ceftazidime–avibactam and metronidazole | 68 (6.5) | Res | 2 (2.9) | - | 2 (100) | [47] |
| Sus | 30 (44.1) | - | 27 (90.0) | ||||
| Meropenem | Res | 4 (5.9) | - | 4 (100) | |||
| Sus | 32 (47.0) | - | 30 (93.8) | ||||
| 16 | Ceftazidime–avibactam | 18 (6.1) | - | 13 (72.2) | 11 (84.6) | 11 (84.6) | [48] |
| Best available treatment | - | 5 (27.8) | 3 (60.0) | 5 (100) | |||
| 17 | Ceftazidime–avibactam | 95 (9.0) | 66.1% Sus | 56 (58.9) | 32 (57.1) | - | [49] |
| Carbapenem comparators | 39 (41.1) | 21 (53.8) | - | ||||
| 18 | Ceftazidime–avibactam | 77 (10.6) | 24.8% Sus | 42 (54.5) | 18 (42.9) | 27 (64.3) | [50] |
| Meropenem | 35 (45.5) | 14 (40.0) | 27 (77.1) |
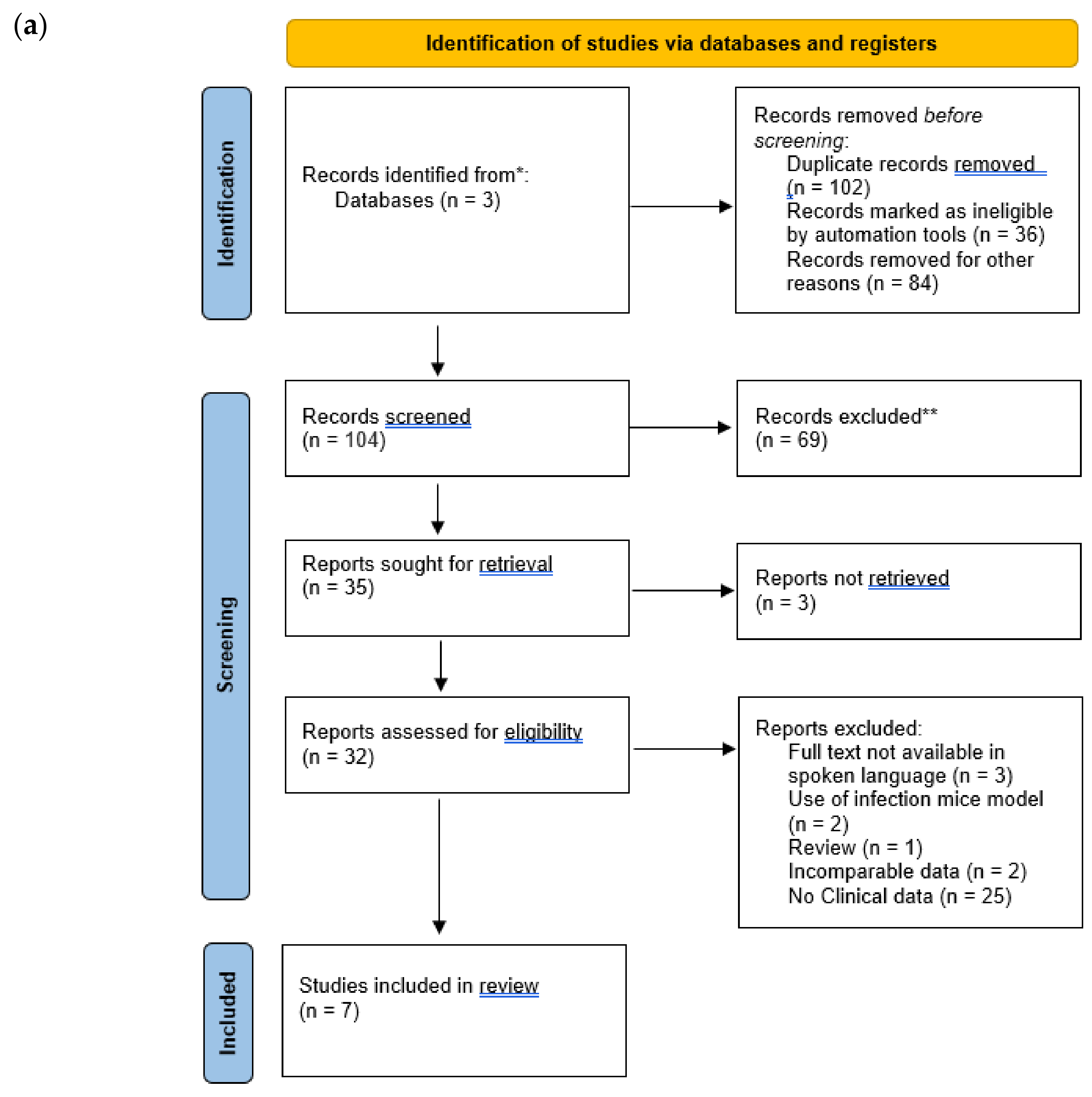
| Search Term 1 | Search Term 2 | Search Term 3 |
|---|---|---|
| Pseudomonas aeruginosa | MDR | Combination therapy |
| XDR | Multi-drug therapy | |
| Carbapenem resistant | ||
| Multi-drug resistant | ||
| Antibiotic resistant | ||
| Extensively drug resistant |
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Title or abstract must include a reference to antibiotic combination therapy or a name of one or more antibiotic combinations | Articles without an available abstract or full text |
| Title or abstract must include a reference to P. aeruginosa | Articles that were not published |
| Title or abstract must include a reference to antibiotic-resistant strains | Articles indicating that P. aeruginosa or antibiotic combination therapy and its efficacy is not the main focus |
| Must include in vitro or in vivo studies | Reviews, conference abstracts etc. |
| Papers that include data on MIC or FIC | Articles that are primarily discussions of the topic of combination therapy and P. aeruginosa |
| Papers that include monotherapy data | All surveys |
| Papers that include clinical cure, mortality rate, or other clinical data | Articles with simulated testing methodologies |
| Papers that include % susceptibility data | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, F.; Hu, Y.; Coates, A. The Efficacy of Using Combination Therapy against Multi-Drug and Extensively Drug-Resistant Pseudomonas aeruginosa in Clinical Settings. Antibiotics 2022, 11, 323. https://doi.org/10.3390/antibiotics11030323
Jones F, Hu Y, Coates A. The Efficacy of Using Combination Therapy against Multi-Drug and Extensively Drug-Resistant Pseudomonas aeruginosa in Clinical Settings. Antibiotics. 2022; 11(3):323. https://doi.org/10.3390/antibiotics11030323
Chicago/Turabian StyleJones, Frank, Yanmin Hu, and Anthony Coates. 2022. "The Efficacy of Using Combination Therapy against Multi-Drug and Extensively Drug-Resistant Pseudomonas aeruginosa in Clinical Settings" Antibiotics 11, no. 3: 323. https://doi.org/10.3390/antibiotics11030323
APA StyleJones, F., Hu, Y., & Coates, A. (2022). The Efficacy of Using Combination Therapy against Multi-Drug and Extensively Drug-Resistant Pseudomonas aeruginosa in Clinical Settings. Antibiotics, 11(3), 323. https://doi.org/10.3390/antibiotics11030323






