Determinants of the Empiric Use of Antibiotics by General Practitioners in South Africa: Observational, Analytic, Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Study Variables and Sample Size
2.3. Questionnaire Design and Analysis
3. Results
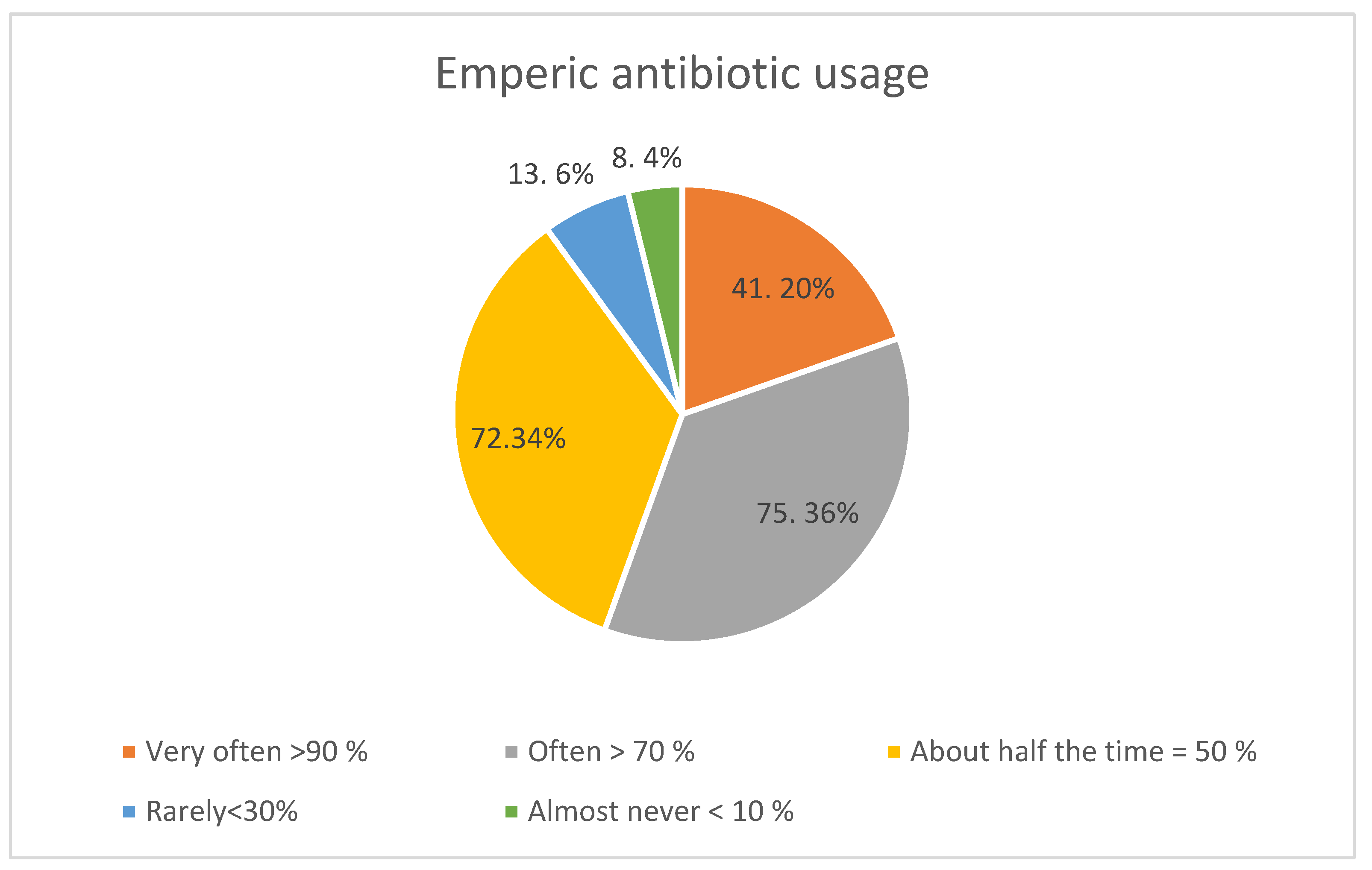
3.1. The Extent of Empiric Prescribing
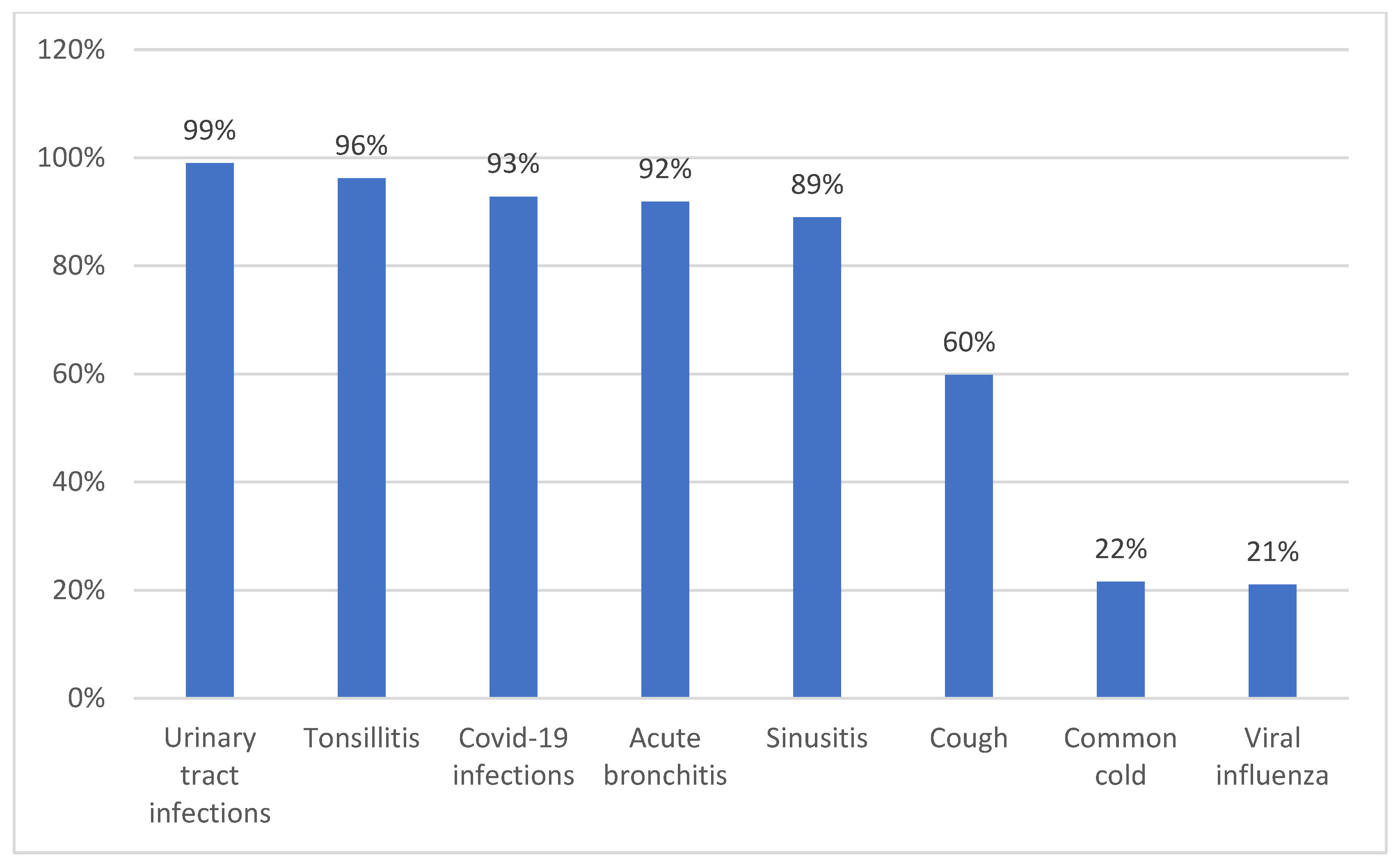
3.2. Prescribing Patterns
3.3. Demographic Factors Associated with Empiric Antibiotic Prescribing
3.4. Environmental Factors Associated with Empiric Antibiotic Prescribing
3.5. Physicians’ Knowledge and Empirical Antibiotic Usage
4. Discussion
Study Limitations
5. Conclusions and Recommendations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cassini, A.; Högberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2019, 19, 56–66. [Google Scholar] [CrossRef]
- Sartelli, M.; Hardcastle, T.C.; Catena, F.; Chichom-Mefire, A.; Coccolini, F.; Dhingra, S.; Haque, M.; Hodonou, A.; Iskandar, K.; Labricciosa, F.M.; et al. Antibiotic Use in Low and Middle-Income Countries and the Challenges of Antimicrobial Resistance in Surgery. Antibiotics 2020, 9, 497. [Google Scholar] [CrossRef]
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Founou, R.C.; Founou, L.L.; Essack, S.Y. Clinical and economic impact of antibiotic resistance in developing countries: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0189621. [Google Scholar] [CrossRef]
- O’Neill, J.C. The Review on Antimicrobial Resistance; Final Report London; HMOS; 2016. Available online: https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf (accessed on 25 August 2022).
- The World Bank. Final Report—Drug-Resistant Infections. A Threat to Our Economic Future March. 2017. Available online: http://documents1.worldbank.org/curated/en/323311493396993758/pdf/final-report.pdf (accessed on 22 August 2022).
- Llor, C.; Bjerrum, L. Antimicrobial resistance: Risk associated with antibiotic overuse and initiatives to reduce the problem. Ther. Adv. Drug Saf. 2014, 5, 229–241. [Google Scholar] [CrossRef]
- Kim, B.; Kim, Y.; Hwang, H.; Kim, J.; Kim, S.-W.; Bae, I.-G.; Choi, W.S.; Jung, S.I.; Jeong, H.W.; Pai, H. Trends and correlation between antibiotic usage and resistance pattern among hospitalized patients at university hospitals in Korea, 2004 to 2012. Medicine 2018, 97, e13719. [Google Scholar] [CrossRef]
- Huemer, M.; Shambat, S.M.; Brugger, S.D.; Zinkernagel, A.S. Antibiotic resistance and persistence—Implications for human health and treatment perspectives. EMBO Rep. 2020, 21, e51034. [Google Scholar] [CrossRef] [PubMed]
- Godman, B.; Egwuenu, A.; Haque, M.; Malande, O.; Schellack, N.; Kumar, S.; Saleem, Z.; Sneddon, J.; Hoxha, I.; Islam, S.; et al. Strategies to Improve Antimicrobial Utilization with a Special Focus on Developing Countries. Life 2021, 11, 528. [Google Scholar] [CrossRef]
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; Levin, S.A.; Goossens, H.; Laxminarayan, R. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463–E3470. [Google Scholar] [CrossRef] [PubMed]
- Torres, N.F.; Chibi, B.; Middleton, L.E.; Solomon, V.; Mashamba-Thompson, T. Evidence of factors influencing self-medication with antibiotics in LMICs: A systematic scoping review protocol. Syst. Rev. 2018, 7, 92–101. [Google Scholar] [CrossRef] [PubMed]
- WHO. Tackling Antimicrobial Resistance (AMR) Together—Working Paper 1.0: Multisectoral coordination. 2018. Available online: https://www.who.int/antimicrobial-resistance/publications/Tackling-AMR-multisectoral-coordination-june2018.pdf?ua=1 (accessed on 22 August 2022).
- Gasson, J.; Blockman, M.; Willems, B. Antibiotic prescribing practice and adherence to guidelines in primary care in the Cape Town Metro District, South Africa. South Afr. Med. J. 2018, 108, 304. [Google Scholar] [CrossRef]
- Sulis, G.; Adam, P.; Nafade, V.; Gore, G.; Daniels, B.; Daftary, A.; Das, J.; Gandra, S.; Pai, M. Antibiotic prescription practices in primary care in low- and middle-income countries: A systematic review and meta-analysis. PLOS Med. 2020, 17, e1003139. [Google Scholar] [CrossRef] [PubMed]
- Godman, B.; Haque, M.; McKimm, J.; Abu Bakar, M.; Sneddon, J.; Wale, J.; Campbell, S.; Martin, A.P.; Hoxha, I.; Abilova, V.; et al. Ongoing strategies to improve the management of upper respiratory tract infections and reduce inappropriate antibiotic use particularly among lower and middle-income countries: Findings and implications for the future. Curr. Med. Res. Opin. 2019, 36, 301–327. [Google Scholar] [CrossRef]
- Havers, F.P.; Hicks, L.A.; Chung, J.R.; Gaglani, M.; Murthy, K.; Zimmerman, R.K.; Jackson, L.A.; Petrie, J.G.; McLean, H.Q.; Nowalk, M.P.; et al. Outpatient Antibiotic Prescribing for Acute Respiratory Infections During Influenza Seasons. JAMA Netw. Open 2018, 1, e180243. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Zhang, Z.; Hicks, J.P.; Walley, J.D.; King, R.; Newell, J.N.; Yin, J.; Zeng, J.; Guo, Y.; Lin, M.; et al. Long-term outcomes of an educational intervention to reduce antibiotic prescribing for childhood upper respiratory tract infections in rural China: Follow-up of a cluster-randomised con-trolled trial. PLoS Med. 2019, 16, e1002733. [Google Scholar] [CrossRef]
- Muwanguzi, T.E.; Yadesa, T.M.; Agaba, A.G. Antibacterial prescription and the associated factors among outpatients diagnosed with respiratory tract infections in Mbarara Municipality, Uganda. BMC Pulm. Med. 2021, 21, 1–11. [Google Scholar] [CrossRef]
- Moore, M.; Stuart, B.; Coenen, S.; Butler, C.C.; Goossens, H.; Verheij, T.J.; Little, P. Amoxicillin for acute lower respiratory tract infection in primary care: Subgroup analysis of potential high-risk groups. Br. J. Gen. Pract. 2014, 64, e75–e80. [Google Scholar] [CrossRef]
- Holloway, K.A.; Ivanovska, V.; Wagner, A.K.; Vialle-Valentin, C.; Ross-Degnan, D. Prescribing for acute childhood infections in developing and transitional countries, 1990–2009. Paediatr. Int. Child Health 2014, 35, 5–13. [Google Scholar] [CrossRef][Green Version]
- Hassali, M.A.; Kamil, T.K.T.; Yusof, F.A.; Alrasheedy, A.A.; Yusoff, Z.M.; Saleem, F.; Al-Tamimi, S.K.; Wong, Z.Y.; Aljadhey, H.; Godman, B. General practitioners’ knowledge, attitude and prescribing of antibiotics for upper respiratory tract infections in Selangor, Malaysia: Findings and implications. Expert Rev. Anti-infective Ther. 2015, 13, 511–520. [Google Scholar] [CrossRef]
- Batura, N.; Cuevas, C.; Khan, M.; Wiseman, V. How effective and cost-effective are behaviour change interventions in improving the prescription and use of antibiotics in low-income and middle-income countries? A protocol for a systematic review. BMJ Open 2018, 8, e021517. [Google Scholar] [CrossRef]
- Farley, E.; Stewart, A.; Davies, M.-A.; Govind, M.; Bergh, D.V.D.; Boyles, T.H. Antibiotic use and resistance: Knowledge, attitudes and perceptions among primary care prescribers in South Africa. South Afr. Med. J. 2018, 108, 763–771. [Google Scholar] [CrossRef] [PubMed]
- WHO. Resource Mobilisation for Antimicrobial Resistance (AMR): Getting AMR into Plans and Budgets of Government and Development Partners—Nigeria Country Level Report. 2018. Available online: https://www.who.int/antimicrobial-resistance/national-action-plans/Nigeria-AMR-country-level-report-WHO-March-2018.pdf?ua=1 (accessed on 25 August 2022).
- Sadak, M.; Cramp, E.; Ashiru-Oredope, D. Antimicrobial Resistance and Stewardship in National Action Plans. Curr. Treat. Options Infect. Dis. 2016, 8, 57–71. [Google Scholar] [CrossRef]
- Godman, B.; Egwuenu, A.; Wesangula, E.; Schellack, N.; Kalungia, A.C.; Tiroyakgosi, C.; Kgatlwane, J.; Mwita, J.C.; Patrick, O.; Niba, L.L.; et al. Tackling antimicrobial resistance across sub-Saharan Africa; current challenges and implications for the future. Expert Opin. Drug Saf. 2022, 21, 1089–1111. [Google Scholar] [CrossRef]
- Department of Health Republic of South Africa. South African Antimicrobial Resistance National Strategy Framework; A One Health Approach–2017–2024. 2017. Available online: https://www.knowledgehub.org.za/system/files/elibdownloads/2020-03/AMR%20National%20Action%20Plan%202018%20-%202024.pdf (accessed on 20 August 2022).
- Engler, D.; Meyer, J.C.; Schellack, N.; Kurdi, A.; Godman, B. Antimicrobial Stewardship Activities in Public Healthcare Facilities in South Africa: A Baseline for Future Direction. Antibiotics 2021, 10, 996. [Google Scholar] [CrossRef] [PubMed]
- Department of Health Republic of South Africa. Antimicrobial Resistance—National Strategy Framework 2014–2024. Pretoria. Available online: https://health-e.org.za/wp-content/uploads/2015/09/Antimicrobial-Resistance-National-Strategy-Framework-2014-2024.pdf (accessed on 20 August 2022).
- Auta, A.; Hadi, M.A.; Oga, E.; Adewuyi, E.O.; Abdu-Aguye, S.N.; Adeloye, D.; Strickland-Hodge, B.; Morgan, D.J. Global access to antibiotics without prescription in community pharmacies: A systematic review and meta-analysis. J. Infect. 2019, 78, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Ocan, M.; Obuku, E.A.; Bwanga, F.; Akena, D.; Richard, S.; Ogwal-Okeng, J.; Obua, C. Household antimicrobial self-medication: A systematic review and meta-analysis of the burden, risk factors and outcomes in developing countries. BMC Public Health 2015, 15, 742. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.P.; Do, T.X.; Nguyen, H.A.; Nguyen, C.T.T.; Meyer, J.C.; Godman, B.; Skosana, P.; Nguyen, B.T. A National Survey of Dispensing Practice and Customer Knowledge on Antibiotic Use in Vietnam and the Implications. Antibiotics 2022, 11, 1091. [Google Scholar] [CrossRef]
- NDOH. National Department of Health—Republic of South Africa. Implementation Plan for Antimicrobial Resistance National Strategy Framework 2014–2019. 2019. Available online: http://www.health.gov.za/index.php/antimicrobial-resistance (accessed on 20 August 2022).
- Torres, N.; Chibi, B. Antibiotic Use and Resistance in South Africa: The Need for Better Data. 2019. Available online: http://www.hsrc.ac.za/en/review/hsrc-review-june-2019/antibiotic-use-and-resistance-in-sa (accessed on 20 August 2022).
- Schellack, N.; Benjamin, D.; Brink, A.; Duse, A.; Faure, K.; Goff, D.; Mendelson, M.; Meyer, J.; Miot, J.; Perovic, O.; et al. A situational analysis of current antimicrobial governance, regulation, and utilization in South Africa. Int. J. Infect. Dis. 2017, 64, 100–106. [Google Scholar] [CrossRef]
- Skosana, P.P.; Schellack, N.; Godman, B.; Kurdi, A.; Bennie, M.; Kruger, D.; Meyer, J.C. A national, multicentre web-based point prevalence survey of antimicrobial use in community healthcare centres across South Africa and the implications. Hosp. Pract. 2022, 50, 306–317. [Google Scholar] [CrossRef]
- Balliram, R.; Sibanda, W.; Essack, S.Y. The knowledge, attitudes and practices of doctors, pharmacists and nurses on antimicrobials, antimicrobial resistance and antimicrobial stewardship in South Africa. South. Afr. J. Infect. Dis. 2021, 36, 15. [Google Scholar] [CrossRef]
- WHO. Antimicrobial Stewardship Programmes in Health-Care Facilities in Low- and Middle-Income Countries: A WHO Practical toolkit. JAC-Antimicrobal Resist. 2019, 1, dlz072. [Google Scholar] [CrossRef]
- Matsitse, T.B.; Helberg, E.; Meyer, J.C.; Godman, B.; Massele, A.; Schellack, N. Compliance with the primary health care treatment guidelines and the essential medicines list in the management of sexually transmitted infections in correctional centres in South Africa: Findings and implications. Expert Rev. Anti-infective Ther. 2017, 15, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Ncube, N.B.Q.; Solanki, G.C.; Kredo, T.; Lalloo, R. Antibiotic prescription patterns of South African general medical practitioners for treatment of acute bronchitis. South Afr. Med. J. 2017, 107, 119–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Manderson, L. Prescribing, care and resistance: Antibiotic use in urban South Africa. Humanit. Soc. Sci. Commun. 2020, 7, 77. [Google Scholar] [CrossRef]
- MedicalBrief. Two-Thirds of SA Patients Prescribed Unnecessary Antibiotics by Their GP. 2019. Available online: https://www.medicalbrief.co.za/two-thirds-sa-patients-prescribed-unnecessary-antibiotics-gp/ (accessed on 25 August 2022).
- Mathibe, L.J.; Zwane, N.P. Unnecessary antimicrobial prescribing for upper respiratory tract infections in children in Pietermar-itzburg, South Africa. Afr. Health Sci. 2020, 20, 1133–1142. [Google Scholar] [CrossRef]
- Grace, C.J.; Robinson, B.; Empiric Antibiotic Selection. Antimicrobe. 2017. Available online: http://www.antimicrobe.org/e62.asp (accessed on 20 August 2022).
- Le Maréchal, M.; Tebano, G.; Monnier, A.A.; Adriaenssens, N.; Gyssens, I.C.; Huttner, B.; Milanic, R.; Schouten, J.; Benic, M.S.; Versporten, A.; et al. Quality indicators assessing antibiotic use in the outpatient setting: A systematic review followed by an international multidisciplinary consensus procedure. J. Antimicrob. Chemother. 2018, 73, vi40–vi49. [Google Scholar] [CrossRef]
- Komen, J.J.; Pottegård, A.; Mantel-Teeuwisse, A.K.; Forslund, T.; Hjemdahl, P.; Wettermark, B.; Hellfritzsch, M.; Hallas, J.; Olesen, M.; Bennie, M.; et al. Persistence and adherence to non-vitamin K antagonist oral anticoagulant treatment in patients with atrial fibrillation across five Western European countries. Europace 2021, 23, 1722–1730. [Google Scholar] [CrossRef]
- Alvarez-Madrazo, S.; McTaggart, S.; Nangle, C.; Nicholson, E.; Bennie, M. Data Resource Profile: The Scottish National Prescribing Information System (PIS). Int. J. Epidemiol. 2016, 45, 714–715f. [Google Scholar] [CrossRef]
- Alvarez-Madrazo, S.; Kavanagh, K.; Siebert, S.; Semple, Y.; Godman, B.; Almeida, A.M.; Acurcio, F.D.A.; Bennie, M. Discontinuation, persistence and adherence to subcutaneous biologics delivered via a homecare route to Scottish adults with rheumatic diseases: A retrospective study. BMJ Open 2019, 9, e027059. [Google Scholar] [CrossRef]
- Wettermark, B.; Godman, B.; Neovius, M.; Hedberg, N.; Mellgren, T.-O.; Kahan, T. Initial effects of a reimbursement restriction to improve the cost-effectiveness of antihypertensive treatment. Health Policy 2010, 94, 221–229. [Google Scholar] [CrossRef]
- Eriksson, I.; Wettermark, B.; Bergfeldt, K. Real-World Use and Outcomes of Olaparib: A Population-Based Cohort Study. Target. Oncol. 2018, 13, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Mashalla, Y.; Setlhare, V.; Massele, A.; Sepako, E.; Tiroyakgosi, C.; Kgatlwane, J.; Chuma, M.; Godman, B. Assessment of prescribing practices at the primary healthcare facilities in Botswana with an emphasis on antibiotics: Findings and implications. Int. J. Clin. Pract. 2017, 71, e13042. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.M.; Godman, B.; Diogene, E.; Fürst, J.; Gustafsson, L.L.; MacBride-Stewart, S.; Malmström, R.E.; Pedersen, H.; Selke, G.; Vlahović-Palčevski, V.; et al. Quality Indicators as a Tool in Improving the Introduction of New Medicines. Basic Clin. Pharmacol. Toxicol. 2014, 116, 146–157. [Google Scholar] [CrossRef]
- Engels, Y.; Campbell, S.; Dautzenberg, M.; Hombergh, P.V.D.; Brinkmann, H.; Szécsényi, J.; Falcoff, H.; Seuntjens, L.; Kuenzi, B.; Grol, R. Developing a framework of, and quality indicators for, general practice management in Europe. Fam. Pract. 2005, 22, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.M.; Ludt, S.; Van Lieshout, J.; Boffin, N.; Wensing, M.; Petek, D.; Grol, R.; Roland, M.O. Quality indicators for the prevention and man-agement of cardiovascular disease in primary care in nine European countries. Eur. J. Prev. Cardiol. 2008, 15, 509–515. [Google Scholar] [CrossRef]
- Campbell, S.M.; Kontopantelis, E.; Hannon, K.; Burke, M.; Barber, A.; Lester, H.E. Framework and indicator testing protocol for de-veloping and piloting quality indicators for the UK quality and outcomes framework. BMC Prim. Care 2011, 12, 85. [Google Scholar]
- Foxlee, N.D.; Townell, N.; Heney, C.; McIver, L.; Lau, C.L. Strategies Used for Implementing and Promoting Adherence to Antibiotic Guidelines in Low- and Lower-Middle-Income Countries: A Systematic Review. Trop. Med. Infect. Dis. 2021, 6, 166. [Google Scholar] [CrossRef]
- Robertson, J.; Iwamoto, K.; Hoxha, I.; Ghazaryan, L.; Abilova, V.; Cvijanovic, A.; Pyshnik, H.; Darakhvelidze, M.; Makalkina, L.; Jakupi, A.; et al. Antimicrobial Medicines Consumption in Eastern Europeand Central Asia—An Updated Cross-National Study and Assessment of QuantitativeMetrics for Policy Action. Front. Pharmacol. 2019, 9, 1156. [Google Scholar] [CrossRef]
- Quality Indicators for Antibiotic Consumption in the Community. Available online: https://www.ecdc.europa.eu/en/antimicrobial-consumption/database/quality-indicators (accessed on 20 August 2022).
- Olaoye, O.; Tuck, C.; Khor, W.P.; McMenamin, R.; Hudson, L.; Northall, M.; Panford-Quainoo, E.; Asima, D.M.; Ashiru-Oredope, E. Improving Access to Antimicrobial Prescribing Guidelines in 4 African Countries: Development and Pilot Implementation of an App and Cross-Sectional Assessment of Attitudes and Behaviour Survey of Healthcare Workers and Patients. Antibiotics 2020, 9, 555. [Google Scholar] [CrossRef]
- Hansen, M.P.; Bjerrum, L.; Gahrn-Hansen, B.; Jarbol, D.E. Quality indicators for diagnosis and treatment of respiratory tract infections in general practice: A modified Delphi study. Scand. J. Prim. Health Care 2010, 28, 4–11. [Google Scholar] [CrossRef]
- De Bie, S.; Kaguelidou, F.; Verhamme, K.M.; De Ridder, M.; Picelli, G.; Straus, S.M.; Giaquinto, C.; Stricker, B.H.; Bielicki, J.; Sharland, M.; et al. Using Prescription Patterns in Primary Care to Derive New Quality Indicators for Childhood Community Antibiotic Prescribing. Pediatr. Infect. Dis. J. 2016, 35, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Sharland, M.; Pulcini, C.; Harbarth, S.; Zeng, M.; Gandra, S.; Mathur, S.; Magrini, N. Classifying antibiotics in the WHO Essential Medicines List for optimal use—Be AWaRe. Lancet Infect. Dis. 2018, 18, 18–20. [Google Scholar] [CrossRef]
- Sharland, M.; Gandra, S.; Huttner, B.; Moja, L.; Pulcini, C.; Zeng, M.; Mendelson, M.; Cappello, B.; Cooke, G.; Magrini, N.; et al. Encouraging AWaRe-ness and discouraging inappropriate antibiotic use—The new 2019 Essential Medicines List becomes a global antibiotic stewardship tool. Lancet Infect. Dis. 2019, 19, 1278–1280. [Google Scholar] [CrossRef]
- Smith, D.R.M.; Dolk, C.; Pouwels, K.; Christie, M.; Robotham, J.; Smieszek, T. Defining the appropriateness and inappropriateness of antibiotic prescribing in primary care. J. Antimicrob. Chemother. 2018, 73, ii11–ii18. [Google Scholar] [CrossRef]
- Adriaenssens, N.; Coenen, S.; Tonkin-Crine, S.; Verheij, T.J.; Little, P.; Goossens, H. European Surveillance of Antimicrobial Con-sumption (ESAC): Disease-Specific Quality Indicators for Outpatient Antibiotic Prescribing. BMJ Qual. Saf. 2011, 20, 764–772. [Google Scholar] [CrossRef]
- Sánchez, X.; Orrico, M.; Morillo, T.; Manzano, A.; Jimbo, R.; Armijos, L. Reducing unnecessary antibiotic prescription through im-plementation of a clinical guideline on self-limiting respiratory tract infections. PLoS ONE 2021, 16, e0249475. [Google Scholar] [CrossRef]
- Niaz, Q.; Godman, B.; Campbell, S.; Kibuule, D. Compliance to prescribing guidelines among public health care facilities in Na-mibia; findings and implications. Int. J. Clin. Pharm. 2020, 42, 1227–1236. [Google Scholar] [CrossRef]
- Hamilton, D.; Bugg, I. Improving antimicrobial stewardship in the outpatient department of a district general hospital in Sierra Leone. BMJ Open Qual. 2018, 7, e000495. [Google Scholar] [CrossRef]
- Hansen, M.P.; Bjerrum, L.; Gahrn-Hansen, B.; Christensen, R.D.-P.; Davidsen, J.R.; Munck, A.; Jarbol, D.E. Quality indicators for treatment of respiratory tract infections? An assessment by Danish general practitioners. Eur. J. Gen. Pract. 2012, 19, 85–91. [Google Scholar] [CrossRef]
- Adriaenssens, N.; Bartholomeeusen, S.; Ryckebosch, P.; Coenen, S. Quality of antibiotic prescription during office hours and out-of-hours in Flemish primary care, using European quality indicators. Eur. J. Gen. Pract. 2013, 20, 114–120. [Google Scholar] [CrossRef]
- Gonzales, R.; Anderer, T.; McCulloch, C.E.; Maselli, J.H.; Bloom, F.J.; Graf, T.R.; Stahl, M.; Yefko, M.; Molecavage, J.; Metlay, J.P. A Cluster Randomized Trial of Decision Support Strategies for Reducing Antibiotic Use in Acute Bronchitis. JAMA Intern. Med. 2013, 173, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Wessell, A.M.; Liszka, H.A.; Nietert, P.J.; Jenkins, R.G.; Nemeth, L.S.; Ornstein, S. Achievable Benchmarks of Care for Primary Care Quality Indicators in a Practice-Based Research Network. Am. J. Med. Qual. 2008, 23, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Department of Health Republic of South Africa. Antimicrobial Resistance—National Strategy Framework; A One Health Approach 2018—2024. Available online: https://cdn.who.int/media/docs/default-source/antimicrobial-resistance/amr-spc-npm/nap-library/south-africa-antimicrobial-resistance-national-action-plan-2018---2024.pdf?sfvrsn=533118b0_1&download=true (accessed on 21 August 2022).
- Langford, B.J.; So, M.; Raybardhan, S.; Leung, V.; Soucy, J.-P.R.; Westwood, D.; Daneman, N.; MacFadden, D.R. Antibiotic prescribing in patients with COVID-19: Rapid review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Al-Hadidi, S.H.; Alhussain, H.; Hadi, H.A.; Johar, A.; Yassine, H.M.; Al Thani, A.A.; Eltai, N.O. The Spectrum of Antibiotic Prescribing During COVID-19 Pandemic: A Systematic Literature Review. Microb. Drug Resist. 2021, 27, 1705–1725. [Google Scholar] [CrossRef]
- Alshaikh, F.S.; Godman, B.; Sindi, O.N.; Seaton, R.A.; Kurdi, A. Prevalence of bacterial coinfection and patterns of antibiotics pre-scribing in patients with COVID-19: A systematic review and meta-analysis. PLoS ONE 2022, 17, e0272375. [Google Scholar] [CrossRef]
- Kimathi, G.; Kiarie, J.; Njarambah, L.; Onditi, J.; Ojakaa, D. A cross-sectional study of antimicrobial use among self-medicating COVID-19 cases in Nyeri County, Kenya. Antimicrob. Resist. Infect. Control 2022, 11, 111. [Google Scholar] [CrossRef]
- Soleymani, F.; Godman, B.; Yarimanesh, P.; Kebriaeezadeh, A. Prescribing patterns of physicians working in both the direct and indirect treatment sectors in Iran; findings and implications. J. Pharm. Health Serv. Res. 2019, 10, 407–413. [Google Scholar] [CrossRef]
- Tiroyakgosi, C.; Matome, M.; Summers, E.; Mashalla, Y.; Paramadhas, B.A.; Souda, S.; Malone, B.; Sinkala, F.; Kgatlwane, J.; Godman, B.; et al. Ongoing initiatives to improve the use of antibiotics in Botswana: University of Botswana symposium meeting report. Expert Rev. Anti-infective Ther. 2018, 16, 381–384. [Google Scholar] [CrossRef]
- Meyer, J.C.; Schellack, N.; Stokes, J.; Lancaster, R.; Zeeman, H.; Defty, D.; Godman, B.; Steel, G. Ongoing Initiatives to Improve the Quality and Effi-ciency of Medicine Use within the Public Healthcare System in South Africa; A Preliminary Study. Front. Pharmacol. 2017, 8, 751. [Google Scholar] [CrossRef]
- Versporten, A.; Zarb, P.; Caniaux, I.; Gros, M.-F.; Drapier, N.; Miller, M.; Jarlier, V.; Nathwani, D.; Goossens, H.; Koraqi, A.; et al. Antimicrobial consumption and resistance in adult hospital inpatients in 53 countries: Results of an internet-based global point prevalence survey. Lancet Glob. 2018, 6, e619–e629. [Google Scholar] [CrossRef]
- Niaz, Q.; Godman, B.; Massele, A.; Campbell, S.; Kurdi, A.; Kagoya, H.R.; Kibuule, D. Validity of World Health Organisation prescribing indicators in Namibia’s primary healthcare: Findings and implications. Int. J. Qual. Health Care 2018, 31, 338–345. [Google Scholar] [CrossRef]
- Ivanovska, V.; Hek, K.; Teeuwisse, A.K.M.; Leufkens, H.G.M.; Nielen, M.M.J.; Van Dijk, L. Antibiotic prescribing for children in primary care and adherence to treatment guidelines. J. Antimicrob. Chemother. 2016, 71, 1707–1714. [Google Scholar] [CrossRef]
- Van den Bergh, D.; Messina, A.P.; Goff, D.A.; van Jaarsveld, A.; Coetzee, R.; de Wet, Y.; Bronkhorst, E.; Brink, A.; Mendelson, M.; Richards, G.A.; et al. A pharmacist-led prospective antibiotic stewardship intervention improves compliance to community-acquired pneumonia guidelines in 39 public and private hos-pitals across South Africa. Int. J. Antimicrob. Agents 2020, 56, 106189. [Google Scholar] [CrossRef] [PubMed]
- Sefah, I.A.; Essah, D.O.; Kurdi, A.; Sneddon, J.; Alalbila, T.M.; Kordorwu, H.; Godman, B. Assessment of adherence to pneumonia guidelines and its determinants in an ambulatory care clinic in Ghana: Findings and implications for the future. JAC-Antimicrob. Resist. 2021, 3, dlab080. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.M.; Meyer, J.; Godman, B. Why Compliance to National Prescribing Guidelines is Important Especially across Sub-Saharan Africa and Suggestions for the Future. Biomed. Pharm. Sci. 2021, 4, 6. [Google Scholar]
- Krejcie, R.V.; Morgan, D.W. Determining Sample Size for Research Activities. Educ. Psychol. Meas. 1970, 30, 607–610. [Google Scholar] [CrossRef]
- Bai, Y.; Wang, S.; Yin, X.; Bai, J.; Gong, Y.; Lu, Z. Factors associated with doctors’ knowledge on antibiotic use in China. Sci. Rep. 2016, 6, 23429. [Google Scholar] [CrossRef]
- Etando, A.; Amu, A.A.; Haque, M.; Schellack, N.; Kurdi, A.; Alrasheedy, A.A.; Timoney, A.; Mwita, J.C.; Rwegerera, G.M.; Patrick, O.; et al. Challenges and Innovations Brought about by the COVID-19 Pandemic Regarding Medical and Pharmacy Education Especially in Africa and Implications for the Future. Healthcare 2021, 9, 1722. [Google Scholar] [CrossRef]
- Ogunleye, O.O.; Godman, B.; Fadare, J.O.; Mudenda, S.; Adeoti, A.O.; Yinka-Ogunleye, A.F.; Ogundele, S.O.; Oyawole, M.R.; Schönfeldt, M.; Rashed, W.M.; et al. Coronavirus Disease 2019 (COVID-19) Pandemic across Africa: Current Status of Vaccinations and Implications for the Future. Vaccines 2022, 10, 1553. [Google Scholar] [CrossRef]
- Ogunleye, O.O.; Basu, D.; Mueller, D.; Sneddon, J.; Seaton, R.A.; Yinka-Ogunleye, A.F.; Wamboga, J.; Miljković, N.; Mwita, J.C.; Rwegerera, G.M.; et al. Response to the Novel Corona Virus (COVID-19) Pandemic Across Africa: Successes, Challenges, and Implications for the Future. Front. Pharmacol. 2020, 11, 11. [Google Scholar] [CrossRef]
- Hoxha, I.; Malaj, A.; Kraja, B.; Bino, S.; Oluka, M.; Marković-Peković, V.; Godman, B. Are pharmacists’ good knowledge and awareness on antibiotics taken for granted? The situation in Albania and future implications across countries. J. Glob. Antimicrob. Resist. 2018, 13, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Babatola, A.O.; Fadare, J.O.; Olatunya, O.S.; Obiako, R.; Enwere, O.; Kalungia, A.; O Ojo, T.; A Sunmonu, T.; Desalu, O.; Godman, B. Addressing antimicrobial resistance in Nigerian hospitals: Exploring physicians prescribing behavior, knowledge, and perception of antimicrobial resistance and stewardship programs. Expert Rev. Anti-infective Ther. 2020, 19, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Department of Health Republic of South Africa—Adult Primary Care: Symptom-Based Integrated Approach to the Adult in Primary Care. 2019/2020. Available online: https://www.knowledgehub.org.za/system/files/elibdownloads/2020-08/APC%202019-20%20eBook.pdf (accessed on 25 August 2022).
- Kotwani, A.; Wattal, C.; Katewa, S.; Joshi, P.C.; Holloway, K. Factors influencing primary care physicians to prescribe antibiotics in Delhi India. Fam. Pract. 2010, 27, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Rezal, R.S.; Hassali, M.A.; Alrasheedy, A.A.; Saleem, F.; Yusof, F.A.M.; Kamal, M.; Din, R.M.; Godman, B. Prescribing patterns for upper respiratory tract infections: A prescription-review of primary care practice in Kedah, Malaysia, and the implications. Expert Rev. Anti-Infect. Ther. 2015, 13, 1547–1556. [Google Scholar] [CrossRef] [PubMed]
- Tsiga, E.; Panagopoulou, E.; Sevdalis, N.; Montgomery, A.; Benos, A. The influence of time pressure on adherence to guidelines in primary care: An experimental study. BMJ Open 2013, 3, e002700. [Google Scholar] [CrossRef]
- Linder, J.A.; Doctor, J.N.; Friedberg, M.W.; Reyes Nieva, H.; Birks, C.; Meeker, D.; Fox, C.R. Time of day and the decision to prescribe anti-biotics. JAMA Intern. Med. 2014, 174, 2029–2031. [Google Scholar] [CrossRef]
- Hyun, D. Why Doctors Prescribe Antibiotics Even When They Shouldn’t—Behavioral science-based strategies can help reduce inappropriate use. 2017. Available online: https://www.pewtrusts.org/en/research-and-analysis/articles/2017/06/30/why-doctors-prescribe-antibiotics-even-when-they-shouldnt (accessed on 28 August 2022).
- Rose, J.; Crosbie, M.; Stewart, A. A qualitative literature review exploring the drivers influencing antibiotic over-prescribing by GPs in primary care and recommendations to reduce unnecessary prescribing. Perspect. Public Health 2019, 141, 19–27. [Google Scholar] [CrossRef]
- Germeni, E.; Frost, J.; Garside, R.; Rogers, M.; Valderas, J.M.; Britten, N. Antibiotic prescribing for acute respiratory tract infections in primary care: An updated and expanded meta-ethnography. Br. J. Gen. Pract. 2018, 68, e633–e645. [Google Scholar] [CrossRef]
- Denyer Willis, L.; Chandler, C. Quick fix for care, productivity, hygiene and inequality: Reframing the entrenched problem of antibiotic overuse. BMJ Glob. Health 2019, 4, e001590. [Google Scholar] [CrossRef]
- Gjelstad, S.; Straand, J.; Dalen, I.; Fetveit, A.; Strøm, H.; Lindbæk, M. Do general practitioners’ consultation rates influence their prescribing patterns of antibiotics for acute respiratory tract infections? J. Antimicrob. Chemother. 2011, 66, 2425–2433. [Google Scholar] [CrossRef]
- Shapiro, D.J.; King, L.M.; Tsay, S.V.; Hicks, L.A.; Hersh, A.L. Association between antibiotic prescribing and visit duration among patients with respiratory tract infections. Infect. Control Hosp. Epidemiol. 2021, 43, 1238–1241. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, C.; Zhang, X.; Liu, C. Does diagnostic uncertainty increase antibiotic prescribing in primary care? NPJ Prim. Care Respir. Med. 2021, 31, 17. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wang, D.; Duan, L.; Zhang, X.; Liu, C. Coping with Diagnostic Uncertainty in Antibiotic Prescribing: A Latent Class Study of Primary Care Physicians in Hubei China. Front. Public Health 2021, 9, 741345. [Google Scholar] [CrossRef] [PubMed]
- Md Rezal, R.S.; Hassali, M.A.; Alrasheedy, A.A.; Saleem, F.; Md Yusof, F.A.; Godman, B. Physicians’ knowledge, perceptions and be-haviour towards antibiotic prescribing: A systematic review of the literature. Expert Rev. Anti-Infect. Ther. 2015, 13, 665–680. [Google Scholar] [CrossRef] [PubMed]
- Ray, M.J.; Tallman, G.B.; Bearden, D.T.; Elman, M.R.; McGregor, J.C. Antibiotic prescribing without documented indication in ambu-latory care clinics: National cross sectional study. BMJ 2019, 367, l6461. [Google Scholar] [CrossRef] [PubMed]
- Vicentini, C.; Vola, L.; Previti, C.; Brescia, V.; Mas, F.D.; Zotti, C.M.; Bert, F. Antimicrobial Stewardship Strategies Including Point-of-Care Testing (POCT) for Pediatric Patients with Upper-Respiratory-Tract Infections in Primary Care: A Systematic Review of Economic Evaluations. Antibiotics 2022, 11, 1139. [Google Scholar] [CrossRef]
- Lubell, Y.; Do, N.T.T.; Nguyen, K.V.; Ta, N.T.D.; Tran, N.T.H.; Than, H.M.; Hoang, L.B.; Shrestha, P.; Van Doorn, R.H.; Nadjm, B.; et al. C-reactive protein point of care testing in the management of acute respiratory infections in the Vietnamese primary healthcare setting—a cost benefit analysis. Antimicrob. Resist. Infect. Control 2018, 7, 119. [Google Scholar] [CrossRef]
- World Bank. Delivering Quality Health Services: A Global Imperative for Universal Health Coverage. 2018. Available online: https://documents.worldbank.org/en/publication/documents-reports/documentdetail/482771530290792652/delivering-quality-health-services-a-global-imperative-for-universal-health-coverage (accessed on 21 August 2022).
- McDonagh, M.S.; Peterson, K.; Winthrop, K.; Cantor, A.; Lazur, B.H.; Buckley, D.I. Interventions to reduce inappropriate prescribing of antibiotics for acute respiratory tract infections: Summary and update of a systematic review. J. Int. Med. Res. 2018, 46, 3337–3357. [Google Scholar] [CrossRef]
- Saleh, H.A.; Borg, M.A.; Stålsby Lundborg, C.; Saliba-Gustafsson, E.A. General Practitioners’, Pharmacists’ and Parents’ Views on Antibiotic Use and Resistance in Malta: An Exploratory Qualitative Study. Antibiotics 2022, 11, 661. [Google Scholar] [CrossRef]
- Tell, D.; Engström, S.; Mölstad, S. Adherence to guidelines on antibiotic treatment for respiratory tract infections in various cat-egories of physicians: A retrospective cross-sectional study of data from electronic patient records. BMJ Open 2015, 5, e008096. [Google Scholar] [CrossRef]
- Orzella, L.; Chini, F.; Rossi, P.G.; Borgia, P. Physician and patient characteristics associated with prescriptions and costs of drugs in the Lazio region of Italy. Health Policy 2010, 95, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, D.; Smit, M.A.M.; Kwestroo, G.A.; Verheij, R.A.; Hek, K.; Kunst, A.E. The influence of gender concordance between general practitioner and patient on antibiotic prescribing for sore throat symptoms: A retrospective study. BMC Fam. Pract. 2018, 19, 175. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, N.; Zhu, H.; Xu, S.; Lu, H.; Feng, Z. Prescription Pattern and Its Influencing Factors in Chinese County Hospitals: A Retrospective Cross-Sectional Study. PLoS ONE 2013, 8, e63225. [Google Scholar] [CrossRef] [PubMed]
- Saleem, Z.; Hassali, M.A.; Godman, B.; Hashmi, F.K.; Saleem, F. Antimicrobial prescribing and determinants of antimicrobial re-sistance: A qualitative study among physicians in Pakistan. Int. J. Clin. Pharm. 2019, 41, 1348–1358. [Google Scholar] [CrossRef]
- O’Doherty, J.; Leader, L.F.W.; O’Regan, A.; Dunne, C.; Puthoopparambil, S.J.; O’Connor, R. Over prescribing of antibiotics for acute respiratory tract infections; a qualitative study to explore Irish general practitioners’ perspectives. BMC Fam. Pract. 2019, 20, 27. [Google Scholar] [CrossRef]
- Lim, J.M.; Singh, S.R.; Duong, M.C.; Legido-Quigley, H.; Hsu, L.Y.; Tam, C.C. Impact of national interventions to promote responsible antibiotic use: A systematic review. J. Antimicrob. Chemother. 2020, 75, 14–29. [Google Scholar] [CrossRef]
- Lee, T.H.; Wong, J.G.; Lye, D.C.; Chen, M.I.; Loh, V.W.; Leo, Y.S.; Lee, L.K.; Chow, A.L. Medical and psychosocial factors associated with antibiotic pre-scribing in primary care: Survey questionnaire and factor analysis. Br. J. Gen. Pract. 2017, 67, e168–e177. [Google Scholar] [CrossRef]
- Liu, C.; Liu, C.; Wang, D.; Zhang, X. Intrinsic and external determinants of antibiotic prescribing: A multi-level path analysis of primary care prescriptions in Hubei, China. Antimicrob. Resist. Infect. Control. 2019, 8, 132. [Google Scholar] [CrossRef]
- Köchling, A.; Löffler, C.; Reinsch, S.; Hornung, A.; Böhmer, F.; Altiner, A.; Chenot, J.-F. Reduction of antibiotic prescriptions for acute res-piratory tract infections in primary care: A systematic review. Implement. Sci. 2018, 13, 47. [Google Scholar] [CrossRef]
- Godman, B.; Shrank, W.; Andersen, M.; Berg, C.; Bishop, I.; Burkhardt, T.; Garuoliene, K.; Herholz, H.; Joppi, R.; Kalaba, M.; et al. Comparing policies to enhance prescribing efficiency in Europe through increasing generic utilization: Changes seen and global implications. Expert Rev. Pharmacoeconomics Outcomes Res. 2010, 10, 707–722. [Google Scholar] [CrossRef]
- Moon, J.; Godman, B.; Petzold, M.; Alvarez-Madrazo, S.; Bennett, K.; Bishop, I.; Bucsics, A.; Hesse, U.; Martin, A.; Simoens, S.; et al. Different initiatives across Europe to enhance losartan utilization post generics: Impact and implications. Front. Pharmacol. 2014, 5, 219. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, L.L.; Wettermark, B.; Godman, B.; Andersén-Karlsson, E.; Bergman, U.; Hasselström, J.; Hensjö, L.-O.; Hjemdahl, P.; Jägre, I.; Julander, M.; et al. The ‘Wise List’- A Comprehensive Concept to Select, Communicate and Achieve Adherence to Recommendations of Essential Drugs in Ambulatory Care in Stockholm. Basic Clin. Pharmacol. Toxicol. 2011, 108, 224–233. [Google Scholar] [CrossRef]
- Björkhem-Bergman, L.; Andersén-Karlsson, E.; Laing, R.; Diogene, E.; Melien, O.; Jirlow, M.; Malmström, R.E.; Vogler, S.; Godman, B.; Gustafsson, L.L. Interface management of pharmacotherapy. Joint hospital and primary care drug recommendations. Eur. J. Clin. Pharmacol. 2013, 69, 73–78. [Google Scholar] [CrossRef]
- Eriksen, J.; Gustafsson, L.L.; Ateva, K.; Bastholm-Rahmner, P.; Ovesjö, M.-L.; Jirlow, M.; Juhasz-Haverinen, M.; Lärfars, G.; E Malmström, R.; Wettermark, B.; et al. High adherence to the ‘Wise List’ treatment recommendations in Stockholm: A 15-year retrospective review of a multifaceted approach promoting rational use of medicines. BMJ Open 2017, 7, e014345. [Google Scholar] [CrossRef] [PubMed]
- Kibuule, D.; Mubita, M.; Naikaku, E.; Kalemeera, F.; Godman, B.B.; Sagwa, E. An analysis of policies for cotrimoxazole, amoxicillin and azithromycin use in Namibia’s public sector: Findings and therapeutic implications. Int. J. Clin. Pract. 2017, 71, e12918. [Google Scholar] [CrossRef] [PubMed]
- NHS Scotland. Secondary Care National Therapeutic Indicators 2018/19. 2018. Available online: https://www.therapeutics.scot.nhs.uk/wp-content/uploads/2018/08/Secondary-Care-National-Therapeutic-Indicators-Version-1.0.pdf (accessed on 20 August 2022).
- Godman, B.; Wettermark, B.; Hoffmann, M.; Andersson, K.; Haycox, A.; Gustafsson, L.L. Multifaceted national and regional drug reforms and initiatives in ambulatory care in Sweden: Global relevance. Expert Rev. Pharmacoeconomics Outcomes Res. 2009, 9, 65–83. [Google Scholar] [CrossRef]
- MacBride-Stewart, M.S.; Kurdi, A.; Sneddon, J.; McBurney, S.; do Nascimento, R.C.R.M.; Mueller, T.; Kwon, H.-Y.; Morton, A.; Seaton, R.A.; Timoney, A.; et al. Initiatives and reforms across Scotland in recent years to improve prescribing; findings and global implications of drug prescriptions. Int. J. Clin. Exp. Med. 2021, 14, 2563–2586. [Google Scholar]
- The WHO Essential Medicines List Antibiotic Book—Draft for Public Comment. 2021. Available online: https://cdn.who.int/media/docs/default-source/essential-medicines/eml-antibiotic-book-draft.pdf?sfvrsn=cb6cb7c2_6&download=true (accessed on 25 August 2022).
- Klein, E.Y.; Milkowska-Shibata, M.; Tseng, K.K.; Sharland, M.; Gandra, S.; Pulcini, C.; Laxminarayan, R. Assessment of WHO antibiotic consumption and access targets in 76 countries, 2000–2015: An analysis of pharmaceutical sales data. Lancet Infect. Dis. 2020, 21, 107–115. [Google Scholar] [CrossRef]
- Oxford Population Health. New Project Aims to Inform National Action Plans to Combat Antimicrobial Resistance. 2022. Available online: https://www.ndph.ox.ac.uk/news/new-project-aims-to-inform-national-action-plans-to-combat-antimicrobial-resistance (accessed on 30 August 2022).
- Skosana, P.; Schellack, N.; Godman, B.; Kurdi, A.; Bennie, M.; Kruger, D.; Meyer, J. A national, multicentre, web-based point prevalence survey of antimicrobial use and quality indices among hospitalised paediatric patients across South Africa. J. Glob. Antimicrob. Resist. 2021, 29, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Kruger, D.; Dlamini, N.; Meyer, J.; Godman, B.; Kurdi, A.; Lennon, M.; Bennie, M.; Schellack, N. Development of a web-based application to improve data collection of antimicrobial utilization in the public health care system in South Africa. Hosp. Pract. 2021, 49, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Nair, M.M.; Mahajan, R.; Burza, S.; Zeegers, M.P. Behavioural interventions to address rational use of antibiotics in outpatient settings of low-income and lower-middle-income countries. Trop. Med. Int. Health 2021, 26, 504–517. [Google Scholar] [CrossRef]
- Do, N.T.T.; Ta, N.T.D.; Tran, N.T.H.; Than, H.M.; Vu, B.T.N.; Hoang, L.; van Doorn, H.R.; Vu, D.T.V.; Cals, J.; Chandna, A.; et al. Point-of-care C-reactive protein testing to reduce inappropriate use of antibiotics for non-severe acute respiratory infections in Vietnamese primary health care: A randomised controlled trial. Lancet Glob. Health. 2016, 4, e633–e641. [Google Scholar] [CrossRef]
- Little, P.; Stuart, B.; Francis, N.; Douglas, E.; Tonkin-Crine, S.; Anthierens, S.; Cals, J.W.; Melbye, H.; Santer, M.; Moore, M.; et al. Effects of internet-based training on antibiotic prescribing rates for acute respiratory-tract infections: A multinational, cluster, randomised, factorial, controlled trial. Lancet 2013, 382, 1175–1182. [Google Scholar] [CrossRef]
- Gjelstad, S.; Høye, S.; Straand, J.; Brekke, M.; Dalen, I.; Lindbæk, M. Improving antibiotic prescribing in acute respiratory tract infections: Cluster randomised trial from Norwegian general practice (prescription peer academic detailing (Rx-PAD) study). BMJ 2013, 347, f4403. [Google Scholar] [CrossRef] [PubMed]
- Gulliford, M.C.; Van Staa, T.; Dregan, A.; McDermott, L.; McCann, G.; Ashworth, M.; Charlton, J.; Little, P.; Moore, M.V.; Yardley, L. Electronic Health Records for Intervention Research: A Cluster Randomized Trial to Reduce Antibiotic Prescribing in Primary Care (eCRT Study). Ann. Fam. Med. 2014, 12, 344–351. [Google Scholar] [CrossRef]
- Meeker, D.; Linder, J.A.; Fox, C.R.; Friedberg, M.W.; Persell, S.D.; Goldstein, N.J.; Knight, T.K.; Hay, J.W.; Doctor, J.N. Effect of Behavioral Interventions on Inappropriate Antibiotic Prescribing Among Primary Care Practices: A Randomized Clinical Trial. JAMA 2016, 315, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, A.K.; Khan, O.F.; Matin, M.A.; Begum, K.; Galib, M.A. Effect of standard treatment guidelines with or without pre-scription audit on prescribing for acute respiratory tract infection (ARI) and diarrhoea in some thana health complexes (THCs) of Bangladesh. Bangladesh Med. Res. Counc. Bull. 2007, 33, 21–30. [Google Scholar] [PubMed]
- Yip, W.; Powell-Jackson, T.; Chen, W.; Hu, M.; Fe, E.; Hu, M.; Hsiao, W.C. Capitation combined with pay-for-performance improves antibiotic prescribing practices in rural China. Health Aff. 2014, 33, 502–510. [Google Scholar] [CrossRef]
- Dehn Lunn, A. Reducing inappropriate antibiotic prescribing in upper respiratory tract infection in a primary care setting in Kolkata, India. BMJ Open Qual. 2018, 7, e000217. [Google Scholar] [CrossRef]
- Korom, R.R.; Onguka, S.; Halestrap, P.; McAlhaney, M.; Adam, M. Brief educational interventions to improve performance on novel quality metrics in ambulatory settings in Kenya: A multi-site pre-post effectiveness trial. PLoS ONE 2017, 12, e0174566. [Google Scholar] [CrossRef]
- Teng, C.L.; I Achike, F.; Phua, K.L.; I Nurjahan, M.; Mastura, I.; Asiah, H.N.; Mariam, A.M.; Narayanan, S.; Norsiah, A.; Sabariah, I.; et al. Modifying antibiotic prescribing: The effectiveness of academic detailing plus information leaflet in a Malaysian primary care setting. Med. J. Malays. 2006, 61, 323–331. [Google Scholar]
- Tay, K.H.; Ariffin, F.; Sim, B.L.; Chin, S.Y.; Sobry, A.C. Multi-Faceted Intervention to Improve the Antibiotic Prescriptions among Doctors for Acute URI and Acute Diarrhoea Cases: The Green Zone Antibiotic Project. Malays. J. Med. Sci. 2019, 26, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, N.; Samir, K.C.; Baltussen, R.; Kafle, K.K.; Bishai, D.; Niessen, L. Practical Approach to Lung Health in Nepal: Better prescribing and reduction of cost. Trop. Med. Int. Health 2006, 11, 765–772. [Google Scholar] [CrossRef]
- Kafle, K.K.; Bhuju, G.B.; Karkee, S.B.; Prasad, R.R.; Shrestha, N.; Shrestha, A.D.; Das, P.L.; Chataut, B.D.; Daud, M. An intervention improving prescribing practices and monitoring drugs availability in a district. Nepal Med. Coll. J. NMCJ 2009, 11, 217–221. [Google Scholar]
- Awad, A.I.; Eltayeb, I.B.; Baraka, O.Z. Changing antibiotics prescribing practices in health centers of Khartoum State, Sudan. Eur. J. Clin. Pharmacol. 2006, 62, 135–142. [Google Scholar] [CrossRef]
- Boonyasiri, A.; Thamlikitkul, V. Effectiveness of multifaceted interventions on rational use of antibiotics for patients with upper respiratory tract infections and acute diarrhea. J. Med. Assoc. Thail. 2014, 97, S13–S19. [Google Scholar]
- Hoa, N.Q.; Lan, P.T.; Phuc, H.D.; Chuc, N.T.K.; Lundborg, C.S. Antibiotic prescribing and dispensing for acute respiratory infections in children: Effectiveness of a multi-faceted intervention for health-care providers in Vietnam. Glob. Health Action 2017, 10, 1327638. [Google Scholar] [CrossRef]
| Guidelines/Contents | Content/Nature |
|---|---|
| Key attributes of quality indicators (adapted from [53,54,55,56]) |
|
| Potential indicators |
| Variable | Criteria |
|---|---|
| Inclusion |
|
| Exclusion |
|
| Variable | Category | Empirical Antibiotic Prescribing (n;%) | Antibiotics Not Prescribed Empirically (n;%) | Total (%) | Unadjusted Odds Ratio (95%CI) | Adjusted Odds Ratio (95%CI) |
|---|---|---|---|---|---|---|
| Age | 25–34 | 11 (5.3%) | 13 (6.2%) | 24 (11.5%) | Reference | |
| 35–44 | 56 (26.8%) | 27 (12.9%) | 83 (39.7%) | 2.45 (0.97–6.18) | 3.38 (1.15–9.88) * | |
| 45–54 | 30 (14.4%) | 33 (15.8%) | 63 (30.1%) | 1.07 (0.42–2.75) | 2.78 (0.77–10.02) | |
| >55 years | 19 (9.1%) | 20 (9.6%) | 39 (18.7%) | 1.12 (0.41–3.11) | 4.75 (1.08–21) * | |
| Gender | Females | 61 (29.2%) | 26 (12.4%) | 87 (41.6%) | ||
| Males | 67 (32.1%) | 55 (26.3%) | 122 (58.4%) | 0.35 (0.18–0.65) ** | 0.46 (0.23–0.90) * | |
| Type of practice | Solo | 64 (30.6%) | 70 (33.5%) | 134 (64.1%) | 0.41 (0.22–0.77) ** | 0.29 (0.14–0.64) ** |
| Group | 52 (24.9%) | 23 (11.0%) | 75 (35.9%) | |||
| No. of consultations per day | <50 | 97 (46.4%) | 75 (35.9%) | 172 (82.3%) | ||
| >50 | 19 (9.1%) | 18 (8.6%) | 37 (17.7%) | 0.83 (0.37–1.77) | 1.04 (0.46–2.35) | |
| Dispensing doctor | Yes | 52 (24.9%) | 49 (23.4%) | 101 (48.3%) | 0.73 (0.41–1.31) | 1.17 (0.6–2.32) |
| No | 64 (30.6%) | 44 (21.1%) | 108 (51.7%) | |||
| Estimated number of antibiotics prescribed in the last week | ≤10 | 28 (13.4%) | 30 (14.4%) | 58 (27.8%) | ||
| >10 | 88 (42.1%) | 63 (30.1%) | 151 (72.3%) | 1.50 (0.78–2.88) | 2.39 (1.15–4.95) * | |
| Years in private practice | ≥15 years | 35 (16.7%) | 47 (22.5%) | 82 (39.2%) | 0.43 (0.23–0.78) * | 0.44 (0.17–1.12) |
| <15 years | 81 (38.8%) | 46 (22.0%) | 127 (60.8%) | |||
| Attended any training on antibiotic prescribing between 2019–2020 | Yes | 90 (43.1%) | 65 (31.1%) | 155 (74.2%) | 1.49 (0.76–2.91) | 1.29 (0.65–2.57) |
| No | 26 (12.4%) | 28 (13.4%) | 54 (25.8%) |
| Variable | Adjusted Odds Ratio (95%CI) |
|---|---|
| To prevent serious complications | 3.25 (1.20–8.81) * |
| Duration of symptoms | 0.64 (0.28–1.45) |
| Patient clinical condition | 0.42 (0.17–1.07) |
| Diagnostic uncertainty | 3.15 (1.40–7.07) ** |
| Type of disease | 2.71 (0.65–11.41) |
| Presence of comorbidities | 0.99 (0.46–2.11) |
| Patient expectation/request | 0.66 (0.129–3.36) |
| Antimicrobial resistance concerns | 0.74 (0.33–1.67) |
| Peers/colleague opinion | 1.16 (0.50–2.70) |
| Workload/Time pressure | 19.35 (2.73–137.19) |
| Medical aid formulary | 2.39 (1.10–5.16) ** |
| International Conferences | 0.27 (0.11–0.65) |
| Lack of resources (access to microbiology laboratory) | 3.11 (0.99–9.71) |
| Pharmaceutical representative | 1.65 (0.66–3.98) |
| Lack of antibiotics prescribing guidelines | 0.018 (0.001–0.18) ** |
| Microbiologist advice | 1.96 (0.89–4.3) |
| Variable | Category | Empiric Antibiotics Use (n;%) | Antibiotics Not Prescribed Empirically (n;%) | Total (%) |
|---|---|---|---|---|
| Dosage reduction of antibiotics is necessary for patients with renal failure? | Yes (Correct) | 108 (51.7%) | 83 (39.7%) | 191 (91.4%) |
| No | 8 (3.8%) | 10 (4.8%) | 18 (8.6%) | |
| Should antibiotics be prescribed for non-febrile diarrhoea? | Yes (Incorrect) | 26 (12.4%) | 13 (6.2%) | 39 (18.7%) |
| No (Correct) | 90 (43.1%) | 80 (38.3%) | 170 (81.4%) | |
| Antibiotics can be used for bacteria pneumonia (including one of the following symptoms: chest in-drawing or stridor, fast breathing). | YES (Correct) | 106 (50.8%) | 84 (40.2%) | 190 (90.9%) |
| NO (Incorrect) | 10 (4.8%) | 9 (4.3%) | 19 (9.1%) | |
| Do antibiotics reduce the duration and the occurrence of complications of upper respiratory tract infections? | YES (Incorrect) | 47 (22.5%) | 49 (23.4%) | 96 (45.9%) |
| NO (Correct) | 69 (33.0%) | 44 (21.1%) | 113 (54.1%) | |
| Is amoxicillin safe to use in pregnancy? | YES (Correct) | 102 (48.8%) | 83 (39.7%) | 185 (88.5%) |
| NO (Incorrect) | 14 (6.7%) | 10 (4.8%) | 24 (11.5%) | |
| Methicillin resistant staphylococcus aureus is resistant to beta-lactam antibiotics? | YES (Correct) | 81 (38.8%) | 72 (34.4%) | 153 (73.2%) |
| NO (Incorrect) | 35 (16.7%) | 21 (10.0%) | 56 (26.8%) | |
| Which of the following antibiotics crosses the blood–brain barrier? | Clindamycin (Incorrect) | 60 (28.7%) | 33 (15.8%) | 93 (44.5%) |
| Vancomycin (Incorrect) | ||||
| Ceftriaxone (Correct) | 44 (18.2%) | 72 (34.4%) | 116 (55.5%) | |
| Methicillin resistant staphylococcus aureus is susceptible to which antibiotics? | Amoxicillin-clavulanic acid (Incorrect) | 57 (27.3%) | 35 (16.7%) | 67 (32.1%) |
| Ceftriaxone (Incorrect) | 11 (5.3%) | |||
| Cefotaxime (Incorrect) | 14 (6.7%) | |||
| None of these antibiotics (Correct) | 59 (28.2%) | 58 (27.8%) | 117 (56.0%) | |
| Use of broad spectrum antibiotics when a narrow spectrum antibiotics (equally effective) are available can increase antibiotics resistance | YES (Correct) | 110 (52.6%) | 6 (2.9%) | 116 (55.5%) |
| NO (Incorrect) | 89 (42.6%) | 4 (1.9%) | 93 (44.55%) | |
| Patients’ non-compliance to antibiotics drives antibiotics resistance | YES (Correct) | 110 (52.6%) | 6 (2.9%) | 116 (55.5%) |
| NO (Incorrect) | 81 (38.8%) | 12 (5.7%) | 93 (44.5%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guma, S.P.; Godman, B.; Campbell, S.M.; Mahomed, O. Determinants of the Empiric Use of Antibiotics by General Practitioners in South Africa: Observational, Analytic, Cross-Sectional Study. Antibiotics 2022, 11, 1423. https://doi.org/10.3390/antibiotics11101423
Guma SP, Godman B, Campbell SM, Mahomed O. Determinants of the Empiric Use of Antibiotics by General Practitioners in South Africa: Observational, Analytic, Cross-Sectional Study. Antibiotics. 2022; 11(10):1423. https://doi.org/10.3390/antibiotics11101423
Chicago/Turabian StyleGuma, Sinenhlanhla Pearl, Brian Godman, Stephen M. Campbell, and Ozayr Mahomed. 2022. "Determinants of the Empiric Use of Antibiotics by General Practitioners in South Africa: Observational, Analytic, Cross-Sectional Study" Antibiotics 11, no. 10: 1423. https://doi.org/10.3390/antibiotics11101423
APA StyleGuma, S. P., Godman, B., Campbell, S. M., & Mahomed, O. (2022). Determinants of the Empiric Use of Antibiotics by General Practitioners in South Africa: Observational, Analytic, Cross-Sectional Study. Antibiotics, 11(10), 1423. https://doi.org/10.3390/antibiotics11101423







