Potential Utility of Ultrasound-Enhanced Delivery of Antibiotics, Anti-Inflammatory Agents, and Nutraceuticals: A Mini Review
Abstract
:1. Introduction
2. Phonophoresis Mechanism of Action
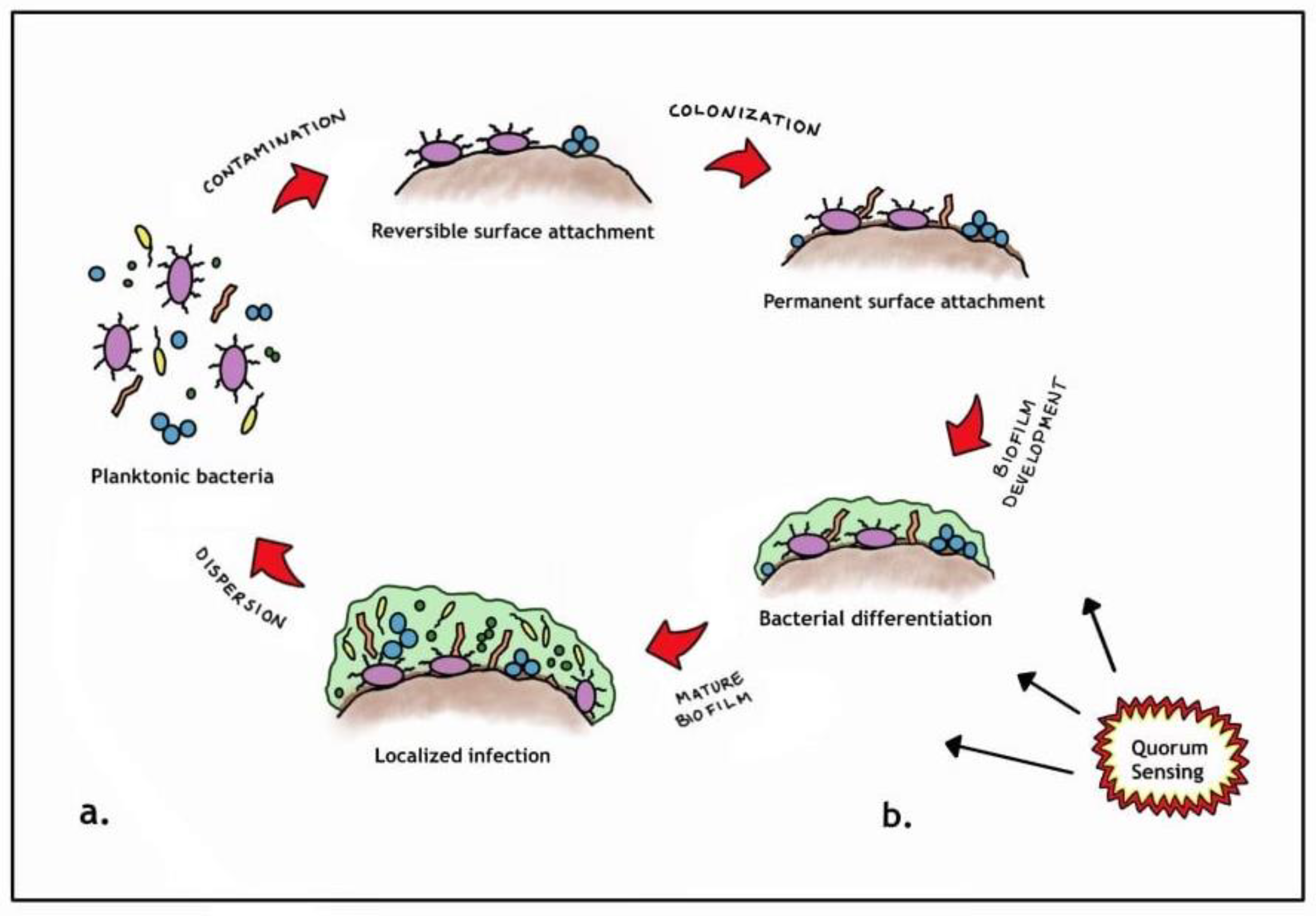
3. Wound Wars: Return of the Biofilm!
- Free floating planktonic bacteria initially disperse but then eventually (reversibly) attach to surface structures [11].
- However, if the planktonic species start to multiply, they become more firmly attached (sessile) and differentiate, consequently changing their genetic makeup to promote survival. This complex communication mechanism is known as quorum sensing [12].
- Once the aggregated planktonic species reach an optimal level, they form an extracellular polymeric substance (EPS) [13].
- The EPS generally is composed of various polysaccharides, proteins, glycolipids and bacterial DNA that essentially functions as a protective barrier [13].
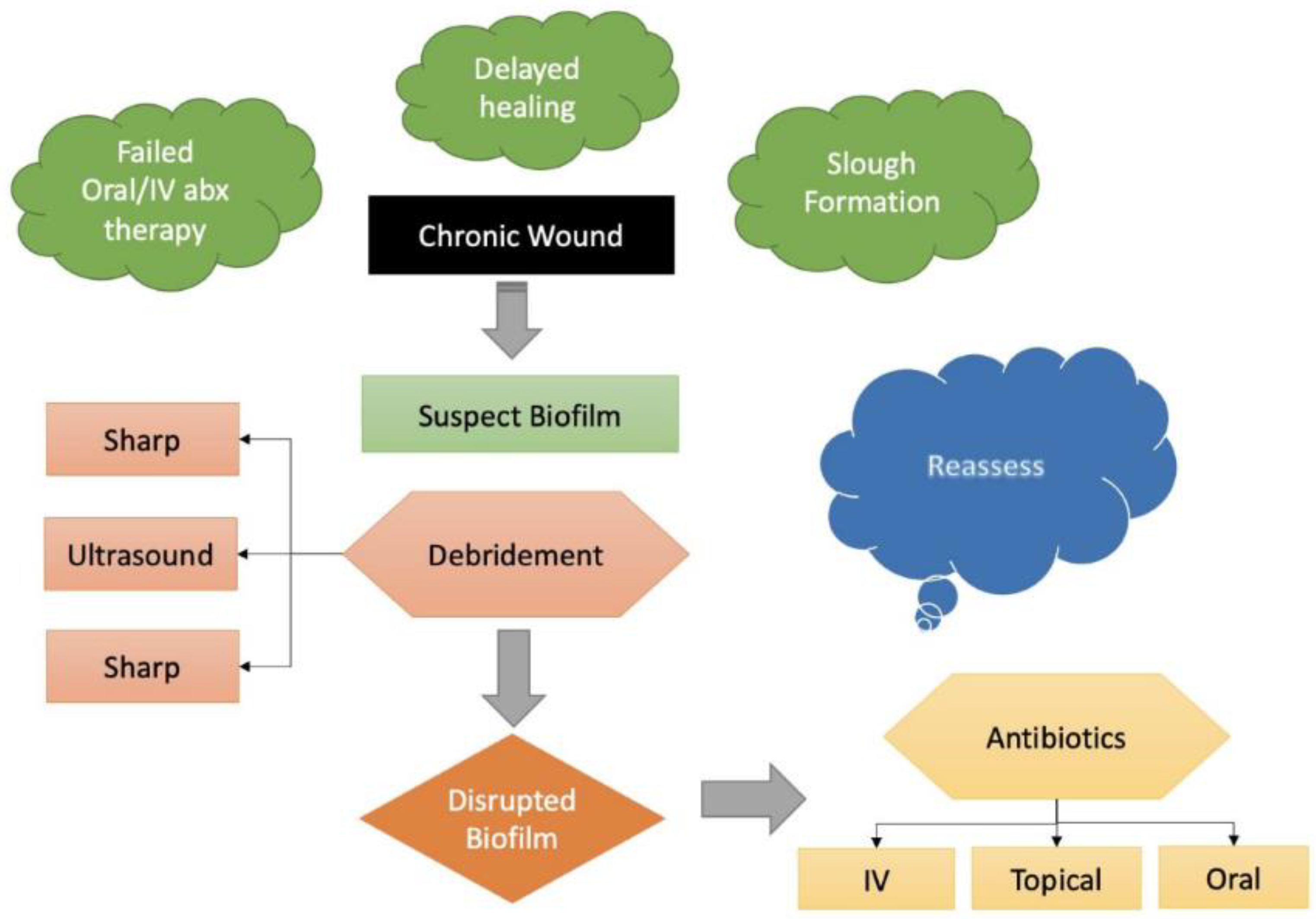
4. Materials and Methods
5. Results
5.1. Phonophoresis with Topical Antibiotics
5.2. Phonophoresis with Topical Anti-Inflammatory Agents
5.3. Phonophoresis with Using Nutraceuticals
| Study | Sample Size | Sample Characteristics | Frequency | US Type and Intensity | Topical Agent | Duration of Treatment |
|---|---|---|---|---|---|---|
| Ansari et al. (2013) [21] | 1 | Animal | 1 MHz | Pulsed, 1.0 W/cm2 | Erythromycin | 5 min, every other day for 10 total treatment sessions |
| Ay et al. (2010) [27] | 20 | Animal | 1.0 MHz | Not Specified, 1.5 W/cm2 | Diclofenac | 10 min, 5 times a week over 3 weeks, 15 total treatment sessions |
| Cagnie et al. (2003) [24] | 20 | Animal | 1.0 MHz | Pulsed & Continuous, 1.5 W/cm2 | Ketoprofen | 5 min |
| Fares et al. (2017) [32] | 6 | Animal | 1.0 MHz | Pulsed, 1.5 W/cm2 | Garlic Extract | 5 min, 3 treatment sessions a week for 3 weeks |
| Saliba et al. (2007) [25] | 10 | Human | 3.0 MHz | Pulsed, 1.0 W/cm2 | Dexamethasone | 5 min |
| Horsely et al. (2019) [19] | 3 | Human | 1.1 MHz | Pulsed with MBs, 2.5 Mpa | Gentamicin | 20 s |
| Dong et al. (2017) [23] | 3 | Human | 1.0 MHz | Pulsed with MBs, 0.5 W/cm2 | Vancomycin | 5 min |
| Cardoso et al. (2019) [28] | 66 | Human | 1.0 MHz | Pulsed, 1.0 W/cm2 | Diclofenac | 1 min |
| Chen et al. (2016) [22] | 41 | Human | 1.0 MHz | Not Specified, 0.75 mW/cm2 | Isoniazid and Rifampin | 30 min |
| Filho et al. (2010) [33] | 20 | Human | 1.0 MHz | Pulsed, 0.5 W/cm2 | Aloe Vera | 2 min, for 7 days |
| Davick et al. (1988) [29] | 9 | In Vitro | 0.87 MHz | Not Specified, 0.5 mW/cm2 | Cortisol Cream | 8 min |
| Luksurapan et al. (2013) [26] | 23 | In Vitro | 1.0 MHz | Continuous, 1.0 W/cm2 | Piroxicam | 10 min, 5 times a week, for 2 weeks |
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kassan, D.G.; Lynch, A.M.; Stiller, M.J. Physical enhancement of dermatologic drug delivery: Iontophoresis and phonophoresis. J. Am. Acad. Dermatol. 1996, 34, 657–666. [Google Scholar] [CrossRef]
- Speed, C.A. Therapeutic ultrasound in soft tissue lesions. Rheumatology 2001, 40, 1331–1336. [Google Scholar] [CrossRef] [PubMed]
- Polat, B.E.; Hart, D.; Langer, R.; Blankschtein, D. Ultrasound-mediated transdermal drug delivery: Mechanisms, scope, and emerging trends. J. Control. Release 2011, 152, 330–348. [Google Scholar] [CrossRef]
- Uhlemann, C.; Heinig, B.; Wollina, U. Therapeutic Ultrasound in Lower Extremity Wound Management. Int. J. Low. Extremity Wounds 2003, 2, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Dragicevic, N.; Maibach, H.I. (Eds.) Percutaneous Penetration Enhancers Physical Methods in Penetration Enhancement, 1st ed.; Springer: Berlin, Germany, 2017. [Google Scholar] [CrossRef]
- Leong, T.; Ashokkumar, M.; Kentish, S. The Growth of Bubbles in an Acoustic Field by Rectified Diffusion. In Handbook of Ultrasonics and Sonochemistry; Springer: Singapore, 2016; pp. 69–98. [Google Scholar] [CrossRef]
- LuTheryn, G.; Glynne-Jones, P.; Webb, J.; Carugo, D. Ultrasound-mediated therapies for the treatment of biofilms in chronic wounds: A review of present knowledge. Microb. Biotechnol. 2020, 13, 613–628. [Google Scholar] [CrossRef]
- Stoodley, P.; Sauer, K.; Davies, D.G.; Costerton, J.W. Biofilms as Complex Differentiated Communities. Annu. Rev. Microbiol. 2002, 56, 187–209. [Google Scholar] [CrossRef]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival Mechanisms of Clinically Relevant Microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef] [PubMed]
- James, G.A.; Swogger, E.; Wolcott, R.; Pulcini, E.D.; Secor, P.; Sestrich, J.; Costerton, J.W.; Stewart, P.S. Biofilms in chronic wounds. Wound Repair Regen. 2008, 16, 37–44. [Google Scholar] [CrossRef]
- Xavier, J.B.; Foster, K.R. Cooperation and conflict in microbial biofilms. Proc. Natl. Acad. Sci. USA 2007, 104, 876–881. [Google Scholar] [CrossRef]
- Hibbing, M.E.; Fuqua, C.; Parsek, M.R.; Peterson, S.B. Bacterial competition: Surviving and thriving in the microbial jungle. Nat. Rev. Microbiol. 2010, 8, 15–25. [Google Scholar] [CrossRef]
- Phillips, P.L.; Wolcott, R.D.; Fletcher, J.; Schultz, G.S. Biofilms Made Easy. Wounds International. 2010. Available online: https://www.woundsinternational.com/resources/details/biofilms-made-easy (accessed on 1 August 2022).
- Hu, Y.; Ruan, X.; Lv, X.; Xu, Y.; Wang, W.; Cai, Y.; Ding, M.; Dong, H.; Shao, J.; Yang, D.; et al. Biofilm microenvironment-responsive nanoparticles for the treatment of bacterial infection. Nano Today 2022, 46, 101602. Available online: https://www.sciencedirect.com/science/article/abs/pii/S1748013222002304?via%3Dihub (accessed on 1 August 2022). [CrossRef]
- Zhu, J.; Tian, J.; Yang, C.; Chen, J.; Wu, L.; Fan, M.; Cai, X. L-Arg-Rich Amphiphilic Dendritic Peptide as a Versatile NO Donor for NO/Photodynamic Synergistic Treatment of Bacterial Infections and Promoting Wound Healing. Small 2021, 17, 2101495. [Google Scholar] [CrossRef]
- Ma, W.; Chen, X.; Fu, L.; Zhu, J.; Fan, M.; Chen, J.; Yang, C.; Yang, G.; Wu, L.; Mao, G.; et al. Ultra-efficient Antibacterial System Based on Photodynamic Therapy and CO Gas Therapy for Synergistic Antibacterial and Ablation Biofilms. ACS Appl. Mater. Interfaces 2020, 12, 22479–22491. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Tian, J.; Zhu, J.; Chen, J.; Li, L.; Yang, C.; Chen, J.; Chen, D. Photodynamic and photothermal co-driven co-enhanced multi-mode synergistic antibacterial nanoplatform to effectively fight against biofilm infections. Chem. Eng. J. 2021, 426, 131919. Available online: https://www.sciencedirect.com/science/article/abs/pii/S1385894721034999 (accessed on 1 August 2022). [CrossRef]
- Kataoka, Y.; Kunimitsu, M.; Nakagami, G.; Koudounas, S.; Weller, C.D.; Sanada, H. Effectiveness of ultrasonic debridement on reduction of bacteria and biofilm in patients with chronic wounds: A scoping review. Int. Wound J. 2021, 18, 176–186. [Google Scholar] [CrossRef]
- Horsley, H.; Owen, J.; Browning, R.; Carugo, D.; Malone-Lee, J.; Stride, E.; Rohn, J. Ultrasound-activated microbubbles as a novel intracellular drug delivery system for urinary tract infection. J. Control. Release 2019, 301, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Römling, U.; Balsalobre, C. Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 2012, 272, 541–561. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.N.; Fathali, M.; Naghdi, S.; Bartley, J.; Rastak, M.S. Treatment of chronic rhinosinusitis using erythromycin phonophoresis. Physiother. Theory Pract. 2013, 29, 159–165. [Google Scholar] [CrossRef]
- Chen, S.; Qin, M.; Han, Y.; Zhao, L.; Fu, Y.; Shang, Y.; Liu, Z.; Huang, H. Assessment of the efficacy of drug transdermal delivery by electro-phonophoresis in treating tuberculous lymphadenitis. Drug Deliv. 2016, 23, 1588–1593. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Xu, Y.; Li, P.; Wang, C.; Cao, Y.; Yu, J. Antibiofilm effect of ultrasound combined with microbubbles against Staphylococcus epidermidis biofilm. Int. J. Med. Microbiol. 2017, 307, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Cagnie, B.; Vinck, E.; Rimbaut, S.; Vanderstraeten, G. Phonophoresis Versus Topical Application of Ketoprofen: Comparison Between Tissue and Plasma Levels. Phys. Ther. 2003, 83, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Saliba, S.; Mistry, D.J.; Perrin, D.H.; Gieck, J.; Weltman, A. Phonophoresis and the absorption of dexamethasone in the presence of an occlusive dressing. J. Athl. Train. 2007, 42, 349–354. [Google Scholar] [PubMed]
- Luksurapan, W.; Boonhong, J. Effects of Phonophoresis of Piroxicam and Ultrasound on Symptomatic Knee Osteoarthritis. Arch. Phys. Med. Rehabil. 2013, 94, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Ay, S.; Doğan, S.K.; Evcik, D.; Başer, O.C. Comparison the efficacy of phonophoresis and ultrasound therapy in myofascial pain syndrome. Rheumatol. Int. 2011, 31, 1203–1208. [Google Scholar] [CrossRef]
- Cardoso, L.; Pinto, N.; Nobre, M.; Silva, M.; Pires, G.; Lopes, M.; Viana, G.; Rodrigues, L. Anti-inflammatory and antinociceptive effects of phonophoresis in animal models: A randomized experimental study. Braz. J. Med. Biol. Res. 2019, 52, e7773. [Google Scholar] [CrossRef]
- Davick, J.P.; Martin, R.K.; Albright, J.P. Distribution and Deposition of Tritiated Cortisol Using Phonophoresis. Phys. Ther. 1988, 68, 1672–1675. [Google Scholar] [CrossRef] [PubMed]
- Klaiman, M.D.; Shrader, J.A.; Danoff, J.V.; Hicks, J.E.; Pesce, W.J.; Ferland, J. Phonophoresis versus ultrasound in the treatment of common musculoskeletal conditions. Med. Sci. Sports Exerc. 1998, 30, 1349–1355. [Google Scholar] [CrossRef]
- Kozanoglu, E.; Basaran, S.; Guzel, R.; Guler-Uysal, F. Short term efficacy of ibuprofen phonophoresis versus continuous ultrasound therapy in knee osteoarthritis. Swiss Med. Wkly. 2003, 133, 333–338. [Google Scholar]
- Fares, A.; Khaled, D.; Khalaf, M.; Elsayed, A.; Ghazal, R. Garlic extract and phonophoresis in wound healing: Histological and immunohistochemical study. Egypt. J. Histol. 2017, 40, 315–327. [Google Scholar] [CrossRef]
- Filho, A.L.M.M.; Villaverde, A.B.; Munin, E.; Aimbire, F.; Albertini, R. Comparative Study of the Topical Application of Aloe Vera Gel, Therapeutic Ultrasound and Phonophoresis on the Tissue Repair in Collagenase-Induced Rat Tendinitis. Ultrasound Med. Biol. 2010, 36, 1682–1690. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ead, J.K.; Sharma, A.; Goransson, M.; Armstrong, D.G. Potential Utility of Ultrasound-Enhanced Delivery of Antibiotics, Anti-Inflammatory Agents, and Nutraceuticals: A Mini Review. Antibiotics 2022, 11, 1290. https://doi.org/10.3390/antibiotics11101290
Ead JK, Sharma A, Goransson M, Armstrong DG. Potential Utility of Ultrasound-Enhanced Delivery of Antibiotics, Anti-Inflammatory Agents, and Nutraceuticals: A Mini Review. Antibiotics. 2022; 11(10):1290. https://doi.org/10.3390/antibiotics11101290
Chicago/Turabian StyleEad, J. Karim, Arjun Sharma, Miranda Goransson, and David G. Armstrong. 2022. "Potential Utility of Ultrasound-Enhanced Delivery of Antibiotics, Anti-Inflammatory Agents, and Nutraceuticals: A Mini Review" Antibiotics 11, no. 10: 1290. https://doi.org/10.3390/antibiotics11101290
APA StyleEad, J. K., Sharma, A., Goransson, M., & Armstrong, D. G. (2022). Potential Utility of Ultrasound-Enhanced Delivery of Antibiotics, Anti-Inflammatory Agents, and Nutraceuticals: A Mini Review. Antibiotics, 11(10), 1290. https://doi.org/10.3390/antibiotics11101290








