Medical-Grade Honey as an Alternative Treatment for Antibiotics in Non-Healing Wounds—A Prospective Case Series
Abstract
:1. Introduction
2. Results
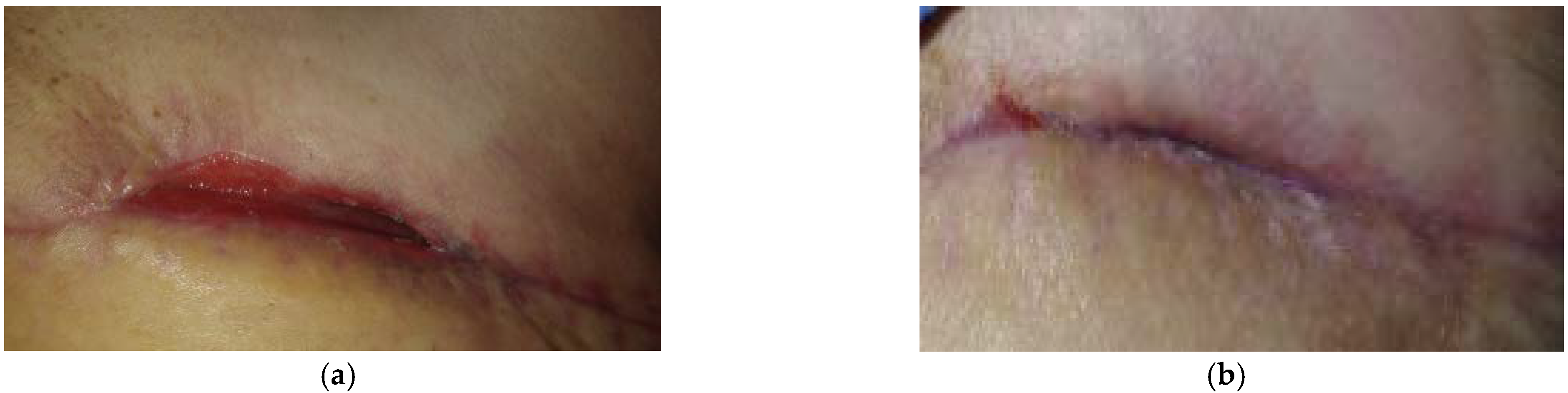
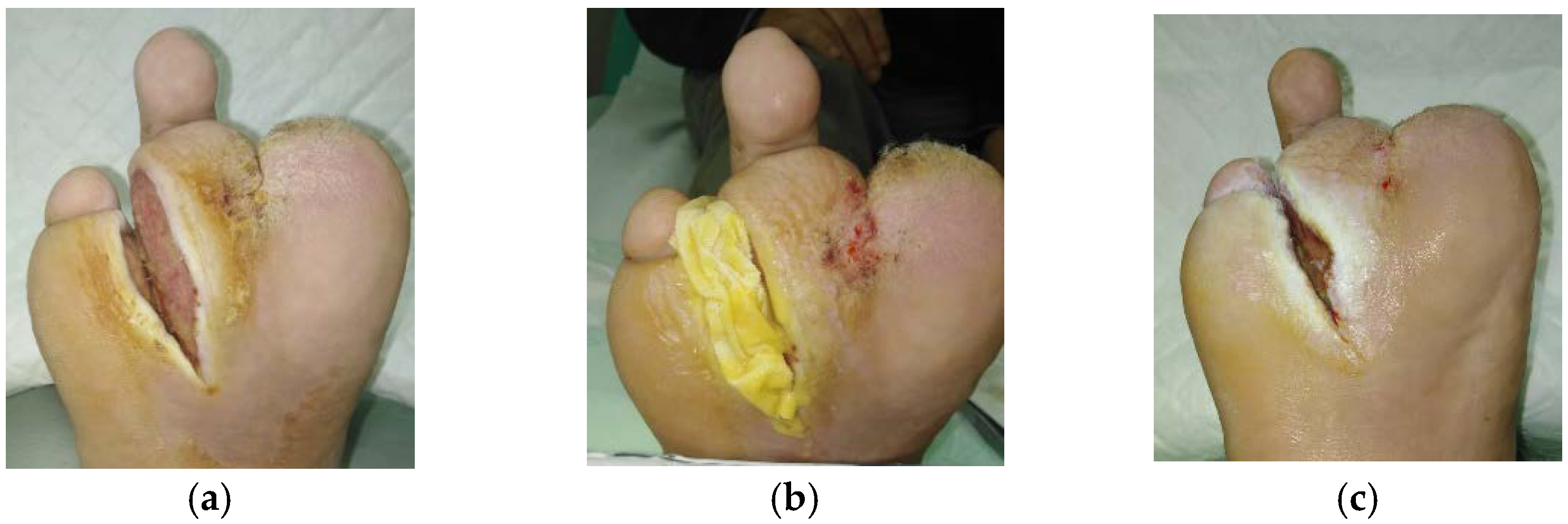
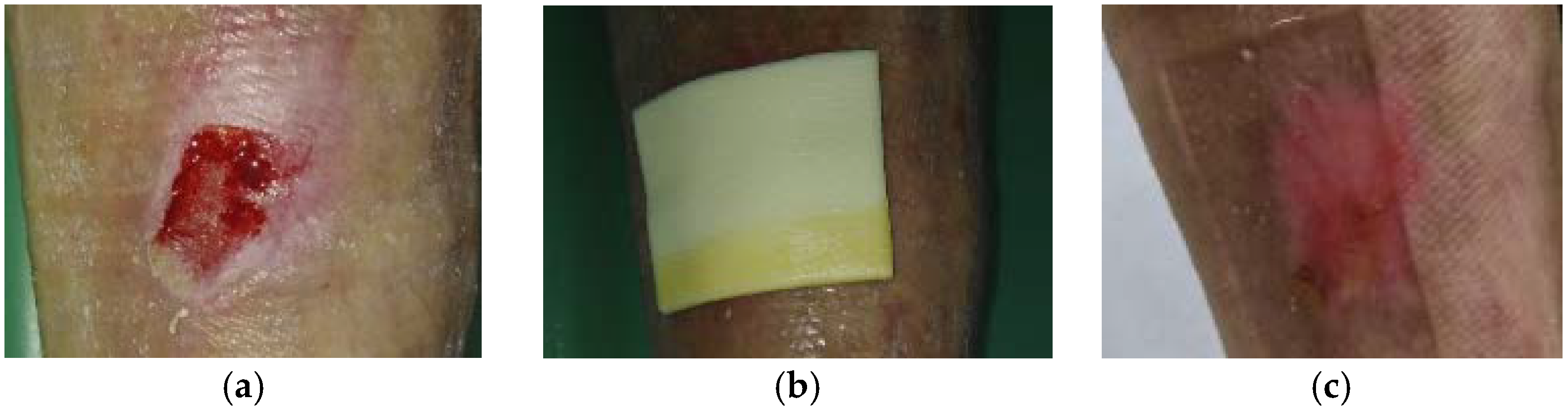
2.1. Case 1: Dehisced Surgical Wound
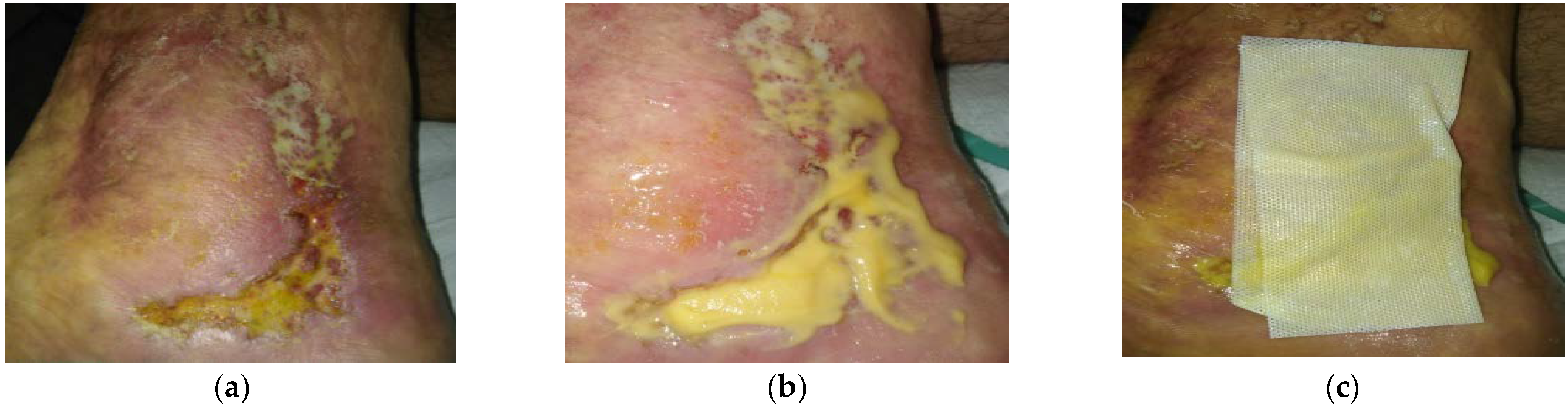
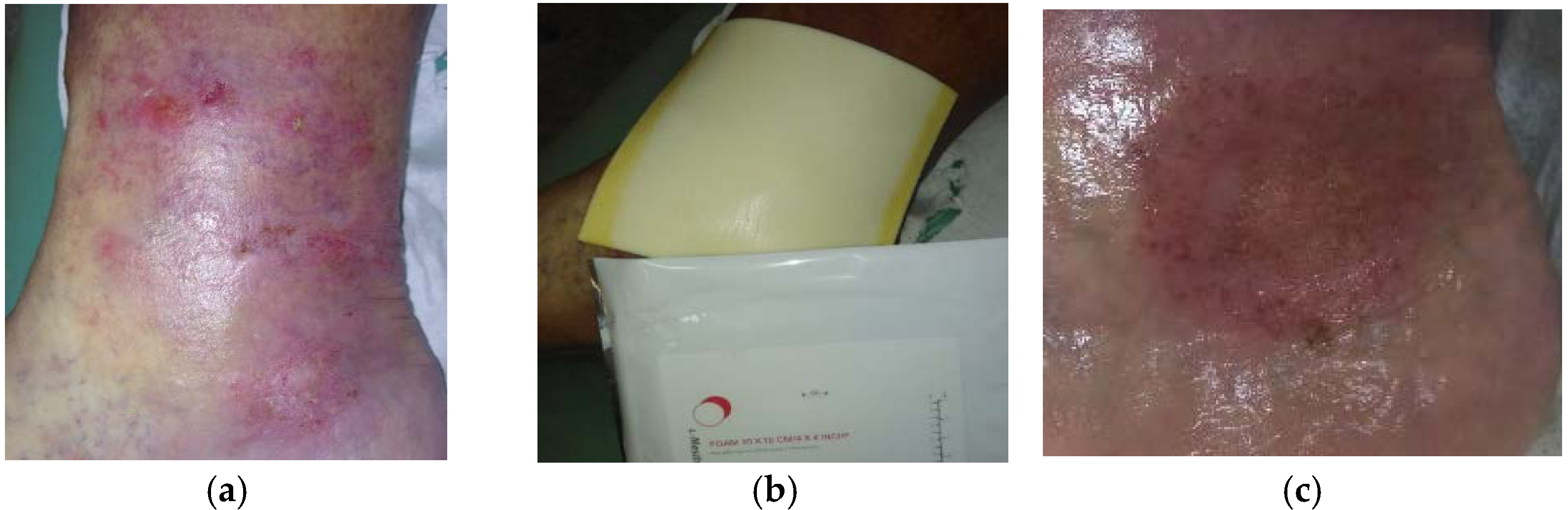
2.2. Case 2: Venous Leg Ulcer
2.3. Case 3: Venous Leg Ulcer
2.4. Case 4: Venous Leg Ulcer
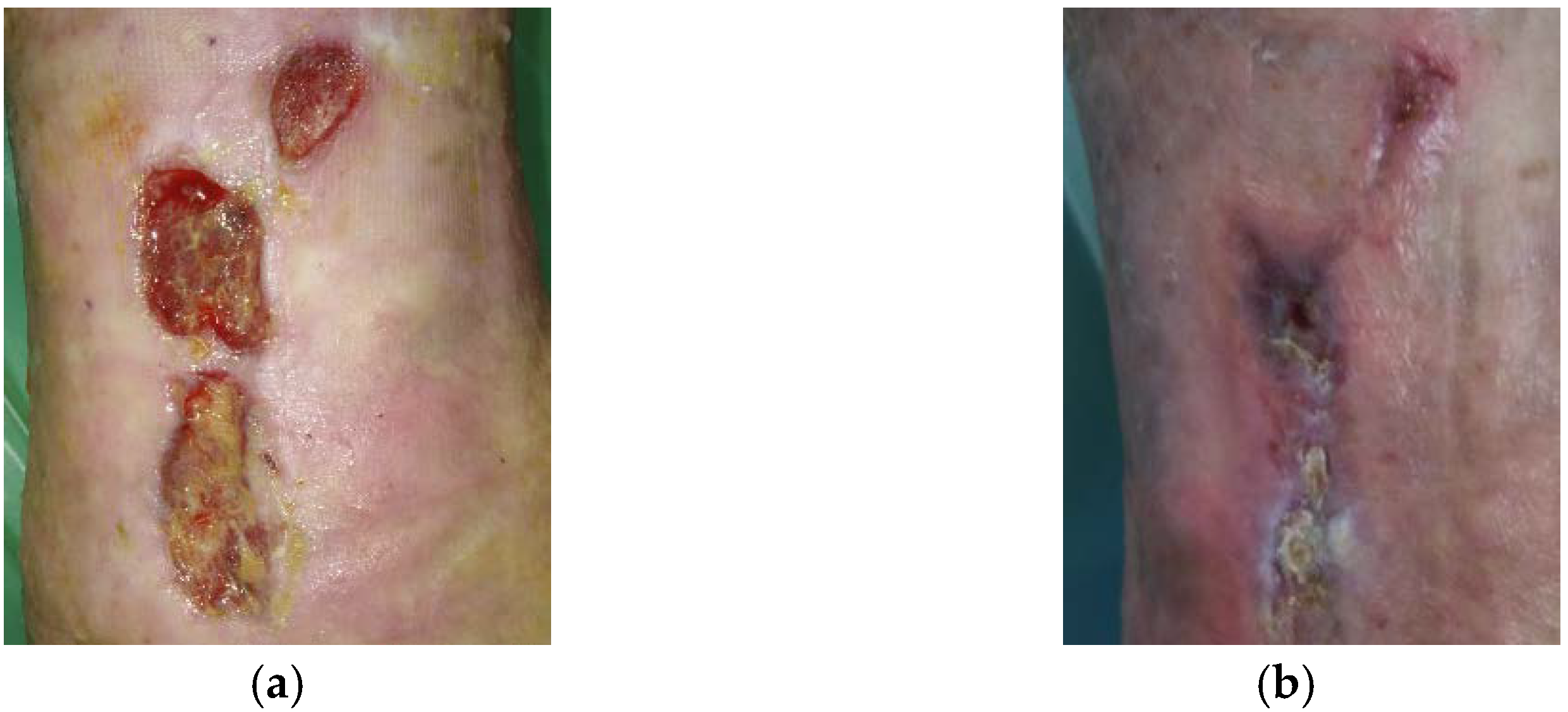
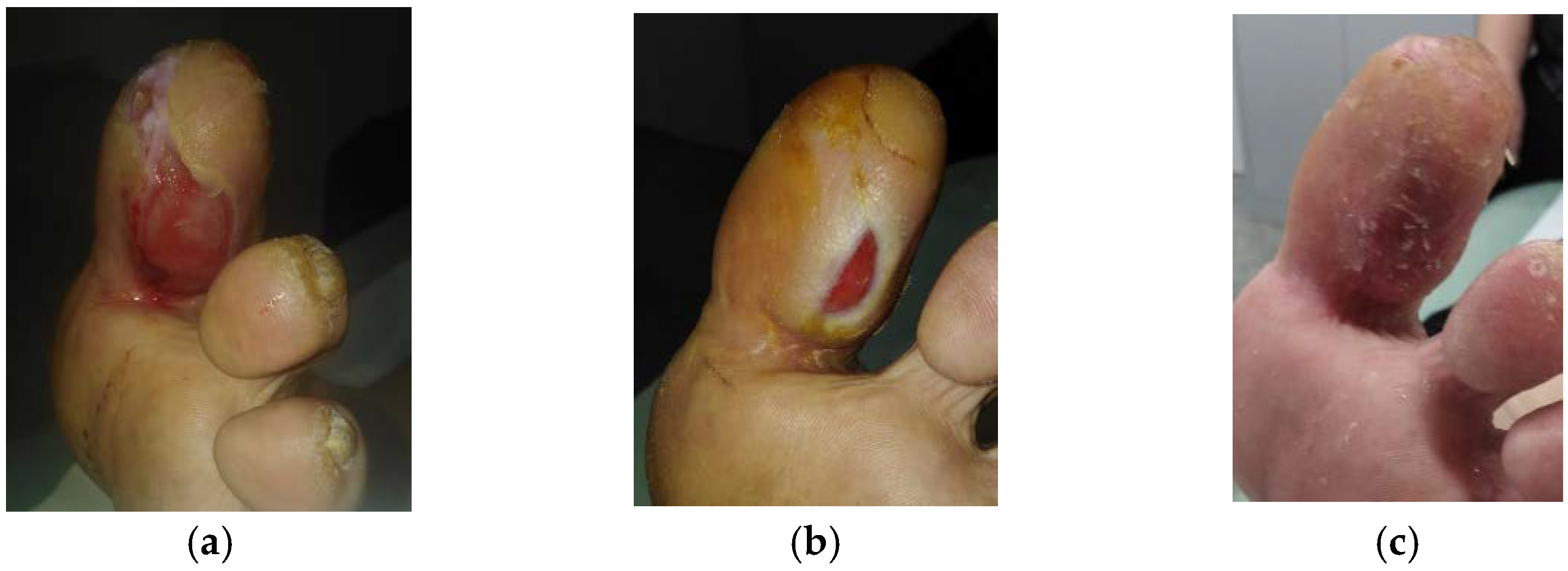
2.5. Case 5: Diabetic Foot Ulcer
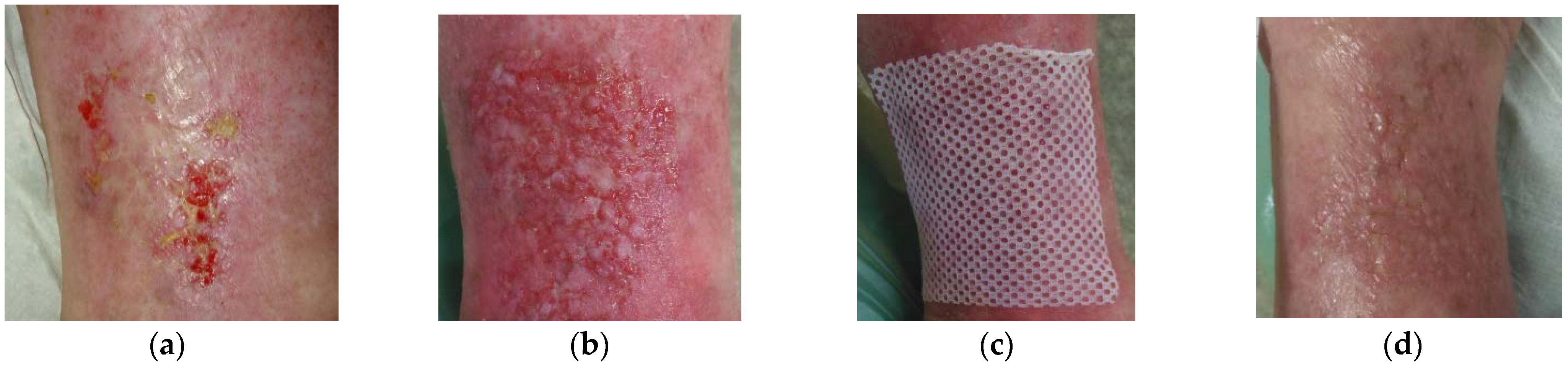
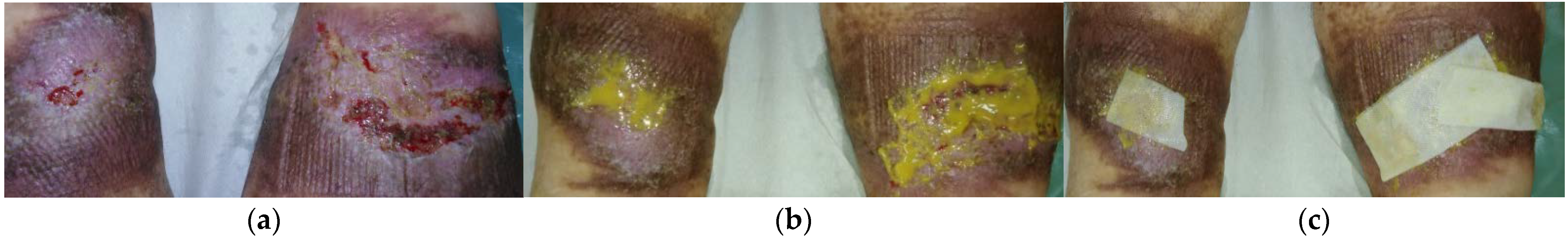
2.6. Case 6: Venous Leg Ulcer
2.7. Case 7: Venous Leg Ulcer
2.8. Case 8: Diabetic Foot Ulcer
2.9. Case 9: Venous Leg Ulcer
3. Discussion
4. Materials and Methods
Patient Selection
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Union of Wound Healing Societies (WUWHS). Florence Congress, Position Document. Advance in Wound Care: The Triangle of Wound Assessment. Wounds Int. 2016. Available online: https://www.coloplast.sg/Documents/Wound/WUWHS_POSITION%20DOCUMENT.pdf (accessed on 27 July 2020).
- Pokorna, A.; Benesova, K.; Muzik, J.; Jarkovsky, J.; Dusek, L. Data Sources for Monitoring of Non-healing Wounds in a National Heal Information System—Epidemiology of Non-healing Wounds Analysis of the National Register of Hospitalized Patients in 2007–2015. Cesk. Slov. Neurol. 2017, 80, 8–17. [Google Scholar] [CrossRef]
- Deufert, D.; Graml, R. Disease-specific, health-related quality of life (HRQoL) of people with chronic wounds—A descriptive cross-sectional study using the Wound-QoL. Wound Med. 2017, 16, 29–33. [Google Scholar] [CrossRef]
- Krupova, L.; Pokorna, A. Quality of life in patient with non-healing wounds, with particular focus on assesment tools-a literature review. Cent. Eur. J. Nurs. Midwifery 2020, 11, 94–103. [Google Scholar] [CrossRef]
- Woo, K.Y.; Krasner, D.L.; Kennedy, B.; Wardle, D.; Moir, O. Palliative wound care management strategies for palliative patients and their circles of care. Adv. Skiin Wound Care 2015, 28, 130–140, quiz 140–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gray, L. Standardising accurate wound assessment and documentation. J. Community Nurs. 2020, 34, 14. [Google Scholar]
- McGuckin, K.R.; IMI National Guidelines A Guide to Good Practice Wound Management Photography. Institute of Medical Illustrators. 2019. Available online: https://www.imi.org.uk/wp-content/uploads/2019/04/2019_Apr_IMINatGuidelines_Wound-Management.pdf (accessed on 10 May 2021).
- Anghel, E.L.; Kumar, A.; Bigham, T.E.; Maselli, K.M.; Steinberg, J.S.; Evans, K.K.; Kim, P.J.; Attinger, C.E. The Reliability of a Novel Mobile 3-dimensional Wound Measurement Device. Wounds 2016, 28, 379–386. [Google Scholar] [PubMed]
- Pokorna, A.; Leaper, D. Assessment and documentation of non-healing, chronic wounds in inpatient health care facilities in the Czech Republic: An evaluation study. Int. Wound J. 2015, 12, 224–231. [Google Scholar] [CrossRef]
- Schreml, S.; Szeimies, R.M.; Prantl, L.; Karrer, S.; Landthaler, M.; Babilas, P. Oxygen in acute and chronic wound healing. Br. J. Derm. 2010, 163, 257–268. [Google Scholar] [CrossRef]
- Guo, S.; Dipietro, L.A. Factors affecting wound healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Dowsett, C.; Hallern, B. The Triangle of Wound Assessment: A holistic framework from wound assessment to management goals and treatments. Wounds Int. 2017, 8, 34–39. [Google Scholar]
- Cremers, N.; Belas, A.; Santos Costa, S.; Couto, I.; de Rooster, H.; Pomba, C. In vitro antimicrobial efficacy of two medical grade honey formulations against common high-risk meticillin-resistant staphylococci and Pseudomonas spp. pathogens. Vet. Derm. 2020, 31, 90–96. [Google Scholar] [CrossRef]
- Smaropoulos, E.; Cremers, N.A.J. Treating severe wounds in pediatrics with medical grade honey: A case series. Clin. Case Rep. 2020, 8, 469–476. [Google Scholar] [CrossRef] [Green Version]
- Bianchi, T.; Wolcott, R.D.; Peghetti, A.; Leaper, D.; Cutting, K.; Polignano, R.; Rosa Rita, Z.; Moscatelli, A.; Greco, A.; Romanelli, M.; et al. Recommendations for the management of biofilm: A consensus document. J. Wound Care 2016, 25, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Sibbald, R.G.; Williamson, D.; Orsted, H.L.; Campbell, K.; Keast, D.; Krasner, D.; Sibbald, D. Preparing the wound bed--debridement, bacterial balance, and moisture balance. Ostomy Wound Manag. 2000, 46, 14–22. [Google Scholar]
- Strohal, R.; Dissemond, J.; Jordan O’Brien, J.; Piaggesi, A.; Rimdeika, R.; Young, T.; Apelqvist, J. EWMA document: Debridement. An updated overview and clarification of the principle role of debridement. J. Wound Care 2013, 22, 5. [Google Scholar] [CrossRef] [Green Version]
- Lumbers, M. Wound debridement: Choices and practice. Br. J. Nurs. 2018, 27, S16–S20. [Google Scholar] [CrossRef] [PubMed]
- Stryja, J. The Importance and Limits of the Pressure Ulcer Surgical Debridement. Česká A Slov. Neurol. A Neurochir. 2016, 79, S25–S27. [Google Scholar] [CrossRef]
- Manna, B.; Nahirniak, P.; Morrison, C.A. Wound Debridement. In StatPearls [Online]; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK507882/ (accessed on 10 June 2021).
- Nair, H.K.R.; Tatavilis, N.; Pospisilova, I.; Kucerova, J.; Cremers, N.A.J. Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series. Antibiotics 2020, 9, 529. [Google Scholar] [CrossRef]
- Hermanns, R.; Mateescu, C.; Thrasyvoulou, A.; Tananaki, C.; Wagener, F.A.D.T.G.; Cremers, N.A.J. Defining the standards for medical grade honey. J. Apic. Res. 2020, 59, 125–135. [Google Scholar] [CrossRef]
- Nolan, V.C.; Harrison, J.; Cox, J.A.G. Dissecting the Antimicrobial Composition of Honey. Antibiotics 2019, 8, 251. [Google Scholar] [CrossRef] [Green Version]
- Gethin, G.; Cowman, S. Bacteriological changes in sloughy venous leg ulcers treated with manuka honey or hydrogel: An RCT. J. Wound Care 2008, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hermanns, R.; Cremers, N.A.J.; Leeming, J.P.; van der Werf, E.T. Sweet Relief: Determining the Antimicrobial Activity of Medical Grade Honey Against Vaginal Isolates of Candida albicans. J. Fungi 2019, 5, 85. [Google Scholar] [CrossRef] [Green Version]
- Oryan, A.; Alemzadeh, E.; Moshiri, A. Biological properties and therapeutic activities of honey in wound healing: A narrative review and meta-analysis. J. Tissue Viability 2016, 25, 98–118. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.C.; Aygin, D. Honey Dressing in Wound Treatment: A Systematic Review. Complement. Med. 2020, 51, 102388. [Google Scholar] [CrossRef]
- Olsson, M.; Jarbrink, K.; Divakar, U.; Bajpai, R.; Upton, Z.; Schmidtchen, A.; Car, J. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair. Regen. 2019, 27, 114–125. [Google Scholar] [CrossRef] [Green Version]
- Kelechi, T.; Prentice, M.; Madisetti, M.; Brunette, G.; Mueller, M. Palliative Care in the Management of Pain, Odor, and Exudate in Chronic Wounds at the End of Life: A Cohort Study. J. Hosp. Palliat. Nurs. 2017, 19, 17–25. [Google Scholar] [CrossRef]
- Lu, J.; Turnbull, L.; Burke, C.M.; Liu, M.; Carter, D.A.; Schlothauer, R.C.; Whitchurch, C.B.; Harry, E.J. Manuka-type honeys can eradicate biofilms produced by Staphylococcus aureus strains with different biofilm-forming abilities. PeerJ 2014, 2, e326. [Google Scholar] [CrossRef] [Green Version]
- Pleeging, C.C.F.; Coenye, T.; Mossialos, D.; De Rooster, H.; Chrysostomou, D.; Wagener, F.A.D.T.G.; Cremers, N.A.J. Synergistic Antimicrobial Activity of Supplemented Medical-Grade Honey against Pseudomonas aeruginosa Biofilm Formation and Eradication. Antibiotics 2020, 9, 866. [Google Scholar] [CrossRef] [PubMed]
- Biglari, B.; Moghaddam, A.; Santos, K.; Blaser, G.; Buchler, A.; Jansen, G.; Langler, A.; Graf, N.; Weiler, U.; Licht, V.; et al. Multicentre prospective observational study on professional wound care using honey (Medihoney). Int. Wound J. 2013, 10, 252–259. [Google Scholar] [CrossRef]
- Halstead, F.D.; Webber, M.A.; Oppenheim, B.A. Use of an engineered honey to eradicate preformed biofilms of important wound pathogens: An in vitro study. J. Wound Care 2017, 26, 442–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.K.; Cheng, N.C.; Cheng, C.M. Biofilms in Chronic Wounds: Pathogenesis and Diagnosis. Trends Biotechnol. 2019, 37, 505–517. [Google Scholar] [CrossRef] [PubMed]
- Burmolle, M.; Thomsen, T.R.; Fazli, M.; Dige, I.; Christensen, L.; Homoe, P.; Tvede, M.; Nyvad, B.; Tolker-Nielsen, T.; Givskov, M.; et al. Biofilms in chronic infections—A matter of opportunity—Monospecies biofilms in multispecies infections. FEMS Immunol. Med. Microbiol. 2010, 59, 324–336. [Google Scholar] [CrossRef] [Green Version]
- Smaropoulos, E.; Cremers, N.A.J. The pro-healing effects of medical grade honey supported by a pediatric case series. Complement. Med. 2019, 45, 14–18. [Google Scholar] [CrossRef]
- Combarros-Fuertes, P.; Fresno, J.M.; Estevinho, M.M.; Sousa-Pimenta, M.; Tornadijo, M.E.; Estevinho, L.M. Honey: Another Alternative in the Fight against Antibiotic-Resistant Bacteria? Antibiotics 2020, 9, 774. [Google Scholar] [CrossRef] [PubMed]
- de Groot, T.; Janssen, T.; Faro, D.; Cremers, N.A.J.; Chowdhary, A.; Meis, J.F. Antifungal Activity of a Medical-Grade Honey Formulation against Candida auris. J. Fungi 2021, 7, 50. [Google Scholar] [CrossRef]
- Smaropoulos, E.; Cremers, N.A.J. Medical grade honey for the treatment of extravasation-induced injuries in preterm neonates—A case series. Adv. Neonatal. Care 2020, in press. [Google Scholar]
- Lu, J.; Cokcetin, N.N.; Burke, C.M.; Turnbull, L.; Liu, M.; Carter, D.A.; Whitchurch, C.B.; Harry, E.J. Honey can inhibit and eliminate biofilms produced by Pseudomonas aeruginosa. Sci. Rep. 2019, 9, 18160. [Google Scholar] [CrossRef] [Green Version]
- Sochor, M.; Slama, O. Management of chronic and acute pain in patients with cancer diseases. Klin. Onkol. 2015, 28, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Juric Vukelic, D.; Juric, J. Hydrocolloid Dressing Application in the Treatment of Chronic Wounds and Relation to Quality of Life. Acta Clin. Croat. 2017, 56, 544–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenbaum, C. An overview of integrative care options for patients with chronic wounds. Ostomy Wound Manag. 2012, 58, 44–51. [Google Scholar]
- Edwards, J. Dealing with wound-related pain at dressing change. J. Community Nurs. 2013, 27, 36–42. [Google Scholar]
- Upton, D.; Solowiej, K.; Hender, C.; Woodyatt, K.Y. Stress and pain associated with dressing change in patients with chronic wounds. J. Wound Care 2012, 21, 53–54, 56,58 passim. [Google Scholar] [CrossRef] [PubMed]
- Alam, F.; Islam, M.A.; Gan, S.H.; Khalil, M.I. Honey: A potential therapeutic agent for managing diabetic wounds. Evid. Based Complement Altern. Med. 2014, 2014, 169130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yapucu Gunes, U.; Eser, I. Effectiveness of a honey dressing for healing pressure ulcers. J. Wound Ostomy Cont. Nurs. 2007, 34, 184–190. [Google Scholar] [CrossRef]
- Molan, P.C. Re-introducing honey in the management of wounds and ulcers—theory and practice. Ostomy Wound Manag. 2002, 48, 28–40. [Google Scholar]
- Labban, L. Honey as a promising treatment for diabetic foot ulcers (DFU). J. Med. Soc. 2014, 28, 64–68. [Google Scholar] [CrossRef] [Green Version]
- Angel, D.E.; Lloyd, P.; Carville, K.; Santamaria, N. The clinical efficacy of two semi-quantitative wound-swabbing techniques in identifying the causative organism(s) in infected cutaneous wounds. Int. Wound J. 2011, 8, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Singhal, N.; Kumar, M.; Kanaujia, P.K.; Virdi, J.S. MALDI-TOF mass spectrometry: An emerging technology for microbial identification and diagnosis. Front. Microbiol. 2015, 6, 791. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holubová, A.; Chlupáčová, L.; Cetlová, L.; Cremers, N.A.J.; Pokorná, A. Medical-Grade Honey as an Alternative Treatment for Antibiotics in Non-Healing Wounds—A Prospective Case Series. Antibiotics 2021, 10, 918. https://doi.org/10.3390/antibiotics10080918
Holubová A, Chlupáčová L, Cetlová L, Cremers NAJ, Pokorná A. Medical-Grade Honey as an Alternative Treatment for Antibiotics in Non-Healing Wounds—A Prospective Case Series. Antibiotics. 2021; 10(8):918. https://doi.org/10.3390/antibiotics10080918
Chicago/Turabian StyleHolubová, Adéla, Lucie Chlupáčová, Lada Cetlová, Niels A. J. Cremers, and Andrea Pokorná. 2021. "Medical-Grade Honey as an Alternative Treatment for Antibiotics in Non-Healing Wounds—A Prospective Case Series" Antibiotics 10, no. 8: 918. https://doi.org/10.3390/antibiotics10080918
APA StyleHolubová, A., Chlupáčová, L., Cetlová, L., Cremers, N. A. J., & Pokorná, A. (2021). Medical-Grade Honey as an Alternative Treatment for Antibiotics in Non-Healing Wounds—A Prospective Case Series. Antibiotics, 10(8), 918. https://doi.org/10.3390/antibiotics10080918






