Evaluation of the Clinical Use of Ceftriaxone among In-Patients in Selected Health Facilities in Uganda
Abstract
1. Introduction
2. Results
2.1. Social Demographic Characteristics
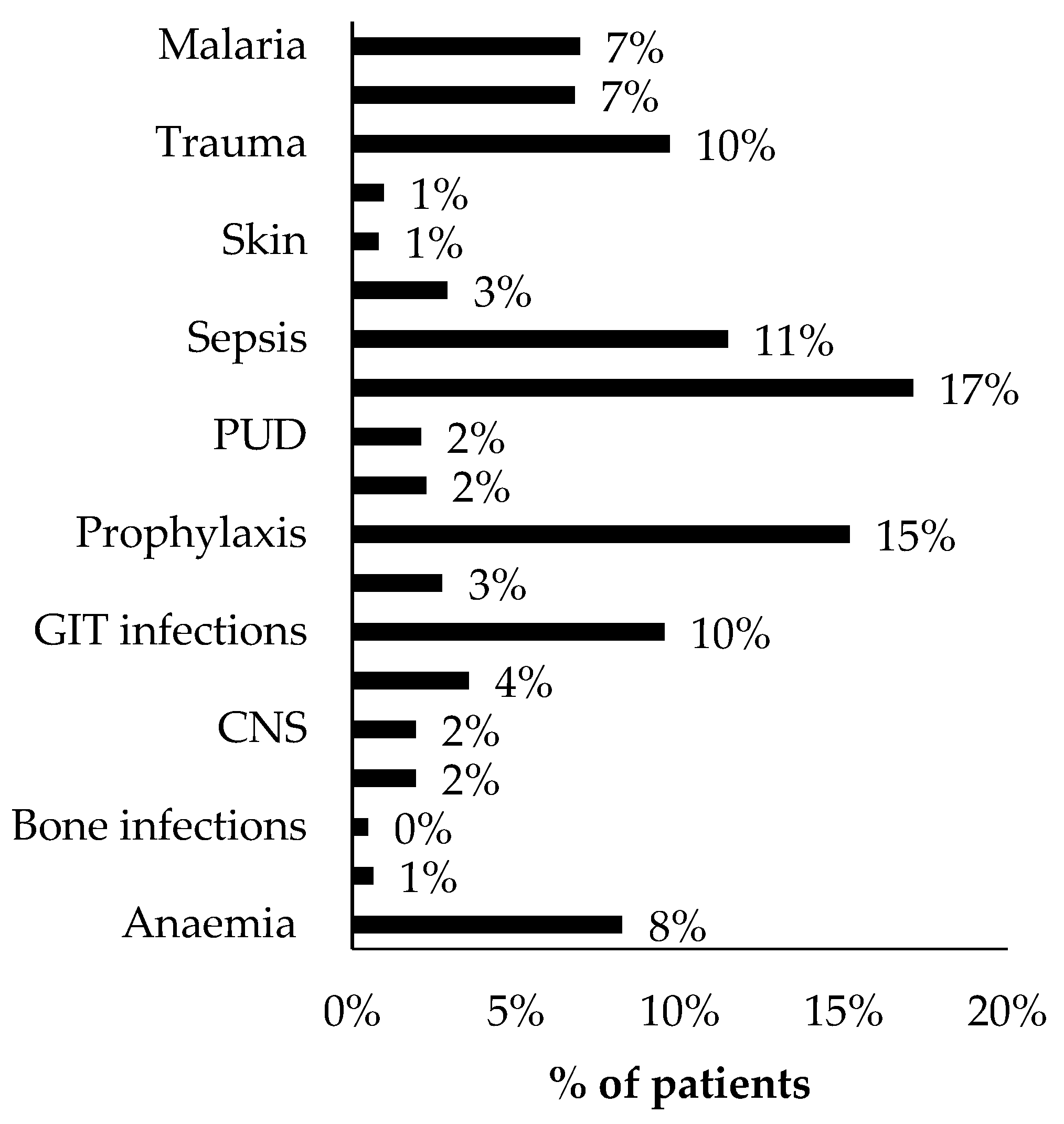
2.2. Appropriateness of Ceftriaxone Use
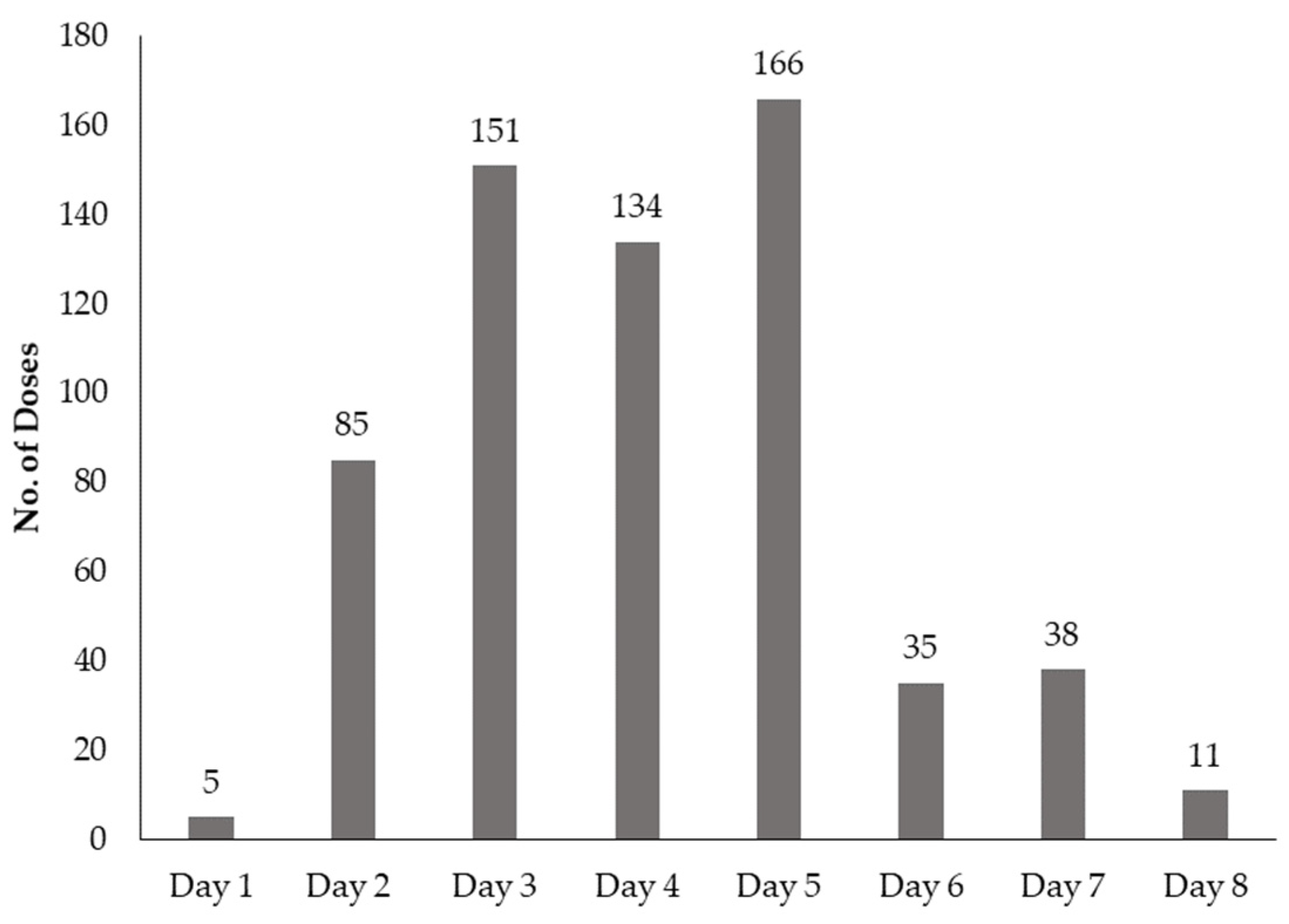
2.3. Ceftriaxone Doses, Dose Frequencies, and Dose Durations Prescribed
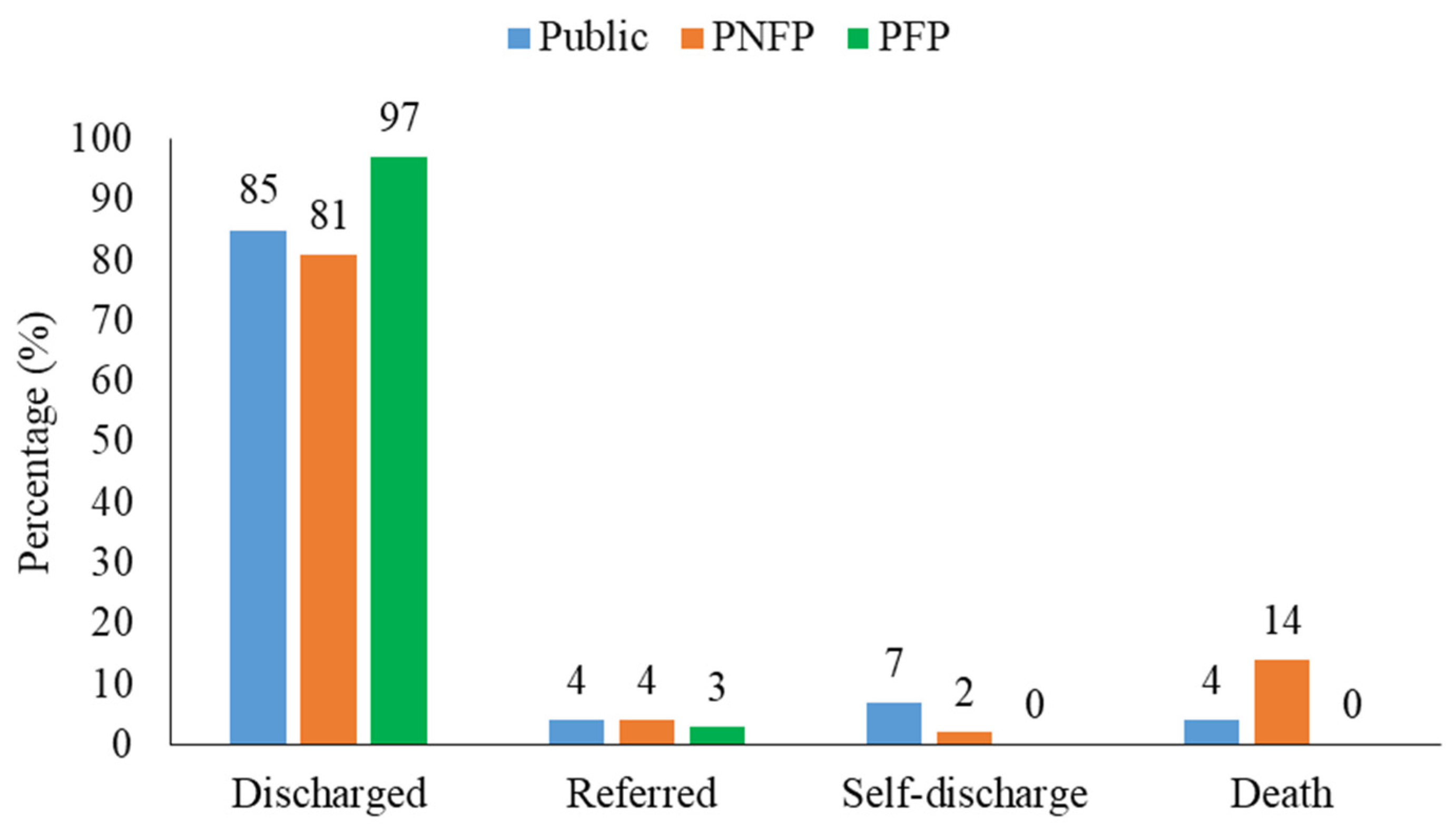
2.4. Treatment Outcomes
2.5. Factors Associated with Appropriate Ceftriaxone Use
3. Discussion
4. Materials and Methods
4.1. Study Setting and Design
4.2. Sample Size Determination and Sampling Procedure
4.3. Data Collection
4.4. Data Handling and Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alvarez-Uria, G.; Zachariah, S.; Thomas, D. High prescription of antimicrobials in a rural district hospital in India. Pharm. Pract. (Granada) 2014, 12, 384. [Google Scholar] [CrossRef]
- Bajis, S.; Van den Bergh, R.; De Bruycker, M.; Mahama, G.; Van Overloop, C.; Satyanarayana, S.; Bernardo, R.S.; Esmati, S.; Reid, A.J. Antibiotic use in a district hospital in Kabul, Afghanistan: Are we overprescribing? Public Health Action 2014, 4, 259–264. [Google Scholar] [CrossRef][Green Version]
- Lee, H.; Jung, D.; Yeom, J.S.; Son, J.S.; Jung, S.I.; Kim, Y.S.; Kim, C.K.; Chang, H.H.; Kim, S.W.; Ki, H.K.; et al. Evaluation of ceftriaxone utilization at multicenter study. Korean J. Intern. Med. 2009, 24, 374–380. [Google Scholar] [CrossRef]
- Saleh, N.; Awada, S.; Awwad, R.; Jibai, S.; Arfoul, C.; Zaiter, L.; Dib, W.; Salameh, P. Evaluation of antibiotic prescription in the Lebanese community: A pilot study. Infect. Ecol. Epidemiol. 2015, 5, 27094. [Google Scholar] [CrossRef]
- Wang, J.; Wang, P.; Wang, X.; Zheng, Y.; Xiao, Y. Use and prescription of antibiotics in primary health care settings in China. JAMA Intern. Med. 2014, 174, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.; Alrowaili, M.; Rauf, A.; Eltom, E. Ceftriaxone Drug Utilization Evaluation (DUE) at Prince Abdulaziz Bin Moussae’ed Hospital, Arar, Kingdom of Saudi Arabia. Sudan J. Med. Sci. 2019. [Google Scholar] [CrossRef]
- Sileshi, A.; Tenna, A.; Feyissa, M.; Shibeshi, W. Evaluation of ceftriaxone utilization in medical and emergency wards of Tikur Anbessa specialized hospital: A prospective cross-sectional study. BMC Pharmacol. Toxicol. 2016, 17, 1–10. [Google Scholar] [CrossRef]
- Uganda Clinical Guidelines (UCG) 2016. Available online: http://health.go.ug/sites/default/files/Uganda%20Clinical%20Guidelines%202016_FINAL.pdf (accessed on 6 February 2021).
- Kiguba, R.; Karamagi, C.; Bird, S.M. Extensive antibiotic prescription rate among hospitalized patients in Uganda: But with frequent missed-dose days. J. Antimicrob. Chemother. 2016, 71, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Muhammed, O.S.; Nasir, B.B. Drug Use Evaluation of Ceftriaxone in Ras-Desta Memorial General Hospital, Ethiopia. Drug Healthc. Patient Saf. 2020, 12, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Eulambius, M.; Osward, N. Evaluation of Ceftriaxone utilization among selected hospitals in Dar-es-Salaam, Tanzania. J. Drug Deliv. Ther. 2019, 9, 7–11. [Google Scholar] [CrossRef]
- Berhe, Y.H.; Amaha, N.D.; Ghebrenegus, A.S. Evaluation of ceftriaxone use in the medical ward of Halibet National Referral and teaching hospital in 2017 in Asmara, Eritrea: A cross sectional retrospective study. BMC Infect. Dis. 2019, 19, 465. [Google Scholar] [CrossRef]
- Manirakiza, L.; Nambasa, V.; Nanyonga, S.; Serwanga, A.; Alphonsus, M.; Denis, N.; Byomire, H. Drug Use Evaluation (DUE) of Ceftriaxone in Mubende Regional Referral Hospital, Uganda: A Cross-Sectional Survey. J. Pharm. Pharmacol. Res. 2019, 3. [Google Scholar] [CrossRef]
- Ayele, A.A.; Gebresillassie, B.M.; Erku, D.A.; Gebreyohannes, E.A.; Demssie, D.G.; Mersha, A.G.; Tegegn, H.G. Prospective evaluation of Ceftriaxone use in medical and emergency wards of Gondar university referral hospital, Ethiopia. Pharmacol. Res. Perspect. 2018, 6, e00383. [Google Scholar] [CrossRef]
- Malik, M.; Alagab, M.; Maatoug, M.; Zahran, F.; Elmubarak, A.; Alhaddad, T.; Omer, S. Evaluation of the Appropriate Use of Ceftriaxone in Internal Medicine Wards of Wad Medani Teaching Hospital in Sudan. Int. J. Med. Sci. Clin. Invent. 2020, 7, 4757–4765. [Google Scholar] [CrossRef]
- Hafte, K.; Tefera, K.; Azeb, W.; Yemsrach, W.; Ayda, R. Assessment of ceftriaxone use in Eastern Ethiopian Referral Hospital: A retrospective study. J. Infect. Dis. Med. Microbiol. 2018, 2, 26–29. [Google Scholar]
- Sewagegn, N.; Ayichew, G.M.; Miskir, A.; Degarege, A.; Mohammed, O.; Andualem, T. Evaluation of Ceftriaxone Use for Hospitalized Patients in Ethiopia: The Case of a Referral Hospital. Int. J. Pharm. Sci. Sci. Res. 2017, 3, 26–31. [Google Scholar]
- Sonda, T.B.; Horumpende, P.G.; Kumburu, H.H.; van Zwetselaar, M.; Mshana, S.E.; Alifrangis, M.; Lund, O.; Aarestrup, F.M.; Chilongola, J.O.; Mmbaga, B.T.; et al. Ceftriaxone use in a tertiary care hospital in Kilimanjaro, Tanzania: A need for a hospital antibiotic stewardship programme. PLoS ONE 2019, 14, e0220261. [Google Scholar] [CrossRef]
- Durham, S.H.; Wingler, M.J.; Eiland, L.S. Appropriate Use of Ceftriaxone in the Emergency Department of a Veteran’s Health Care System. J. Pharm. Technol. 2017, 33, 215–218. [Google Scholar] [CrossRef]
- WHO. Antibacterial Resistance. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 10 May 2021).
- WHO. Global Action on Antimicrobial Resistance 2015. Available online: https://apps.who.int/iris/bitstream/handle/10665/193736/9789241509763_eng.pdf?sequence=1 (accessed on 5 February 2021).
- Tadesse, B.T.; Ashley, E.A.; Ongarello, S.; Havumaki, J.; Wijegoonewardena, M.; González, I.J.; Dittrich, S. Antimicrobial resistance in Africa: A systematic review. BMC Infect. Dis. 2017, 17, 616. [Google Scholar] [CrossRef]
- Di Gennaro, F.; Marotta, C.; Pisani, L.; Veronese, N.; Pisani, V.; Lippolis, V.; Pellizer, G.; Pizzol, D.; Tognon, F.; Bavaro, D.F.; et al. Maternal caesarean section infection (MACSI) in Sierra Leone: A case-control study. Epidemiol. Infect. 2020, 148, e40. [Google Scholar] [CrossRef]
- Hickson, E.; Harris, J.; Brett, D. A journey to zero: Reduction of post-operative cesarean surgical site infections over a five-year period. Surg. Infect. (Larchmt) 2015, 16, 174–177. [Google Scholar] [CrossRef]
- Rickard, J.; Beilman, G.; Forrester, J.; Sawyer, R.; Stephen, A.; Weiser, T.G.; Valenzuela, J. Surgical Infections in Low- and Middle-Income Countries: A Global Assessment of the Burden and Management Needs. Surg. Infect. 2019, 21, 478–494. [Google Scholar] [CrossRef]
- Bonet, M.; Brizuela, V.; Abalos, E.; Cuesta, C.; Baguiya, A.; Chamillard, M.; Fawole, B.; Knight, M.; Kouanda, S.; Lumbiganon, P.; et al. Frequency and management of maternal infection in health facilities in 52 countries (GLOSS): A 1-week inception cohort study. Lancet Glob. Health 2020, 8, e661–e671. [Google Scholar] [CrossRef]
- Government of Uganda. Antimicrobial Resistance National Action Plan: 2018–2023. Available online: https://cddep.org/wp-content/uploads/2018/12/GoU_AMR-NAP.pdf (accessed on 2 January 2021).
- Ampaire, L.; Muhindo, A.; Orikiriza, P.; Mwanga-Amumpaire, J.; Bebell, L.; Boum, Y. A review of antimicrobial resistance in East Africa. Afr. J. Lab. Med. 2016, 5, 432. [Google Scholar] [CrossRef]
- Musaba, M.W.; Kagawa, M.N.; Kiggundu, C.; Kiondo, P.; Wandabwa, J. Cervicovaginal Bacteriology and Antibiotic Sensitivity Patterns among Women with Premature Rupture of Membranes in Mulago Hospital, Kampala, Uganda: A Cross-Sectional Study. Infect. Dis. Obstet. Gynecol. 2017, 2017, 9264571. [Google Scholar] [CrossRef]
- Kajumbula, H.; Fujita, A.W.; Mbabazi, O.; Najjuka, C.; Izale, C.; Akampurira, A.; Aisu, S.; Lamorde, M.; Walwema, R.; Bahr, N.C.; et al. Antimicrobial Drug Resistance in Blood Culture Isolates at a Tertiary Hospital, Uganda. Emerg. Infect. Dis. 2018, 24, 174–175. [Google Scholar] [CrossRef]
- MSH; WHO. Drug and Therapeutics Committee Training Course: Session 11: Drug Use Evaluation; Participants’ Guide; Management Sciences for Health and World Health Organization: Arlington, Virginia, 2007. [Google Scholar]
- WHO. Drug and Therapeutics Committees—A Practical Guide 2003. Available online: https://apps.who.int/medicinedocs/en/d/Js4882e/8.5.html (accessed on 3 April 2020).
- WHO. WHO Model Prescribing Information: Drugs used in Bacterial Infections: Drugs: Ceftriaxone. Available online: https://apps.who.int/medicinedocs/en/d/Js5406e/16.11.html (accessed on 12 February 2021).
- Afriyie, D.K.; Amponsah, S.K.; Dogbey, J.; Agyekum, K.; Kesse, S.; Truter, I.; Meyer, J.C.; Godman, B. A pilot study evaluating the prescribing of ceftriaxone in hospitals in Ghana: Findings and implications. Hosp. Pract. (1995) 2017, 45, 143–149. [Google Scholar] [CrossRef]
- ASHP. Criteria for Drug Use Evaluation. Available online: https://www.ashp.org/-/media/assets/policy-guidelines/docs/guidelines/medication-use-evaluation-current.ashx (accessed on 20 September 2020).
- Guarner, J.; Amukele, T.; Mehari, M.; Gemechu, T.; Woldeamanuel, Y.; Winkler, A.M.; Asrat, D.; Wilson, M.L.; del Rio, C. Building capacity in laboratory medicine in Africa by increasing physician involvement: A laboratory medicine course for clinicians. Am. J. Clin. Pathol. 2015, 143, 405–411. [Google Scholar] [CrossRef]
- Opintan, J.A.; Newman, M.J.; Arhin, R.E.; Donkor, E.S.; Gyansa-Lutterodt, M.; Mills-Pappoe, W. Laboratory-based nationwide surveillance of antimicrobial resistance in Ghana. Infect. Drug Resist. 2015, 8, 379–389. [Google Scholar] [CrossRef]
- Ombelet, S.; Ronat, J.B.; Walsh, T.; Yansouni, C.P.; Cox, J.; Vlieghe, E.; Martiny, D.; Semret, M.; Vandenberg, O.; Jacobs, J. Clinical bacteriology in low-resource settings: Today’s solutions. Lancet Infect. Dis. 2018, 18, e248–e258. [Google Scholar] [CrossRef]
- WHO; FAO; WOAH. Taking a Multisectoral, One Health Approach: A Tripartite Guide to Addressing Zoonotic Diseases in Countries; World Health Organization, Food and Agricultural Organization, World Organisation for Animal Health: Geneva, Switzerland, 2019. [Google Scholar]
- Jonas, O.B.; Irwin, A.; Berthe, F.C.J.; Le Gall, F.G.; Marquez, P.V. Drug-Resistant Infections: A Threat to Our Economic Future: Final Report; HNP/Agriculture Global Antimicrobial Resistance Initiative: Washington, DC, USA, 2017. [Google Scholar]
- Rousham, E.K.; Unicomb, L.; Islam, M.A. Human, animal and environmental contributors to antibiotic resistance in low-resource settings: Integrating behavioural, epidemiological and One Health approaches. Proc. Biol. Sci. 2018, 285, 20180332. [Google Scholar] [CrossRef]
- Kiwanuka, S.N.; Namuhani, N.; Akulume, M.; Kalyesubula, S.; Bazeyo, W.; Kisakye, A.N. Uganda’s laboratory human resource in the era of global health initiatives: Experiences, constraints and opportunities—an assessment of 100 facilities. Hum. Resour. Health 2020, 18, 13. [Google Scholar] [CrossRef]
- Steadman, E.; Raisch, D.W.; Bennett, C.L.; Esterly, J.S.; Becker, T.; Postelnick, M.; McKoy, J.M.; Trifilio, S.; Yarnold, P.R.; Scheetz, M.H. Evaluation of a potential clinical interaction between ceftriaxone and calcium. Antimicrob. Agents Chemother. 2010, 54, 1534–1540. [Google Scholar] [CrossRef]
- Bhagavathula, A.S.; Berhanie, A.; Tigistu, H.; Abraham, Y.; Getachew, Y.; Khan, T.M.; Unakal, C. Prevalence of potential drug–drug interactions among internal medicine ward in University of Gondar Teaching Hospital, Ethiopia. Asian Pac. J. Trop. Biomed. 2014, 4, S204–S208. [Google Scholar] [CrossRef]
- Ministry of Health (MoH) Uganda. Hospitals. Available online: https://www.health.go.ug/hospitals/#:~:text=The%20total%20number%20of%20hospitals,PNFP%20and%2027%20are%20private (accessed on 17 July 2019).
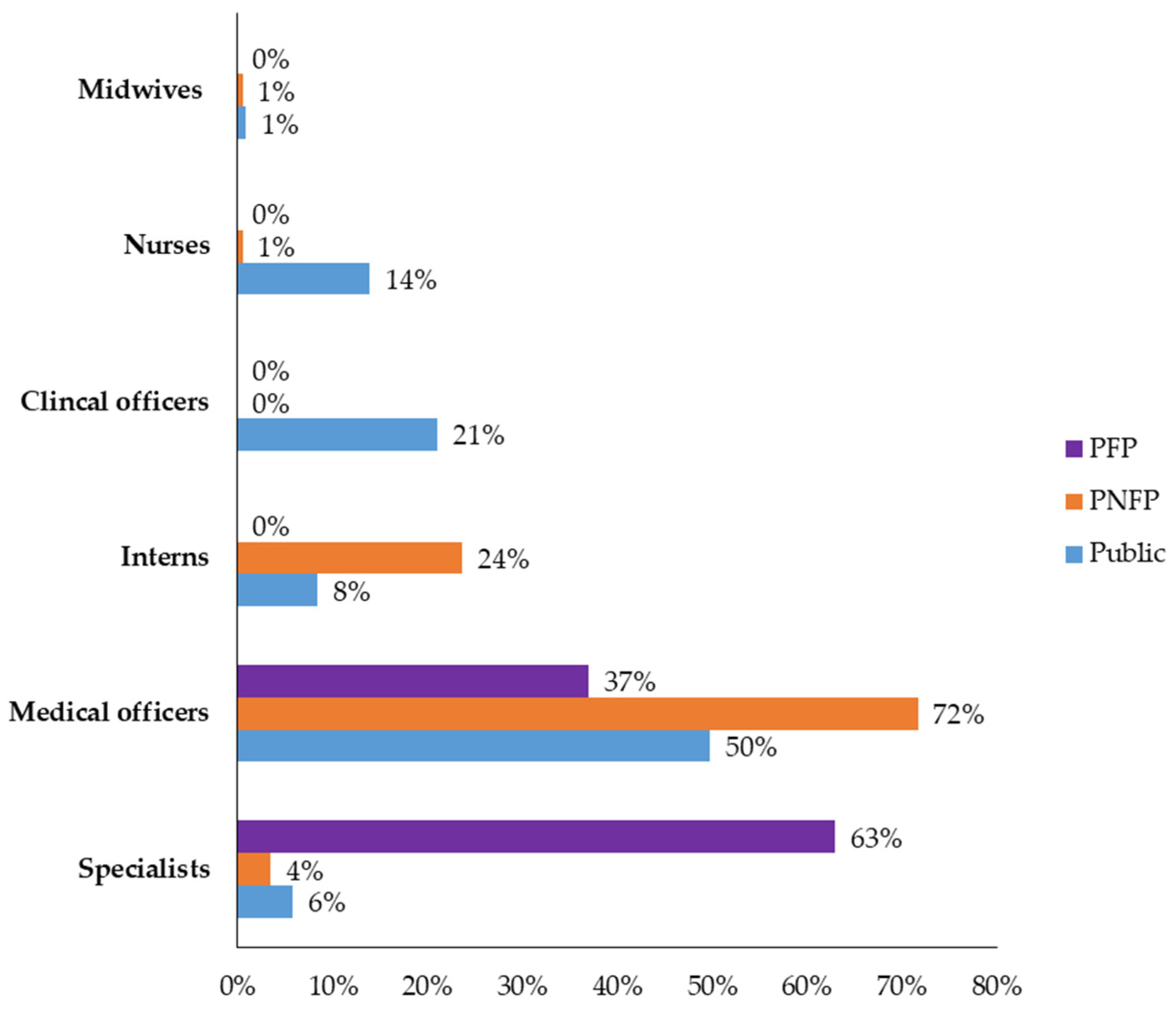
| Prescriber Category | Number of Prescriptions | Percentage |
|---|---|---|
| Medical officers | 470 | 53.3 |
| Clinical officers | 123 | 13.9 |
| Specialists | 104 | 11.8 |
| Medical interns | 96 | 10.9 |
| Nurses | 82 | 9.3 |
| Midwives | 7 | 0.8 |
| Drug Use Indicators | Average Percentage Score (± SD) | Overall Average Percentage Score (± SD) | WHO Target (%) * |
|---|---|---|---|
| Appropriateness of indication | |||
| PNFP | 85.5 (2.0) | ||
| PFP | 97.0 (0.0) | 83.3 (5.6) | 90 |
| Public | 80.5 (2.4) | ||
| Appropriateness of dose | |||
| PNFP | 89.5 (0.5) | ||
| PFP | 92.0 (0.0) | 88.8 (3.2) | 95 |
| Public | 88.0 (3.6) | ||
| Appropriateness of duration | |||
| PNFP | 86.5 (1.5) | ||
| PFP | 90.0 (0.0) | 84.1 (5.7) | 95 |
| Public | 82.3 (6.1) | ||
| Generic prescribing | |||
| PNFP | 66.9 (33.2) | ||
| PFP | 87.0 (0.0) | 87.1 (21.7) | 100 |
| Public | 93.5 (12.7) | ||
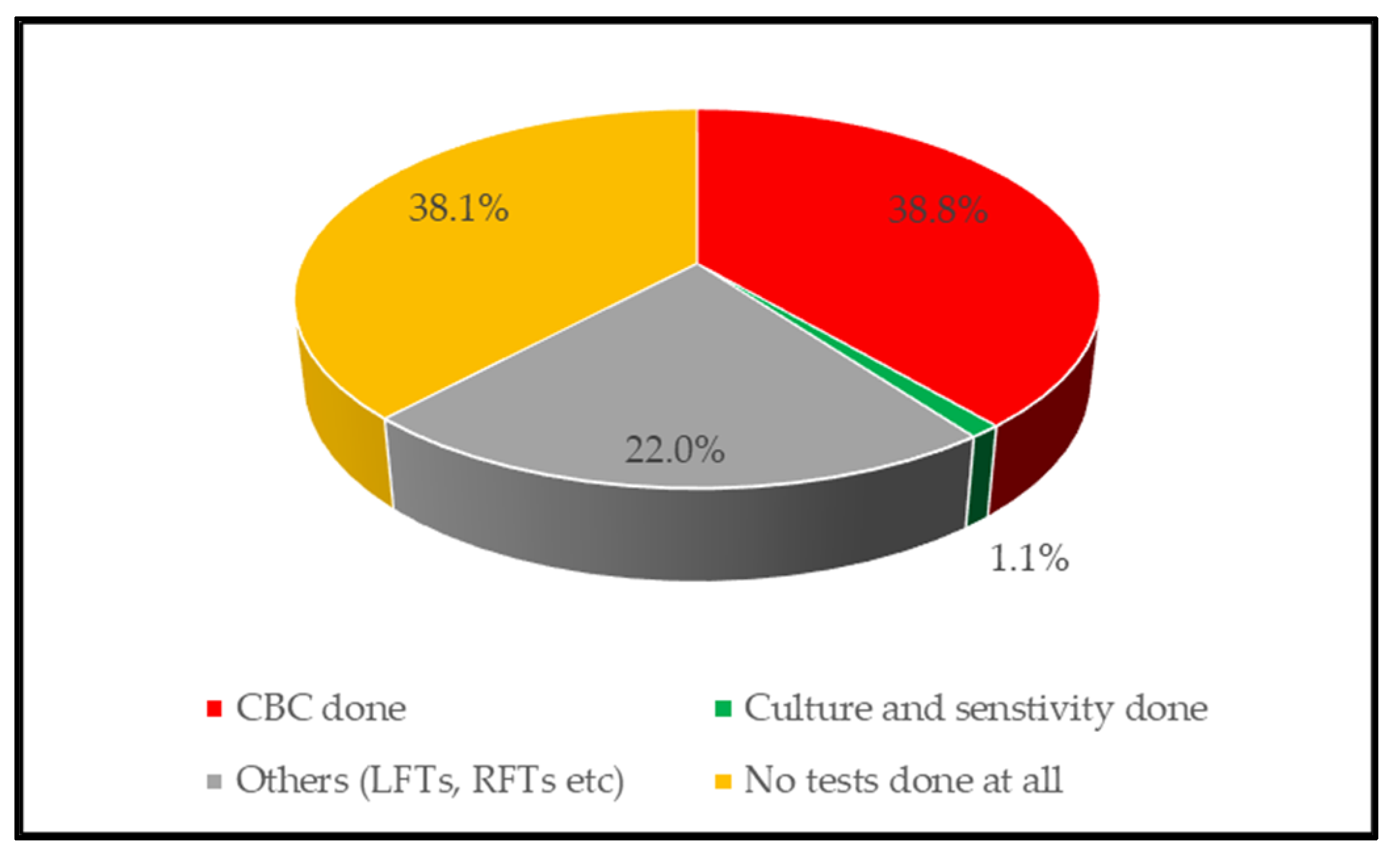
| Appropriateness of testing | |||
| PNFP | 96.5 (0.65) | ||
| PFP | 77.0 (0.0) | 62.2 (28.4) | 100 |
| Public | 48.3 (24.2) | ||
| Drug interactions | |||
| PNFP | 3.5 (2.5) | ||
| PFP | 2.0 (0.0) | 4.1 (4.1) | 0 |
| Public | 4.6 (4.9) | ||
| Appropriateness of dispensing | |||
| PNFP | 30.9 (3.5) | ||
| PFP | 96.0 (0.0) | 58.2 (26.3) | 95 |
| Public | 61.0 (23.1) | ||
| Dose Completion | Ceftriaxone Doses n (%) | ||||
|---|---|---|---|---|---|
| 250 mg | 500 mg | 1 g | 2 g | Other | |
| Prescribed dose not completed | 6 (20.0%) | 25 (34.3%) | 88 (34.5%) | 158 (35.7%) | 45 (53.6%) |
| Prescribed dose completed | 23 (76.7%) | 39 (53.4%) | 161 (63.1%) | 258 (58.2%) | 35 (41.7%) |
| Unspecified dose duration | 1 (3.3%) | 9 (12.3%) | 6 (2.4%) | 27 (6.1%) | 4 (4.8%) |
| Total | 30 | 73 | 255 | 443 | 84 |
| Category | Ceftriaxone Dose Prescribed n (%) | ||||
|---|---|---|---|---|---|
| 250 mg | 500 mg | 1 g | 2 g | other | |
| Facility type | |||||
| Public facilities (PF) | 26 (4.4%) | 55 (9.4%) | 156 (25.6%) | 285 (48.6%) | 65 (11.1%) |
| Private not-for-profit (PNFP) | 4 (2.0%) | 15 (7.6%) | 72 (36.4%) | 89 (44.9%) | 18 (9.1%) |
| Private for-profit (PFP) | 0 (0%) | 3 (3%) | 27 (27%) | 69 (69%) | 1 (1%) |
| Level of care | |||||
| General hospital (GH) | 2 (1.0%) | 11 (6.6%) | 30 (20.9%) | 42 (55.6%) | 13 (15.8%) |
| Regional referral Hospital (RRH) | 5 (1.1%) | 24 (4.5%) | 159 (29.6%) | 305 (56.7%) | 45 (8.4%) |
| Health center IV (HCIV) | 23 (9.2%) | 38 (15.3%) | 66 (26.5%) | 96 (38.6) | 26 (10.4) |
| Prescriber | Ceftriaxone Dose Prescribed n (%) | ||||
|---|---|---|---|---|---|
| 250 mg | 500 mg | 1 g | 2 g | Other | |
| Unspecified prescriber | 0 (0%) | 0 (0%) | 0 (0%) | 1 (100%) | 0 (0%) |
| Intern | 1 (1.0%) | 6 (6.3%) | 26 (27.1%) | 50 (52.1%) | 13 (13.5%) |
| Medical officer | 12 (2.6%) | 36 (7.7%) | 118 (25.1%) | 269 (57.2%) | 35 (7.5%) |
| Specialist | 1 (1.0%) | 4 (3.9%) | 33 (31.7%) | 58 (55.8%) | 8 (7.7%) |
| Clinical officer | 6 (4.9%) | 6 (4.9%) | 45 (36.6%) | 54 (43.9%) | 12 (9.8%) |
| Nurse | 9 (11.0%) | 19 (23.2%) | 30 (36.6%) | 9 (11.0%) | 15 (18.3%) |
| Midwife | 0 (0%) | 2 (33.3%) | 1 (16.7%) | 2 (33.3%) | 1 (16.9%) |
| Variables | Appropriateness | Crude Analysis | Adjusted Analysis | |||
|---|---|---|---|---|---|---|
| No | Yes | Odds Ratio (CI) | p-Value | Adj. Odds Ratio (CI) | p-Value | |
| Gender | ||||||
| Male | 299 (73.8%) | 106 (26.2%) | ||||
| Female | 398 (82.9%) | 82 (17.1%) | 0.6 (0.4–0.8) | 0.001 | 0.2 (0.1–1.0) | 0.045 |
| Pregnancy | ||||||
| Not pregnant | 263 (79.2%) | 69 (20.8%) | ||||
| Pregnant | 132 (89.2%) | 16 (10.8%) | 0.5 (0.3–0.8) | 0.009 | 0.4 (0.2–0.8) | 0.006 |
| Days of hospitalization | ||||||
| 1–3 days | 312 (83.9%) | 60 (16.1%) | ||||
| 4–7 days | 307 (76.2%) | 96 (23.8%) | 1.6 (1.1–2.3) | 0.008 | 2.0 (1.2–2.5) | 0.002 |
| Above 7 days | 74 (69.8%) | 32 (30.2%) | 2.3 (1.4–3.7) | 0.001 | 2.2 (1.2–3.4) | 0.010 |
| Level of care | ||||||
| Regional referral | 183 (93.4%) | 13 (6.6%) | ||||
| General hospital | 312 (70.9%) | 128 (29.1%) | 5.8 (3.2–10.5) | 0.000 | 3.4 (1.6–7.3) | 0.002 |
| HC IV | 202 (81.1%) | 47 (18.9%) | 3.3 (1.7–6.2) | 0.000 | 3.6 (1.7–7.5) | 0.001 |
| Facility type | ||||||
| Public | 496 (84.5%) | 91 (15.5%) | ||||
| PNFP | 164 (82.8%) | 34 (17.2%) | 1.1 (0.73–1.7) | 0.579 | ||
| PFP | 37 (37%) | 63 (63%) | 9.3 (5.8–14.8) | 0.000 | 7.4 (3.5–15.8) | 0.000 |
| Prescriber category | ||||||
| Medical intern | 93 (96.9%) | 3 (3.1%) | ||||
| Medical officer | 370 (78.6%) | 101 (21.4%) | 8.5 (2.6–27.3) | 0.000 | 4.8 (1.5–16) | 0.010 |
| Specialist | 62 (59.6%) | 42 (40.4%) | 21.0 (6.2–70.7) | 0.000 | 3.6 (0.9–14.0) | 0.063 |
| Clinical officer | 97 (78.9%) | 26 (21.1%) | 8.3 (2.4–28.4) | 0.001 | 4.3 (1.1–16.1) | 0.033 |
| Nurse | 68 (82.9%) | 14 (17.1%) | 6.4 (1.8–23.1) | 0.005 | 4.1 (1.0–16.8) | 0.051 |
| Midwife | 4 (66.7%) | 2 (33.3%) | 15.5 (2.0–120.4) | 0.009 | 8.9 (1.1–75.0) | 0.044 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kutyabami, P.; Munanura, E.I.; Kalidi, R.; Balikuna, S.; Ndagire, M.; Kaggwa, B.; Nambatya, W.; Kamba, P.F.; Musiimenta, A.; Kesi, D.N.; et al. Evaluation of the Clinical Use of Ceftriaxone among In-Patients in Selected Health Facilities in Uganda. Antibiotics 2021, 10, 779. https://doi.org/10.3390/antibiotics10070779
Kutyabami P, Munanura EI, Kalidi R, Balikuna S, Ndagire M, Kaggwa B, Nambatya W, Kamba PF, Musiimenta A, Kesi DN, et al. Evaluation of the Clinical Use of Ceftriaxone among In-Patients in Selected Health Facilities in Uganda. Antibiotics. 2021; 10(7):779. https://doi.org/10.3390/antibiotics10070779
Chicago/Turabian StyleKutyabami, Paul, Edson Ireeta Munanura, Rajab Kalidi, Sulah Balikuna, Margaret Ndagire, Bruhan Kaggwa, Winnie Nambatya, Pakoyo Fadhiru Kamba, Allan Musiimenta, Diana Nakitto Kesi, and et al. 2021. "Evaluation of the Clinical Use of Ceftriaxone among In-Patients in Selected Health Facilities in Uganda" Antibiotics 10, no. 7: 779. https://doi.org/10.3390/antibiotics10070779
APA StyleKutyabami, P., Munanura, E. I., Kalidi, R., Balikuna, S., Ndagire, M., Kaggwa, B., Nambatya, W., Kamba, P. F., Musiimenta, A., Kesi, D. N., Nambasa, V., Serwanga, A., & Ndagije, H. B. (2021). Evaluation of the Clinical Use of Ceftriaxone among In-Patients in Selected Health Facilities in Uganda. Antibiotics, 10(7), 779. https://doi.org/10.3390/antibiotics10070779







