Population Pharmacokinetics and Significant Under-Dosing of Anti-Tuberculosis Medications in People with HIV and Critical Illness
Abstract
1. Introduction
2. Results
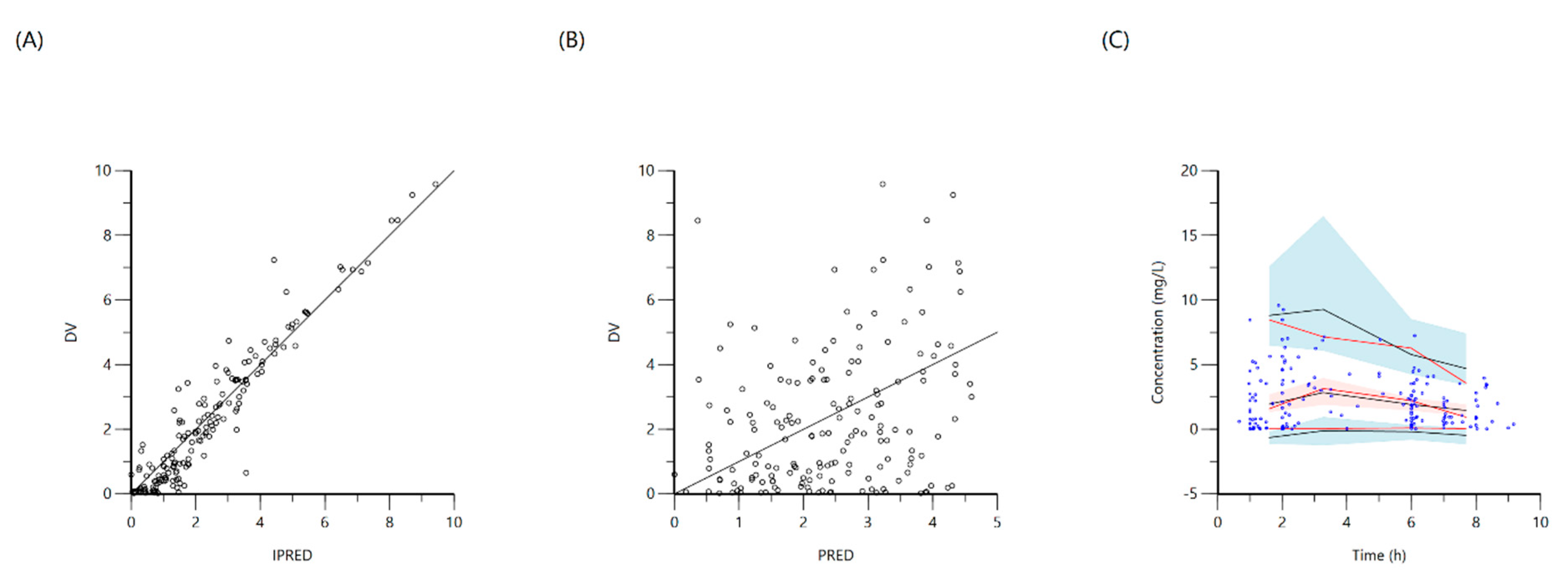
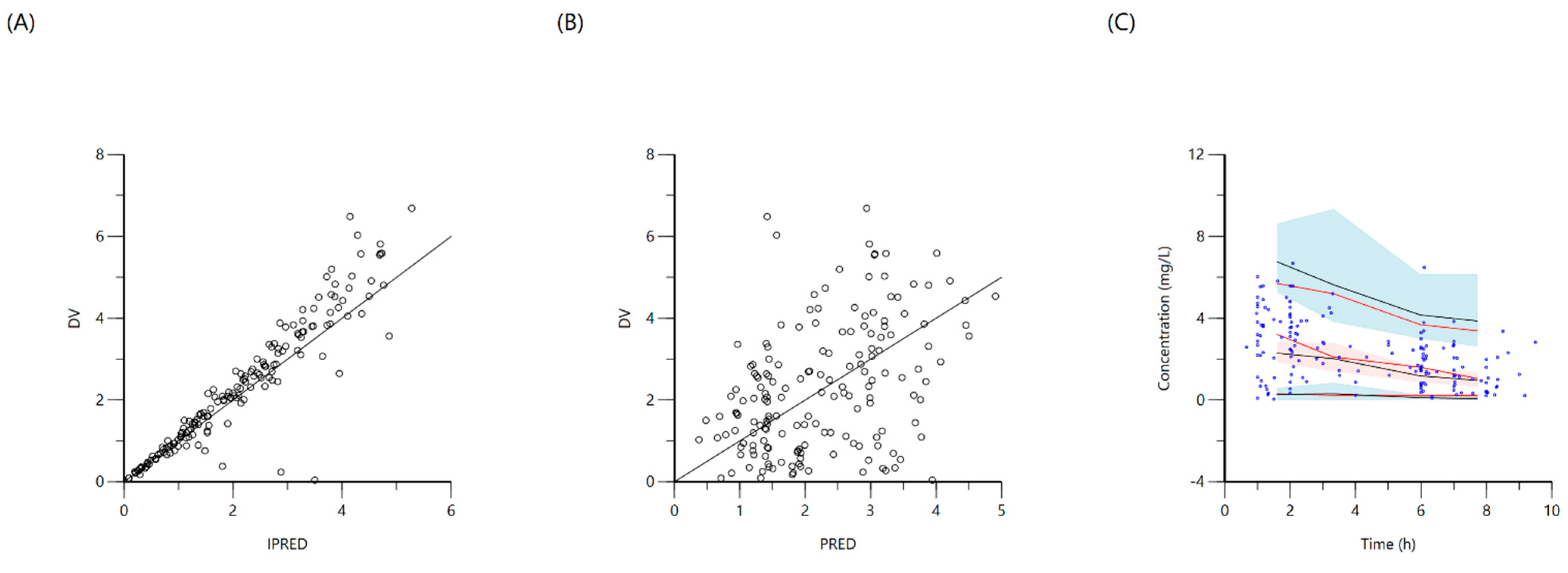
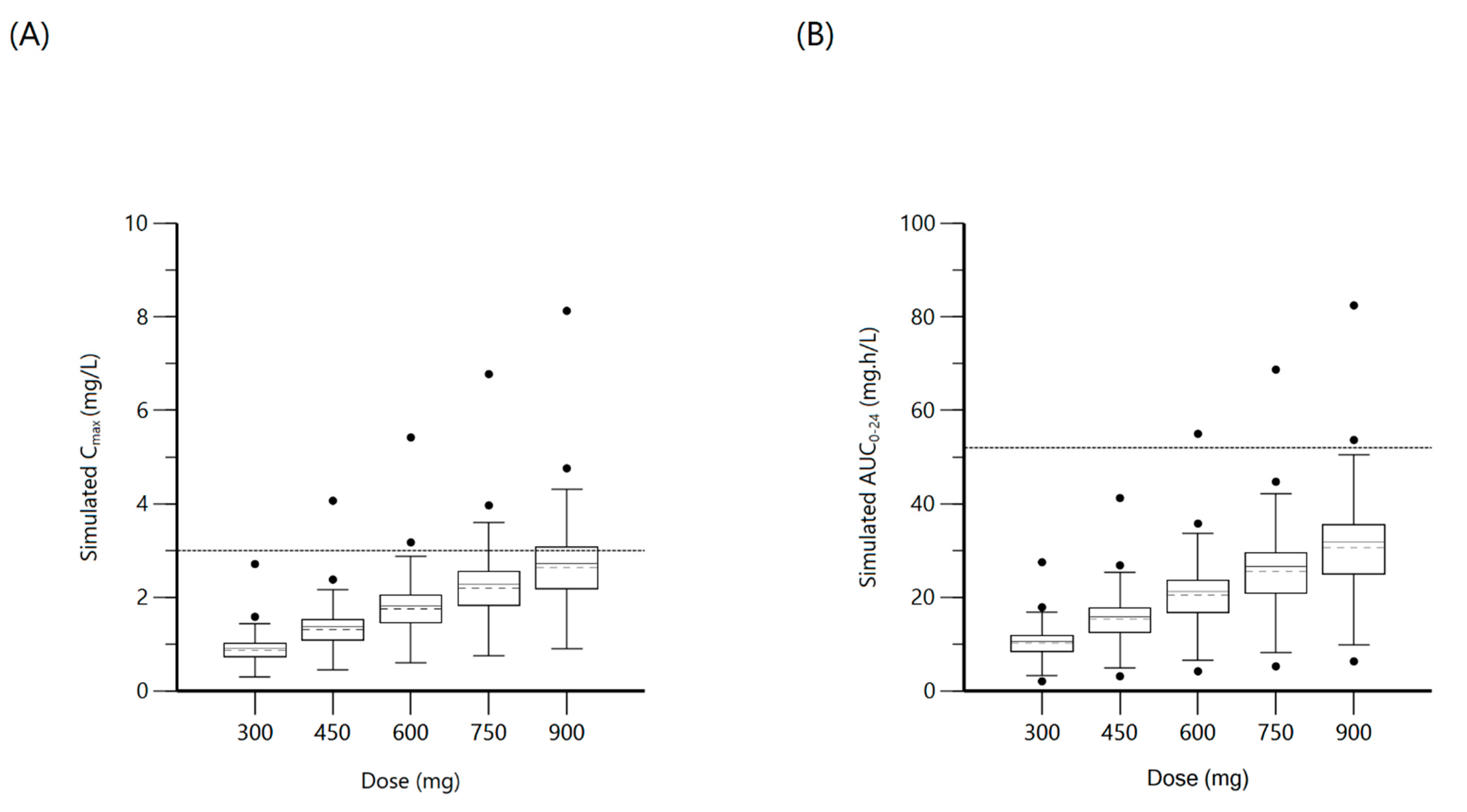
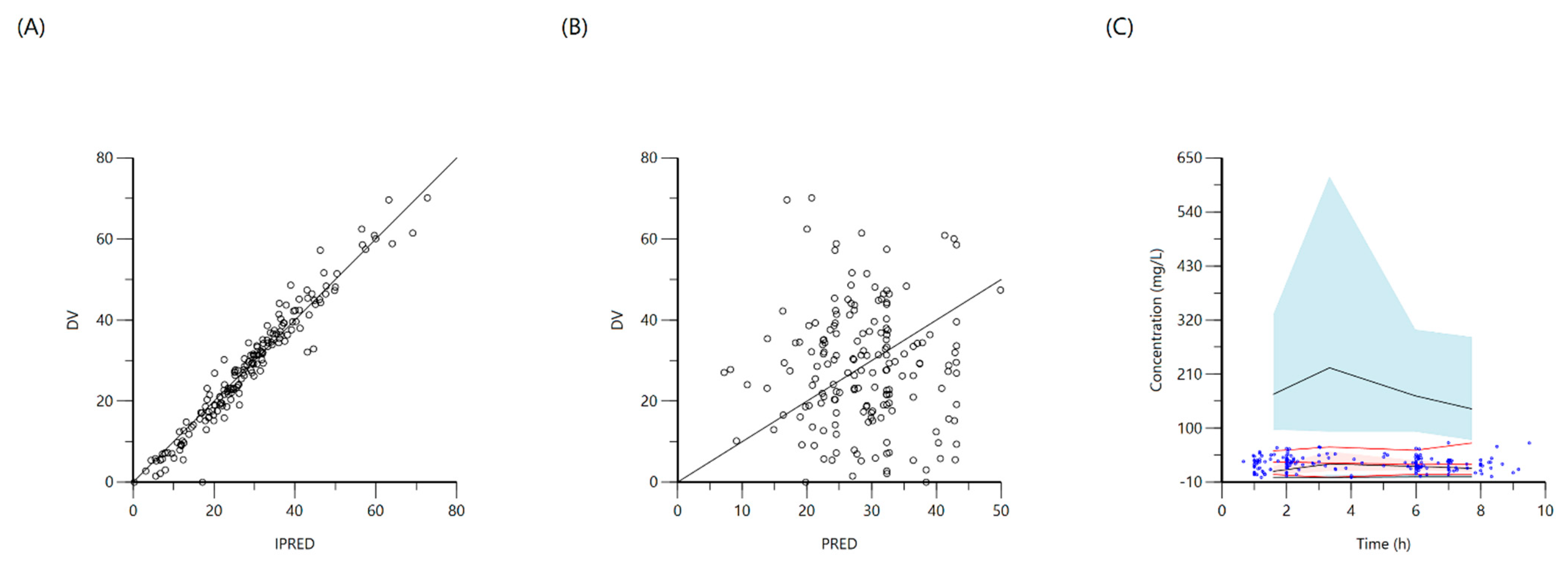
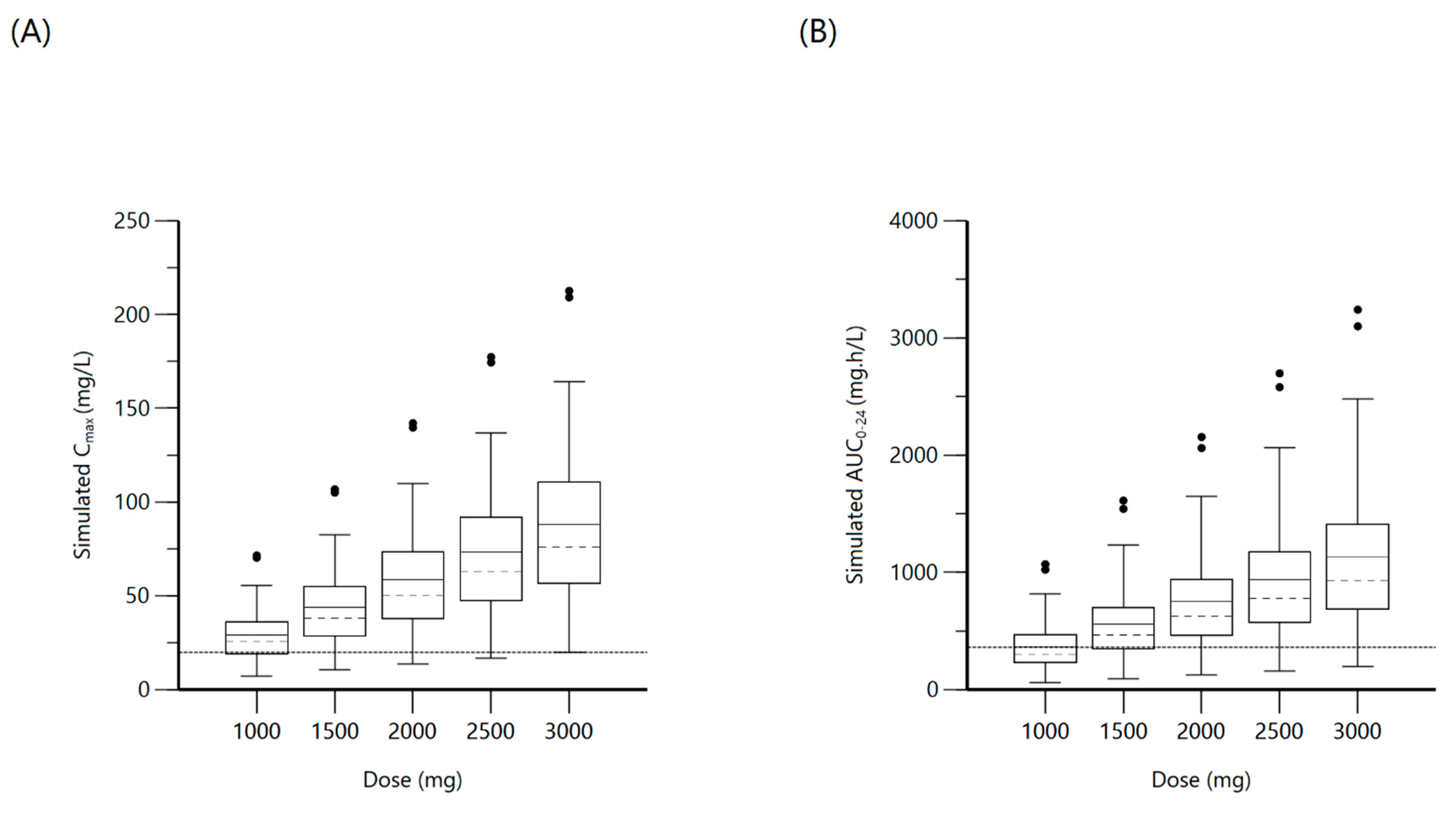
2.1. Rifampin
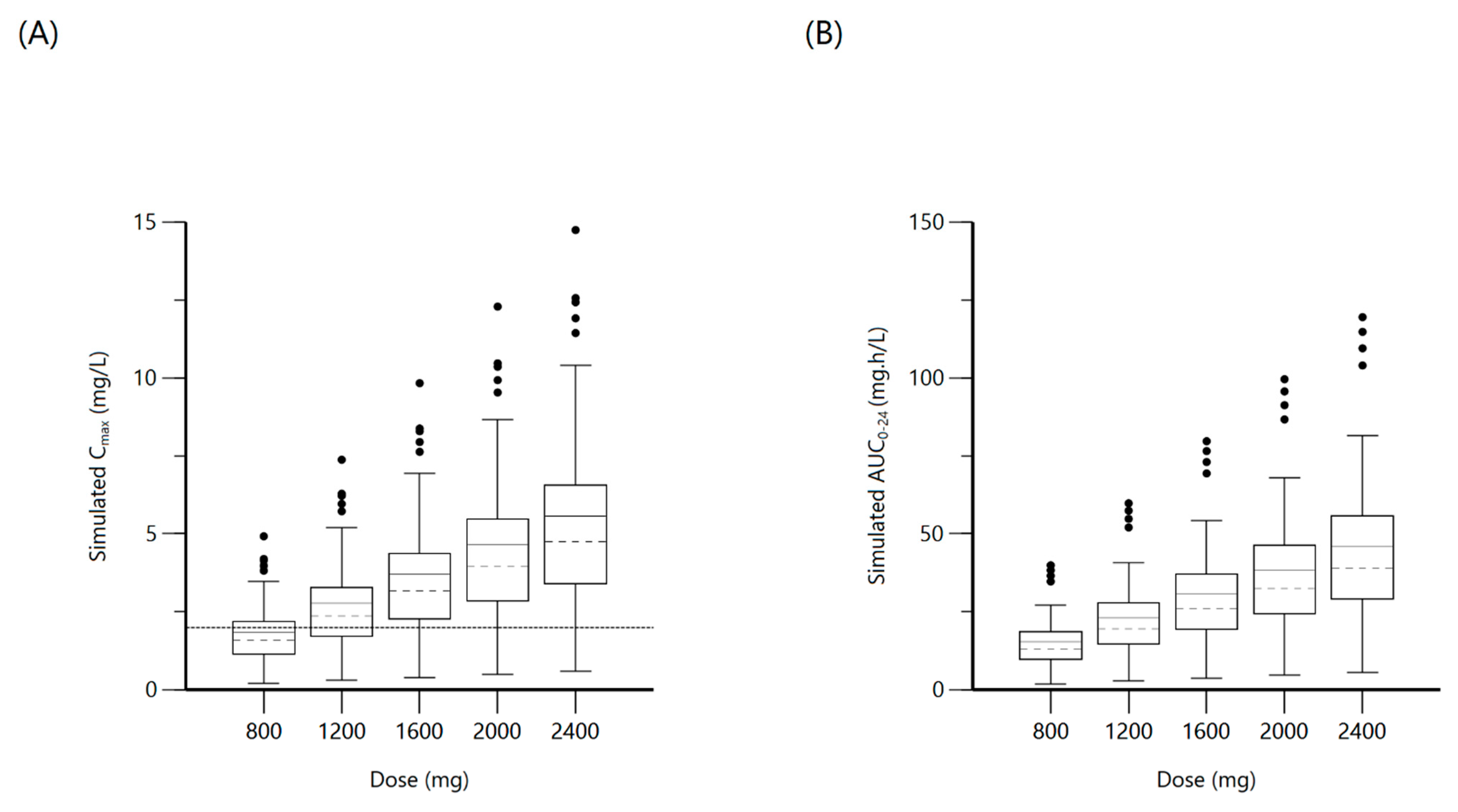
2.2. Isoniazid
2.3. Pyrazinamide
2.4. Ethambutol
3. Discussion
4. Materials and Methods
4.1. Patient Population
4.2. NAT2 Testing
4.3. Anti-TB Therapy and PK Study Methods
4.4. Population Pharmacokinetics, Model Development, and Simulation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ART | Anti-Retroviral Therapy |
| AUC | Area Under Curve |
| CD4 | Cluster of Differentiation 4 |
| CI | Confidence Interval |
| Cl | Systemic clearance |
| Cl2 | Intercompartmental clearance |
| Cmax | Maximum serum Concentration |
| CV | Coefficient of Variation |
| DV | Dependent Variable (observed concentrations) |
| HIV | Human Immunodeficiency Virus |
| IIV | Inter Individual Variability |
| IPRED | Individual Predicted Concentrations |
| IQR | Inter Quartile Range |
| Ka | Absorption rate constant |
| LAM | Lipoarabinomannan |
| LC-MS-MS | Liquid Chromatography-Mass Spectrometry-Mass Spectrometry |
| MUAC | Mid-Upper Arm Circumference |
| NAT2 | N-Acetyl Transferase 2 |
| NLME | Non-Linear Mixed Effects |
| PCR | Polymerase Chain Reaction |
| PD | Pharmacodynamic |
| PK | Pharmacokinetic |
| PLWH | People Living With HIV |
| PRED | Population Predicted Concentrations |
| TAC | Taqman Array Card |
| TB | Tuberculosis |
| V2 | Volume of distribution of the peripheral compartment |
| V | Volume of distribution in the central compartment |
| VPC | Visual Predictive Check |
References
- Adhikari, N.K.; Fowler, R.A.; Bhagwanjee, S.; Rubenfeld, G.D. Critical care and the global burden of critical illness in adults. Lancet 2010, 376, 1339–1346. [Google Scholar] [CrossRef]
- Rudd, K.E.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.S.; Kissoon, N.; Finfer, S.; et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: Analysis for the Global Burden of Disease Study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef]
- Barr, D.A.; Lewis, J.M.; Feasey, N.; Schutz, C.; Kerkhoff, A.D.; Jacob, S.T.; Andrews, B.; Kelly, P.; Lakhi, S.; Muchemwa, L.; et al. Mycobacterium tuberculosis bloodstream infection prevalence, diagnosis, and mortality risk in seriously ill adults with HIV: A systematic review and meta-analysis of individual patient data. Lancet Infect. Dis. 2020, 742–752. [Google Scholar] [CrossRef]
- Cummings, M.J.; O’Donnell, M.R. Inverting the pyramid: Increasing awareness of mycobacterial sepsis in sub-Saharan Af-rica. Int. J. Tuberc. Lung Dis. 2015, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.C.; Jacob, S.T.; Banura, P.; Zhang, J.; Stroup, S.; Boulware, D.R.; Scheld, W.M.; Houpt, E.R.; Liu, J. Etiology of Sepsis in Uganda using a Quantitative PCR-based TaqMan Array Card. Clin. Infect. Dis. 2019, 68, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.M.; Feasey, N.A.; Rylance, J. Aetiology and outcomes of sepsis in adults in sub-Saharan Africa: A systematic review and meta-analysis. Crit. Care 2019, 23, 212. [Google Scholar] [CrossRef]
- Van den Broek, J.; Mfinanga, S.; Moshiro, C.; O’Brien, R.; Mugomela, A.; Lefi, M. Impact of human immunodeficiency virus infection on the outcome of treatment and survival of tuberculosis patients in Mwanza, Tanzania. Int. J. Tuberc. Lung Dis. 1998, 2, 547–552. [Google Scholar] [PubMed]
- Weiner, M.; Burman, W.; Vernon, A.; Benator, D.; Peloquin, C.A.; Khan, A.; Weis, S.; King, B.; Shah, N.; Hodge, T.; et al. Low isoniazid concentrations and outcome of tuberculosis treatment with once-weekly isoniazid and rifapentine. Am. J. Respir. Crit Care Med. 2003, 167, 1341–1347. [Google Scholar] [CrossRef]
- Weiner, M.; Benator, D.; Burman, W.; Peloquin, C.A.; Khan, A.; Vernon, A.; Jones, B.; Silva-Trigo, C.; Zhao, Z.; Hodge, T.; et al. Association between acquired rifamycin resistance and the pharmacokinetics of rifabutin and isoniazid among patients with HIV and tuberculosis. Clin. Infect. Dis. 2005, 40, 1481–1491. [Google Scholar] [CrossRef] [PubMed]
- Tappero, J.W.; Bradford, W.Z.; Agerton, T.B.; Hopewell, P.; Reingold, A.L.; Lockman, S.; Oyewo, A.; Talbot, E.A.; Kenyon, T.A.; Moeti, T.L.; et al. Serum concentrations of antimycobacterial drugs in patients with pulmonary tuberculosis in Bot-swana. Clin Infect. Dis. 2005, 41, 461–469. [Google Scholar] [CrossRef]
- Chideya, S.; Winston, C.A.; Peloquin, C.A.; Bradford, W.Z.; Hopewell, P.C.; Wells, C.D.; Reingold, A.L.; Kenyon, T.A.; Moeti, T.L.; Tappero, J.W. Isoniazid, rifampin, ethambutol, and pyrazinamide pharmacokinetics and treatment outcomes among a predominantly HIV-infected cohort of adults with tuberculosis from Botswana. Clin. Infect. Dis. 2009, 48, 1685–1694. [Google Scholar] [CrossRef]
- Pasipanodya, J.G.; McIlleron, H.; Burger, A.; Wash, P.A.; Smith, P.; Gumbo, T. Serum drug concentrations predictive of pul-monary tuberculosis outcomes. J. Infect. Dis. 2013, 208, 1464–1473. [Google Scholar] [CrossRef]
- Sekaggya-Wiltshire, C.; von Braun, A.; Lamorde, M.; Ledergerber, B.; Buzibye, A.; Henning, L.; Musaazi, J.; Gutteck, U.; Denti, P.; de Kock, M.; et al. Delayed Sputum Culture Conversion in Tuberculosis-Human Immunodeficiency Vi-rus-Coinfected Patients with Low Isoniazid and Rifampicin Concentrations. Clin. Infect. Dis. 2018, 67, 708–716. [Google Scholar] [CrossRef]
- Heysell, S.K.; Mtabho, C.; Mpagama, S.; Mwaigwisya, S.; Pholwat, S.; Ndusilo, N.; Gratz, J.; Aarnoutse, R.E.; Kibiki, G.S.; Houpt, E.R. Plasma drug activity assay for treatment optimization in tuberculosis patients. Antimicrob. Agents Chemother. 2011, 5819–5825. [Google Scholar] [CrossRef]
- Alffenaar, J.C.; Gumbo, T.; Dooley, K.E.; Peloquin, C.A.; Mcilleron, H.; Zagorski, A.; Cirillo, D.M.; Heysell, S.K.; Silva, D.R.; Migliori, G.B. Integrating Pharmacokinetics and Pharmacodynamics in Operational Research to End Tuberculosis. Clin. Infect. Dis. 2020, 70, 1774–1780. [Google Scholar] [CrossRef]
- Boeree, M.J.; Heinrich, N.; Aarnoutse, R.; Diacon, A.H.; Dawson, R.; Rehal, S.; Kibiki, G.S.; Churchyard, G.; Sanne, I.; Ntinginya, N.E.; et al. High-dose rifampicin, moxifloxacin, and SQ109 for treating tuberculosis: A multi-arm, multi-stage randomised controlled trial. Lancet Infect. Dis. 2017, 39–49. [Google Scholar] [CrossRef]
- Zuur, M.A.; Pasipanodya, J.G.; van, S.D.; van der Werf, T.S.; Gumbo, T.; Alffenaar, J.C. Intermediate Susceptibility Dose-Dependent Breakpoints for High-Dose Rifampin, Isoniazid, and Pyrazinamide Treatment in Multidrug-Resistant Tu-berculosis Programs. Clin. Infect. Dis. 2018, 67, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- De, P.P.; Belpaire, F.M.; Buylaert, W.A. Pharmacokinetic and pharmacodynamic considerations when treating patients with sepsis and septic shock. Clin. Pharmacokinet. 2002, 41, 1135–1151. [Google Scholar] [CrossRef]
- Hazard, R.H.; Kagina, P.; Kitayimbwa, R.; Male, K.; McShane, M.; Mubiru, D.; Welikhe, E.; Moore, C.C.; Abdallah, A. Effect of Empiric Anti-Mycobacterium tuberculosis Therapy on Survival Among Human Immunodeficiency Virus-Infected Adults Admitted With Sepsis to a Regional Referral Hospital in Uganda. Open Forum Infect. Dis. 2019, 6, ofz140. [Google Scholar] [CrossRef]
- Horita, Y.; Alsultan, A.; Kwara, A.; Antwi, S.; Enimil, A.; Ortsin, A.; Dompreh, A.; Yang, H.; Wiesner, L.; Peloquin, C.A. Evaluation of the Adequacy of WHO Revised Dosages of the First-Line Antituberculosis Drugs in Children with Tuberculo-sis Using Population Pharmacokinetic Modeling and Simulations. Antimicrob. Agents Chemother. 2018, 62, e00008-18. [Google Scholar] [CrossRef] [PubMed]
- Alsultan, A.; Peloquin, C.A. Therapeutic drug monitoring in the treatment of tuberculosis: An update. Drugs 2014, 839–854. [Google Scholar] [CrossRef]
- Patel, K.B.; Belmonte, R.; Crowe, H.M. Drug malabsorption and resistant tuberculosis in HIV-infected patients. N. Eng. J. Med. 1995, 332, 336–337. [Google Scholar] [CrossRef]
- Hemanth Kumar, A.K.; Kannan, T.; Chandrasekaran, V.; Sudha, V.; Vijayakumar, A.; Ramesh, K.; Lavanya, J.; Swaminathan, S.; Ramachandran, G. Pharmacokinetics of thrice-weekly rifampicin, isoniazid and pyrazinamide in adult tuberculosis pa-tients in India. Int. J. Tuberc. Lung Dis. 2016, 1236–1241. [Google Scholar] [CrossRef] [PubMed]
- McIlleron, H.; Rustomjee, R.; Vahedi, M.; Mthiyane, T.; Denti, P.; Connolly, C.; Rida, W.; Pym, A.; Smith, P.J.; Onyebujoh, P.C. Reduced antituberculosis drug concentrations in HIV-infected patients who are men or have low weight: Implications for international dosing guidelines. Antimicrob. Agents Chemother. 2012, 3232–3238. [Google Scholar] [CrossRef] [PubMed]
- Hong, B.L.; D’Cunha, R.; Li, P.; Al-Shaer, M.H.; Alghamdi, W.A.; An, G.; Peloquin, C. A Systematic Review and Me-ta-analysis of Isoniazid Pharmacokinetics in Healthy Volunteers and Patients with Tuberculosis. Clin. Ther. 2020, e220–e241. [Google Scholar] [CrossRef]
- Subbarao, S.; Wilkinson, K.A.; van Halsema, C.L.; Rao, S.S.; Boyles, T.; Utay, N.S.; Wilkinson, R.J.; Meintjes, G. Raised Ve-nous Lactate and Markers of Intestinal Translocation Are Associated With Mortality Among In-Patients With HIV-Associated TB in Rural South Africa. J. Acquir. Immune Defic. Syndr. 2015, 70, 406–413. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schutz, C.; Barr, D.; Andrade, B.B.; Shey, M.; Ward, A.; Janssen, S.; Burton, R.; Wilkinson, K.A.; Sossen, B.; Fukutani, K.F.; et al. Clinical, microbiologic, and immunologic determinants of mortality in hospitalized patients with HIV-associated tu-berculosis: A prospective cohort study. PLoS Med. 2019, 16, e1002840. [Google Scholar] [CrossRef]
- Te Brake, L.H.M.; de Jager, V.; Narunsky, K.; Vanker, N.; Svensson, E.M.; Phillips, P.P.J.; Gillespie, S.H.; Heinrich, N.; Hoelscher, M.; Dawson, R.; et al. Increased bactericidal activity but dose-limiting intolerability at 50 mg·kg-1 rifampicin. Eur. Respir. J. 2021, 2000955. [Google Scholar] [CrossRef]
- Dooley, K.E. High-Dose Rifampin: Shall We Be Bolder? Am. J. Respir. Crit. Care Med. 2018, 198, 558–560. [Google Scholar] [CrossRef]
- Aarnoutse, R.E.; Kibiki, G.S.; Reither, K.; Semvua, H.H.; Haraka, F.; Mtabho, C.M.; Mpagama, S.G.; van den Boogaard, J.; Sumari-de Boer, I.M.; Magis-Escurra, C.; et al. Pharmacokinetics, Tolerability, and Bacteriological Response of Rifampin Administered at 600, 900, and 1200 Milligrams Daily in Patients with Pulmonary Tuberculosis. Antimicrob. Agents Chemother. 2017, 61, e01054-17. [Google Scholar] [CrossRef]
- Walsh, K.F.; Vilbrun, S.C.; Souroutzidis, A.; Delva, S.; Joissaint, G.; Mathurin, L.; Ocheretina, O.; Cremieux, P.; Pape, J.W.; Koenig, S.P. Improved Outcomes With High-dose Isoniazid in Multidrug-resistant Tuberculosis Treatment in Haiti. Clin. Infect. Dis. 2019, 69, 717–719. [Google Scholar] [CrossRef]
- Diacon, A.; Miyahara, S.; Dawson, R.; Sun, X.; Hogg, E.; Donahue, K.; Urbanowski, M.; De Jager, V.; Fletcher, C.V.; Hafner, R.; et al. Assessing whether isoniazid is essential during the first 14 days of tuberculosis therapy: A phase 2a, open-label, randomised controlled trial. Lancet Microbe 2020, e84–e92. [Google Scholar] [CrossRef]
- Pasipanodya, J.G.; Gumbo, T. Clinical and toxicodynamic evidence that high-dose pyrazinamide is not more hepatotoxic than the low doses currently used. Antimicrob. Agents Chemother. 2010, 2847–2854. [Google Scholar] [CrossRef]
- Levy, M.M.; Fink, M.P.; Marshall, J.C.; Abraham, E.; Angus, D.; Cook, D.; Cohen, J.; Opal, S.M.; Vincent, J.L.; Ramsay, G. SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit. Care Med. 2003, 1250–1256. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ochieng, C.; Wiersma, S.; Ströher, U.; Towner, J.S.; Whitmer, S.; Nichol, S.T.; Moore, C.C.; Kersh, G.J.; Kato, C.; et al. Development of a TaqMan Array Card for Acute-Febrile-Illness Outbreak Investigation and Surveillance of Emerging Pathogens, Including Ebola Virus. J. Clin. Microbiol. 2016, 49–58. [Google Scholar] [CrossRef]
- Justine, M.; Yeconia, A.; Nicodemu, I.; Augustino, D.; Gratz, J.; Mduma, E.; Heysell, S.K.; Kivuyo, S.; Mfinanga, S.; Peloquin, C.A.; et al. Pharmacokinetics of First-Line Drugs Among Children With Tuberculosis in Rural Tanzania. J. Pediatric. Infect. Dis. Soc. 2020, 9, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Heysell, S.K.; Moore, J.L.; Keller, S.J.; Houpt, E.R. Therapeutic drug monitoring for slow response to tuberculosis treatment in a state control program, Virginia, USA. Emerg. Infect. Dis. 2010, 16, 1546–1553. [Google Scholar] [CrossRef] [PubMed]


| Characteristic | N (%) or Median (IQR) N = 81 |
|---|---|
| Age (years) | 36 (30–43.5) |
| Female | 29 (36.3%) |
| Weight (kg) | 52.5 (45.3–58) |
| MUAC (cm) | 22 (20.4–24) |
| CD4 count (cells/mL3) | 169 |
| Prior TB | 7 (9%) |
| Concurrent meningitis | 27 (33%) |
| Duration of Illness (days) | 30 (14–90) |
| Cough >2 weeks | 63 (79%) |
| Fever >2 weeks | 75 (94%) |
| Night sweats >2 weeks | 67 (86%) |
| Loss of appetite >2 weeks | 78 (96.3%) |
| Loss of wt >2 weeks | 48 (58.3%) |
| Vital Status at Week 2 PK Testing | All N = 81 | Confirmed TB N = 36 | Clinical TB N = 45 |
|---|---|---|---|
| Survived (%N) | 49 (60%) | 26 (72%) | 23 (51%) |
| Sepsis | 32 | 21 | 11 |
| Meningitis | 17 | 5 | 12 |
| Died (%N) | 18 (23%) | 5 (14%) | 13 (29%) |
| Sepsis | 13 | 4 | 9 |
| Meningitis | 5 | 1 | 4 |
| Lost to follow-up, withdrew or incomplete PK testing (%N) | 14 (17%) | 5 (17%) | 9 (20%) |
| Sepsis | 9 | 5 | 4 |
| Meningitis | 5 | 0 | 5 |
| Drug | Parameter | Final Model | |
|---|---|---|---|
| Estimate (%CV) | IIV (Shrinkage) | ||
| Rifampin | Ka (1/h) | 0.3 (13.5) | 0.01 (0.9) |
| V (L) | 25.3 (33.3) | 3.2 (0.2) | |
| Cl (L/h) | 0.1 (49.6) | 0.2 (0.3) | |
| Tlag (h) | 0.9 (9.1) | 0.003 (0.8) | |
| Isoniazid | Ka (1/h) | 0.9 (0.9) | 0.9 (0.3) |
| V (L) | 2.9 (3) | 0.07 (0.6) | |
| V2 (L) | 32.5 (5.7) | 0.5 (0.7) | |
| Cl (L/h) | 9.2 (8.7) | 0.8 (0.1) | |
| Cl2 (L/h) | 9.6 (3) | 0.0001 (0.9) | |
| Tlag (h) | 0.4 (8.6) | 0.5 (0.5) | |
| Pyrazinamide | Ka (1/h) | 0.08 (13) | 0.1-0.11 |
| V (L) | 1.5 (16.3) | 0.9 (0.4) | |
| Cl (L/h) | 2.6 (14.5) | 0.4 (0.05) | |
| Tlag (h) | 0.2 (7.1) | 1.3 (0.4) | |
| Ethambutol | Ka (1/h) | 0.15 (21.8) | 0.6 (0.4) |
| V (L) | 75.17 (23.7) | 0.4 (0.1) | |
| Cl (L/h) | 51.6 (12.2) | 0.3 (0.3) | |
| Tlag (h) | 0.4 (12.3) | 0.6 (0.4) | |
| Drug | Simulated Dose (mg) | Target | Cmax (mg/L) | AUC0–24 (mg·h/L) | ||
|---|---|---|---|---|---|---|
| Median (IQR) | Attaining Target (%) | Median (IQR) | Attaining Target (%) | |||
| Rifampin | 600 | Cmax: 8 mg/L | 4.6 (3.4–6.4) | 16.30 | 34.5 (26.3–42.2) | 43.6 |
| 900 | 6.8 (4.8–9.6) | 34.7 | 52.3 (36.6–63.5) | 78 | ||
| 1200 | 9 (6.3–12.7) | 57.1 | 71.2 (48.1–84) | 92.7 | ||
| 1500 | AUC0–24: 35.4 mg·h/L | 11.2 (7.9–16) | 73.5 | 90.6 (66.7–105.6) | 97.7 | |
| 1800 | 13.3 (9.5–19.2) | 81.6 | 108.8 (79–123.9) | 100 | ||
| Isoniazid | 300 | Cmax: 3 mg/L | 0.9 (0.7–1) | 0 | 10.2 (8.7–11.8) | 0 |
| 450 | 1.3 (1.1–1.5) | 0 | 15.3 (13.1–17.7) | 0 | ||
| 600 | 1.7 (1.5–2) | 4.1 | 20.5 (17.4–23.6) | 2.3 | ||
| 750 | AUC0–24: 52 mg·h/L | 2.2 (1.8–2.5) | 8.2 | 25.6 (21.7–29-6) | 2.3 | |
| 900 | 2.6 (2.2–3.1) | 28.6 | 30.7 (26.1–35.5) | 4.5 | ||
| Pyrazinamide | 1000 | Cmax: 20 mg/L | 25.7 (19.1–36) | 69.4 | 303.8 (231.6–466.1) | 42.9 |
| 1500 | 38 (28.7–54.3) | 89.8 | 466 (351.1–702.1) | 71.4 | ||
| 2000 | 50.4 (38.1–72.6) | 91.8 | 624.7 (469.5–934.8) | 85.1 | ||
| 2500 | AUC0–24: 363 mg·h/L | 63.1 (47.4–90.8) | 96 | 778.5 (588–1167.8) | 89.6 | |
| 3000 | 75.8 (56.7–109.2) | 98 | 933 (706.6–1403.9) | 91.7 | ||
| Ethambutol | 800 | Cmax: 2 mg/L | 1.6 (1.2–2.2) | 32.6 | 13 (10–18.6) | |
| 1200 | 2.4 (1.7–3.3) | 63.2 | 19.5 (15–28) | |||
| 1600 | 3.2 (2.3–4.4) | 89.8 | 26 (20–37.1) | |||
| 2000 | 3.9 (2.9–5.5) | 93.8 | 32.5 (25–46.4) | |||
| 2400 | 4.7 (3.5–6.5) | 93.8 | 39 (30–55.7) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, P.S.; Moore, C.C.; Mbonde, A.A.; Nuwagira, E.; Orikiriza, P.; Nyehangane, D.; Al-Shaer, M.H.; Peloquin, C.A.; Gratz, J.; Pholwat, S.; et al. Population Pharmacokinetics and Significant Under-Dosing of Anti-Tuberculosis Medications in People with HIV and Critical Illness. Antibiotics 2021, 10, 739. https://doi.org/10.3390/antibiotics10060739
Rao PS, Moore CC, Mbonde AA, Nuwagira E, Orikiriza P, Nyehangane D, Al-Shaer MH, Peloquin CA, Gratz J, Pholwat S, et al. Population Pharmacokinetics and Significant Under-Dosing of Anti-Tuberculosis Medications in People with HIV and Critical Illness. Antibiotics. 2021; 10(6):739. https://doi.org/10.3390/antibiotics10060739
Chicago/Turabian StyleRao, Prakruti S., Christopher C. Moore, Amir A. Mbonde, Edwin Nuwagira, Patrick Orikiriza, Dan Nyehangane, Mohammad H. Al-Shaer, Charles A. Peloquin, Jean Gratz, Suporn Pholwat, and et al. 2021. "Population Pharmacokinetics and Significant Under-Dosing of Anti-Tuberculosis Medications in People with HIV and Critical Illness" Antibiotics 10, no. 6: 739. https://doi.org/10.3390/antibiotics10060739
APA StyleRao, P. S., Moore, C. C., Mbonde, A. A., Nuwagira, E., Orikiriza, P., Nyehangane, D., Al-Shaer, M. H., Peloquin, C. A., Gratz, J., Pholwat, S., Arinaitwe, R., Boum, Y., Mwanga-Amumpaire, J., Houpt, E. R., Kagan, L., Heysell, S. K., & Muzoora, C. (2021). Population Pharmacokinetics and Significant Under-Dosing of Anti-Tuberculosis Medications in People with HIV and Critical Illness. Antibiotics, 10(6), 739. https://doi.org/10.3390/antibiotics10060739





