Prolonged Infusion of β-Lactams Decreases Mortality in Patients with Septic Shock: A Retrospective before-and-after Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Antimicrobial Treatment, Clinical Scores, Laboratory Data
2.3. Clinical Outcome Measures
2.4. Statistical Methods
3. Results
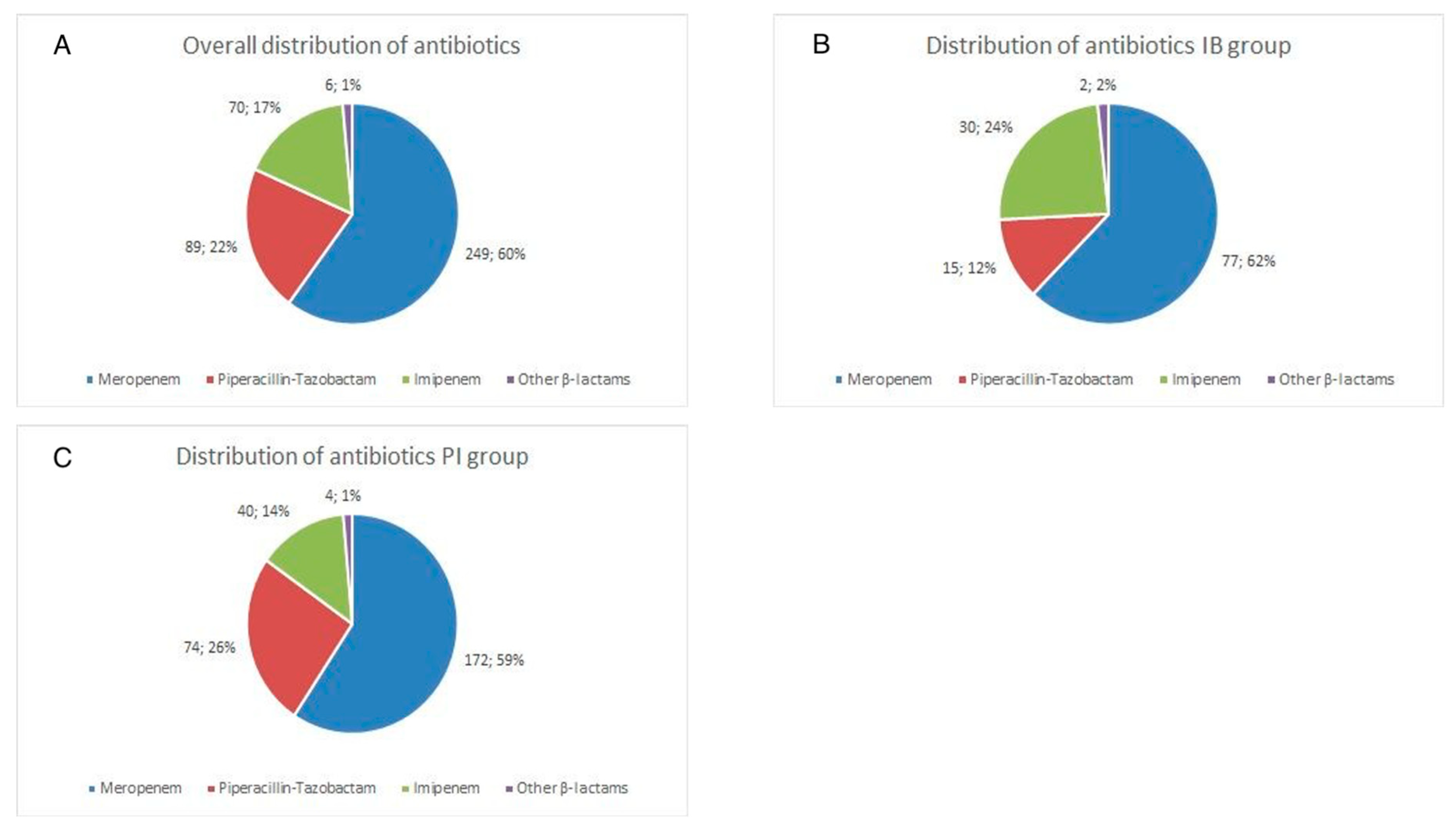
3.1. Baseline Characteristics
| TZB: | PI group 0.2 g/kg/d (0.1) | vs. IB group 0.19 g/kg/d (0.06) |
| Meropenem: | PI group 0.05 g/kg/d (0.03) | vs. IB group 0.04 g/kg/d (0.01) |
| Imipenem: | PI group 0.04 g/kg/d (0.02) | vs. IB group 0.04 g/kg/d (0.01) |
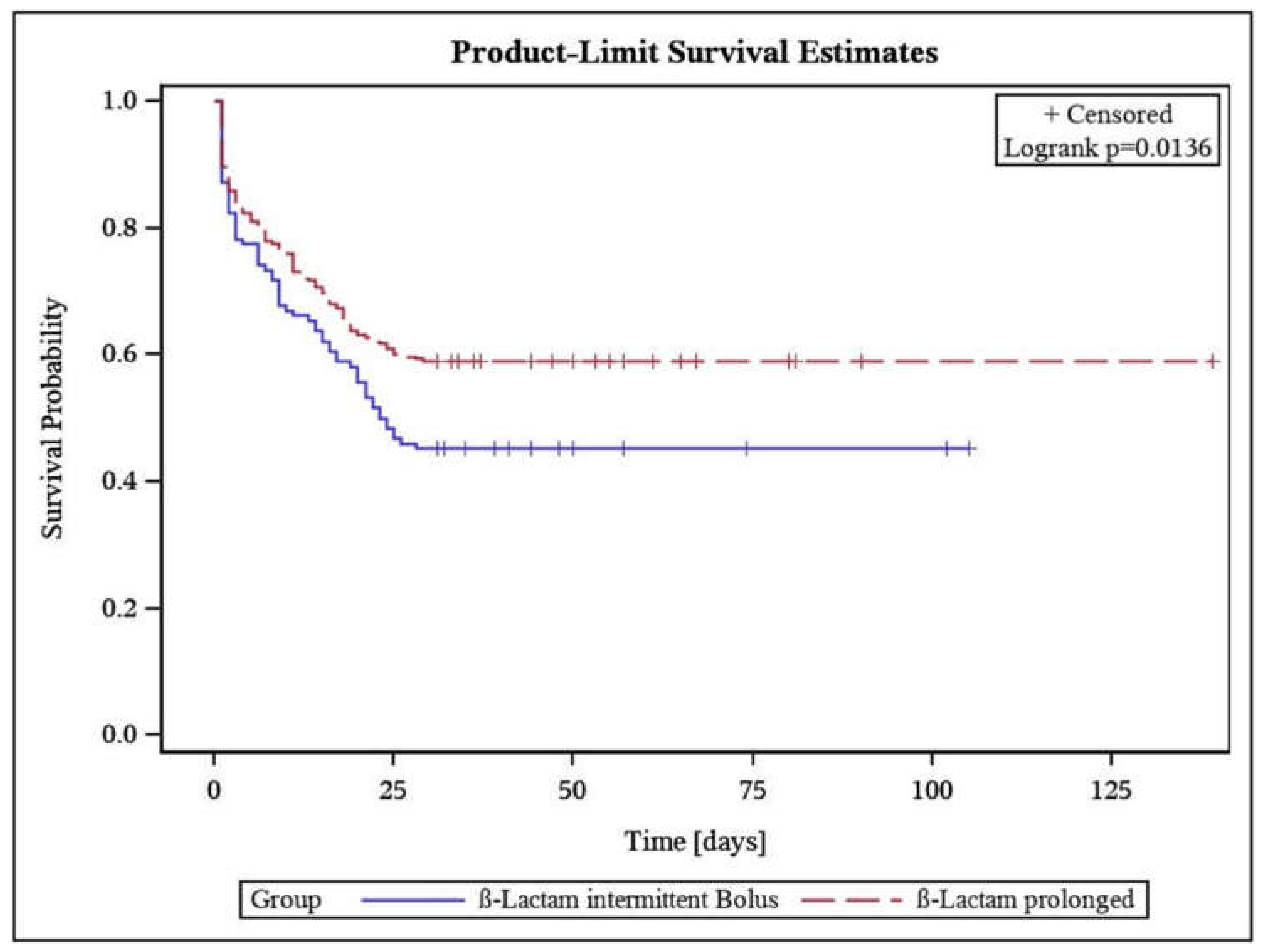
3.2. Primary Outcome
Prolonged β-Lactam Administration Was Associated with Lower Mortality
3.3. Secondary Outcome Parameters
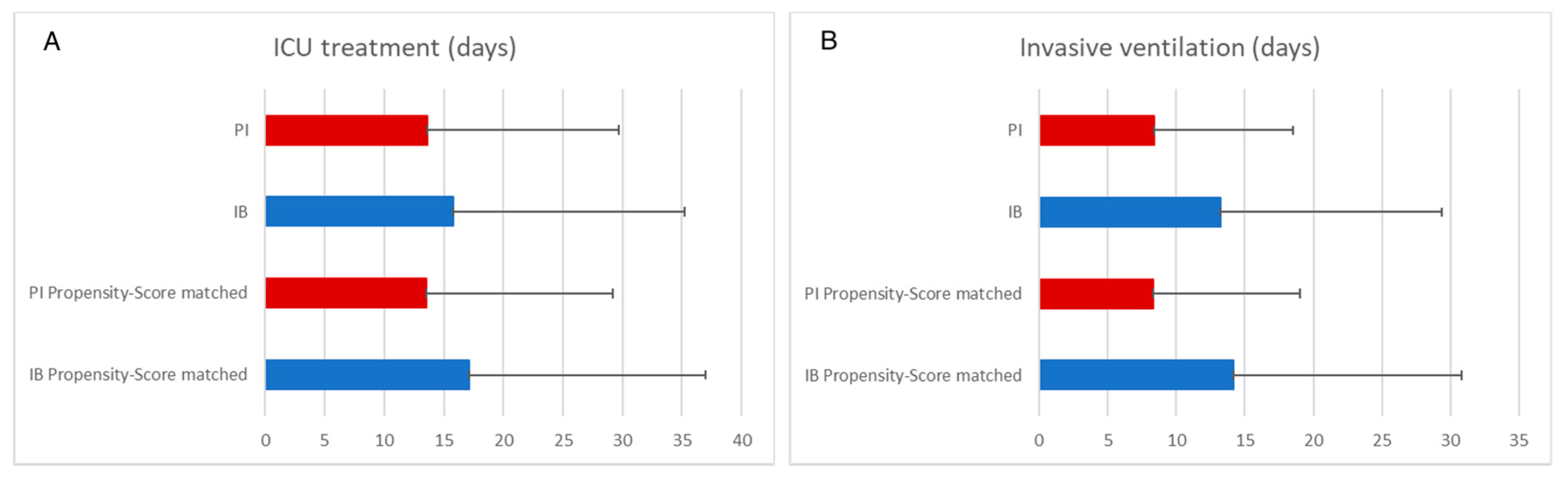
3.3.1. Prolonged β-Lactam Administration Was Associated with Shorter Duration of Invasive Ventilation
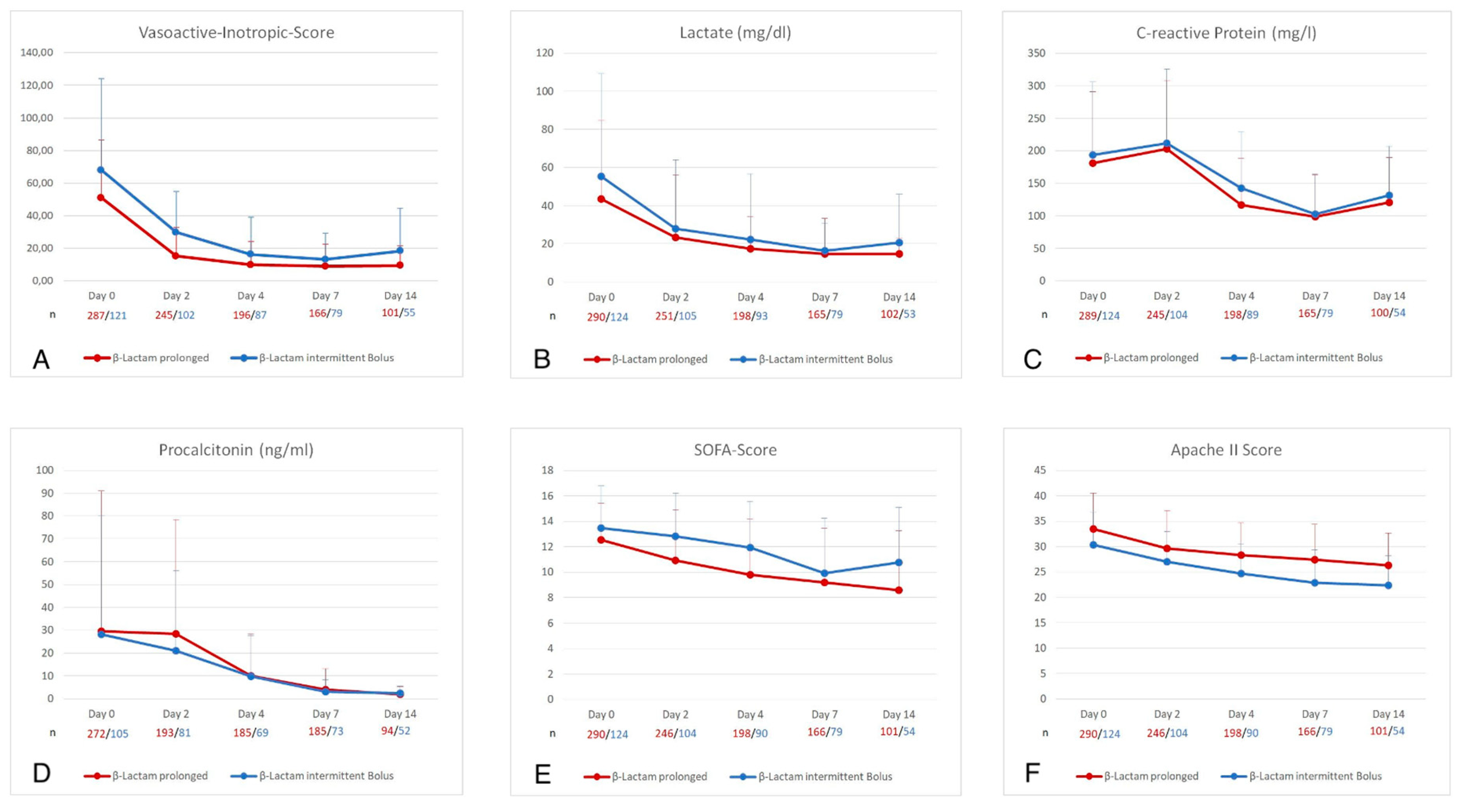
3.3.2. PI of β-Lactams Was Associated with Faster Improvement of Organ Failure
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fleischmann, C.; Scherag, A.; Adhikari, N.K.J.; Hartog, C.S.; Tsaganos, T.; Schlattman, P.; Angus, D.C.; Reinhart, K.; International Forum of Acute Care Trialists. Assessment of global incidence and mortality of hospital-treated sepsis. Current estimates and limitations. Am. J. Respir. Crit. Care Med. 2016, 193, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, C.; Thomas–Rueddel, D.O.; Hartmann, M.; Hartog, C.S.; Welte, T.; Heublein, S.; Dennler, U.; Reinhart, K. Hospital incidence and mortality rates of sepsis. Dtsch. Aerzteblatt Int. 2016, 113, 159–166. [Google Scholar] [CrossRef]
- Group, S.C.C.T. Incidence of severe sepsis and septic shock in German intensive care units: The prospective, multicentre INSEP study. Intensive Care Med. 2016, 42, 1980–1989. [Google Scholar]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelly, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunally, M.E.; et al. Surviving sepsis campaign: International guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 2017, 43, 304–377. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Roberts, D.; Wood, K.E.; Light, B.; Parrillo, J.E.; Sharma, S.; Suppes, R.; Feinstein, D.; Zannoti, S.; Taiberg, L.; et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit. Care Med. 2006, 34, 1589–1596. [Google Scholar] [CrossRef]
- Ferrer, R.; Martin-Loeches, I.; Phillips, G.; Osborn, T.M.; Townsend, S.; Dellinger, R.P.; Artigas, A.; Schorr, C.; Levy, M.M. Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: Results from a guideline-based performance improvement program. Crit. Care Med. 2014, 42, 1749–1755. [Google Scholar] [CrossRef]
- Díaz-Martín, A.; Martínez-González, M.L.; Ferrer, R.; Ortiz-Leyba, C.; Piacentini, E.; Lopez-Pueyo, M.J.; Martín-Loeches, I.; Levy, M.M.; Artigas, A.; Garnacho-Montero, J.; et al. Antibiotic prescription patterns in the empiric therapy of severe sepsis: Combination of antimicrobials with different mechanisms of action reduces mortality. Crit. Care 2012, 16, R223. [Google Scholar] [CrossRef]
- Magill, S.S.; Edwards, J.R.; Beldavs, Z.G.; Dumyati, G.; Janelle, S.J.; Kainer, M.A.; Lynfield, R.; Nadle, J.; Neuhauser, M.M.; Ray, S.M.; et al. Prevalence of antimicrobial use in US acute care hospitals, May–September 2011. JAMA 2014, 312, 1438–1446. [Google Scholar] [CrossRef]
- Roberts, J.A.; Abdul-Aziz, M.H.; Lipman, J.; Mouton, J.W.; Vinks, A.A.; Felton, T.W.; Hope, W.W.; Farkas, A.; Neely, M.N.; Schentag, J.J.; et al. Individualised antibiotic dosing for patients who are critically ill: Challenges and potential solutions. Lancet Infect. Dis. 2014, 14, 498–509. [Google Scholar] [CrossRef]
- Claus, B.O.; Hoste, E.A.; Colpaert, K.; Robays, H.; Decruyenaere, J.; De Waele, J.J. Augmented renal clearance is a common finding with worse clinical outcome in critically ill patients receiving antimicrobial therapy. J. Crit. Care 2013, 28, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Sime, F.; Udy, A.; Roberts, J.A. Augmented renal clearance in critically ill patients: Etiology, definition and implications for beta-lactam dose optimization. Curr. Opin. Pharmacol. 2015, 24, 1–6. [Google Scholar] [CrossRef]
- Udy, A.A.; Varghese, J.M.; Altukroni, M.; Briscoe, S.; McWhinney, B.; Ungerer, J.P.; Lipman, J.; Roberts, J.A. Subtherapeutic initial beta-lactam concentrations in select critically ill patients: Association between augmented renal clearance and low trough drug concentrations. Chest 2012, 142, 30–39. [Google Scholar] [CrossRef]
- Udy, A.A.; Lipman, J.; Jarrett, P.; Klein, K.; Wallis, S.C.; Patel, K.; Kirkpatrick, C.M.; Kruger, P.S.; Paterson, D.L.; Roberts, M.S.; et al. Are standard doses of piperacillin sufficient for critically ill patients with augmented creatinine clearance? Crit. Care 2015, 19, 28. [Google Scholar] [CrossRef]
- Petersson, J.; Giske, C.; Eliasson, E. Standard dosing of piperacillin and meropenem fail to achieve adequate plasma con-centrations in ICU patients. Acta Anaesthesiol. Scand. 2016, 60, 1425–1436. [Google Scholar] [CrossRef] [PubMed]
- Zander, J.; Döbbeler, G.; Nagel, D.; Maier, B.; Scharf, C.; Huseyn-Zada, M.; Jung, J.; Frey, L.; Vogeser, M.; Zoller, M. Piperacillin concentration in relation to therapeutic range in critically ill patients–A prospective observational study. Crit. Care 2016, 20, 79. [Google Scholar] [CrossRef]
- De Waele, J.J.; Lipman, J.; Akova, M.; Bassetti, M.; Dimopoulos, G.; Kaukonen, M.; Koulenti, D.; Martin, C.; Montravers, P.; Rello, J.; et al. Risk factors for target non-attainment during empirical treatment with β-lactam antibiotics in critically ill patients. Intensive Care Med. 2014, 40, 1340–1351. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves-Pereira, J.; Silva, N.E.; Mateus, A.; Pinho, C.; Póvoa, P. Assessment of pharmacokinetic changes of meropenem during therapy in septic critically ill patients. BMC Pharmacol. Toxicol. 2014, 15, 21. [Google Scholar] [CrossRef] [PubMed]
- Kalil, A.C.; Metersky, M.L.; Klompas, M.; Muscedere, J.; Sweeney, D.A.; Palmer, L.B.; Napolitano, L.M.; O’Grady, N.; Bartlett, J.G.; Carratalà, J.; et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin. Infect. Dis. 2016, 63, e61–e111. [Google Scholar] [CrossRef]
- Abdul-Aziz, M.H.; The Infection Section of European Society of Intensive Care Medicine (ESICM); Alffenaar, J.-W.C.; Bassetti, M.; Bracht, H.; Dimopoulos, G.; Marriott, D.; Neely, M.N.; Paiva, J.-A.; Pea, F.; et al. Antimicrobial therapeutic drug monitoring in critically ill adult patients: A Position Paper#. Intensive Care Med. 2020, 46, 1127–1153. [Google Scholar] [CrossRef] [PubMed]
- Veiga, R.P.; Paiva, J.-A. Pharmacokinetics–pharmacodynamics issues relevant for the clinical use of beta-lactam antibiotics in critically ill patients. Crit. Care 2018, 22, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Sinnollareddy, M.G.; Roberts, M.S.; Lipman, J.; Roberts, J.A. β-L actam pharmacokinetics and pharmacodynamics in critically ill patients and strategies for dose optimization: A structured review. Clin. Exp. Pharmacol. Physiol. 2012, 39, 489–496. [Google Scholar] [CrossRef]
- Roberts, J.A.; Paul, S.K.; Akova, M.; Bassetti, M.; De Waele, J.J.; Dimopoulos, G.; Kaukonen, K.-M.; Koulenti, D.; Martin, C.; Montravers, P.; et al. DALI: Defining antibiotic levels in intensive care unit patients: Are current beta-lactam antibiotic doses sufficient for critically ill patients? Clin. Infect. Dis. 2014, 58, 1072–1083. [Google Scholar] [CrossRef]
- Abdul-Aziz, M.H.; Lipman, J.; Akova, M.; Bassetti, M.; De Waele, J.J.; Dimopoulos, G.; Dulhunty, J.; Kaukonen, K.-M.; Koulenti, D.; Martin, C.; et al. Is prolonged infusion of piperacillin/tazobactam and meropenem in critically ill patients associated with improved pharmacokinetic/pharmacodynamic and patient outcomes? An observation from the Defining Antibiotic Levels in Intensive care unit patients (DALI) cohort. J. Antimicrob. Chemother. 2015, 71, 196–207. [Google Scholar] [CrossRef]
- Rhodes, N.J.; Liu, J.; O’Donnell, J.N.; Dulhunty, J.M.; Abdul-Aziz, M.H.; Berko, P.Y.; Nadler, B.; Lipman, J.; Roberts, J.A. Prolonged infusion piperacillin-tazobactam decreases mortality and improves outcomes in severely ill patients: Results of a systematic review and meta-analysis. Crit. Care Med. 2018, 46, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Tansarli, G.S.; Ikawa, K.; Vardakas, K.Z. Clinical outcomes with extended or continuous versus short-term intravenous infusion of carbapenems and piperacillin/tazobactam: A systematic review and meta-analysis. Clin. Infect. Dis. 2012, 56, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Brunkhorst, F.; Weigand, M.A.; Pletz, M.; Gastmeier, P.; Lemmen, S.W.; Meier-Hellmann, A.; Ragaller, M.; Weyland, A.; Marx, G.; Bucher, M.; et al. S3-leitlinie sepsis–prävention, diagnose, therapie und nachsorge. Med. Klin. Intensivmed. Notf. 2020, 115, 37–109. [Google Scholar] [CrossRef] [PubMed]
- Butts, R.J.; Scheurer, M.A.; Atz, A.M.; Zyblewski, S.C.; Hulsey, T.C.; Bradley, S.M.; Graham, E.M. Comparison of maximum vasoactive inotropic score and low cardiac output syndrome as markers of early postoperative outcomes after neonatal cardiac surgery. Pediatr. Cardiol. 2012, 33, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.-L.; Moreno, R.; Takala, J.; Willats, S.; De Mendonça, A.; Bruining, H.; Reinhart, C.K.; Suter, P.M.; Thijs, L.G. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef]
- Knaus, W.A.; Draper, E.A.; Wagner, D.P.; Zimmerman, J.E. APACHE II: A severity of disease classification system. Crit. Care Med. 1985, 13, 818–829. [Google Scholar] [CrossRef]
- Alobaid, A.S.; Hites, M.; Lipman, J.; Taccone, F.S.; Roberts, J.A. Effect of obesity on the pharmacokinetics of antimicrobials in critically ill patients: A structured review. Int. J. Antimicrob. Agents 2016, 47, 259–268. [Google Scholar] [CrossRef]
- Cotta, M.; Roberts, J.; Lipman, J. We need to optimize piperacillin-tazobactam dosing in critically ill patients—But how? Crit. Care 2016, 20, 163. [Google Scholar] [CrossRef]
- Brinkmann, A.; Röhr, A.C.; Köberer, A.; Fuchs, T.; Krüger, W.A.; König, C.; Richter, D.; Weingand, M.A.; Frey, O.R. Adäquate antiinfektivatherapie. Der Anaesthesist 2018, 67, 461–476. [Google Scholar] [CrossRef]
- Craig, W.A. State-of-the-art clinical article: Pharmacokinetic/pharmacodynamic parameters: Rationale for antibacterial dosing of mice and men. Clin. Infect. Dis. 1998, 26, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.A.; Abdul-Aziz, M.-H.; Davis, J.S.; Dulhunty, J.M.; Cota, M.O.; Myburgh, J.; Bellomo, R.; Lipman, J. Continuous versus Intermittent beta-lactam infusion in severe sepsis: A meta-analysis of individual patient data from randomized trials. Am. J. Respir. Crit. Care Med. 2016, 194, 681–691. [Google Scholar] [CrossRef]
- Vardakas, K.Z.; Voulgaris, G.L.; Maliaros, A.; Samonis, G.; Falagas, M.E. Prolonged versus short-term intravenous infusion of antipseudomonal β-lactams for patients with sepsis: A systematic review and meta-analysis of randomised trials. Lancet Infect. Dis. 2018, 18, 108–120. [Google Scholar] [CrossRef]
- Richter, D.C.; Frey, O.; Röhr, A.; Roberts, J.A.; Köberer, A.; Fuchs, T.; Papadimas, N.; Heinzel-Gutenbrunner, M.; Brenner, T.; Lichtenstern, C.; et al. Therapeutic drug monitoring-guided continuous infusion of piperacillin/tazobactam significantly improves pharmacokinetic target attainment in critically ill patients: A retrospective analysis of four years of clinical experience. Infection 2019, 47, 1001–1011. [Google Scholar] [CrossRef]
- Zembles, T.N.; Schortemeyer, R.; Kuhn, E.M.; Bushee, G.; Thompson, N.E.; Mitchell, M.L. Extended infusion of beta-lactams is associated with improved outcomes in pediatric patients. J. Pediatr. Pharmacol. Ther. 2021, 26, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Aziz, M.H.; Sulaiman, H.; Nor, M.B.M.; Rai, V.; Wong, K.K.; Hasan, M.S.; Rahman, A.N.A.; Jamal, J.A.; Wallis, S.C.; Lipman, J.; et al. Beta-lactam infusion in severe sepsis (BLISS): A prospective, two-centre, open-labelled randomised controlled trial of continuous versus intermittent beta-lactam infusion in critically ill patients with severe sepsis. Intensive Care Med. 2016, 42, 1535–1545. [Google Scholar] [CrossRef] [PubMed]
- Dulhunty, J.M.; Roberts, J.A.; Davis, J.S.; Webb, S.A.R.; Bellomo, R.; Gomersall, C.; Shirwadkar, C.; Eastwood, G.M.; Myburgh, J.; Paterson, D.; et al. A multicenter randomized trial of continuous versus intermittent β-lactam infusion in severe sepsis. Am. J. Respir. Crit. Care Med. 2015, 192, 1298–1305. [Google Scholar] [CrossRef]
- Dulhunty, J.M.; Roberts, J.; Davis, J.S.; Webb, S.A.R.; Bellomo, R.; Gomersall, C.; Shirwadkar, C.; Eastwood, G.M.; Myburgh, J.; Paterson, D.; et al. Continuous infusion of beta-lactam antibiotics in severe sepsis: A Multicenter double-blind, randomized controlled trial. Clin. Infect. Dis. 2012, 56, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.E.; Trzeciak, S.; Kline, J.A. The Sequential Organ Failure Assessment score for predicting outcome in patients with severe sepsis and evidence of hypoperfusion at the time of emergency department presentation*. Crit. Care Med. 2009, 37, 1649–1654. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Aziz, M.H.; Portunato, F.; Roberts, J.A. Prolonged infusion of beta-lactam antibiotics for Gram-negative infections: Rationale and evidence base. Curr. Opin. Infect. Dis. 2020, 33, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Bloos, F.; Rüddel, H.; Thomas-Rüddel, D.; Schwarzkopf, D.; Pausch, C.; Harbarth, S.; Schreiber, T.; Gründling, M.; Marshall, J.; Simon, P.; et al. Effect of a multifaceted educational intervention for anti-infectious measures on sepsis mortality: A cluster ran-domized trial. Intensive Care Med. 2017, 43, 1602–1612. [Google Scholar] [CrossRef]
- Bloos, F.; Thomas-Rüddel, D.; Rüddel, H.; Engel, C.; Schwarzkopf, D.; Marshall, J.C.; Harbarth, S.; Simon, P.; Riessen, R.; Keh, D.; et al. Impact of compliance with infection management guidelines on outcome in patients with severe sepsis: A prospective observational multi-center study. Crit. Care 2014, 18, R42. [Google Scholar] [CrossRef]
- Reignier, J.; Mercier, E.; La Gouge, A.; Boulain, T.; Desachy, A.; Bellec, F.; Clavel, M.; Frat, J.-P.; Plantefeve, G.; Quenot, J.-P.; et al. Effect of not monitoring residual gastric volume on risk of ventilator-associated pneumonia in adults receiving mechanical ventilation and early enteral feeding: A randomized controlled trial. JAMA 2013, 309, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Blot, S.I.; Poelaert, J.; Kollef, M. How to avoid microaspiration? A key element for the prevention of ventilator-associated pneumonia in intubated ICU patients. BMC Infect. Dis. 2014, 14, 1–6. [Google Scholar] [CrossRef]
- Sager, R.; Kutz, A.; Mueller, B. Procalcitonin-guided diagnosis and antibiotic stewardship revisited. BMC Med. 2017, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wen, Q.; Yao, F.; Xu, D.; Huang, Y.; Wang, J. Gut–lung axis: The microbial contributions and clinical implications. Crit. Rev. Microbiol. 2017, 43, 81–95. [Google Scholar] [CrossRef]
- Roberts, J.A.; Joynt, G.M.; Lee, A.; Choi, G.; Bellomo, R.; Kanji, S.; Mudaliar, M.Y.; Peake, S.L.; Stephens, D.; Taccone, F.S.; et al. The effect of renal replacement therapy and antibiotic dose on antibiotic concentrations in critically ill patients: Data from the multinational SMARRT Study. Clin. Infect. Dis. 2020, 72, 1369–1378. [Google Scholar] [CrossRef]
- Beumier, M.; Casu, G.S.; Hites, M.; Wolff, F.; Cotton, F.; Vincent, J.L.; Jacobs, F.; Taccone, F.S. Elevated beta-lactam concentrations are associated with neurological deterioration in ICU septic patients. Minerva Anestesiol. 2015, 81, 497–506. [Google Scholar]
- Imani, S.; Buscher, H.; Marriott, D.; Gentili, S.; Sandaradura, I. Too much of a good thing: A retrospective study of β-lactam concentration–toxicity relationships. J. Antimicrob. Chemother. 2017, 72, 2891–2897. [Google Scholar] [CrossRef]
- Beumier, M.; Legros, B.; Wolff, F.; Cotton, F.; Jacobs, F.; Vincent, J.L.; Taccone, F.S. 933: Beta-lactams overdosing is associated with electroencephalographic abnormalities during sepsis. Crit. Care Med. 2013, 41, A234. [Google Scholar] [CrossRef]
- Chow, K.M.; Szeto, C.C.; Hui, A.C.-F.; Wong, T.Y.-H.; Li, P.K.-T. Retrospective review of neurotoxicity induced by cefepime and ceftazidime. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2003, 23, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Hagel, S.; on behalf of the TARGET Study Group; Fiedler, S.; Hohn, A.; Brinkmann, A.; Frey, O.R.; Hoyer, H.; Schlattmann, P.; Kiehntopf, M.; Roberts, J.A.; et al. Therapeutic drug monitoring-based dose optimisation of piperacillin/tazobactam to improve outcome in patients with sepsis (TARGET): A prospective, multi-centre, randomised controlled trial. Trials 2019, 20, 330. [Google Scholar] [CrossRef] [PubMed]
| Baseline (n = 414) | Propensity-Score Matched (n = 224) | |||||
|---|---|---|---|---|---|---|
| Intermittent Bolus (n = 124) | Prolonged Infusion (n = 290) | p-Value | Intermittent Bolus (n = 112) | Prolonged Infusion (n = 112) | p-Value | |
| Demographic data | ||||||
| Male sex | 86 (69%) | 189 (65%) | 0.4091 | 81 (72%) | 81 (72%) | 1.0000 |
| Tumor disease | 68 (55%) | 150 (52%) | 0.5610 | 63 (56%) | 48 (43%) | 0.0450 |
| Organ transplant | 16 (13%) | 28 (10%) | 0.4926 | 16 (14%) | 7 (6%) | 0.1323 |
| Age | 65 (13) | 66 (13) | 0.3792 | 64 (13) | 64 (13) | 0.9309 |
| BMI [kg/m2] | 29 (8) | 29 (9) | 0.9718 | 29 (8) | 28 (10) | 0.3679 |
| Clinical condition | ||||||
| SOFA score | 13.5 (3.3) | 12.6 (2.9) | 0.0069 | 13.2 (3.0) | 12.4 (3.1) | 0.041 |
| APACHE II Score GFR (Cockroft-Gault) | 30.3 (6.4) 56 (46) | 33.5 (7.1) 56 (40) | <0.0001 0.8994 | 29.9 (6.3) 60 (47) | 33.7 (6.6) 57 (50) | <0.0001 0.6158 |
| VIS [μg/kg/min] | 68 (56) | 51 (35) | 0.0073 | 57 (37) | 56 (40) | 0.5616 |
| Creatinine [mg/dL] | 2.1 (1.2) | 2.0 (1.2) | 0.457 | 2.1 (1.2) | 2.0 (1.3) | 0.4988 |
| Serum lactate [mg/dL] | 55 (54) | 44 (41) | 0.0428 | 51 (49) | 46 (47) | 0.5728 |
| Leukocytes [n/mL] | 17 (12) | 17 (12) | 0.6721 | 17 (12) | 15 (11) | 0.5173 |
| CRP [mg/L] | 193 (113) | 181 (182) | 0.1036 | 194 (110) | 193 (256) | 0.1355 |
| Procalcitonin [ng/mL] | 28 (52) | 30 (62) | 0.0534 | 26 (45) | 30 (50) | 0.7274 |
| SOT | % (n) 13 (16/124) | % (n) 10 (28/290) | 0.5 | % (n) 14 (16/112) | % (n) 6 (7/112) | 0.1323 |
| Liver | 63 (10/16) | 46 (13/28) | 63 (10/16) | 71 (5/7) | ||
| Kidney | 19 (3/16) | 36 (10/28) | 19 (3/16) | 14 (1/7) | ||
| Others | 19 (3/16) | 18 (5/28) | 19 (3/16) | 14 (1/7) | ||
| Focus | 0.04 * | 0.108 | ||||
| Abdominal | 66 (82/124) | 76 (221/290) | 65 (73/112) | 78 (87/112) | ||
| Pulmonary | 17 (21/124) | 11 (31/290) | 18 (20/112) | 10 (11/112) | ||
| UTI | 6 (7/124) | 7 (20/290) | 5 (6/112) | 6 (7/112) | ||
| Intermittent Bolus (n = 179) | Prolonged Infusion (n = 451) | ||||
|---|---|---|---|---|---|
| Lead Pathogen (n = 112) | Contributing Pathogen (n = 67) | Lead Pathogen (n = 265) | Contributing Pathogen (n = 186) | ||
| % (n) | % (n) | % (n) | % (n) | ||
| Gram-negative | E. coli | 25 (28) | 20.9 (14) | 29.8 (79) | 16.1 (30) |
| MDR—E. coli * | 1.8 (2) | --- | --- | 0.5 (1) | |
| K. pneumoniae | 9 (10) | 3 (2) | 6.4 (17) | 4.3 (8) | |
| MDR—K. pneumoniae * | none | 1.5 (1) | 0.4 (1) | --- | |
| K. oxytoca | 2.7 (3) | --- | 3.4 (9) | 4.3 (8) | |
| Klebsiella spp. | --- | --- | 0.4 (1) | 1.1 (2) | |
| Citrobacter freundii | 1.8 (2) | 3 (2) | 0.4 (1) | 0.5 (1) | |
| Citrobacter spp. | 1.8 (2) | --- | --- | 1.1 (2) | |
| Serratia marscescens | 2.7 (3) | --- | 0.8 (2) | 0.5 (1) | |
| Serratia spp. | 0.9 (1) | --- | 0.4 (1) | --- | |
| Proteus mirabilis | 2.7 (3) | 1.5 (1) | 4.5 (12) | 1.6 (3) | |
| Proteus spp. | --- | 1.5 (1) | 0.4 (1) | 0.5 (1) | |
| Enterobacter spp. | 3.6 (4) | --- | 2.3 (6) | 5.4 (10) | |
| Pseudomonas aeruginosa | 6.3 (7) | 6 (4) | --- | 5.9 (11) | |
| MDR—P. aeruginosa # | 0.9 (1) | 1.5 (1) | --- | --- | |
| Bacteroides fragilis | 3.6 (4) | 7.5 (5) | 1.5 (4) | 1.1 (2) | |
| Gram-positive | Enterococcus spp. | 4.5 (5) | 10.4 (7) | 3 (8) | 5.9 (11) |
| E. faecium | 9 (10) | 13.4 (9) | 10.9 (29) | 6.5 (12) | |
| E. faecalis | --- | 4.5 (3) | 1.1 (3) | 3.8 (7) | |
| VRE | 8 (9) | 3 (2) | 7.5 (20) | 10.8 (20) | |
| Staphylococcus spp. | 7.1 (8) | 4.5 (3) | 7.5 (25) | 4.3 (8) | |
| MRSA | 0.9 (1) | 3 (2) | --- | --- | |
| Streptococcus spp. | 1.8 (2) | 1.5 (1) | 3.4 (9) | 4.3 (8) | |
| Clostridium spp. | 0.9 (1) | 1.5 (1) | 2.6 (7) | 1.6 (3) | |
| Others | 46 (83/179) | 15 (67/451) | |||
| Baseline (n = 414) Propensity-Score Matched (n = 224) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Intermittent Bolus | Prolonged Infusion | d% | p | Intermittent Bolus | Prolonged Infusion | d% | p | |
| Hospital mortality, % (n) | 64.5 (80/124) | 48.3 (140/290) | 16.2 | 0.0024 | 62.5 (70/112) | 50.9 (57/112) | 11.6 | n.s. |
| ICU mortality, % (n) | 57.3 (71/124) | 43.5 (126/290) | 13.8 | 0.01 | 54.5 (61/112) | 44.6 (50/112) | 9.9 | n.s. |
| 30-day mortality % (n) | 54.8 (68/124) | 41 (119/290) | 13.8 | 0.0097 | 52.7 (59/112) | 42 (47/112) | 10.7 | n.s. |
| 90-day mortality % (n) | 62.9 (78/124) | 47.9 (139/290) | 15 | 0.005 | 60.7 (68/112) | 50.9 (57/112) | 9.8 | n.s. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richter, D.C.; Dietrich, M.; Lalev, L.D.; Schmitt, F.C.F.; Fiedler, M.O.; Bruckner, T.; Stoerzinger, D.; Chiriac, U.; Klein, S.; Hackert, T.; et al. Prolonged Infusion of β-Lactams Decreases Mortality in Patients with Septic Shock: A Retrospective before-and-after Study. Antibiotics 2021, 10, 687. https://doi.org/10.3390/antibiotics10060687
Richter DC, Dietrich M, Lalev LD, Schmitt FCF, Fiedler MO, Bruckner T, Stoerzinger D, Chiriac U, Klein S, Hackert T, et al. Prolonged Infusion of β-Lactams Decreases Mortality in Patients with Septic Shock: A Retrospective before-and-after Study. Antibiotics. 2021; 10(6):687. https://doi.org/10.3390/antibiotics10060687
Chicago/Turabian StyleRichter, Daniel Christoph, Maximilian Dietrich, Lazar Detelinov Lalev, Felix C. F. Schmitt, Mascha Onida Fiedler, Thomas Bruckner, Dominic Stoerzinger, Ute Chiriac, Sabrina Klein, Thilo Hackert, and et al. 2021. "Prolonged Infusion of β-Lactams Decreases Mortality in Patients with Septic Shock: A Retrospective before-and-after Study" Antibiotics 10, no. 6: 687. https://doi.org/10.3390/antibiotics10060687
APA StyleRichter, D. C., Dietrich, M., Lalev, L. D., Schmitt, F. C. F., Fiedler, M. O., Bruckner, T., Stoerzinger, D., Chiriac, U., Klein, S., Hackert, T., Brenner, T., Brinkmann, A., & Weigand, M. A. (2021). Prolonged Infusion of β-Lactams Decreases Mortality in Patients with Septic Shock: A Retrospective before-and-after Study. Antibiotics, 10(6), 687. https://doi.org/10.3390/antibiotics10060687











