Clinical and Microbiological Evaluation of Local Doxycycline and Antimicrobial Photodynamic Therapy during Supportive Periodontal Therapy: A Randomized Clinical Trial
Abstract
1. Introduction
2. Results
3. Discussion
4. Material and Methods
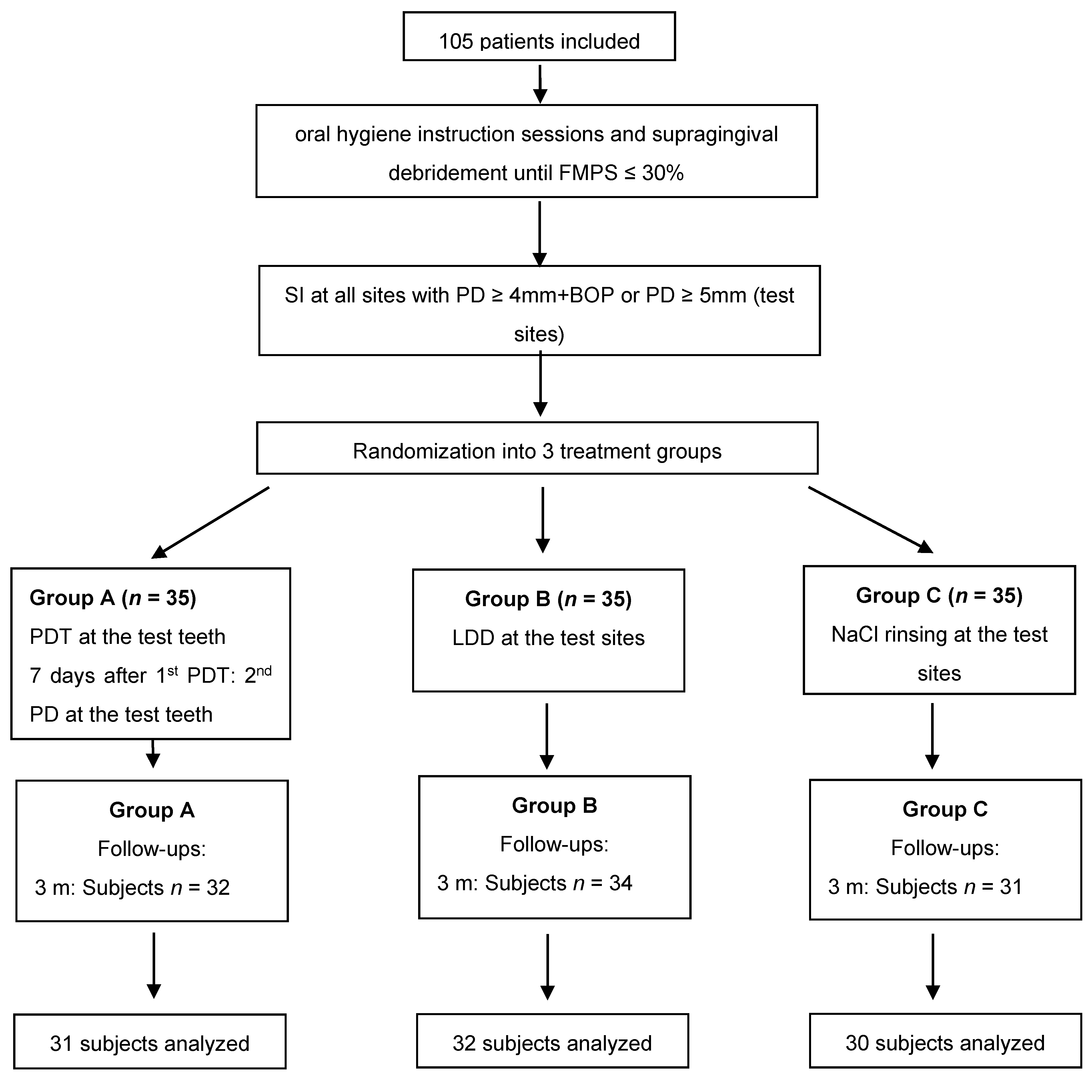
4.1. Study Protocol and Participants
4.2. Clinical Protocol
- Group A:
- session 1: SI plus PDTsession 2: PDT (7 days later)
- Group B:
- session 1: SI plus LDD
- Group C:
- session 1: SI alone
- Session 1: Five minutes after SI and ceasing of bleeding, the photosensitizer (HELBO Blue Photosensitizer, Bredent medical, Senden, Germany) was applied at 6 sites per test tooth from the bottom to the top of the pocket and left in situ for 3 min. Subsequently, the dye (phenothiazine chloride) was rinsed off with sterile saline solution and each site of the treated tooth was exposed to laser light for 10 s (HELBO TheraLite Laser, HELBO 3D Pocket Probe, Bredent medical, Senden, Germany) at a wavelength of 660 nm and an output power of 100 mW.
- Session 2: after 1 week, PDT was repeated at all test teeth as previously described.
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cohen, R.E.; Research, S.; Therapy Committee, A.A.O.P. Position paper: Periodontal maintenance. J. Periodontol. 2003, 74, 1395–1401. [Google Scholar] [CrossRef]
- Sanz, M.; Herrera, D.; Kebschull, M.; Chapple, I.; Jepsen, S.; Beglundh, T.; Sculean, A.; Tonetti, M.S.; Participants, E.F.P.W.; Methodological, C. Treatment of stage I-III periodontitis-The EFP S3 level clinical practice guideline. J. Clin. Periodontol. 2020, 47, 4–60. [Google Scholar] [CrossRef] [PubMed]
- Lang, N.P.; Tonetti, M.S. Periodontal risk assessment (PRA) for patients in supportive periodontal therapy (SPT). Oral Health Prev. Dent. 2003, 1, 7–16. [Google Scholar]
- Matuliene, G.; Pjetursson, B.E.; Salvi, G.E.; Schmidlin, K.; Brägger, U.; Zwahlen, M.; Lang, N.P. Influence of residual pockets on progression of periodontitis and tooth loss: Results after 11 years of maintenance. J. Clin. Periodontol. 2008, 35, 685–695. [Google Scholar] [CrossRef]
- Bogren, A.; Teles, R.P.; Torresyap, G.; Haffajee, A.D.; Socransky, S.S.; Wennstrom, J.L. Locally delivered doxycycline during supportive periodontal therapy: A 3-year study. J. Periodontol. 2008, 79, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Campos, G.N.; Pimentel, S.P.; Ribeiro, F.V.; Casarin, R.C.; Cirano, F.R.; Saraceni, C.H.; Casati, M.Z. The adjunctive effect of photodynamic therapy for residual pockets in single-rooted teeth: A randomized controlled clinical trial. Lasers Med. Sci. 2013, 28, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Chondros, P.; Nikolidakis, D.; Christodoulides, N.; Rossler, R.; Gutknecht, N.; Sculean, A. Photodynamic therapy as adjunct to non-surgical periodontal treatment in patients on periodontal maintenance: A randomized controlled clinical trial. Lasers Med. Sci. 2009, 24, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Dannewitz, B.; Lippert, K.; Lang, N.P.; Tonetti, M.S.; Eickholz, P. Supportive periodontal therapy of furcation sites: Non-surgical instrumentation with or without topical doxycycline. J. Clin. Periodontol. 2009, 36, 514–522. [Google Scholar] [CrossRef]
- Kolbe, M.F.; Ribeiro, F.V.; Luchesi, V.H.; Casarin, R.C.; Sallum, E.A.; Nociti, F.H., Jr.; Ambrosano, G.M.; Cirano, F.R.; Pimentel, S.P.; Casati, M.Z. Photodynamic therapy during supportive periodontal care: Clinical, microbiologic, immunoinflammatory, and patient-centered performance in a split-mouth randomized clinical trial. J. Periodontol. 2014, 85, 277–286. [Google Scholar] [CrossRef]
- Bonito, A.J.; Lux, L.; Lohr, K.N. Impact of local adjuncts to scaling and root planing in periodontal disease therapy: A systematic review. J. Periodontol. 2005, 76, 1227–1236. [Google Scholar] [CrossRef] [PubMed]
- Eickholz, P.; Kim, T.S.; Schacher, B.; Reitmeir, P.; Burklin, T.; Ratka-Kruger, P. Subgingival topical doxycycline versus mechanical debridement for supportive periodontal therapy: A single blind randomized controlled two-center study. Am. J. Dent. 2005, 18, 341–346. [Google Scholar]
- Chan, Y.; Lai, C.H. Bactericidal effects of different laser wavelengths on periodontopathic germs in photodynamic therapy. Lasers Med. Sci. 2003, 18, 51–55. [Google Scholar] [CrossRef]
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic therapy. J. Natl. Cancer Inst. 1998, 90, 889–905. [Google Scholar] [CrossRef] [PubMed]
- Soukos, N.S.; Goodson, J.M. Photodynamic therapy in the control of oral biofilms. Periodontol. 2000 2011, 55, 143–166. [Google Scholar] [CrossRef] [PubMed]
- Von Tappeiner, H.J. Über die Wirkung der photodynamischen (fluorescierenden) Stoffe auf Protozoen und Enzyme. Dtsch. Arch. Klin. Med. 1904, 39, 427–487. [Google Scholar]
- Sarkar, S.; Wilson, M. Lethal photosensitization of bacteria in subgingival plaque from patients with chronic periodontitis. J. Periodontal. Res. 1993, 28, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Dobson, J.; Wilson, M. Sensitization of oral bacteria in biofilms to killing by light from a low-power laser. Arch. Oral Biol. 1992, 37, 883–887. [Google Scholar] [CrossRef]
- Pfitzner, A.; Sigusch, B.W.; Albrecht, V.; Glockmann, E. Killing of periodontopathogenic bacteria by photodynamic therapy. J. Periodontol. 2004, 75, 1343–1349. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.; Dobson, J.; Harvey, W. Sensitization of oral bacteria to killing by low-power laser radiation. Curr. Microbiol. 1992, 25, 77–81. [Google Scholar] [CrossRef]
- Luchesi, V.H.; Pimentel, S.P.; Kolbe, M.F.; Ribeiro, F.V.; Casarin, R.C.; Nociti, F.H., Jr.; Sallum, E.A.; Casati, M.Z. Photodynamic therapy in the treatment of class II furcation: A randomized controlled clinical trial. J. Clin. Periodontol. 2013, 40, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Theodoro, L.H.; Silva, S.P.; Pires, J.R.; Soares, G.H.; Pontes, A.E.; Zuza, E.P.; Spolidorio, D.M.; de Toledo, B.E.; Garcia, V.G. Clinical and microbiological effects of photodynamic therapy associated with nonsurgical periodontal treatment. A 6-month follow-up. Lasers Med. Sci. 2012, 27, 687–693. [Google Scholar] [CrossRef]
- Arweiler, N.B.; Pietruska, M.; Pietruski, J.; Skurska, A.; Dolinska, E.; Heumann, C.; Auschill, T.M.; Sculean, A. Six-month results following treatment of aggressive periodontitis with antimicrobial photodynamic therapy or amoxicillin and metronidazole. Clin. Oral Investig. 2014, 18, 2129–2135. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Atieh, M.A. Photodynamic therapy as an adjunctive treatment for chronic periodontitis: A meta-analysis. Lasers Med. Sci. 2010, 25, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Azarpazhooh, A.; Shah, P.S.; Tenenbaum, H.C.; Goldberg, M.B. The effect of photodynamic therapy for periodontitis: A systematic review and meta-analysis. J. Periodontol. 2010, 81, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Sgolastra, F.; Severino, M.; Gatto, R.; Monaco, A. Effectiveness of diode laser as adjunctive therapy to scaling root planning in the treatment of chronic periodontitis: A meta-analysis. Lasers Med. Sci. 2013, 28, 1393–1402. [Google Scholar] [CrossRef] [PubMed]
- Salvi, G.E.; Stahli, A.; Schmidt, J.C.; Ramseier, C.A.; Sculean, A.; Walter, C. Adjunctive laser or antimicrobial photodynamic therapy to non-surgical mechanical instrumentation in patients with untreated periodontitis: A systematic review and meta-analysis. J. Clin. Periodontol. 2020, 47, 176–198. [Google Scholar] [CrossRef] [PubMed]
- Konopka, K.; Goslinski, T. Photodynamic therapy in dentistry. J. Dent. Res. 2007, 86, 694–707. [Google Scholar] [CrossRef]
- Sculean, A.; Deppe, H.; Miron, R.; Schwarz, F.; Romanos, G.; Cosgarea, R. Effectiveness of Photodynamic Therapy in the Treatment of Periodontal and Peri-Implant Diseases. Monogr. Oral Sci. 2021, 29, 133–143. [Google Scholar] [CrossRef]
- Tabenski, L.; Moder, D.; Cieplik, F.; Schenke, F.; Hiller, K.A.; Buchalla, W.; Schmalz, G.; Christgau, M. Antimicrobial photodynamic therapy vs. local minocycline in addition to non-surgical therapy of deep periodontal pockets: A controlled randomized clinical trial. Clin. Oral Investig. 2017, 21, 2253–2264. [Google Scholar] [CrossRef]
- Bassetti, M.; Schar, D.; Wicki, B.; Eick, S.; Ramseier, C.A.; Arweiler, N.B.; Sculean, A.; Salvi, G.E. Anti-infective therapy of peri-implantitis with adjunctive local drug delivery or photodynamic therapy: 12-month outcomes of a randomized controlled clinical trial. Clin. Oral Implant. Res. 2014, 25, 279–287. [Google Scholar] [CrossRef]
- Petelin, M.; Perkic, K.; Seme, K.; Gaspirc, B. Effect of repeated adjunctive antimicrobial photodynamic therapy on subgingival periodontal pathogens in the treatment of chronic periodontitis. Lasers Med. Sci. 2015, 30, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Ruhling, A.; Fanghanel, J.; Houshmand, M.; Kuhr, A.; Meisel, P.; Schwahn, C.; Kocher, T. Photodynamic therapy of persistent pockets in maintenance patients-a clinical study. Clin. Oral Investig. 2010, 14, 637–644. [Google Scholar] [CrossRef]
- Siva, N.T.D.; Silva, D.N.A.; Azevedo, M.; Silva Junior, F.L.D.; Almeida, M.L.; Longo, J.P.F.; Moraes, M.; Gurgel, B.C.V.; de Aquino Martins, A.R.L. The effectiveness of photodynamic therapy as a complementary therapy to mechanical instrumentation on residual periodontal pocket clinical parameters: A clinical split-mouth test. Photodiagn. Photodyn. 2020, 29, 101565. [Google Scholar] [CrossRef] [PubMed]
- Lulic, M.; Leiggener Gorog, I.; Salvi, G.E.; Ramseier, C.A.; Mattheos, N.; Lang, N.P. One-year outcomes of repeated adjunctive photodynamic therapy during periodontal maintenance: A proof-of-principle randomized-controlled clinical trial. J. Clin. Periodontol. 2009, 36, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Muller Campanile, V.S.; Giannopoulou, C.; Campanile, G.; Cancela, J.A.; Mombelli, A. Single or repeated antimicrobial photodynamic therapy as adjunct to ultrasonic debridement in residual periodontal pockets: Clinical, microbiological, and local biological effects. Lasers Med. Sci. 2015, 30, 27–34. [Google Scholar] [CrossRef]
- Grzech-Lesniak, K.; Gaspirc, B.; Sculean, A. Clinical and microbiological effects of multiple applications of antibacterial photodynamic therapy in periodontal maintenance patients. A randomized controlled clinical study. Photodiagn. Photodyn. 2019, 27, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Cappuyns, I.; Cionca, N.; Wick, P.; Giannopoulou, C.; Mombelli, A. Treatment of residual pockets with photodynamic therapy, diode laser, or deep scaling. A randomized, split-mouth controlled clinical trial. Lasers Med. Sci. 2012, 27, 979–986. [Google Scholar] [CrossRef]
- Killeen, A.C.; Harn, J.A.; Erickson, L.M.; Yu, F.; Reinhardt, R.A. Local Minocycline Effect on Inflammation and Clinical Attachment During Periodontal Maintenance: Randomized Clinical Trial. J. Periodontol. 2016, 87, 1149–1157. [Google Scholar] [CrossRef]
- Tomasi, C.; Koutouzis, T.; Wennstrom, J.L. Locally delivered doxycycline as an adjunct to mechanical debridement at retreatment of periodontal pockets. J. Periodontol. 2008, 79, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Lang, N.P.; Cortellini, P.; Suvan, J.E.; Eickholz, P.; Fourmousis, I.; Topoll, H.; Vangsted, T.; Wallkamm, B. Effects of a single topical doxycycline administration adjunctive to mechanical debridement in patients with persistent/recurrent periodontitis but acceptable oral hygiene during supportive periodontal therapy. J. Clin. Periodontol. 2012, 39, 475–482. [Google Scholar] [CrossRef]
- Tomasi, C.; Wennstrom, J.L. Locally delivered doxycycline as an adjunct to mechanical debridement at retreatment of periodontal pockets: Outcome at furcation sites. J. Periodontol. 2011, 82, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Mombelli, A. Microbial colonization of the periodontal pocket and its significance for periodontal therapy. Periodontol. 2000 2018, 76, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Schär, D.; Ramseier, C.A.; Eick, S.; Arweiler, N.B.; Sculean, A.; Salvi, G.E. Anti-infective therapy of peri-implantitis with adjunctive local drug delivery or photodynamic therapy: Six-month outcomes of a prospective randomized clinical trial. Clin. Oral Implant. Res. 2013, 24, 104–110. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, T.J.; Drake, R.B.; Naylor, J.E. The plaque control record. J. Periodontol. 1972, 43, 38. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Pini-Prato, G.; Cortellini, P. Effect of cigarette smoking on periodontal healing following GTR in infrabony defects. A preliminary retrospective study. J. Clin. Periodontol. 1995, 22, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Ainamo, J.; Bay, I. Problems and proposals for recording gingivitis and plaque. Int. Dent. J. 1975, 25, 229–235. [Google Scholar] [PubMed]
- Jentsch, H.F.R.; Heusinger, T.; Weickert, A.; Eick, S. Professional tooth cleaning prior to non-surgical periodontal therapy: A randomized clinical trial. J. Periodontol. 2020, 91, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Sculean, A.; Rothamel, D.; Schwenzer, K.; Georg, T.; Becker, J. Clinical evaluation of an Er:YAG laser for nonsurgical treatment of peri-implantitis: A pilot study. Clin. Oral Implant. Res. 2005, 16, 44–52. [Google Scholar] [CrossRef]
| Parameters | All | Group A | Group B | Group C | Group Comparisons | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | % | Mean ± SD | n | % | Mean ± SD | n | % | Mean ± SD | n | % | Mean ± SD | p-Value | |
| n | 105 | 100 | 35 | 33.33 | 35 | 33.33 | 35 | 33.33 | |||||
| Gender (female/male) | 64/41 | 61/39 | 21 | 60.00 | 23 | 65.71 | 20 | 57.14 | 0.756 (¶) | ||||
| Age | 46 ± 10 | 46 ± 11 | 48 ± 9 | 44 ± 9.15 | 0.480 (§) | ||||||||
| Diagnosis | |||||||||||||
| Stage I grade A | 3 | 2.86 | 1 | 2.86 | 0 | 0.00 | 2 | 5.71 | |||||
| Stage II grade A | 1 | 0.95 | 0 | 0.00 | 1 | 2.86 | 0 | 0.00 | |||||
| Stage II grade B | 16 | 15.24 | 6 | 17.14 | 1 | 2.86 | 9 | 25.71 | |||||
| Stage III grade A | 2 | 1.90 | 1 | 2.86 | 0 | 0.00 | 1 | 2.86 | |||||
| Stage III grade B | 44 | 41.90 | 17 | 48.57 | 15 | 42.86 | 12 | 34.29 | |||||
| Stage III grade C | 19 | 18.10 | 5 | 14.29 | 6 | 17.14 | 8 | 22.86 | |||||
| Stage IV grade B | 10 | 9.52 | 3 | 8.57 | 6 | 17.14 | 1 | 2.86 | |||||
| Stage IV grade C | 10 | 9.52 | 2 | 5.71 | 6 | 17.14 | 2 | 5.71 | |||||
| Non-smokers | 76 | 72.38 | 30 | 85.71 | 23 | 65.71 | 23 | 65.71 | |||||
| Smokers | 23 | 21.90 | 2 | 5.71 | 9 | 25.71 | 12 | 34.29 | 0.018 (£) | ||||
| Former smokers | 6 | 5.71 | 3 | 8.57 | 3 | 8.57 | 0 | 0.00 | |||||
| Parameters | All | Group A | Group B | Group C | Inter-Group | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | min | max | Mean | SD | min | max | Mean | SD | min | max | Mean | SD | min | max | p-Value (£) | |
| BOP (BL) | 16.82 | 8.67 | 3.33 | 42.3 | 16.52 | 8.31 | 4.16 | 42.3 | 17.36 | 9.33 | 4.2 | 41.1 | 16.57 | 8.57 | 3.33 | 39.8 | 0.949 |
| BOP (3 MO) | 13.37 | 7.54 | 1.6 | 42.7 | 12.29 | 6.1 | 2.5 | 23.9 | 13.9 | 8.17 | 1.6 | 42.7 | 13.92 | 8.28 | 2.4 | 38.7 | 0.779 |
| BOP (6 MO) | 12.66 | 7.6 | 0 | 41.6 | 10.75 | 7.12 | 0 | 26.6 | 13.23 | 9.11 | 0 | 41.6 | 14.04 | 6.07 | 1.8 | 25.9 | 0.133 |
| p-value (*) | 0.003 | 0.038 | 0.113 | 0.312 | |||||||||||||
| GBI (BL) | 3.67 | 6.07 | 0 | 28 | 2.56 | 4.5 | 0 | 22.4 | 4.39 | 6.75 | 0 | 27.7 | 4.07 | 6.71 | 0 | 28 | 0.829 |
| GBI (3 MO) | 4.77 | 5.61 | 0 | 21.3 | 3.4 | 4.41 | 0 | 15.4 | 5.37 | 6.02 | 0 | 21.3 | 5.54 | 6.17 | 0 | 17.8 | 0.237 |
| GBI (6 MO) | 4.59 | 5.99 | 0 | 22.8 | 3.85 | 5.54 | 0 | 22.8 | 4.92 | 5.65 | 0 | 19 | 5 | 6.86 | 0 | 22.3 | 0.713 |
| p-value (*) | 0.007 | 0.282 | 0.094 | 0.227 | |||||||||||||
| FMPS (BL) | 18.43 | 6.97 | 1.7 | 33 | 18.32 | 7.32 | 4.2 | 27.3 | 18.39 | 6.2 | 7 | 33 | 18.57 | 7.51 | 1.7 | 29 | 0.736 |
| FMPS (3 MO) | 22.69 | 12.34 | 0 | 59.7 | 21.51 | 11.76 | 4.3 | 59.7 | 24.94 | 12.48 | 3.8 | 52.7 | 21.4 | 12.84 | 0 | 50.8 | 0.401 |
| FMPS (6 MO) | 24.71 | 14.64 | 0 | 66 | 21.88 | 10.9 | 2.5 | 53.8 | 24.53 | 14.22 | 0 | 64.4 | 27.83 | 17.94 | 0 | 66 | 0.472 |
| p-value (*) | 0.002 | 0.407 | 0.038 | 0.0223 | |||||||||||||
| Groups | Baseline | 3 Months | 6 Months | Intra-Group | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n pat. | Mean n Teeth (±SD) | Mean BoP+ Sites/Tooth (±SD) | min | max | n pat. | Mean n Teeth (±SD) | Mean BoP+ Sites/Tooth (±SD) | min | max | n pat. | Mean n Teeth (±SD) | Mean BoP+ Sites/Tooth (±SD) | min | max | p-Value | ||
| Group A | 35 | 5.43 (±1.96) | 1.71 (±0.69) | 0.38 | 3.20 | 32 | 4.91 (±2.43) | 1.21 (±0.75) | 0.00 | 3.50 | 31 | 4.89 (±2.62) | 0.17 (±0.12) | 0.00 | 0.52 | <0.001 | (*) |
| Group B | 35 | 5.34 (±2.13) | 1.82 (±0.78) | 0.50 | 3.60 | 34 | 4.97 (±1.87) | 1.12 (±0.68) | 0.25 | 3.00 | 32 | 4.97 (±2.61) | 0.16 (±0.12) | 0.00 | 0.43 | <0.001 | (*) |
| Group C | 35 | 5.49 (±2.17) | 1.92 (±0.91) | 0.60 | 4.25 | 31 | 5.00 (±2.80) | 1.12 (±0.70) | 0.00 | 3.50 | 30 | 5.11 (±3.35) | 0.20 (±0.11) | 0.04 | 0.47 | <0.001 | (*) |
| p-value (intergroup) | 0.778 | (£) | 0.835 | (£) | 0.182 | (£) | |||||||||||
| Groups | Baseline | 3 Months | 6 Months | Intra-Group | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n pat. | Mean n Teeth (±SD) | Mean PD (±SD) | min | max | n pat. | Mean n Teeth (±SD) | Mean PD (±SD) | min | max | n pat. | Mean n Teeth (±SD) | Mean PD (±SD) | min | max | p-Value | ||
| Group A | 35 | 5.43 (±1.96) | 2.96 (±0.30) | 2.37 | 3.67 | 32 | 4.91 (±2.43) | 2.72 (±0.30) | 2.23 | 3.63 | 31 | 4.89 (±2.62) | 2.75 (±0.39) | 2.06 | 3.46 | 0.001 | (*) |
| Group B | 35 | 5.34 (±2.13) | 2.94 (±0.20) | 2.50 | 3.38 | 34 | 4.97 (±1.87) | 2.64 (±0.26) | 2.17 | 3.33 | 32 | 4.97 (±2.61) | 2.66 (±0.28) | 2.11 | 3.30 | <0.001 | (*) |
| Group C | 35 | 5.49 (±2.17) | 2.97 (±0.24) | 2.63 | 3.54 | 31 | 5.00 (±2.80) | 2.66 (±0.30) | 2.21 | 3.49 | 30 | 5.11 (±3.35) | 2.71 (±0.34) | 2.17 | 3.80 | <0.001 | (*) |
| p-value (intergroup) | 0.969 | (£) | 0.453 | (£) | 0.641 | (£) | |||||||||||
| Groups | Baseline | 3 Months | 6 Months | Intra-Group | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n pat. | Mean n Teeth (±SD) | Mean CAL (±SD) | min | max | n pat. | Mean n Teeth (±SD) | Mean CAL (±SD) | min | max | n pat. | Mean n Teeth (±SD) | Mean CAL (±SD) | min | max | p-Value | ||
| Group A | 35 | 5.43 (±1.96) | 3.67 (±0.81) | 2.33 | 5.82 | 32 | 4.91 (±2.43) | 3.48 (±0.90) | 2.08 | 5.99 | 31 | 4.89 (±2.62) | 3.53 (±0.86) | 1.96 | 5.99 | 0.004 | (*) |
| Group B | 35 | 5.34 (±2.13) | 4.13 (±0.97) | 2.88 | 7.00 | 34 | 4.97 (±1.87) | 3.91 (±1.12) | 2.54 | 7.00 | 32 | 4.97 (±2.61) | 3.83 (±1.10) | 2.42 | 7.25 | <0.001 | (*) |
| Group C | 35 | 5.49 (±2.17) | 3.66 (±0.83) | 2.39 | 6.15 | 31 | 5.00 (±2.80) | 3.38 (±0.84) | 2.31 | 6.12 | 30 | 5.11 (±3.35) | 3.51 (±0.88) | 2.19 | 5.58 | <0.001 | (*) |
| p-value (intergroup) | 0.041 | (£) | 0.064 | (£) | 0.409 | ||||||||||||
| Variables | Group A | Group B | Group C | Inter-Group p-Value (£) |
|---|---|---|---|---|
| A. actinomycetemecomitans (log10) | ||||
| Baseline | 0.74 ± 1.67 | 0.68 ± 1.72 | 0.63 ± 1.79 | 0.899 |
| 3 m | 0.51 ± 1.37 | 0.70 ± 1.59 | 0.47 ± 1.47 | 0.713 |
| 6 m | 0.31 ± 1.15 | 0.70 ± 1.58 | 0.20 ± 1.07 | 0.235 |
| p value (*) | 0.094 | 0.580 | 0.905 | |
| P. gingivalis (log10) | ||||
| Baseline | 3.45 ± 2.97 | 3.69 ± 2.97 | 3.84 ± 2.83 | 0.919 |
| 3 months | 3.66 ± 3.12 | 2.92 ± 2.72 a | 3.85 ± 2.96 | 0.201 |
| 6 months | 3.74 ± 2.96 | 2.64 ± 3.04 | 4.26 ± 3.11 c | 0.103 |
| p value (*) | 0.314 | 0.170 | 0.158 | |
| T. denticola (log10) | ||||
| Baseline | 3.78 ± 2.96 | 3.06 ± 2.90 | 3.63 ± 2.50 | 0.611 |
| 3 m | 3.78 ± 2.80 | 2.13 ± 2.51 a | 3.63 ± 2.60 a | 0.013 s |
| 6 m | 3.66 ± 2.78 c | 2.29 ± 2.65 | 4.11 ± 2.55 a,b | 0.015 s |
| p value (*) | 0.291 | 0.014 | 0.026 | |
| T. forsythia (log10) | ||||
| Baseline | 4.88 ± 2.36 | 4.72 ± 2.53 | 4.61 ± 2.68 | 1.000 |
| 3 m | 5.30 ± 2.02 | 4.04 ± 2.63 a | 4.93 ± 2.31 | 0.034 |
| 6 m | 5.04 ± 2.32 | 3.48 ± 2.89 b | 5.14 ± 2.29 | 0.020 s |
| p value (*) | 0.664 | 0.026 s | 0.508 | |
| P. intermedia (log10) | ||||
| Baseline | 2.96 ± 3.00 | 2.19 ± 2.97 | 2.33 ± 2.88 | 0.481 |
| 3 m | 3.81 ± 3.15 a | 1.87 ± 2.66 | 2.11 ± 2.76 | 0.011 s |
| 6 m | 3.88 ± 2.93 | 1.48 ± 2.51 | 2.19 ± 2.95 | 0.011 s |
| p value (*) | 0.076 | 0.538 | 0.360 | |
| F. nucleatum (log10) | ||||
| Baseline | 6.87 ± 0.98 | 6.53 ± 1.91 | 6.49 ± 1.28 | 0.455 |
| 3 m | 6.87 ± 1.13 | 6.42 ± 1.59 | 6.74 ± 1.11 | 0.456 |
| 6 m | 6.71 ± 1.03 | 6.51 ± 1.03 | 6.99 ± 0.95 b | 0.194 |
| p value (*) | 0.432 | 0.764 | 0.012 s | |
| C. rectus (log10) | ||||
| Baseline | 4.35 ± 2.78 | 4.15 ± 3.02 | 3.66 ± 3.02 | 0.534 |
| 3 m | 4.21 ± 3.09 | 2.96 ± 2.97 a | 3.90 ± 2.85 | 0.129 |
| 6 m | 3.99 ± 3.07 c | 2.38 ± 3.14 b | 4.48 ± 3.02 | 0.056 |
| p value (*) | 0.202 | 0.055 | 0.404 | |
| F. allocis (log10) | ||||
| Baseline | 5.23 ± 2.38 | 4.98 ± 2.60 | 4.92 ± 2.74 | 0.959 |
| 3 m | 4.93 ± 2.75 | 3.88 ± 2.59 a | 5.21 ± 2.34 | 0.025 s |
| 6 m | 4.99 ± 2.72 | 3.31 ± 3.13 b | 5.37 ± 2.55 | 0.016 s |
| p value (*) | 0.180 | 0.016 s | 0.318 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cosgarea, R.; Eick, S.; Batori-Andronescu, I.; Jepsen, S.; Arweiler, N.B.; Rößler, R.; Conrad, T.; Ramseier, C.A.; Sculean, A. Clinical and Microbiological Evaluation of Local Doxycycline and Antimicrobial Photodynamic Therapy during Supportive Periodontal Therapy: A Randomized Clinical Trial. Antibiotics 2021, 10, 277. https://doi.org/10.3390/antibiotics10030277
Cosgarea R, Eick S, Batori-Andronescu I, Jepsen S, Arweiler NB, Rößler R, Conrad T, Ramseier CA, Sculean A. Clinical and Microbiological Evaluation of Local Doxycycline and Antimicrobial Photodynamic Therapy during Supportive Periodontal Therapy: A Randomized Clinical Trial. Antibiotics. 2021; 10(3):277. https://doi.org/10.3390/antibiotics10030277
Chicago/Turabian StyleCosgarea, Raluca, Sigrun Eick, Ionela Batori-Andronescu, Søren Jepsen, Nicole B. Arweiler, Ralf Rößler, Torsten Conrad, Christoph A. Ramseier, and Anton Sculean. 2021. "Clinical and Microbiological Evaluation of Local Doxycycline and Antimicrobial Photodynamic Therapy during Supportive Periodontal Therapy: A Randomized Clinical Trial" Antibiotics 10, no. 3: 277. https://doi.org/10.3390/antibiotics10030277
APA StyleCosgarea, R., Eick, S., Batori-Andronescu, I., Jepsen, S., Arweiler, N. B., Rößler, R., Conrad, T., Ramseier, C. A., & Sculean, A. (2021). Clinical and Microbiological Evaluation of Local Doxycycline and Antimicrobial Photodynamic Therapy during Supportive Periodontal Therapy: A Randomized Clinical Trial. Antibiotics, 10(3), 277. https://doi.org/10.3390/antibiotics10030277








