An Outbreak of tet(X6)-Carrying Tigecycline-Resistant Acinetobacter baumannii Isolates with a New Capsular Type at a Hospital in Taiwan
Abstract
:1. Introduction
2. Results
2.1. Screening of tet(X) Variants
2.2. Antimicrobial Susceptibility of Strains Carrying tet(X) Variants
2.3. The Genomes of A. baumannii Isolates Carrying tet(X) Variants Are Highly Similar and Carry Two Plasmids
2.4. Tigecycline-Resistant A. baumannii Isolates Carrying tet(X) Variants Also Carry Other Antimicrobial Resistance Genes
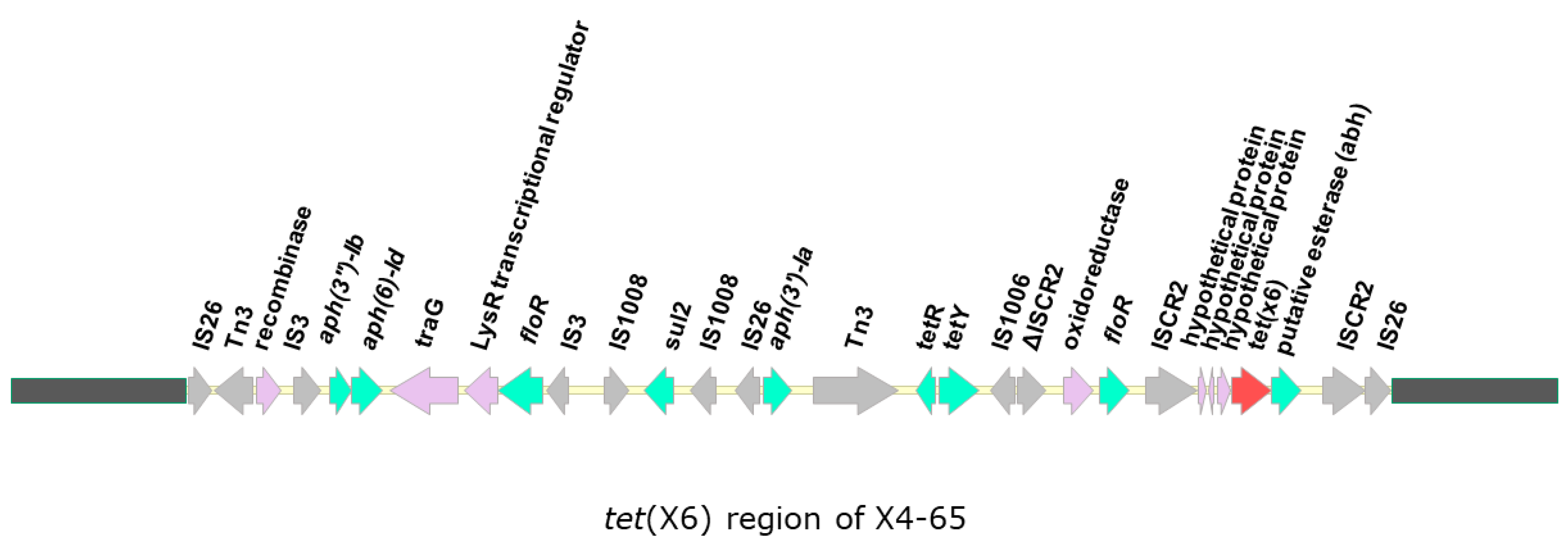
2.5. The Tigecycline-Resistant A. baumannii Isolates Carry tet(X6) Genes
2.6. The A. baumannii Isolates Showed No Evidence of Conjugal Transfer of tet(X6)
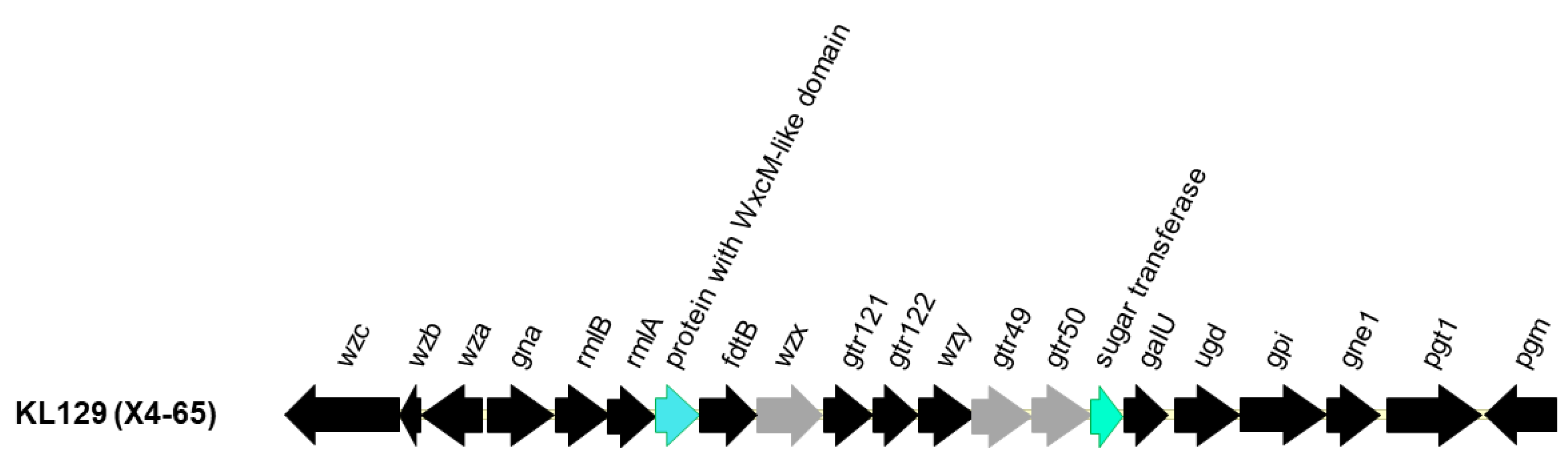
2.7. K Type and Sequence-Tyype (ST) of the Tigecycline-Resistant A. baumannii Isolates Carrying tet(X6)
2.8. Nosocomial Spread of Tigecycline-Resistant A. baumannii Isolates Carrying tet(X6)
3. Discussion
4. Materials and Methods
4.1. Tigecycline-Non-Susceptible A. baumannii Isolate Collection
4.2. PCR Detection of tet(X) Variants
4.3. Bacterial Genome Sequencing and Analysis
4.4. Multilocus Sequence Typing (MLST) Analysis
4.5. Conjugation Assay
4.6. In Silico Analysis of PFGE
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ayobami, O.; Willrich, N.; Harder, T.; Okeke, I.N.; Eckmanns, T.; Markwart, R. The incidence and prevalence of hospital-acquired (carbapenem-resistant) Acinetobacter baumannii in Europe, Eastern Mediterranean and Africa: A systematic review and meta-analysis. Emerg. Microbes Infect. 2019, 8, 1747–1759. [Google Scholar] [CrossRef] [Green Version]
- Mohd Sazlly Lim, S.; Zainal Abidin, A.; Liew, S.M.; Roberts, J.A.; Sime, F.B. The global prevalence of multidrug-resistance among Acinetobacter baumannii causing hospital-acquired and ventilator-associated pneumonia and its associated mortality: A systematic review and meta-analysis. J. Infect. 2019, 79, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Joly-Guillou, M.L. Clinical impact and pathogenicity of Acinetobacter. Clin. Microbiol. Infect. 2005, 11, 868–873. [Google Scholar] [CrossRef] [Green Version]
- Jerassy, Z.; Yinnon, A.M.; Mazouz-Cohen, S.; Benenson, S.; Schlesinger, Y.; Rudensky, B.; Raveh, D. Prospective hospital-wide studies of 505 patients with nosocomial bacteraemia in 1997 and 2002. J. Hosp. Infect. 2006, 62, 230–236. [Google Scholar] [CrossRef]
- Wong, D.; Nielsen, T.B.; Bonomo, R.A.; Pantapalangkoor, P.; Luna, B.; Spellberg, B. Clinical and Pathophysiological Overview of Acinetobacter Infections: A Century of Challenges. Clin. Microbiol. Rev. 2017, 30, 409–447. [Google Scholar] [CrossRef] [Green Version]
- Morris, F.C.; Dexter, C.; Kostoulias, X.; Uddin, M.I.; Peleg, A.Y. The Mechanisms of Disease Caused by Acinetobacter baumannii. Front. Microbiol. 2019, 10, 1601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathlouthi, N.; El Salabi, A.A.; Ben Jomaa-Jemili, M.; Bakour, S.; Al-Bayssari, C.; Zorgani, A.A.; Kraiema, A.; Elahmer, O.; Okdah, L.; Rolain, J.M.; et al. Early detection of metallo-beta-lactamase NDM-1- and OXA-23 carbapenemase-producing Acinetobacter baumannii in Libyan hospitals. Int. J. Antimicrob. Agents 2016, 48, 46–50. [Google Scholar] [CrossRef]
- Munoz-Price, L.S.; Weinstein, R.A. Acinetobacter infection. N. Engl. J. Med. 2008, 358, 1271–1281. [Google Scholar] [CrossRef]
- Su, C.H.; Wang, J.T.; Hsiung, C.A.; Chien, L.J.; Chi, C.L.; Yu, H.T.; Chang, F.Y.; Chang, S.C. Increase of carbapenem-resistant Acinetobacter baumannii infection in acute care hospitals in Taiwan: Association with hospital antimicrobial usage. PLoS ONE 2012, 7, e37788. [Google Scholar] [CrossRef] [Green Version]
- Kempf, M.; Rolain, J.M. Emergence of resistance to carbapenems in Acinetobacter baumannii in Europe: Clinical impact and therapeutic options. Int. J. Antimicrob. Agents 2012, 39, 105–114. [Google Scholar] [CrossRef] [Green Version]
- Bertrand, X.; Dowzicky, M.J. Antimicrobial susceptibility among gram-negative isolates collected from intensive care units in North America, Europe, the Asia-Pacific Rim, Latin America, the Middle East, and Africa between 2004 and 2009 as part of the Tigecycline Evaluation and Surveillance Trial. Clin. Ther. 2012, 34, 124–137. [Google Scholar] [CrossRef]
- Kim, U.J.; Kim, H.K.; An, J.H.; Cho, S.K.; Park, K.H.; Jang, H.C. Update on the Epidemiology, Treatment, and Outcomes of Carbapenem-resistant Acinetobacter infections. Chonnam Med. J. 2014, 50, 37–44. [Google Scholar] [CrossRef] [Green Version]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Trebosc, V.; Gartenmann, S.; Totzl, M.; Lucchini, V.; Schellhorn, B.; Pieren, M.; Lociuro, S.; Gitzinger, M.; Tigges, M.; Bumann, D.; et al. Dissecting Colistin Resistance Mechanisms in Extensively Drug-Resistant Acinetobacter baumannii Clinical Isolates. mBio 2019, 10. [Google Scholar] [CrossRef] [Green Version]
- Cafiso, V.; Stracquadanio, S.; Lo Verde, F.; Gabriele, G.; Mezzatesta, M.L.; Caio, C.; Pigola, G.; Ferro, A.; Stefani, S. Colistin Resistant A. baumannii: Genomic and Transcriptomic Traits Acquired Under Colistin Therapy. Front. Microbiol. 2018, 9, 3195. [Google Scholar] [CrossRef]
- Bakour, S.; Olaitan, A.O.; Ammari, H.; Touati, A.; Saoudi, S.; Saoudi, K.; Rolain, J.M. Emergence of Colistin- and Carbapenem-Resistant Acinetobacter baumannii ST2 Clinical Isolate in Algeria: First Case Report. Microb. Drug Resist. 2015, 21, 279–285. [Google Scholar] [CrossRef]
- Costello, S.E.; Gales, A.C.; Morfin-Otero, R.; Jones, R.N.; Castanheira, M. Mechanisms of Resistance, Clonal Expansion, and Increasing Prevalence of Acinetobacter baumannii Strains Displaying Elevated Tigecycline MIC Values in Latin America. Microb. Drug Resist. 2016, 22, 253–258. [Google Scholar] [CrossRef]
- Noskin, G.A. Tigecycline: A new glycylcycline for treatment of serious infections. Clin. Infect. Dis. 2005, 41 (Suppl. 5), S303–S314. [Google Scholar] [CrossRef]
- Sun, Y.; Cai, Y.; Liu, X.; Bai, N.; Liang, B.; Wang, R. The emergence of clinical resistance to tigecycline. Int. J. Antimicrob. Agents 2013, 41, 110–116. [Google Scholar] [CrossRef]
- Beabout, K.; Hammerstrom, T.G.; Perez, A.M.; Magalhaes, B.F.; Prater, A.G.; Clements, T.P.; Arias, C.A.; Saxer, G.; Shamoo, Y. The ribosomal S10 protein is a general target for decreased tigecycline susceptibility. Antimicrob. Agents Chemother. 2015, 59, 5561–5566. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Hu, D.; Zhang, Q.; Liao, X.P.; Liu, Y.H.; Sun, J. Efflux Pump Overexpression Contributes to Tigecycline Heteroresistance in Salmonella enterica serovar Typhimurium. Front. Cell Infect. Microbiol. 2017, 7, 37. [Google Scholar] [CrossRef]
- Zhong, X.; Xu, H.; Chen, D.; Zhou, H.; Hu, X.; Cheng, G. First emergence of acrAB and oqxAB mediated tigecycline resistance in clinical isolates of Klebsiella pneumoniae pre-dating the use of tigecycline in a Chinese hospital. PLoS ONE 2014, 9, e115185. [Google Scholar] [CrossRef]
- Ruzin, A.; Keeney, D.; Bradford, P.A. AdeABC multidrug efflux pump is associated with decreased susceptibility to tigecycline in Acinetobacter calcoaceticus-Acinetobacter baumannii complex. J. Antimicrob. Chemother. 2007, 59, 1001–1004. [Google Scholar] [CrossRef] [Green Version]
- Linkevicius, M.; Sandegren, L.; Andersson, D.I. Potential of Tetracycline Resistance Proteins To Evolve Tigecycline Resistance. Antimicrob. Agents Chemother. 2016, 60, 789–796. [Google Scholar] [CrossRef] [Green Version]
- Moore, I.F.; Hughes, D.W.; Wright, G.D. Tigecycline is modified by the flavin-dependent monooxygenase TetX. Biochemistry 2005, 44, 11829–11835. [Google Scholar] [CrossRef]
- Speer, B.S.; Salyers, A.A. Characterization of a novel tetracycline resistance that functions only in aerobically grown Escherichia coli. J. Bacteriol. 1988, 170, 1423–1429. [Google Scholar] [CrossRef] [Green Version]
- Park, B.H.; Levy, S.B. The cryptic tetracycline resistance determinant on Tn4400 mediates tetracycline degradation as well as tetracycline efflux. Antimicrob. Agents Chemother. 1988, 32, 1797–1800. [Google Scholar] [CrossRef] [Green Version]
- Zeng, Y.; Dong, N.; Zhang, R.; Liu, C.; Sun, Q.; Lu, J.; Shu, L.; Cheng, Q.; Chan, E.W.; Chen, S. Emergence of an Empedobacter falsenii strain harbouring a tet(X)-variant-bearing novel plasmid conferring resistance to tigecycline. J. Antimicrob. Chemother. 2020, 75, 531–536. [Google Scholar] [CrossRef]
- Whittle, G.; Hund, B.D.; Shoemaker, N.B.; Salyers, A.A. Characterization of the 13-kilobase ermF region of the Bacteroides conjugative transposon CTnDOT. Appl. Environ. Microbiol. 2001, 67, 3488–3495. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Liu, D.; Lv, Y.; Cui, L.; Li, Y.; Li, T.; Song, H.; Hao, Y.; Shen, J.; Wang, Y.; et al. Novel Plasmid-Mediated tet(X5) Gene Conferring Resistance to Tigecycline, Eravacycline, and Omadacycline in a Clinical Acinetobacter baumannii Isolate. Antimicrob. Agents Chemother. 2019, 64. [Google Scholar] [CrossRef]
- Sun, J.; Chen, C.; Cui, C.Y.; Zhang, Y.; Liu, X.; Cui, Z.H.; Ma, X.Y.; Feng, Y.; Fang, L.X.; Lian, X.L.; et al. Plasmid-encoded tet(X) genes that confer high-level tigecycline resistance in Escherichia coli. Nat. Microbiol. 2019, 4, 1457–1464. [Google Scholar] [CrossRef]
- Sun, C.; Cui, M.; Zhang, S.; Wang, H.; Song, L.; Zhang, C.; Zhao, Q.; Liu, D.; Wang, Y.; Shen, J.; et al. Plasmid-mediated tigecycline-resistant gene tet(X4) in Escherichia coli from food-producing animals, China, 2008-2018. Emerg. Microbes Infect. 2019, 8, 1524–1527. [Google Scholar] [CrossRef] [Green Version]
- Speer, B.S.; Bedzyk, L.; Salyers, A.A. Evidence that a novel tetracycline resistance gene found on two Bacteroides transposons encodes an NADP-requiring oxidoreductase. J. Bacteriol. 1991, 173, 176–183. [Google Scholar] [CrossRef] [Green Version]
- Peng, K.; Li, R.; He, T.; Liu, Y.; Wang, Z. Characterization of a porcine Proteus cibarius strain co-harbouring tet(X6) and cfr. J. Antimicrob. Chemother. 2020, 75, 1652–1654. [Google Scholar] [CrossRef]
- Liu, D.; Zhai, W.; Song, H.; Fu, Y.; Schwarz, S.; He, T.; Bai, L.; Wang, Y.; Walsh, T.R.; Shen, J. Identification of the novel tigecycline resistance gene tet(X6) and its variants in Myroides, Acinetobacter and Proteus of food animal origin. J. Antimicrob. Chemother. 2020, 75, 1428–1431. [Google Scholar] [CrossRef]
- Li, R.; Liu, Z.; Peng, K.; Liu, Y.; Xiao, X.; Wang, Z. Co-occurrence of two tet(X) variants in an Empedobacter brevis of shrimp origin. Antimicrob. Agents Chemother. 2019. [Google Scholar] [CrossRef]
- He, T.; Wang, R.; Liu, D.; Walsh, T.R.; Zhang, R.; Lv, Y.; Ke, Y.; Ji, Q.; Wei, R.; Liu, Z.; et al. Emergence of plasmid-mediated high-level tigecycline resistance genes in animals and humans. Nat. Microbiol. 2019, 4, 1450–1456. [Google Scholar] [CrossRef]
- He, D.; Wang, L.; Zhao, S.; Liu, L.; Liu, J.; Hu, G.; Pan, Y. A novel tigecycline resistance gene, tet(X6), on an SXT/R391 integrative and conjugative element in a Proteus genomospecies 6 isolate of retail meat origin. J. Antimicrob. Chemother. 2020, 75, 1159–1164. [Google Scholar] [CrossRef]
- Cui, C.Y.; Chen, C.; Liu, B.T.; He, Q.; Wu, X.T.; Sun, R.Y.; Zhang, Y.; Cui, Z.H.; Guo, W.Y.; Jia, Q.L.; et al. Co-occurrence of Plasmid-Mediated Tigecycline and Carbapenem Resistance in Acinetobacter spp. from Waterfowls and Their Neighboring Environment. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Gasparrini, A.J.; Markley, J.L.; Kumar, H.; Wang, B.; Fang, L.; Irum, S.; Symister, C.T.; Wallace, M.; Burnham, C.D.; Andleeb, S.; et al. Tetracycline-inactivating enzymes from environmental, human commensal, and pathogenic bacteria cause broad-spectrum tetracycline resistance. Commun. Biol. 2020, 3, 241. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, Y.; Liu, Y.; Guo, Y.; Zhou, Y.; Xiao, T.; Zhang, S.; Xu, H.; Chen, Y.; Shan, T.; et al. Identification of novel tetracycline resistance gene tet(X14) and its co-occurrence with tet(X2) in a tigecycline-resistant and colistin-resistant Empedobacter stercoris. Emerg. Microbes Infect. 2020, 9, 1843–1852. [Google Scholar] [CrossRef]
- Leski, T.A.; Bangura, U.; Jimmy, D.H.; Ansumana, R.; Lizewski, S.E.; Stenger, D.A.; Taitt, C.R.; Vora, G.J. Multidrug-resistant tet(X)-containing hospital isolates in Sierra Leone. Int. J. Antimicrob. Agents 2013, 42, 83–86. [Google Scholar] [CrossRef]
- Eitel, Z.; Soki, J.; Urban, E.; Nagy, E.; ESCMID Study Group on Anaerobic Infection. The prevalence of antibiotic resistance genes in Bacteroides fragilis group strains isolated in different European countries. Anaerobe 2013, 21, 43–49. [Google Scholar] [CrossRef]
- Deng, M.; Zhu, M.H.; Li, J.J.; Bi, S.; Sheng, Z.K.; Hu, F.S.; Zhang, J.J.; Chen, W.; Xue, X.W.; Sheng, J.F.; et al. Molecular epidemiology and mechanisms of tigecycline resistance in clinical isolates of Acinetobacter baumannii from a Chinese university hospital. Antimicrob. Agents Chemother. 2014, 58, 297–303. [Google Scholar] [CrossRef] [Green Version]
- Bird, K.; Boopathy, R.; Nathaniel, R.; LaFleur, G. Water pollution and observation of acquired antibiotic resistance in Bayou Lafourche, a major drinking water source in Southeast Louisiana, USA. Environ. Sci. Pollut Res. Int. 2019, 26, 34220–34232. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Z.; Hao, Q.; Wu, J.; Xiao, J.; Jing, H. Complete Genome Sequence of Acinetobacter baumannii ZW85-1. Genome Announc. 2014, 2. [Google Scholar] [CrossRef] [Green Version]
- Rao, J.; Susanti, D.; Childress, J.C.; Mitkos, M.C.; Brima, J.K.; Baffoe-Bonnie, A.W.; Pearce, S.N.; Grgurich, D.; Fernandez-Cotarelo, M.J.; Kerkering, T.M.; et al. Tn2008-driven carbapenem resistance in Acinetobacter baumannii isolates from a period of increased incidence of infections in a Southwest Virginia hospital (USA). J. Glob. Antimicrob. Resist. 2018, 12, 79–87. [Google Scholar] [CrossRef]
- Tada, T.; Miyoshi-Akiyama, T.; Shimada, K.; Shimojima, M.; Kirikae, T. Dissemination of 16S rRNA methylase ArmA-producing Acinetobacter baumannii and emergence of OXA-72 carbapenemase coproducers in Japan. Antimicrob. Agents Chemother. 2014, 58, 2916–2920. [Google Scholar] [CrossRef] [Green Version]
- Chen, F.J.; Huang, W.C.; Liao, Y.C.; Wang, H.Y.; Lai, J.F.; Kuo, S.C.; Lauderdale, T.L.; Sytwu, H.K. Molecular Epidemiology of Emerging Carbapenem Resistance in Acinetobacter nosocomialis and Acinetobacter pittii in Taiwan, 2010 to 2014. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Peng, K.; Li, Y.; Liu, Y.; Wang, Z. Exploring tet(X)-bearing tigecycline-resistant bacteria of swine farming environments. Sci. Total Environ. 2020, 733, 139306. [Google Scholar] [CrossRef]
- Zheng, X.R.; Zhu, J.H.; Zhang, J.; Cai, P.; Sun, Y.H.; Chang, M.X.; Fang, L.X.; Sun, J.; Jiang, H.X. A novel plasmid-borne tet(X6) variant co-existing with blaNDM-1 and blaOXA-58 in a chicken Acinetobacter baumannii isolate. J. Antimicrob. Chemother. 2020, 75, 3397–3399. [Google Scholar] [CrossRef]
- Wyres, K.L.; Cahill, S.M.; Holt, K.E.; Hall, R.M.; Kenyon, J.J. Identification of Acinetobacter baumannii loci for capsular polysaccharide (KL) and lipooligosaccharide outer core (OCL) synthesis in genome assemblies using curated reference databases compatible with Kaptive. Microb. Genom. 2020, 6. [Google Scholar] [CrossRef]
- Zhang, R.; Dong, N.; Shen, Z.; Zeng, Y.; Lu, J.; Liu, C.; Zhou, H.; Hu, Y.; Sun, Q.; Cheng, Q.; et al. Epidemiological and phylogenetic analysis reveals Flavobacteriaceae as potential ancestral source of tigecycline resistance gene tet(X). Nat. Commun. 2020, 11, 4648. [Google Scholar] [CrossRef]
- Karah, N.; Sundsfjord, A.; Towner, K.; Samuelsen, O. Insights into the global molecular epidemiology of carbapenem non-susceptible clones of Acinetobacter baumannii. Drug Resist. Updatess Rev. Comment. Antimicrob. Anticancer. Chemother. 2012, 15, 237–247. [Google Scholar] [CrossRef]
- Adams, M.D.; Wright, M.S.; Karichu, J.K.; Venepally, P.; Fouts, D.E.; Chan, A.P.; Richter, S.S.; Jacobs, M.R.; Bonomo, R.A. Rapid Replacement of Acinetobacter baumannii Strains Accompanied by Changes in Lipooligosaccharide Loci and Resistance Gene Repertoire. mBio 2019, 10. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, Y.C.; Wang, S.H.; Chen, Y.Y.; Lin, T.L.; Shie, S.S.; Huang, C.T.; Lee, C.H.; Chen, Y.C.; Quyen, T.L.T.; Pan, Y.J. Association of capsular types with carbapenem resistance, disease severity, and mortality in Acinetobacter baumannii. Emerg. Microbes Infect. 2020, 9, 2094–2104. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Wu, H.; Wang, Z.Y.; Shen, P.C.; Tian, Y.Q.; Sun, F.; Pan, Z.M.; Jiao, X. Coexistence of blaOXA-58 and tet(X) on a Novel Plasmid in Acinetobacter sp. From Pig in Shanghai, China. Front. Microbiol. 2020, 11, 578020. [Google Scholar] [CrossRef]
- Li, R.; Lu, X.; Peng, K.; Liu, Z.; Li, Y.; Liu, Y.; Xiao, X.; Wang, Z. Deciphering the Structural Diversity and Classification of the Mobile Tigecycline Resistance Gene tet(X)-Bearing Plasmidome among Bacteria. mSystems 2020, 5. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of MICs and Zone Diameters. Version 8.1. 2018. Available online: http://www.eucast.org (accessed on 5 February 2019).
- Diancourt, L.; Passet, V.; Nemec, A.; Dijkshoorn, L.; Brisse, S. The population structure of Acinetobacter baumannii: Expanding multiresistant clones from an ancestral susceptible genetic pool. PLoS ONE 2010, 5, e10034. [Google Scholar] [CrossRef] [Green Version]
- Bartual, S.G.; Seifert, H.; Hippler, C.; Luzon, M.A.; Wisplinghoff, H.; Rodríguez-Valera, F. Development of a multilocus sequence typing scheme for characterization of clinical isolates of Acinetobacter baumannii. J. Clin. Microbiol. 2005, 43, 4382–4390. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.M.; Guo, M.K.; Ke, S.C.; Lin, Y.P.; Li, C.R.; Vy Nguyen, H.T.; Wu, L.T. Emergence and nosocomial spread of ST11 carbapenem-resistant Klebsiella pneumoniae co-producing OXA-48 and KPC-2 in a regional hospital in Taiwan. J. Med. Microbiol. 2018, 67, 957–964. [Google Scholar] [CrossRef]
- Vy Nguyen, H.T. Characteristics of Clinical Carbapenem—Resistant Enterobacteriaceae Isolates in Central Taiwan. Master’s Thesis, China Medical University Taichung, Taichung, Taiwan, 2019. [Google Scholar]
- Vijayashree Priyadharsini, J.; Smiline Girija, A.S.; Paramasivam, A. In silico analysis of virulence genes in an emerging dental pathogen A. baumannii and related species. Arch. Oral Biol. 2018, 94, 93–98. [Google Scholar] [CrossRef]
- Murtagh, F.; Legendre, P. Ward’s Hierarchical Agglomerative Clustering Method: Which Algorithms Implement Ward’s Criterion? J. Classif. 2014, 31, 274–295. [Google Scholar] [CrossRef] [Green Version]
- Dice, L.R. Measures of the amount of ecologic association between species. Ecology 1945, 26, 297–302. [Google Scholar] [CrossRef]


| Strain Name | Bacteria Species | Description | Carbapenem/Tigecycline Resistance Genes | MIC (mg/L) | |
|---|---|---|---|---|---|
| IMP | TIG | ||||
| J53 | E. coli | Recipient | - | 0.125 (S) | 0.125 (S) |
| 17CRE24 | K. pneumoniae | Donor | blaOXA-48 | >16 (R) | ND |
| J53-blaOXA-48 | E. coli | No. 1 transconjugant | blaOXA-48 | 8 (R) | ND |
| E. coli | No. 2 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 3 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 4 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 5 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 6 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 7 transconjugant | blaOXA-48 | >8 (R) | ND | |
| E. coli | No. 8 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 9 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 10 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 11 transconjugant | blaOXA-48 | 8 (R) | ND | |
| E. coli | No. 12 transconjugant | blaOXA-48 | 8 (R) | ND | |
| X4-65 | A. baumannii | Donor | tet(X6) | ND | 8 (R) |
| X4-107 | A. baumannii | Donor | tet(X6) | ND | 8 (R) |
| X4-136 | A. baumannii | Donor | tet(X6) | ND | 8 (R) |
| X4-201 | A. baumannii | Donor | tet(X6) | ND | 8 (R) |
| X4-300 | A. baumannii | Donor | tet(X6) | ND | 8 (R) |
| X4-584 | A. baumannii | Donor | tet(X6) | ND | 8 (R) |
| X4-705 | A. baumannii | Donor | tet(X6) | ND | 4 (R) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsieh, Y.-C.; Wu, J.-W.; Chen, Y.-Y.; Quyen, T.L.T.; Liao, W.-C.; Li, S.-W.; Chen, Y.-C.; Pan, Y.-J. An Outbreak of tet(X6)-Carrying Tigecycline-Resistant Acinetobacter baumannii Isolates with a New Capsular Type at a Hospital in Taiwan. Antibiotics 2021, 10, 1239. https://doi.org/10.3390/antibiotics10101239
Hsieh Y-C, Wu J-W, Chen Y-Y, Quyen TLT, Liao W-C, Li S-W, Chen Y-C, Pan Y-J. An Outbreak of tet(X6)-Carrying Tigecycline-Resistant Acinetobacter baumannii Isolates with a New Capsular Type at a Hospital in Taiwan. Antibiotics. 2021; 10(10):1239. https://doi.org/10.3390/antibiotics10101239
Chicago/Turabian StyleHsieh, Yu-Chia, Jia-Wen Wu, Yi-Yin Chen, Tran Lam Tu Quyen, Wei-Chao Liao, Shiao-Wen Li, Yin-Cheng Chen, and Yi-Jiun Pan. 2021. "An Outbreak of tet(X6)-Carrying Tigecycline-Resistant Acinetobacter baumannii Isolates with a New Capsular Type at a Hospital in Taiwan" Antibiotics 10, no. 10: 1239. https://doi.org/10.3390/antibiotics10101239
APA StyleHsieh, Y.-C., Wu, J.-W., Chen, Y.-Y., Quyen, T. L. T., Liao, W.-C., Li, S.-W., Chen, Y.-C., & Pan, Y.-J. (2021). An Outbreak of tet(X6)-Carrying Tigecycline-Resistant Acinetobacter baumannii Isolates with a New Capsular Type at a Hospital in Taiwan. Antibiotics, 10(10), 1239. https://doi.org/10.3390/antibiotics10101239






