It’s Not Easy Being Green: A Narrative Review on the Microbiology, Virulence and Therapeutic Prospects of Multidrug-Resistant Pseudomonas aeruginosa
Abstract
1. Introduction, Taxonomy, and Microbiology of Pseudomonas aeruginosa
1.1. General Concepts
1.2. Taxonomy and Phenotypic Characteristics of P. aeruginosa
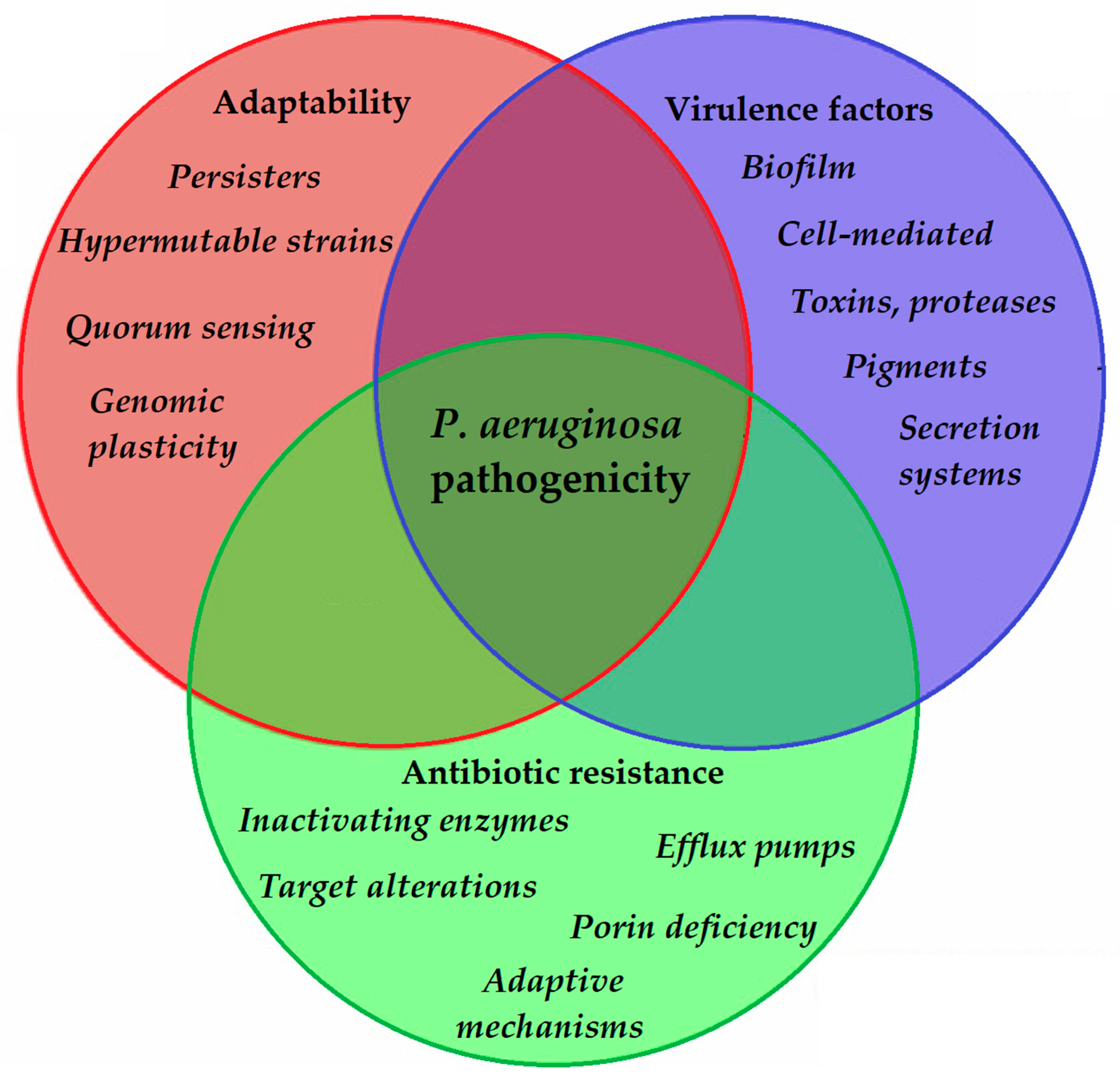
2. Virulence Determinants of P. aeruginosa and Modulation of Virulence Factor Expression
2.1. Genome of P. aeruginosa
2.2. Virulence Factors of P. aeruginosa
2.3. Typing Methods for the Differentiation of P. aeruginosa Clones, Global Dissemination
2.4. Biofilm Formation
2.5. Quorum Sensing (QS)-Mediated Control of Virulence Factor Expression in P. aeruginosa
2.6. Clinical Relevance of P. aeruginosa
3. Antibiotic Resistance in P. aeruginosa: Therapeutic Alternatives
3.1. General Concepts Related to MDR Pathogens
3.2. Intrinsic Resistance and Main Therapeutic Alternatives in P. aeruginosa Infections
3.3. Main Mechanisms of Resistance in P. aeruginosa to Antibiotics Other Than β-Lactams
3.4. Main Mechanisms of β-Lactam-Resistance in P. aeruginosa
3.5. Carbapenem-Resistant but Cephalosporin-Susceptible P. aeruginosa (Car-R Ceph-S)
4. Emerging Therapeutic Options for Pseudomonas Infections
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sobel, J.D.; Kaye, D. Urinary Tract Infections. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 8th ed.; Bennett, J.E., Dolin, R., Blaser, M.J., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; pp. 886–913. [Google Scholar]
- Tohamy, S.T.; Aboshanab, K.M.; El-Mahallawy, H.A.; El-Ansary, M.R.; Afifi, S.S. Prevalence of Multidrug-Resistant Gram-Negative Pathogens Isolated from Febrile Neutropenic Cancer Patients with Bloodstream Infections in Egypt and New Synergistic Antibiotic Combinations. Infect. Drug Res. 2018, 11, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Chawla, K.; Vishwanath, S.; Munim, F.C. Nonfermenting Gram-negative Bacilli other than Pseudomonas aeruginosa and Acinetobacter Spp. Causing Respiratory Tract Infections in a Tertiary Care Center. J. Glob. Infect. Dis. 2013, 5, 144–148. [Google Scholar]
- Smith, S.M.; Cundy, K.R.; Gilardi, G.L.; Wong, W. Evaluation of the AutoMicrobic System for Identification of Glucose-Nonfermenting Gram-Negative Rods. J. Clin. Microbiol. 1982, 15, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Whistler, T.; Sangwichian, O.; Jorakate, P.; Sawatwong, P.; Surin, U.; Piralam, B.; Thamthitiwat, S.; Promkong, C.; Peruski, L. Identification of Gram negative non-fermentative bacteria: How hard can it be? PLoS Neglect. Pathog. 2019, 13, e0007729. [Google Scholar] [CrossRef] [PubMed]
- Malini, A.; Deepa, E.K.; Gokul, B.N.; Prasad, S.R. Nonfermenting Gram-Negative Bacilli Infections in a Tertiary Care Hospital in Kolar, Karnataka. J. Lab. Physicians 2009, 1, 62–66. [Google Scholar] [CrossRef]
- Gilardi, G.L. Pseudomonas species in clinical microbiology. Mt. Sinai J. Med. 1976, 43, 710–726. [Google Scholar] [PubMed]
- Palleroni, N.J. Pseudomonas classification. A new case history in the taxonomy of gram-negative bacteria. Antonie Van Leeuwenhoek 1993, 64, 231–351. [Google Scholar] [CrossRef] [PubMed]
- Palleroni, N.J. The Pseudomonas Story. Environ. Microbiol. 2010, 12, 1377–1383. [Google Scholar] [CrossRef]
- Gumey, J.; Pradier, L.; Griffin, J.S.; Gougat-Barbera, C.; Chan, B.K.; Turner, P.E.; Kaltz, O.; Hochberg, M.E. Phage steering of antibiotic-resistance evolution in the bacterial pathogen, Pseudomonas aeruginosa. Evol. Med. Pub. Health 2020, 1, 148–157. [Google Scholar]
- Blanc, D.S.; Francioli, P.; Zanetti, G. Molecular Epidemiology of Pseudomonas aeruginosa in the Intensive Care Units—A Review. Open Microbiol. J. 2007, 1, 8–11. [Google Scholar] [CrossRef]
- Klockgether, J.; Tömmler, B. Recent advances in understanding Pseudomonas aeruginosa as a pathogen. F100Research 2017, 6, e1261. [Google Scholar] [CrossRef] [PubMed]
- Maenni, M.; Bour, M.; Chatre, P.; Madec, J.Y.; Plésiat, P.; Jeannot, K. Resistance of Animal Strains of Pseudomonas aeruginosa to Carbapenems. Front. Microbiol. 2017, 8, e1841. [Google Scholar]
- Algammal, A.M.; Mabrok, M.; Sivaramasamy, E.; Youssef, F.M.; Atwa, M.H.; El-Kholy, A.W.; Hetta, H.F.; Hozzein, W.N. Emerging MDR-Pseudomonas aeruginosa in fish commonly harbor oprL and toxA virulence genes and blaTEM, blaCTX-M, and tetA antibiotic-resistance genes. Sci. Rep. 2020, 10, e15961. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Roldán, L.; Rojo-Bezares, B.; de Toro, M.; López, M.; Toledano, P.; Lozano, C.; Chichón, G.; Alvarez-Erviti, L.; Torres, C.; Sáenz, Y. Antimicrobial resistance and virulence of Pseudomonas spp. among healthy animals: Concern about exolysin ExlA detection. Sci. Rep. 2020, 10, e11667. [Google Scholar]
- Fernández, M.; Porcel, M.; de la Torre, J.; Molina-Henares, M.A.; Daddaoua, A.; Llamas, M.A.; Roca, A.; Carriel, V.; Garzón, I.; Ramos, J.L.; et al. Analysis of the pathogenic potential of nosocomial Pseudomonas putida strains. Front. Microbiol. 2015, 6, e871. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Barranquero, J.A.; Cazorla, F.M.; de Vincente, A. Pseudomonas syringae pv. syringae Associated with Mango Trees, a Particular Pathogen Within the “Hodgepodge” of the Pseudomonas syringae Complex. Front. Plant Sci. 2019, 10, e570. [Google Scholar]
- Shariff, A.M.; Beri, K. Exacerbation of bronchiectasis by Pseudomonas monteilii: A case report. BMC Infect. Dis. 2017, 17, e511. [Google Scholar]
- Brown, V.I.; Lowbury, E.J.L. Use of an improved cetrimide agar medium and other culture methods for Pseudomonas aeruginosa. J. Clin. Pathol. 1965, 18, 752–756. [Google Scholar] [CrossRef]
- Gajdács, M.; Burián, K.; Terhes, G. Resistance Levels and Epidemiology of Non-Fermenting Gram-Negative Bacteria in Urinary Tract Infections of Inpatients and Outpatients (RENFUTI): A 10-Year Epidemiological Snapshot. Antibiotics 2019, 8, 143. [Google Scholar] [CrossRef]
- Clark, S.T.; Caballero, J.D.; Cheang, M.; Coburn, B.; Wang, P.W.; Donaldson, S.L.; Zhang, Y.; Liu, M.; Keshavjee, S.; Yau, Y.C.W.; et al. Phenotypic diversity within a Pseudomonas aeruginosa population infecting an adult with cystic fibrosis. Sci. Rep. 2015, 5, 10932. [Google Scholar] [CrossRef]
- Behzadi, P.; Behzadi, E. The microbial agents of urinary tract infections at central laboratory of Dr. Shariati Hospital, Tehran, Iran. Turk. Klin. Tip Bilim. 2008, 28, e445. [Google Scholar]
- Hall, S.; McDermott, C.; Anoopkumar-Dukie, S.; McFarland, A.J.; Forbes, A.; Perkis, A.V.; Davey, A.K.; Chess-Williams, R.; Kiefel, M.J.; Arora, D.; et al. Cellular Effects of Pyocyanin, a Secreted Virulence Factor of Pseudomonas aeruginosa. Toxins 2016, 8, 236. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.M.; Neely, A.; Stintzi, A.; Georges, C.; Holder, A.I. Pyoverdin is essential for virulence of Pseudomonas aeruginosa. Infect. Immun. 1996, 64, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Alyydice-Francis, K.; Brown, P.D. Diversity of Antimicrobial Resistance and Virulence Determinants in Pseudomonas aeruginosa Associated with Fresh Vegetables. Int. J. Microbiol. 2012, 2012, e426241. [Google Scholar]
- El-Fouly, M.Z.; Sharaf, A.M.; Shahin, A.A.M.; El-Bialy, H.A.; Omara, A.M.A. Biosynthesis of pyocyanin pigment by Pseudomonas aeruginosa. J. Rad. Res. Appl. Sci. 2015, 8, 36–48. [Google Scholar] [CrossRef]
- Behzadi, P.; Behzadi, E. A study on apoptosis inducing effects of UVB irradiation in Pseudomonas aeruginosa. Roum. Arch. Microbiol. Immunol. 2011, 70, 74–77. [Google Scholar]
- Azuama, O.C.; Ortiz, S.; Quirós-Guerrero, L.; Bouffartigues, E.; Tortuel, D.; Maillot, O.; Feuilloley, M.; Cornelis, P.; Lesouhaitier, O.; Grougnet, R.; et al. Tackling Pseudomonas aeruginosa Virulence by Mulinane-Like Diterpenoids from Azorella atacamensis. Biomolecules 2020, 10, 1626. [Google Scholar] [CrossRef]
- Shaw, E.; Wuest, W.M. Virulence attenuating combination therapy: A potential multi-target synergy approach to treat Pseudomonas aeruginosa infections in cystic fibrosis patients. RSC Med. Chem. 2020, 11, e358. [Google Scholar] [CrossRef]
- Silby, M.W.; Winstanley, C.; Godfrey, S.A.C.; Levy, S.B.; Jackson, R.W. Pseudomonas genomes: Diverse and adaptable. FEMS Microbiol. Rev. 2011, 35, 652–680. [Google Scholar] [CrossRef]
- Vodovar, N.; Vallenet, D.; Cruveiller, S.; Rouy, Z.; Barbe, V.; Acosta, C.; Cattolico, L.; Jubin, C.; Lajus, A.; Segurens, B.; et al. Complete genome sequence of the entomopathogenic and metabolically versatile soil bacterium Pseudomonas entomophila. Nat. Biotechnol. 2006, 24, 673–679. [Google Scholar] [CrossRef]
- Stover, C.K.; Pham, X.Q.; Erwin, A.L.; Mizoguchi, S.D.; Warrener, P.; Hickey, M.J.; Brinkman, F.S.L.; Hufnagle, W.O.; Lagrou, M.; Garber, R.L.; et al. Complete genome sequence of Pseudomonas aeruginosa PAO1, an opportunistic pathogen. Nature 2006, 406, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Hozzari, A.; Behzadi, P.; Khiabani, P.K.; Sholeh, M.; Sabokroo, N. Clinical cases, drug resistance, and virulence genes profiling in Uropathogenic Escherichia coli. J. Appl Gen. 2020, 61, 265–273. [Google Scholar] [CrossRef]
- Issakhanian, L.; Behzadi, P. Antimicrobial agents and urinary tract infections. Curr. Pharm. Des. 2019, 25, 1409–1423. [Google Scholar] [CrossRef] [PubMed]
- de Lorenzo, V. Pseudomonas aeruginosa: The making of a pathogen. Environ. Microbiol. 2015, 17, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Suenaga, H.; Fujihara, H.; Kimura, N.; Hirose, J.; Watanabe, T.; Futagami, T.; Goto, M.; Shimodaira, J.; Furukawa, K. Insights into the genomic plasticity of Pseudomonas putida KF715, a strain with unique biphenyl-utilizing activity and genome instability properties. Environ. Microbiol. Rep. 2017, 9, 589–598. [Google Scholar] [CrossRef]
- Rumbaugh, K.P. Genomic complexity and plasticity ensurePseudomonas success. FEMS Microbiol. Lett. 2014, 356, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Brüggemann, H.; Migliorini, L.B.; de Sales, R.O.; Koga, P.C.M.; de Sousa, A.V.; Jensen, A.; Poehlein, A.; Brzuszkiewicz, E.; Doi, A.M.; Pasternak, J.; et al. Comparative Genomics of Nonoutbreak Pseudomonas aeruginosa Strains Underlines Genome Plasticity and Geographic Relatedness of the Global Clone ST235. Genome Biol. Evol. 2018, 10, 1852–1857. [Google Scholar] [CrossRef]
- Bao, Z.; Stodghill, P.V.; Myers, C.R.; Lam, H.; Wei, H.L.; Charavarthy, S.; Kvitko, B.H.; Collmer, A.; Cartinhour, S.W.; Schweitzer, P.; et al. Genomic Plasticity Enables Phenotypic Variation of Pseudomonas syringae pv. tomato DC3000. PLoS ONE 2014, 9, e8662. [Google Scholar] [CrossRef]
- Jansen, G.; Mahrt, N.; Tueffers, L.; Barbosa, C.; Harjes, M.; Adolph, G.; Friedrichs, A.; Krenz-Weinreich, A.; Rosenstiel, P.; Schulenburg, H. Association between clinical antibiotic resistance and susceptibility of Pseudomonas in the cystic fibrosis lung. Evol. Med. Pub. Health 2016, 1, 182–194. [Google Scholar] [CrossRef]
- Liao, C.H.; McCallus, D.E. Biochemical and Genetic Characterization of an Extracellular Protease from Pseudomonas fluorescens CY091. Appl. Environ. Microbiol. 1988, 64, 914–921. [Google Scholar] [CrossRef]
- Bonomo, R.A.; Szabó, D. Mechanisms of Multidrug Resistance in Acinetobacter Species and Pseudomonas aeruginosa. Clin. Infect. Dis. 2006, 43, S49–S56. [Google Scholar] [CrossRef] [PubMed]
- Hogardt, M.; Heesemann, J. Microevolution of Pseudomonas aeruginosa to a chronic pathogen of the cystic fibrosis lung. Curr. Top. Microbiol. Immunol. 2013, 358, 91–118. [Google Scholar] [PubMed]
- Rees, V.E.; Lucas, D.S.D.; López-Causapé, C.; Huang, Y.; Kotsimbos, T.; Bulitta, J.B.; Rees, M.C.; Barugahare, A.; Peleg, A.Y.; Nation, R.L.; et al. Characterization of Hypermutator Pseudomonas aeruginosa Isolates from Patients with Cystic Fibrosis in Australia. Antmicrob. Agents Chemother. 2019, 63, e02538-18. [Google Scholar] [CrossRef]
- Ciofu, O.; Fussing, V.; Bagge, N.; Koch, C.; Hřiby, N. Characterization of paired mucoid/non-mucoid Pseudomonas aeruginosa isolates from Danish cystic fibrosis patients: Antibiotic resistance, beta-lactamase activity and RiboPrinting. J. Antimicrob. Chemother. 2001, 48, 391–396. [Google Scholar] [CrossRef]
- Lee, B.; Schjerling, C.K.; Kirkby, N.; Hoffmann, N.; Borup, R.; Molin, S.; Hoiby, N.; Coifu, O. Mucoid Pseudomonas aeruginosa isolates maintain the biofilm formation capacity and the gene expression profiles during the chronic lung infection of CF patients. APMIS 2011, 119, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Behrens, A.J.; Kaever, V.; Kazmierczak, B.I. Type IV Pilus Assembly in Pseudomonas aeruginosa over a Broad Range of Cyclic di-GMP Concentrations. J. Bacteriol. 2012, 194, 4285–4294. [Google Scholar] [CrossRef]
- Lam, J.S.; Taylor, V.L.; Islam, S.T.; Hao, Y.; Kocincová, D. Genetic and functional diversity of Pseudomonas aeruginosa lipopolysaccharide. Front. Microbiol. 2011, 2, e118. [Google Scholar] [CrossRef]
- Pan, X.; Cui, X.; Zhang, F.; He, Y.; Li, L.; Yang, H. Genetic Evidence for O-Specific Antigen as Receptor of Pseudomonas aeruginosa Phage K8 and Its Genomic Analysis. Front. Microbiol. 2016, 7, e252. [Google Scholar] [CrossRef]
- Kazmierczak, B.I.; Schnierderberend, M.; Jain, R. Cross-regulation of Pseudomonas motility systems: The intimate relationship between flagella, pili and virulence. Curr. Opin. Microbiol. 2015, 28, 78–82. [Google Scholar] [CrossRef]
- Chemani, C.; Imberty, A.; de Bentzmann, S.; Pierre, M.; Wimmerová, M.; Guery, B.P.; Faure, K. Role of LecA and LecB Lectins in Pseudomonas aeruginosa-Induced Lung Injury and Effect of Carbohydrate Ligands. Infect. Immun. 2009, 77, 2065–2075. [Google Scholar] [CrossRef]
- Thuenauer, R.; Landi, A.; Trefzer, A.; Altmann, S.; Wehrum, S.; Elerhoff, T.; Diedrich, B.; Dengjel, J.; Nyström, A.; Imberty, A.; et al. The Pseudomonas aeruginosa Lectin LecB Causes Integrin Internalization and Inhibits Epithelial Wound Healing. mBio 2020, 11, e03260-19. [Google Scholar] [CrossRef] [PubMed]
- Gilboa-Garber, N.; Avichezer, D. Effects of Pseudomonas aeruginosa PA-I and PA-II Lectins on Tumoral Cells. In Lectins and Glycobiology; Gabius, H.J., Gabius, S., Eds.; Springer: Berlin, Germany, 1993. [Google Scholar]
- Szabó, D.; Szentandrássy, J.; Juhász, Z.; Katona, K.; Nagy, K.; Rókusz, L. Imported PER-1 producing Pseudomonas aeruginosa, PER-1 producing Acinetobacter baumannii and VIM-2 producing Pseudomonas aeruginosa strains in Hungary. Ann. Clin. Microbiol. Antimicrob. 2008, 7, e12. [Google Scholar] [CrossRef] [PubMed]
- Vanderwoude, J.; Fleming, D.; Azimi, S.; Trivedi, U.; Rumbaugh, K.R.; Diggle, S.P. The evolution of virulence in Pseudomonas aeruginosa during chronic wound infection. Proc. R. Soc. B 2020, 287, e20202272. [Google Scholar] [CrossRef] [PubMed]
- Veesenmeyer, J.L.; Hauser, A.R.; Lisboa, T.; Rello, J. Pseudomonas aeruginosa virulence and therapy: Evolving translational strategies. Crit. Care Med. 2009, 37, 1777–1786. [Google Scholar] [CrossRef] [PubMed]
- Amsalu, A.; Sapula, S.A.; Lopes, M.D.B.; Hart, B.J.; Nguyen, A.H.; Drigo, B.; Turnidge, J.; Leong, L.E.; Venter, H. Eux Pump-Driven Antibiotic and Biocide Cross-Resistance in Pseudomonas aeruginosa Isolated from Dierent Ecological Niches: A Case Study in the Development of Multidrug Resistance in Environmental Hotspots. Microorganisms 2020, 8, 1647. [Google Scholar] [CrossRef]
- Crousilles, A.; Maunders, E.; Bartlett, S.; Fan, C.; Emem-Fong, U.; Abdelhamid, Y.; Baker, Y.; Floto, A.; Spring, D.R.; Welch, M. Which microbial factors really are important in Pseudomonas aeruginosa infections? Future Microbiol. 2015, 10, 1825–1836. [Google Scholar] [CrossRef]
- Cabot, G.; Ocampo-Sosa, A.A.; Domínguez, M.A.; Gago, J.F.; Juan, C.; Tubau, F.; Rodriguez, C.; Moyá, C.; Martínez-Martínez, L.; Oliver, A. Spanish Network for Research in Infectious Diseases (REIPI). Genetic markers of widespread extensively drug-resistant Pseudomonas aeruginosa high-risk clones. Antimicrob. Agents Chemother. 2012, 56, 6349–6357. [Google Scholar] [CrossRef]
- Manos, J.; Hu, H.; Rose, B.R.; Wainwright, C.E.; Zablotska, I.B.; Cheney, J.; Turnbull, L.; Whitchurch, C.B.; Grimwood, K.; Harmer, C.; et al. Virulence factor expression patterns in Pseudomonas aeruginosa strains from infants with cystic fibrosis. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1583–1592. [Google Scholar] [CrossRef]
- Moscoso, J.A.; Mikkelsen, H.; Heeb, S.; Williams, P.; Filloux, A. The Pseudomonas aeruginosa sensor RetS switches type III and type VI secretion via c-di-GMP signalling. Environ. Microbiol. 2011, 13, 3128–3138. [Google Scholar] [CrossRef]
- Chung, J.C.S.; Rzhepishevska, O.; Ramstedt, M.; Welch, M. Type III secretion system expression in oxygen-limited Pseudomonas aeruginosa cultures is stimulated by isocitrate lyase activity. Open Biol. 2013, 3, e120131. [Google Scholar] [CrossRef]
- Yahr, T.L.; Wolfgang, M.C. Transcriptional regulation of the Pseudomonas aeruginosa type III secretion system. Mol. Microbiol. 2006, 62, 631–640. [Google Scholar] [CrossRef]
- Waters, V.; Zlosnik, J.E.A.; Yau, Y.C.W.; Speert, D.P.; Aaron, S.D.; Guttman, D.S. Comparison of three typing methods for Pseudomonas aeruginosa isolates from patients with cystic fibrosis. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 3341–3350. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, B.; Valot, B.; Abdelbary, M.H.H.; Prod’hom, G.; Greub, G.; Senn, L.; Blanc, D.S. Combining Standard Molecular Typing and Whole Genome Sequencing to Investigate Pseudomonas aeruginosa Epidemiology in Intensive Care Units. Front. Public Health 2020, 8, e3. [Google Scholar] [CrossRef] [PubMed]
- Hernandes, J.; Ferrus, M.A.; Hernandez, M.; Alonso, J.L. Comparison of six different methods for typing Pseudomonas aeruginosa strains isolated from bottled and well waters. Water Res. 1997, 31, 3169–3174. [Google Scholar] [CrossRef]
- Ballarini, A.; Scarlet, G.; Kos, M.; Cramer, N.; Wiehlmann, L.; Jousson, O. Molecular typing and epidemiological investigation of clinical populations of Pseudomonas aeruginosa using an oligonucleotide-microarray. BMC Microbiol. 2012, 12, e152. [Google Scholar] [CrossRef] [PubMed]
- MacCannell, D. Bacterial strain typing. Clin. Lab. Med. 2013, 33, 629–650. [Google Scholar] [CrossRef] [PubMed]
- Oliver, A.; Mulet, X.; López-Causapé, C.; Juan, C. The increasing threat of Pseudomonas aeruginosa high-risk clones. Drug Res. Update 2015, 21, 41–59. [Google Scholar] [CrossRef]
- Treepong, P.; Kos, V.N.; Goyeux, C.; Blanc, D.S.; Bertand, X.; Valot, B.; Hocquet, D. Global emergence of the widespread Pseudomonas aeruginosa ST235 clone. Clin. Microbiol. Infect. 2018, 24, 258–266. [Google Scholar] [CrossRef]
- Wi, Y.M.; Choi, J.Y.; Lee, J.Y.; Kang, C.I.; Chung, D.R.; Peck, K.R.; Song, J.H.; Ko, K.S. Emergence of colistin resistance in Pseudomonas aeruginosa ST235 clone in South Korea. Int. J. Antimicrob. Agents 2017, 49, 767–769. [Google Scholar] [CrossRef]
- Reboud, E.; Basso, P.; Maillard, A.P.; Huber, P.; Attrée, I. Exolysin Shapes the Virulence of Pseudomonas aeruginosa Clonal Outliers. Toxins 2017, 9, 364. [Google Scholar] [CrossRef]
- Khan, M.; Stapleton, F.; Summers, S.; Rice, S.A.; Willcox, M.D.P. Antibiotic Resistance Characteristics of Pseudomonas aeruginosa Isolated from Keratitis in Australia and India. Antibiotics 2020, 9, 600. [Google Scholar] [CrossRef] [PubMed]
- Subedi, D.; Vijay, A.K.; Kohli, G.S.; Rice, S.A.; Willcox, M. Association between possession of ExoU and antibiotic resistance in Pseudomonas aeruginosa. PLoS ONE 2018, 13, e0204936. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Delcaru, C.; Alexandru, I.; Podgoreanu, P.; Grosu, M.; Stavropoulos, E.; Chifiriuc, M.C.; Lazăr, V. Microbial Biofilms in Urinary Tract Infections and Prostatitis: Etiology, Pathogenicity, and Combating strategies. Pathogens 2016, 5, 65. [Google Scholar] [CrossRef] [PubMed]
- Maurice, N.M.; Bedi, B.; Sadikot, R.T. Pseudomonas aeruginosa Biofilms: Host Response and Clinical Implications in Lung Infections. Am. J. Respir. Cell Mol. Biol. 2018, 58, 428–439. [Google Scholar] [CrossRef]
- Behzadi, P.; Urbán, E.; Gajdács, M. Association between Biofilm-Production and Antibiotic Resistance in Uropathogenic Escherichia coli (UPEC): An In Vitro Study. Diseases 2020, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Lebeaux, D.; Ghigo, J.-M.; Beloin, C. Biofilm-Related Infections: Bridging the Gap between Clinical Management and Fundamental Aspects of Recalcitrance toward Antibiotics. Microbiol. Mol. Boil. Rev. 2014, 78, 510–543. [Google Scholar] [CrossRef]
- Gajdács, M.; Urbán, E. Epidemiological Trends and Resistance Associated with Stenotrophomonas maltophilia Bacteremia: A 10-Year Retrospective Cohort Study in a Tertiary-Care Hospital in Hungary. Diseases 2019, 7, 41. [Google Scholar] [CrossRef]
- Mulchany, L.R.; Isabella, V.M.; Lewis, K. Pseudomonas aeruginosa biofilms in disease. Microb. Ecol. 2014, 68, 1–12. [Google Scholar]
- Ciszek-Lenda, M.; Strus, M.; Walczewska, M.; Majka, G.; Machul-Żwirbla, A.; Mikolajczyk, D.; Górska, S.; Gamian, A.; Chain, B.; Marcinkiewicz, J. Pseudomonas aeruginosa biofilm is a potent inducer of phagocyte hyperinflammation. Inflamm. Res. 2019, 68, 397–413. [Google Scholar] [CrossRef]
- Ciofu, O.; Tolker-Nielsen, T. Tolerance and Resistance of Pseudomonas aeruginosa Biofilms to Antimicrobial Agents—How P. aeruginosa Can Escape Antibiotics. Front. Microbiol. 2019, 10, e913. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wubbolts, R.W.; Haagsman, H.P.; Weldhuizen, E.J.A. Inhibition and Eradication of Pseudomonas aeruginosa Biofilms by Host Defence Peptides. Sci. Rep. 2018, 8, e10446. [Google Scholar] [CrossRef] [PubMed]
- Barrak, I.; Baráth, Z.; Tián, T.; Venkei, A.; Gajdács, M.; Urbán, E.; Stájer, A. Effects of different decontaminating solutions used for the treatment of peri-implantitis on the growth of Porphyromonas gingivalis—An in vitro study. Acta Microbiol. Immunol. Hung. 2020. [Google Scholar] [CrossRef] [PubMed]
- Stájer, A.; Barrak, I.; Gajdács, M.; Urbán, E.; Baráth, Z. Diagnosis and Management of Cervicofacial Actinomycosis: Lessons from Two Distinct Clinical Cases. Antibiotics 2020, 9, 139. [Google Scholar] [CrossRef] [PubMed]
- Pye, C. Pseudomonas otitis externa in dogs. Can. Vet. J. 2018, 59, 1231–1234. [Google Scholar] [PubMed]
- Martínez-Alemán, S.; Bustamante, A.E.; Jimenez-Valdes, R.J.; González, G.M.; Sánchez-González, A. Pseudomonas aeruginosa isolates from cystic fibrosis patients induce neutrophil extracellular traps with different morphologies that could correlate with their disease severity. Int. J. Med. Microbiol. 2020, 310, e151451. [Google Scholar] [CrossRef]
- Schinner, S.; Engelhardt, F.; Preusse, M.; Thöming, J.G.; Tomasch, J.; Haussler, S. Genetic determinants of Pseudomonas aeruginosa fitness during biofilm growth. Biofilm 2020, 1, e100023. [Google Scholar] [CrossRef]
- Hoiby, N.; Ciofu, O.; Bjarnsholt, T. Pseudomonas aeruginosa biofilms in cystic fibrosis. Future Microbiol. 2010, 5, 1663–1674. [Google Scholar] [CrossRef]
- Trunk, K.; Benkert, B.; Quack, N.; Münch, R.; Scheer, M.; Garbe, J.; Jansch, L.; Trost, M.; Wehland, J.; Buer, J.; et al. Anaerobic adaptation in Pseudomonas aeruginosa: Definition of the Anr and Dnr regulons. Environ. Microbiol. 2010, 12, 1719–1733. [Google Scholar] [CrossRef]
- Grassi, L.; Di Luca, M.; Maisetta, G.; Rinaldi, A.C.; Esin, S.; Trampuz, A.; Batoni, G. Generation of Persister Cells of Pseudomonas aeruginosa and Staphylococcus aureus by Chemical Treatment and Evaluation of Their Susceptibility to Membrane-Targeting Agents. Front. Microbiol. 2017, 8, e1917. [Google Scholar] [CrossRef]
- Balogh, E.P.; Faludi, I.; Virók, D.P.; Endrész, V.; Burián, K. Chlamydophila pneumoniae induces production of the defensin-like MIG/CXCL9, which has in vitro antichlamydial activity. Int. J. Med. Microbiol. 2011, 301, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Burián, K.; Berencsi, K.; Endrész, V.; Gyulai, Z.; Vályi-Nagy, T.; Bakay, M.; Geng, Y.; Virók, D.; Kari, L.; Hajnal-Papp, R.; et al. Chlamydia pneumoniae Exacerbates Aortic Inflammatory Foci Caused by Murine Cytomegalovirus Infection in Normocholesterolemic Mice. Clin. Vaccine Immunol. 2001, 8, 1263–1266. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Möker, N.; Dean, C.R.; Tao, J. Pseudomonas aeruginosa Increases Formation of Multidrug-TolerantPersister Cells in Response to Quorum-Sensing Signaling Molecule. J. Bacteriol. 2010, 192, 1946–1955. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Long, Y.; Liu, Y.; Liu, Y.; Chen, R.; Zhang, L.; Jin, Y.; Yang, L.; Bai, F.; Jin, S.; et al. HigB of Pseudomonas aeruginosa Enhances Killing of Phagocytes by Up-Regulating the Type III Secretion System in Ciprofloxacin Induced Persister Cells. Front. Cell. Infect. Microbiol. 2016, 6, e125. [Google Scholar] [CrossRef] [PubMed]
- Gnanadhas, D.P.; Elango, M.; Datey, A.; Chakravortty, D. Chronic lung infection by Pseudomonas aeruginosa biofilm is cured by L-Methionine in combination with antibiotic therapy. Sci. Rep. 2015, 5, e16043. [Google Scholar] [CrossRef] [PubMed]
- Alenanizy, F.S.; Alqahtani, F.Y.; Eltayb, E.K.; Alrumikan, N.; Almebki, R.; Alhossan, A.; Almangour, T.A.; Al-Qahtani, H. Evaluating the effect of antibiotics sub-inhibitory dose on Pseudomonas aeruginosa quorum sensing dependent virulence and its phenotypes. Saudi J. Biol. Sci. 2020. [Google Scholar] [CrossRef]
- Hemati, S.; Kouhsari, E.; Sadeghifard, N.; Maleki, A.; Omidi, N.; Mahdavi, Z.; Pakzad, I. Sub-minimum inhibitory concentrations of biocides induced biofilm formation in Pseudomonas aeruginosa. New Microbe New Infect. 2020, 38, e100794. [Google Scholar] [CrossRef]
- Navidifar, T.; Amin, M.; Rashno, M. Effects of sub-inhibitory concentrations of meropenem and tigecycline on the expression of genes regulating pili, efflux pumps and virulence factors involved in biofilm formation by Acinetobacter baumannii. Infect. Drug Resist. 2019, 12, 1099–1111. [Google Scholar] [CrossRef]
- Subedi, D.; Vijay, A.K.; Wilcox, M. Study of Disinfectant Resistance Genes in Ocular Isolates of Pseudomonas aeruginosa. Antibiotics 2018, 7, 88. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Lin, J.; Ma, J.; Cronan, J.E.; Wang, H. Triclosan Resistance of Pseudomonas aeruginosa PAO1 Is Due to FabV, a Triclosan-Resistant Enoyl-Acyl Carrier Protein Reductase. Antimicrob. Agents Chemother. 2010, 54, 689–698. [Google Scholar] [CrossRef]
- Gobbetti, M.; De Angelis, M.; Di Cagno, R.; Minervini, F.; Limitone, A. Cell–cell communication in food related bacteria. Int. J. Food Microbiol. 2007, 120, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.B.; Bassler, B.L. Quorum sensing in bacteria. Ann. Rev. Microbiol. 2001, 55, 165–199. [Google Scholar] [CrossRef] [PubMed]
- Pesci, E.C.; Iglewski, B.H. The chain of command in Pseudomonas quorum sensing. Trends Microbiol. 1997, 2, 132–134. [Google Scholar] [CrossRef]
- Turan, N.B.; Chormey, D.S.; Büyükpinar, C.; Engin, G.O.; Bakirdere, S. Quorum sensing: Little talks for an effective bacterial coordination. Trends Anal. Chem. 2017, 91, 1–11. [Google Scholar] [CrossRef]
- Rampioni, G.; Leoni, L.; Williams, P. The art of antibacterial warfare: Deception through interference with quorum sensing–mediated communication. Bioorg. Chem. 2014, 55, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Fuse, K.; Fujimura, S.; Kikuchi, T.; Gomi, K.; Iida, Y.; Nukiwa, T.; Watanabe, A. Reduction of virulence factor pyocyanin production in multidrug-resistant Pseudomonas aeruginosa. J. Infect. Chemother. 2013, 19, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Whiteley, M.; Lee, K.M.; Greenberg, E. Identification of genes controlled by quorum sensing in Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 1999, 96, 13904–13909. [Google Scholar] [CrossRef] [PubMed]
- Venturi, V. Regulation of quorum sensing in Pseudomonas. FEMS Microbiol. Rev. 2006, 30, 274–291. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.S.; Iglewski, B.H. Pseudomonas aeruginosa quorum sensing as a potential antimicrobial target. J. Clin. Investig. 2003, 112, 1460–1465. [Google Scholar] [CrossRef]
- Duan, K.; Surette, M.G. Environmental Regulation of Pseudomonas aeruginosa PAO1 Las and Rhl Quorum-Sensing Systems. J. Bacteriol. 2007, 189, 4827–4836. [Google Scholar] [CrossRef]
- Lee, J.; Zhang, L. The hierarchy quorum sensing network in Pseudomonas aeruginosa. Protein Cell 2015, 6, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Postat, J.; Bousso, P. Quorum Sensing by Monocyte-Derived Populations. Front. Immunol. 2019, 10, e2140. [Google Scholar] [CrossRef] [PubMed]
- Shih, P.C.; Huang, C.T. Effects of quorum-sensing deficiency on Pseudomonas aeruginosa biofilm formation and antibiotic resistance. J. Antimicrob. Chemother. 2002, 49, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Geisinger, E.; Isberg, R.R. Interplay between Antibiotic Resistance and Virulence During Disease Promoted by Multidrug-Resistant Bacteria. J. Infect. Dis. 2017, 215, S9–S17. [Google Scholar] [CrossRef]
- Rumbaugh, K.P.; Griswold, J.A.; Iglewski, B.H.; Hamood, A.N. Contribution of Quorum Sensing to the Virulence of Pseudomonas aeruginosa in Burn Wound Infections. Infect Immun. 1999, 67, 5854–5862. [Google Scholar] [CrossRef]
- Krzyzek, P. Challenges and Limitations of Anti-quorum Sensing Therapies. Front. Microbiol. 2019, 10, e2473. [Google Scholar] [CrossRef]
- Rada, B.; Leto, T.L. Pyocyanin effects on respiratory epithelium: Relevance in Pseudomonas aeruginosa airway infections. Trends Microbiol. 2013, 21, 73–81. [Google Scholar] [CrossRef]
- Song, F.; Wang, H.; Sauer, K.; Ren, D. Cyclic-di-GMP and oprF Are Involved in the Response of Pseudomonas aeruginosa to Substrate Material Stiffness during Attachment on Polydimethylsiloxane (PDMS). Front. Microbiol. 2018, 9, 110. [Google Scholar] [CrossRef]
- Valentini, M.; Filloux, A. Biofilms and Cyclic di-GMP (c-di-GMP) Signaling: Lessons from Pseudomonas aeruginosa and Other Bacteria. J. Biol. Chem. 2016, 291, 12547–12555. [Google Scholar] [CrossRef]
- Sadikot, R.T.; Blackwell, T.S.; Chrsitman, J.W.; Prince, A.S. Pathogen–Host Interactions in Pseudomonas aeruginosa Pneumonia. Am. J. Respir. Crit. Care Med. 2005, 171, 1209–1223. [Google Scholar] [CrossRef]
- Gales, A.C.; Jones, R.N.; Forward, K.R.; Linares, J.; Sader, H.S.; Verhoef, J. Emerging importance of multidrug-resistant Acinetobacter species and Stenotrophomonas maltophilia as pathogens in seriously ill patients: Geographic patterns, epidemiological features, and trends in the SENTRY Antimicrobial Surveillance Program (1997–1999). Clin. Infect. Dis. 2001, 32, S104–S113. [Google Scholar] [CrossRef]
- Pourcel, C.; Midoux, C.; Vergnaud, C.; Latino, L. The Basis for Natural Multiresistance to Phage in Pseudomonas aeruginosa. Antibiotics 2020, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Bentzmann, S.; Plésiat, P. The Pseudomonas aeruginosa opportunistic pathogen and human infections. Environ. Microbiol. 2011, 13, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- Pressler, T.; Bohmova, C.; Conway, S.; Dumcius, S.; Hjelte, L.; Hoiby, N.; Kollberg, H.; Tümmler, B.; Varvova, V. Chronic Pseudomonas aeruginosa infection definition: EuroCareCF Working Group report. J. Cyst. Fibros. 2011, 10, S75–S78. [Google Scholar] [CrossRef]
- Moradali, M.F.; Ghods, S.; Rehm, B.H. Pseudomonas aeruginosa lifestyle: A paradigm for adaptation, survival, and persistence. Front. Cell Infect. Microbiol. 2017, 7, e39. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.W.; Floyd, R.V.; Fothergill, J.L. The contribution of Pseudomonas aeruginosa virulence factors and host factors in the establishment of urinary tract infections. FEMS Microbiol. Lett. 2017, 364, e15. [Google Scholar] [CrossRef] [PubMed]
- Navon-Venezia, S.; Ben-Ami, R.; Carmeli, Y. Update on Pseudomonas aeruginosa and Acinetobacter baumannii infections in the healthcare setting. Curr. Opin. Infect. Dis. 2005, 18, 306–313. [Google Scholar] [CrossRef]
- Zilahi, G.; Artigas, A.; Loeches-Martin, I. What’s new in multidrug-resistant pathogens in the ICU? Ann. Intensive Care 2016, 6, e96. [Google Scholar] [CrossRef]
- Parcell, B.J.; Oravoca, K.; Pinheiro, M.; Holden, M.T.G.; Phillips, G.; Turton, J.F.; Giellspie, S.H. Pseudomonas aeruginosa intensive care unit outbreak: Winnowing of transmissions with molecular and genomic typing. J. Hosp. Infect. 2018, 98, 282–288. [Google Scholar] [CrossRef]
- Tarafdar, F.; Jafari, B.; Azimi, T. Evaluating the antimicrobial resistance patterns and molecular frequency of blaoxa-48 and blaGES-2 genes in Pseudomonas aeruginosa and Acinetobacter baumannii strains isolated from burn wound infection in Tehran, Iran. New Microbe New Infect. 2020, 37, e100686. [Google Scholar] [CrossRef]
- Gould, I.M.; Wise, R. Pseudomonas aeruginosa: Clinical manifestations and management. Lancet 1985, 326, 1224–1227. [Google Scholar] [CrossRef]
- Hue, B.; Doat, M.; Renald, G.; Brandely, M.L.; Chast, F. Severe Keratitis Caused by Pseudomonas aeruginosa Successfully Treated with Ceftazidime Associated with Acetazolamide. J. Opthtalmol. 2009, 2009, e794935. [Google Scholar]
- Willcox, M.D.P. Management and treatment of contact lens-related Pseudomonas keratitis. Clin. Opthalmol. 2012, 6, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, Y.; Wan, Q. Pseudomonas aeruginosa bacteremia among liver transplant recipients. Infect. Drug Res. 2018, 11, 2345–2356. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.; Zeng, J.; Chang, Y.; Han, S.; Zhao, J.; Fan, Y.; Xiong, Z.; Zou, X.; Wang, C.; et al. Risk Factors for Mortality of Inpatients with Pseudomonas aeruginosa Bacteremia in China: Impact of Resistance Profile in the Mortality. Infect. Drug Res. 2020, 13, 4115–4123. [Google Scholar] [CrossRef]
- Rojas, A.; Palacios-Baena, Z.R.; López-Cortés, L.E.; Rodriguez-Bano, J. Rates, predictors and mortality of community-onset bloodstream infections due to Pseudomonas aeruginosa: Systematic review and meta-analysis. Clin. Microbiol. Infect. 2019, 25, 964–970. [Google Scholar] [CrossRef]
- Chai, Y.H.; Xu, J.F. How does Pseudomonas aeruginosa affect the progression of bronchiectasis? Clin. Microbiol. Infect. 2019, 26, 313–318. [Google Scholar] [CrossRef]
- Xu, M.; Zhang, H.; Yu, N.; Dong, Y.; Wang, W.; Chen, Y.; Kang, J. Cigarette smoke extract induces the Pseudomonas aeruginosa nfxC drug-resistant phenotype. J. Inf. Chemother. 2020, 26, 1278–1282. [Google Scholar] [CrossRef]
- Stefani, S.; Campana, S.; Lariani, L.; Carnovale, V.; Colombo, C.; Lleo, M.M.; Iula, V.D.; Minicucci, L.; Morelli, P.; Pizzamiglio, G.; et al. Relevance of multidrug-resistant Pseudomonas aeruginosa infections in cystic fibrosis. Int. J. Med. Microbiol. 2017, 307, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Stableforth, D. The treatment of respiratory pseudomonas infection in cystic fibrosis: What drug and which way? Drugs 2000, 60, 1053–1064. [Google Scholar] [CrossRef]
- Carmeli, Y.; Troillet, N.; Karchmer, A.W.; Samore, M.H. Health and economic outcomes of antibiotic resistance in Pseudomonas aeruginosa. Arch. Intern. Med. 1999, 159, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Zhen, X.; Lundborg, C.S.; Sun, X.; Gu, S.; Dong, H. Clinical and Economic Burden of Carbapenem-Resistant Infection or Colonization Caused by Klebsiella pneumoniae, Pseudomonas aeruginosa, Acinetobacter baumannii: A Multicenter Study in China. Antibiotics 2020, 9, 514. [Google Scholar] [CrossRef] [PubMed]
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, G.C.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Lijequist, B.; et al. Multidrug-resistant, Extensively Drug-Resistant and Pandrug-Resistant Bacteria: An International Expert Proposal for Interim Standard Definitions for Acquired Resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Antimicrobial Resistance: Global Report on Surveillance. Available online: https://www.who.int/iris/bitstream/10665/112642/1/9789241564748_eng.pdf?ua=1 (accessed on 8 December 2020).
- ECDC/EMEA Joint Technical Report: The Bacterial Challenge: Time to React. Available online: https://www.ecdc.europa.eu/en/publications-data/ecdcemea-joint-technical-report-bacterial-challenge-time-react (accessed on 8 December 2020).
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. Available online: https://www.jpiamr.eu/wp-content/uploads/2014/12/AMR-Review-Paper-Tackling-a-crisis-for-the-health-and-wealth-of-nations_1-2.pdf (accessed on 8 December 2020).
- CDC. Antibiotic/Antimicrobial Resistance (AR/AMR). Available online: https://www.cdc.gov/drugresistance/biggest_threats.html (accessed on 8 December 2020).
- Shallcross, L.J.; Howard, S.J.; Fowler, T.; Davies, S.C. Tackling the threat of antimicrobial resistance: From policy to sustainable action. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140082. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M. The Concept of an Ideal Antibiotic: Implications for Drug Design. Molecules 2019, 24, 892. [Google Scholar] [CrossRef] [PubMed]
- van Duin, D.; Paterson, D. Multidrug Resistant Bacteria in the Community: Trends and Lessons Learned. Infect. Dis. Clin. N. Am. 2016, 30, 377–390. [Google Scholar] [CrossRef]
- Cassini, A.; Högberg, D.L.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.N.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Burden of the AMR Collaborative Group. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2019, 19, 55–56. [Google Scholar] [CrossRef]
- United Nations. Sustainable Development Goals (SDGs). Available online: https://www.un.org/sustainabledevelopment/sustainable-development-goals/ (accessed on 8 December 2020).
- Boucher, H.W.; Talbot, G.H.; Bradley, J.S.; Edwards, J.E.; Gilbert, D.; Rice, L.B.; Scheld, M.; Spellberg, B.; Bartlett, J. Bad Bugs, No Drugs: No ESKAPE! An Update from the Infectious Diseases Society of America. Clin. Infect. Dis. 2009, 48, 1–12. [Google Scholar] [CrossRef]
- World Health Organisation. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics; World Health Organisation: Geneva, Switzerland, 2017; pp. 1–7. [Google Scholar]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef]
- Ranjan, V.K.; Mukherjee, S.; Thakur, S.; Gupta, K.; Chakraborty, R. Pandrug-resistant Pseudomonas spp. expresses New Delhi metallo-β-lactamase-1 and consumes ampicillin as sole carbon source. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Bassetti, M.; Vena, A.; Croxatto, A.; Righi, E.; Guery, B. How to manage Pseudomonas aeruginosa infections. Drugs Context 2018, 7, e212527. [Google Scholar] [CrossRef] [PubMed]
- Hurley, M.; Cámara, M.; Smyth, A.R. Novel approaches to the treatment of Pseudomonas aeruginosa infections in cystic fibrosis. Eur. Resp. J. 2012, 40, 1014–1023. [Google Scholar] [CrossRef] [PubMed]
- López-Causapé, C.; Cabot, G.; del Barrio-Tofino, E.; Oliver, A. The Versatile Mutational Resistome of Pseudomonas aeruginosa. Front. Microbiol. 2018, 9, e685. [Google Scholar] [CrossRef] [PubMed]
- Talebi-Taher, M.; Majidpour, A.; Gholami, A.; Rasouli-Kouli, S.; Adabi, M. Role of efflux pump inhibitor in decreasing antibiotic cross-resistance of Pseudomonas aeruginosa in a burn hospital in Iran. J. Infect. Dev. Ctries. 2016, 10, 600–604. [Google Scholar] [CrossRef]
- Muderris, T.; Durmaz, R.; Ozdem, B.; Dal, T.; Undaldi, O.; Aydogan, S.; Celikbilek, N.; Acikgoz, Z.C. Role of efflux pump and OprD porin expression in carbapenem resistance of Pseudomonas aeruginosa clinical isolates. J. Infect. Dev. Ctries. 2018, 12, 1–8. [Google Scholar] [CrossRef]
- Kos, V.; Déraspe, M.; McLaughin, R.E.; Whiteaker, J.D.; Roy, P.H.; Alm, R.A.; Corbeil, J.; Gardner, H. The Resistome of Pseudomonas aeruginosa in Relationship to Phenotypic Susceptibility. Antimicrob. Agents Chemother. 2015, 59, 427–436. [Google Scholar] [CrossRef]
- Gajdács, M. Intravenous or oral antibiotic therapy: Sophie’s choice? Gen. Int Med. Clin. Innov. 2019, 4. [Google Scholar] [CrossRef]
- Saikawa, I.; Minami, S.; Watanabe, Y. Development of β-lactams with antipseudomonal activity. J. Infect. Chemother. 1996, 2, 53–64. [Google Scholar] [CrossRef]
- Paul, M.; Yahav, D.; Bivas, A.; Fraser, A.; Leibovci, L. Anti-pseudomonal beta-lactams for the initial, empirical, treatment of febrile neutropenia: Comparison of beta-lactams. Cochrane Database Syst. Rev. 2010, 11, CD005197. [Google Scholar] [CrossRef]
- Chaudhary, A.S. A review of global initiatives to fight antibiotic resistance and recent antibiotics’ discovery. Acta Pharm. Sin. B 2016, 6, 552–556. [Google Scholar] [CrossRef]
- Gajdács, M.; Bátori, Z.; Ábrók, M.; Lázár, A.; Burián, K. Characterization of resistance in gram-negative urinary isolates using existing and novel indicators of clinical relevance: A 10-year data analysis. Life 2020, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Kadri, S.S.; Adjemian, J.; Lai, Y.L.; Spaulding, A.B.; Ricotta, E.; Prevots, D.R.; Powers, J.H., III. Difficult-to-treat resistance in gram-negative bacteremia at 173 US hospitals: Retrospective cohort analysis of prevalence, predictors, and outcome of resistance to all first-line agents. Clin. Infect. Dis. 2018, 67, 1803–1814. [Google Scholar] [CrossRef] [PubMed]
- Pachori, P.; Gothalwal, R.; Gandhi, P. Emergence of antibiotic resistance Pseudomonas aeruginosa in intensive care unit; A critical review. Genes Dis. 2016, 6, e109. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Gao, X.; Gui, H.; Ding, X.; Lu, Y.; An, S.; Liu, Q. Proteomic Analysis of Proteins Associated with Inhibition of Pseudomonas aeruginosa Resistance to Imipenem Mediated by the Chinese Herbal Medicine Qi Gui Yin. Microb. Drug Res. 2020. [Google Scholar] [CrossRef]
- Raman, G.; Avendano, E.E.; Chan, J.; Merchant, S.; Puzniak, L. Risk factors for hospitalized patients with resistant or multidrug-resistant Pseudomonas aeruginosa infections: A systematic review and meta-analysis. Antimicrob. Res. Infect. Control 2018, 7, e79. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Zeng, J.; Wang, X.; Drlica, K.; Zhao, X. Post-stress bacterial cell death mediated by reactive oxygen species. Proc. Natl. Acad. Sci. USA 2019, 116, 10064–10071. [Google Scholar] [CrossRef]
- Liu, J.; Gefen, O.; Ronin, I.; Bar-Meir, M.; Balaban, N.Q. Effect of tolerance on the evolution of antibiotic resistance under drug combinations. Science 2020, 367, 200–204. [Google Scholar]
- Silhavy, T.J.; Kahne, D.; Walker, S. The Bacterial Cell Envelope. Cold Spring Harb. Perspect. Biol. 2010, 2, a000414. [Google Scholar] [CrossRef]
- Hassuna, N.A.; Darwish, M.K.; Sayed, M.; Ibrahem, R.A. Molecular Epidemiology and Mechanisms of High-Level Resistance to Meropenem and Imipenem in Pseudomonas aeruginosa. Infect. Drug Res. 2020, 13, 285–293. [Google Scholar] [CrossRef]
- Mirzaei, B.; Bazgir, Z.N.; Goli, H.R.; Iranpour, F.; Mohammadi, F.; Babaei, R. Prevalence of multi-drug resistant (MDR) and extensively drug-resistant (XDR) phenotypes of Pseudomonas aeruginosa and Acinetobacter baumannii isolated in clinical samples from Northeast of Iran. BMC Res. Notes 2020, 13, e380. [Google Scholar] [CrossRef]
- Ferreiro, J.L.L.; Otero, J.Á.; González, L.G.; Lamazares, L.N.; Blanco, A.A.; Sanjurjo, J.R.B.; Conde, I.R.; Soneira, M.F.; Aguado, J.F. Pseudomonas aeruginosa urinary tract infections in hospitalized patients: Mortality and prognostic factors. PLoS ONE 2017, 12, e0178178. [Google Scholar] [CrossRef]
- Chevalier, S.; Bouffartigues, E.; Bodilis, J.; Maillot, O.; Lesouhaitier, O.; Feuilloley, M.G.J.; Orange, N.; Dufour, A.; Cornelis, P. Structure, function and regulation of Pseudomonas aeruginosa porins. FEMS Microbiol. Rev. 2017, 41, 698–722. [Google Scholar] [CrossRef] [PubMed]
- Shariati, A.; Azimi, T.; Ardebili, A.; Chirani, A.S.; Bahramian, A.; Pormohammad, A.; Sardedinamin, M.; Erfanimanesh, S.; Bostanghadiri, N.; Shams, S.; et al. Insertional inactivation of oprD in carbapenem-resistant Pseudomonas aeruginosa strains isolated from burn patients in Tehran, Iran. New Microbe New Infect. 2018, 21, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Kaier, K.; Heister, T.; Götting, T.; Wolkewitz, M.; Mutters, N.T. Measuring the in-hospital costs of Pseudomonas aeruginosa pneumonia: Methodology and results from a German teaching hospital. BMC Infect. Dis. 2019, 19, e1028. [Google Scholar] [CrossRef]
- Yoneyama, H.; Nakae, T. Mechanism of Efficient Elimination of Protein D2 in Outer Membrane of Imipenem-Resistant Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 1993, 37, 2385–2390. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.E.E.; Zhong, L.L.; Shen, C.; Yang, Y.; Doi, Y.; Tian, G.B. Colistin and its role in the Era of antibiotic resistance: An extended review (2000–2019). Emerg. Microbes Infect. 2020, 9, 868–885. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.J.; Deng, Z.Q.; Yan, A.X. Bacterial multidrug efflux pumps: Mechanisms, physiology and pharmacological exploitations. Biochem. Bioph. Res. Commun. 2014, 453, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Blair, J.M.A.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J.V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015, 13, 42–51. [Google Scholar] [CrossRef]
- Piddock, L.J.V. Clinically relevant chromosomally encoded multidrug resistance efflux pumps in bacteria. Clin. Microbiol. Rev. 2006, 19, 382–402. [Google Scholar] [CrossRef]
- Martinez, J.L.; Sanchez, M.B.; Martinez-Solano, L.; Hernandez, A.; Garmendia, L.; Fajardo, A.; Alvarez-Ortega, C. Functional role of bacterial multidrug efflux pumps in microbial natural ecosystems. FEMS Microbiol. Rev. 2009, 33, 430–449. [Google Scholar] [CrossRef]
- Nicasio, A.M.; Kuti, J.L.; Nicolau, D.P. The current state of multidrug-resistant gram-negative bacilli in North America. Pharmacotherapy 2008, 28, 235–249. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. Mechanisms of Resistance to Quinolones. Clin. Infect. Dis. 2005, 41, S120–S126. [Google Scholar] [CrossRef] [PubMed]
- Garneau-Tsodikova, S.; Labby, K.J. Mechanisms of Resistance to Aminoglycoside Antibiotics: Overview and Perspectives. MedChemComm 2016, 7, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Ullmann, I.F.; Tunsjo, H.S.; Andreassen, M.; Nielsen, K.M.; Lund, V.; Charnock, C. Detection of Aminoglycoside Resistant Bacteria in Sludge Samples from Norwegian Drinking Water Treatment Plants. Front. Microbiol. 2019, 10, e487. [Google Scholar] [CrossRef] [PubMed]
- Olaitan, A.O.; Morand, S.; Rolain, J.M. Mechanisms of polymyxin resistance: Acquired and intrinsic resistance in bacteria. Front. Microbiol. 2014, 5, e643. [Google Scholar] [CrossRef]
- Lin, J.; Xu, C.; Fang, R.; Cao, J.; Zhang, X.; Zhao, Y.; Dong, G.; Sun, Y.; Zhou, T. Resistance and Heteroresistance to Colistin in Pseudomonas aeruginosa Isolates from Wenzhou, China. Antimicrob. Agents Chemother. 2019, 63, e00556-19. [Google Scholar] [CrossRef]
- El-Baky, A.R.M.; Masoud, S.M.; Mohamed, D.S.; Waly, N.F.G.M.; Shafik, E.A.; Mohareb, D.A.; Elkady, A.; Elbadr, M.M.; Hetta, H.F. Prevalence and Some Possible Mechanisms of Colistin Resistance Among Multidrug-Resistant and Extensively Drug-Resistant Pseudomonas aeruginosa. Infect. Drug Res. 2020, 13, 323–332. [Google Scholar] [CrossRef]
- Das, P.; Sengupta, K.; Goel, G.; Bhattacharya, S. Colistin: Pharmacology, drug resistance and clinical applications. J. Acad. Clin. Microbiol. 2017, 19, 77–85. [Google Scholar] [CrossRef]
- Munita, J.M.; Arias, C.A. Mechanisms of antibiotic resistance. Virulence mechanisms of bacterial pathogens. Microbiol. Spectr. 2016, 2016, 481–511. [Google Scholar]
- Falagas, M.; Blizotis, I.A.; Kasiakou, S.A.; Samonis, G.; Athanassopoulou, P.; Michalopoulos, A. Outcome of infections due to pandrug-resistant (PDR) Gram-negative bacteria. BMC Infect. Dis. 2005, 5, e24. [Google Scholar] [CrossRef]
- Kempf, M.; Rolain, J.M. Emergence of resistance to carbapenems in Acinetobacter baumannii in Europe: Clinical impact and therapeutic options. Int. J. Antimicrob. Agents 2012, 39, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, I.; Hackel, M.; Badal, R.; Bouchillon, S.; Hawser, S.; Biedenbach, D. A Review of Ten Years of the Study for Monitoring Antimicrobial Resistance Trends (SMART) from 2002 to 2011. Pharmaceuticals 2013, 6, 1335–1346. [Google Scholar] [CrossRef] [PubMed]
- Azuma, M.; Murakami, K.; Murata, R.; Kataoka, K.; Fujii, H.; Miyake, Y.; Nishioka, Y. Clinical Significance of Carbapenem-Tolerant Pseudomonas aeruginosa Isolated in the Respiratory Tract. Antibiotics 2020, 9, 626. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M.; Urbán, E. Prevalence and Antibiotic Resistance of Stenotrophomonas maltophilia in Respiratory Tract Samples: A 10-Year Epidemiological Snapshot. Health Serv. Res. Manag. Epidemiol. 2019, 6, 2333392819870774. [Google Scholar] [CrossRef]
- Kong, K.F.; Schneper, L.; Mathee, K. Beta-lactam Antibiotics: From Antibiosis to Resistance and Bacteriology. APMIS 2010, 118, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.Y.; Cao, J.M.; Yang, Q.; Chen, S.; Lv, H.Y.; Zhou, H.W.; Zhang, R. Risk Factors for Carbapenem-Resistant Pseudomonas aeruginosa, Zhejiang Province, China. Emerg. Infect. Dis. 2019, 25, 1861–1867. [Google Scholar] [CrossRef] [PubMed]
- Poole, K. Pseudomonas aeruginosa: Resistance to the max. Front. Microbiol. 2011, 2, e65. [Google Scholar] [CrossRef]
- Bradford, P.A. Extended-spectrum b-lactamases in the 21st century: Characterization, epidemiology, and detection of this important resistance threat. Clin. Microbiol. Rev. 2001, 14, 933–951. [Google Scholar] [CrossRef]
- Stehling, E.G.; da Silveira, W.D.; Leite, D.D.S. Study of biological characteristics of Pseudomonas aeruginosa strains isolated from patients with cystic fibrosis and from patients with extra-pulmonary infections. Braz. J. Infect. Dis 2008, 12, 86–88. [Google Scholar] [CrossRef]
- Stewart, P.S.; Costerton, J.W. Antibiotic resistance of bacteria in biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef]
- Fowler, R.C.; Hanson, N.D. Emergence of Carbapenem Resistance Due to the Novel Insertion Sequence ISPa8 in Pseudomonas aeruginosa. PLoS ONE 2014, 9, e91299. [Google Scholar] [CrossRef] [PubMed]
- Pai, H.; Kim, J.W.; Kim, J.; Lee, J.H.; Choe, K.W.; Gotoh, N. Carbapenem Resistance Mechanisms inPseudomonasaeruginosaClinical Isolates. Antimicrob. Agents Chemother. 2001, 45, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Fernández, L.; Hancock, R.E. Adaptive and mutational resistance: Role of porins and efflux pumps in drug resistance. Clin. Microbiol. Rev. 2012, 25, 661–681. [Google Scholar] [CrossRef] [PubMed]
- Sahuquillo-Arce, J.M.; Hernández-Cabezas, A.; Yarad-Auad, F.; Ibanez-Martinez, E.; Falomir-Salcedo, P.; Ruiz-Gaitán, A. Carbapenemases: A worldwide threat to antimicrobial therapy. World J. Pharm. 2015, 4, 75–95. [Google Scholar] [CrossRef]
- Behzadi, P.; García-Perdomo, H.A.; Karpiński, T.M.; Issakhanian, L. Metallo-ß-lactamases: A review. Mol. Biol. Rep. 2020. [Google Scholar] [CrossRef]
- Bush, K.; Jacoby, G.A. Updated Functional Classification of β-Lactamases. Antimicrob. Agents Chemother. 2010, 54, 969–976. [Google Scholar] [CrossRef]
- Papp-Wallace, K.M.; Endimiani, A.; Taracila, M.A.; Bonomo, R.A. Carbapenems: Past, Present, and Future. Antimicrob. Agent Chemother. 2011, 55, 4943–4960. [Google Scholar] [CrossRef]
- van Duin, D.; Kaye, K.S.; Neuner, E.A.; Bonomo, R.A. Carbapenem-resistant Enterobacteriaceae: A review of treatment and outcomes. Diagn. Microbiol. Infect. Dis 2013, 75, 115–120. [Google Scholar] [CrossRef]
- Meletis, G. Carbapenem resistance: Overview of the problem and future perspectives. Adv. Infect. Dis. 2016, 3, 15–21. [Google Scholar] [CrossRef]
- Hong, D.J.; Bae, I.K.; Jang, I.H.; Jeong, S.K.; Kang, K.H.; Lee, K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect. Chemother. 2015, 47, 81–97. [Google Scholar] [CrossRef]
- Li, C.; Li, Y.; Zhao, Z.; Liu, Q.; Li, B. Treatment options and clinical outcomes for carbapenem-resistant Enterobacteriaceae bloodstream infection in a Chinese university hospital. J. Infect. Public Health 2019, 12, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Memon, U.A.; Ahmed, A.R.; Khalid, M.; Quadir, K.; Jabbar, N.; Junejo, S.; Hauque, A. Clinical profile and outcome of carbapenem-resistant gram negative bacteremia in children with cancer in pediatric intensive care unit of a resource-limited country. Ped. Hemat. Oncol. J. 2020, 5, 65–68. [Google Scholar] [CrossRef]
- Shin, D.H.; Shin, D.Y.; Kang, C.K.; Park, S.; Park, J.; Jun, K.I.; Kim, T.S.; Koh, Y.; Hong, J.S.; Choe, P.G.; et al. Risk factors for and clinical outcomes of carbapenem non-susceptible gram negative bacilli bacteremia in patients with acute myelogenous leukemia. BMC Infect. Dis. 2020, 20, e404. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zeng, W.; Chen, T.; Liao, W.; Qian, J.; Lin, J.; Zhou, C.; Tian, X.; Cao, J.; Zhou, T. Mechanisms of Heteroresistance and Resistance to Imipenem in Pseudomonas aeruginosa. Infect. Drug Res. 2020, 13, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y. Treatment Options for Carbapenem-resistant Gram-negative Bacterial Infections. Clin. Infect. Dis. 2019, 69, S565–S575. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for the Prevention and Control of Carbapenem-Resistant Enterobacteriaceae, Acinetobacter baumannii and Pseudomonas aeruginosa in Health Care Facilities. Available online: https://www.who.int/infection-prevention/publications/guidelines-cre/en/ (accessed on 20 May 2020).
- CDC. Tracking Carbapenem-Resistant Pseudomonas Aeruginosa. Available online: https://www.cdc.gov/hai/organisms/pseudomonas/tracking.html (accessed on 20 May 2020).
- European Centre for Disease Prevention and Control. Surveillance Atlas of Infectious Diseases. Available online: http://atlas.ecdc.europa.eu/public/index.aspx (accessed on 20 May 2020).
- Zhang, D.; Cui, K.; Wang, T.; Shan, Y.; Dong, H.; Feng, W.; Ma, C.; Dong, Y. Risk factors for carbapenem-resistant Pseudomonas aeruginosa infection or colonization in a Chinese teaching hospital. J. Infect. Dev. Ctries 2018, 12, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Elshafiee, E.A.; Nader, S.M.; Dorgham, S.M.; Hamza, D.A. Carbapenem-resistant Pseudomonas Aeruginosa Originating from Farm Animals and People in Egypt. J. Vet. Res. 2019, 63, 333–337. [Google Scholar] [CrossRef]
- Adesida, S.A.; Coker, A.O.; Smith, S.I. Resistotyping of Campylobacter jejuni. Niger Postgrad Med. J. 2003, 10, 211–215. [Google Scholar]
- Shigemura, K.; Arakawa, S.; Sakai, Y.; Kinoshita, S.; Tanaka, K.; Fujisawa, M. Complicated urinary tract infection caused by Pseudomonas aeruginosa in a single institution (1999–2003). Int. J. Urol. 2006, 13, 538–542. [Google Scholar] [CrossRef]
- Zeng, Z.R.; Wang, W.P.; Huang, M.; Shi, L.N.; Wang, Y.; Shao, H.F. Mechanisms of carbapenem resistance in cephalosporin-susceptible Pseudomonas aeruginosa in China. Diagn. Microbiol. Infect. Dis. 2014, 78, 268–270. [Google Scholar] [CrossRef]
- Li, S.; Jia, X.; Zou, H.; Liu, H.; Guo, Y.; Zhang, L. Carbapenem-resistant and cephalosporin susceptible Pseudomonas aeruginosa: A notable phenotype in patients with bacteremia. Infect. Drug Res. 2018, 11, 1225–1235. [Google Scholar] [CrossRef] [PubMed]
- Wi, Y.M.; Choi, J.Y.; Lee, J.Y.; Kang, C.I.; Chung, D.R.; Peck, R.K.; Song, J.H.; Ko, K.S. Antimicrobial Effects of Beta-Lactams on Imipenem-Resistant Ceftazidime-Susceptible Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2017, 61, e00054-17. [Google Scholar] [CrossRef] [PubMed]
- Khuntayaporn, P.; Montakantikul, P.; Santanirand, P.; Kirasitin, P.; Chomnawang, M.T. Molecular investigation of carbapenem resistance among multidrug-resistant Pseudomonas aerugonisa isolated clinically in Thailand. Microbiol. Immunol. 2013, 57, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Campana, E.H.; Xavier, D.E.; Petrolini, F.V.; Codeiro-Moura, J.R.; de Araujo, M.R.E.; Gales, A.C. Carbapenem-resistant and cephalosporin-susceptible: A worrisome phenotype among Pseudomonas aeruginosa clinical isolates in Brazil. Braz. J. Infect. Dis. 2017, 21, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Zaidenstein, R.; Miller, A.; Tal-Jasper, R.; Ofer-Friedman, H.; Sklarz, M.; Ketz, D.E.; Lazarovitch, T.; Lephart, P.R.; Mengehsa, B.; Tzuman, O.; et al. Therapeutic Management of Pseudomonas aeruginosa Bloodstream Infection Non-Susceptible to Carbapenems but Susceptible to “Old” Cephalosporins and/or to Penicillins. Microorganisms 2018, 6, 9. [Google Scholar] [CrossRef]
- Tsai, H.M.; Wu, T.L.; Su, L.H.; Su, L.H.; Lo, W.L.; Chen, C.L.; Liang, Y.H.; Chiu, C.H. Carbapenem-resistant-only Pseudomonas aeruginosa infection in patients formerly infected by carbapenem-susceptible strains. Int. J. Antimicrob. Agents 2014, 44, 541–545. [Google Scholar] [CrossRef]
- Pena, C.; Suarez, C.; Tubau, F.; Juan, C.; Moya, M.A.; Dominguez, M.A.; Oliver, A.; Pujol, M.; Ariza, J. Nosocomial Outbreak of a Non-Cefepime-Susceptible Ceftazidime-Susceptible Pseudomonas aeruginosa Strain Overexpressing MexXY-OprM and Producing an Integron-Borne PSE-1 β-Lactamase. J. Clin. Microbiol. 2009, 47, 2381–2387. [Google Scholar] [CrossRef]
- Pournaras, S.; Maniati, M.; Spanakis, N.; Ikonomidis, A.; Tassios, P.T.; Tsakris, A.; Legakis, N.J.; Maniatis, A.N. Spread of efflux pump-overexpressing, non-metallo-b-lactamaseproducing, meropenem-resistant but ceftazidime-susceptible Pseudomonas aeruginosa in a region with blaVIM endemicity. J. Antimicrob. Chemother. 2005, 56, 761–764. [Google Scholar] [CrossRef]
- Khalili, Y.; Yekani, M.; Goli, H.R.; Memar, M.Y. Characterization of carbapenem-resistant but cephalosporin-susceptible Pseudomonas aeruginosa. Acta Microbiol. Immunol. Hung. 2019, 66, 529–540. [Google Scholar] [CrossRef]
- Lee, C.H.; Su, T.Y.; Ye, J.J.; Hsu, P.C.; Kuo, A.J.; Chia, J.S.; Lee, M.H. Risk factors and clinical significance of bacteremia caused by Pseudomonas aeruginosa resistant only to carbapenems. J. Microbiol. Immunol. Infect. 2014, 50, 677–683. [Google Scholar] [CrossRef]
- Wafy, A.A.; Wageih, S.E.; Atef, A.; Taha, A.; Gamil, T. Evaluation of Simple Screening Tests in the Diagnosis of Non-Fermentative Gram Negative bacilli, A Prospective Study. Egypt. J. Med. Microbiol. 2013, 22, 41–49. [Google Scholar] [CrossRef]
- Rodulfo, H.; Arcia, A.; Hernández, A.; Michelli, E.; Martinez, D.V.; Guzman, M.; Sharma, A.; De Donato, M. Virulence factors and integrons are associated with MDR and XDR phenotypes in nosocomial strains of Pseudomonas aeruginosa in a Venezuelan university hospital. Rev. Inst. Med. Trop. Sao Paolo 2019, 61, e20. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M. Carbapenem-Resistant but Cephalosporin-Susceptible Pseudomonas aeruginosa in Urinary Tract Infections: Opportunity for Colistin Sparing. Antibiotics 2020, 9, 153. [Google Scholar] [CrossRef] [PubMed]
- Libisch, B.; Gacs, M.; Csiszár, K.; Muzslay, M.; Rókusz, L.; Füzi, M. Isolation of an Integron-Borne blaVIM-4 Type Metallo—Lactamase Gene from a Carbapenem-Resistant Pseudomonas aeruginosa Clinical Isolate in Hungary. Antimicrob. Agent Chemother. 2004, 48, 3576–3578. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stultz, J.S.; Arnold, S.R.; Shelton, C.M.; Bagga, B.; Lee, K.R. Antimicrobial stewardship impact on Pseudomonas aeruginosa susceptibility to meropenem at a tertiary pediatric institution. Am. J. Infect. Ctrl. 2019. [Google Scholar] [CrossRef]
- Sader, H.S.; Castanheira, M.; Duncan, L.R.; Flamm, R.K. Antimicrobial Susceptibility of Enterobacteriaceae and Pseudomonas aeruginosa Isolates from United States Medical Centers Stratified by Infection Type: Results from the International Network for Optimal Resistance Monitoring (INFORM) Surveillance Program, 2015–2016. Diagn. Microbiol. Infect. Dis. 2018, 92, 69–74. [Google Scholar]
- El-Gamal, M.I.; Brahim, I.; Hisham, N.; Aladdin, R.; Mohammed, H.; Bahaaeldin, A. Recent updates of carbapenem antibiotics. Eur. J. Med. Chem. 2017, 131, 185–195. [Google Scholar] [CrossRef]
- Bassetti, M.; Righi, E.; Viscoli, C. Novel beta-lactam antibiotics and inhibitor combinations. Expert Opin. Investig. Drugs 2008, 17, 196–285. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M.; Albericio, F. Antibiotic Resistance: From the Bench to Patients. Antibiotics 2019, 8, 129. [Google Scholar] [CrossRef]
- Gajdács, M. Extra deaths due to pandrug resistant bacteria: A survey of the literature. Egészségfejlesztés 2019, 60, 31–36. [Google Scholar]
- Giacobbe, D.R.; Giani, T.; Bassetti, M.; Marchese, A.; Viscoli, C.; Rossolini, G.M. Rapid microbiological tests for bloodstream infections due to multidrug resistant Gram-negative bacteria: Therapeutic implications. Clin. Microbiol. Infect. 2020, 26, 73–722. [Google Scholar] [CrossRef] [PubMed]
- Asmepa, T.E.; Nicolau, D.P.; Kuti, J.L. Carbapenem-Nonsusceptible Pseudomonas aeruginosa Isolates from Intensive Care Units in the United States: A Potential Role for New Beta-Lactam Combination Agents. J. Clin. Microbiol. 2019, 57, e00535-19. [Google Scholar] [CrossRef] [PubMed]
- Halat, D.H.; Moubareck, C.A. The Current Burden of Carbapenemases: Review of Significant Properties and Dissemination among Gram-Negative Bacteria. Antibiotics 2020, 9, 186. [Google Scholar] [CrossRef] [PubMed]
- Idowu, T.; Zhanel, G.G.; Schwelzer, F. A Dimer, but Not Monomer, of Tobramycin Potentiates Ceftolozane against Multidrug-Resistant and Extensively Drug-Resistant Pseudomonas aeruginosa and Delays Resistance Development. Antimicrob. Agents Chemother. 2020, 64, e02055-19. [Google Scholar] [CrossRef]
- Sarshar, M.; Behzadi, P.; Ambrosi, C.; Zagaglia, C.; Palamara, A.T.; Scribano, D. FimH and Anti-Adhesive Therapeutics: A Disarming Strategy Against Uropathogens. Antibiotics 2020, 9, 397. [Google Scholar] [CrossRef]
- Chegini, Z.; Khosbayan, A.; Moghadam, M.T.; Farahani, I.; Jazieiran, P.; Shariati, A. Bacteriophage therapy against Pseudomonas aeruginosa biofilms: A review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, e45. [Google Scholar] [CrossRef]
- Castello, V.; Edwards-Gayle, J.C.C.; Hamley, I.W.; Barrett, G.; Seisonen, J.; Ruokolainen, J.; de Mello, L.R.; da Silva, E.R. Model self-assembling arginine-based tripeptides show selective activity against Pseudomonas bacteria. Chem. Commun. 2020, 56, e615. [Google Scholar] [CrossRef]
- Cernohorska, L.; Votava, M. Antibiotic Synergy against Biofilm-Forming Pseudomonas aeruginosa. Folia Microbiol. 2008, 53, 57–60. [Google Scholar] [CrossRef]
- Savoia, D. New perspectives in the management of Pseudomonas aeruginosa infections. Future Microbiol. 2014, 9, 917–928. [Google Scholar] [CrossRef]
- Sharma, G.; Rao, S.; Bansal, A.; Dang, S.; Gupta, S.; Gabrani, R. Pseudomonas aeruginosa biofilm: Potential therapeutic targets. Biologicals 2014, 42, 1–7. [Google Scholar] [CrossRef]
- Knezevic, P.; Curcin, S.; Aleksic, V.; Petrusic, M.; Vlaski, L. Phage-antibiotic synergism: A possible approach to combatting Pseudomonas aeruginosa. Res. Microbiol. 2013, 164, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Amorese, V.; Donadu, M.; Usai, D.; Sanna, A.; Milia, F.; Pisanu, F.; Molicotti, P.; Zanetti, S.; Doria, C. In vitro activity of essential oils against Pseudomonas aeruginosa isolated from infected hip implants. J. Infect. Dev. Ctries. 2018, 12, 996–1001. [Google Scholar] [CrossRef] [PubMed]
- Man, A.; Santacroce, L.; Jacob, R.; Mare, A.; Man, L. Antimicrobial Activity of Six Essential Oils Against a Group of Human Pathogens: A Comparative Study. Pathogens 2019, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Elcocks, E.R.; Spencer-Phillips, P.T.N.; Adukwu, E.C. Rapid bactericidal effect of cinnamon bark essential oil against Pseudomonas aeruginosa. J. Appl. Microbiol. 2020, 128, 1025–1037. [Google Scholar] [CrossRef]
- Stájer, A.; Kajári, S.; Gajdács, M.; Musah-Eroje, A.; Baráth, Z. Utility of Photodynamic Therapy in Dentistry: Current Concepts. Dent. J. 2020, 8, 43. [Google Scholar] [CrossRef]
- DeLeon, K.; Balldin, F.; Watters, C.; Hamood, A.; Griswold, J.; Sreedharan, S.; Rumbaugh, K.P. Gallium maltolate treatment eradicates Pseudomonas aeruginosa infection in thermally injured mice. Antimicrob. Agents Chemother. 2009, 53, 1331–1337. [Google Scholar] [CrossRef]
- Bhardway, A.K.; Vinothkumar, K.; Rajpara, N. Bacterial quorum sensing inhibitors: Attractive alternatives for control of infectious pathogens showing multiple drug resistance. Recent Pat. Antiinfect. Drug Discov. 2013, 8, 68–83. [Google Scholar] [CrossRef]
- Antunes, L.S.C.; Imperi, F.; Mindandri, F.; Visca, P. In Vitro and In Vivo Antimicrobial Activities of Gallium Nitrate against Multidrug-Resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2012, 56, 5961–5970. [Google Scholar] [CrossRef]
- Zeng, J.; Wu, L.; Liu, Z.; Lv, Y.; Feng, J.; Wang, W.; Xue, Y.; Wang, D.; Li, J.; Drlica, K.; et al. Gain-of-function mutations in acid stress response (evgS) protect Escherichia coli from killing by gallium nitrate, an antimicrobial candidate. Antimicrob. Agents Chemother. 2020. [Google Scholar] [CrossRef]
- Codjoe, F.S.; Donkor, E.S. Carbapenem Resistance: A Review. Med. Sci. 2018, 6, 1. [Google Scholar] [CrossRef]
- Gonçalves, I.R.; Dantas, R.C.; Ferreira, M.L.; Batistão, D.W.; Gontijo-Filho, P.P.; Ribas, R.M. Carbapenem-resistant Pseudomonas aeruginosa: Association with virulence genes and biofilm formation. Braz. J. Microbiol. 2017, 48, 211–217. [Google Scholar] [CrossRef] [PubMed]
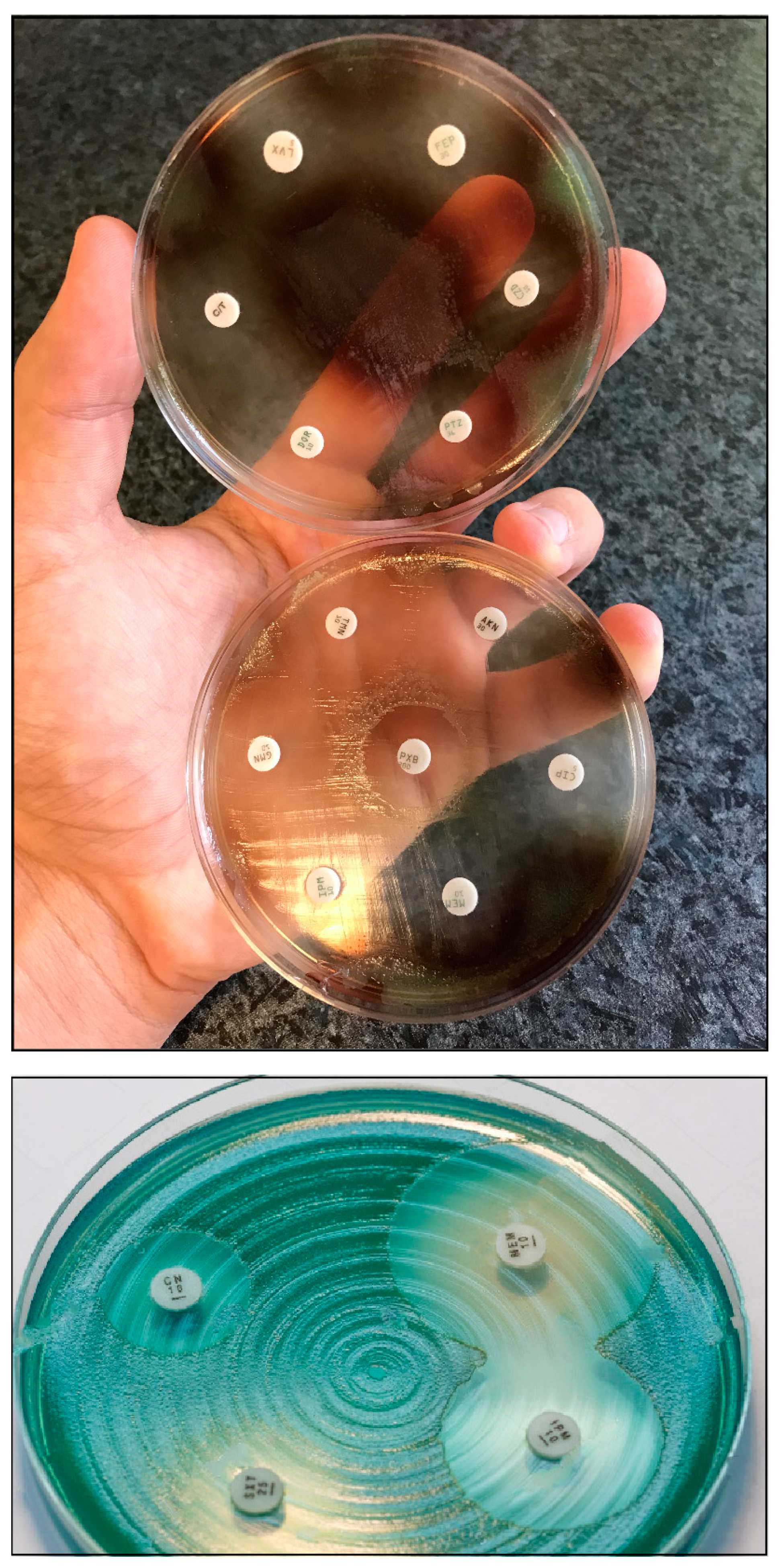
| Hospitalization |
| Mechanical ventilation |
| Innate or acquired immunodeficiencies (neutropenia, human immunodeficiency virus [HIV]/acquired immunodeficiency syndrome [AIDS], malignancy) |
| Invasive medical procedures (surgery, transplantation) |
| Catheterization (urinary, central venous) |
| Burns, severe external injuries |
| Immunosuppressive therapy |
| Cancer chemotherapy |
| Radiotherapy |
| Diseases of the cardiovascular system |
| Diseases of the respiratory system (e.g., chronic obstructive pulmonary disease, cystic fibrosis) |
| Diabetes mellitus |
| Patients living in poor living conditions, malnutrition |
| Intravenous drug use |
| Antibiotics to Which Pseudomonas Species Are Intrinsically Resistant | Antibiotics Relevant in the Therapy of Pseudomonas Infections |
|---|---|
| Glycopeptides (e.g., vancomycin) | β-lactam antibiotics: third and fourth generation parenterally administered cephalosporins (ceftazidime, cefepime, piperacillin/tazobactam), monobactams (aztreonam), carbapenems (imipenem, meropenem and doripenem), novel β-lactam/β-lactamase inhibitor combinations (ceftolozane/tazobactam, ceftazidime/avibactam, imipenem/cilastatin/relebactam, meropenem/vaborbactam) |
| Daptomycin | |
| Oxazolidinones (e.g., linezolid) | |
| Macrolides (e.g., azithromycin) | |
| Lincosamides (e.g., clindamycin) | |
| Streptogramins (e.g., quinpristin-dalfopristin) | |
| Rifampin | |
| Trimethoprim-sulfamethoxazole | Fluoroquinolones: ciprofloxacin, levofloxacin, moxifloxacin, delafloxacin |
| Tetracycline | |
| Aminopenicillins | Aminoglycosides, neoglycosides: gentamicin, tobramycin, amikacin, plazmomicin |
| Aminopenicillin/β-lactamase-inhibitor combinations | |
| I–II. generation cepalosporins | Polymyxins: colistin |
| Orally administered III generation cephalosporins |
| Emerging therapeutic strategy | Description (when relevant) |
| Novel antibiotics, antibiotic combination therapy | Ceftolozane/tazobactam, ceftazidim/avibactam, imipenem/cilastatin/relebactam, meropenem/vaborbactam, plazmomicin, delafloxacin |
| Existing drugs in novel formulations | Nebulized or liposomal tobramycin, levofloxacin, aztreonam lysine, fosfomycin, colistin, and lyposimal used to treat P. aeruginosa in the lungs of CF patients |
| Phage therapy, endolysins | Bactericidal, highly specific to target bacteria without affecting the commensal bacteria, effective against MDR isolates, synergistic activity with antibiotics, may penetrate dense biofilms. Endolysins: they degrade the bacterial peptidoglycan from the inside of the cell during the lytic cycle of phages. |
| Siderophores, iron chelation | Perturbation of irom metabolism has been proposed as an emerging therapeutic strategy. Gallium (Ga3+): clinical trials include iv. gallium-nitrate (GaNite) and co-encapsulation of Ga-gentamicin in CF patients. The exact mechanism of action for Ga is still poorly understood. Some studies propose that Ga interferes with iron (Fe) uptake, Fe metabolism, and inhibits the function of Fe-containing respiratory enyzmes; however, this explanation was deemed unstatisfactory, as most compounds affecting Fe-metabolism or acting through chelation are bacteriostatic, while Ga has rapid bactericidal activity. Newer studies suggest that Ga treatment acts through the generation of ROS and the inhibition of antioxidant defence mechanisms in bacteria. |
| Lectin inhibition | Inhibition of LecA/LecB binding to lung epithelial cells. |
| Quorum sensing (QS) inhibition, virulence inhibition | Inhibition of signal molecule synthesis or sensing, which may hinder bacteria from adapting to diverse ecological niches, evading the immune system and producing virulence factors. Virulence inhibitors may “disarm” bacteria; therefore, they will not be able to induce their characteristic pathologies in vivo. In addition, as QS and virulence inhibitors do not target essential cellular components (which leads to high levels of selection pressure and the emergence of resistant mutants), it is unlikely that the host microbiome will be affected or that rapid resistance against these agents will occur. |
| Efflux pump inhibitors | |
| Antimicrobial peptides (AMPs) | |
| Photodynamic therapy | |
| Vaccine development | |
| Nanoparticles (NPs) | |
| Monoclonal antibodies | |
| Conjugates | |
| Natural compounds, essential oils |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behzadi, P.; Baráth, Z.; Gajdács, M. It’s Not Easy Being Green: A Narrative Review on the Microbiology, Virulence and Therapeutic Prospects of Multidrug-Resistant Pseudomonas aeruginosa. Antibiotics 2021, 10, 42. https://doi.org/10.3390/antibiotics10010042
Behzadi P, Baráth Z, Gajdács M. It’s Not Easy Being Green: A Narrative Review on the Microbiology, Virulence and Therapeutic Prospects of Multidrug-Resistant Pseudomonas aeruginosa. Antibiotics. 2021; 10(1):42. https://doi.org/10.3390/antibiotics10010042
Chicago/Turabian StyleBehzadi, Payam, Zoltán Baráth, and Márió Gajdács. 2021. "It’s Not Easy Being Green: A Narrative Review on the Microbiology, Virulence and Therapeutic Prospects of Multidrug-Resistant Pseudomonas aeruginosa" Antibiotics 10, no. 1: 42. https://doi.org/10.3390/antibiotics10010042
APA StyleBehzadi, P., Baráth, Z., & Gajdács, M. (2021). It’s Not Easy Being Green: A Narrative Review on the Microbiology, Virulence and Therapeutic Prospects of Multidrug-Resistant Pseudomonas aeruginosa. Antibiotics, 10(1), 42. https://doi.org/10.3390/antibiotics10010042







