A Multi-Wavelength Opto-Electronic Patch Sensor to Effectively Detect Physiological Changes against Human Skin Types
Abstract
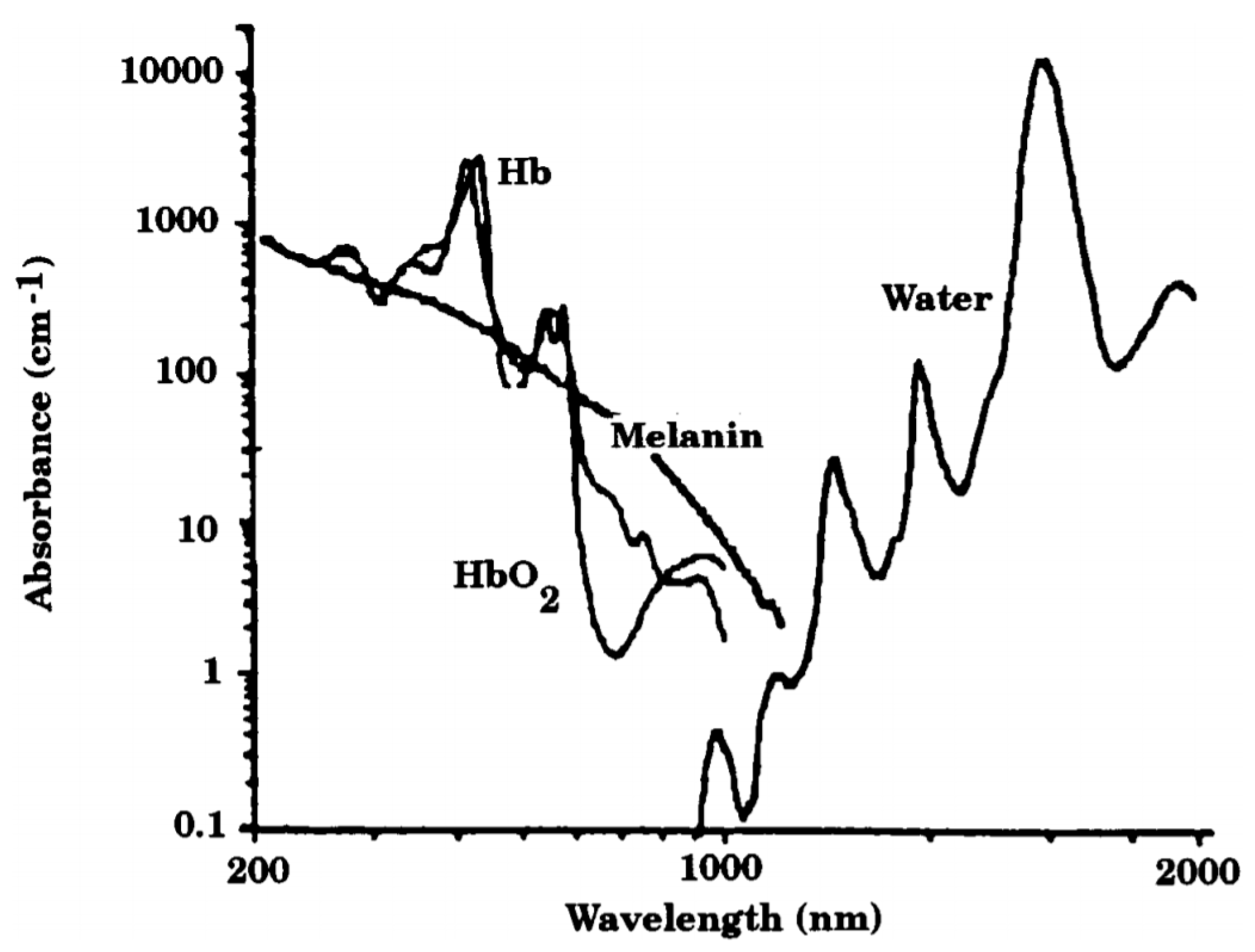
:1. Introduction
2. Materials and Methods
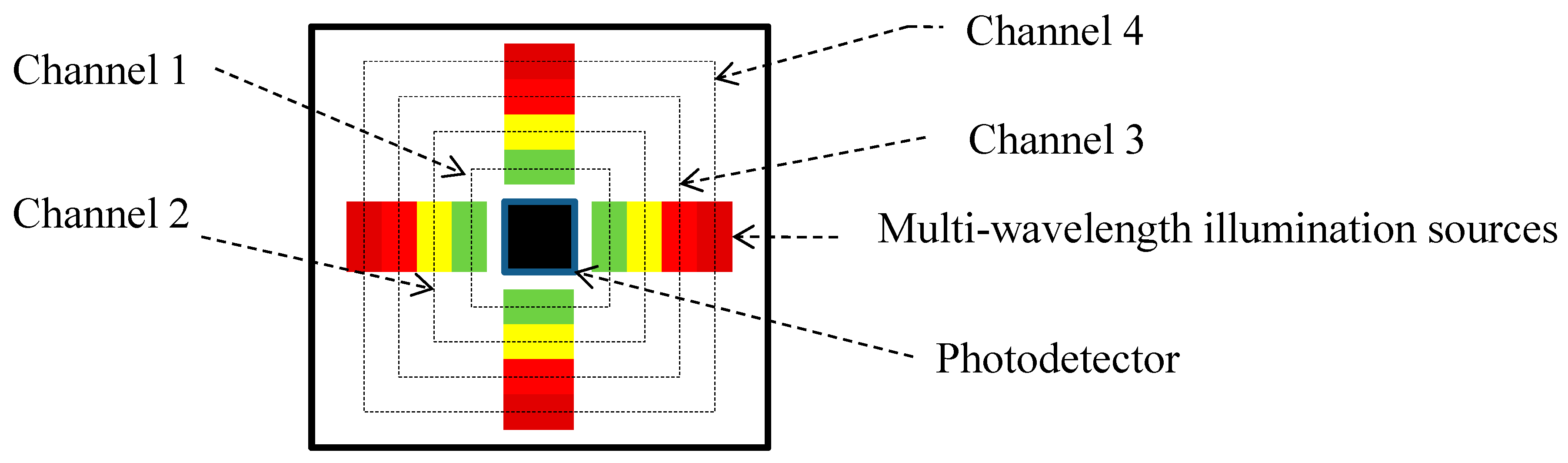
2.1. OEPS Configuration
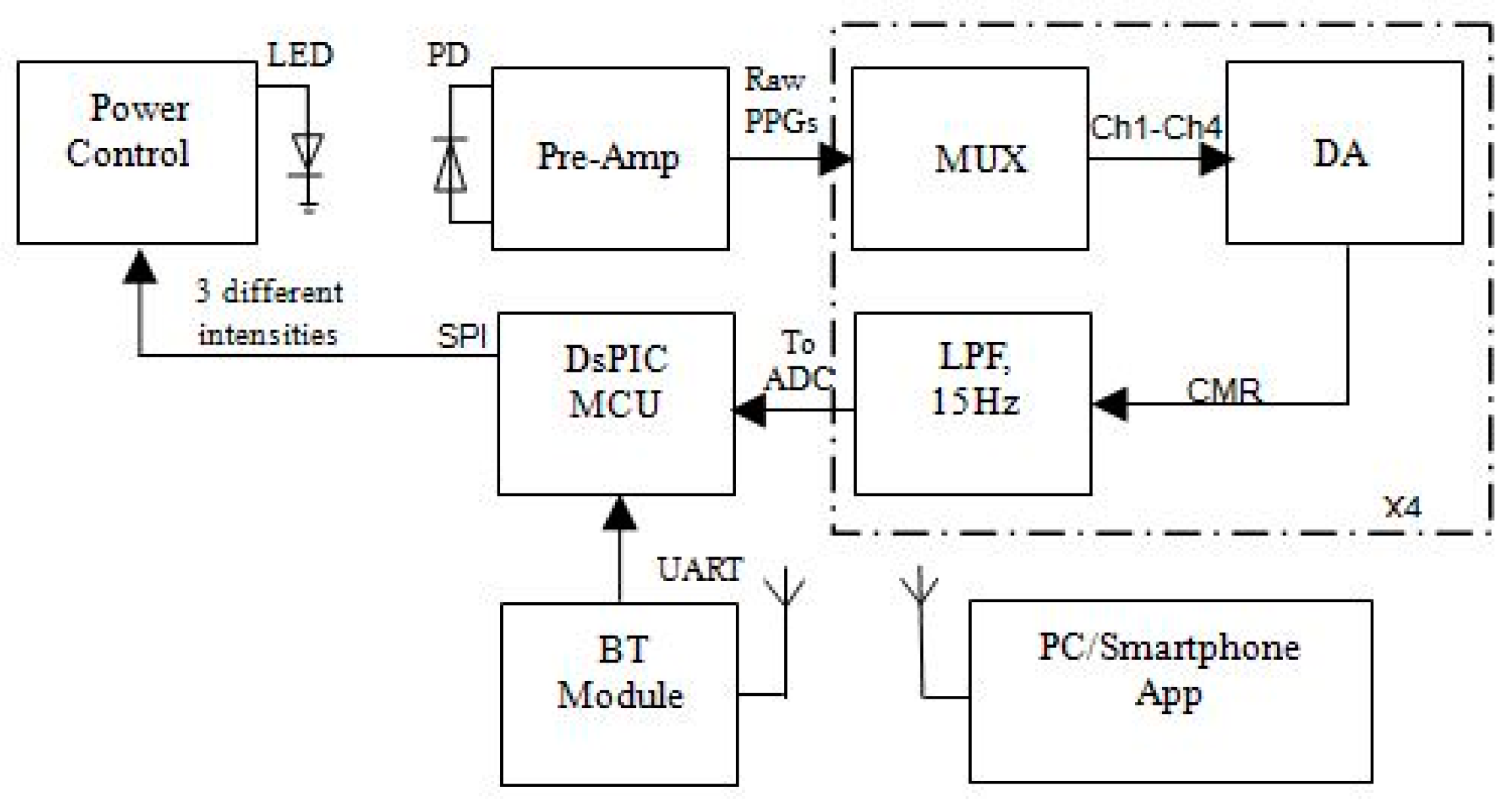
2.2. Electronic Composition
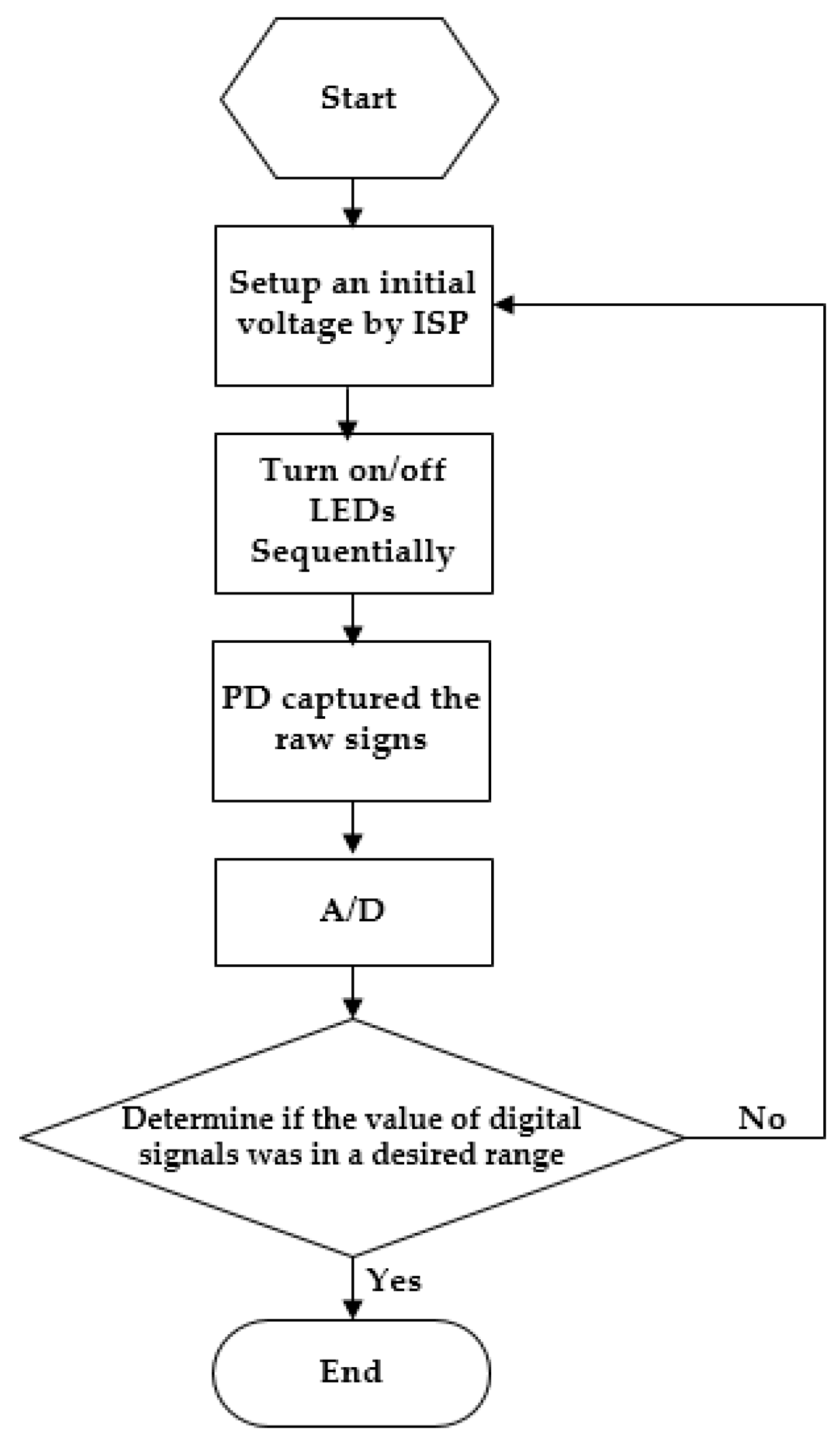
2.3. Execution of OEPS System
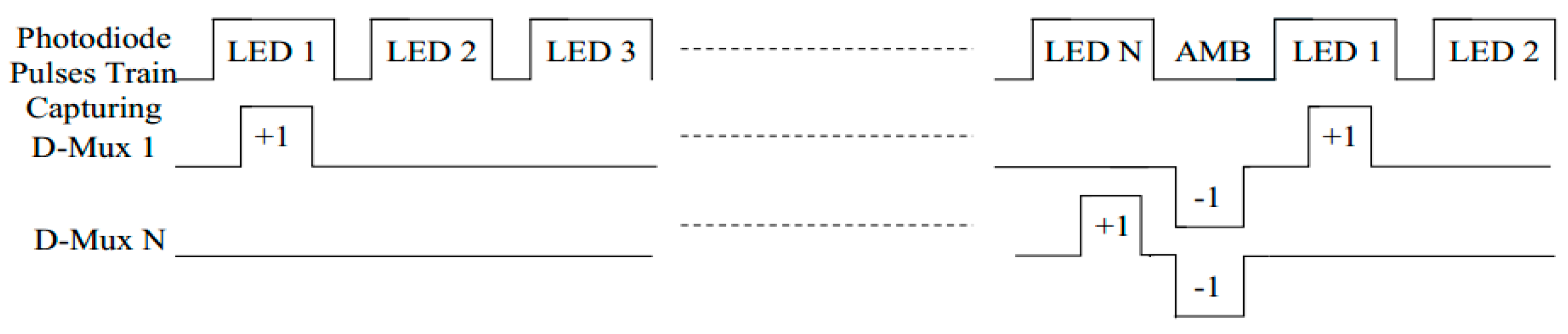
(1) Time sequence of multiplexing LED and demultiplexing signals from the PD.
(2) Auto adaption for LED illumination intensity.
(3) OEPS measurement procedures.
2.4. Experimental Protocol
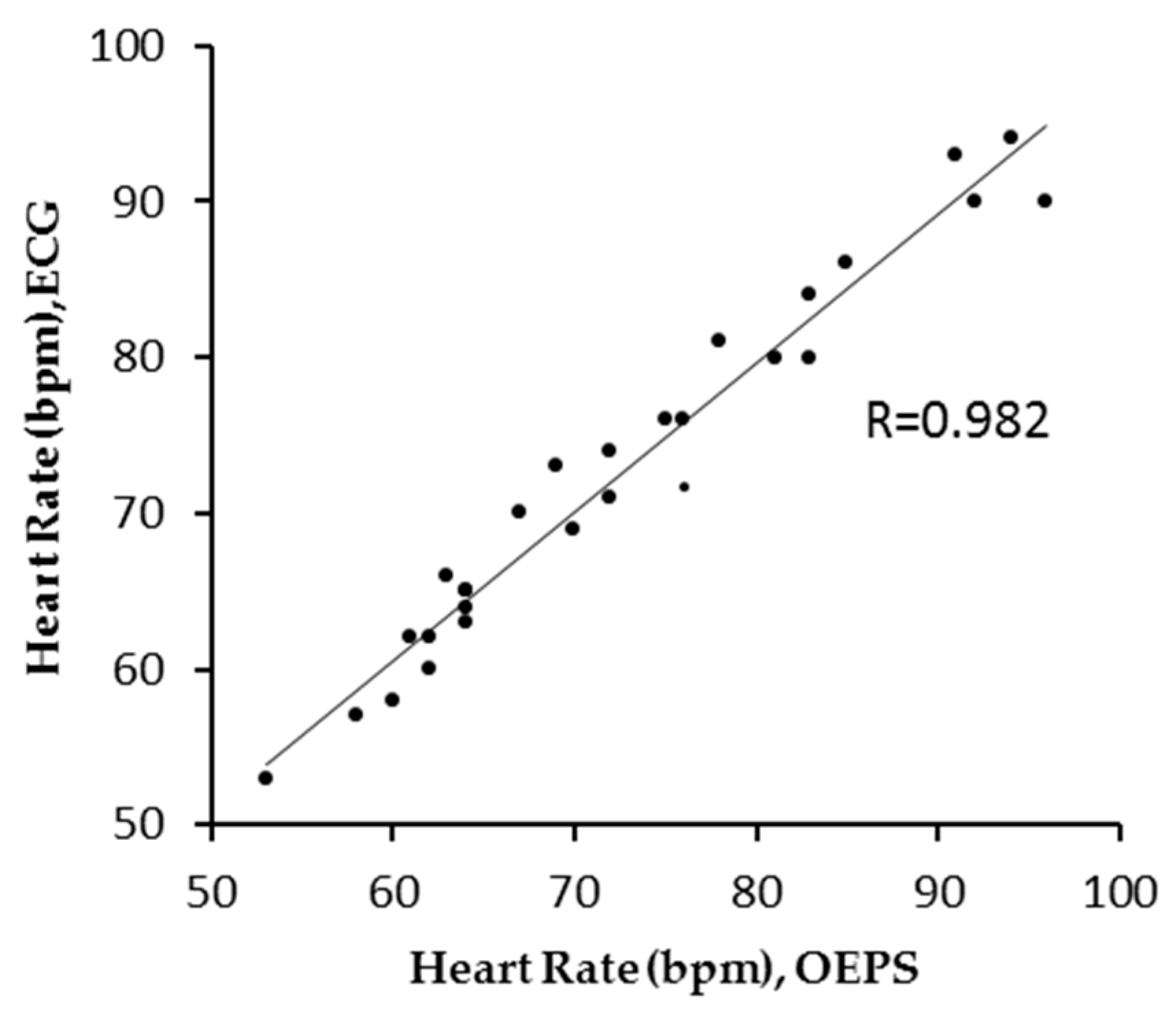
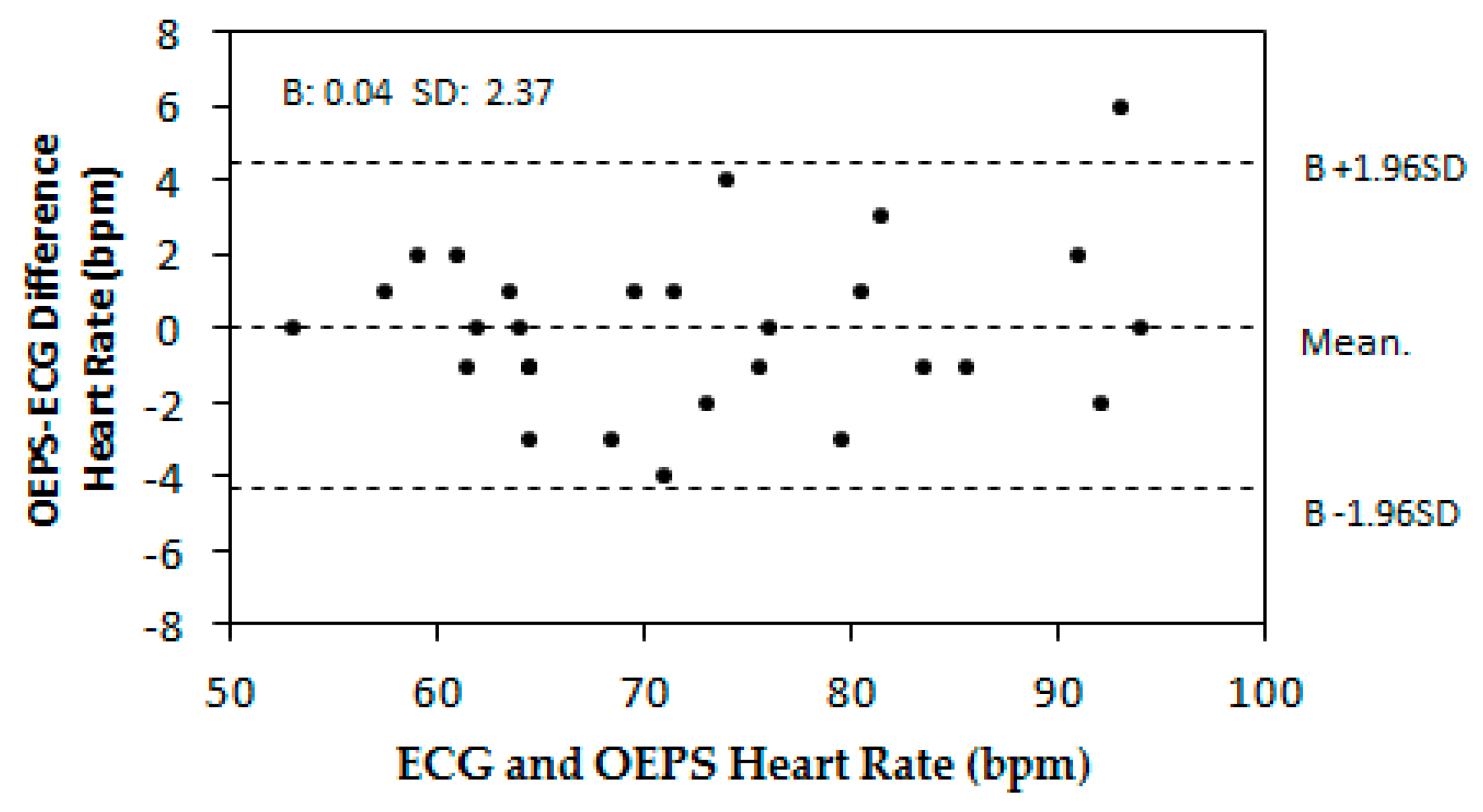
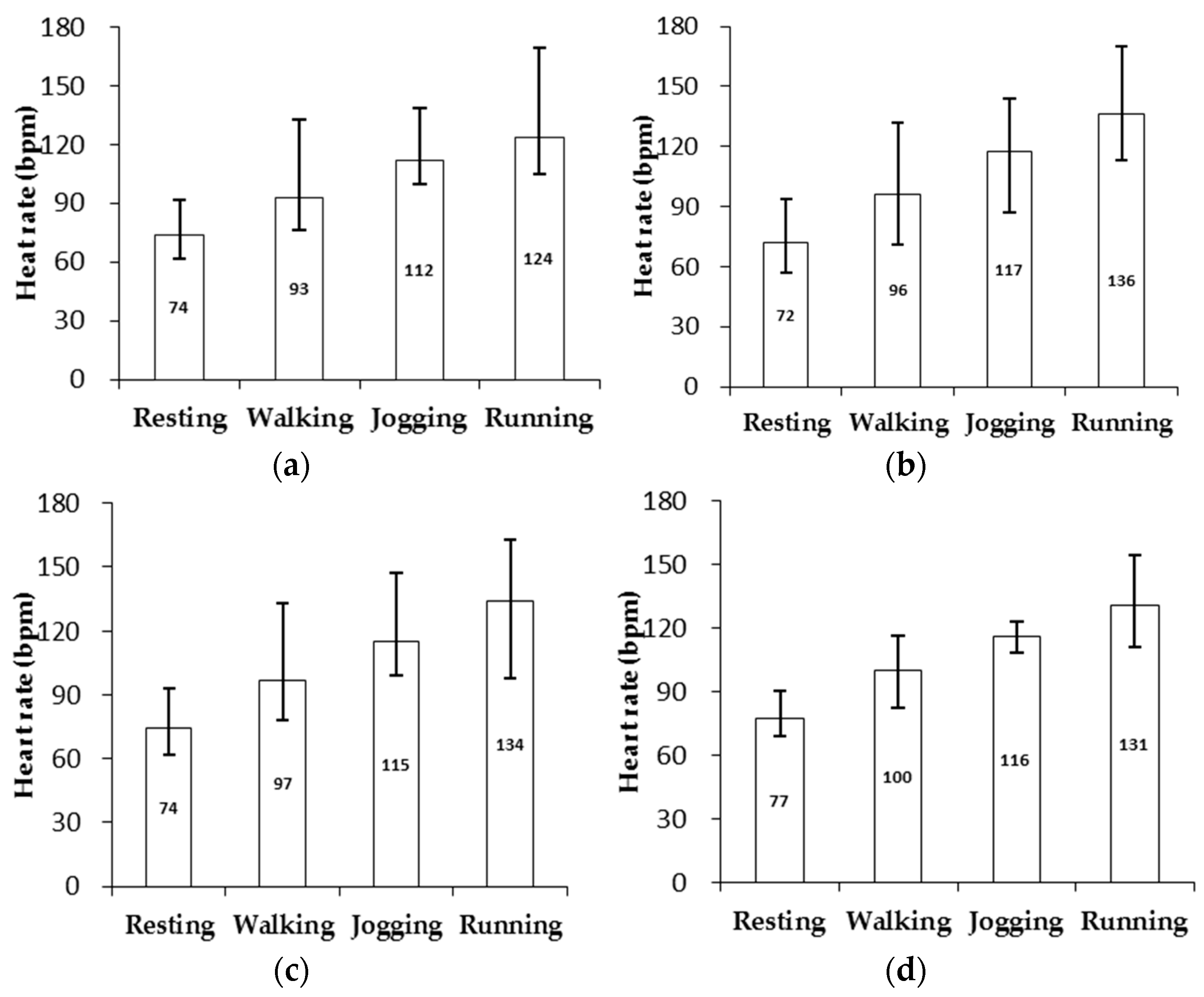
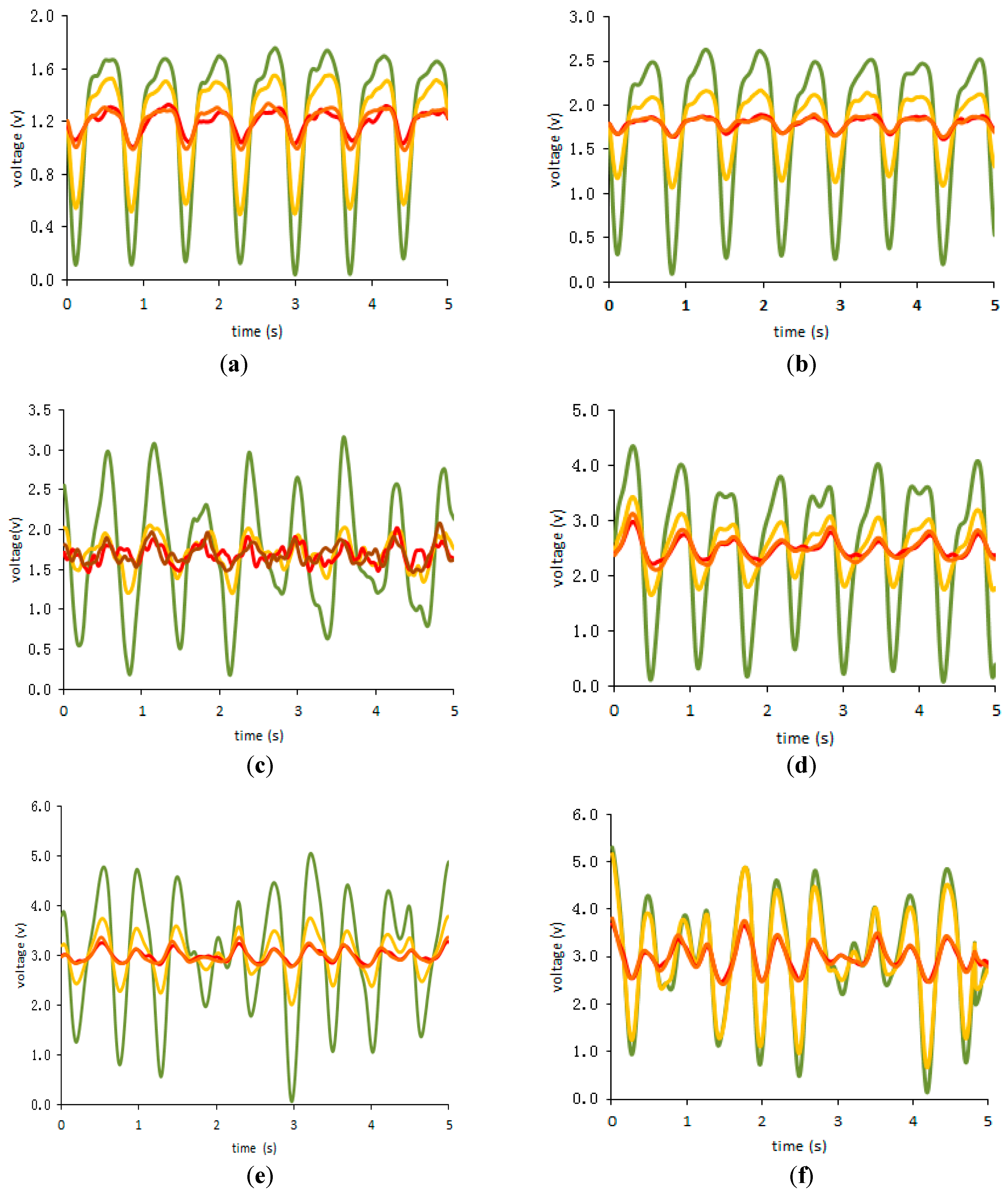
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gertrude, C.; Vincent, J.; Hearing, K. Human skin pigmentation: Melanocytes modulate skin color in response to stress. FASEB J. Rev. 2007, 21, 976–994. [Google Scholar]
- Zonios, G.; Bykowski, J.; Kollias, N. Skin melanin, hemoglobin, and light scattering properties can be quantitatively assessed in vivo using diffuse reflecatance spectroscopy. J. Investig. Dermatol. 2001, 117, 1452–1457. [Google Scholar] [CrossRef] [PubMed]
- Jubran, A.; Tobin, M.J. Reliability of pulse oximetry in titrating supplemental oxygen therapy in ventilator-dependent patients. Chest 1990, 97, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Emery, J.R. Skin pigmentation as an influence on the accuracy of pulse oximetry. J. Perinatol. 1987, 7, 329–330. [Google Scholar] [PubMed]
- Ralston, A.C.; Webb, K.; Runciman, W.B. Potential errors in pulse oximetry. III: Effects of interferences, dyes, dyshaemoglobins and other pigments. Anaesthesia 1991, 5, 291–295. [Google Scholar] [CrossRef]
- Fallow, B.A.; Tarumi, T.; Tanaka, H. Influence of skin type and wavelength on light wave reflectance. J. Clin. Monit. Comput. 2013, 27, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Abbott, A.L.; Doerzaph, Z.; Sykes, K. Evaluation of video magnification for nonintrusive heart rate measurement. In Proceedings of the 2016 IEEE First International Conference Control, Measurement and Instrumentation (CMI), Kolkata, India, 8 January 2016; pp. 494–498. [Google Scholar]
- Chanchal, D.K.; Swarnlata, S. Skin care assessment on the basis of skin hydration, melanin, erythema and sebum at various body sites. Int. J. Pharm. Pharm. Sci. 2011, 3, 209–213. [Google Scholar]
- Fitzpatrick, T.B. The Sun and the skin (Soleil et peau). J. Med. Esthet. 1975, 2, 33–34. [Google Scholar]
- Fitzpatrick, T.B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Spigulis, J.; Gailite, L.; Alexey, L.; Renars, E. Multi-wavelength photoplethysmography for simultaneous recording of skin blood pulsations at different vascular depths. Appl. Opt. 2007, 46, 1754–1759. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.W.; Soter, N.A. Clinical Photomedicine; CRC Press: New York, NY, USA, 1993. [Google Scholar]
- Anderson, R.; Parrish, J. The optics of human skin. J. Investig. Dermatol. 1981, 77, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Azorin-Peris, V. Opto-Physiological Sensor and Method of Assembly. WO 2015056007 A1, 23 April 2015. [Google Scholar]
- Meglinski, I.V.; Matcher, S.J. Computer simulation of the skin reflectance spectra. Comput. Methods Programs Biomed. 2003, 70, 179–186. [Google Scholar] [CrossRef]
- Kurylyak, Y.; Lamonaca, F.; Grimaldi, D. Smartphone-Based photoplethysmogram measurement. In Digital Image and Signal Processing for Measurement Systems; Richard, J.D, Femando, L.P., Eds.; River Publishers: Aalborg, Denmark, 2012; pp. 135–164. [Google Scholar]
- Alzahrani, A.A. A Reflectance Opto-Electronic Patch Sensor for Physiological Monitoring with Motion Artefacts Reduction. Ph.D. Thesis, Loughborough University, Leicestershire, UK, 2016. [Google Scholar]
- Weinschenk, S.W.; Beise, R.D.; Lorenz, J. Heart rate variability (HRV) in deep breathing tests and 5-min short-term recordings: Agreement of ear photoplethysmography with ECG measurements, in 343 subjects. Eur. J. Appl. Physiol. 2016, 116, 1527–1535. [Google Scholar] [CrossRef] [PubMed]

| Type | Color/Description | Sun-burning | VLCS | Region/Area |
|---|---|---|---|---|
| I & II | Light/White | Often/Usually | 1–10 | Caucasus/Europe |
| III | Medium, white to light brown | Rarely | 11–15 | Asia |
| IV | Olive, moderate brown | Rarely | 16–21 | Middle East |
| V | Brown, dark brown | Very rarely | 22–28 | Africa |
| Wavelength (nm) | 500 | 600 | 700 | 800 | 1000 | 1200 |
|---|---|---|---|---|---|---|
| Depth (µm) | 230 | 550 | 750 | 1200 | 1600 | 2200 |
| Skin Type | Resting | Walking: 3 km/h | Jogging: 6 km/h | Running: 9 km/h |
|---|---|---|---|---|
| I & II (n = 11) | 62~92 | 77~133 | 100~139 | 104~170 |
| (1.0~4.3) | (1.7~5.4) | (4.2~11.9) | (4.6~12.8) | |
| III (n = 10) | 57~94 | 71~132 | 87~144 | 113~170 |
| (1.3~3.4) | (1.3~7.2) | (1.9~10.2) | (6.1~20.8) | |
| IV (n = 7) | 62~93 | 78~133 | 99~147 | 98~163 |
| (1.7~5.1) | (1.3~6.3) | (2.4~8.1) | (3.6~21.9 *) | |
| V (n = 5) | 73~90 | 106~116 | 122~123 | 131~154 |
| (3.7~5.9) | (1.9~4.1) | (5.4~10.4) | (3.1~5.3) |
| Skin Type | Resting | Walking: 3 km/h | Jogging: 6 km/h | Running: 9 km/h |
|---|---|---|---|---|
| I & II (n = 11) | 74(2.2) | 93(3.5) | 112(7.1) | 124(8.8) |
| III (n = 10) | 72(2.3) | 96(3.1) | 117(5.0) | 136(13.4) |
| IV (n = 7) | 74(2.6) | 97(4.9) | 115(5.1) | 134(9.4) |
| V (n = 5) | 77(4.8) | 100(3.0) | 116(7.9) | 131(4.2) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, L.; Hu, S.; Alzahrani, A.; Alharbi, S.; Blanos, P. A Multi-Wavelength Opto-Electronic Patch Sensor to Effectively Detect Physiological Changes against Human Skin Types. Biosensors 2017, 7, 22. https://doi.org/10.3390/bios7020022
Yan L, Hu S, Alzahrani A, Alharbi S, Blanos P. A Multi-Wavelength Opto-Electronic Patch Sensor to Effectively Detect Physiological Changes against Human Skin Types. Biosensors. 2017; 7(2):22. https://doi.org/10.3390/bios7020022
Chicago/Turabian StyleYan, Liangwen, Sijung Hu, Abdullah Alzahrani, Samah Alharbi, and Panagiotis Blanos. 2017. "A Multi-Wavelength Opto-Electronic Patch Sensor to Effectively Detect Physiological Changes against Human Skin Types" Biosensors 7, no. 2: 22. https://doi.org/10.3390/bios7020022
APA StyleYan, L., Hu, S., Alzahrani, A., Alharbi, S., & Blanos, P. (2017). A Multi-Wavelength Opto-Electronic Patch Sensor to Effectively Detect Physiological Changes against Human Skin Types. Biosensors, 7(2), 22. https://doi.org/10.3390/bios7020022







