Sensitivity of Electrocardiogram on Electrode-Pair Locations for Wearable Devices: Computational Analysis of Amplitude and Waveform Distortion
Abstract
1. Introduction
2. Materials and Methods
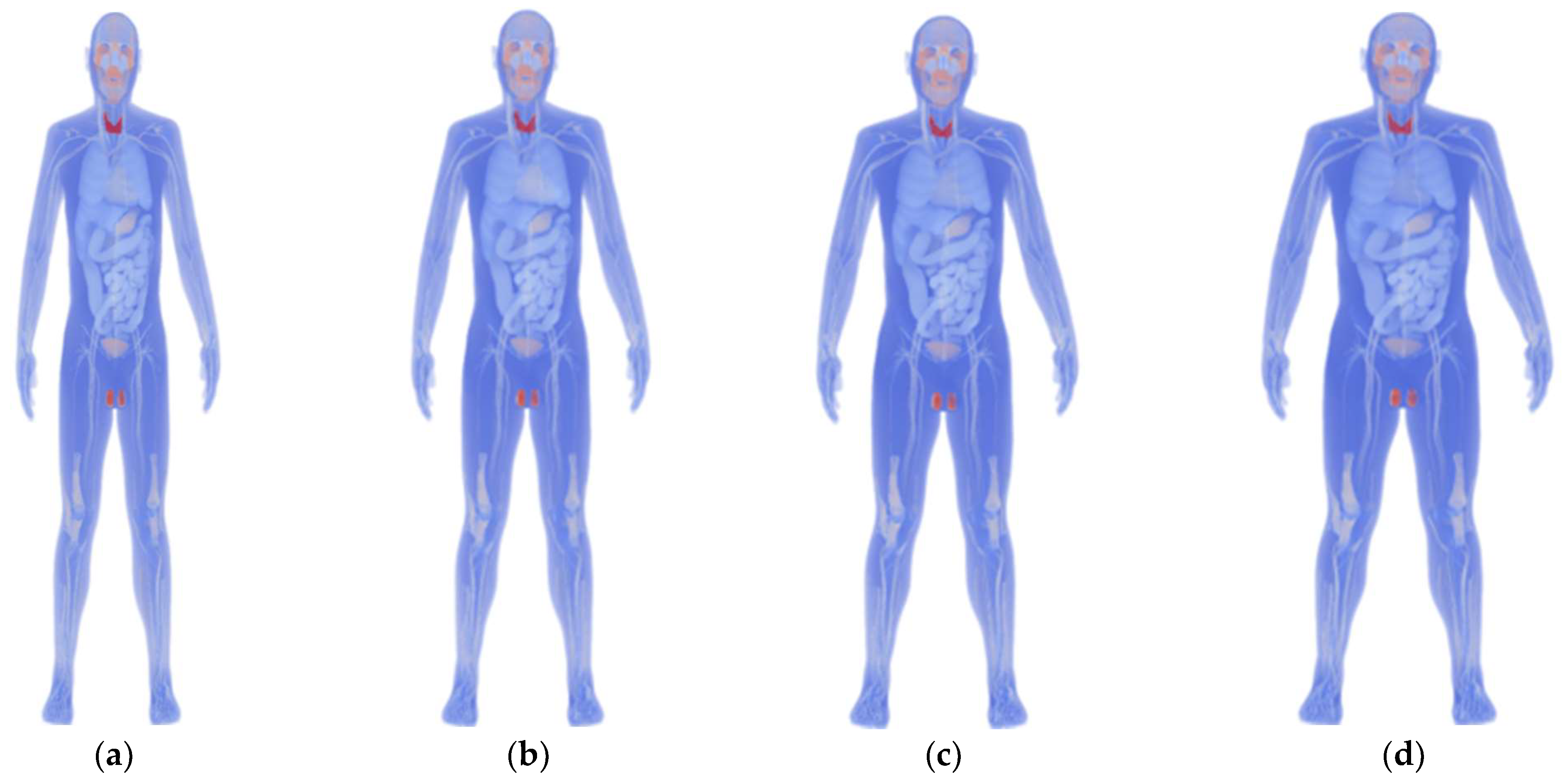
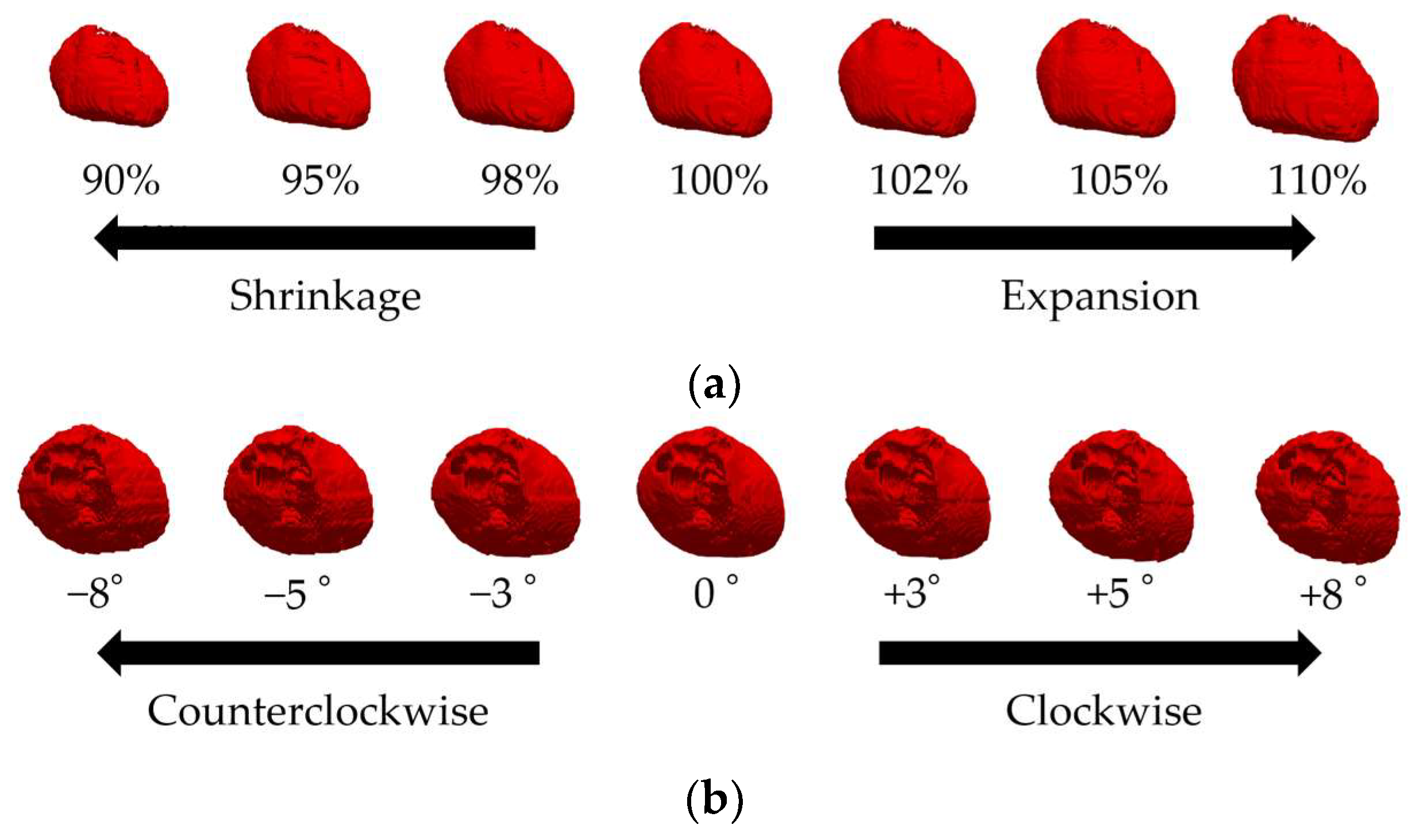
2.1. Anatomical Human Model
2.2. Bipolar Electrode Pairs
2.3. Scalar-Potential Finite-Difference Methods
2.4. Quasi-Static Finite-Difference Time-Domain Methods
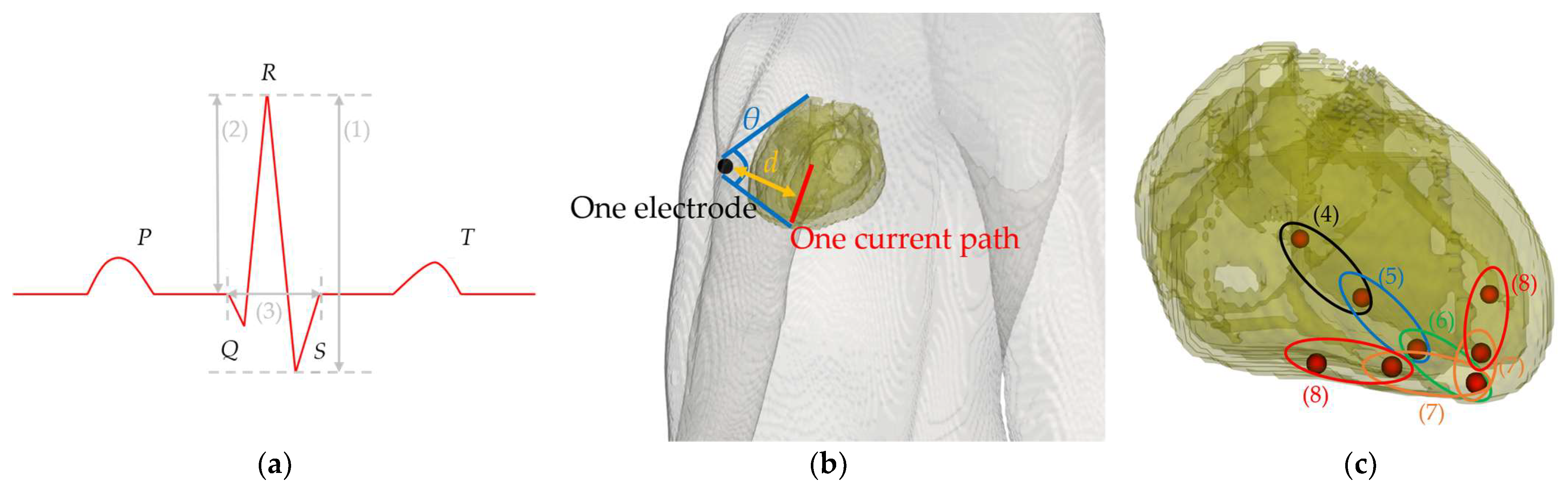
2.5. Modeling Cardiac Potentials and Construction of the ECG
2.6. Dynamic Time Warping Methods
2.7. Multivariate Analysis
2.8. Evaluation Procedure
3. Results
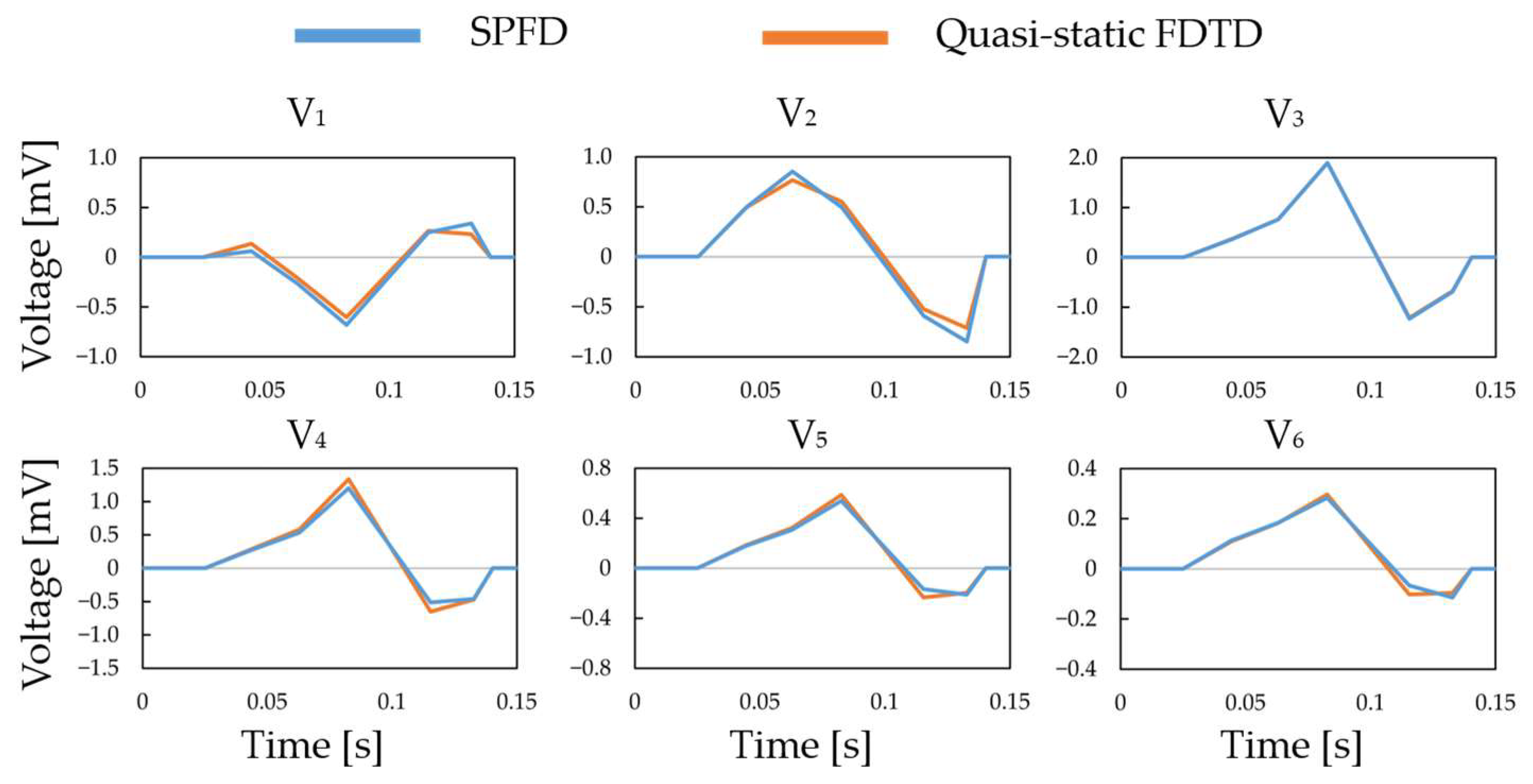
3.1. Verification in the Construction of the ECG Waveform Using the 12-Lead ECG
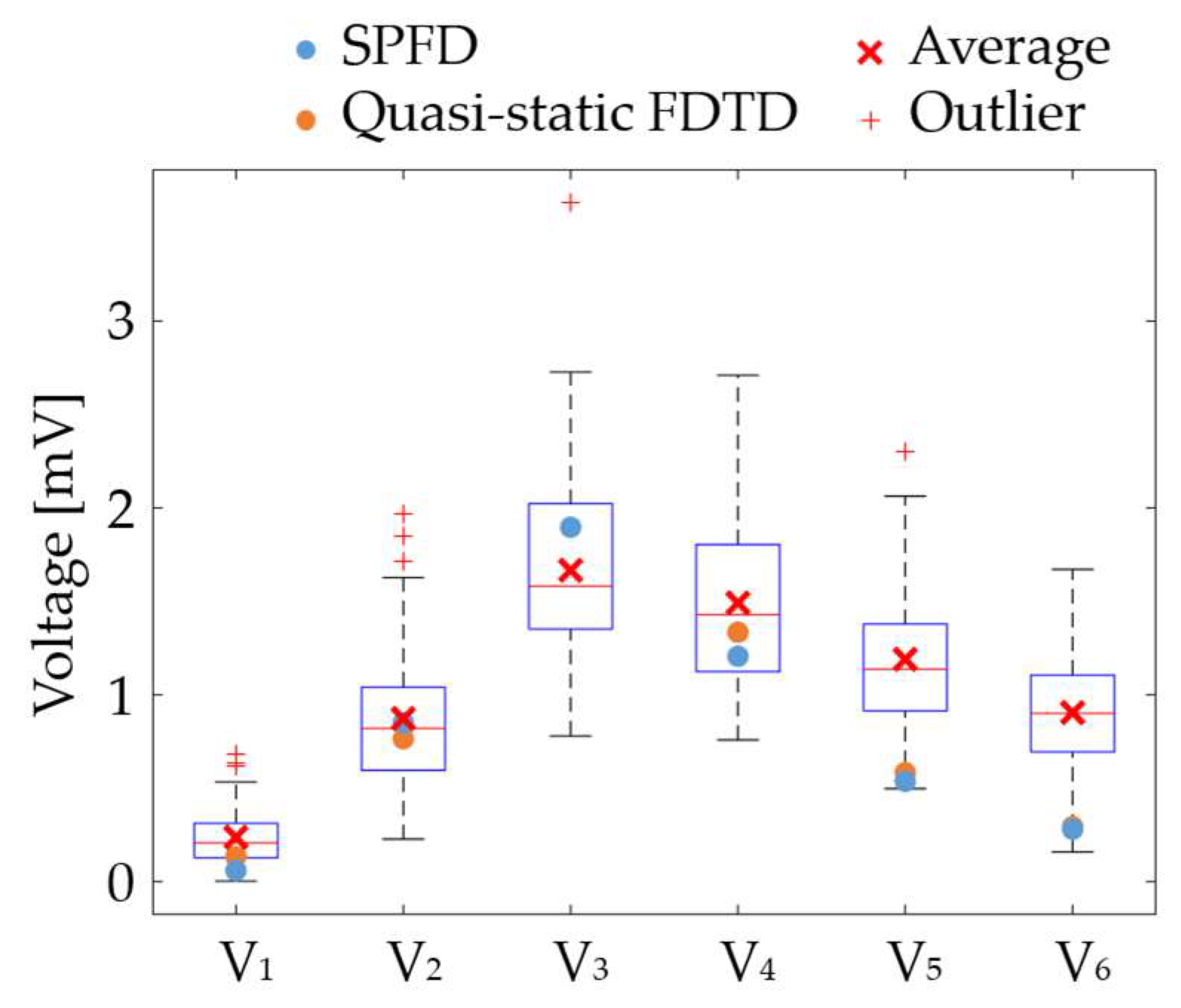
3.2. Validation in the Signal Amplitude of the QRS Wave
3.3. Variation in DTW and Normalized DTW
3.4. Statistical Analysis of the Geometrical Factors
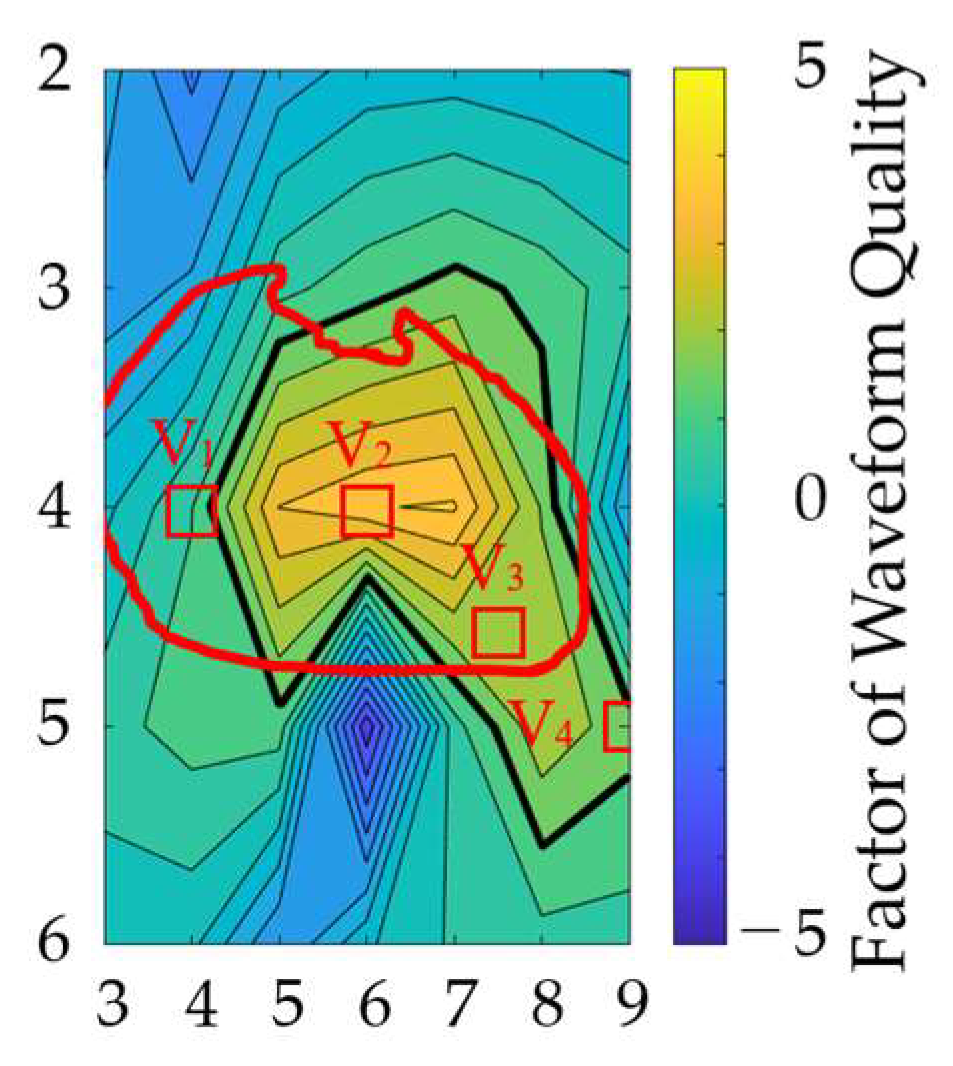
3.5. Optimal Position of the Electrodes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Malmivuo, J.; Plonsey, R. Bioelectromagnetism Principles and Applications of Bioelectric; Oxford University Press: Oxford, UK, 1995; p. 512. [Google Scholar]
- Garcia, T.B. 12-Lead ECG: The Art of Interpretation; Jones & Bartlett Learning: Burlington, MA, USA, 2013. [Google Scholar]
- Montague, T.J.; Smith, E.R.; Cameron, D.A.; Rautaharju, P.M.; Klassen, G.A.; Felmington, C.S.; Horacek, B.M. Isointegral Analysis of Body Surface Maps: Surface Distribution and Temporal Variability in Normal Subjects. Circulation 1981, 63, 1166–1172. [Google Scholar] [CrossRef]
- Bergquist, J.; Rupp, L.; Zenger, B.; Brundage, J.; Busatto, A.; MacLeod, R.S. Body Surface Potential Mapping: Contemporary Applications and Future Perspectives. Hearts 2021, 2, 514–542. [Google Scholar] [CrossRef]
- Rahul, J.; Sora, M.; Sharma, L.D. A Novel and Lightweight P, QRS, and T Peaks Detector Using Adaptive Thresholding and Template Waveform. Comput. Biol. Med. 2021, 132, 104307. [Google Scholar] [CrossRef] [PubMed]
- Boineau, J.P.; Spach, M.S. The Relationship between the Electrocardiogram and the Electrical Activity of the Heart. J. Electrocardiol. 1968, 1, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterom, A.; Hoekema, R.; Uijen, G.J.H. Geometrical Factors Affecting the Interindividual Variability of the ECG and the VCG. J. Electrocardiol. 2000, 33, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Mincholé, A.; Zacur, E.; Ariga, R.; Grau, V.; Rodriguez, B. MRI-Based Computational Torso/Biventricular Multiscale Models to Investigate the Impact of Anatomical Variability on the ECG QRS Complex. Front. Physiol. 2019, 10, 1103. [Google Scholar] [CrossRef] [PubMed]
- Huiskamp, G.J.M.; van Oosterom, A. Heart Position and Orientation in Forward and Inverse Electrocardiography. Med. Biol. Eng. Comput. 1992, 30, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Sameni, R.; Clifford, G.D. A Review of Fetal ECG Signal Processing Issues and Promising Directions. Open Pacing Electrophysiol. Ther. J. 2010, 3, 4–20. [Google Scholar] [CrossRef]
- Sharma, M.; Dhiman, H.S.; Acharya, U.R. Automatic Identification of Insomnia Using Optimal Antisymmetric Biorthogonal Wavelet Filter Bank with ECG Signals. Comput. Biol. Med. 2021, 131, 104246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Ma, C.; Sun, Y.; Fan, G.; Song, F.; Feng, Y.; Zhang, G. Global Hybrid Multi-Scale Convolutional Network for Accurate and Robust Detection of Atrial Fibrillation Using Single-Lead ECG Recordings. Comput. Biol. Med. 2021, 139, 104880. [Google Scholar] [CrossRef]
- Kania, M.; Rix, H.; Fereniec, M.; Zavala-Fernandez, H.; Janusek, D.; Mroczka, T.; Stix, G.; Maniewski, R. The Effect of Precordial Lead Displacement on ECG Morphology. Med. Biol. Eng. Comput. 2014, 52, 109–119. [Google Scholar] [CrossRef]
- Georgiou, K.; Larentzakis, A.V.; Khamis, N.N.; Alsuhaibani, G.I.; Alaska, Y.A.; Giallafos, E.J. Can Wearable Devices Accurately Measure Heart Rate Variability? A Systematic Review. Folia Med. 2018, 60, 7–20. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Sato, R.; Takagahara, K.; Ishihara, T.; Watanabe, K.; Togo, H. Validation of Wearable Device Consisting of a Smart Shirt with Built-In Bioelectrodes and a Wireless Transmitter for Heart Rate Monitoring in Light to Moderate Physical Work. Sensors 2022, 22, 9241. [Google Scholar] [CrossRef]
- Puurtinen, M.; Viik, J.; Hyttinen, J. Best Electrode Locations for a Small Bipolar ECG Device: Signal Strength Analysis of Clinical Data. Ann. Biomed. Eng. 2009, 37, 331–336. [Google Scholar] [CrossRef]
- Noh, H.W.; Jang, Y.; Lee, I.B.; Song, Y.; Jeong, J.W.; Lee, S. A Preliminary Study of the Effect of Electrode Placement in Order to Define a Suitable Location for Two Electrodes and Obtain Sufficiently Reliable ECG Signals When Monitoring with Wireless System. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 2124–2127. [Google Scholar] [CrossRef]
- Geneser, S.E.; Kirby, R.M.; MacLeod, R.S. Application of Stochastic Finite Element Methods to Study the Sensitivity of ECG Forward Modeling to Organ Conductivity. IEEE Trans. Biomed. Eng. 2008, 55, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Farina, D.; Jiang, Y.; Dössel, O. Acceleration of FEM-Based Transfer Matrix Computation for Forward and Inverse Problems of Electrocardiography. Med. Biol. Eng. Comput. 2009, 47, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Fischer, G.; Tilg, B.; Modre, R.; Huiskamp, G.J.M.; Fetzer, J.; Rucker, W.; Wach, P. Bidomain Model Based BEM-FEM Coupling Formulation for Anisotropic Cardiac Tissue. Ann. Biomed. Eng. 2000, 28, 1229–1243. [Google Scholar] [CrossRef] [PubMed]
- Nakane, T.; Ito, T.; Matsuura, N.; Togo, H.; Hirata, A. Forward Electrocardiogram Modeling by Small Dipoles Based on Whole-Body Electric Field Analysis. IEEE Access 2019, 7, 123463–123472. [Google Scholar] [CrossRef]
- Nakano, Y.; Rashed, E.A.; Nakane, T.; Laakso, I.; Hirata, A. Ecg Localization Method Based on Volume Conductor Model and Kalman Filtering. Sensors 2021, 21, 4275. [Google Scholar] [CrossRef] [PubMed]
- Segars, W.P.; Mahesh, M.; Beck, T.J.; Frey, E.C.; Tsui, B.M.W. Realistic CT Simulation Using the 4D XCAT Phantom. Med. Phys. 2008, 35, 3800–3808. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, S.; Lau, R.W.; Gabriel, C. The Dielectric Properties of Biological Tissues: III. Parametric Models for the Dielectric Spectrum of Tissues. Phys. Med. Biol. 1996, 41, 2271–2293. [Google Scholar] [CrossRef]
- Dimbylow, P.J. Induced Current Densities from Low-Frequency Magnetic Fields in a 2 Mm Resolution, Anatomically Realistic Model of the Body. Phys. Med. Biol. 1998, 43, 221–230. [Google Scholar] [CrossRef]
- Carlsson, M.; Cain, P.; Holmqvist, C.; Stahlberg, F.; Lundback, S.; Arheden, H. Total Heart Volume Variation throughout the Cardiac Cycle in Humans. Am. J. Physiol. Circ. Physiol. 2004, 287, H243–H250. [Google Scholar] [CrossRef]
- Kawai, N.; Sotobata, I.; Noda, S.; Okada, M.; Kondo, T.; Yokota, M.; Yamauchi, K.; Tsuzuki, J. Correlation between the Direction of the Interventricular Septum Estimated with Transmission Computed Tomography and the Initial QRS Vectors. J. Electrocardiol. 1984, 17, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, A.V.; Savard, P. Forward Problem of Electrocardiography: Construction of Human Torso Models and Field Calculations Using Finite Element Method. Med. Biol. Eng. Comput. 1994, 32, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Dawson, T.W. Analytic Validation of A Three-Dimensional Solar-Potential Finite- Difference Code for Low-Frequency Magnetic Induction. Appl. Comput. Electromagn. Soc. J. 2022, 11, 72–81. [Google Scholar]
- Hirata, A.; Takano, Y.; Nagai, T. Quasi-Static FDTD Method for Dosimetry in Human Due to Contact Current. IEICE Trans. Electron. 2010, E93-C, 60–65. [Google Scholar] [CrossRef]
- Rajbhandary, P.L.; Nallathambi, G.; Selvaraj, N.; Tran, T.; Colliou, O. ECG Signal Quality Assessments of a Small Bipolar Single-Lead Wearable Patch Sensor. Cardiovasc. Eng. Technol. 2022, 13, 783–796. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, B.S.; Bera, D.; Bopardikar, A.S.; Narayanan, R. Cardiac Arrhythmia Detection Using Dynamic Time Warping of ECG Beats in E-Healthcare Systems. In Proceedings of the 2011 IEEE International Symposium on a World of Wireless, Mobile and Multimedia Networks, Lucca, Italy, 20–24 June 2011; pp. 1–6. [Google Scholar] [CrossRef]
- Yang, H.; Wei, Z. Arrhythmia Recognition and Classification Using Combined Parametric and Visual Pattern Features of ECG Morphology. IEEE Access 2020, 8, 47103–47117. [Google Scholar] [CrossRef]
- Müller, M. Information Retrieval for Music and Motion; Springer: Berlin/Heidelberg, Germany, 2007; pp. 1–313. [Google Scholar] [CrossRef]
- Senin, P. Dynamic Time Warping Algorithm Review. Science 2008, 2007, 1–23. [Google Scholar]
- Rencher, A.C.; Christensen, W.F. Methods of Multivariate Analysis; Wiley Online Library: Hoboken, NJ, USA, 2012; ISBN 9781118391686. [Google Scholar]
- Wagner, P.; Strodthoff, N.; Bousseljot, R.D.; Kreiseler, D.; Lunze, F.I.; Samek, W.; Schaeffter, T. PTB-XL, a Large Publicly Available Electrocardiography Dataset. Sci. Data 2020, 7, 154. [Google Scholar] [CrossRef] [PubMed]
- Shiroma, E.J.; Lee, I.-M. Physical Activity and Cardiovascular Health. Circulation 2010, 122, 743–752. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanjo, K.; Hebiguchi, K.; Tang, C.; Rashed, E.A.; Kodera, S.; Togo, H.; Hirata, A. Sensitivity of Electrocardiogram on Electrode-Pair Locations for Wearable Devices: Computational Analysis of Amplitude and Waveform Distortion. Biosensors 2024, 14, 153. https://doi.org/10.3390/bios14030153
Sanjo K, Hebiguchi K, Tang C, Rashed EA, Kodera S, Togo H, Hirata A. Sensitivity of Electrocardiogram on Electrode-Pair Locations for Wearable Devices: Computational Analysis of Amplitude and Waveform Distortion. Biosensors. 2024; 14(3):153. https://doi.org/10.3390/bios14030153
Chicago/Turabian StyleSanjo, Kiyoto, Kazuki Hebiguchi, Cheng Tang, Essam A. Rashed, Sachiko Kodera, Hiroyoshi Togo, and Akimasa Hirata. 2024. "Sensitivity of Electrocardiogram on Electrode-Pair Locations for Wearable Devices: Computational Analysis of Amplitude and Waveform Distortion" Biosensors 14, no. 3: 153. https://doi.org/10.3390/bios14030153
APA StyleSanjo, K., Hebiguchi, K., Tang, C., Rashed, E. A., Kodera, S., Togo, H., & Hirata, A. (2024). Sensitivity of Electrocardiogram on Electrode-Pair Locations for Wearable Devices: Computational Analysis of Amplitude and Waveform Distortion. Biosensors, 14(3), 153. https://doi.org/10.3390/bios14030153






