Nanoisland SERS-Substrates for Specific Detection and Quantification of Influenza A Virus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Aptamer Assembly
2.3. Viruses
2.4. SERS-Substrates Production
2.5. Scanning Electron Microscopy
2.6. Spectroscopy of Surface Plasmon Resonance
2.7. Atomic Force Microscopy
2.8. Initial Setup of Aptasensor
2.9. Initial Setup of Aptasensor with Displacement of the Buffer
2.10. Novel Setup of the Aptasensor
3. Results
3.1. IAV Distorts the Nanostructured Surface of the Sensor
3.2. The Distortion of the Nanostructured Surface Is Caused by the Buffer Solution
3.3. Optimization of Stability of Substrate Nanostructure
3.4. Optimization of the Sensor Setup
3.5. Analytical Performance of the New Sensor
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chan, K.-H.; To, K.K.W.; Chan, J.F.W.; Li, C.P.Y.; Chen, H.; Yuen, K.-Y. Analytical sensitivity of seven point-of-care influenza virus detection tests and two molecular tests for detection of avian origin H7N9 and swine origin H3N2 variant influenza A viruses. J. Clin. Microbiol. 2013, 51, 3160–3161. [Google Scholar] [CrossRef] [PubMed]
- Instructions for Use of QuickVue SARS Antigen Test. Available online: https://www.fda.gov/media/144668/download (accessed on 3 August 2023).
- Kim, H.N.; Yoon, S.Y.; Lim, C.S.; Yoon, J. Comparison of three molecular diagnostic assays for SARS-CoV-2 detection: Evaluation of analytical sensitivity and clinical performance. Clin. Lab. Anal. 2022, 36, e24242. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, B.; Larsson, C.; Zweygberg, B.W. Simultaneous detection and typing of influenza viruses A and B by a nested reverse transcription-PCR: Comparison to virus isolation and antigen detection by immunofluorescence and optical immunoassay (FLU OIA). J. Clin. Microbiol. 2001, 39, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Han, Y.; Zhang, R.; Zhang, R.; Li, J. Comparison of analytical sensitivity of SARS-CoV-2 molecular detection kits. Int. J. Infect. Dis. 2021, 111, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Xie, Z.; Xie, L.; Liu, J.; Xie, Z.; Deng, X.; Huang, L.; Huang, J.; Zeng, T.; Khan, M.I. Reverse-transcription, loop-mediated isothermal amplification assay for the sensitive and rapid detection of H10 subtype avian influenza viruses. Virol. J. 2015, 12, 145. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Cui, J.; Huang, L.; Du, B.; Chen, L.; Xue, G.; Li, S.; Zhang, W.; Zhao, L.; Sun, Y.; et al. Rapid and visual detection of 2019 novel coronavirus (SARS-CoV-2) by a reverse transcription loop-mediated isothermal amplification assay. Clin. Microbiol. Infect. 2020, 26, 773–779. [Google Scholar] [CrossRef]
- Elskens, J.P.; Elskens, J.M.; Madder, A. Chemical modification of aptamers for increased binding affinity in diagnostic applications: Current status and future prospects. Int. J. Mol. Sci. 2020, 21, 4522. [Google Scholar] [CrossRef]
- Chan, K.Y.; Kinghorn, A.B.; Hollenstein, M.; Tanner, J.A. Chemical modifications for a next generation of nucleic acid aptamers. ChemBioChem 2022, 23, e202200006. [Google Scholar] [CrossRef]
- Amini, R.; Zhang, Z.; Li, J.; Gu, J.; Brennan, J.D.; Li, Y. Aptamers for SARS-CoV-2: Isolation, characterization, and diagnostic and therapeutic developments. Anal. Sens. 2022, 2, e202200012. [Google Scholar] [CrossRef]
- Zhang, Y.; Juhas, M.; Kwok, C.K. Aptamers targeting SARS-CoV-2: A promising tool to fight against COVID-19. Trends Biotechnol. 2023, 41, 528–544. [Google Scholar] [CrossRef]
- Zavyalova, E.; Kopylov, A. Aptamers to hemagglutinin: A novel tool for influenza virus recognition and neutralization. Curr. Pharm. Des. 2016, 22, 4835–4853. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Kim, J.; Noh, S.; Sohn, H.; Lee, T. Recent development of aptasensor for influenza virus detection. BioChip J. 2020, 14, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Bala, J.; Chinnapaiyan, S.; Dutta, R.K.; Unwalla, H. Aptamers in HIV research diagnosis and therapy. RNA Biol. 2018, 15, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Gruenke, P.R.; Aneja, R.; Welbourn, S.; Ukah, O.B.; Sarafianos, S.G.; Burke, D.H.; Lange, M.J. Selection and identification of an RNA aptamer that specifically binds the HIV-1 capsid lattice and inhibits viral replication. Nucleic Acids Res. 2022, 50, 1701–1717. [Google Scholar] [CrossRef] [PubMed]
- Mirian, M.; Khanahmad, H.; Darzi, L.; Salehi, M.; Sadeghi-Aliabadi, H. Oligonucleotide aptamers: Potential novel molecules against viral hepatitis. Res. Pharm. Sci. 2017, 12, 88–98. [Google Scholar] [PubMed]
- Torres-Vázquez, B.; de Lucas, A.M.; García-Crespo, C.; García-Martín, J.A.; Fragoso, A.; Fernández-Algar, M.; Perales, C.; Domingo, E.; Moreno, M.; Briones, C. In vitro selection of high affinity DNA and RNA aptamers that detect hepatitis C virus core protein of genotypes 1 to 4 and inhibit virus production in cell culture. J. Mol. Biol. 2022, 434, 167501. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Le, C.; Tyrrell, D.L.; Le, X.C.; Li, X.F. Aptamer binding assay for the E antigen of hepatitis B using modified aptamers with G-quadruplex structures. Anal. Chem. 2020, 92, 6495–6501. [Google Scholar] [CrossRef]
- Lou, B.; Liu, Y.; Shi, M.; Chen, J.; Li, K.; Tan, Y.; Chen, L.; Wu, Y.; Wang, T.; Liu, X.; et al. Aptamer-based biosensors for virus protein detection. Trends Anal. Chem. 2022, 157, 116738. [Google Scholar] [CrossRef]
- Divya; Dkhar, D.S.; Kumari, R.; Mahapatra, S.; Kumar, R.; Chandra, P. Ultrasensitive aptasensors for the detection of viruses based on opto-electrochemical readout systems. Biosensors 2022, 12, 81. [Google Scholar] [CrossRef]
- Kukushkin, V.; Ambartsumyan, O.; Subekin, A.; Astrakhantseva, A.; Gushchin, V.; Nikonova, A.; Dorofeeva, A.; Zverev, V.; Keshek, A.; Meshcheryakova, N.; et al. Multiplex lithographic SERS aptasensor for detection of several respiratory viruses in one pot. Int. J. Mol. Sci. 2023, 24, 8081. [Google Scholar] [CrossRef]
- Kukushkin, V.; Kristavchuk, O.; Andreev, E.; Meshcheryakova, N.; Zaborova, O.; Gambaryan, A.; Nechaev, A.; Zavyalova, E. Aptamer-coated track-etched membranes with a nanostructured silver layer for single virus detection in biological fluids. Front. Bioeng. Biotechnol. 2023, 10, 1076749. [Google Scholar] [CrossRef] [PubMed]
- Kukushkin, V.I.; Kristavchuk, O.V.; Zhdanov, G.A.; Keshek, A.K.; Gambaryan, A.S.; Andreev, Y.V.; Nechaev, A.N.; Zavyalova, E.G. Aptasensors based on track-etched membranes coated with a nanostructured silver layer for influenza A and B virus detection. Bull. Russ. Acad. Sci. Phys. 2023, 87, 172–177. [Google Scholar] [CrossRef]
- Zhdanov, G.; Nyhrikova, E.; Meshcheryakova, N.; Kristavchuk, O.; Akhmetova, A.; Andreev, E.; Rudakova, E.; Gambaryan, A.; Yaminsky, I.; Aralov, A.; et al. A combination of membrane filtration and Raman-active DNA ligand greatly enhances sensitivity of SERS-based aptasensors for influenza A virus. Front. Chem. 2022, 10, 937180. [Google Scholar] [CrossRef] [PubMed]
- Kukushkin, V.I.; Ivanov, N.M.; Novoseltseva, A.A.; Gambaryan, A.S.; Yaminsky, I.V.; Kopylov, A.M.; Zavyalova, E.G. Highly sensitive detection of influenza virus with SERS aptasensor. PLoS ONE 2019, 14, e0216247. [Google Scholar] [CrossRef] [PubMed]
- Subekin, A.; Alieva, R.; Kukushkin, V.; Oleynikov, I.; Zavyalova, E. Rapid SERS detection of botulinum neurotoxin type A. Nanomaterials 2023, 13, 2531. [Google Scholar] [CrossRef] [PubMed]
- Shiratori, I.; Akitomi, J.; Boltz, D.A.; Horii, K.; Furuichi, M.; Waga, I. Selection of DNA aptamers that bind to influenza A viruses with high affinity and broad subtype specificity. Biochem. Biophys. Res. Commun. 2014, 443, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Killian, M.L. Hemagglutination assay for influenza virus. Methods Mol. Biol. 2014, 1161, 3–9. [Google Scholar]
- Noda, T. Native morphology of influenza virions. Front. Microbiol. 2012, 2, 269. [Google Scholar] [CrossRef]
- Touizer, E.; Sieben, C.; Henriques, R.; Marsh, M.; Laine, R.F. Application of super-resolution and advanced quantitative microscopy to the spatio-temporal analysis of influenza virus replication. Viruses 2021, 13, 233. [Google Scholar] [CrossRef]
- Colas, F.; Barchiesi, D.; Kessentini, S.; Toury, T.; Lamy de la Chapelle, M. Comparison of adhesion layers of gold on silicate glasses for SERS detection. J. Opt. 2015, 17, 114010. [Google Scholar] [CrossRef]
- Yilmaz, A.; Yılmaz, Ü.; Çakır, M.T. Silver films on glass with very thin chromium adherence interlayers; reflectance and adherence at film-glass interface. Optik 2023, 274, 170530. [Google Scholar] [CrossRef]
- Davies, M.P.; McKinney, C.S.; Serrano, J.M.; Mullen, T.J.; Stagon, S.P. Effects of adhesion layer on Ag nanorod growth mode and morphology using glancing angle physical vapor deposition. Appl. Phys. Lett. 2017, 110, 053109. [Google Scholar] [CrossRef]
- Moskovits, M. Surface-enhanced spectroscopy. Rev. Mod. Phys. 1985, 57, 783. [Google Scholar] [CrossRef]
- Chen, X.F.; Zhao, X.; Yang, Z. Aptasensors for the detection of infectious pathogens: Design strategies and point-of-care testing. Microchim. Acta 2022, 189, 443. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yuan, J.; Khan, I.M.; Zhang, L.; Ma, P.; Wang, Z. Research progress of aptasensor technology in the detection of foodborne pathogens. Food Control 2023, 153, 109891. [Google Scholar] [CrossRef]
- Trunzo, N.E.; Hong, K.L. Recent progress in the identification of aptamers against bacterial origins and their diagnostic applications. Int. J. Mol. Sci. 2020, 21, 5074. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Gao, S.; Wu, J.; Hu, X. Recent advances in aptamer-based biosensors for detection of Pseudomonas aeruginosa. Front. Microbiol. 2020, 11, 605229. [Google Scholar] [CrossRef]
- Klimstra, W.B.; Tilston-Lunel, N.L.; Nambulli, S.; Boslett, J.; McMillen, C.M.; Gilliland, T.; Dunn, M.D.; Sun, C.; Wheeler, S.E.; Wells, A.; et al. SARS-CoV-2 growth, furin-cleavage-site adaptation and neutralization using serum from acutely infected hospitalized COVID-19 patients. J. Gen. Virol. 2020, 101, 1156–1169. [Google Scholar] [CrossRef]
- Kramberger, P.; Ciringer, M.; Štrancar, A.; Peterka, M. Evaluation of nanoparticle tracking analysis for total virus particle determination. Virol. J. 2012, 9, 265. [Google Scholar] [CrossRef]
- McCormick, W.; Mermel, L.A. The basic reproductive number and particle-to-plaque ratio: Comparison of these two parameters of viral infectivity. Virol. J. 2021, 18, 92. [Google Scholar] [CrossRef]
- Yeh, Y.-T.; Gulino, K.; Zhang, Y.H.; Sabestien, A.; Chou, T.-W.; Zhou, B.; Lin, Z.; Albert, I.; Lu, H.; Swaminathan, V.; et al. A rapid and label-free platform for virus capture and identification from clinical samples. Proc. Nat. Acad. Sci. USA 2020, 117, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Shanmukh, S.; Jones, L.; Driskell, J.; Zhao, Y.; Dluhy, R.; Tripp, R.A. Rapid and sensitive detection of respiratory virus molecular signatures using a silver nanorod array SERS substrate. Nano Lett. 2006, 6, 2630–2636. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Park, S.G.; Choi, N.; Moon, J.I.; Dang, H.; Das, A.; Lee, S.; Kim, D.G.; Chen, L.; Choo, J. SERS imaging-based aptasensor for ultrasensitive and reproducible detection of influenza virus A. Biosens. Bioelectron. 2020, 167, 112496. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Park, S.K.; Joung, Y.; Kang, T.; Lee, M.K.; Choo, J. SERS-based dual-mode DNA aptasensors for rapid classification of SARS-CoV-2 and influenza A/H1N1 infection. Sens. Actuators B Chem. 2022, 355, 131324. [Google Scholar] [CrossRef]
- Moon, J.; Yi, S.Y.; Hwang, A.; Eom, G.; Sim, J.; Jeong, J.; Lim, E.-K.; Chung, B.H.; Kim, B.; Jung, J.; et al. Facile and sensitive detection of influenza viruses using SERS antibody probes. RSC Adv. 2016, 6, 84415–84419. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Wu, C.-Y.; Li, T.; Hsiao, P.-W.; Chang, D.-K. A rapid and sensitive early diagnosis of influenza virus subtype via surface enhanced Raman scattering. J. Biosens. Bioelectron. 2014, 5, 2. [Google Scholar]
- Lee, J.H.; Kim, B.C.; Oh, B.K.; Choi, J.W. Rapid and sensitive determination of HIV-1 virus based on surface enhanced Raman spectroscopy. J. Biomed. Nanotechnol. 2015, 11, 2223–2230. [Google Scholar] [CrossRef]

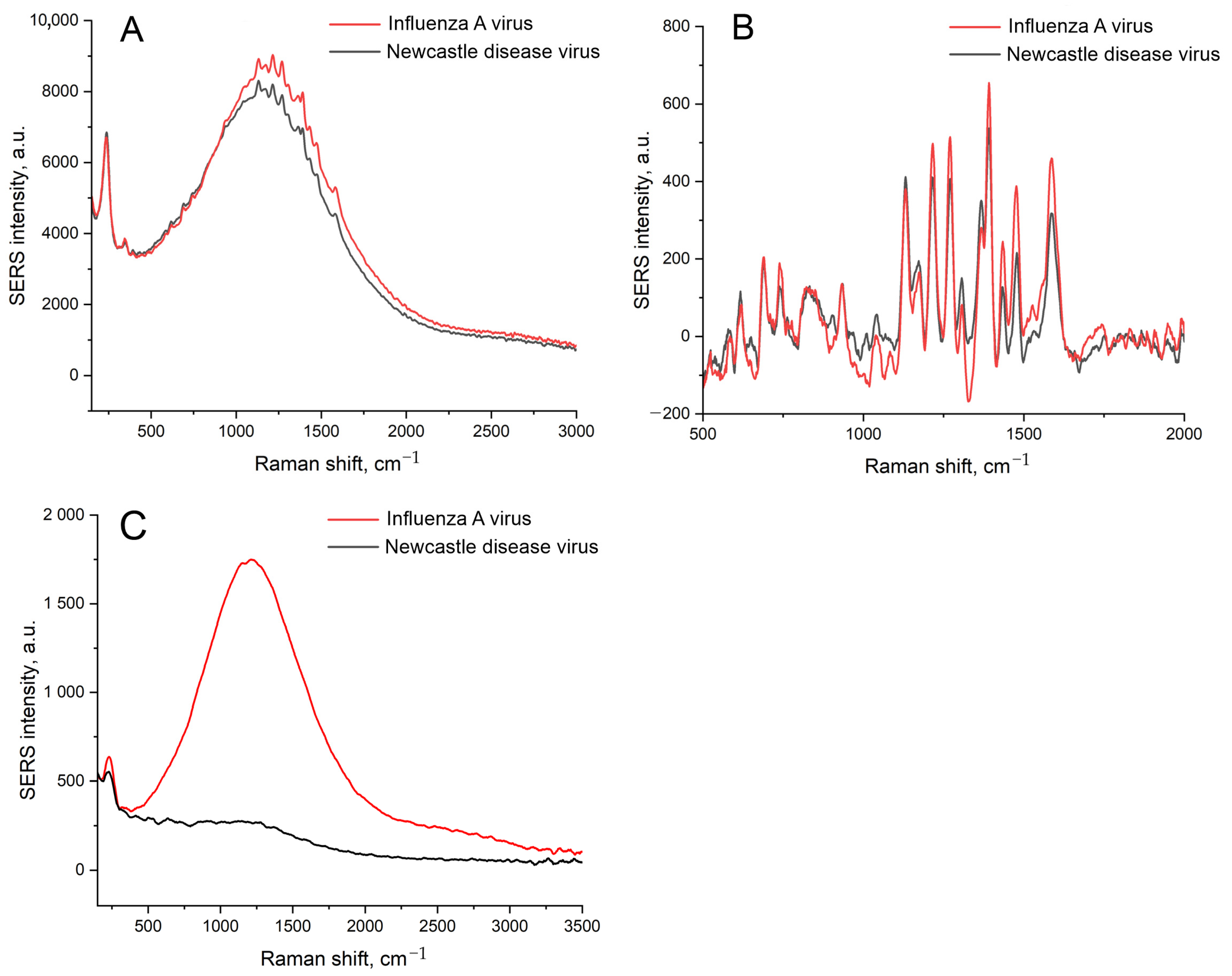
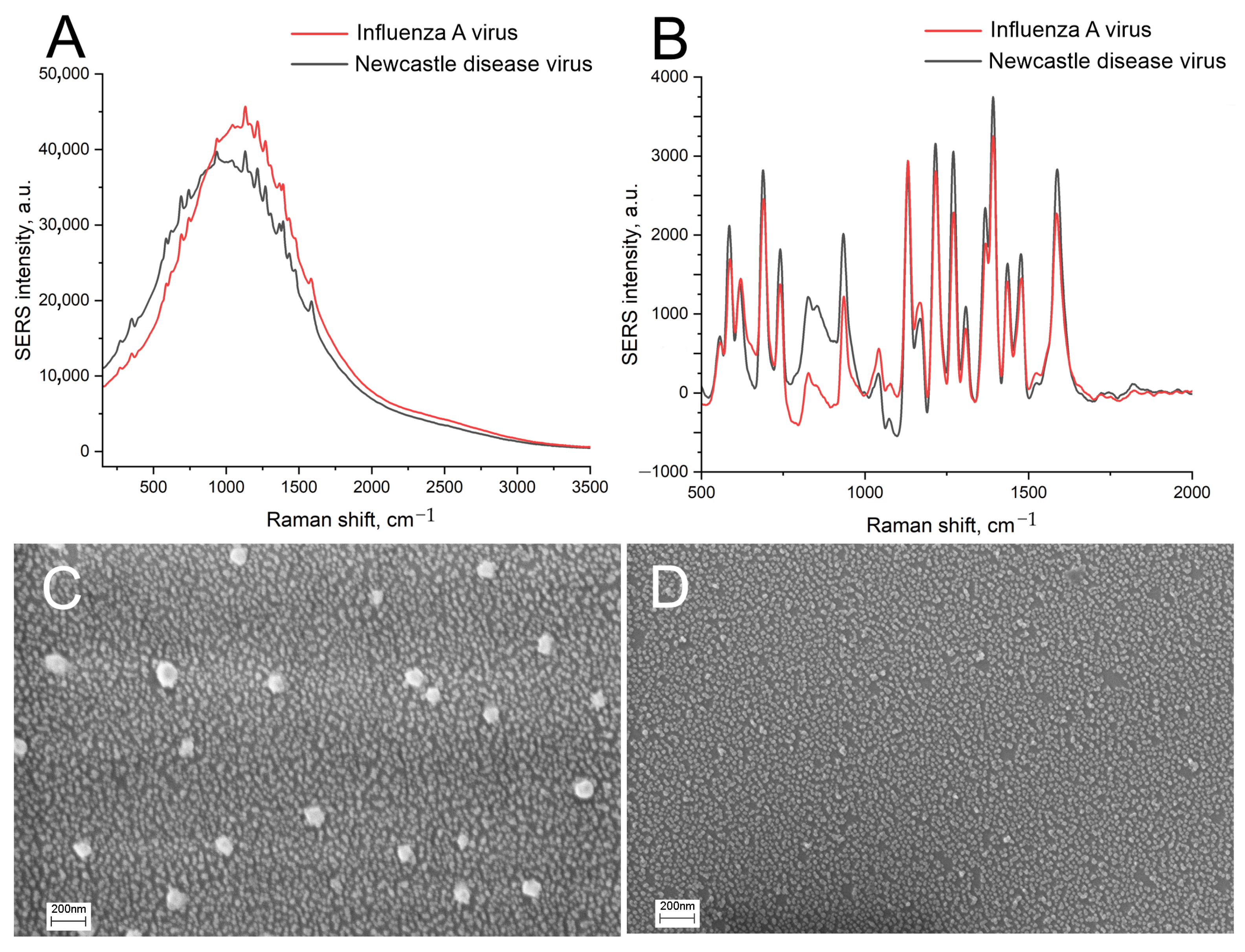




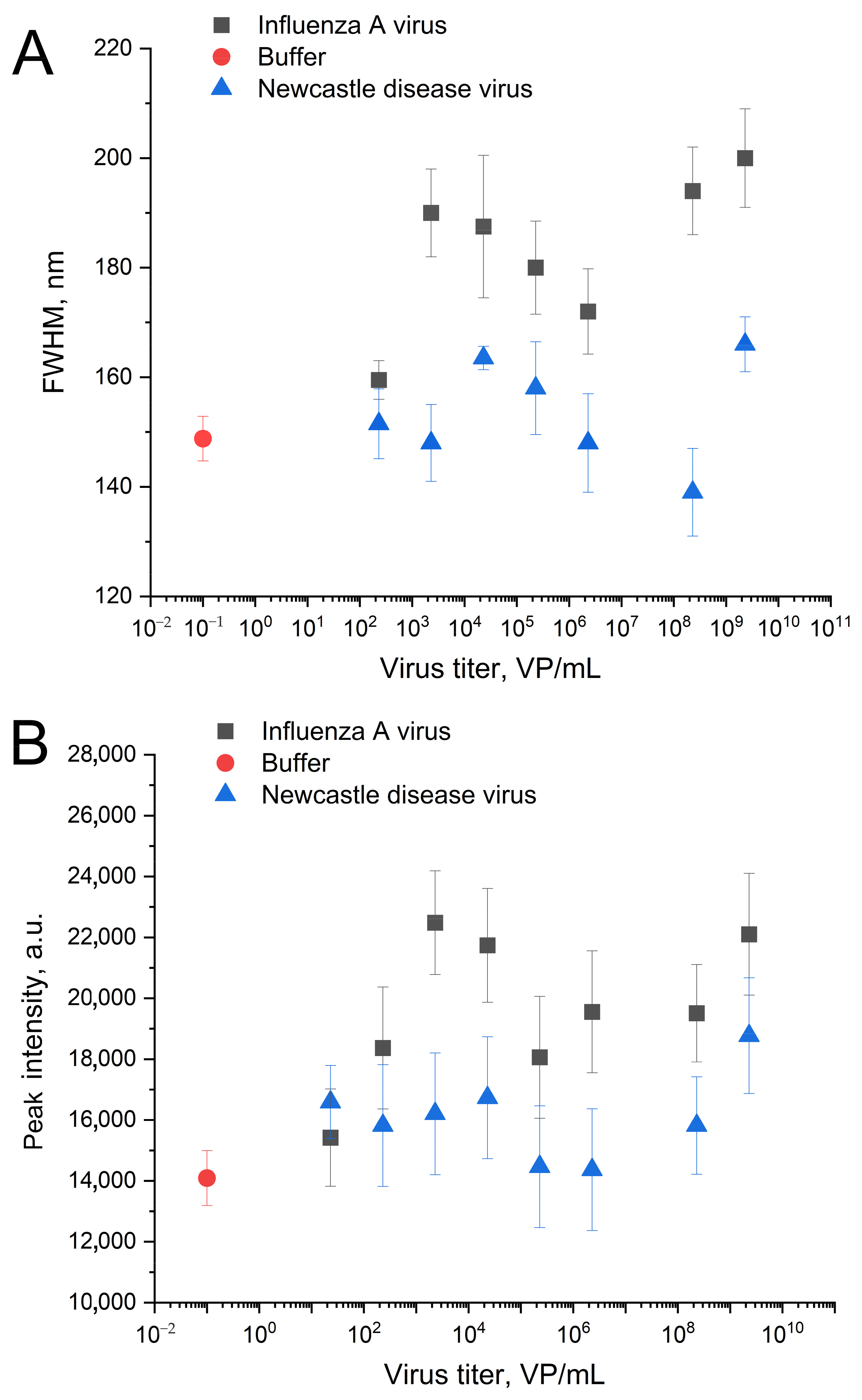

| Substrate | Peak Position, nm | Peak Intensity, a.u. | FWHM, nm |
|---|---|---|---|
| Initial | 628 | 5400 | 150 |
| Aptamer in PBS | 535 | 1100 | 50 |
| Aptamer in TrisNa | 577 | 6200 | 130 |
| RHA0385-SH Concentration, nM | RBD-1C-SH Concentration, nM | SEL(IAV)/SEL(NDV) | SERS(IAV)/SERS(NDV) |
|---|---|---|---|
| 2 | - | n.d. | 1.2 ± 0.1 |
| 20 | - | 2.0 ± 0.2 | 2.4 ± 0.2 |
| 200 | - | 1.1 ± 0.1 | 2.2 ± 0.2 |
| - | 20 | 0.9 ± 0.1 | 0.9 ± 0.1 |
| Time of Immobilization, min | SERS, a.u. |
|---|---|
| 3 | 50 |
| 5 | 400 |
| 15 | 1500 |
| 30 | 1100 |
| Recognizing Element | Virus Type | Type of the SERS-Substrate | Analytical Performance | Refs | ||
|---|---|---|---|---|---|---|
| Limit of Detection | Quantification Range | Time of Analysis | ||||
| None | Rheovirus, rinovirus, influenza A, parainfluenza | Gold nanoparticles onto carbon nanotube arrays | 102 EID50/mL (104 VP/mL) * | No | 15 min | [42] |
| Respiratory syncytial virus | Silver nanorod array | 100 pfu/mL (3 × 105 VP/mL) * | 3 × 105–3 × 106 VP/mL | 1 h | [43] | |
| Circovirus, parvovirus, and pseudorabies | Silver nanoparticles onto porous carbon films | 1 × 107 VP/mL | No | 15 min | [6] | |
| Aptamer | Influenza A | Silver nanoparticles | 2.2 × 10−5 HAU/mL (103 VP/mL) * | No | 15 min | [24] |
| Gold nanopopcorn | 97 pfu/mL (104 VP/mL) * | 104–106 VP/mL | 20 min | [44] | ||
| Silver nanoislands onto silica oxide | 5 × 10−4 HAU/mL (2 × 104 VP/mL) * | No | 12 min | [25] | ||
| Silver nanoparticles onto the membrane | 10 VP/mL | 10–5 × 103 VP/mL | 15 min | [22] | ||
| Silver nanoislands onto silica oxide with chromium | 190 VP/mL | 190–5 × 106 VP/mL | 6 min | This work | ||
| Gold nanopopcorn | 1.06 HAU/mL (5 × 107 VP/mL) * | 5 × 107–5 × 109 VP/mL | 15 min | [45] | ||
| SARS-CoV-2 | 0.95 pfu/mL (7 × 105 VP/mL) * | 7 × 105–7 × 107 VP/mL | ||||
| Silver/Chromium/Gold lithographic nanocolumns | 100 copies/mL (100 VP/mL) | 100–107 VP/mL | 15 min | [21] | ||
| Antibody | Influenza A | Core-shell nanoparticles loaded with a dye | 4 × 103 TCID50/mL (4 × 105 VP/mL) | 4 × 105–109 VP/mL | 3.5 h | [46] |
| Gold nanoparticles | 30 ng/mL (2 × 108 VP/mL) ** | 2 × 108–2 × 1012 VP/mL | 2 h | [47] | ||
| Human immunodeficiency virus | Gold nanostructured surface | 350 fg/mL (2 × 103 VP/mL) ** | 2 × 103–2 × 106 VP/mL | 12 h | [48] | |
| RT PCR | Influenza A | N/A | 3 × 102–1.2 × 103 VP/mL | Yes, the range is not provided | 2–3 h | [4] |
| SARS-CoV-2 | N/A | 1.2 × 103 VP/mL | 103–107 VP/mL | 2–3 h | [3] | |
| RT LAMP | Influenza A | N/A | 104 VP/mL | 104–107 VP/mL | 1 h | [6] |
| SARS-CoV-2 | N/A | 2 × 104 VP/mL | 2 × 104–1011 VP/mL | 1 h | [7] | |
| Anti-body-based test strip | Influenza A | N/A | 1 × 106 VP/mL 20 TCID50/mL | No | 10–15 min | [1] |
| SARS-CoV-2 | N/A | 7.6 × 103 TCID50/mL (5 × 108 VP/mL) * | No | 15 min | [2] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhdanov, G.; Gambaryan, A.; Akhmetova, A.; Yaminsky, I.; Kukushkin, V.; Zavyalova, E. Nanoisland SERS-Substrates for Specific Detection and Quantification of Influenza A Virus. Biosensors 2024, 14, 20. https://doi.org/10.3390/bios14010020
Zhdanov G, Gambaryan A, Akhmetova A, Yaminsky I, Kukushkin V, Zavyalova E. Nanoisland SERS-Substrates for Specific Detection and Quantification of Influenza A Virus. Biosensors. 2024; 14(1):20. https://doi.org/10.3390/bios14010020
Chicago/Turabian StyleZhdanov, Gleb, Alexandra Gambaryan, Assel Akhmetova, Igor Yaminsky, Vladimir Kukushkin, and Elena Zavyalova. 2024. "Nanoisland SERS-Substrates for Specific Detection and Quantification of Influenza A Virus" Biosensors 14, no. 1: 20. https://doi.org/10.3390/bios14010020
APA StyleZhdanov, G., Gambaryan, A., Akhmetova, A., Yaminsky, I., Kukushkin, V., & Zavyalova, E. (2024). Nanoisland SERS-Substrates for Specific Detection and Quantification of Influenza A Virus. Biosensors, 14(1), 20. https://doi.org/10.3390/bios14010020





