Mode-Coupling Generation Using ITO Nanodisk Arrays with Au Substrate Enabling Narrow-Band Biosensing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
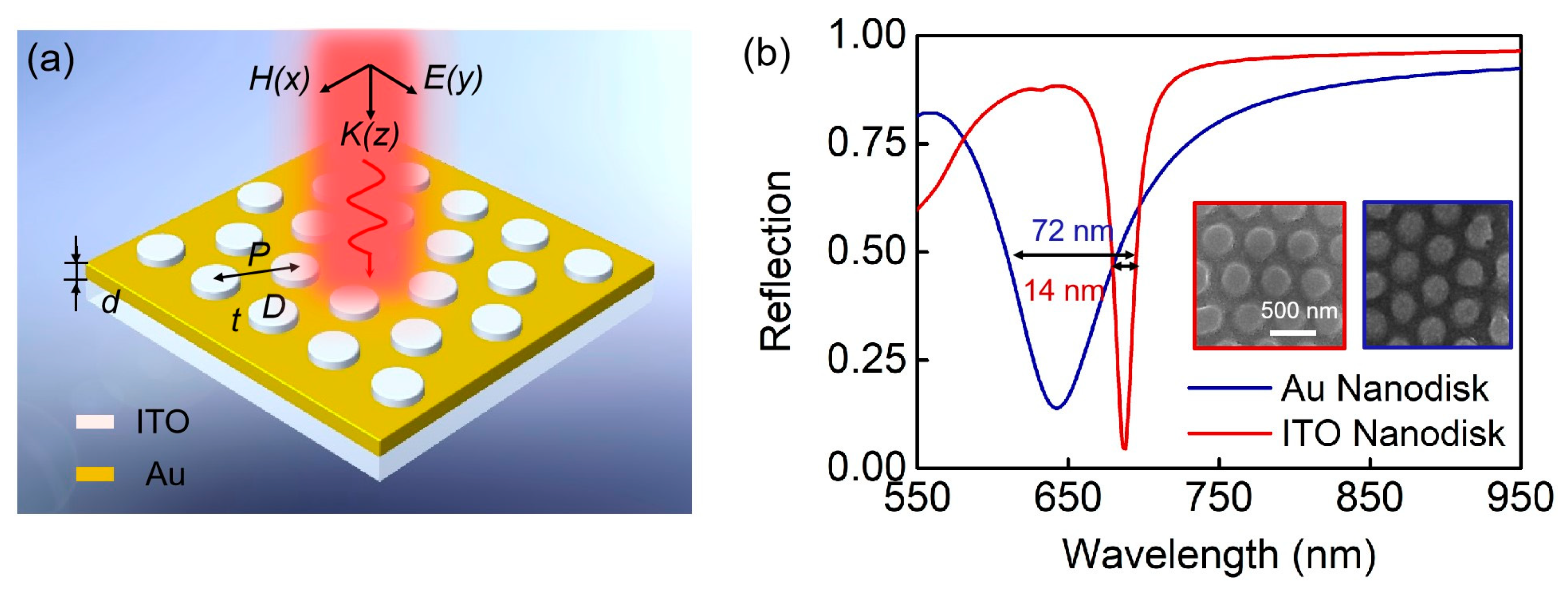
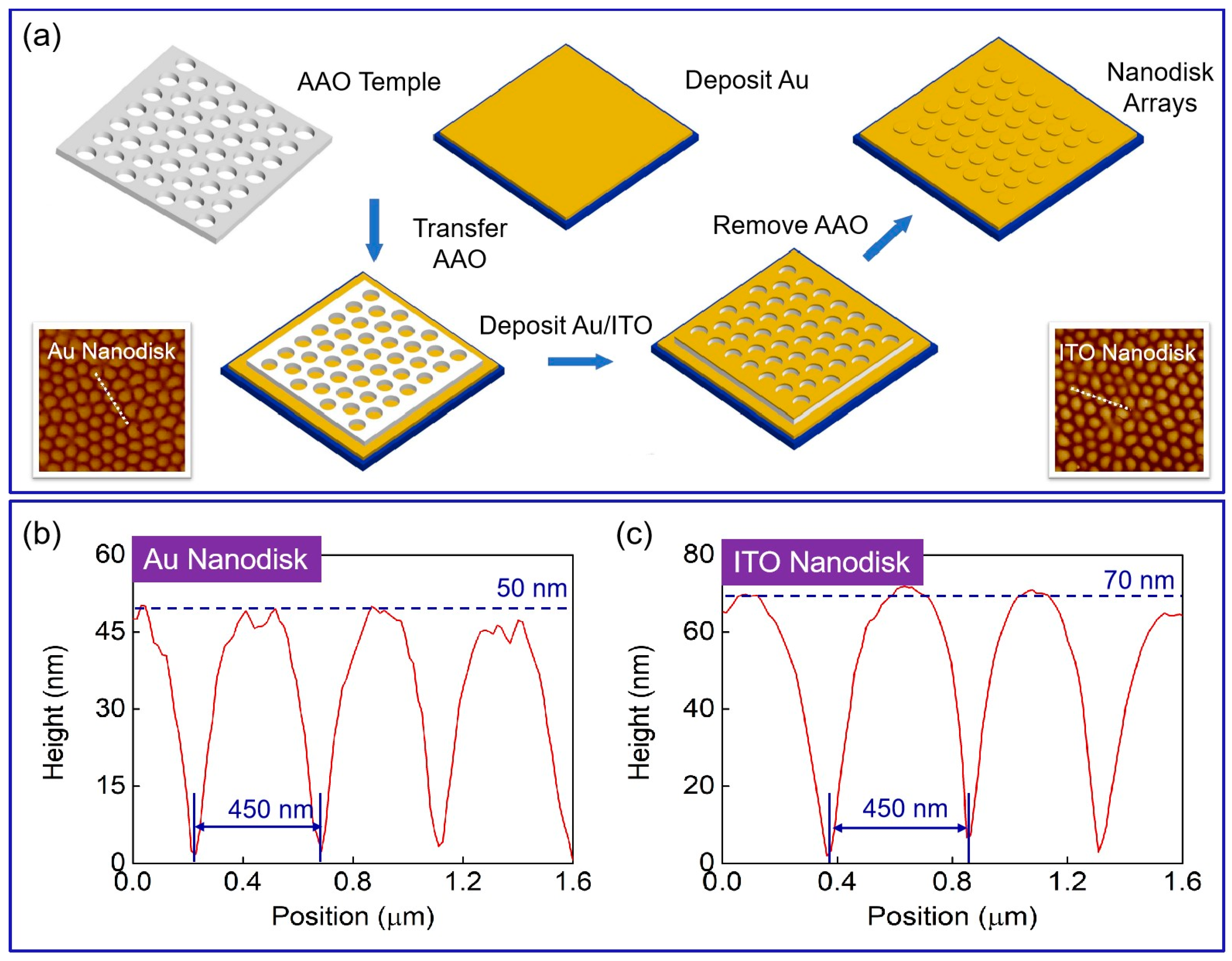
2.2. Preparation of Plasmonic Device Based on a Nanodisk Array Deposited on Au Substrate
2.3. Optical Settings
2.4. Software
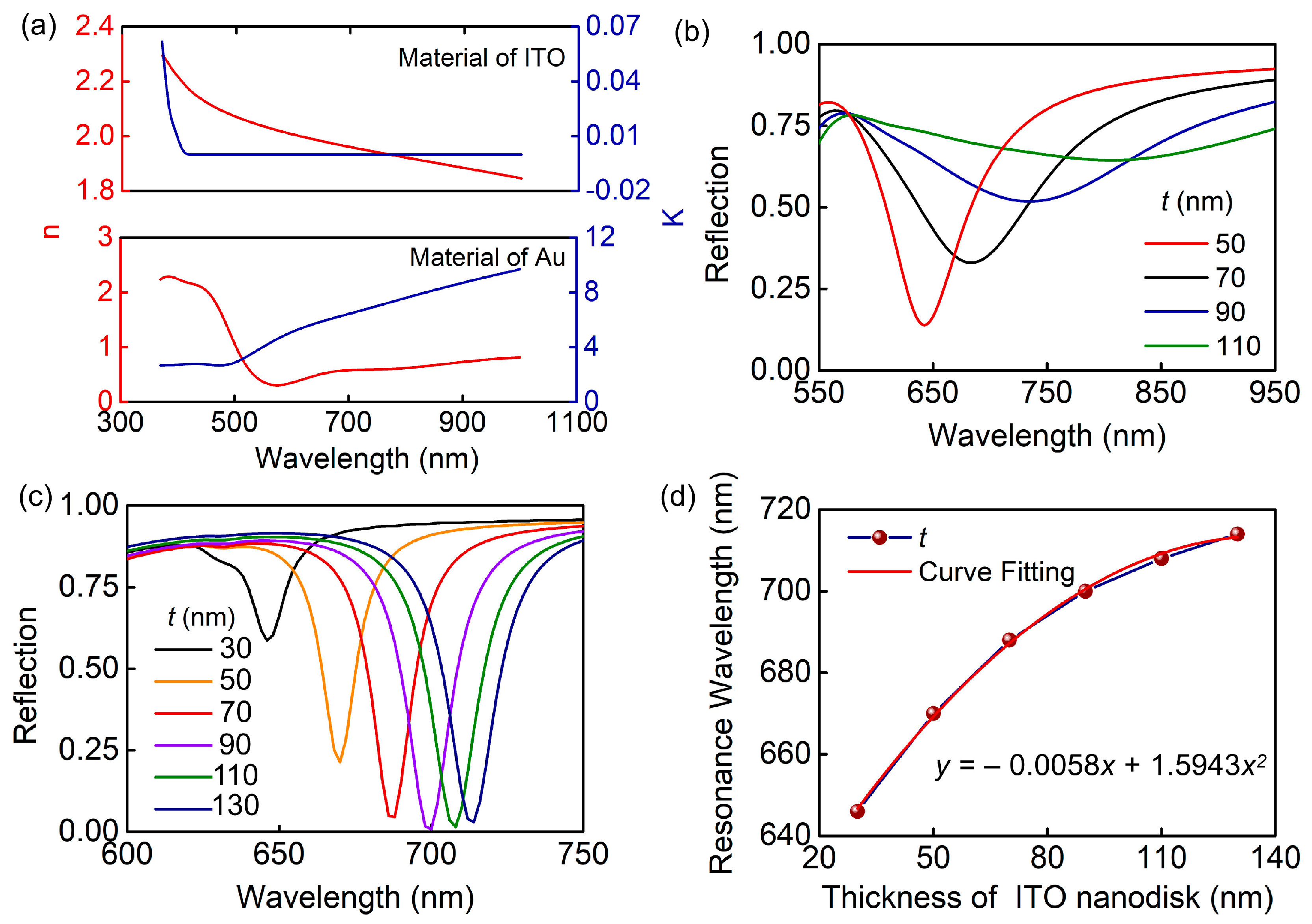
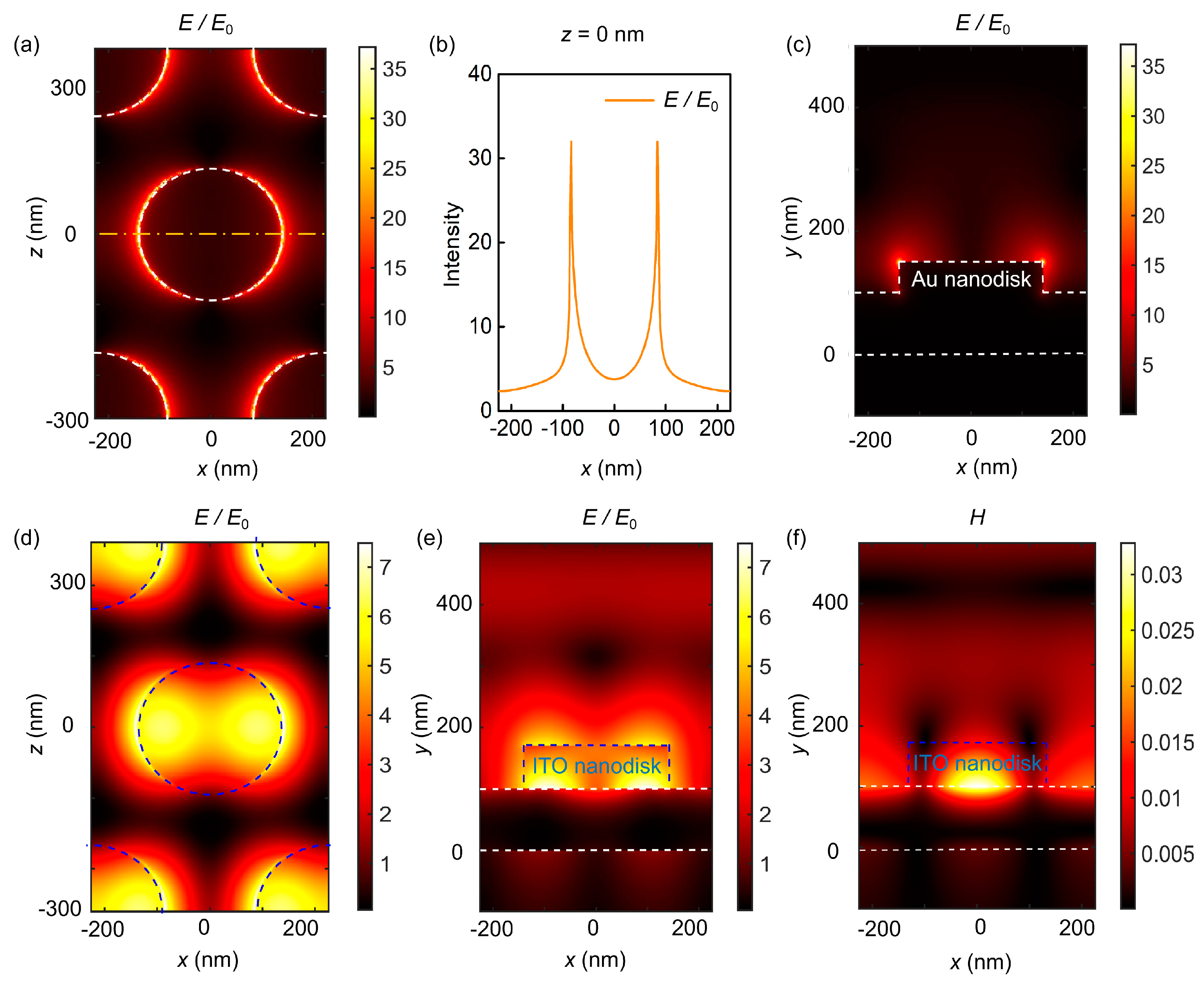
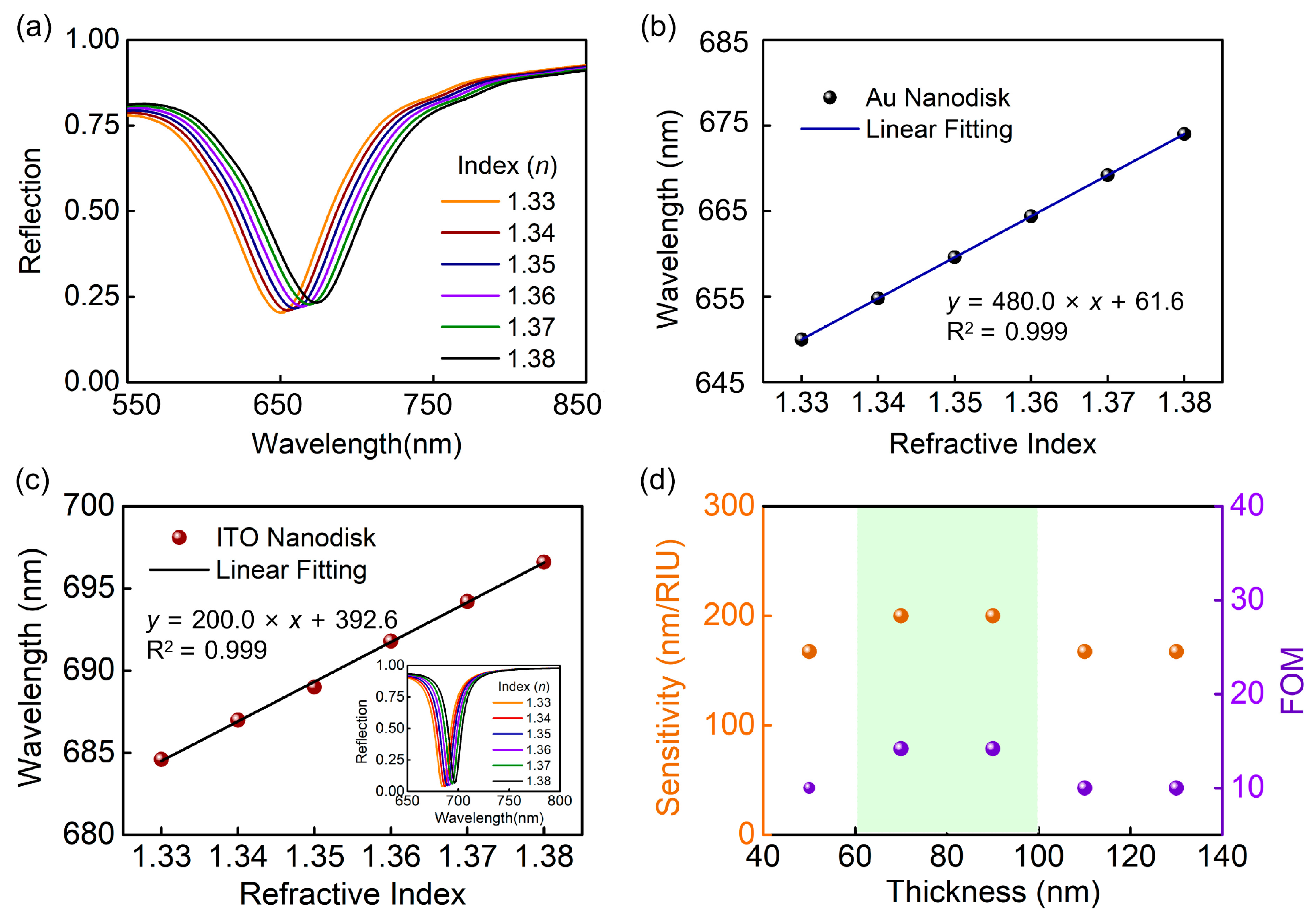
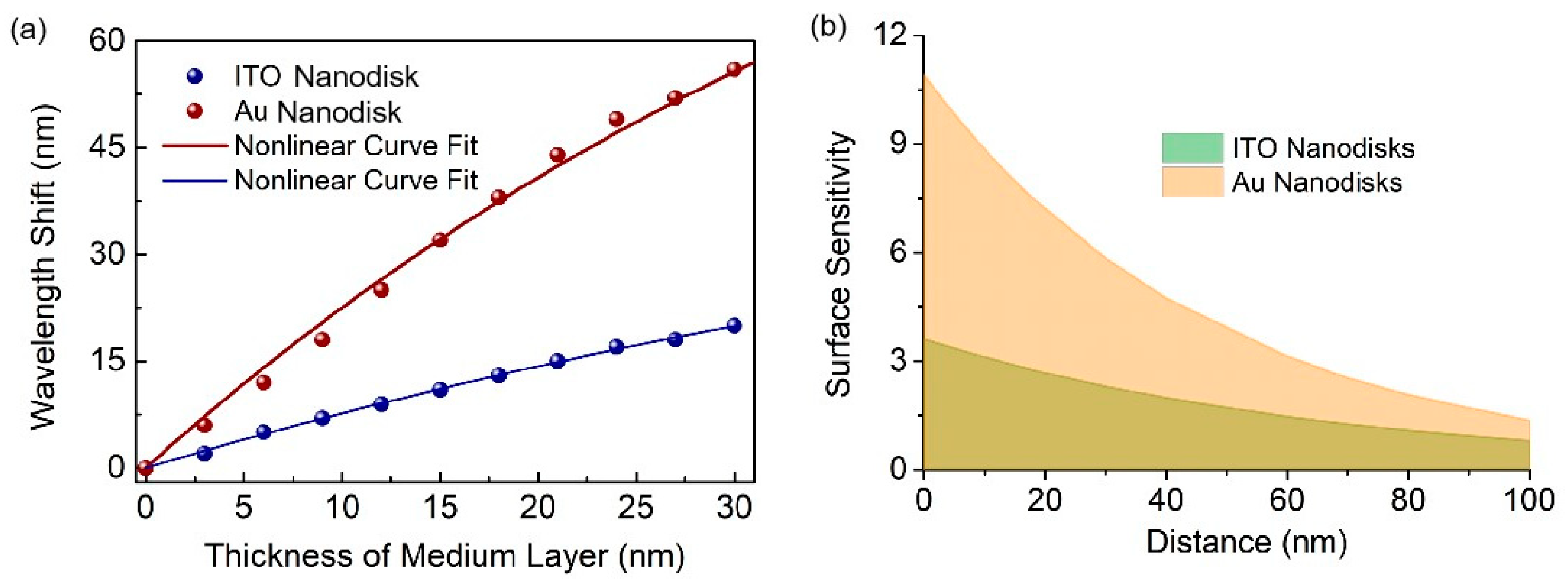
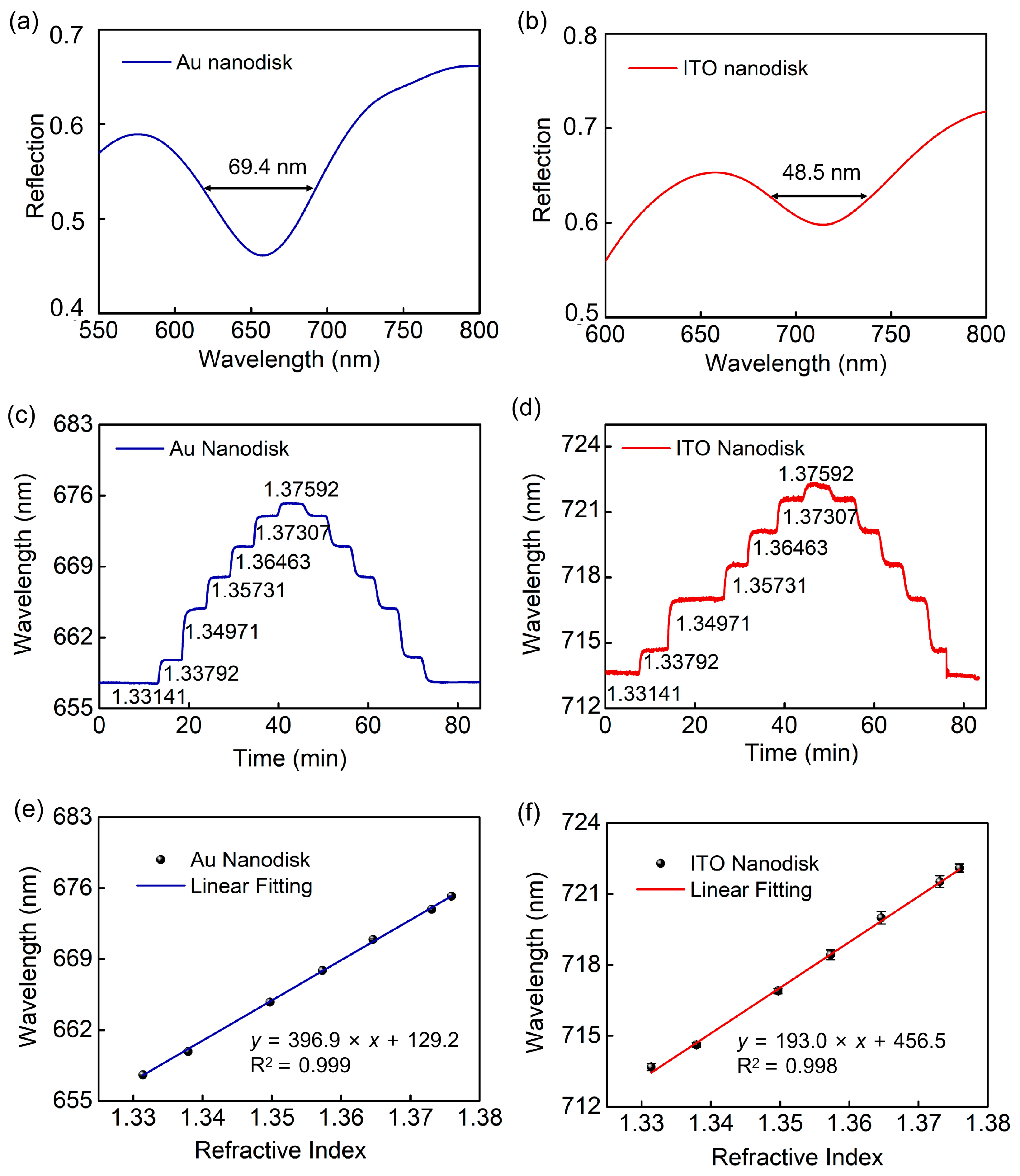
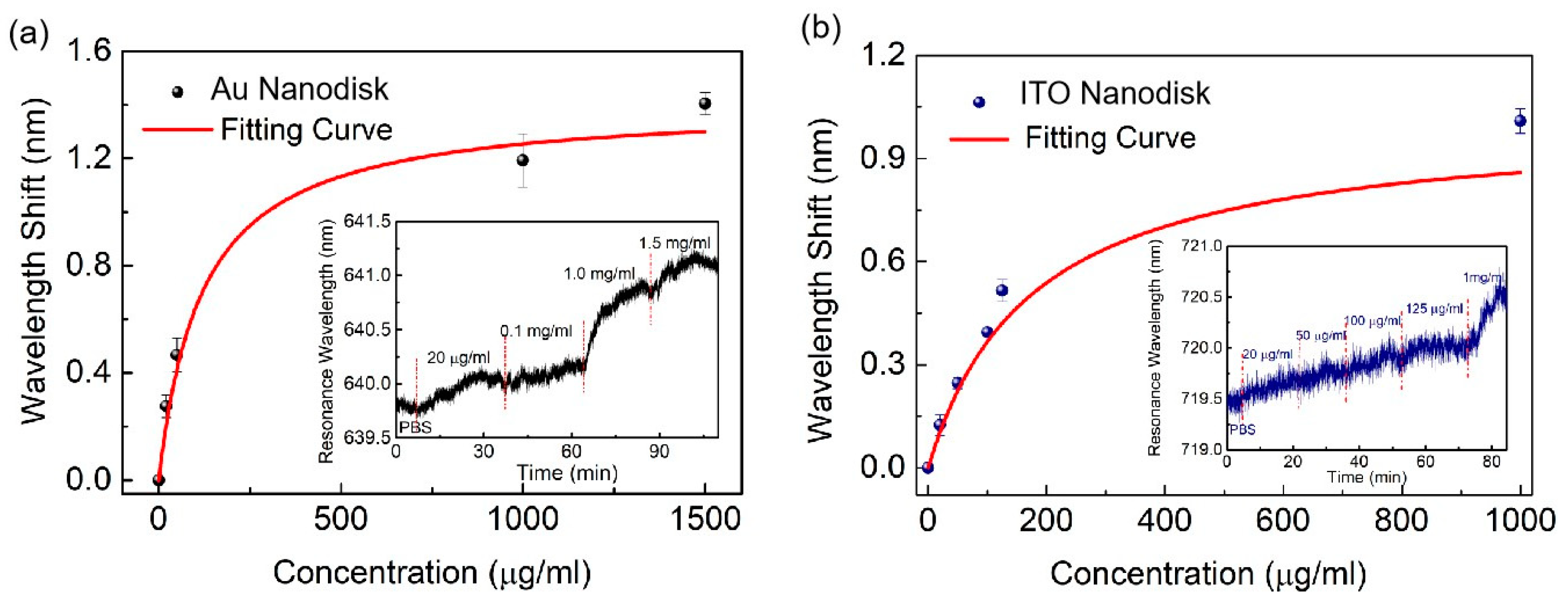
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gu, Y.; Zhang, L.; Yang, J.K.; Yeo, S.P.; Qiu, C.W. Color generation via subwavelength plasmonic nanostructures. Nanoscale 2015, 7, 6409–6419. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Xiao, S.; Song, Q.; Liu, Y.; Wu, Y.; Wang, S.; Yu, J.; Han, J.; Tsai, D.P. All-dielectric metasurface for high-performance structural color. Nat. Commun. 2020, 11, 1864. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Liao, Z.F.; Liu, Z.Q.; Liu, X.S.; Zhou, J.; Liu, G.Q.; Yi, Z.; Wang, J.Q. Recent progresses on metamaterials for optical absorption and sensing: A review. J. Phys. D Appl. Phys. 2021, 54, 113002. [Google Scholar] [CrossRef]
- Yu, P.; Besteiro, L.V.; Huang, Y.J.; Wu, J.; Fu, L.; Tan, H.H.; Jagadish, C.; Wiederrecht, G.P.; Govorov, A.O.; Wang, Z.M. Broadband metamaterial absorbers. Adv. Opt. Mater. 2018, 7, 1800995. [Google Scholar] [CrossRef]
- Nie, J.; Yu, J.; Liu, W.; Yu, T.; Gao, P. Ultra-narrowband perfect absorption of monolayer two-dimensional materials enabled by all-dielectric subwavelength gratings. Opt. Express 2020, 28, 38592–38602. [Google Scholar] [CrossRef]
- Bi, W.; Zhang, X.; Yan, M.; Zhao, L.; Ning, T.; Huo, Y. Low-threshold and controllable nanolaser based on quasi-BIC supported by an all-dielectric eccentric nanoring structure. Opt. Express 2021, 29, 12634–12643. [Google Scholar] [CrossRef]
- Zhu, W.; Xu, T.; Wang, H.; Zhang, C.; Deotare, P.B.; Agrawal, A.; Lezec, H.J. Surface plasmon polariton laser based on a metallic trench Fabry-Perot resonator. Sci. Adv. 2017, 3, 2375–2548. [Google Scholar] [CrossRef]
- Minopoli, A.; Acunzo, A.; Della, V.B.; Velotta, R. Nanostructured surfaces as plasmonic biosensors: A review. Adv. Mater. Interfaces 2021, 9, 2101133. [Google Scholar] [CrossRef]
- Mauriz, E.; Lechuga, L.M. Plasmonic biosensors for single-molecule biomedical analysis. Biosensors 2021, 11, 123. [Google Scholar] [CrossRef]
- Sharma, A.K.; Pandey, A.K. Self-referenced plasmonic sensor with TiO2 grating on thin Au layer: Simulated performance analysis in optical communication band. J. Opt. Soc. Am. B 2019, 36, F25–F31. [Google Scholar] [CrossRef]
- Akiyoshi, K.; Tanaka, Y.Y.; Ishida, T.; Shimura, T.; Tatsuma, T. Plasmonic-diffractive hybrid sensors based on a gold nanoprism array. ACS Appl. Nano Mater. 2018, 1, 5994–5999. [Google Scholar] [CrossRef]
- Ameen, A.; Hackett, L.P.; Seo, S.; Dar, F.K.; Gartia, M.R.; Goddard, L.L.; Liu, G.L. Plasmonic sensing of oncoproteins without resonance shift using 3D periodic nanocavity in nanocup arrays. Adv. Opt. Mater. 2017, 5, 1601051. [Google Scholar] [CrossRef]
- Kuo, C.W.; Wang, S.H.; Lo, S.C.; Yong, W.H.; Ho, Y.L.; Delaunay, J.J.; Tsai, W.S.; Wei, P.K. Sensitive Oligonucleotide Detection Using Resonant Coupling between Fano Resonance and Image Dipoles of Gold Nanoparticles. ACS Appl. Mater. Interfaces 2022, 14, 14012–14024. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Liang, Z.Z.; Meng, D.J.; Qin, Z.; Fan, Y.D.; Shi, X.Y.; Smith, D.R.; Hou, E.Z. All-dielectric refractive index sensor based on Fano resonance with high sensitivity in the mid-infrared region. Results Phys. 2021, 24, 104129. [Google Scholar] [CrossRef]
- Wang, B.Q.; Yu, P.; Wang, W.H.; Zhang, X.T.; Kuo, H.C.; Xu, H.X.; Wang, Z.M. High-Q Plasmonic Resonances: Fundamentals and Applications. Adv. Opt. Mater. 2021, 9, 2001520. [Google Scholar] [CrossRef]
- Kasani, S.; Curtin, K.; Wu, N. A review of 2D and 3D plasmonic nanostructure array patterns: Fabrication, light management and sensing applications. Nanophotonics 2019, 8, 2065–2089. [Google Scholar] [CrossRef]
- Yang, K.; Yao, X.; Liu, B.; Ren, B. Metallic plasmonic array structures: Principles, fabrications, properties, and applications. Adv. Mater. 2021, 33, e2007988. [Google Scholar] [CrossRef]
- Liang, Y.; Cui, W.; Li, L.; Yu, Z.; Peng, W.; Xu, T. Large-scale plasmonic nanodisk structures for a high sensitivity biosensing platform fabricated by transfer nanoprinting. Adv. Opt. Mater. 2019, 7, 1801269. [Google Scholar] [CrossRef]
- Chu, S.W.; Liang, Y.Z.; Yuan, H.Z.; Yu, L.; Liu, Q.; Peng, W. Ultranarrow linewidth coupling resonance in flexible plasmonic nanopillar array for enhanced biomolecule detection. Adv. Mater. Interfaces 2022, 9, 8. [Google Scholar] [CrossRef]
- Liu, B.; Chen, S.; Zhang, J.; Yao, X.; Zhong, J.H.; Lin, H.X.; Huang, T.X.; Yang, Z.L.; Zhu, J.F.; Liu, S.; et al. A Plasmonic Sensor Array with Ultrahigh Figures of Merit and Resonance Linewidths down to 3 nm. Adv. Mater. 2018, 30, 1706031. [Google Scholar] [CrossRef]
- Lu, X.; Zhang, L.; Zhang, T. Nanoslit-microcavity-based narrow band absorber for sensing applications. Opt. Express 2015, 23, 20715–20720. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zheng, L.; Ma, P.; Zhong, Y.; Liu, B.; Chen, X.; Liu, H. Metasurface generated polarization insensitive Fano resonance for high-performance refractive index sensing. Opt. Express 2019, 27, 13252–13262. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Peng, C.; Qi, S.B.; Zhang, Q.; Tang, C.J.; Shen, X.Y.; Da, H.X.; Wang, L.H.; Park, G.S. Photonic microcavity-enhanced magnetic plasmon resonance of metamaterials for sensing applications. IEEE Photon. Technol. Lett. 2019, 31, 113–116. [Google Scholar] [CrossRef]
- Hong, Q.; Luo, J.; Wen, C.C.; Zhang, J.F.; Zhu, Z.H.; Qin, S.Q.; Yuan, X.D. Hybrid metal-graphene plasmonic sensor for multi-spectral sensing in both near- and mid-infrared ranges. Opt. Express 2019, 27, 35914–35924. [Google Scholar] [CrossRef] [PubMed]
- Vala, M.; Ertsgaard, C.T.; Wittenberg, N.J.; Oh, S.H. Plasmonic sensing on symmetric nanohole arrays supporting high-Q hybrid modes and reflection geometry. ACS Sens. 2019, 4, 3265–3274. [Google Scholar] [CrossRef]
- Spackova, B.; Wrobel, P.; Bockova, M.; Homola, J. Optical biosensors Based on plasmonic nanostructures: A review. Proc. IEEE 2016, 104, 2380–2408. [Google Scholar] [CrossRef]
- Conteduca, D.; Barth, I.; Pitruzzello, G.; Reardon, C.P.; Martins, E.R.; Krauss, T.F. Dielectric nanohole array metasurface for high-resolution near-field sensing and imaging. Nat. Commun. 2021, 12, 3293. [Google Scholar] [CrossRef]
- Yesilkoy, F.; Arvelo, E.R.; Jahani, Y.; Liu, M.K.; Tittl, A.; Cevher, V.; Kivshar, Y.; Altug, H. Ultrasensitive hyperspectral imaging and biodetection enabled by dielectric metasurfaces. Nat. Photonics 2019, 13, 390–396. [Google Scholar] [CrossRef]
- Caruso, F.; Niikura, K.; Furlong, D.N.; Okahata, Y. 2. Assembly of alternating polyelectrolyte and protein multilayer films for immunosensing. Langmuir 1997, 13, 3427–3433. [Google Scholar] [CrossRef]
- Wang, G.L.; Shu, J.X.; Dong, Y.M.; Xu, X.M.; Li, Z.J. An ultrasensitive and universal photoelectrochemical immunoassay based on enzyme mimetics enhanced signal amplification. Biosens. Bioelectron. 2015, 66, 283–289. [Google Scholar] [CrossRef]
| Δλ∞ | ld | |
|---|---|---|
| Full-metal nanodisk arrays | 119.8 nm | 48.0 nm |
| ITO nanodisk arrays | 54.8 nm | 66.2 nm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, S.; Liang, Y.; Lu, M.; Yuan, H.; Han, Y.; Masson, J.-F.; Peng, W. Mode-Coupling Generation Using ITO Nanodisk Arrays with Au Substrate Enabling Narrow-Band Biosensing. Biosensors 2023, 13, 649. https://doi.org/10.3390/bios13060649
Chu S, Liang Y, Lu M, Yuan H, Han Y, Masson J-F, Peng W. Mode-Coupling Generation Using ITO Nanodisk Arrays with Au Substrate Enabling Narrow-Band Biosensing. Biosensors. 2023; 13(6):649. https://doi.org/10.3390/bios13060649
Chicago/Turabian StyleChu, Shuwen, Yuzhang Liang, Mengdi Lu, Huizhen Yuan, Yi Han, Jean-Francois Masson, and Wei Peng. 2023. "Mode-Coupling Generation Using ITO Nanodisk Arrays with Au Substrate Enabling Narrow-Band Biosensing" Biosensors 13, no. 6: 649. https://doi.org/10.3390/bios13060649
APA StyleChu, S., Liang, Y., Lu, M., Yuan, H., Han, Y., Masson, J.-F., & Peng, W. (2023). Mode-Coupling Generation Using ITO Nanodisk Arrays with Au Substrate Enabling Narrow-Band Biosensing. Biosensors, 13(6), 649. https://doi.org/10.3390/bios13060649






