Abstract
The ultimate objective of this research work is to design a sensitive and selective electrochemical sensor for the efficient detection of ascorbic acid (AA), a vital antioxidant found in blood serum that may serve as a biomarker for oxidative stress. To achieve this, we utilized a novel Yb2O3.CuO@rGO nanocomposite (NC) as the active material to modify the glassy carbon working electrode (GCE). The structural properties and morphological characteristics of the Yb2O3.CuO@rGO NC were investigated using various techniques to ensure their suitability for the sensor. The resulting sensor electrode was able to detect a broad range of AA concentrations (0.5–1571 µM) in neutral phosphate buffer solution, with a high sensitivity of 0.4341 µAµM−1cm−2 and a reasonable detection limit of 0.062 µM. The sensor’s great sensitivity and selectivity allowed it to accurately determine the levels of AA in human blood serum and commercial vitamin C tablets. It demonstrated high levels of reproducibility, repeatability, and stability, making it a reliable and robust sensor for the measurement of AA at low overpotential. Overall, the Yb2O3.CuO@rGO/GCE sensor showed great potential in detecting AA from real samples.
1. Introduction
Ascorbic acid (AA) is a vital biomolecule that is present in a variety of naturally occurring sources, including fruits and vegetables, and that functions as a nutrient and antioxidant [1]. It is essential to numerous bodily metabolic activities including activating the immune system, aiding in wound healing, helping with the absorption of iron, and protecting against damage to bones and teeth [2]. Additionally, AA serves as a cofactor during the synthesis of collagen and carnitine [3]. In addition, AA has been demonstrated to provide protective effects against oxidative illnesses such as heart disease, several cancers, AIDS, the common cold, etc. [4]. However, there is no AA produced by the human body and it can only be obtained through the consumption of foods and medicines [5]. AA is a crucial ingredient in dietary and pharmaceutical supplements [6]. Human blood serum typically contains between 28.5 and 85.2 µM of AA, and the amount of AA in blood serum can provide information about a person’s general state of health [7]. Scurvy and anemia can result from an AA deficit in blood serum, while an excess of AA can lead to gastric irritation or diarrhea [8]. Therefore, it is crucial to have precise and efficient techniques for figuring out how much AA is present in foods, medications, and blood serum.
Direct titration [9], chromatography [10], spectrophotometry [11], and solid-phase spectrophotometry [12] are currently used methods for AA measurement. However, such methods are costly and require skilled personnel as well as challenging analytical measures for multi-sample preparation. To address these issues, researchers are working to develop efficient and cost-effective methods for real-time and in situ AA determination. The advantages of electrochemical detection include quicker measurements, reduced sample size, reduced costs, and an absence of pre-concentration processes, making them handy, portable, and simple to use with miniaturized electrodes [13]. However, due to its irreversible nature and high overpotential requirements, the electrochemical AA oxidation at a bare electrode might have negative effects on selectivity, electrode fouling, and repeatability. Therefore, it is indispensable to fabricate an electrode surface that enables efficient AA detection with less overpotential. In recent years, to detect AA, researchers have proposed several sensors, including electrodes modified with metals and metal oxides [14], alkylimidazolium salt [15], graphene derivatives [16], carbon nanotubes [14], and polymers [17].
Due to their potential technological uses and intriguing optical and structural characteristics, semiconducting doped nanostructured materials comprising transition metal oxides have drawn a lot of attention. Due to their size, shape, and surface, these materials have distinct physical and chemical properties that make them relevant in a variety of study fields and applications for industry. In particular, metal oxide-based sensors have been explored for their diverse uses in areas such as the protection of the environment, chemical process management, personal security, healthcare, and in the military [18,19,20]. These sensors have several advantages, including their compact size, affordable price, lower power consumption, straightforward processing, and good stability [21]. Previously, researchers have investigated various types of metal oxides, such as CuO [22,23], MnO2 [24], NiO [25], Fe2O3 [26], and ZnO [27], as electron mediators for sensing applications. Additionally, doped metal oxides, such as NiO.CoO nanocomposites [28], CdO.SnO2.V2O5 [29], CuO.In2O3 [30], CuO.Nd2O5 [31], CuO.NiO [32], and CuO.ZnO [33], have been studied as efficient sensing materials with higher sensitivity, small detection limits, wide linear dynamic ranges, and quick response times. CuO, a p-type semiconductor, has shown particularly good performance as an electrocatalyst in sensing applications [34]. To improve the performance of CuO, researchers have also investigated using other semiconductor metal oxides such as In2O3 [30], Nd2O5 [31], NiO [32], TiO2 [35], and SnO2 [36] in combination with CuO as bimetallic oxide pairs. Ytterbium oxide (Yb2O3) has also been explored for use in sensing applications [37,38,39]. Yb2O3 is a lanthanide-based C-type sesquioxide that can exhibit electrochemical redox characteristics [40]. In addition, the electrical as well as structural and morphological properties of the CuO can be engineered through doping of the CuO matrix with suitable dopants. Many trivalent lanthanide ions and their oxides have been reported as excellent dopants for CuO with modified and enhanced electrical properties. However, a detailed literature survey revealed very few reports for the synthesis of Yb2O3-doped CuO structures. Thus, here we have synthesized the Yb2O3-doped CuO nanostructures through the coprecipitation method. Furthermore, this Yb2O3.CuO was combined with rGO to obtain the ternary Yb2O3.CuO@rGO nanocomposite for the AA sensing with improved sensor parameters.
A lot of research has been carried out on the applicability of carbonaceous nanomaterials for sensing applications, including reduced graphene oxide, activated carbon, mesoporous carbon, and carbon nanotubes. In particular, graphene, a sheet of sp2-bonded carbon atoms, with a particular surface area, low density, outstanding electrical conductivity, and great mechanical properties, has drawn a lot of interest [41,42]. Graphene-based 3D nanomaterials have also generated huge interest due to their high surface area, lower density, better electrical conductivity, and exceptional mechanical properties [43,44]. Composite materials made of metal oxides and graphene have recently been explored for their stability, long-term storage, and photo-catalytic capabilities [45,46]. Many graphene-based nanomaterials have been studied in sensing applications [47,48,49]. However, rare earth oxide-transition metal oxide-reduced graphene nanocomposite has hardly been studied in sensing applications. Hence, in this work, we developed and examined Yb2O3.CuO@rGO nanocomposite (NC) as the active sensing material for AA detection.
Inspired by previous works available in the literature, we synthesized Yb2O3-doped CuO nanoparticles to improve stability, sensitivity, and selectivity, and then used a simple sonication technique to synthesize the Yb2O3.CuO@rGO NC sensing material. This study presents a simple method for preparing an electrochemical AA sensor using Yb2O3.CuO@rGO NC that offered improved selectivity and sensitivity. As far as we are aware, this will be the maiden article utilizing Yb2O3.CuO@rGO NC to develop an enzyme-less AA electrochemical sensor.
2. Materials and Methods
2.1. Materials
All necessary chemicals, including ascorbic acid, copper (II) nitrate, ytterbium (III) nitrate, sodium hydroxide, reduced graphene oxide, NaH2PO4, Na2HPO4, citric acid, glucose, uric acid, dopamine, sodium chloride, and calcium nitrate, were purchased from Sigma–Aldrich, and utilized exactly as they were given. All solutions were made using double-distilled water. The XPS investigation of Yb2O3.CuO@rGO was performed using a MgKα spectrometer (JEOL, JPS 9200) in the subsequent circumstances: pass energy = 50 eV (wide-scan) and 30 eV (narrow-scan), Voltage = 10 kV, Current = 20 mA. A PANalytical X-ray diffractometer was used to acquire X-ray diffraction (XRD) spectra with Cu Kα1/2, λα1 = 154.060 pm, λα2 = 154.439 pm radiation. A “Raman station 400 (Perkin Elmer)” spectrometer was used to acquire the Raman spectra. A FE-SEM (JEOL-6300F, 5 kV) was used to analyze the morphology and structural characteristics of Yb2O3.CuO@rGO. EDS (JEOL) was used to investigate the elemental composition of the Yb2O3.CuO@rGO. A JEOL JEM-2100F-UHR field emission apparatus fitted with a Gatan GIF 2001 energy filter and a 1 k-CCD camera was used to capture transmission electron microscopy (TEM) micrographs at 200 kV. Electrochemical measurements were conducted using a Zahner Zennium potentiostat.
2.2. Synthesis of CuO, Yb2O3, Yb2O3.CuO, and Yb2O3.CuO@rGO Nanocomposite
To synthesize the CuO, Yb2O3, Yb2O3.CuO, and Yb2O3.CuO@rGO nanocomposites, the following process was followed: firstly, equimolar Cu(NO3)2 and Yb(NO3)3 solutions were mixed in a beaker and stirred for half an hour at 70 °C. This mixture was then combined with NaOH and stirred vigorously at 80 °C for 8 h. Afterwards, the ensuing dark precipitate was cleaned with distilled water and ethanol to remove contaminants and the resulting black precipitate was dried at 80 °C. This as-grown Yb2O3.CuO nanoparticle (NP) was then calcined by heating it for six hours at 500 °C in a furnace. During this synthesis process, the following chemical reactions occurred:
Cu(NO3)2 + 2NaOH → Cu(OH)2 + 2NaCl
Yb(NO3)3 + 3NaOH → Yb(OH)3 + 3NaCl
Cu(OH)2 + 2Yb(OH)3 → Yb2O3.CuO + 4H2O
Precursors, Yb3+ and Cu2+ ions are soluble in NaOH solution, where NaOH keeps the pH constant during the reaction and continuously releases OH−. The development of the Cu(OH)2 nucleus starts when the ionic product of Cu2+ and OH− exceeds the Ksp value. Similarly, Yb(OH)3 was also produced. Cu2+ ions easily incorporate themselves into the Yb2O3 lattice because of the similar ionic radii. On heating, hydroxides decompose to produce respective oxides. Similarly, CuO and Yb2O3 NPs were also synthesized.
To synthesize the Yb2O3.CuO@rGO nanocomposite, 0.5 g of Yb2O3.CuO NPs and 0.025 g of reduced graphene oxide (rGO) were mixed followed by 40 min of sonication in 80 mL distilled water. This resulting mixture was then filtered and had 12 h of drying in an oven at 70 °C.
2.3. Glassy Carbon Electrode Modification Using Yb2O3.CuO@rGO Nanocomposite
Glassy carbon electrodes (GCEs) (diameter = 3 mm; BAS Inc., Sumida-Ku, Japan) were cleaned using a 1 µm diamond past, followed by a 0.05 µm alumina slurry using the commercially available polishing pads. Next, the GCE was fabricated utilizing Yb2O3.CuO@rGO nanocomposite using a Nafion solution. During the fabrication process, 4.0 mg of Yb2O3.CuO@rGO was uniformly mixed with 0.05 mL Nafion and 0.45 mL propan-2-ol, and then 2 µL of this suspension was carefully applied to a pre-cleaned GCE and dried at 60 °C for 20 min. Such a fabricated GCE was labeled as the Yb2O3.CuO@rGO/GCE. Control experiments were also conducted, in which CuO/GCE, Yb2O3/GCE, rGO/GCE, and Yb2O3.CuO/GCE were fabricated using similar procedures. The electrochemical investigations of AA (0.5–1744 µM) were carried out in a typical three-electrode electrochemical cell at ambient conditions in 0.1 M PBS (pH 7.0), a Yb2O3.CuO@rGO/GCE, Ag/AgCl, and a platinum spiral were served as the working, reference, and counter electrodes, respectively.
3. Results and Discussion
3.1. Characterization of Yb2O3.CuO@rGO Nanocomposite
The elemental compositions and structure of Yb2O3.CuO@rGO were examined using XPS. It is evident from the XPS analysis shown in Figure 1a–e that Yb2O3.CuO@rGO nanocomposite is composed of Yb, Cu, O, and C atoms only. The Yb4d5/2 spectrum has three clearly defined peaks appearing at energies of 187.2, 188.4, and 189.1, which are compatible with Yb4d (Figure 1b) [50]. In the deconvoluted Cu2p spectrum in Figure 1c, there are two peaks at 937.1 and 956.8 eV that may be related to Cu2p3/2 and Cu2p1/2, respectively [51]. In between these two peaks, there are some satellite peaks that appeared that are also consistent with the literature [52]. Figure 1d shows two peaks from the fine-scan O1s spectra that are associated with the Yb–O and Cu–O bonds, respectively, at 533.3 and 535.2 eV [13]. Three peaks are shown in the fine-scan C1s spectrum in Figure 1e at energies of 284.6, 287.2, and 289.1 eV. The peaks at 284.6 and 287.2 eV may be attributed to C–C and C–O–H bonds, respectively [53], and the remaining peak at 289.1 eV can be correlated to COOH [54].
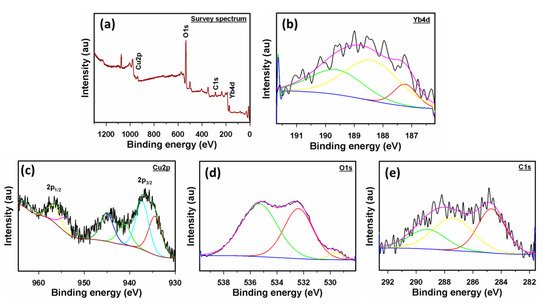
Figure 1.
(a) Survey XPS spectrum of Yb2O3.CuO@rGO NC, (b) deconvoluted spectra of Yb4d, (c) Cu2p, (d) O1s, and (e) C1s of Yb2O3.CuO@rGO nanocomposite.
XRD patterns in Figure 2a showed diffraction bands at 2θ = 20.80, 29.50, 34.30, 36.50, 40.60, 44.00, 47.50, 49.20, 51.00, 54.10, 57.10, 58.50, 60.00, and 61.50, which are related to the (211), (222), (400), (411), (332), (134), (125), (440), (443), (611), (145), (662), (136), and (444) planes for Yb2O3 NPs (JCPDS#65-3173), respectively [50]. The diffraction bands at 35.40, 38.60, 48.60, 58.20, 61.60, 66.30, and 68.10 can be related to (002), (111), (−202), (202), (−113), (−311), and (220) planes of CuO NPs ((JCPDS#45-0937), respectively [55]. The Yb2O3.CuO@rGO contains the rGO peak connected to carbon that is often appearing at 2θ = 24.30 which is correlated to (002) plane [56] but is not easily visible in Figure 2a due to low intensity. However, the presence of carbon in Yb2O3.CuO@rGO was established by XPS, EDS, SEM, and TEM. Figure 2b shows the Raman spectra, where bands at 359.3, 718, and 1060 cm−1 can be related to Yb2O3, while bands at 328 and 850 cm−1 were connected to CuO [57]. The characteristic carbon bands at 1344 and 1676 cm−1 are related to the D and G bands of rGO [58].
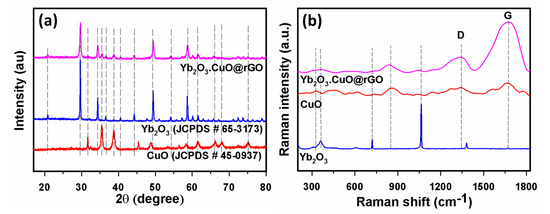
Figure 2.
(a) XRD patterns and (b) Raman spectra of CuO, Yb2O3, and Yb2O3.CuO@rGO NC.
FESEM was employed to analyze the morphological and surface structure of CuO, Yb2O3, Yb2O3.CuO, and Yb2O3.CuO@rGO nanocomposite as presented in Figure 3a–d. The Yb2O3.CuO@rGO nanocomposite was found to be made up of Yb2O3.CuO composites that were randomly distributed over the graphene sheets. EDS was used to determine the Yb2O3.CuO@rGO nanocomposite’s elemental composition (Figure 3e), and the results showed that the nanocomposite is exclusively made of Yb, Cu, O, and C with their respective weight percentages as 39.37%, 17.02%, 29.27%, and 14.34%. This elemental composition agrees with the findings of XPS and XRD. A more thorough morphology of CuO, Yb2O3, Yb2O3.CuO, and Yb2O3.CuO@rGO nanocomposite was provided by the TEM images in Figure 3f–i that show a collection of spherical Yb2O3 and elongated CuO NPs dispersed on sheet-like structures of rGO. Figure 3j presents an HR-TEM image of the Yb2O3.CuO@rGO nanocomposite and Figure 3k displays the selected area electron diffraction (SAED) patterns, which unequivocally reveal that the composite is polycrystalline.
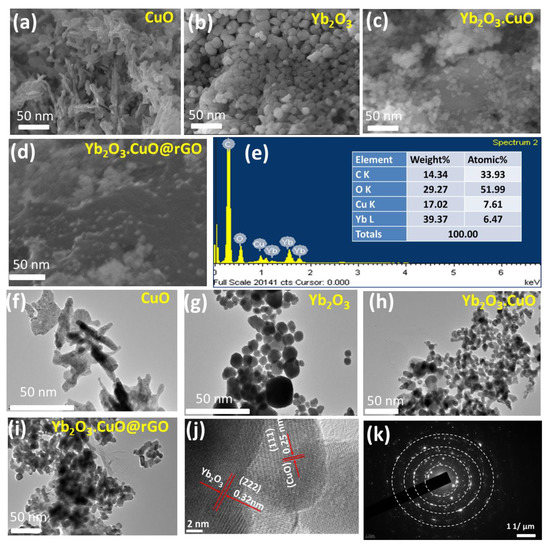
Figure 3.
FESEM image: (a) CuO, (b) Yb2O3, (c) Yb2O3.CuO, (d) Yb2O3.CuO@rGO, (e) EDS spectrum of Yb2O3.CuO@rGO; TEM micrograph from (f) CuO, (g) Yb2O3, (h) Yb2O3.CuO, (i) Yb2O3.CuO@rGO, (j) HR-TEM image, and (k) SAED patterns of Yb2O3.CuO@rGO nanocomposite.
3.2. Ascorbic Acid Sensor Development
3.2.1. Electrochemical Study of Yb2O3.CuO@rGO/GCE Assembly
We evaluated the electro-chemical activity of the modified electrodes through cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS). Figure 4a illustrates the feeble CV response from a bare GCE in the presence of 40 µM AA at +0.52 V; however, the CuO/GCE and Yb2O3.CuO/GCE showed enhanced CV outputs at +0.41 V and 0.28 V, respectively. For the Yb2O3/GCE and rGO/GCE electrodes, no CV response was detected. A significantly improved CV result at a low potential of +0.25 V was obtained from the Yb2O3.CuO@rGO/GCE. This demonstrates that this Yb2O3.CuO@rGO/GCE assembly possessed the greatest electrocatalytic performance during AA determination in comparison to other the electrodes shown in Figure 4a. Therefore, we designated the Yb2O3.CuO@rGO/GCE assembly as an AA sensor in this investigation. Additionally, a definite CV peak was produced for the Yb2O3.CuO@rGO/GCE sensor with 40 µM AA while, in the absence of AA, no CV response was seen (Figure 4b), further emphasizing the effective electro-chemical properties of Yb2O3.CuO@rGO/GCE as an AA sensor. Figure 4c displays EIS Nyquist plots of bare GCE, CuO/GCE, Yb2O3/GCE, Yb2O3.CuO/GCE, and Yb2O3.CuO@rGO/GCE, and a relevant equivalent circuit is presented in the inset. The Yb2O3.CuO@rGO/GCE electrode was found to have the shortest semicircle diameter, which indicates that its charge transfer resistance (Rct = 9.2 kΩ) value is lower than that of other electrodes including bare GCE (75.2 kΩ), CuO/GCE (35.2 kΩ), Yb2O3/GCE (94.7 kΩ), and Yb2O3.CuO/GCE (22.9 kΩ), which were acquired through fitting utilizing the EIS Spectrum Analyzer Software. The smallest semicircular diameter of the Yb2O3.CuO@rGO/GCE electrode suggests that the fabrication process lowered its Rct value. We therefore draw the conclusion that the Yb2O3.CuO@rGO/GCE electrode provided improved electron transfer performance than the other modified electrodes shown in Figure 4a.
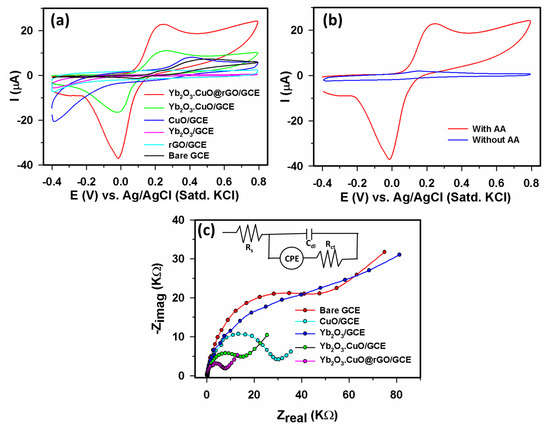
Figure 4.
CVs recorded at scan rate 0.05 Vs−1 in 0.1 M PBS (pH 7.0) (a) CVs from bare GCE, CuO/GCE, Yb2O3/GCE, rGO/GCE, Yb2O3.CuO/GCE, Yb2O3.CuO@rGO/GCE with 40 µM AA, (b) CVs from the Yb2O3.CuO@rGO/GCE with 40 µM AA and without AA, and (c) EIS Nyquist plots acquired using various electrodes in 1.0 mM [Fe(CN)6]3−/4− in 0.1 M KCl at +0.50 V, at signal amplitude 10 mV, and with frequency ranging from 0.1 Hz to 100 KHz with a relevant equivalent circuit in the inset.
We investigated the impact of a pH between 6.0 and 8.0 with 40 µM AA to better understand the electrochemical AA oxidation. Figure 5a,b shows that, for a pH of 6.0 to 7.0, the anodic peak current (Ipa) value steadily increased and, for a pH 7.0 to 8.0, a declining trend was seen. The extreme Ipa was seen at a pH ~ 7.0, as shown in Figure 5b. As a result, pH 7.0 was set as the standard for the remaining tests in this paper. Figure 5c displayed a straight-line plot for anodic peak potential (Epa) vs. pH having a regression Equation (1):
Epa(V) = 0.5614 − 0.0467pH (R2 = 0.9750)
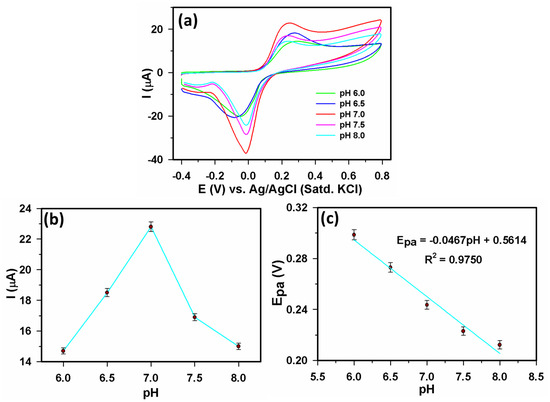
Figure 5.
(a) CVs recorded using 40 µM AA in 0.1 M PBS at varying pH (6.0–8.0) at 0.05 Vs−1 scan rate, (b) Ipa vs. pH, and (c) Epa vs. pH.
Figure 5c showed that the gradient of −56 mV per pH unit over the selected pH range is extremely near to the predicted value of −59, demonstrating that the quantity of transported protons and electrons involved in this AA oxidation are equal [13,19].
Scan rate (v) analysis in Figure 6a shows the CVs of 40 µM AA acquired using different scan rates (20–200 mVs−1) using a Yb2O3.CuO@rGO/GCE sensor. The Ipa value in Figure 6a was rising as v increased, although the Epa value only marginally changed in a positive way. The nonlinear change in Ipa vs. v in Figure 6b suggested that AA oxidation is not a surface-controlled process [59] while, in Figure 6c, a linear Ipa vs. v1/2 curve was seen, validating a diffusion-controlled process [60] using Equation (2) below.
Ipa(µA) = 190.3043 v1/2 (V1/2s−1/2) − 9.5808 (R2 = 0.9978)
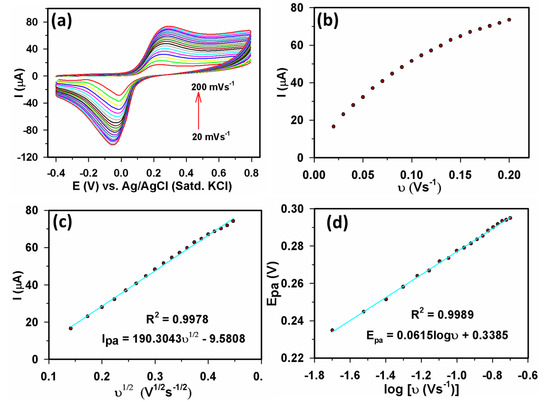
Figure 6.
Investigation of scan rate effect of Yb2O3.CuO@rGO/GCE sensor: (a) CVs recorded at different scan rates (20–200 mVs−1) with 40 µM AA in 0.1 M PBS (b) Ipa vs. v, (c) Ipa vs. , and (d) Epa vs. log(v).
Additionally, in Figure 6d, a straight line from Epa vs. log(v) plot was seen using Equation (3).
Epa (V) = 0.0615 log[v (Vs−1)] + 0.3385 (R2 = 0.9989)
Figure 6a exhibited that for v > 70 mVs−1, the value of [Epa − Epc]/2 remained essentially unchanged. Hence, at 100 mVs−1 scan rate, the [Epa − Epc]/2 value assume to be 90.5/nα mV [61], consequently, it was determined that there were 2.29 ≈ 2 transferred electrons (nα). Therefore, it is established that AA oxidation at the Yb2O3.CuO@rGO/GCE surface was a two-electron-transfer system. Overall, the scan rate and pH investigations determined that AA oxidation at the Yb2O3.CuO@rGO/GCE surface is a combined two-electrons and two-protons reaction, which is consistent with the literature [13].
3.2.2. Sensor Parameters Determination
We used amperometry for evaluating the sensor performance of the Yb2O3.CuO@rGO/GCE sensor. An amperometric response was acquired at +0.3 V after adding AA of varying concentrations (0.5–1744 µM) at consecutive time intervals. Figure 7a displays the amperometric responses achieved from the Yb2O3.CuO@rGO/GCE sensor for AA additions. Here, the current response in each AA addition increased to around 95% of its maximum current in just 4 s. Figure 7b shows a linear segment of calibration plot for 0.5–1744 µM AA using the Equation (4).
I (μA) = 0.0214 [AA] (µM) + 0.1527 (R2 = 0.9989)
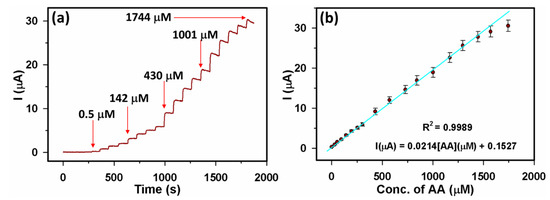
Figure 7.
(a) Yb2O3.CuO@rGO/GCE sensor’s amperometric response for AA (0.5–1744 µM) at +0.3 V potential, and (b) related calibration plot.
As a result, the Yb2O3.CuO@rGO/GCE sensor’s linear detection range (LDR) was determined to be 0.5–1571 μM. Additionally, the Yb2O3.CuO@rGO/GCE sensor’s estimated sensitivity value was found to be 0.4341 μAμM−1cm−2 and limit of detection (LOD) and limit of quantification (LOQ) were determined to be ~0.062 μM (S/N = 3) and 0.1887 µM, respectively. The sensitivity was calculated using the equation, sensitivity = S/Aeff [62], where Aeff stands for the surface area of the modified electrode (0.0493 cm2), as provided in the electronic Supplementary Materials of Figure S1 [19,63,64]. The equations were used to calculate LOD and LOQ are LOD = 3.3(Sb/S) and LOQ = 10(Sb/S), respectively [65]; here, Sb (0.000403) stands for relative standard deviation (RSD) related to five blank responses, and S stands for calibration curve’s slope.
The electrocatalytic performance is dependent on two variables: (i) increase in Ipa and (ii) decreased Epa. Hence, attempts have been made to improve the electrocatalytic activity of GCEs by fabricating them using Yb2O3.CuO@rGO NC. The achieved results showed that the Yb2O3.CuO@rGO/GCE sensor successfully satisfied both of the aforementioned requirements. Figure 4a showed a substantial negative shift of Epa and a significant increase in Ipa from the Yb2O3.CuO@rGO/GCE sensor compared to other electrodes used in this study. We achieved about a three-fold Ipa from the Yb2O3.CuO@rGO/GCE compared to a bare GCE during AA oxidation.
3.2.3. Selectivity, Repeatability, Reproducibility, and Stability
To test the Yb2O3.CuO@rGO/GCE sensor’s selectivity, we used common interfering chemicals such as uric acid (UA), glucose (Glc), citric acid (CA), dopamine (DA), Cl- ions, and NO3- ions. Here, 90 μM AA and an equal concentration of each interfering chemical were used to record the amperometric response (Figure 8a). While AA addition generated a significant amperometric response, no response was observed for the interfering chemicals. This confirms the selectivity of the Yb2O3.CuO@rGO/GCE assembly during the AA detection. Furthermore, the various sensor characteristics of Yb2O3.CuO@rGO/GCE were also investigated using CV with 40 M AA. A freshly fabricated Yb2O3.CuO@rGO/GCE assembly was employed to measure 40 M AA for the repeatability study shown in Figure 8b. Five runs with a 4.2% RSD and with nearly similar CV responses showed good repeatability. Figure 8c showed the reproducibility study of the Yb2O3.CuO@rGO/GCE assembly that used five newly modified Yb2O3.CuO@rGO/GCE electrodes (E1–E5). The Ipa variations in CV responses revealed a 4.7% RSD, demonstrating remarkable reproducibility. In addition, we recorded CV responses every fourth day for a newly modified Yb2O3.CuO@rGO/GCE sensor to assess its stability while keeping it at room temperature. Figure 8d displays the stability investigation bar graph. It demonstrates that the Ipa value in CVs was retained at approximately 81% of its initial value after being stored for 20 days at ambient conditions and that the Yb2O3.CuO@rGO/GCE sensor surface remained undamaged.
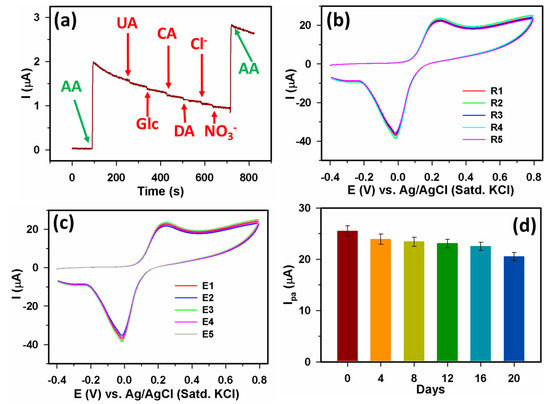
Figure 8.
(a) Amperometric (i–t) response at +0.3 V from Yb2O3.CuO@rGO/GCE sensor upon successive additions of 90 µM of AA, UA, Glc, CA, DA, Cl−, NO3−, and AA, (b) repeatability, (c) reproducibility, and (d) stability investigations.
When the AA molecule touches the Yb2O3.CuO@rGO surface, an electro-oxidation reaction occurs. AA molecules release electrons to the conduction-band of the Yb2O3.CuO@rGO nanocomposite that ultimately enhance the conductivity of the Yb2O3.CuO@rGO/GCE sensor and, hence, an enhanced CV response can be obtained. In comparison to other AA sensors, the Yb2O3.CuO@rGO/GCE sensor demonstrated a greater sensitivity for AA detection (Table 1) [13,17,39,65,66,67,68,69,70,71,72,73].

Table 1.
Comparative ascorbic acid sensor performance employing various electrodes.
Considering the experimental findings stated above, we may say that AA oxidation at the Yb2O3.CuO@rGO NC is a combined two-electrons and two-protons transfer reaction and, in this AA oxidation, the Yb2O3.CuO@rGO NC is exceedingly active. The Yb2O3.CuO@rGO/GCE sensor’s appropriateness in detecting AA can be attributed to the effective electrode-analyte interaction. Scheme 1 shows a concise model for electrochemical AA oxidation at this novel Yb2O3.CuO@rGO/GCE sensor.
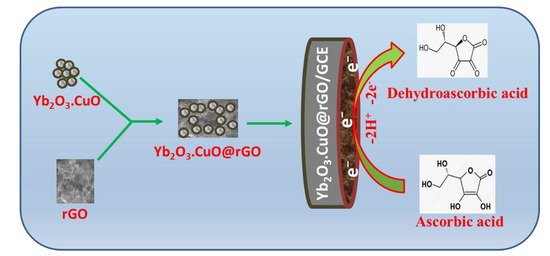
Scheme 1.
Schematic representation for Yb2O3.CuO@rGO/GCE−based ascorbic acid sensor.
3.3. Analyses of Real Samples: AA Detection from Blood Serum and Vitamin C Tablet
The developed suggested Yb2O3.CuO@rGO/GCE sensor’s efficacy was tested by measuring AA in blood serums and vitamin C tablets utilizing the standard addition method. Firstly, we measured the Yb2O3.CuO@rGO/GCE sensor’s (i–t) response at +0.3 V in 10 mL PBS with 200 µL of undiluted blood serum (BS1) and then three repeated injections of 50 µL 0.01 M AA. Such processes were carried out three times under the same circumstances. Next, we performed the same standard addition procedure using the second blood serum (BS2). Furthermore, we used a dissolved Vitamin C 1000 tablet (Vit-C) from Dallah Pharma Factory, KSA as the real sample, as in our previous report [13]. Finally, we repeated the whole standard addition process using 100 µL of Vitamin C and then three repeated injections of 100 µL 0.01 M AA. Table 2 summarizes the outcomes of the real sample investigations. These results indicate that, with approximately 100% quantitative recovery, this novel Yb2O3.CuO@rGO/GCE sensor can be utilized to precisely assess the presence of AA in real samples. Additionally, the measured level of AA in blood serums is within AA levels typically found in adults (28.5–85.2 µM) [7] and, for the Vitamin C tablets, the calculated AA amount was 98.1% of the manufacturer’s specification, confirming that the newly-developed Yb2O3.CuO@rGO/GCE sensor is appropriately validated.

Table 2.
AA Detection from commercial vitamin C tablets and blood serums (BS1 and BS2) using the Yb2O3.CuO@rGO/GCE sensor.
4. Conclusions
Herein, we successfully synthesized and characterized the Yb2O3.CuO@rGO nanocomposite. This nanocomposite material was then used to design a sensitive, selective, and reusable electrochemical AA sensor. This AA sensor was developed by a facile technique and is able to measure both high and low levels of AA because of its broad linear dynamic range and high sensitivity. Additionally, this AA sensor demonstrated a minimal interference effect, a fast response time, a reasonable limit of detection, excellent stability, reproducibility, and repeatability. These features make it a promising tool for detecting AA. To further validate the Yb2O3.CuO@rGO/GCE sensor’s accuracy, it was tested utilizing blood serums and vitamin C tablets, and the results were consistent and encouraging. Overall, the method of sensor fabrication presented in this study offers a promising platform for developing a highly efficient AA sensor in the future.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/bios13060588/s1, Figure S1: (a) CVs recorded with 5 mM [Fe(CN)6]3−/4− in 0.1 M KCl using the Yb2O3.CuO@rGO/GCE assembly for scan rates ranging from 20 to 120 mVs−1 (b) Ipa vs. ν1/2.
Author Contributions
Conceptualization, J.A. and F.A.H.; methodology, J.A., M.F., J.S.A. and M.A.A.; validation, J.A., M.F., J.S.A., M.A.A. and S.A.A.; formal analysis, J.A., M.F. and J.S.A.; investigation, J.A., M.A.A. and F.A.H.; resources, J.S.A. and F.A.H.; data curation, J.A., M.F., J.S.A., M.A.A. and F.A.H.; writing—original draft preparation, J.A.; writing—review and editing, M.F., J.S.A., M.A.A., S.A.A. and F.A.H.; supervision, F.A.H.; project administration, J.S.A. and F.A.H.; funding acquisition, M.A.A. and F.A.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the grant no. (NU/IFC/2/SERC/-/15) under the Institutional Funding Committee at Najran University, Kingdom of Saudi Arabia.
Institutional Review Board Statement
The study was conducted according to the guidelines of the ethical standards and approved by the Research Ethics Committee of Najran University (reference No. 010509-023062-DS).
Informed Consent Statement
The verbal informed consent of the participants was obtained.
Data Availability Statement
Data will be available upon request.
Acknowledgments
The authors would like to acknowledge the support of the Deputyship for Research and Innovation-Ministry of Education, Kingdom of Saudi Arabia for this research through a grant (NU/IFC/2/SERC/-/15) under the Institutional Funding Committee at Najran University, Kingdom of Saudi Arabia.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bilal, S.; Akbar, A.; Shah, A.U.H.A. Highly Selective and Reproducible Electrochemical Sensing of Ascorbic Acid through a Conductive Polymer Coated Electrode. Polymers 2019, 11, 346. [Google Scholar] [CrossRef]
- Qu, C.; Li, H.; Zhou, S.; Li, G.; Wang, C.; Snyders, R.; Bittencourt, C.; Li, W. Bi2S3/RGO Composite Based Electrochemical Sensor for Ascorbic Acid Detection. Chemosensors 2021, 9, 190. [Google Scholar] [CrossRef]
- Tkachenko, A.B.; Onizhuk, M.O.; Tkachenko, O.S.; Arenas, L.T.; Benvenutti, E.V.; Gushikem, Y.; Panteleimonov, A.V. An Electrochemical Sensor Based on Graphite Electrode Modified with Silica Containing 1-n-Propyl-3-Methylimidazolium Species for Determination of Ascorbic Acid. Methods Objects Chem. Anal. 2019, 14, 5–14. [Google Scholar] [CrossRef]
- Chen, F.; Li, Q.; Yu, Y.; Yang, W.; Shi, F.; Qu, Y. Association of Vitamin C, Vitamin D, Vitamin E and Risk of Bladder Cancer: A Dose-Response Meta-Analysis. Sci. Rep. 2015, 5, 9599. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Jiang, X.; Yang, M. Electrochemical Sensing of L-Ascorbic Acid by Using a Glassy Carbon Electrode Modified with a Molybdophosphate Film. Microchim. Acta 2019, 186, 445. [Google Scholar] [CrossRef] [PubMed]
- Mandl, J.; Szarka, A.; Bánhegyi, G. Vitamin C: Update on Physiology and Pharmacology. Br. J. Pharmacol. 2009, 157, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Hagel, A.F.; Albrecht, H.; Dauth, W.; Hagel, W.; Vitali, F.; Ganzleben, I.; Schultis, H.W.; Konturek, P.C.; Stein, J.; Neurath, M.F.; et al. Plasma Concentrations of Ascorbic Acid in a Cross Section of the German Population. J. Int. Med. Res. 2018, 46, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Massey, L.K.; Liebman, M.; Kynast-Gales, S.A. Ascorbate Increases Human Oxaluria and Kidney Stone Risk. J. Nutr. 2005, 135, 1673–1677. [Google Scholar] [CrossRef]
- Lenghor, N.; Jakmunee, J.; Vilen, M.; Sara, R.; Christian, G.D.; Grudpan, K. Sequential Injection Redox or Acid-Base Titration for Determination of Ascorbic Acid or Acetic Acid. Talanta 2002, 58, 1139–1144. [Google Scholar] [CrossRef]
- Klimczak, I.; Gliszczyńska-Świgło, A. Comparison of UPLC and HPLC Methods for Determination of Vitamin C. Food Chem. 2015, 175, 100–105. [Google Scholar] [CrossRef]
- Gómez Ruiz, B.; Roux, S.; Courtois, F.; Bonazzi, C. Spectrophotometric Method for Fast Quantification of Ascorbic Acid and Dehydroascorbic Acid in Simple Matrix for Kinetics Measurements. Food Chem. 2016, 211, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Morosanova, M.A.; Morosanova, E.I. Silica-Titania Xerogel Doped with Mo,P-Heteropoly Compounds for Solid Phase Spectrophotometric Determination of Ascorbic Acid in Fruit Juices, Pharmaceuticals, and Synthetic Urine. Chem. Cent. J. 2017, 11, 3. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Faisal, M.; Harraz, F.A.; Jalalah, M.; Alsareii, S.A. Porous Silicon-Mesoporous Carbon Nanocomposite Based Electrochemical Sensor for Sensitive and Selective Detection of Ascorbic Acid in Real Samples. J. Taiwan Inst. Chem. Eng. 2021, 125, 360–371. [Google Scholar] [CrossRef]
- Hameed, S.; Munawar, A.; Khan, W.S.; Mujahid, A.; Ihsan, A.; Rehman, A.; Ahmed, I.; Bajwa, S.Z. Assessing Manganese Nanostructures Based Carbon Nanotubes Composite for the Highly Sensitive Determination of Vitamin C in Pharmaceutical Formulation. Biosens. Bioelectron. 2017, 89, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Arabali, V.; Ebrahimi, M.; Abbasghorbani, M.; Gupta, V.K.; Farsi, M.; Ganjali, M.R.; Karimi, F. Electrochemical Determination of Vitamin C in the Presence of NADH Using a CdO Nanoparticle/Ionic Liquid Modified Carbon Paste Electrode as a Sensor. J. Mol. Liq. 2016, 213, 312–316. [Google Scholar] [CrossRef]
- Tadayon, F.; Vahed, S.; Bagheri, H. Au-Pd/Reduced Graphene Oxide Composite as a New Sensing Layer for Electrochemical Determination of Ascorbic Acid, Acetaminophen and Tyrosine. Mater. Sci. Eng. C 2016, 68, 805–813. [Google Scholar] [CrossRef]
- Yan, C.; Liu, X.; Zhang, R.; Chen, Y.; Wang, G. A Selective Strategy for Determination of Ascorbic Acid Based on Molecular Imprinted Copolymer of O-Phenylenediamine and Pyrrole. J. Electroanal. Chem. 2016, 780, 276–281. [Google Scholar] [CrossRef]
- Ahmed, J.; Faisal, M.; Alsareii, S.A.; Harraz, F.A. Highly Sensitive and Selective Non-Enzymatic Uric Acid Electrochemical Sensor Based on Novel Polypyrrole-Carbon Black-Co3O4 Nanocomposite. Adv. Compos. Hybrid Mater. 2022, 5, 920–933. [Google Scholar] [CrossRef]
- Ahmed, J.; Faisal, M.; Jalalah, M.; Alsaiari, M.; Alsareii, S.A.; Harraz, F.A. An Efficient Amperometric Catechol Sensor Based on Novel Polypyrrole-Carbon Black Doped α-Fe2O3 Nanocomposite. Colloids Surfaces A Physicochem. Eng. Asp. 2021, 619, 126469. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ahmed, J.; Asiri, A.M.; Alamry, K.A. Fabrication of a Hydrazine Chemical Sensor Based on Facile Synthesis of Doped NZO Nanostructure Materials. New J. Chem. 2020, 44, 13018–13029. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ahmed, J.; Asiri, A.M.; Alfaifi, S.Y. Ultra-Sensitive, Selective and Rapid Carcinogenic Bisphenol A Contaminant Determination Using Low-Dimensional Facile Binary Mg-SnO2 Doped Microcube by Potential Electro-Analytical Technique for the Safety of Environment. J. Ind. Eng. Chem. 2022, 109, 147–154. [Google Scholar] [CrossRef]
- Mahmoud, B.G.; Khairy, M.; Rashwan, F.A.; Foster, C.W.; Banks, C.E. Self-Assembly of Porous Copper Oxide Hierarchical Nanostructures for Selective Determinations of Glucose and Ascorbic Acid. RSC Adv. 2016, 6, 14474–14482. [Google Scholar] [CrossRef]
- Wang, X.; Han, Q.; Cai, S.; Wang, T.; Qi, C.; Yang, R.; Wang, C. Excellent Peroxidase Mimicking Property of CuO/Pt Nanocomposites and Their Application as an Ascorbic Acid Sensor. Analyst 2017, 142, 2500–2506. [Google Scholar] [CrossRef]
- Šljukić, B.R.; Kadara, R.O.; Banks, C.E. Disposable Manganese Oxide Screen Printed Electrodes for Electroanalytical Sensing. Anal. Methods 2011, 3, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Xie, A.; Wei, B.; Tao, X.; Zhang, J.; Peng, W.; Liu, C.; Gu, L.; Xu, C.; Luo, S. Construction and Application of a Nonenzymatic Ascorbic Acid Sensor Based on a NiO1.0/Polyaniline3.0 hybrid. New J. Chem. 2020, 44, 9288–9297. [Google Scholar] [CrossRef]
- Yin, Y.; Zhao, J.; Qin, L.; Yang, Y.; He, L. Synthesis of an Ordered Nanoporous Fe2O3/Au Film for Application in Ascorbic Acid Detection. RSC Adv. 2016, 6, 63358–63364. [Google Scholar] [CrossRef]
- Pan, Y.; Zuo, J.; Hou, Z.; Huang, Y.; Huang, C. Preparation of Electrochemical Sensor Based on Zinc Oxide Nanoparticles for Simultaneous Determination of AA, DA, and UA. Front. Chem. 2020, 8, 592538. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, S.; He, W.; Uyama, H.; Xie, Q.; Zhang, L.; Yang, F. Electrochemical Sensor Based on Carbon-Supported NiCoO2 Nanoparticles for Selective Detection of Ascorbic Acid. Biosens. Bioelectron. 2014, 55, 446–451. [Google Scholar] [CrossRef]
- Alam, M.M.; Asiri, A.M.; Rahman, M.M.; Islam, M.A. Selective Detection of Ascorbic Acid with Wet-Chemically Prepared CdO/SnO2/V2O5 Micro-Sheets by Electrochemical Approach. SN Appl. Sci. 2020, 2, 1953. [Google Scholar] [CrossRef]
- Xing, R.; Sheng, K.; Xu, L.; Liu, W.; Song, J.; Song, H. Three-Dimensional In2O3-CuO Inverse Opals: Synthesis and Improved Gas Sensing Properties towards Acetone. RSC Adv. 2016, 6, 57389–57395. [Google Scholar] [CrossRef]
- Purbia, R.; Kwon, Y.M.; Choi, S.Y.; Kim, S.H.; Lee, Y.S.; Ahi, Z.B.; Park, H.; Baik, J.M. A Thermodynamic Approach toward Selective and Reversible Sub-Ppm H2S Sensing Using Ultra-Small CuO Nanorods Impregnated with Nb2O5 nanoparticles. J. Mater. Chem. A 2021, 9, 17425–17433. [Google Scholar] [CrossRef]
- Hossain, R.; Hassan, K.; Sahajwalla, V. Utilising Problematic Waste to Detect Toxic Gas Release in the Environment: Fabricating a NiO Doped CuO Nanoflake Based Ammonia Sensor from e-Waste. Nanoscale Adv. 2022, 4, 4066–4079. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Shen, Z.; Jia, Q.; Zhao, J.; Zhao, Z.; Ji, H. A CuO-ZnO Nanostructured p-n Junction Sensor for Enhanced N-Butanol Detection. RSC Adv. 2016, 6, 2504–2511. [Google Scholar] [CrossRef]
- Hussain, M.M.; Hussain, M.M.; Hussain, M.M.; Asiri, A.M.; Rahman, M.M.; Rahman, M.M.; Hussain, M.M. A Non-Enzymatic Electrochemical Approach for l-Lactic Acid Sensor Development Based on CuO·MWCNT Nanocomposites Modified with a Nafion Matrix. New J. Chem. 2020, 44, 9775–9787. [Google Scholar] [CrossRef]
- Tobaldi, D.M.; Espro, C.; Leonardi, S.G.; Lajaunie, L.; Seabra, M.P.; Calvino, J.J.; Marini, S.; Labrincha, J.A.; Neri, G. Photo-Electrochemical Properties of CuO-TiO2heterojunctions for Glucose Sensing. J. Mater. Chem. C 2020, 8, 9529–9539. [Google Scholar] [CrossRef]
- Sun, G.J.; Choi, S.W.; Katoch, A.; Wu, P.; Kim, S.S. Bi-Functional Mechanism of H2S Detection Using CuO-SnO2 Nanowires. J. Mater. Chem. C 2013, 1, 5454–5462. [Google Scholar] [CrossRef]
- Rahman, M.M. Rapid and Sensitive Detection of Selective 1,2-Diaminobenzene Based on Facile Hydrothermally Prepared Doped Co3O4/Yb2O3 Nanoparticles. PLoS ONE 2021, 16, e0246756. [Google Scholar] [CrossRef]
- Rahman, M.M.; Alam, M.M.; Asiri, A.M.; Islam, M.A. Ethanol Sensor Development Based on Ternary-Doped Metal Oxides (CdO/ZnO/Yb2O3) Nanosheets for Environmental Safety. RSC Adv. 2017, 7, 22627–22639. [Google Scholar] [CrossRef]
- Rahman, M.M.; Alam, M.M.; Asiri, A.M.; Awual, M.R. Fabrication of 4-Aminophenol Sensor Based on Hydrothermally Prepared ZnO/Yb2O3 Nanosheets. New J. Chem. 2017, 41, 9159–9169. [Google Scholar] [CrossRef]
- Umar, A.; Ibrahim, A.A.; Kumar, R.; Almas, T.; Al-Assiri, M.S.; Baskoutas, S. Nitroaniline Chemi-Sensor Based on Bitter Gourd Shaped Ytterbium Oxide (Yb2O3) Doped Zinc Oxide (ZnO) Nanostructures. Ceram. Int. 2019, 45, 13825–13831. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, L.; Sun, F.; Li, T.; Zhang, T.; Qin, S. Humidity-Insensitive NO2 Sensors Based on SnO2/RGO Composites. Front. Chem. 2021, 9, 681313. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Hawari, H.F.; Kumar, P.; Burhanudin, Z.A.; Tansu, N. Functionalized Reduced Graphene Oxide Thin Films for Ultrahigh Co2 Gas Sensing Performance at Room Temperature. Nanomaterials 2021, 11, 623. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Chen, X.; Wang, T.; Li, B.; Zeng, M.; Yang, J.; Hu, N.; Su, Y.; Zhou, Z.; Yang, Z. Enhancing Room-Temperature NO2 gas Sensing Performance Based on a Metal Phthalocyanine/Graphene Quantum Dot Hybrid Material. RSC Adv. 2021, 11, 5618–5628. [Google Scholar] [CrossRef] [PubMed]
- Feizollahi, A.; Rafati, A.A.; Assari, P.; Asadpour Joghani, R. Development of an Electrochemical Sensor for the Determination of Antibiotic Sulfamethazine in Cow Milk Using Graphene Oxide Decorated with Cu-Ag Core-Shell Nanoparticles. Anal. Methods 2021, 13, 910–917. [Google Scholar] [CrossRef]
- Yu, H.; Guo, W.; Lu, X.; Xu, H.; Yang, Q.; Tan, J.; Zhang, W. Reduced Graphene Oxide Nanocomposite Based Electrochemical Biosensors for Monitoring Foodborne Pathogenic Bacteria: A Review. Food Control 2021, 127, 108117. [Google Scholar] [CrossRef]
- Fauzi, A.S.A.; Hamidah, N.L.; Kitamura, S.; Kodama, T.; Sonda, K.; Putri, G.K.; Shinkai, T.; Ahmad, M.S.; Inomata, Y.; Quitain, A.T.; et al. Electrochemical Detection of Ethanol in Air Using Graphene Oxide Nanosheets Combined with Au-WO3. Sensors 2022, 22, 3194. [Google Scholar] [CrossRef]
- Wu, S.; Liang, Y.; Xu, Y.; Tong, Y.; Chen, Y.; Chen, X. Graphene-Based Electrochemical Sensor for Detection of Hepatocellular Carcinoma Markers. Front. Chem. 2022, 10, 883627. [Google Scholar] [CrossRef]
- Boroujerdi, R.; Paul, R. Graphene-Based Electrochemical Sensors for Psychoactive Drugs. Nanomaterials 2022, 12, 2250. [Google Scholar] [CrossRef]
- Fu, L.; Mao, S.; Chen, F.; Zhao, S.; Su, W.; Lai, G.; Yu, A.; Lin, C.-T. Graphene-Based Electrochemical Sensors for Antibiotic Detection in Water, Food and Soil: A Scientometric Analysis in CiteSpace (2011–2021). Chemosphere 2022, 297, 134127. [Google Scholar] [CrossRef]
- Saravanan, T.; Anandan, P.; Shanmugam, M.; Azhagurajan, M.; Mohamed Ismail, M.; Arivanandhan, M.; Hayakawa, Y.; Jayavel, R. Facile Synthesis of Yb2O3–Graphene Nanocomposites for Enhanced Energy and Environmental Applications. Polym. Bull. 2020, 77, 3891–3906. [Google Scholar] [CrossRef]
- Aftab, U.; Tahira, A.; Mazzaro, R.; Abro, M.I.; Baloch, M.M.; Willander, M.; Nur, O.; Yu, C.; Ibupoto, Z.H. The Chemically Reduced CuO-Co3O4 Composite as a Highly Efficient Electrocatalyst for Oxygen Evolution Reaction in Alkaline Media. Catal. Sci. Technol. 2019, 9, 6274–6284. [Google Scholar] [CrossRef]
- Baig, N.; Saleh, T.A. Superhydrophobic Polypropylene Functionalized with Nanoparticles for Efficient Fast Static and Dynamic Separation of Spilled Oil from Water. Glob. Chall. 2019, 3, 1800115. [Google Scholar] [CrossRef] [PubMed]
- Gongalsky, M.B.; Kargina, J.V.; Cruz, J.F.; Sánchez-Royo, J.F.; Chirvony, V.S.; Osminkina, L.A.; Sailor, M.J. Formation of Si/SiO2 Luminescent Quantum Dots From Mesoporous Silicon by Sodium Tetraborate/Citric Acid Oxidation Treatment. Front. Chem. 2019, 7, 165. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Zhang, P.; Dai, H.; Chen, L.; Ma, H.; Lin, M.; Shen, D. An Electrochemical Sensor Based on Co3O4 Nanosheets for Lead Ions Determination. RSC Adv. 2017, 7, 39611–39616. [Google Scholar] [CrossRef]
- Eslami, A.; Juibari, N.M.; Hosseini, S.G.; Abbasi, M. Synthesis and Characterization of CuO Nanoparticles by the Chemical Liquid Deposition Method and Investigation of Its Catalytic Effect on the Thermal Decomposition of Ammonium Perchlorate. Cent. Eur. J. Energetic Mater. 2017, 14, 152–168. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ahmed, J.; Asiri, A.M. Ultra-Sensitive, Selective, and Rapid Carcinogenic 1,2-Diaminobenzene Chemical Determination Using Sol–Gel Coating Low-Dimensional Facile CuS Modified-CNT Nanocomposites by Electrochemical Approach. Microchem. J. 2022, 175, 107230. [Google Scholar] [CrossRef]
- Cao, Z.; An, S.; Song, X. Effect of Thermal Treatment at High Temperature on Phase Stability and Transformation of Yb2O3 and Y2O3 Co-Doped ZrO2 Ceramics. Sci. Rep. 2022, 12, 9955. [Google Scholar] [CrossRef]
- Wang, J.; Salihi, E.C.; Šiller, L. Green Reduction of Graphene Oxide Using Alanine. Mater. Sci. Eng. C 2017, 72, 1–6. [Google Scholar] [CrossRef]
- El-Raheem, H.A.; Hassan, R.Y.A.; Khaled, R.; Farghali, A.; El-Sherbiny, I.M. New Sensing Platform of Poly(Ester-Urethane)Urea Doped with Gold Nanoparticles for Rapid Detection of Mercury Ions in Fish Tissue. RSC Adv. 2021, 11, 31845–31854. [Google Scholar] [CrossRef]
- Ahmed, J.; Rahman, M.M.; Siddiquey, I.A.; Asiri, A.M.; Hasnat, M.A. Efficient Bisphenol-A Detection Based on the Ternary Metal Oxide (TMO) Composite by Electrochemical Approaches. Electrochim. Acta 2017, 246, 597–605. [Google Scholar] [CrossRef]
- Ahmed, J.; Rahman, M.M.; Siddiquey, I.A.; Asiri, A.M.; Hasnat, M.A. Efficient Hydroquinone Sensor Based on Zinc, Strontium and Nickel Based Ternary Metal Oxide (TMO) Composites by Differential Pulse Voltammetry. Sens. Actuators B Chem. 2018, 256, 383–392. [Google Scholar] [CrossRef]
- Ahmed, J.; Faisal, M.; Jalalah, M.; Alsareii, S.A.; Harraz, F.A. Novel Polypyrrole-Carbon Black Doped ZnO Nanocomposite for Efficient Amperometric Detection of Hydroquinone. J. Electroanal. Chem. 2021, 898, 115631. [Google Scholar] [CrossRef]
- Ahmed, J.; Faisal, M.; Alsareii, S.A.; Jalalah, M.; Harraz, F.A. A Novel Gold-Decorated Porous Silicon-Poly(3-Hexylthiophene) Ternary Nanocomposite as a Highly Sensitive and Selective Non-Enzymatic Dopamine Electrochemical Sensor. J. Alloys Compd. 2023, 931, 167403. [Google Scholar] [CrossRef]
- Ahmed, J.; Rashed, M.A.; Faisal, M.; Harraz, F.A.; Jalalah, M.; Alsareii, S.A. Novel SWCNTs-Mesoporous Silicon Nanocomposite as Efficient Non-Enzymatic Glucose Biosensor. Appl. Surf. Sci. 2021, 552, 149477. [Google Scholar] [CrossRef]
- Ahmed, J.; Faisal, M.; Alsareii, S.A.; Jalalah, M.; Alsaiari, M.; Harraz, F.A. Mn2O3 Nanoparticle-Porous Silicon Nanocomposite Based Amperometric Sensor for Sensitive Detection and Quantification of Acetaminophen in Real Samples. Ceram. Int. 2023, 49, 933–943. [Google Scholar] [CrossRef]
- Xu, C.; Liu, B.; Ning, W.; Wang, X. Highly Sensitive Ascorbic Acid Sensor Based on Ionic Liquid Functionalized Graphene Oxide Nanocomposite. Int. J. Electrochem. Sci. 2019, 14, 1670–1683. [Google Scholar] [CrossRef]
- Roy, P.R.; Saha, M.S.; Okajima, T.; Ohsaka, T. Electrooxidation and Amperometric Detection of Ascorbic Acid at GC Electrode Modified by Electropolymerization of N,N-Dimethylaniline. Electroanalysis 2004, 16, 289–297. [Google Scholar] [CrossRef]
- Kong, Y.; Shan, X.; Ma, J.; Chen, M.; Chen, Z. A Novel Voltammetric Sensor for Ascorbic Acid Based on Molecularly Imprinted Poly(o-Phenylenediamine-Co-o-Aminophenol). Anal. Chim. Acta 2014, 809, 54–60. [Google Scholar] [CrossRef]
- Salahandish, R.; Ghaffarinejad, A.; Naghib, S.M.; Niyazi, A.; Majidzadeh-A, K.; Janmaleki, M.; Sanati-Nezhad, A. Sandwich-Structured Nanoparticles-Grafted Functionalized Graphene Based 3D Nanocomposites for High-Performance Biosensors to Detect Ascorbic Acid Biomolecule. Sci. Rep. 2019, 9, 1226. [Google Scholar] [CrossRef]
- Qi, S.; Zhao, B.; Tang, H.; Jiang, X. Determination of Ascorbic Acid, Dopamine, and Uric Acid by a Novel Electrochemical Sensor Based on Pristine Graphene. Electrochim. Acta 2015, 161, 395–402. [Google Scholar] [CrossRef]
- Zhu, H.; Xu, G. Electrochemical Determination of Ascorbic Acid Based on Hydrothermal Synthesized ZnO Nanoparticles. Int. J. Electrochem. Sci. 2017, 12, 3873–3882. [Google Scholar] [CrossRef]
- Yang, L.; Liu, D.; Huang, J.; You, T. Simultaneous Determination of Dopamine, Ascorbic Acid and Uric Acid at Electrochemically Reduced Graphene Oxide Modified Electrode. Sens. Actuators B Chem. 2014, 193, 166–172. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, N.; Zhang, L.; Wang, H.; Shi, H.; Liu, Q. Simultaneous Voltammetric Detection of Dopamine, Ascorbic Acid and Uric Acid Using a Poly(2-(N-Morpholine)Ethane Sulfonic Acid)/RGO Modified Electrode. RSC Adv. 2018, 8, 5280–5285. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Furtado, V.L.; Gonçalves, J.M.; Bannitz-Fernandes, R.; Netto, L.E.S.; Araki, K.; Bertotti, M. Amperometric Microsensor Based on Nanoporous Gold for Ascorbic Acid Detection in Highly Acidic Biological Extracts. Anal. Chim. Acta 2020, 1095, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Du, X. Sensitive Electrochemical Sensors for Simultaneous Determination of Ascorbic Acid, Dopamine, and Uric Acid Based on Au@Pd-Reduced Graphene Oxide Nanocomposites. Nanoscale 2014, 6, 11303–11309. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).