Complementary Strategy Enhancing Broad-Specificity for Multiplexed Immunoassay of Adulterant Sulfonylureas in Functional Food
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Instruments
2.3. Buffers and Solutions
2.4. Conjugation
2.4.1. Hapten 1-OVA/BSA
2.4.2. Hapten 2-OVA/BSA
2.4.3. Hapten 3-OVA
2.4.4. Hapten 4-OVA/BSA
2.5. Antibody Generation
2.6. ELISA Procedure
2.7. Specificity
2.8. Sample Preparation
2.9. Matrix Effect
2.10. Recovery and Confirmation
2.11. Analysis of Blind Samples
3. Results
3.1. Antibody Production
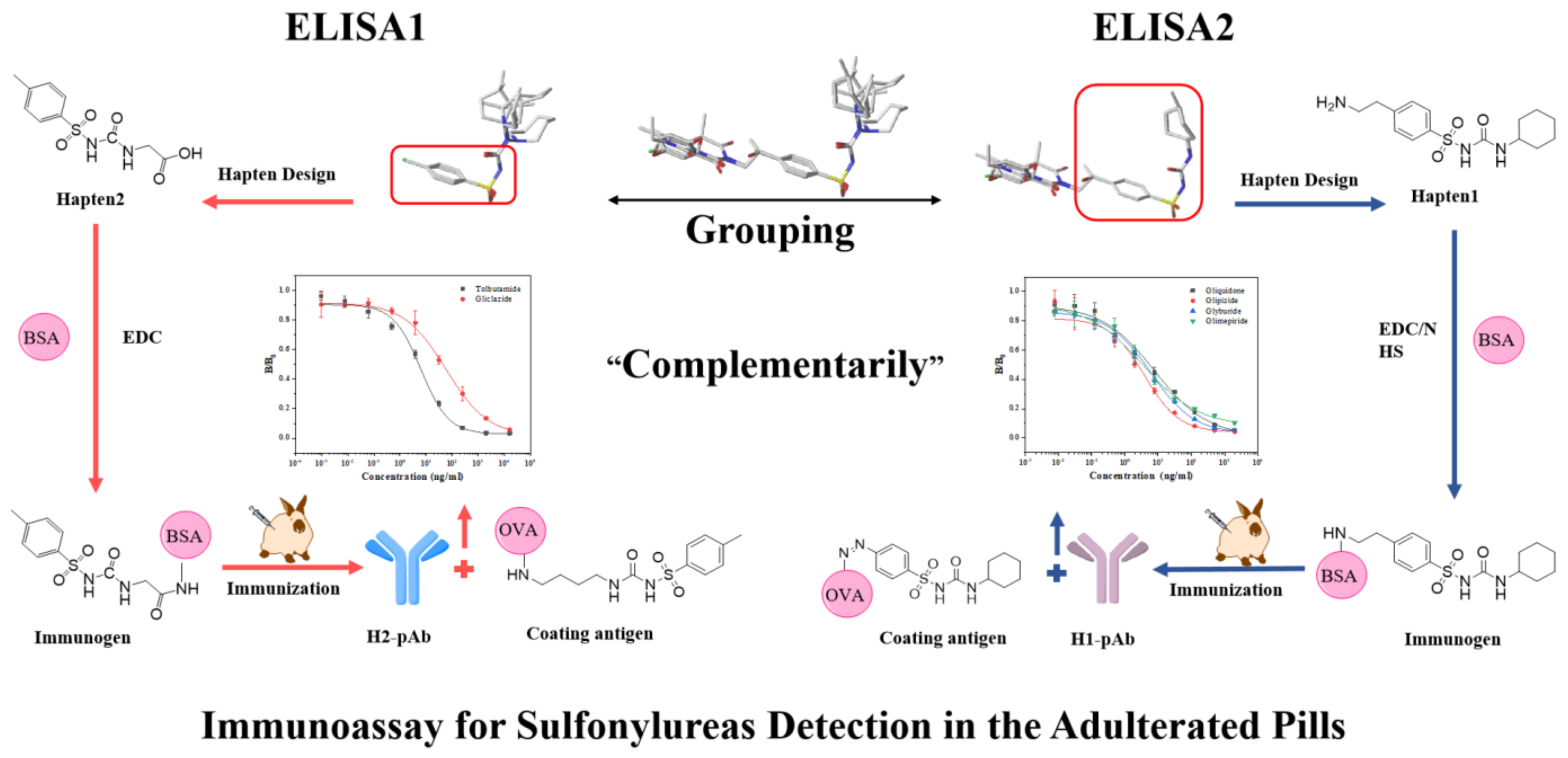
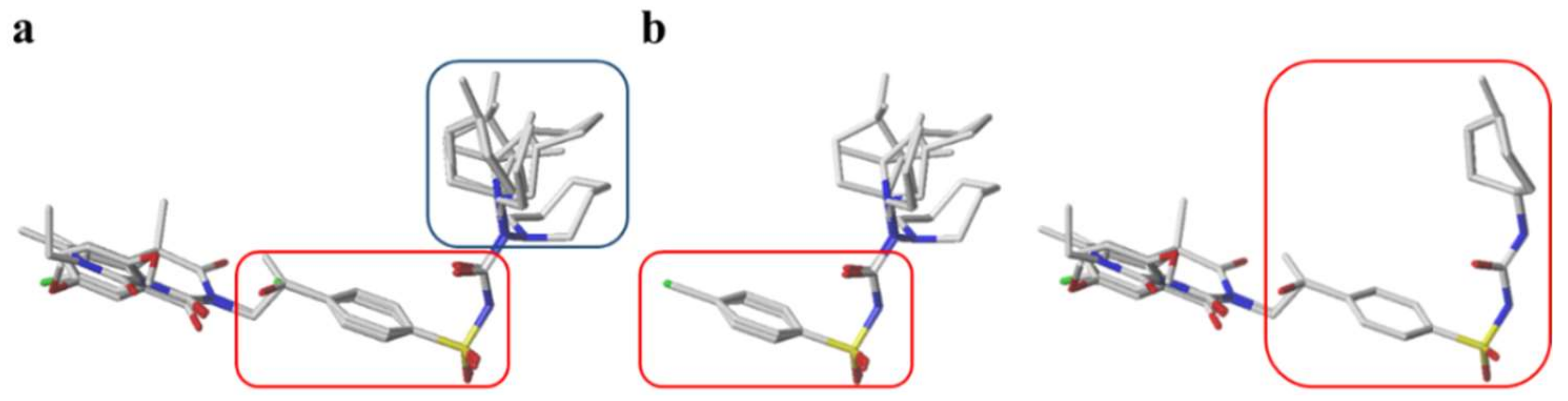
3.1.1. Hapten Design
3.1.2. Conjugate Preparation
3.1.3. ELISA Optimization
3.1.4. Broad-Specificity
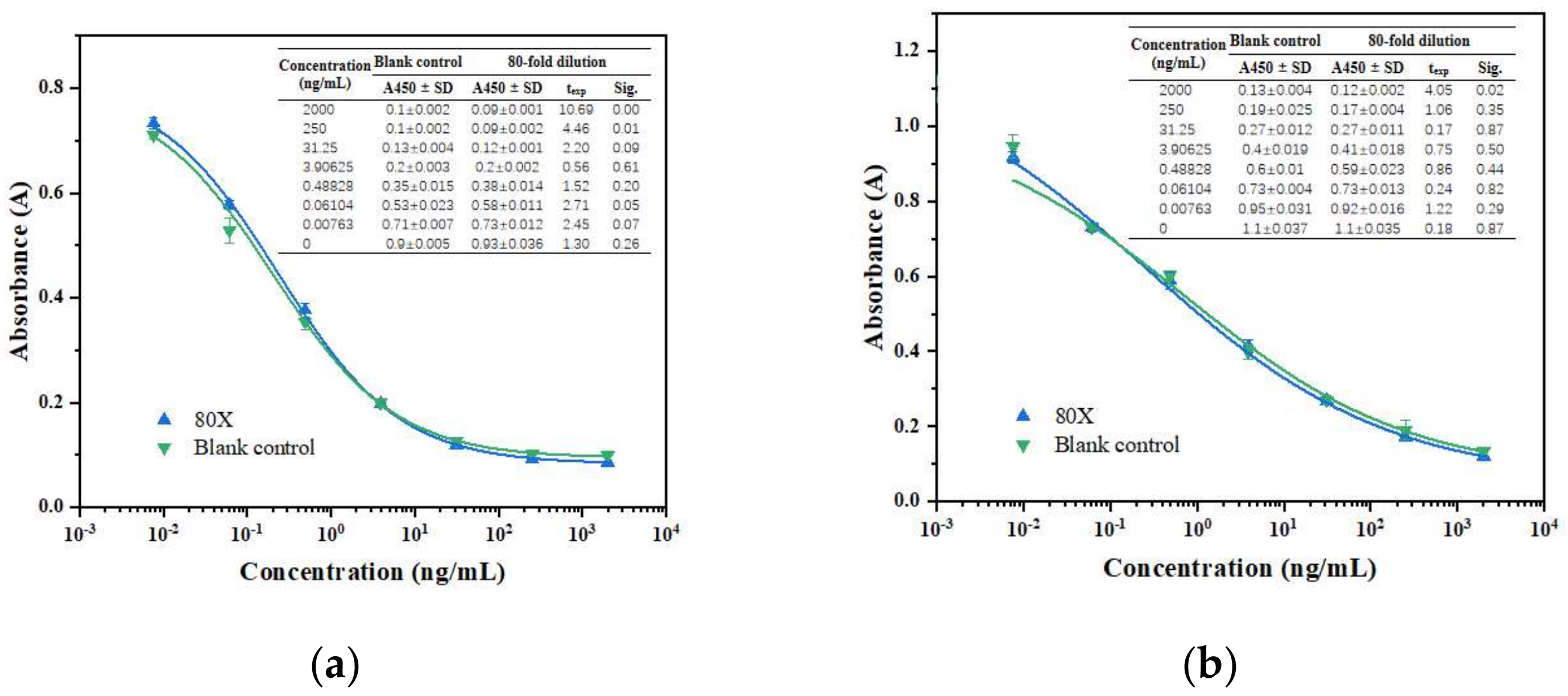
3.2. Matrix Effect
3.3. Recovery and Confirmation
3.4. Analysis of Blind Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- International Diabetes Federation. DIABETES ATLAS 9th Edition. Available online: https://diabetesatlas.org/en/resources (accessed on 18 March 2021).
- Emoto, M.; Nishizawa, Y.; Maekawa, K.; Hiura, Y.; Kanda, H.; Kawagishi, T.; Shoji, T.; Okuno, Y.; Morii, H. Homeostasis model assessment as a clinical index of insulin resistance in type 2 diabetic patients treated with sulfonylureas. Diabetes Care 1999, 22, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Mahwish Saeed, F.; Arshad, M.S.; Nisa, M.U.; Nadeem, M.T.; Arshad, M.U. Hypoglycemic and hypolipidemic effects of different parts and formulations of bitter gourd (Momordica charantia). Lipids Health Dis. 2017, 16, 211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calahan, J.; Howard, D.; Almalki, A.J.; Gupta, M.P.; Calderón, A.I. Chemical adulterants in herbal medicinal products: A review. Planta Med. 2016, 82, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Sola, D.; Rossi, L.; Schianca, G.P.C.; Maffioli, P.; Bigliocca, M.; Mella, R.; Corlianò, F.; Fra, G.P.; Bartoli, E.; Derosa, G. Sulfonylureas and their use in clinical practice. Arch. Med. Sci. 2015, 11, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Jiang, Q.; Li, R.; Xu, Q.; Li, H. Nanofibers mat as sampling module of direct analysis in real time mass spectrometry for sensitive and high-throughput screening of illegally adulterated sulfonylureas in antidiabetic health-care teas. Talanta 2019, 204, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.S.; Kim, K.Y.; Yoo, G.J.; Lee, J.H.; Park, H.N.; Park, S.-K.; Baek, S.Y. Determination of 26 anti-diabetic compounds in dietary supplements using a validated UPLC method. Food Addit. Contam. Part A-Chem. 2018, 35, 387–394. [Google Scholar] [CrossRef]
- Ma, J.; Pawar, R.S.; Grundel, E. Validation of an LC-MS/MS method for analysis of anti-diabetic drugs in botanical dietary supplements labeled for blood sugar management. Drug Test. Anal. 2018, 10, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, J.; Zhang, W.; Bai, Y.; Liu, H. Rapid screening for synthetic antidiabetic drug adulteration in herbal dietary supplements using direct analysis in real time mass spectrometry. Analyst 2011, 136, 2613–2618. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Xu, X.; Chen, G.; Bashir, K.; Shu, H.; Ge, Y.; Jing, W.; Luo, Z.; Chang, C.; Fu, Q. On-Line two dimensional liquid chromatography based on skeleton type molecularly imprinted column for selective determination of sulfonylurea additive in Chinese patent medicines or functional foods. J. Pharm. Biomed. Anal. 2017, 146, 292–301. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Yang, J.; Zou, T.; Xu, Z.; Tian, Y.; Sun, W.; Wang, H.; Sun, Y.; Lei, H.; Chen, Z.; et al. A gold nanoparticle-based immunochromatographic assay for simultaneous detection of multiplex sildenafil adulterants in health food by only one antibody. Anal. Chim. Acta. 2021, 1141, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Suryoprabowo, S.; Liu, L.; Kuang, H.; Cui, G.; Xu, C. Fluorescence based immunochromatographic sensor for rapid and sensitive detection of tadalafil and comparison with a gold lateral flow immunoassay. Food Chem. 2021, 342, 128255. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Chen, J.; Zhang, C.; Huang, X.-A.; Sun, Y.; Xu, Z.; Lei, H. Broad-Specificity Chemiluminescence Enzyme Immunoassay for (Fluoro)quinolones: Hapten Design and Molecular Modeling Study of Antibody Recognition. Anal. Chem. 2016, 88, 3909–3916. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, L.; Shen, X.; Wei, X.; Huang, X.; Liu, Y.; Sun, X.; Wang, Z.; Sun, Y.; Xu, Z. Broad-specificity immunoassay for simultaneous detection of ochratoxins A, B, and C in millet and maize. J. Agric. Food Chem. 2017, 65, 4830–4838. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Xu, Z.-L.; Yang, J.-Y.; Xiao, Z.-L.; Li, Y.-J.; Beier, R.C.; Sun, Y.-M.; Lei, H.-T.; Wang, H.; Shen, Y.-D. Synthesis of novel haptens and development of an enzyme-linked immunosorbent assay for quantification of histamine in foods. J. Agric. Food Chem. 2014, 62, 12299–12308. [Google Scholar] [CrossRef] [PubMed]
- Page, M.; Thorpe, R. Purification of IgG by precipitation with sodium sulfate or ammonium sulfate. In The Protein Protocols Handbook; Springer: Berlin/Heidelberg, Germany, 1996; pp. 721–722. [Google Scholar]
- Xie, H.; Li, Y.; Wang, J.; Lei, Y.; Koidis, A.; Li, X.; Shen, X.; Xu, Z.; Lei, H. Broad-specific immunochromatography for simultaneous detection of various sulfonylureas in adulterated multi-herbal tea. Food Chem. 2022, 370, 131055. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.-L.; Zeng, D.-P.; Yang, J.-Y.; Shen, Y.-D.; Beier, R.C.; Lei, H.-T.; Wang, H.; Sun, Y.-M. Monoclonal antibody-based broad-specificity immunoassay for monitoring organophosphorus pesticides in environmental water samples. J. Environ. Monit. 2011, 13, 3040–3048. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-J.; Wu, H.-L.; Xiao, Z.-L.; Fu, H.-J.; Shen, Y.-D.; Luo, L.; Wang, H.; Lei, H.-T.; Hongsibsong, S.; Xu, Z.-L. Rational hapten design to produce high-quality antibodies against carbamate pesticides and development of immunochromatographic assays for simultaneous pesticide screening. J. Hazard. Mater. 2021, 412, 125241. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, F.; Zhao, L.; Yang, Z. Development of a broad-specificity immunoassay for determination of organophosphorus pesticides using dual-generic hapten antigens. Food Anal. Method. 2015, 8, 420–427. [Google Scholar] [CrossRef]
- Supervision, G.A.O.Q. Detection of 75 Illegally Added Chemical Drugs in Health Food. 2017; BJS 201710. Available online: https://www.samr.gov.cn/spcjs/bcjyff/201905/P020190516332773491737.doc (accessed on 26 July 2022).
- Ross, J.; Downs, J.; Bazydlo, L.; Bordwine, P.H.; Gineste, C.E.; Kopatic, M.C.; Rege, A.V.; Saady, D.M.; Utah, O.F.; Wyatt, S.A. Outbreak of Severe Hypoglycemia After Ingestion of a Male Enhancement Supplement-Virginia, August November 2019. Morb. Mortal. Wkly. Rep. 2020, 69, 740–743. [Google Scholar] [CrossRef] [PubMed]

| Name | General Structural | R1 a | R2 b | Sulfonylureas | R1 | R2 |
|---|---|---|---|---|---|---|
| Glyburide | 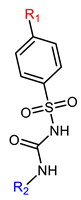 |  | 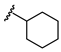 | Tolbutamide |  | 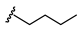 |
| Glipizide |  |  | Carbutamide | 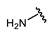 |  | |
| Glimepiride | 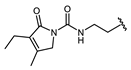 | 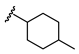 | Gliclazide | 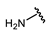 | 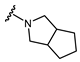 | |
| Gliquidone |  |  | Tolazamide |  | 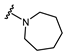 | |
| Acetohexamide |  |  | Glibornuride |  | 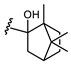 | |
| Chlorpropamide | 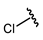 | 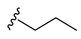 | ||||
| Hapten | Hapten 1 | 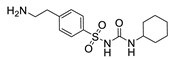 | Hapten 2 | 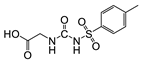 | ||
| Hapten 3 | 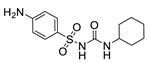 | Hapten 4 | 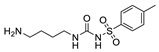 | |||
| Name | H1-Ab | H2-Ab | ||||||
|---|---|---|---|---|---|---|---|---|
| LOD a nmol/μL | Dynamic Working Range (nmol/μL) | IC50 b nmol/μL | CR c % | LOD nmol/μL | Dynamic Working Range nmol/μL | IC50 nmol/μL | CR % | |
| Glimepiride | 0.1 | 0.6–102.5 | 7.1 | 100.0 | 1672.4 | - | 17,228.5 | 0.1 d (0.04) e |
| Glipizide | 0.7 | 1.6–45.8 | 8.5 | 83.6 | 1823.3 | - | >10,000 | <0.01 |
| Glyburide | 0.4 | 1.6–96.4 | 12.1 | 58.7 | 3788.8 | - | >10,000 | <0.01 |
| Gliquidone | 0.04 | 1.1–190.7 | 14.8 | 48.3 | 2455.7 | - | >10,000 | <0.01 |
| Acetohexamide | 2.1 | 5.3–161.9 | 29.4 | 24.2 | 23.5 | - | 4173.0 | 0.6 (0.2) |
| Tolazamide | 7.7 | 19.9–530.5 | 103.1 | 6.9 | 2.2 | 11.2–3221.6 | 189.5 | 12.3 (3.8) |
| Tolbutamide | 32.2 | 81.7–1947.8 | 398.7 | 1.8 | 1.5 | 4.4–127.2 | 23.3 | 100 (30.6) |
| Carbutamide | 66.3 | 161.1–3331.6 | 732.3 | 1.0 | 15.8 | 62.7–6911.3 | 658.6 | 3.5 (1.1) |
| Gliclazide | 37.7 | 115.3–5224.3 | 775.8 | 0.9 | 3.1 | 13.9–2347.8 | 179.6 | 13.0 (4.0) |
| Chlorpropamide | 80.2 | 267.8–16,444.3 | 2098.7 | 0.3 | 0.7 | 6.9–163.7 | 33.6 | 69.3 (21.2) |
| Glibornuride | 228.1 | - | >10,000 | <0.01 | 9.8 | 52.4–15,498.0 | 901.0 | 2.6 (0.8) |
| Repaglinide | - | - | >10,000 | <0.01 | - | - | >10,000 | <0.01 |
| Rosiglitazone | - | - | >10,000 | <0.01 | - | - | >10,000 | <0.01 |
| Phenformin | - | - | >10,000 | <0.01 | - | - | >10,000 | <0.01 |
| Metformin hydrochloride | - | - | >10,000 | <0.01 | - | - | >10,000 | <0.01 |
| Name | Spiked Level (μg/kg) | ELISA | LC-MS/MS | R2 b | ||||
|---|---|---|---|---|---|---|---|---|
| Observed Level (μg/kg) | Average Recovery % | CV a % | Observed Level (μg/kg) | Average Recovery % | CV % | |||
| Glipizide | 320 | 259.5 | 81.1 | 10.9 | 244.3 | 76.4 | 1.0 | 0.995 |
| 1600 | 1272.0 | 79.5 | 7.9 | 1543.5 | 96.5 | 0.8 | ||
| 4800 | 4434.7 | 92.4 | 4.1 | 4701.4 | 98.0 | 2.4 | ||
| Glimepiride | 320 | 318.8 | 99.6 | 10.8 | 253.5 | 79.2 | 9.4 | 0.994 |
| 1600 | 1548.8 | 96.8 | 18.5 | 1340.3 | 83.8 | 3.1 | ||
| 6400 | 5526.3 | 86.3 | 14.2 | 5745.4 | 89.8 | 1.6 | ||
| Gliquidone | 640 | 703.8 | 110.0 | 12.4 | 647.8 | 101.2 | 0.4 | 0.995 |
| 3200 | 3092.0 | 96.6 | 5.7 | 3366.3 | 105.2 | 2.1 | ||
| 12,800 | 11,472.8 | 89.6 | 7.4 | 12,304.1 | 96.1 | 1.8 | ||
| Glyburide | 320 | 301.9 | 94.3 | 6.8 | 267.4 | 83.6 | 10.4 | 0.978 |
| 1600 | 1554.4 | 97.1 | 6.2 | 1409.5 | 88.1 | 1.3 | ||
| 6400 | 5621.5 | 87.8 | 3.7 | 6363.2 | 99.4 | 2.8 | ||
| Tolbutamide | 320 | 319.2 | 99.7 | 10.4 | 315.9 | 98.7 | 5.8 | 0.999 |
| 1600 | 1550.3 | 96.9 | 9.0 | 1722.5 | 107.7 | 0.9 | ||
| 6400 | 6494.9 | 101.5 | 6.5 | 6539.4 | 102.2 | 0.7 | ||
| Gliclazide | 3200 | 2534.1 | 79.2 | 18.1 | 3013.1 | 94.2 | 2.7 | 0.994 |
| 16,000 | 16,034.9 | 100.2 | 6.8 | 14,888.0 | 93.1 | 1.1 | ||
| 64,000 | 67,399.1 | 105.3 | 4.1 | 71,709.9 | 112.1 | 3.8 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Xie, H.; Fu, T.; Li, Y.; Shen, X.; Li, X.; Lei, Y.; Yao, X.; Koidis, A.; Liu, Y.; et al. Complementary Strategy Enhancing Broad-Specificity for Multiplexed Immunoassay of Adulterant Sulfonylureas in Functional Food. Biosensors 2022, 12, 591. https://doi.org/10.3390/bios12080591
Li Z, Xie H, Fu T, Li Y, Shen X, Li X, Lei Y, Yao X, Koidis A, Liu Y, et al. Complementary Strategy Enhancing Broad-Specificity for Multiplexed Immunoassay of Adulterant Sulfonylureas in Functional Food. Biosensors. 2022; 12(8):591. https://doi.org/10.3390/bios12080591
Chicago/Turabian StyleLi, Zhaodong, Haihuan Xie, Tingdan Fu, Yingying Li, Xing Shen, Xiangmei Li, Yi Lei, Xiaojun Yao, Anastasios Koidis, Yingju Liu, and et al. 2022. "Complementary Strategy Enhancing Broad-Specificity for Multiplexed Immunoassay of Adulterant Sulfonylureas in Functional Food" Biosensors 12, no. 8: 591. https://doi.org/10.3390/bios12080591
APA StyleLi, Z., Xie, H., Fu, T., Li, Y., Shen, X., Li, X., Lei, Y., Yao, X., Koidis, A., Liu, Y., Huang, X., & Lei, H. (2022). Complementary Strategy Enhancing Broad-Specificity for Multiplexed Immunoassay of Adulterant Sulfonylureas in Functional Food. Biosensors, 12(8), 591. https://doi.org/10.3390/bios12080591







