Microbial Fuel Cell Based on Nitrogen-Fixing Rhizobium anhuiense Bacteria
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cultivation of R. anhuiense Bacteria
2.3. Preparation of Carbon Felt-Based Electrodes
2.4. Microbial Fuel Cell Set-Up
2.5. Electrochemical Characterization of MFC
3. Results
3.1. Evaluation of R. anhuiense Bacteria Morphology and Cells Growth Kinetics
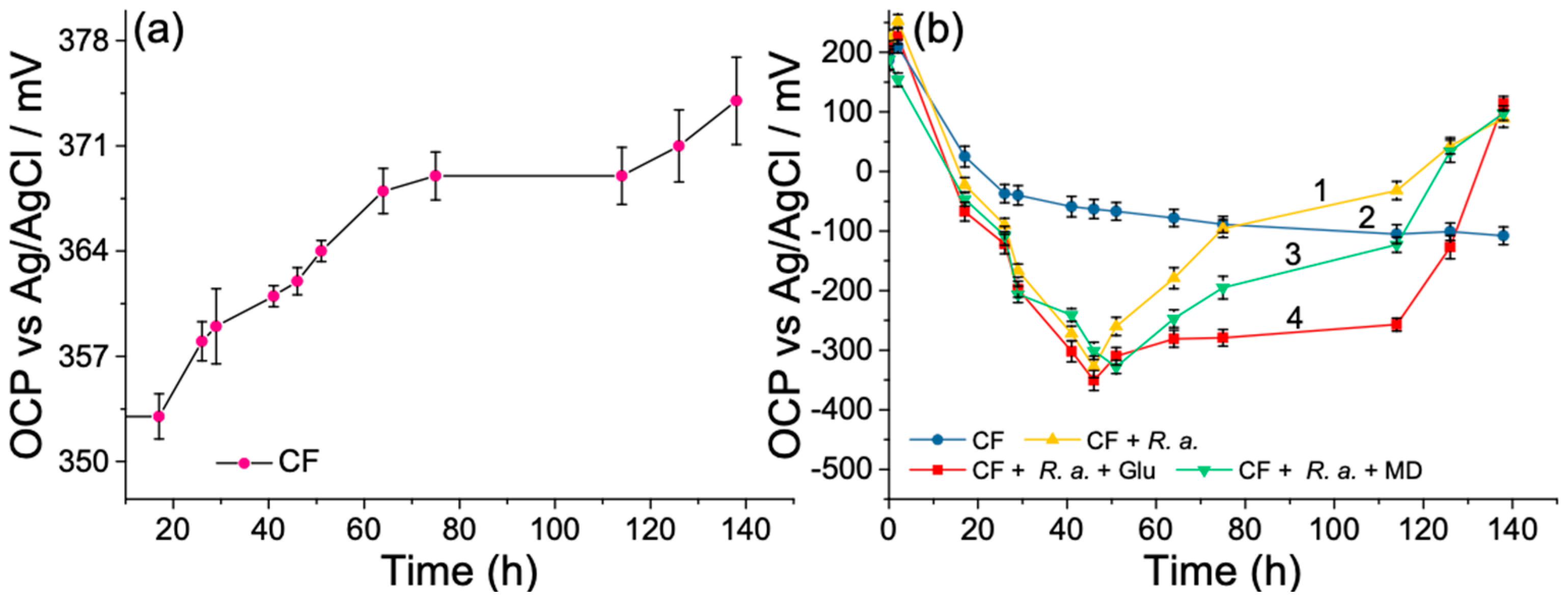
3.2. Open-Circuit Potential Investigations of MFC
3.3. Power Output of Dual-Chamber MFC
3.4. Investigation of Interference Reactions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Robertson, G.P.; Vitousek, P.M. Nitrogen in agriculture: Balancing the cost of an essential resource. Annu. Rev. Environ. Resour. 2009, 34, 97–125. [Google Scholar]
- Stuart, D.; Schewe, R.L.; McDermott, M. Reducing nitrogen fertilizer application as a climate change mitigation strategy: Understanding farmer decision-making and potential barriers to change in the US. Land Use Policy 2014, 36, 210–218. [Google Scholar] [CrossRef]
- Dang, P.; Li, C.; Huang, T.; Lu, C.; Li, Y.; Qin, X.; Siddique, K.H.M. Effects of different continuous fertilizer managements on soil total nitrogen stocks in China: A meta-analysis. Pedosphere 2022, 32, 39–48. [Google Scholar]
- Samanta, P.; Shin, S.; Jang, S.; Song, Y.C.; Oh, S.; Kim, J.K. Stable carbon and nitrogen isotopic characterization and tracing nutrient sources of Ulva blooms around Jeju coastal areas. Environ. Pollut. 2019, 254, 113033–113043. [Google Scholar] [CrossRef]
- Olesen, A.J.; Harðardóttir, S.; Daugbjerg, N.; Andersen, P.; Lyngsgaard, M.; Krock, B.; Lundholm, N. The impact of urea on toxic diatoms—Potential effects of fertilizer silo breakdown on a Pseudo-nitzschia bloom. Harmful Algae 2020, 98, 101817–101829. [Google Scholar]
- Thorup-Kristensen, K.; Magid, J.; Jensen, L.S. Catch crops and green manures as biological tools in nitrogen management in temperate zones. Adv. Agron. 2003, 79, 227–302. [Google Scholar]
- Cheng, L.; Ye, Z.; Cheng, S.; Guo, X. Agricultural ammonia emissions and its impact on PM2.5 concentrations in the Beijing–Tianjin–Hebei region from 2000 to 2018. Environ. Pollut. 2021, 291, 118162–118171. [Google Scholar]
- Kelleghan, D.B.; Hayes, E.T.; Everard, M.; Keating, P.; Lesniak-Podsiadlo, A.; Curran, T.P. Atmospheric ammonia and nitrogen deposition on Irish Natura 2000 sites: Implications for Irish agriculture. Atmos. Environ. 2021, 261, 118611–118624. [Google Scholar]
- Insausti, M.; Timmis, R.; Kinnersley, R.; Rufino, M.C. Advances in sensing ammonia from agricultural sources. Sci. Total Environ. 2020, 706, 135124–135136. [Google Scholar] [CrossRef] [PubMed]
- Leite, R.d.A.; Martins, L.C.; Ferreira, L.V.d.S.F.; Barbosa, E.S.; Alves, B.J.R.; Zilli, J.E.; Araújo, A.P.; Jesus, E.d.C. Co-inoculation of Rhizobium and Bradyrhizobium promotes growth and yield of common beans. Appl. Soil Ecol. 2022, 172, 104356–104365. [Google Scholar]
- Tong, W.; Li, X.; Huo, Y.; Zhang, L.; Cao, Y.; Wang, E.; Chen, W.; Tao, S.; Wei, G. Genomic insight into the taxonomy of Rhizobium genospecies that nodulate Phaseolus vulgaris. Syst. Appl. Microbiol. 2018, 41, 300–310. [Google Scholar] [CrossRef]
- Wolińska, A. Metagenomic achievements in microbial diversity determination in croplands: A review. Microb. Divers Genomic. Era. 2019, 15–35. [Google Scholar]
- Chen, J.S. Nitrogen fixation in the Clostridia. Genet. Regul. Nitrogen Fixat. Free Bact. 2004, 2, 53–64. [Google Scholar]
- Andrade-Linares, D.R.; Zistl-Schlingmann, M.; Foesel, B.; Dannenmann, M.; Schulz, S.; Schloter, M. Short term effects of climate change and intensification of management on the abundance of microbes driving nitrogen turnover in montane grassland soils. Sci. Total Environ. 2021, 780, 146672–146685. [Google Scholar] [CrossRef]
- Gómez-Godínez, L.J.; Martínez-Romero, E.; Banuelos, J.; Arteaga-Garibay, R.I. Tools and challenges to exploit microbial communities in agriculture. Curr. Res. Microb. Sci. 2021, 2, 100062–100069. [Google Scholar] [CrossRef] [PubMed]
- Harindintwali, J.D.; Zhou, J.; Yu, X. Lignocellulosic crop residue composting by cellulolytic nitrogen-fixing bacteria: A novel tool for environmental sustainability. Sci. Total Environ. 2020, 715, 136912–136925. [Google Scholar] [CrossRef]
- Hu, X.; Vandamme, P.; Boon, N. Co-cultivation enhanced microbial protein production based on autotrophic nitrogen-fixing hydrogen-oxidizing bacteria. Chem. Eng. J. 2022, 429, 132535–132545. [Google Scholar] [CrossRef]
- Potter, M.C. Electrical effects accompanying the decomposition of organic compounds. Proc. R. Soc. Lond. B 1911, 84, 260–276. [Google Scholar]
- Kumar, R.; Singh, L.; Wahid, Z.A.; Din, M.F.M. Exoelectrogens in microbial fuel cells toward bioelectricity generation: A review. Int. J. Energy Res. 2015, 39, 1048–1067. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, S.; Guo, S.; Wan, D.; Xu, H.; Yan, W.; Jin, X.; Feng, J. New insights in light-assisted microbial fuel cells for wastewater treatment and power generation: A win-win cooperation. J. Power Sources 2021, 501, 230000–230018. [Google Scholar] [CrossRef]
- Fadzli, F.S.; Rashid, M.; Yaqoob, A.A.; Ibrahim, M.N.M. Electricity generation and heavy metal remediation by utilizing yam (Dioscorea alata) waste in benthic microbial fuel cells (BMFCs). Biochem. Eng. J. 2021, 172, 108067–108076. [Google Scholar] [CrossRef]
- Nguyen, T.; Arias-Thode, Y.M.; Obraztsova, A.; Sarmiento, A.; Stevens-Bracy, A.; Grbovic, D.; Kartalov, E.P. Proof-of-concept for a novel application for in situ microfluidic benthic microbial fuel cell device (MBMFC). J. Environ. Chem. Eng. 2021, 9, 105659–105666. [Google Scholar] [CrossRef]
- Michel, R.F.M.; Schaefer, C.E.G.R.; Dias, L.E.; Simas, F.N.B.; Benites, V.d.M.; Mendonça, E.d.S. Ornithogenic gelisols (cryosols) from maritime Antarctica. Soil Sci. Soc. Am. J. 2006, 70, 1370–1376. [Google Scholar] [CrossRef]
- Gul, H.; Raza, W.; Lee, J.; Azam, M.; Ashraf, M.; Kim, K.H. Progress in microbial fuel cell technology for wastewater treatment and energy harvesting. Chemosphere 2021, 281, 130828–130840. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Duan, C.; Duan, W.; Sun, F.; Cui, H.; Zhang, S.; Chen, X. Role of electrode materials on performance and microbial characteristics in the constructed wetland coupled microbial fuel cell (CW-MFC): A review. J. Clean. Prod. 2021, 301, 126951–126963. [Google Scholar] [CrossRef]
- Sophia, A.C.; Sreeja, S. Green energy generation from plant microbial fuel cells (PMFC) using compost and a novel clay separator. Sustain. Energy Technol. Assess. 2017, 21, 59–66. [Google Scholar] [CrossRef]
- He, Z. Development of microbial fuel cells needs to go beyond “power density”. ACS Energy Lett. 2017, 2, 700–702. [Google Scholar] [CrossRef]
- Jung, S.P.; Pandit, S. Important factors influencing microbial fuel cell performance. Microb. Electrochem. Technol. 2019, 377–406. [Google Scholar]
- Mehboob, I.; Naveed, M.; Zahir, Z.A.; Sessitsch, A. Potential of Rhizosphere bacteria for improving Rhizobium-Legume symbiosis. In Plant Microbe Symbiosis: Fundamentals and Advances; Springer: Berlin/Heidelberg, Germany, 2013; pp. 305–349. [Google Scholar]
- Bouizgarne, B.; Oufdou, K.; Ouhdouch, Y. Actinorhizal and Rhizobial-Legume symbioses for alleviation of abiotic stresses. Plant Microbes Symbiosis Appl. Facet 2015, 273–295. [Google Scholar]
- Zhang, L.; Jiang, M.; Zhou, S. Conversion of nitrogen and carbon in enriched paddy soil by denitrification coupled with anammox in a bioelectrochemical system. J. Environ. Sci. 2022, 111, 197–207. [Google Scholar] [CrossRef]
- Tkach, O.; Sangeetha, T.; Maria, S.; Wang, A. Performance of low temperature microbial fuel cells (MFCs) catalyzed by mixed bacterial consortia. J. Environ. Sci. 2017, 52, 284–292. [Google Scholar] [CrossRef] [Green Version]
- Kabutey, F.T.; Zhao, Q.; Wei, L.; Ding, J.; Antwi, P.; Quashie, F.K.; Wang, W. An overview of plant microbial fuel cells (PMFCs): Configurations and applications. Renew Sustain. Energy Rev. 2019, 110, 402–414. [Google Scholar] [CrossRef]
- Nawaz, A.; Hafeez, A.; Abbas, S.Z.; Haq, I.; Mukhtar, H.; Rafatullah, M. A state of the art review on electron transfer mechanisms, characteristics, applications and recent advancements in microbial fuel cells technology. Green Chem. Lett. Rev. 2020, 13, 365–381. [Google Scholar] [CrossRef]
- Luo, J.; Li, M.; Zhou, M.; Hu, Y. Characterization of a novel strain phylogenetically related to Kocuria rhizophila and its chemical modification to improve performance of microbial fuel cells. Biosens. Bioelectron. 2015, 69, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Nandy, A.; Jana, S.; Khamrai, M.; Kumar, V.; Mukherjee, S.; Bhattacharyya, A.; Kundu, P.P. Cloning and expression of α-amylase in E. coli: Genesis of a superior biocatalyst for substrate-specific MFC. Int. J. Green Energy 2019, 16, 309–316. [Google Scholar] [CrossRef]
- Jorgensen, J.H.; Pfaller, M.A.; Carroll, K.C.; Funke, G.; Landry, M.L.; Richter, S.S.; Warnock, D.W. Manual of Clinical Microbiology, 11th ed.; ASM Press: Washington, DC, USA, 2015; Volume 1, pp. 226–237. [Google Scholar]
- Sijilmassi, B.; Filali-Maltouf, A.; Boulahyaoui, H.; Kricha, A.; Boubekri, K.; Udupa, S.; Kumar, S.; Amri, A. Assessment of genetic diversity and symbiotic efficiency of selected Rhizobia strains nodulating lentil (Lens culinaris Medik.). Plants 2021, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Petroniene, J.; Morkvenaite-Vilkonciene, I.; Miksiunas, R.; Bironaite, D.; Ramanaviciene, A.; Rucinskas, K.; Janusauskas, V.; Ramanavicius, A. Scanning electrochemical microscopy for the investigation of redox potential of human myocardium-derived mesenchymal stem cells grown at 2D and 3D conditions. Electrochim. Acta 2020, 360, 136956–136965. [Google Scholar] [CrossRef]
- Heiskanen, A.; Yakovleva, J.; Spegel, C.; Taboryski, R.; Koudelka-Hep, M.; Emneus, J.; Ruzgas, T. Amperometric monitoring of redox activity in living yeast cells: Comparison of menadione and menadione sodium bisulfite as electron transfer mediators. Electrochem. Commun. 2004, 6, 219–224. [Google Scholar] [CrossRef]
- Fang, D.; Gao, G.; Yang, Y.; Wang, Y.; Gao, L.; Zhi, J. Redox mediator-based microbial biosensors for acute water toxicity assessment: A critical review. ChemElectroChem 2020, 7, 2513–2526. [Google Scholar] [CrossRef]
- Hidalgo, D.; Tommasi, T.; Bocchini, S.; Chiolerio, A.; Chiodoni, A.; Mazzarino, I.; Ruggeri, B. Surface modification of commercial carbon felt used as anode for microbial fuel cells. Energy 2016, 99, 193–201. [Google Scholar] [CrossRef]
- Yang, S.Y.; Ma, C.C.M.; Teng, C.C.; Huang, Y.W.; Liao, S.H.; Huang, Y.L.; Tien, H.W.; Lee, T.M.; Chiou, K.C. Effect of functionalized carbon nanotubes on the thermal conductivity of epoxy composites. Carbon 2010, 48, 592–603. [Google Scholar] [CrossRef]
- Žalnėravičius, R.; Klimas, V.; Paškevičius, A.; Grincienė, G.; Karpicz, R.; Jagminas, A.; Ramanavičius, A. Highly efficient antimicrobial agents based on sulfur-enriched, hydrophilic molybdenum disulfide nano/microparticles and coatings functionalized with palladium nanoparticles. J. Colloid Interface Sci. 2021, 591, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Hays, M.P.; Hardwidge, P.R.; Kim, J. Surface characteristics influencing bacterial adhesion to polymeric substrates. RSC Adv. 2017, 7, 14254–14261. [Google Scholar] [CrossRef] [Green Version]
- Ma, J.; Chen, D.; Zhang, W.; An, Z.; Zeng, K.; Yuan, M.; Shen, J. Enhanced performance and degradation of wastewater in microbial fuel cells using titanium dioxide nanowire photocathodes. RSC Adv. 2021, 11, 2242–2252. [Google Scholar] [CrossRef]
- Rossi, R.; Logan, B.E. Impact of external resistance acclimation on charge transfer and diffusion resistance in bench-scale microbial fuel cells. Bioresour. Technol. 2020, 318, 123921–123926. [Google Scholar] [CrossRef]
- Logan, B. Exoelectrogenic bacteria that power microbial fuel cells. Nat. Rev. Microbiol. 2009, 7, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Little, B.J.; Hinks, J.; Blackwood, D.J. Microbially influenced corrosion: Towards an interdisciplinary perspective on mechanisms. Int. Biodeterior. 2020, 154, 105062–105069. [Google Scholar] [CrossRef]
- Kracke, F.; Vassilev, I.; Krömer, J.O. Microbial electron transport and energy conservation—The foundation for optimizing bioelectrochemical systems. Front. Microbiol. 2015, 6, 575–593. [Google Scholar] [CrossRef] [Green Version]
- Ruiz, Y.; Ribot-Llobet, E.; Baeza, J.A.; Guisasola, A. Conditions for high resistance to starvation periods in bioelectrochemical systems. Bioelectrochemistry 2015, 106, 328–334. [Google Scholar] [CrossRef]
- Kolthoff, I.M.; Pearson, E.A. Stability of potassium ferrocyanide solutions. Ind. Eng. Chem. Anal. Ed. 1931, 3, 381–382. [Google Scholar] [CrossRef]
- Wang, X.; Falk, M.; Ortiz, R.; Matsumura, H.; Bobacka, J.; Ludwig, R.; Bergelin, M.; Gorton, L.; Shleev, S. Mediatorless sugar/oxygen enzymatic fuel cells based on gold nanoparticle-modified electrodes. Biosens. Bioelectron. 2012, 31, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Rozene, J.; Morkvenaite-Vilkonciene, I.; Bruzaite, I.; Zinovicius, A.; Ramanavicius, A. Baker’s yeast-based microbial fuel cell mediated by 2-methyl1,4-maphthoquinone. Membranes 2021, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Shreeram, D.D.; Hassett, D.J.; Schaefer, D.W. Urine-powered microbial fuel cell using a hyperpiliated pilT mutant of Pseudomonas aeruginosa. J. Ind. Microbiol. 2016, 43, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.T.; Chen, W.J.; Huang, R.Y. Influence of growth curve phase on electricity performance of microbial fuel cell by Escherichia coli. Int. J. Hydrogen Energy 2010, 35, 7217–7223. [Google Scholar] [CrossRef]
- Sayed, E.T.; Tsujiguchi, T.; Nakagawa, N. Catalytic activity of baker’s yeast in a mediatorless microbial fuel cell. Bioelectrochemistry 2012, 86, 97–101. [Google Scholar] [CrossRef]
- Sayed, E.T.; Barakat, N.A.M.; Abdelkareem, M.A.; Fouad, H.; Nakagawa, N. Yeast extract as effective and safe mediator for the baker’s yeast-based microbial fuel cell. Ind. Eng. Chem. Res. 2015, 54, 3116–3122. [Google Scholar] [CrossRef]
- Kim, M.S.; Cha, J.; Kim, D.H. Enhancing factors of electricity generation in a microbial fuel cell using Geobacter sulfurreducens. J. Microbiol. Biotechnol. 2012, 22, 1395–1400. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Deng, F.; Hu, Y.; Sun, J.; Yang, Y. Antibacterial activity of graphene-modified anode on Shewanella oneidensis MR-1 biofilm in microbial fuel cell. J. Power Sources 2015, 290, 80–86. [Google Scholar] [CrossRef]
- Kasem, E.T.; Tsujiguchi, T.; Nakagawa, N. Effect of metal modification to carbon paper anodes on the performance of yeast-based microbial fuel cells part Ι: In the case without exogenous mediator. Key Eng. Mater. 2013, 534, 76–81. [Google Scholar] [CrossRef]
- Islam, M.A.; Ethiraj, B.; Cheng, C.K.; Yousuf, A.; Thiruvenkadam, S.; Prasad, R.; Khan, M.M.R. Enhanced current generation using mutualistic interaction of yeast-bacterial co-culture in dual chamber microbial fuel cell. Ind. Eng. Chem. Res. 2018, 57, 813–821. [Google Scholar] [CrossRef]
- Yamashoji, S. Different characteristics between menadione and menadione sodium bisulfite as redox mediator in yeast cell suspension. Biochem. Biophys. Rep. 2016, 6, 88–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruzgas, T. Enzyme-based (bio)fuel cells—Bilirubin oxidase use. In Encyclopedia of Interfacial Chemistry: Surface Science and Electrochemistry; Elsevier: Oxford, UK, 2018; pp. 209–216. [Google Scholar]
- Doyle, L.E.; Marsili, E. Weak electricigens: A new avenue for bioelectrochemical research. Bioresour. Technol. 2018, 258, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Schaetzle, O.; Barriere, F.; Baronian, K. Bacteria and yeasts as catalysts in microbial fuelcells: Electron transfer from micro-organisms to electrodes for green electricity. Energy Environ. Sci. 2008, 1, 607–620. [Google Scholar] [CrossRef]
- Heart, E.; Palo, M.; Womack, T.; Smith, P.J.S.; Gray, J.P. The level of menadione redox-cycling in pancreatic β-cells is proportional to the glucose concentration: Role of NADH and consequences for insulin secretion. Toxicol. Appl. Pharmacol. 2012, 258, 216–225. [Google Scholar] [CrossRef] [Green Version]
- Kumar, R.; Singh, L.; Wahid, Z.A. Role of microorganisms in microbial fuel cells for bioelectricity production. In Microbial Factories; Springer: New York City, NY, USA, 2015; pp. 136–154. [Google Scholar]
- Din, M.I.; Nabi, A.G.; Hussain, Z.; Khalid, R.; Iqbal, M.; Arshad, M.; Mujahid, A.; Hussain, T. Microbial fuel cells—A preferred technology to prevail energy crisis. Int. J. Energy Res. 2021, 45, 8370–8388. [Google Scholar] [CrossRef]
- Andrews, M.; Andrews, M.E. Specificity in Legume-Rhizobia symbioses. Int. J. Mol. Sci. 2017, 18, 705. [Google Scholar] [CrossRef] [Green Version]






| Reported List of Exoelectrogens | References |
|---|---|
| Firmicutes, Proteobacteria, Acidobacteria, fungi, and algae | [19] |
| Proteobacteria, Bacteroidetes, Chloroflexi, Acidobacteria, Firmicutes, and Nitrospirae | [31] |
| Geobacter psychrophilus, Pseudomonas caeni, Simplicispira sychrophile, Comamonas badia, and Geobacter chapelle | [32] |
| Clostridium butyricum, Rhodoferax ferrireducens, Shewanella sp., Geobacter spp., and Aeromonas hydrophila | [28] |
| Geobacter sulfurreducens | [24] |
| Natronocella acetinitrilica, Beijerinckiaceae, Rhizobiales, and Rhodobacter gluconicum | [33] |
| MFC Set-Up | Anode Compartment | Cathode Compartment | Carbon Source | Open Circuit Voltage (OCV), mV | Maximal Power Output (Pmax), mW m−2 | Reference |
|---|---|---|---|---|---|---|
| H-type cell | Graphite felt/P. aeruginosa (pilT) mutant | Pt/carbon cloth + 50 mM of K3[Fe(CN)6] | Urea | 720 | 54.16 | [55] |
| Graphite felt/P. aeruginosa (wild-type) | 750 | 20.0 | ||||
| Batch-type | Graphite carbon cloth/E. coli (gene type 1) + 11.5 mM of methylene blue | Graphite carbon cloth + 0.1 M of K3[Fe(CN)6] | Beef extract; peptone. | 309 * | 21.7 * | [56] |
| Graphite carbon cloth/E. coli (gene type 2) + 11.5 mM of methylene blue | 470 ** | 8.36 ** | ||||
| Graphite carbon cloth/E. coli (gene type 3) + 11.5 mM of methylene blue | Yeast extract; tryptone | 570 *** | 134 *** | |||
| Single chamber, air-cathode | Carbon paper/S. cerevisiae | Carbon paper with Pt catalyst (1 mg cm−2) | Glucose | 550 | 3.0 | [57] |
| Single chamber, open-air cathode | Gold-sputtered carbon paper/S. cerevisiae + 20 g L−1 of yeast extract | Carbon paper with Pt catalyst (1 mg cm−2) | Glucose; Peptone | 910 | 70.0 | [58] |
| Batch-type | Carbon paper/G. sulfurreducens + 50 mM of fumarate | Carbon paper | Acetate | - | 16.2 | [59] |
| Single-chamber, air-cathode | Graphite block modified by graphene/S. oneidensis | Carbon paper with Pt catalyst (0.5 mg cm−2) | Sodium lactate | 780 | 102 | [60] |
| Dual chamber | Carbon felt/Cystobasidium slooffiae | Carbon felt + 50 mM of K3[Fe(CN)6] | Xylose | 540 | 67 | [61] |
| Single-chamber, open-air cathode | Au-sputtered carbon paper + S. cerevisiae | Carbon paper with Pt catalyst (1 mg cm−2) | Glucose | 600 | 2 | [62] |
| Dual chamber | Stainless-steel/L. starkeyi | Stainless-steel + KMnO4 | Effluent | 900 | 47.6 | [63] |
| Stainless-steel/K. pneumonia | 800 | 19.77 | ||||
| Stainless-steel/co-culture of L. starkeyi and K. pneumonia | 750 | 10.98 | ||||
| H-type cell | Carbon felt/R. anhuiense | Bare carbon felt + 40 of mM K3[Fe(CN)6] | Glucose | 635 | 2.59 | This work |
| Carbon felt/R. anhuiense + 5 μM of menadione | 683 | 4.93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Žalnėravičius, R.; Paškevičius, A.; Samukaitė-Bubnienė, U.; Ramanavičius, S.; Vilkienė, M.; Mockevičienė, I.; Ramanavičius, A. Microbial Fuel Cell Based on Nitrogen-Fixing Rhizobium anhuiense Bacteria. Biosensors 2022, 12, 113. https://doi.org/10.3390/bios12020113
Žalnėravičius R, Paškevičius A, Samukaitė-Bubnienė U, Ramanavičius S, Vilkienė M, Mockevičienė I, Ramanavičius A. Microbial Fuel Cell Based on Nitrogen-Fixing Rhizobium anhuiense Bacteria. Biosensors. 2022; 12(2):113. https://doi.org/10.3390/bios12020113
Chicago/Turabian StyleŽalnėravičius, Rokas, Algimantas Paškevičius, Urtė Samukaitė-Bubnienė, Simonas Ramanavičius, Monika Vilkienė, Ieva Mockevičienė, and Arūnas Ramanavičius. 2022. "Microbial Fuel Cell Based on Nitrogen-Fixing Rhizobium anhuiense Bacteria" Biosensors 12, no. 2: 113. https://doi.org/10.3390/bios12020113
APA StyleŽalnėravičius, R., Paškevičius, A., Samukaitė-Bubnienė, U., Ramanavičius, S., Vilkienė, M., Mockevičienė, I., & Ramanavičius, A. (2022). Microbial Fuel Cell Based on Nitrogen-Fixing Rhizobium anhuiense Bacteria. Biosensors, 12(2), 113. https://doi.org/10.3390/bios12020113







