Conducting Polymer-Infused Electrospun Fibre Mat Modified by POEGMA Brushes as Antifouling Biointerface
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of (3,4-Ethylenedioxythiophene) Methyl 2-Bromopropanoate (EDOTBr)
2.2. Sulfonation of Polystyrene-Block-Poly(Ethylene-Ran-Butylene)-Block-Polystyrene (SEBS)
2.3. Fabrication of Electrospun Fibre Mats
2.4. Chemical Oxidative Polymerisation of EDOT on the Electrospun Fibre Mats
2.5. Electropolymerisation of P(EDOT-co-EDOTBr) on the sSEBS-PEDOT Fibre Mats
2.6. Grafting POEGMA Brushes on the sSEBS-PEDOT/P(EDOT-EDOTBr) Fibre Mat
2.7. Protein Adsorption Test
2.8. Cell Viability and Proliferation
2.9. Scanning Electron Microscopy (SEM)
2.10. Four-Point Probe Conductivity Meter
2.11. Raman Spectroscopy
2.12. X-ray Photoelectron Spectroscopy (XPS)
2.13. 1H NMR Measurement
2.14. Water Contact Angle Measurement
3. Results and Discussion
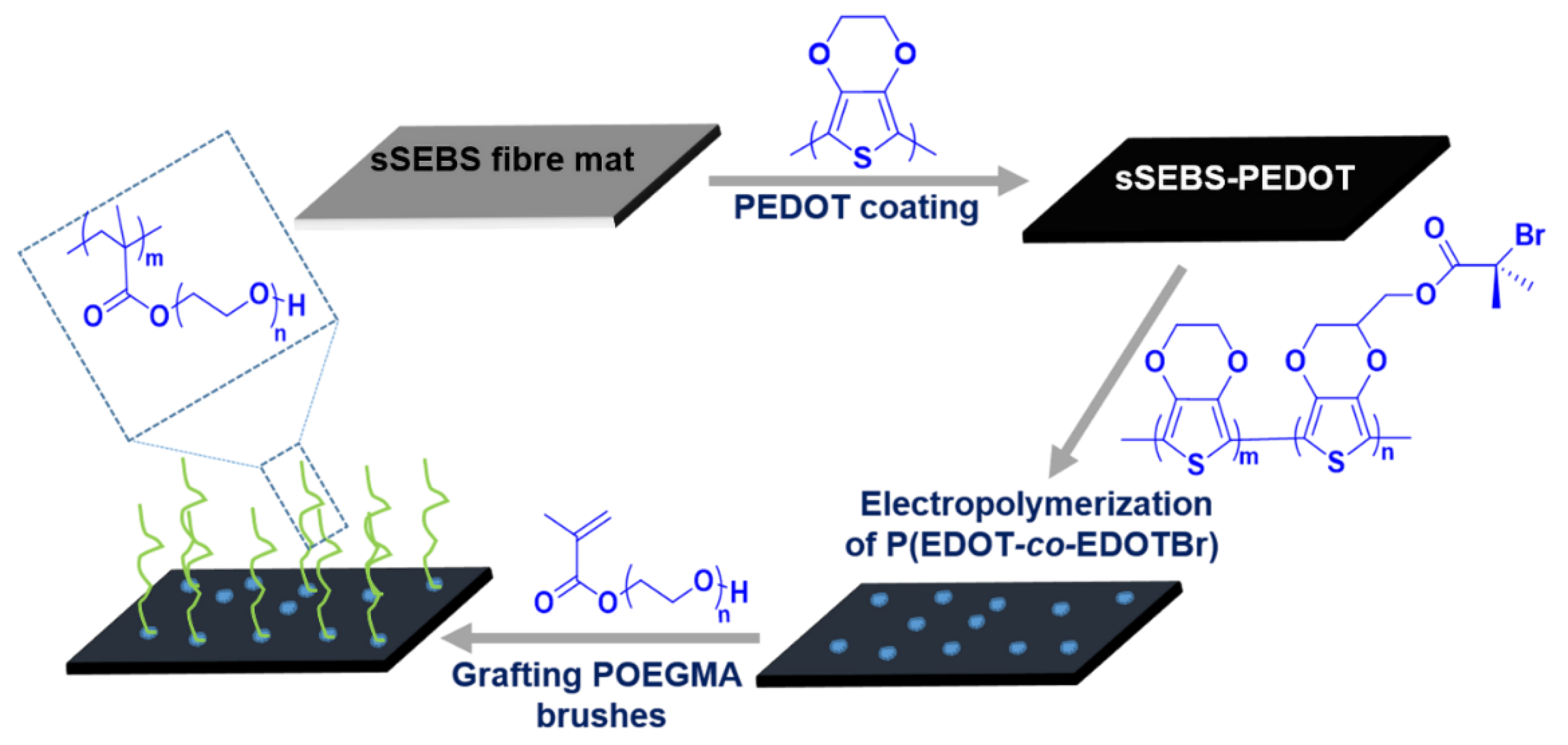
3.1. Fabrication of POEGMA Brush Grafted Electrospun Fibre Mat
3.2. Characterization of Electrospun Conducting sSEBS-PEDOT Fibre Mat
3.3. Electrochemical Copolymerisation of P(EDOT-co-EDOTBr) on sSEBS-PEDOT Fibre Mat
3.4. Grafting POEGMA Brushes from sSEBS-PEDOT/P(EDOT-co-EDOTBr) Fibre Mat
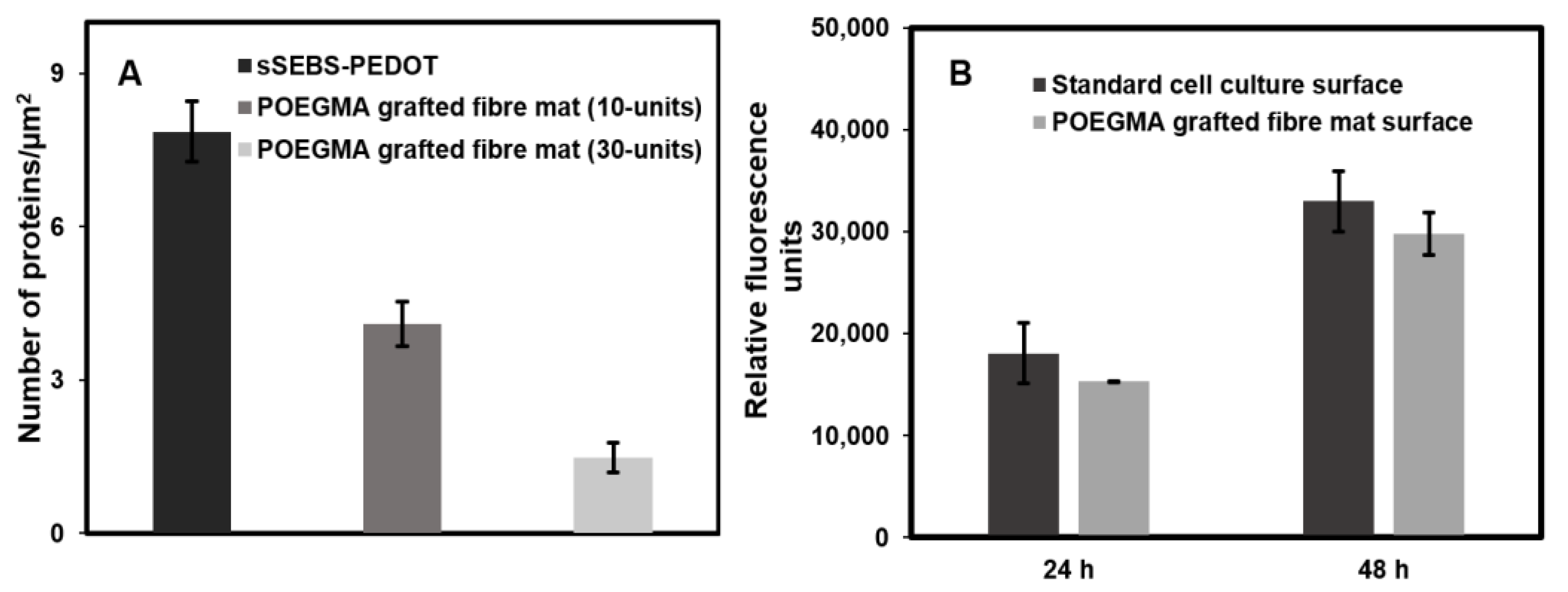
3.5. Antifouling Properties of sSEBS-PEDOT/P(EDOT-co-EDOTBr)-g-POEGMA Fibre Mat
3.6. Cell Viability and Proliferation Assessment Using PrestoBlueTM
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mauriz, E. Low-Fouling Substrates for Plasmonic Sensing of Circulating Biomarkers in Biological Fluids. Biosensors 2020, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Lin, Z.; Feng, Y.; Tan, H.; Xu, X.; Luo, J.; Li, J. Zwitterionic PMCP-Modified Polycaprolactone Surface for Tissue Engineering: Antifouling, Cell Adhesion Promotion, and Osteogenic Differentiation Properties. Small 2019, 15, 1903784. [Google Scholar] [CrossRef]
- Prakash, A.; Baer, M.D.; Mundy, C.J.; Pfaendtner, J. Peptoid Backbone Flexibilility Dictates Its Interaction with Water and Surfaces: A Molecular Dynamics Investigation. Biomacromolecules 2018, 19, 1006–1015. [Google Scholar] [CrossRef]
- Riedel, T.; Májek, P.; Riedelová-Reicheltová, Z.; Vorobii, M.; Houska, M.; Rodriguez-Emmenegger, C. Total removal of intact blood plasma proteins deposited on surface-grafted polymer brushes. Anal. Methods 2016, 8, 6415–6419. [Google Scholar] [CrossRef]
- Riedel, T.; Majek, P.; Rodriguez-Emmenegger, C.; Brynda, E. Surface plasmon resonance: Advances of label-free approaches in the analysis of biological samples. Bioanalysis 2014, 6, 3325–3336. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Tang, P.; Nie, C.; Hou, Y.; Haag, R. Well-Defined Nanostructured Biointerfaces: Strengthened Cellular Interaction for Circulating Tumor Cells Isolation. Adv. Healthc. Mater. 2021, 10, 2002202. [Google Scholar] [CrossRef] [PubMed]
- Akbarinejad, A.; Hisey, C.L.; Brewster, D.; Ashraf, J.; Chang, V.; Sabet, S.; Nursalim, Y.; Lucarelli, V.; Blenkiron, C.; Chamley, L.; et al. Novel Electrochemically Switchable, Flexible, Microporous Cloth that Selectively Captures, Releases, and Concentrates Intact Extracellular Vesicles. ACS Appl. Mater. Interfaces 2020, 12, 39005–39013. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Shi, Q.; Ye, W.; Fan, Q.; Shi, H.; Wong, S.-C.; Xu, X.; Yin, J. A novel hydrophilic polymer-brush pattern for site-specific capture of blood cells from whole blood. Chem. Commun. 2014, 51, 4200–4203. [Google Scholar] [CrossRef]
- Sakala, G.P.; Reches, M. Peptide-based approaches to fight biofouling. Adv. Mater. Interfaces 2018, 5, 1800073. [Google Scholar] [CrossRef]
- Goh, S.; Luan, Y.; Wang, X.; Du, H.; Chau, C.; Schellhorn, H.; Brash, J.; Chen, H.; Fang, Q. Polydopamine–polyethylene glycol–albumin antifouling coatings on multiple substrates. J. Mater. Chem. B 2018, 6, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Lowe, S.; O'Brien-Simpson, N.M.; Connal, L.A. Antibiofouling polymer interfaces: Poly(ethylene glycol) and other promising candidates. Polym. Chem. 2014, 6, 198–212. [Google Scholar] [CrossRef] [Green Version]
- Izquierdo-Barba, I.; Colilla, M.; Vallet-Regí, M. Zwitterionic ceramics for biomedical applications. Acta Biomater. 2016, 40, 201–211. [Google Scholar] [CrossRef]
- Yeh, S.-L.; Wang, T.-C.; Yusa, S.-I.; Thissen, H.; Tsai, W.-B. Conjugation of Polysulfobetaine via Poly(pyrogallol) Coatings for Improving the Antifouling Efficacy of Biomaterials. ACS Omega 2021, 6, 3517–3524. [Google Scholar] [CrossRef]
- Kuzmyn, A.R.; Teunissen, L.W.; Fritz, P.; van Lagen, B.; Smulders, M.M.; Zuilhof, H. Diblock and Random Antifouling Bioactive Polymer Brushes on Gold Surfaces by Visible-Light-Induced Polymerization (SI-PET-RAFT) in Water. Adv. Mater. Interfaces 2022, 9, 2101784. [Google Scholar] [CrossRef]
- Zhang, S.; Cao, J.; Ma, N.; You, M.; Wang, X.; Meng, J. Fast and facile fabrication of antifouling and hemocompatible PVDF membrane tethered with amino-acid modified PEG film. Appl. Surf. Sci. 2018, 428, 41–53. [Google Scholar] [CrossRef]
- Wu, J.; Zhao, C.; Lin, W.; Hu, R.; Wang, Q.; Chen, H.; Li, L.; Chen, S.; Zheng, J. Binding characteristics between polyethylene glycol (PEG) and proteins in aqueous solution. J. Mater. Chem. B 2014, 2, 2983–2992. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Su, Y.; Zhao, X.; Li, Y.; Zhang, R.; Ma, T.; Liu, Y.; Jiang, Z. Manipulating the segregation behavior of polyethylene glycol by hydrogen bonding interaction to endow ultrafiltration membranes with enhanced antifouling performance. J. Membr. Sci. 2015, 499, 56–64. [Google Scholar] [CrossRef]
- Liu, N.; Xu, Z.; Morrin, A.; Luo, X. Low fouling strategies for electrochemical biosensors targeting disease biomarkers. Anal. Methods 2019, 11, 702–711. [Google Scholar] [CrossRef]
- Mousavi, M.; Ghaleh, H.; Jalili, K.; Abbasi, F. Multi-layer PDMS films having antifouling property for biomedical applications. J. Biomater. Sci. Polym. Ed. 2020, 32, 678–693. [Google Scholar] [CrossRef] [PubMed]
- Venault, A.; Chang, Y.; Hsu, H.-H.; Jhong, J.-F.; Yang, H.-S.; Wei, T.-C.; Tung, K.-L.; Higuchi, A.; Huang, J. Biofouling-resistance control of expanded poly(tetrafluoroethylene) membrane via atmospheric plasma-induced surface PEGylation. J. Membr. Sci. 2013, 439, 48–57. [Google Scholar] [CrossRef]
- Matyjaszewski, K. Atom Transfer Radical Polymerization (ATRP): Current Status and Future Perspectives. Macromolecules 2012, 45, 4015–4039. [Google Scholar] [CrossRef]
- Kang, C.; Crockett, R.M.; Spencer, N.D. Molecular-Weight Determination of Polymer Brushes Generated by SI-ATRP on Flat Surfaces. Macromolecules 2013, 47, 269–275. [Google Scholar] [CrossRef]
- Yan, W.; Fantin, M.; Spencer, N.D.; Matyjaszewski, K.; Benetti, E.M. Translating Surface-Initiated Atom Transfer Radical Polymerization into Technology: The Mechanism of Cu0-Mediated SI-ATRP under Environmental Conditions. ACS Macro Lett. 2019, 8, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Bansal, A.; Behera, B.; Jain, S.L.; Ray, S.S. Ternary hybrid polymeric nanocomposites through grafting of polystyrene on graphene oxide-TiO2 by surface initiated atom transfer radical polymerization (SI-ATRP). Mater. Chem. Phys. 2016, 172, 189–196. [Google Scholar] [CrossRef]
- Rodda, A.E.; Ercole, F.; Nisbet, D.R.; Forsythe, J.S.; Meagher, L. Optimization of Aqueous SI-ATRP Grafting of Poly(Oligo(Ethylene Glycol) Methacrylate) Brushes from Benzyl Chloride Macroinitiator Surfaces. Macromol. Biosci. 2015, 15, 799–811. [Google Scholar] [CrossRef]
- Albers, R.F.; Magrini, T.; Romio, M.; Leite, E.R.; Libanori, R.; Studart, A.R.; Benetti, E.M. Fabrication of Three-Dimensional Polymer-Brush Gradients within Elastomeric Supports by Cu0-Mediated Surface-Initiated ATRP. ACS Macro Lett. 2021, 10, 1099–1106. [Google Scholar] [CrossRef]
- Aydemir, N.; Malmström, J.; Travas-Sejdic, J. Conducting polymer based electrochemical biosensors. Phys. Chem. Chem. Phys. 2016, 18, 8264–8277. [Google Scholar] [CrossRef] [PubMed]
- Baek, P.; Voorhaar, L.; Barker, D.; Travas-Sejdic, J. Molecular Approach to Conjugated Polymers with Biomimetic Properties. Acc. Chem. Res. 2018, 51, 1581–1589. [Google Scholar] [CrossRef]
- Hackett, A.J.; Malmström, J.; Travas-Sejdic, J. Functionalization of conducting polymers for biointerface applications. Prog. Polym. Sci. 2017, 70, 18–33. [Google Scholar] [CrossRef]
- Oh, B.; George, P. Conductive polymers to modulate the post-stroke neural environment. Brain Res. Bull. 2019, 148, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jang, L.K.; Jang, M.; Lee, S.; Hardy, J.G.; Lee, J.Y. Electrically Conductive Polydopamine–Polypyrrole as High Performance Biomaterials for Cell Stimulation in Vitro and Electrical Signal Recording in Vivo. ACS Appl. Mater. Interfaces 2018, 10, 33032–33042. [Google Scholar] [CrossRef] [Green Version]
- Ramanavicius, S.; Samukaite-Bubniene, U.; Ratautaite, V.; Bechelany, M.; Ramanavicius, A. Electrochemical molecularly imprinted polymer based sensors for pharmaceutical and biomedical applications (review). J. Pharm. Biomed. Anal. 2022, 215, 114739. [Google Scholar] [CrossRef]
- Puiggalí-Jou, A.; del Valle, L.J.; Alemán, C. Drug delivery systems based on intrinsically conducting polymers. J. Control. Release 2019, 309, 244–264. [Google Scholar] [CrossRef] [PubMed]
- Stříteský, S.; Marková, A.; Víteček, J.; Šafaříková, E.; Hrabal, M.; Kubáč, L.; Kubala, L.; Weiter, M.; Vala, M. Printing inks of electroactive polymer PEDOT: PSS: The study of biocompatibility, stability, and electrical properties. J. Biomed. Mater. Res. Part A 2018, 106, 1121–1128. [Google Scholar] [CrossRef]
- Wang, G.; Han, R.; Su, X.; Li, Y.; Xu, G.; Luo, X. Zwitterionic peptide anchored to conducting polymer PEDOT for the development of antifouling and ultrasensitive electrochemical DNA sensor. Biosens. Bioelectron. 2017, 92, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Malmström, J.; Nieuwoudt, M.K.; Strover, L.T.; Hackett, A.; Laita, O.; Brimble, M.A.; Williams, D.E.; Travas-Sejdic, J. Grafting from Poly(3,4-ethylenedioxythiophene): A Simple Route to Versatile Electrically Addressable Surfaces. Macromolecules 2013, 46, 4955–4965. [Google Scholar] [CrossRef]
- Zhao, H.; Zhu, B.; Luo, S.-C.; Lin, H.-A.; Nakao, A.; Yamashita, Y.; Yu, H. Controlled protein absorption and cell adhesion on polymer-brush-grafted poly (3, 4-ethylenedioxythiophene) films. ACS Appl. Mater. Interfaces 2013, 5, 4536–4543. [Google Scholar] [CrossRef] [PubMed]
- Boncler, M.; Różalski, M.; Krajewska, U.; Podsędek, A.; Watala, C. Comparison of PrestoBlue and MTT assays of cellular viability in the assessment of anti-proliferative effects of plant extracts on human endothelial cells. J. Pharmacol. Toxicol. Methods 2014, 69, 9–16. [Google Scholar] [CrossRef]
- Zhao, Q.; Jamal, R.; Zhang, L.; Wang, M.; Abdiryim, T. The structure and properties of PEDOT synthesized by template-free solution method. Nanoscale Res. Lett. 2014, 9, 557. [Google Scholar] [CrossRef] [Green Version]
- Nagasaki, Y. Construction of a densely poly(ethylene glycol)-chain-tethered surface and its performance. Polym. J. 2011, 43, 949–958. [Google Scholar] [CrossRef]
- Dong, H.-B.; Xu, Y.-Y.; Yi, Z.; Shi, J.-L. Modification of polysulfone membranes via surface-initiated atom transfer radical polymerization. Appl. Surf. Sci. 2009, 255, 8860–8866. [Google Scholar] [CrossRef]
- Shen, C.; Bian, L.; Zhang, P.; An, B.; Cui, Z.; Wang, H.; Li, J. Microstructure evolution of bonded water layer and morphology of grafting membrane with different polyethylene glycol length and their influence on permeability and anti-fouling capacity. J. Membr. Sci. 2020, 601, 117949. [Google Scholar] [CrossRef]
- Tomaskovic-Crook, E.; Zhang, P.; Ahtiainen, A.; Kaisvuo, H.; Lee, C.-Y.; Beirne, S.; Aqrawe, Z.; Svirskis, D.; Hyttinen, J.; Wallace, G.G.; et al. Human Neural Tissues from Neural Stem Cells Using Conductive Biogel and Printed Polymer Microelectrode Arrays for 3D Electrical Stimulation. Adv. Healthc. Mater. 2019, 8, e1900425. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Travas-Sejdic, J.; O'Grady, G.; Du, P. Comparison of gold and PEDOT:PSS contacts for high-resolution gastric electrical mapping using flexible printed circuit arrays. In Proceedings of the 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Guadalajara, Mexico, 1–5 November 2021; pp. 6937–6940. [Google Scholar] [CrossRef]
- Jiang, C.; Wang, G.; Hein, R.; Liu, N.; Luo, X.; Davis, J.J. Antifouling strategies for selective in vitro and in vivo sensing. Chem. Rev. 2020, 120, 3852–3889. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Z.; Chen, J.; Lian, C.; Han, X.; Liu, H. Conformation-dominated surface antifouling and aqueous lubrication. Colloids Surf. B Biointerfaces 2022, 214, 112452. [Google Scholar] [CrossRef]
- Yan, H.; Wu, Q.; Yu, C.; Zhao, T.; Liu, M. Recent Progress of Biomimetic Antifouling Surfaces in Marine. Adv. Mater. Interfaces 2020, 7, 202000966. [Google Scholar] [CrossRef]
- Alejo, T.; Prieto, M.; García-Juan, H.; Andreu, V.; Mendoza, G.; Sebastián, V.; Arruebo, M. A facile method for the controlled polymerization of biocompatible and thermoresponsive oligo(ethylene glycol) methyl ether methacrylate copolymers. Polym. J. 2018, 50, 203–211. [Google Scholar] [CrossRef]
- Lishchynskyi, O.; Stetsyshyn, Y.; Raczkowska, J.; Awsiuk, K.; Orzechowska, B.; Abalymov, A.; Skirtach, A.; Bernasik, A.; Nastyshyn, S.; Budkowski, A. Fabrication and Impact of Fouling-Reducing Temperature-Responsive POEGMA Coatings with Embedded CaCO3 Nanoparticles on Different Cell Lines. Materials 2021, 14, 1417. [Google Scholar] [CrossRef]
- Xu, F.; Lam, A.; Pan, Z.; Randhawa, G.; Lamb, M.; Sheardown, H.; Hoare, T. Fast Thermoresponsive Poly(oligoethylene glycol methacrylate) (POEGMA)-Based Nanostructured Hydrogels for Reversible Tuning of Cell Interactions. ACS Biomater. Sci. Eng. 2021, 7, 4258–4268. [Google Scholar] [CrossRef]
- Tomšík, E.; Laishevkina, S.; Svoboda, J.; Gunar, K.; Hromádková, J.; Shevchenko, N. Preparation of Smart Surfaces Based on PNaSS@ PEDOT Microspheres: Testing of E. coli Detection. Sensors 2022, 22, 2784. [Google Scholar] [CrossRef]
- Wang, W.; Cui, M.; Song, Z.; Luo, X. An antifouling electrochemical immunosensor for carcinoembryonic antigen based on hyaluronic acid doped conducting polymer PEDOT. RSC Adv. 2016, 6, 88411–88416. [Google Scholar] [CrossRef]
- Yang, X.; Kirsch, J.; Olsen, E.V.; Fergus, J.; Simonian, A.L. Anti-fouling PEDOT:PSS modification on glassy carbon electrodes for continuous monitoring of tricresyl phosphate. Sens. Actuators B Chem. 2012, 177, 659–667. [Google Scholar] [CrossRef]
- Hsu, C.-C.; Cheng, Y.-W.; Liu, C.-C.; Peng, X.-Y.; Yung, M.-C.; Liu, T.-Y. Anti-Bacterial and Anti-Fouling Capabilities of Poly(3,4-Ethylenedioxythiophene) Derivative Nanohybrid Coatings on SUS316L Stainless Steel by Electrochemical Polymerization. Polymers 2020, 12, 1467. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Sheng, G.; Cui, Y.; Li, M.; Song, Z.; Ding, C.; Luo, X. Low fouling electrochemical sensing in complex biological media by using the ionic liquid-doped conducting polymer PEDOT: Application to voltammetric determination of dopamine. Microchim. Acta 2019, 186, 220. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashraf, J.; Lau, S.; Akbarinejad, A.; Evans, C.W.; Williams, D.E.; Barker, D.; Travas-Sejdic, J. Conducting Polymer-Infused Electrospun Fibre Mat Modified by POEGMA Brushes as Antifouling Biointerface. Biosensors 2022, 12, 1143. https://doi.org/10.3390/bios12121143
Ashraf J, Lau S, Akbarinejad A, Evans CW, Williams DE, Barker D, Travas-Sejdic J. Conducting Polymer-Infused Electrospun Fibre Mat Modified by POEGMA Brushes as Antifouling Biointerface. Biosensors. 2022; 12(12):1143. https://doi.org/10.3390/bios12121143
Chicago/Turabian StyleAshraf, Jesna, Sandy Lau, Alireza Akbarinejad, Clive W. Evans, David E. Williams, David Barker, and Jadranka Travas-Sejdic. 2022. "Conducting Polymer-Infused Electrospun Fibre Mat Modified by POEGMA Brushes as Antifouling Biointerface" Biosensors 12, no. 12: 1143. https://doi.org/10.3390/bios12121143
APA StyleAshraf, J., Lau, S., Akbarinejad, A., Evans, C. W., Williams, D. E., Barker, D., & Travas-Sejdic, J. (2022). Conducting Polymer-Infused Electrospun Fibre Mat Modified by POEGMA Brushes as Antifouling Biointerface. Biosensors, 12(12), 1143. https://doi.org/10.3390/bios12121143





