Capture-SELEX: Selection Strategy, Aptamer Identification, and Biosensing Application
Abstract
1. Introduction
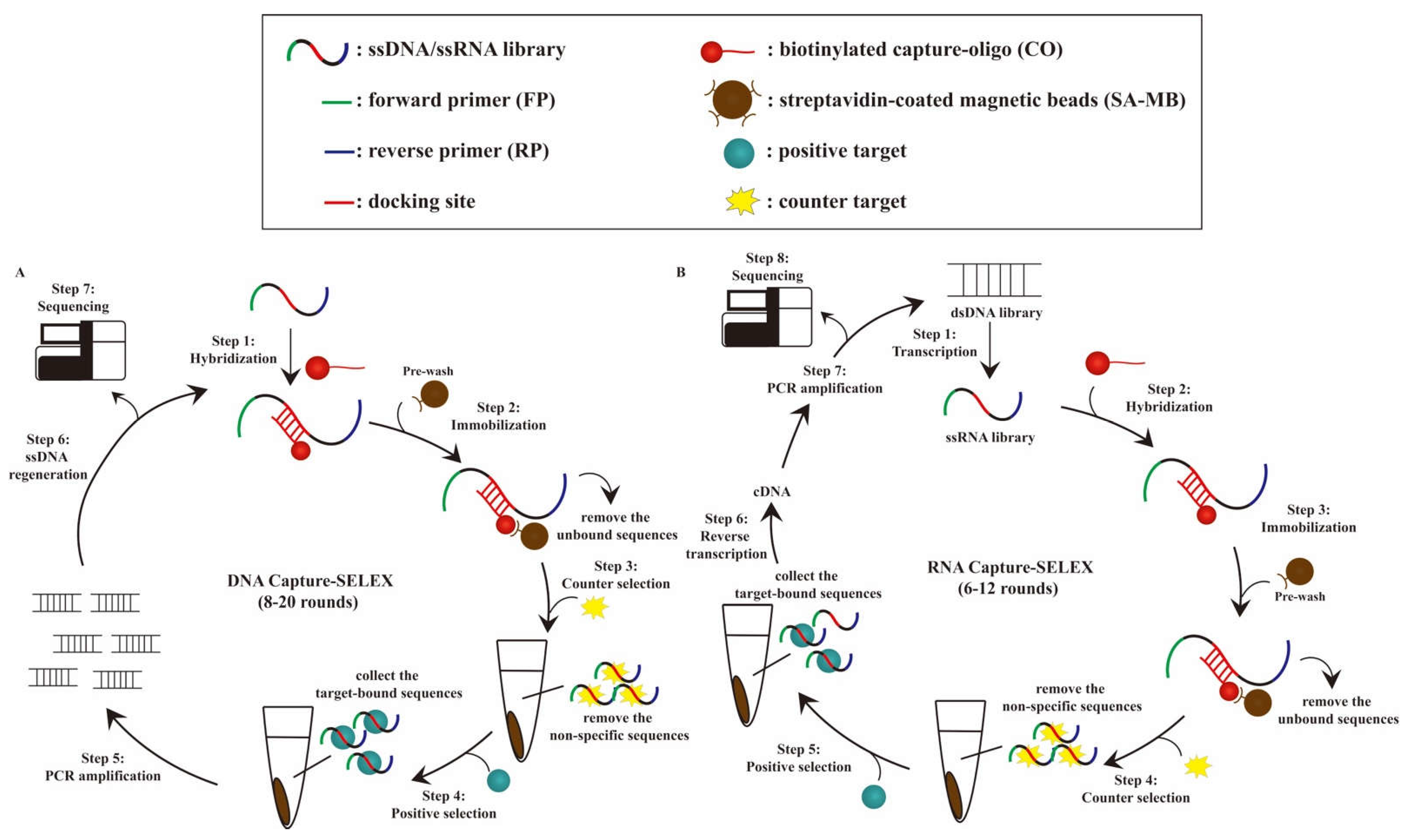
2. Principle and General Procedure of Capture-SELEX for Aptamer Selection
2.1. Overview of DNA and RNA Capture-SELEX
2.2. Sanger and Next-Generation Sequencing and Analysis
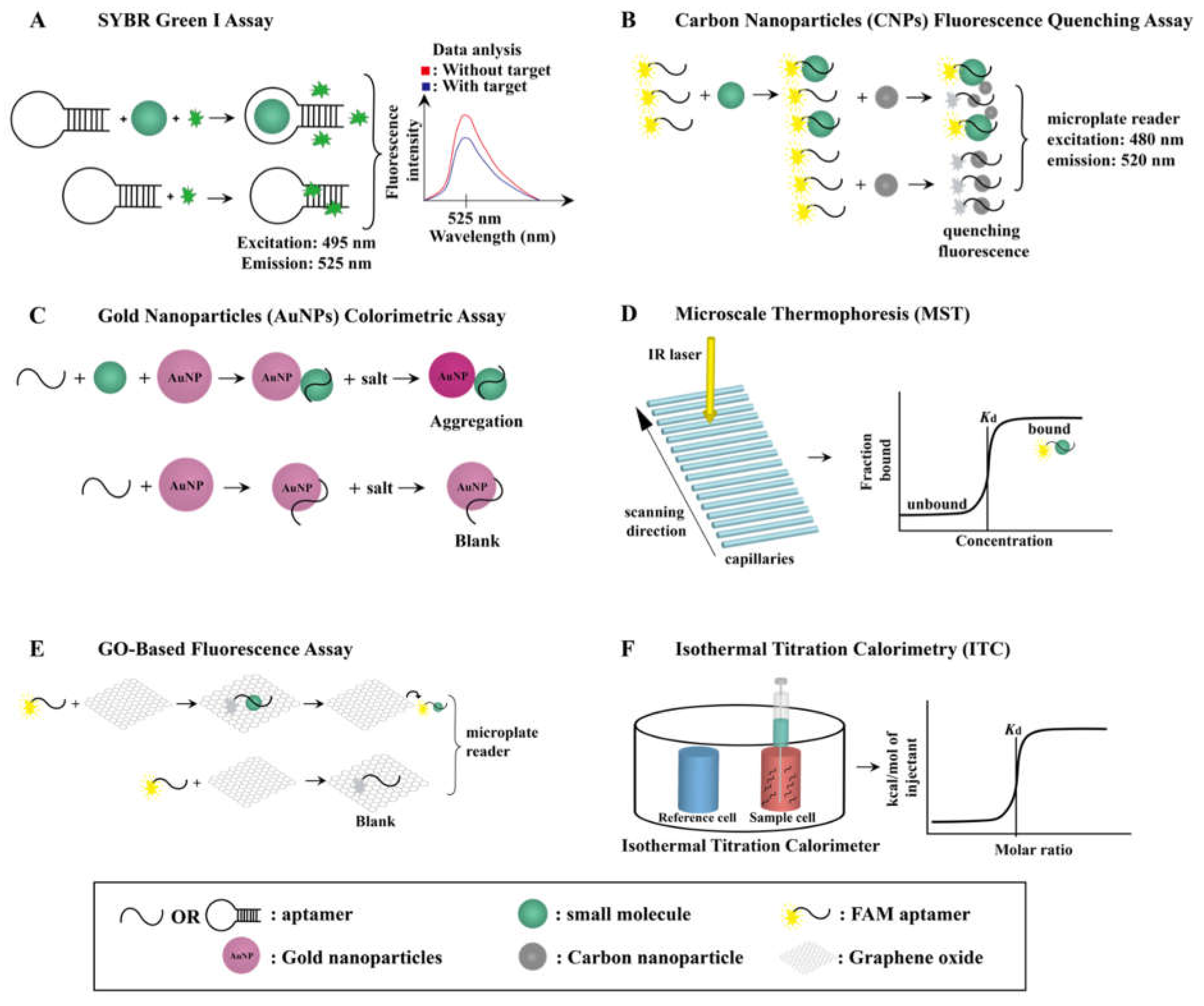
3. Small-Molecule–Aptamer Interaction and Characterization
3.1. SYBR Green I (SGI) Assay
3.2. Carbon Nanoparticle (CNP) Fluorescence Quenching Assay
3.3. Gold Nanoparticle (AuNP) Colorimetric Assay
3.4. Microscale Thermophoresis (MST) Assay
3.5. GO-Based Fluorescent Assay
3.6. Isothermal Titration Calorimetry (ITC) Assay
4. Development of Aptamer-Based Biosensors
4.1. Biolayer Interferometry (BLI)-Based Aptasensor
4.2. Lateral Flow Aptasensors (LFAs)
5. Biosensor Applications
5.1. Food Safety Analysis
5.1.1. Veterinary Drug Residues and Pesticides
5.1.2. Food Additives and Flavoring Agents
5.2. Aquatic Environment
5.2.1. Veterinary Drug Residues and Pesticides
5.2.2. Toxins and Plasticizers
5.3. Other Potential Applications
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Minh, T.B.; Leung, H.W.; Loi, I.H.; Chan, W.H.; So, M.K.; Mao, J.Q.; Choi, D.; Lam, J.C.; Martin, M.; Lee, J.H.W.; et al. Antibiotics in the Hong Kong metropolitan area: Ubiquitous distribution and fate in Victoria Harbour. Mar. Pollut. Bull. 2009, 58, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Gothwal, R.; Shashidhar, T. Antibiotic Pollution in the Environment: A Review. CLEAN–Soil. Air Water 2015, 43, 479–489. [Google Scholar] [CrossRef]
- Parra-Arroyo, L.; Gonzalez-Gonzalez, R.B.; Castillo-Zacarias, C.; Melchor Martinez, E.M.; Sosa-Hernandez, J.E.; Bilal, M.; Iqbal, H.M.N.; Barcelo, D.; Parra-Saldivar, R. Highly hazardous pesticides and related pollutants: Toxicological, regulatory, and analytical aspects. Sci. Total Environ. 2022, 807, 151879. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Zeng, D.; Ding, N.; Luo, K.; Man, Y.B.; Zeng, L.; Zhang, Q.; Luo, J.; Kang, Y. Percutaneous Penetration and Metabolism of Plasticizers by Skin Cells and Its Implication in Dermal Exposure to Plasticizers by Skin Wipes. Environ. Sci. Technol. 2020, 54, 10181–10190. [Google Scholar] [CrossRef] [PubMed]
- Eales, J.; Bethel, A.; Galloway, T.; Hopkinson, P.; Morrissey, K.; Short, R.E.; Garside, R. Human health impacts of exposure to phthalate plasticizers: An overview of reviews. Environ. Int. 2022, 158, 106903. [Google Scholar] [CrossRef]
- Qadeer, A.; Kirsten, K.L.; Ajmal, Z.; Jiang, X.; Zhao, X. Alternative Plasticizers As Emerging Global Environmental and Health Threat: Another Regrettable Substitution? Environ. Sci. Technol. 2022, 56, 1482–1488. [Google Scholar] [CrossRef]
- Schuster, C.; Sterz, S.; Teupser, D.; Brugel, M.; Vogeser, M.; Paal, M. Multiplex Therapeutic Drug Monitoring by Isotope-dilution HPLC-MS/MS of Antibiotics in Critical Illnesses. J. Vis. Exp. 2018. [Google Scholar] [CrossRef]
- Li, S.; Wnag, L.; Wu, J.; Inoue, H. Syntheses and spectra of Mn(III)-chlorophyll-a and Mn(II)-chlorophyll-a. Guang Pu Xue Yu Guang Pu Fen Xi 1997, 17, 55–59. [Google Scholar]
- Sheng, W.; Xia, X.; Wei, K.; Li, J.; Li, Q.X.; Xu, T. Determination of marbofloxacin residues in beef and pork with an enzyme-linked immunosorbent assay. J. Agric. Food Chem. 2009, 57, 5971–5975. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, H.C.; Liu, J.; Wang, J.P. A receptor-based chemiluminescence enzyme linked immunosorbent assay for determination of tetracyclines in milk. Anal. Biochem. 2019, 564-565, 40–46. [Google Scholar] [CrossRef]
- Dahal, U.P.; Jones, J.P.; Davis, J.A.; Rock, D.A. Small molecule quantification by liquid chromatography-mass spectrometry for metabolites of drugs and drug candidates. Drug Metab. Dispos. 2011, 39, 2355–2360. [Google Scholar] [CrossRef]
- Li, B.; Van Schepdael, A.; Hoogmartens, J.; Adams, E. Characterization of impurities in tobramycin by liquid chromatography-mass spectrometry. J. Chromatogr. A 2009, 1216, 3941–3945. [Google Scholar] [CrossRef]
- Zhou, J.; Rossi, J. Aptamers as targeted therapeutics: Current potential and challenges. Nat. Rev. Drug Discov. 2017, 16, 440. [Google Scholar] [CrossRef]
- Thiviyanathan, V.; Gorenstein, D.G. Aptamers and the next generation of diagnostic reagents. Proteom. Clin. Appl. 2012, 6, 563–573. [Google Scholar] [CrossRef]
- He, Y.; Zhou, L.; Deng, L.; Feng, Z.; Cao, Z.; Yin, Y. An electrochemical impedimetric sensing platform based on a peptide aptamer identified by high-throughput molecular docking for sensitive l-arginine detection. Bioelectrochemistry 2021, 137, 107634. [Google Scholar] [CrossRef]
- Lin, N.; Wu, L.; Xu, X.; Wu, Q.; Wang, Y.; Shen, H.; Song, Y.; Wang, H.; Zhu, Z.; Kang, D.; et al. Aptamer Generated by Cell-SELEX for Specific Targeting of Human Glioma Cells. ACS Appl. Mater. Interfaces 2021, 13, 9306–9315. [Google Scholar] [CrossRef]
- Chen, X.; Qiu, L.; Cai, R.; Cui, C.; Li, L.; Jiang, J.H.; Tan, W. Aptamer-Directed Protein-Specific Multiple Modifications of Membrane Glycoproteins on Living Cells. ACS Appl. Mater. Interfaces 2020, 12, 37845–37850. [Google Scholar] [CrossRef]
- Lohlamoh, W.; Soontornworajit, B.; Rotkrua, P. Anti-Proliferative Effect of Doxorubicin-Loaded AS1411 Aptamer on Colorectal Cancer Cell. Asian Pac. J. Cancer Prev. 2021, 22, 2209–2219. [Google Scholar] [CrossRef]
- Amraee, M.; Oloomi, M.; Yavari, A.; Bouzari, S. DNA aptamer identification and characterization for E. coli O157 detection using cell based SELEX method. Anal. Biochem. 2017, 536, 36–44. [Google Scholar] [CrossRef]
- Chinnappan, R.; Eissa, S.; Alotaibi, A.; Siddiqua, A.; Alsager, O.A.; Zourob, M. In vitro selection of DNA aptamers and their integration in a competitive voltammetric biosensor for azlocillin determination in waste water. Anal. Chim. Acta 2020, 1101, 149–156. [Google Scholar] [CrossRef]
- Paniel, N.; Istamboulie, G.; Triki, A.; Lozano, C.; Barthelmebs, L.; Noguer, T. Selection of DNA aptamers against penicillin G using Capture-SELEX for the development of an impedimetric sensor. Talanta 2017, 162, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Stoltenburg, R.; Nikolaus, N.; Strehlitz, B. Capture-SELEX: Selection of DNA Aptamers for Aminoglycoside Antibiotics. J. Anal. Methods Chem. 2012, 2012, 415697. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, A.; Komarova, N.; Andrianova, M.; Grudtsov, V.; Kuznetsov, E. Aptamer based vanillin sensor using an ion-sensitive field-effect transistor. Mikrochim. Acta 2017, 185, 3. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Liu, D.; Wang, M.; Guo, F.; Lin, J.S. Preparation of DNA aptamer and development of lateral flow aptasensor combining recombinase polymerase amplification for detection of erythromycin. Biosens. Bioelectron. 2021, 181, 113157. [Google Scholar] [CrossRef] [PubMed]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef]
- Robertson, D.L.; Joyce, G.F. Selection in vitro of an RNA enzyme that specifically cleaves single-stranded DNA. Nature 1990, 344, 467–468. [Google Scholar] [CrossRef]
- Yang, J.; Bowser, M.T. Capillary electrophoresis-SELEX selection of catalytic DNA aptamers for a small-molecule porphyrin target. Anal. Chem. 2013, 85, 1525–1530. [Google Scholar] [CrossRef]
- Eaton, R.M.; Shallcross, J.A.; Mael, L.E.; Mears, K.S.; Minkoff, L.; Scoville, D.J.; Whelan, R.J. Selection of DNA aptamers for ovarian cancer biomarker HE4 using CE-SELEX and high-throughput sequencing. Anal. Bioanal. Chem. 2015, 407, 6965–6973. [Google Scholar] [CrossRef]
- Zhu, C.; Li, L.; Fang, S.; Zhao, Y.; Zhao, L.; Yang, G.; Qu, F. Selection and characterization of an ssDNA aptamer against thyroglobulin. Talanta 2021, 223, 121690. [Google Scholar] [CrossRef]
- Kou, Q.; Wu, P.; Sun, Q.; Li, C.; Zhang, L.; Shi, H.; Wu, J.; Wang, Y.; Yan, X.; Le, T. Selection and truncation of aptamers for ultrasensitive detection of sulfamethazine using a fluorescent biosensor based on graphene oxide. Anal. Bioanal. Chem. 2021, 413, 901–909. [Google Scholar] [CrossRef]
- Ozyurt, C.; Canbay, Z.C.; Dinckaya, E.; Evran, S. A highly sensitive DNA aptamer-based fluorescence assay for sarcosine detection down to picomolar levels. Int. J. Biol. Macromol. 2019, 129, 91–97. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, J.; Lee, B.H.; Song, C.S.; Gu, M.B. Specific detection of avian influenza H5N2 whole virus particles on lateral flow strips using a pair of sandwich-type aptamers. Biosens. Bioelectron. 2019, 134, 123–129. [Google Scholar] [CrossRef]
- Wang, G.; Liu, J.; Chen, K.; Xu, Y.; Liu, B.; Liao, J.; Zhu, L.; Hu, X.; Li, J.; Pu, Y.; et al. Selection and characterization of DNA aptamer against glucagon receptor by cell-SELEX. Sci. Rep. 2017, 7, 7179. [Google Scholar] [CrossRef]
- Yilmaz, D.; Muslu, T.; Parlar, A.; Kurt, H.; Yuce, M. SELEX against whole-cell bacteria resulted in lipopolysaccharide binding aptamers. J. Biotechnol. 2022, 354, 10–20. [Google Scholar] [CrossRef]
- Wan, J.; Ye, L.; Yang, X.; Guo, Q.; Wang, K.; Huang, Z.; Tan, Y.; Yuan, B.; Xie, Q. Cell-SELEX based selection and optimization of DNA aptamers for specific recognition of human cholangiocarcinoma QBC-939 cells. Analyst 2015, 140, 5992–5997. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Y.; Yang, H.; Qin, M.; Ding, X.; Liu, R.; Jiang, Y. In Vivo SELEX of an Inhibitory NSCLC-Specific RNA Aptamer from PEGylated RNA Library. Mol. Ther. Nucleic Acids 2018, 10, 187–198. [Google Scholar] [CrossRef]
- Huang, X.; Zhong, J.; Ren, J.; Wen, D.; Zhao, W.; Huan, Y. A DNA aptamer recognizing MMP14 for in vivo and in vitro imaging identified by cell-SELEX. Oncol. Lett. 2019, 18, 265–274. [Google Scholar] [CrossRef]
- Reinemann, C.; Freiin von Fritsch, U.; Rudolph, S.; Strehlitz, B. Generation and characterization of quinolone-specific DNA aptamers suitable for water monitoring. Biosens. Bioelectron. 2016, 77, 1039–1047. [Google Scholar] [CrossRef]
- Stoltenburg, R.; Reinemann, C.; Strehlitz, B. FluMag-SELEX as an advantageous method for DNA aptamer selection. Anal. Bioanal. Chem. 2005, 383, 83–91. [Google Scholar] [CrossRef]
- Lyu, C.; Khan, I.M.; Wang, Z. Capture-SELEX for aptamer selection: A short review. Talanta 2021, 229, 122274. [Google Scholar] [CrossRef] [PubMed]
- Jenison, R.D.; Gill, S.C.; Pardi, A.; Polisky, B. High-resolution molecular discrimination by RNA. Science 1994, 263, 1425–1429. [Google Scholar] [CrossRef]
- Shkembi, X.; Skouridou, V.; Svobodova, M.; Leonardo, S.; Bashammakh, A.S.; Alyoubi, A.O.; Campas, M.; CK, O.S. Hybrid Antibody-Aptamer Assay for Detection of Tetrodotoxin in Pufferfish. Anal. Chem. 2021. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Duan, N.; Wu, S.; Wang, Z. Selection and application of ssDNA aptamers against spermine based on Capture-SELEX. Anal. Chim. Acta 2019, 1081, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Lyu, C.; Duan, N.; Wu, S.; Khan, I.M.; Wang, Z. The isolation of high-affinity ssDNA aptamer for the detection of ribavirin in chicken. Anal. Methods 2021, 13, 3110–3117. [Google Scholar] [CrossRef]
- Wang, T.; Chen, C.; Larcher, L.M.; Barrero, R.A.; Veedu, R.N. Three decades of nucleic acid aptamer technologies: Lessons learned, progress and opportunities on aptamer development. Biotechnol. Adv. 2019, 37, 28–50. [Google Scholar] [CrossRef]
- Spiga, F.M.; Maietta, P.; Guiducci, C. More DNA-Aptamers for Small Drugs: A Capture-SELEX Coupled with Surface Plasmon Resonance and High-Throughput Sequencing. ACS Comb. Sci. 2015, 17, 326–333. [Google Scholar] [CrossRef]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- Crossley, B.M.; Bai, J.; Glaser, A.; Maes, R.; Porter, E.; Killian, M.L.; Clement, T.; Toohey-Kurth, K. Guidelines for Sanger sequencing and molecular assay monitoring. J. Vet. Diagn. Investig. 2020, 32, 767–775. [Google Scholar] [CrossRef]
- Levy, S.E.; Myers, R.M. Advancements in Next-Generation Sequencing. Annu. Rev. Genom. Hum. Genet. 2016, 17, 95–115. [Google Scholar] [CrossRef]
- Balasubramanian, S. Solexa sequencing: Decoding genomes on a population scale. Clin. Chem. 2015, 61, 21–24. [Google Scholar] [CrossRef]
- Slatko, B.E.; Gardner, A.F.; Ausubel, F.M. Overview of Next-Generation Sequencing Technologies. Curr. Protoc. Mol. Biol. 2018, 122, e59. [Google Scholar] [CrossRef]
- Eisold, A.; Labudde, D. Detailed Analysis of 17beta-Estradiol-Aptamer Interactions: A Molecular Dynamics Simulation Study. Molecules 2018, 23, 1690. [Google Scholar] [CrossRef]
- Bavi, R.; Liu, Z.; Han, Z.; Zhang, H.; Gu, Y. In silico designed RNA aptamer against epithelial cell adhesion molecule for cancer cell imaging. Biochem. Biophys. Res. Commun. 2019, 509, 937–942. [Google Scholar] [CrossRef]
- Ahmad, N.A.; Mohamed Zulkifli, R.; Hussin, H.; Nadri, M.H. In silico approach for Post-SELEX DNA aptamers: A mini-review. J. Mol. Graph Model 2021, 105, 107872. [Google Scholar] [CrossRef]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef]
- Sievers, F.; Higgins, D.G. Clustal omega. Curr. Protoc. Bioinform. 2014, 48, 3–13. [Google Scholar] [CrossRef]
- Bailey, T.L.; Boden, M.; Buske, F.A.; Frith, M.; Grant, C.E.; Clementi, L.; Ren, J.; Li, W.W.; Noble, W.S. MEME SUITE: Tools for motif discovery and searching. Nucleic Acids Res. 2009, 37, W202–W208. [Google Scholar] [CrossRef]
- Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 2003, 31, 3406–3415. [Google Scholar] [CrossRef]
- Xayaphoummine, A.; Bucher, T.; Isambert, H. Kinefold web server for RNA/DNA folding path and structure prediction including pseudoknots and knots. Nucleic Acids Res. 2005, 33, W605–W610. [Google Scholar] [CrossRef]
- Xu, Z.Z.; Mathews, D.H. Experiment-Assisted Secondary Structure Prediction with RNAstructure. Methods Mol. Biol. 2016, 1490, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Hu, L.; Zhang, B.T.; Lu, A.; Wang, Y.; Yu, Y.; Zhang, G. Artificial Intelligence in Aptamer-Target Binding Prediction. Int. J. Mol. Sci. 2021, 22, 3605. [Google Scholar] [CrossRef] [PubMed]
- Soon, S.; Nordin, N.A. In silico predictions and optimization of aptamers against streptococcus agalactiae surface protein using computational docking. Mater. Today-Proc. 2019, 16, 2096–2100. [Google Scholar] [CrossRef]
- Douaki, A.; Garoli, D.; Inam, A.; Angeli, M.A.C.; Cantarella, G.; Rocchia, W.; Wang, J.; Petti, L.; Lugli, P. Smart Approach for the Design of Highly Selective Aptamer-Based Biosensors. Biosensors 2022, 12, 574. [Google Scholar] [CrossRef] [PubMed]
- Mousivand, M.; Anfossi, L.; Bagherzadeh, K.; Barbero, N.; Mirzadi-Gohari, A.; Javan-Nikkhah, M. In silico maturation of affinity and selectivity of DNA aptamers against aflatoxin B1 for biosensor development. Anal. Chim. Acta 2020, 1105, 178–186. [Google Scholar] [CrossRef]
- Kadam, U.S.; Hong, J.C. Advances in aptameric biosensors designed to detect toxic contaminants from food, water, human fluids, and the environment. Trends Environ. Anal. Chem. 2022, 36, e00184. [Google Scholar] [CrossRef]
- Tian, Y.; Wang, Y.; Sheng, Z.; Li, T.; Li, X. A colorimetric detection method of pesticide acetamiprid by fine-tuning aptamer length. Anal. Biochem. 2016, 513, 87–92. [Google Scholar] [CrossRef]
- Komarova, N.; Andrianova, M.; Glukhov, S.; Kuznetsov, A. Selection, Characterization, and Application of ssDNA Aptamer against Furaneol. Molecules 2018, 23, 3159. [Google Scholar] [CrossRef]
- Song, M.; Li, C.; Wu, S.; Duan, N. Screening of specific aptamers against chlorpromazine and construction of novel ratiometric fluorescent aptasensor based on metal-organic framework. Talanta 2022, 252, 123850. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, M.; Zhang, Y.; Chen, H.; Su, Y.; Wang, Y.; Lin, J.S. Preparation and characterization of DNA aptamers against roxithromycin. Anal. Chim. Acta 2021, 1164, 338509. [Google Scholar] [CrossRef]
- Lu, Q.; Liu, X.; Hou, J.; Yuan, Q.; Li, Y.; Chen, S. Selection of Aptamers Specific for DEHP Based on ssDNA Library Immobilized SELEX and Development of Electrochemical Impedance Spectroscopy Aptasensor. Molecules 2020, 25, 747. [Google Scholar] [CrossRef]
- Ren, L.; Qi, S.; Khan, I.M.; Wu, S.; Duan, N.; Wang, Z. Screening and application of a broad-spectrum aptamer for acyclic guanosine analogues. Anal. Bioanal. Chem. 2021, 413, 4855–4863. [Google Scholar] [CrossRef]
- Wu, C.; Barkova, D.; Komarova, N.; Offenhausser, A.; Andrianova, M.; Hu, Z.; Kuznetsov, A.; Mayer, D. Highly selective and sensitive detection of glutamate by an electrochemical aptasensor. Anal. Bioanal. Chem. 2021, 414, 1609–1622. [Google Scholar] [CrossRef]
- Li, C.; Song, M.; Wu, S.; Wang, Z.; Duan, N. Selection of aptamer targeting levamisole and development of a colorimetric and SERS dual-mode aptasensor based on AuNPs/Cu-TCPP(Fe) nanosheets. Talanta 2023, 251, 123739. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, Y.; Deng, B.; Hu, B.; Zhao, L.; Guo, H.; Yang, C.; Ma, Z.; Sun, M.; Jiao, B.; et al. Selection, Characterization, and Optimization of DNA Aptamers against Challenging Marine Biotoxin Gymnodimine-A for Biosensing Application. Toxins 2022, 14, 195. [Google Scholar] [CrossRef]
- Yang, Y.; Tang, Y.; Wang, C.; Liu, B.; Wu, Y. Selection and identification of a DNA aptamer for ultrasensitive and selective detection of lambda-cyhalothrin residue in food. Anal. Chim. Acta 2021, 1179, 338837. [Google Scholar] [CrossRef]
- Xie, M.; Chen, Z.; Zhao, F.; Lin, Y.; Zheng, S.; Han, S. Selection and Application of ssDNA Aptamers for Fluorescence Biosensing Detection of Malachite Green. Foods 2022, 11, 801. [Google Scholar] [CrossRef]
- Wu, W.; Sun, Q.; Li, T.; Liu, K.; Jiang, Y.; Wang, Y.; Yang, Y. Selection and characterization of bispecific aptamers against malachite green and leucomalachite green. Anal. Biochem. 2022, 658, 114849. [Google Scholar] [CrossRef]
- Wang, L.; Liu, X.; Zhang, Q.; Zhang, C.; Liu, Y.; Tu, K.; Tu, J. Selection of DNA aptamers that bind to four organophosphorus pesticides. Biotechnol. Lett. 2012, 34, 869–874. [Google Scholar] [CrossRef]
- Martin, J.A.; Chavez, J.L.; Chushak, Y.; Chapleau, R.R.; Hagen, J.; Kelley-Loughnane, N. Tunable stringency aptamer selection and gold nanoparticle assay for detection of cortisol. Anal. Bioanal. Chem. 2014, 406, 4637–4647. [Google Scholar] [CrossRef]
- Duan, N.; Gong, W.; Wu, S.; Wang, Z. Selection and Application of ssDNA Aptamers against Clenbuterol Hydrochloride Based on ssDNA Library Immobilized SELEX. J. Agric. Food Chem. 2017, 65, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Duan, N.; Gong, W.; Wu, S.; Wang, Z. An ssDNA library immobilized SELEX technique for selection of an aptamer against ractopamine. Anal. Chim. Acta 2017, 961, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Abraham, K.M.; Roueinfar, M.; Ponce, A.T.; Lussier, M.E.; Benson, D.B.; Hong, K.L. In Vitro Selection and Characterization of a Single-Stranded DNA Aptamer Against the Herbicide Atrazine. ACS Omega 2018, 3, 13576–13583. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, T.; Wang, Y.; Diao, C.; Zhou, Y.; Zhao, L.; Chen, H. Selection of a DNA Aptamer against Zearalenone and Docking Analysis for Highly Sensitive Rapid Visual Detection with Label-Free Aptasensor. J. Agric. Food Chem. 2018, 66, 12102–12110. [Google Scholar] [CrossRef] [PubMed]
- Trinh, K.H.; Kadam, U.S.; Song, J.; Cho, Y.; Kang, C.H.; Lee, K.O.; Lim, C.O.; Chung, W.S.; Hong, J.C. Novel DNA Aptameric Sensors to Detect the Toxic Insecticide Fenitrothion. Int. J. Mol. Sci. 2021, 22, 846. [Google Scholar] [CrossRef] [PubMed]
- Kadam, U.S.; Trinh, K.H.; Kumar, V.; Lee, K.W.; Cho, Y.; Can, M.T.; Lee, H.; Kim, Y.; Kim, S.; Kang, J.; et al. Identification and structural analysis of novel malathion-specific DNA aptameric sensors designed for food testing. Biomaterials 2022, 287, 121617. [Google Scholar] [CrossRef]
- Trinh, K.H.; Kadam, U.S.; Rampogu, S.; Cho, Y.; Yang, K.A.; Kang, C.H.; Lee, K.W.; Lee, K.O.; Chung, W.S.; Hong, J.C. Development of novel fluorescence-based and label-free noncanonical G4-quadruplex-like DNA biosensor for facile, specific, and ultrasensitive detection of fipronil. J. Hazard Mater. 2022, 427, 127939. [Google Scholar] [CrossRef]
- Can, M.T.; Kadam, U.S.; Trinh, K.H.; Cho, Y.; Lee, H.; Kim, Y.; Kim, S.; Kang, C.H.; Kim, S.H.; Chung, W.S.; et al. Engineering Novel Aptameric Fluorescent Biosensors for Analysis of the Neurotoxic Environmental Contaminant Insecticide Diazinon from Real Vegetable and Fruit Samples. Front Biosci. (Landmark Ed.) 2022, 27, 92. [Google Scholar] [CrossRef]
- Xia, L.; Yang, Y.; Yang, H.; Tang, Y.; Zhou, J.; Wu, Y. Screening and identification of an aptamer as novel recognition molecule in the test strip and its application for visual detection of ethyl carbamate in liquor. Anal. Chim. Acta 2022, 1226, 340289. [Google Scholar] [CrossRef]
- Yu, H.; Yang, W.; Alkhamis, O.; Canoura, J.; Yang, K.A.; Xiao, Y. In vitro isolation of small-molecule-binding aptamers with intrinsic dye-displacement functionality. Nucleic Acids Res. 2018, 46, e43. [Google Scholar] [CrossRef]
- Zhang, W.; Li, D.; Zhang, J.; Jiang, L.; Li, Z.; Lin, J.S. Preparation and Characterization of Aptamers Against O,p’-DDT. Int. J. Mol. Sci. 2020, 21, 2211. [Google Scholar] [CrossRef]
- Matsenko, N.U.; Rijikova, V.S.; Kovalenko, S.P. Comparison of SYBR Green I and TaqMan real-time PCR formats for the analysis of her2 gene dose in human breast tumors. Bull. Exp. Biol. Med. 2008, 145, 240–244. [Google Scholar] [CrossRef]
- Marinowic, D.R.; Zanirati, G.; Rodrigues, F.V.F.; Grahl, M.V.C.; Alcara, A.M.; Machado, D.C.; Da Costa, J.C. A new SYBR Green real-time PCR to detect SARS-CoV-2. Sci. Rep. 2021, 11, 2224. [Google Scholar] [CrossRef]
- Schnerr, H.; Niessen, L.; Vogel, R.F. Real time detection of the tri5 gene in Fusarium species by lightcycler-PCR using SYBR Green I for continuous fluorescence monitoring. Int. J. Food Microbiol. 2001, 71, 53–61. [Google Scholar] [CrossRef]
- Vitzthum, F.; Geiger, G.; Bisswanger, H.; Brunner, H.; Bernhagen, J. A quantitative fluorescence-based microplate assay for the determination of double-stranded DNA using SYBR Green I and a standard ultraviolet transilluminator gel imaging system. Anal. Biochem. 1999, 276, 59–64. [Google Scholar] [CrossRef]
- Iida, R.; Yasuda, T.; Tsubota, E.; Nakashima, Y.; Sawazaki, K.; Aoyama, M.; Matsuki, T.; Kishi, K. Detection of isozymes of deoxyribonucleases I and II on electrophoresed gels with picogram sensitivity using SYBR Green I. Electrophoresis 1998, 19, 2416–2418. [Google Scholar] [CrossRef]
- Izumiyama, S.; Omura, M.; Takasaki, T.; Ohmae, H.; Asahi, H. Plasmodium falciparum: Development and validation of a measure of intraerythrocytic growth using SYBR Green I in a flow cytometer. Exp. Parasitol. 2009, 121, 144–150. [Google Scholar] [CrossRef]
- Assuncao, P.; Rosales, R.S.; Rifatbegovic, M.; Antunes, N.T.; de la Fe, C.; Ruiz de Galarreta, C.M.; Poveda, J.B. Quantification of mycoplasmas in broth medium with sybr green-I and flow cytometry. Front. Biosci. 2006, 11, 492–497. [Google Scholar] [CrossRef][Green Version]
- Dragan, A.I.; Pavlovic, R.; McGivney, J.B.; Casas-Finet, J.R.; Bishop, E.S.; Strouse, R.J.; Schenerman, M.A.; Geddes, C.D. SYBR Green I: Fluorescence properties and interaction with DNA. J. Fluoresc. 2012, 22, 1189–1199. [Google Scholar] [CrossRef]
- Wang, Y.; Bao, L.; Liu, Z.; Pang, D.W. Aptamer biosensor based on fluorescence resonance energy transfer from upconverting phosphors to carbon nanoparticles for thrombin detection in human plasma. Anal. Chem. 2011, 83, 8130–8137. [Google Scholar] [CrossRef]
- Li, H.L.; Zhang, Y.W.; Wang, L.; Tian, J.Q.; Sun, X.P. Nucleic acid detection using carbon nanoparticles as a fluorescent sensing platform. Chem. Commun. 2011, 47, 961–963. [Google Scholar] [CrossRef]
- Kimling, J.; Maier, M.; Okenve, B.; Kotaidis, V.; Ballot, H.; Plech, A. Turkevich method for gold nanoparticle synthesis revisited. J. Phys. Chem. B 2006, 110, 15700–15707. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhan, S.; Wang, L.; Zhou, P. Selection of a DNA aptamer for cadmium detection based on cationic polymer mediated aggregation of gold nanoparticles. Analyst 2014, 139, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Alba-Molina, D.; Martin-Romero, M.T.; Camacho, L.; Giner-Casares, J.J. Ion-Mediated Aggregation of Gold Nanoparticles for Light-Induced Heating. Appl. Sci. 2017, 7, 916. [Google Scholar] [CrossRef]
- Zhao, W.; Chiuman, W.; Lam, J.C.; McManus, S.A.; Chen, W.; Cui, Y.; Pelton, R.; Brook, M.A.; Li, Y. DNA aptamer folding on gold nanoparticles: From colloid chemistry to biosensors. J. Am. Chem. Soc. 2008, 130, 3610–3618. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.T.; LaChance, A.M.; Zeng, S.; Liu, B.; Sun, L. Synthesis, properties, and applications of graphene oxide/reduced graphene oxide and their nanocomposites. Nano Mater. Sci. 2019, 1, 31–47. [Google Scholar] [CrossRef]
- Offeman, W.S.H.J.a.R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Lakowicz, J.R.; Weber, G. Quenching of fluorescence by oxygen. A probe for structural fluctuations in macromolecules. Biochemistry 1973, 12, 4161–4170. [Google Scholar] [CrossRef]
- Duan, N.; Song, M.; Mi, W.; Wang, Z.; Wu, S. Effectively Selecting Aptamers for Targeting Aromatic Biogenic Amines and Their Application in Aptasensing Establishment. J. Agric. Food Chem. 2021. [Google Scholar] [CrossRef]
- Lin, K.; Wu, G. Isothermal Titration Calorimetry Assays to Measure Binding Affinities In Vitro. Methods Mol. Biol. 2019, 1893, 257–272. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, H.; Wang, J.; Xu, L.; Chen, H.; Pei, R. Selection and characterization of DNA aptamers for the development of light-up biosensor to detect Cd(II). Talanta 2016, 154, 498–503. [Google Scholar] [CrossRef]
- Overacker, R.D.; Plitzko, B.; Loesgen, S. Biolayer interferometry provides a robust method for detecting DNA binding small molecules in microbial extracts. Anal. Bioanal. Chem. 2021, 413, 1159–1171. [Google Scholar] [CrossRef]
- Ciesielski, G.L.; Hytonen, V.P.; Kaguni, L.S. Biolayer Interferometry: A Novel Method to Elucidate Protein-Protein and Protein-DNA Interactions in the Mitochondrial DNA Replisome. Methods Mol. Biol. 2016, 1351, 223–231. [Google Scholar] [CrossRef]
- Teague, J.L.; Barrows, J.K.; Baafi, C.A.; Van Dyke, M.W. Discovering the DNA-Binding Consensus of the Thermus thermophilus HB8 Transcriptional Regulator TTHA1359. Int. J. Mol. Sci. 2021, 22, 42. [Google Scholar] [CrossRef]
- Kamat, V.; Rafique, A. Designing binding kinetic assay on the bio-layer interferometry (BLI) biosensor to characterize antibody-antigen interactions. Anal. Biochem. 2017, 536, 16–31. [Google Scholar] [CrossRef]
- Wartchow, C.A.; Podlaski, F.; Li, S.; Rowan, K.; Zhang, X.; Mark, D.; Huang, K.S. Biosensor-based small molecule fragment screening with biolayer interferometry. J. Comput. Aided Mol. Des. 2011, 25, 669–676. [Google Scholar] [CrossRef]
- Cusano, A.M.; Aliberti, A.; Cusano, A.; Ruvo, M. Detection of small DNA fragments by biolayer interferometry. Anal. Biochem. 2020, 607, 113898. [Google Scholar] [CrossRef]
- Mahmoudinobar, F.; Britton, D.; Montclare, J.K. Protein-based lateral flow assays for COVID-19 detection. Protein Eng. Des. Sel. 2021, 34, gzab010. [Google Scholar] [CrossRef]
- Sachdeva, S.; Davis, R.W.; Saha, A.K. Microfluidic Point-of-Care Testing: Commercial Landscape and Future Directions. Front. Bioeng. Biotechnol. 2020, 8, 602659. [Google Scholar] [CrossRef]
- Boehringer, H.R.; O’Farrell, B.J. Lateral Flow Assays in Infectious Disease Diagnosis. Clin. Chem. 2021, 68, 52–58. [Google Scholar] [CrossRef]
- Wang, L.; Wang, R.; Wei, H.; Li, Y. Selection of aptamers against pathogenic bacteria and their diagnostics application. World J. Microbiol. Biotechnol. 2018, 34, 149. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Zhao, Y.; Yan, M.; Huang, Y.; Yan, J.; Bai, W.; Chen, A. A sandwich dipstick assay for ATP detection based on split aptamer fragments. Anal. Bioanal. Chem. 2016, 408, 4151–4158. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, L.; Chikkanna, A.; Chen, S.; Brusius, I.; Sbuh, N.; Veedu, R.N. Development of nucleic acid aptamer-based lateral flow assays: A robust platform for cost-effective point-of-care diagnosis. Theranostics 2021, 11, 5174–5196. [Google Scholar] [CrossRef] [PubMed]
- Petrakova, A.V.; Urusov, A.E.; Zherdev, A.V.; Dzantiev, B.B. Gold nanoparticles of different shape for bicolor lateral flow test. Anal. Biochem. 2019, 568, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.; Abazari, M.; Nourozi, M. Effects of two beta-adrenergic agonists on adipose tissue, plasma hormones and metabolites of Moghani ewes. Small Ruminant. Res. 2006, 63, 84–90. [Google Scholar] [CrossRef]
- Fan, F.S. Assessing the Possible Influence of Residues of Ractopamine, a Livestock Feed Additive, in Meat on Alzheimer Disease. Dement. Geriatr. Cogn. Dis. Extra 2021, 11, 110–113. [Google Scholar] [CrossRef]
- Wang, M.Y.; Zhu, W.; Ma, L.; Ma, J.J.; Zhang, D.E.; Tong, Z.W.; Chen, J. Enhanced simultaneous detection of ractopamine and salbutamol—Via electrochemical-facial deposition of MnO2 nanoflowers onto 3D RGO/Ni foam templates. Biosens. Bioelectron. 2016, 78, 259–266. [Google Scholar] [CrossRef]
- Bownik, A.; Kowalczyk, M.; Banczerowski, J. Lambda-cyhalothrin affects swimming activity and physiological responses of Daphnia magna. Chemosphere 2019, 216, 805–811. [Google Scholar] [CrossRef]
- Djouaka, R.; Soglo, M.F.; Kusimo, M.O.; Adeoti, R.; Talom, A.; Zeukeng, F.; Paraiso, A.; Afari-Sefa, V.; Saethre, M.G.; Manyong, V.; et al. The Rapid Degradation of Lambda-Cyhalothrin Makes Treated Vegetables Relatively Safe for Consumption. Int. J. Environ. Res. Public Health 2018, 15, 1536. [Google Scholar] [CrossRef]
- Bradberry, S.M.; Cage, S.A.; Proudfoot, A.T.; Vale, J.A. Poisoning due to pyrethroids. Toxicol. Rev. 2005, 24, 93–106. [Google Scholar] [CrossRef]
- Banerjee, G.; Chattopadhyay, P. Vanillin biotechnology: The perspectives and future. J. Sci. Food Agric. 2019, 99, 499–506. [Google Scholar] [CrossRef]
- Mourtzinos, I.; Konteles, S.; Kalogeropoulos, N.; Karathanos, V.T. Thermal oxidation of vanillin affects its antioxidant and antimicrobial properties. Food Chem. 2009, 114, 791–797. [Google Scholar] [CrossRef]
- Yadav, M.; Pandey, R.; Chauhan, N.S. Catabolic Machinery of the Human Gut Microbes Bestow Resilience Against Vanillin Antimicrobial Nature. Front. Microbiol. 2020, 11, 588545. [Google Scholar] [CrossRef]
- Schwab, W. Natural 4-hydroxy-2,5-dimethyl-3(2H)-furanone (Furaneol(R)). Molecules 2013, 18, 6936–6951. [Google Scholar] [CrossRef]
- Prester, L. Biogenic amines in fish, fish products and shellfish: A review. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess 2011, 28, 1547–1560. [Google Scholar] [CrossRef]
- Bogdanovic, T.; Petricevic, S.; Brkljaca, M.; Listes, I.; Pleadin, J. Biogenic amines in selected foods of animal origin obtained from the Croatian retail market. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess 2020, 37, 815–830. [Google Scholar] [CrossRef]
- Alvarez-Elcoro, S.; Enzler, M.J. The macrolides: Erythromycin, clarithromycin, and azithromycin. Mayo. Clin. Proc. 1999, 74, 613–634. [Google Scholar] [CrossRef]
- Ashraf, A.; Liu, G.; Yousaf, B.; Arif, M.; Ahmed, R.; Irshad, S.; Cheema, A.I.; Rashid, A.; Gulzaman, H. Recent trends in advanced oxidation process-based degradation of erythromycin: Pollution status, eco-toxicity and degradation mechanism in aquatic ecosystems. Sci. Total Environ. 2021, 772, 145389. [Google Scholar] [CrossRef]
- Harju, K.; Koskela, H.; Kremp, A.; Suikkanen, S.; de la Iglesia, P.; Miles, C.O.; Krock, B.; Vanninen, P. Identification of gymnodimine D and presence of gymnodimine variants in the dinoflagellate Alexandrium ostenfeldii from the Baltic Sea. Toxicon 2016, 112, 68–76. [Google Scholar] [CrossRef]
- Rowdhwal, S.S.S.; Chen, J. Toxic Effects of Di-2-ethylhexyl Phthalate: An Overview. Biomed. Res. Int. 2018, 2018, 1750368. [Google Scholar] [CrossRef]
- Deigan, K.E.; Ferre-D’Amare, A.R. Riboswitches: Discovery of drugs that target bacterial gene-regulatory RNAs. Acc. Chem. Res. 2011, 44, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Born, J.; Weitzel, K.; Suess, B.; Pfeifer, F. A Synthetic Riboswitch to Regulate Haloarchaeal Gene Expression. Front Microbiol. 2021, 12, 696181. [Google Scholar] [CrossRef] [PubMed]
- Topp, S.; Reynoso, C.M.; Seeliger, J.C.; Goldlust, I.S.; Desai, S.K.; Murat, D.; Shen, A.; Puri, A.W.; Komeili, A.; Bertozzi, C.R.; et al. Synthetic riboswitches that induce gene expression in diverse bacterial species. Appl. Environ. Microbiol. 2010, 76, 7881–7884. [Google Scholar] [CrossRef] [PubMed]
- Lotz, T.S.; Suess, B. Small-Molecule-Binding Riboswitches. Microbiol. Spectr. 2018, 6, RWR-0025-2018. [Google Scholar] [CrossRef]
- Groher, F.; Bofill-Bosch, C.; Schneider, C.; Braun, J.; Jager, S.; Geissler, K.; Hamacher, K.; Suess, B. Riboswitching with ciprofloxacin-development and characterization of a novel RNA regulator. Nucleic Acids Res. 2018, 46, 2121–2132. [Google Scholar] [CrossRef]
- Boussebayle, A.; Torka, D.; Ollivaud, S.; Braun, J.; Bofill-Bosch, C.; Dombrowski, M.; Groher, F.; Hamacher, K.; Suess, B. Next-level riboswitch development-implementation of Capture-SELEX facilitates identification of a new synthetic riboswitch. Nucleic Acids Res. 2019, 47, 4883–4895. [Google Scholar] [CrossRef]
- Mally, A.; Solfrizzo, M.; Degen, G.H. Biomonitoring of the mycotoxin Zearalenone: Current state-of-the art and application to human exposure assessment. Arch. Toxicol. 2016, 90, 1281–1292. [Google Scholar] [CrossRef]
- Liew, W.P.; Mohd-Redzwan, S. Mycotoxin: Its Impact on Gut Health and Microbiota. Front. Cell. Infect. Microbiol. 2018, 8, 60. [Google Scholar] [CrossRef]
- Saber, T.M.; Abd El-Aziz, R.M.; Ali, H.A. Quercetin mitigates fenitrothion-induced testicular toxicity in rats. Andrologia 2016, 48, 491–500. [Google Scholar] [CrossRef]

| Name | Class | Aptamer Sequence (5′–3′) | nt | Dissociation Constant (Kd) | Ref. |
|---|---|---|---|---|---|
| Kanamycin A CAS no.: 59-01-8 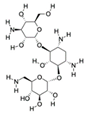 | Aminoglycoside antibiotic | ATACCAGCTTATTCAATTAGCCCGGTATTGAGGTCGATCTCTTATCCTATGGCTTGTCCCCCATGGCTCGGTTATATCCAGATAGTAAGTGCAATCT | 97 | M | [22] |
| Ofloxacin CAS no.: 82419-36-1 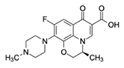 | Antibiotic | ATACCAGCTTATTCAATTGCAGGGTATCTGAGGCTTGATCTACTAAATGTCGTGGGGCATTGCTATTGGCGTTGATACGTACAATCGTAATCAGTTAG | 98 | 0.11 ± 0.06 nM | [39] |
| Furaneol CAS no.: 3658-77-3  | Aroma compound | CGACCAGCTCATTCCTCACCACGAGAAAGGAGCTCGATGAACTGCGAGCCGGATTCGACCCTATGCGAGTAGGTGGTGGATCCGAGCTCACCAGTC | 96 | 1.1 ± M | [68] |
| Vanillin CAS no.: 121-33-5 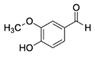 | Flavoring | CGACCAGCTCATTCCTCAGGAGAAACATGGAGTCTCGATGATGTAGGAGCGGCGGAACGTAGGAAGAGAGGATGACGGAGGATCCGAGCTCACCAGTC | 98 | 0.9 ± M | [23] |
| Erythromycin CAS no.: 114-07-8  | Antibiotic | AGGAATTCACGTCTCACTGGATTCACGCACGCCAAGGACTGCACTTAAGGTTAGATAGCCCCATGCAGTGAGTCAGGATATCG | 83 | 20 ± 9 nM | [24] |
| Spermine CAS no.: 71-44-3 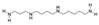 | Biogenic amine | TATGAACGATTTACTCGTACAGACGACACTTATCATTTGC | 40 | 9.648 ± 0.896 nM | [44] |
| Tetrodotoxin CAS no.: 4368-28-9  | Toxin | ATACCAGCTTATTCAATTTAATGCGGGGTGAGGCTCAATCAAGGAAAGATATAAGTAAGCAAAAAGGTCAAACAAGGGCGAGATAGTAAGTGCAATCT | 98 | 7 ± 1 nM | [43] |
| Chlorpromazine CAS no.: 50-53-3 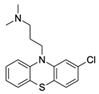 | Phenothiazine | TCGGAGGGAAGTGCACCCATTCTTGGAAACAGGAGCTCCTGAACCGCCCACACGC | 55 | 69.8 ± 9.81 nM | [69] |
| Roxithromycin CAS no.: 80214-83-1 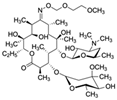 | Antibiotic | ATTGGCACTCCACGCATAGGCACACCCACCGGCCTAGCCACACCATGCTGCTGTTGCCCACCTATGCGTGCTACCGTGAA | 80 | 0.46 ± | [70] |
| Di(2-ethylhexyl) phthalate (DEHP) CAS no.: 117-81-7 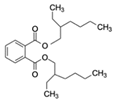 | Plasticizer | ACGCATAGGGTGCGACCACATACGCCCCATGTATGTCCCTTGGTTGTGCCCTATGCGT | 58 | 2.26 ± 0.06 nM | [71] |
| Acyclovir (ACV) CAS no.: 59277-89-3  | Aminoglycoside Antibiotics | TGAGCCCAAGCCCTGGTATGTGAAAACATACTAGACGTGGCTATGTATTTTTAAATCAATGGCAGGTCTACTTTGGGATC | 80 | 32.67 ± 4.127 nM | [72] |
| Glutamate CAS no.: 56-86-0 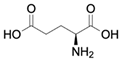 | Excitatory neurotransmitter | GCATCAGTCCACTCGTGAGGTCGACTGATGAGGCTCGATCAGGAGCGCCGCTCGATCGCACTTTCACAGGATAGTAGTTGGTAGCGACCTCTGCTAGA | 98 | 12 ± | [73] |
| Famciclovir (FCV) CAS no.: 104227-87-4 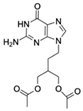 | Aminoglycoside Antibiotics | TGAGCCCAAGCCCTGGTATGTGAAAACATACTAGACGTGGCTATGTATTTTTAAATCAATGGCAGGTCTACTTTGGGATC | 80 | 47.35 ± 10.42 nM | [72] |
| Ganciclovir (GCV) CAS no.: 82410-32-0  | Aminoglycoside Antibiotics | TGAGCCCAAGCCCTGGTATGTGAAAACATACTAGACGTGGCTATGTATTTTTAAATCAATGGCAGGTCTACTTTGGGATC | 80 | 47.91 ± 13.47 nM | [72] |
| Penciclovir (PCV) CAS no.: 39809-25-1  | Aminoglycoside Antibiotics | TGAGCCCAAGCCCTGGTATGTGAAAACATACTAGACGTGGCTATGTATTTTTAAATCAATGGCAGGTCTACTTTGGGATC | 80 | 33.29 ± 5.851 nM | [72] |
| Levamisole (LEV) CAS no.: 14769-73-4 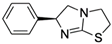 | Veterinary drug | AATCAAACGCTAAGGTCAAGGGAGAGTGCACCCATTCTTGGGGCCCCGGGCCAGCCCCGACACGCCGCCGAAGCTTGGTACCCGTATCGT | 90 | 66.15 ± 11.86 nM | [74] |
| Valaciclovir (VACV) CAS no.: 124832-26-4  | Aminoglycoside Antibiotics | TGAGCCCAAGCCCTGGTATGTGAAAACATACTAGACGTGGCTATGTATTTTTAAATCAATGGCAGGTCTACTTTGGGATC | 80 | 44.26 ± 6.744 nM | [72] |
| Tetrodotoxin CAS no.: 4368-28-9  | Toxin | ATACCAGCTTATTCAATTTAATGCGGGGTGAGGCTCAATCAAGGAAAGATATAAGTAAGCAAAAAGGTCAAACAAGGGCGAGATAGTAAGTGCAATCT | 98 | 7 ± 1 nM | [43] |
| Ribavirin CAS no.: 36791-04-5 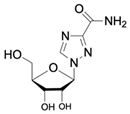 | Antiviral agent | AAAGTAATGCCCGGTAGTTATTCAAAGATGAGTAGGAAAAGA | 42 | 61.19 ± 21.48 nM | [45] |
| Gymnodimine-A CAS no.: 173792-58-0 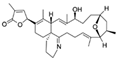 | Toxin | GCGACCGAAGTGAGGCTCGATCCAAGGTGGACGGGAGGTTGGATTGTGCGTG | 52 | 34.5 ± 1.72 nM | [75] |
| -cyhalothrin CAS no.: 91456-08-6  | Pesticide | AGGGGAAGCACGGGCGGGCG | 20 | 10.27 ± 1.33 nM | [76] |
| Malachite green (MG) CAS no.: 569-64-2 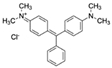 | Veterinary drug | (1) CGCAGCGCGGCAGACAGTCAGGCTCAGCACGTGGCA | 36 | 102.46 μM | [77] |
| (2) CACTCCACGCATAGGGACGCGAATTGCGGACCTATGTGTGGTGTG | 45 | 2.3 ± 0.2 μM | [78] | ||
| Leucomalachite green (LMG) CAS no.: 129-73-7  | Antimicrobial | CACTCCACGCATAGGGACGCGAATTGCGGACCTATGTGTGGTGTG | 45 | 8.2 ± 1.2 μM | [78] |
| Phorate CAS no.: 298-02-2  | Organothiophosphate insecticide | AAGCTTGCTTTATAGCCTGCAGCGATTCTTGATCGGAAAAGGCTGAGAGCTACGC | 55 | 1.11 μM | [79] |
| Profenofos CAS no.: 41198-08-7 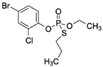 | Organothiophosphate insecticide | AAGCTTGCTTTATAGCCTGCAGCGATTCTTGATCGGAAAAGGCTGAGAGCTACGC | 55 | 1 μM | [79] |
| Isocarbophos CAS no.: 24353-61-5 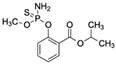 | Organothiophosphate insecticide | AAGCTTGCTTTATAGCCTGCAGCGATTCTTGATCGGAAAAGGCTGAGAGCTACGC | 55 | 0.83 μM | [79] |
| Omethoate CAS no.: 1113-02-6 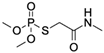 | Organothiophosphate insecticide | AAGCTTTTTTGACTGACTGCAGCGATTCTTGATCGCCACGGTCTGGAAAAAGAG | 54 | 2 μM | [79] |
| Cortisol CAS no.: 50-23-7 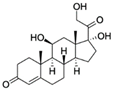 | Steroid hormone | GGAATGGATCCACATCCATGGATGGGCAATGCGGGGTGGAGAATGGTTGCCGCA CTTCGGCTTCACTGCAGA CTTGACGAAGCTT | 85 | 6.9 ± 2.8 nM | [80] |
| Tobramycin CAS no.: 32986-56-4  | Aminoglycoside antibiotic | CCATGATTCAACTTTACTGGTCTTGTCTTGGCTAGTCGTGTGTCATTCCCGTAAGGG | 57 | 200 nM | [47] |
| Clenbuterol hydrochloride (CLB) CAS no.: 21898-19-1  | beta-adrenergic agonist | AGCAGCACAGAGGTCAGATGTCATCTGAAGTGAATGAAGGTAAACATTATTTCATTAACACCTATGCGTGCTACCGTGAA | 80 | 76.61 ± 12.7 nM | [81] |
| Ractopamine CAS no.: 97825-25-7 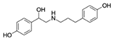 | Veterinary drug | AGCAGCACAGAGGTCAGATGGTCTCTACTAAAAGTTTTGATCATACCGTTCACTAATTGACCTATGCGTGCTACCGTGAA | 80 | 54.22 ± 8.02 nM | [82] |
| Atrazine CAS no.: 1912-24-9  | Herbicide | TGTACCGTCTGAGCGATTCGTACTTTATTCGGGAAGGGTATCAGCGGGGTTCAACAAGCCAGTCAGTCAGTGTTAAGGAGTGC | 83 | 3.7 nM | [83] |
| Zearalenone CAS no.: 17924-92-4 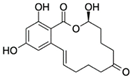 | Mycotoxin | ATACCAGCTTATTCAATTCTACCAGCTTTGAGGCTCGATCCAGCTTATTCAATTATACCAGCTTATTCAATTATACCAGCACAATCGTAATCAGTTAG | 98 | 15.2 ± 3.4 nM | [84] |
| Fenitrothion CAS no.: 122-14-5 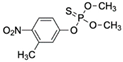 | Phosphorothioate insecticide | CTCTCGGGACGACGGGCCGAGTAGTCTCCACGATTGATCGGAAGTCGTCCC | 51 | 33.57 nM | [85] |
| Malathion CAS no.: 121-75-5  | Organophosphate insecticide | GGGATACAGGTAGTATGGCATGTGCTAGCGGTTGCA | 36 | 22.56 nM | [86] |
| Fipronil CAS no.: 120068-37-3 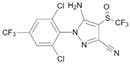 | Insecticide | ACGACAGATAGTGTGTACATGAAGGGTCGT | 30 | 15 nM | [87] |
| Diazinon CAS no.: 333-41-5  | Organophosphate insecticide | TTCCGATCAATCGTGGAGACTACTCGGCCC | 30 | 4.571 ± 0.714 µM | [88] |
| Ethyl carbamate (EC) CAS no.: 51-79-6 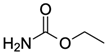 | Organic compound | GGGGGCACGGGAGGT | 15 | 17.97 ± 0.98 nM | [89] |
| 3,4-methylenedioxypyrov-alerone (MDPV) CAS no.: 687603-66-3 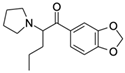 | Synthetic cathinone | CTTACGACTCAGGCATTTTGCCGGGTAACGAAGTTACTGTCGTAAG | 46 | 6.1 ± 0.2 μM | [90] |
| Dichlorodiphenyltrichloroethane (o,p’—DDT) CAS no.: 789-02-6 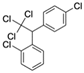 | Insecticide | TCCAGCACTCCACGCATAACGAATTGTGCTCAATGCGCCCCTGCAGTGAATGTGGAATTTGTTATGCGTGCGACGGTGAA | 80 | 412.3 ± 124.6 nM | [91] |
| Penicillin G CAS no.: 61-33-6 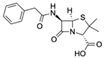 | -lactam antibiotic | GGGAGGACGAAGCGGAACGAGATGTAGATGAGGCTCGATCCGAATGCGTGACGTCTATCGGAATACTCGTTTTTACGCCTCAGAAGACACGCCCGACA | 98 | -- | [21] |
| Types | Aptamer Targets | Limit of Detection (LOD) | Detection Range | Ref. |
|---|---|---|---|---|
| Ion-sensitive field-effect transistor (ISFET) | Vanillin | 0.155 μM | 0.155–1.0 μM | [23] |
| Furaneol | -- | 0.1–10 μM | [68] | |
| Electrochemical impedance spectroscopy (EIS) | Penicillin G | 0.00051 μM | 1.2 nM–2.99 μM | [21] |
| Di(2-ethylhexyl) phthalate (DEHP) | 0.264 pM | -- | [71] | |
| Glutamate | 0.0013 pM | 0.01 pM–1 nM | [73] | |
| Fluorescent | Spermine | 0.052 nM | 0.1–20 nM | [44] |
| Tyramine (TYR) | 2.48 nM | 3.64–728.97 nM | [109] | |
| β-phenethylamine | 3.22 nM | 4.13–825.22 nM | [109] | |
| Acyclovir | 2.13 nM | 8.88–444.03 nM | [72] | |
| Famciclovir | 1.74 nM | 6.22–311.2 nM | [72] | |
| Ganciclovir | 2.08 nM | 7.84–391.80 nM | [72] | |
| Penciclovir | 1.97 nM | 7.9–394.85 nM | [72] | |
| Valaciclovir | 1.17 nM | 6.17–308.32 nM | [72] | |
| Ribavirin | 2.74 nM | 4.09–204.75 nM | [45] | |
| Malachite Green | 5.84 nM | 4.69 nM–2.35 μM | [77] | |
| Cadmium ions (Cd(II)) | 40 nM | 0–1000 nM | [111] | |
| Clenbuterol hydrochloride (CLB) | 0.22 nM | 0.32–159.44 nM | [81] | |
| Ractopamine | 0.13 nM | 0.33–331.79 nM | [82] | |
| Chlorpromazine | 0.67 nM | 1–100 nM | [69] | |
| Fenitrothion | 14 nM | 0–80 nM | [85] | |
| Malathion | 6.08 nM | -- | [86] | |
| Fipronil | 3.4 nM | 0–70 nM | [87] | |
| Diazinon | 148 nM | 0.1–25 μM | [88] | |
| Lateral flow aptasensor (LFA) | Erythromycin | 3 pM | 1 pM–10 nM | [24] |
| Ethyl carbamate | 0.024 μM | 0.11–0.67 μM | [89] | |
| Colorimetric | Roxithromycin | 0.077 μM | 0–4.44 μM | [70] |
| -cyhalothrin | 41 nM | 0.22–1.11 μM | [76] | |
| Zearalenone | 12.5 nM | 12.5–402.1 nM | [84] | |
| Levamisole | 1.12 nM | 1–200 nM | [74] | |
| Biolayer interferometry (BLI) | Gymnodimine-A | 6.21 nM | 55–1400 nM | [75] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lam, S.Y.; Lau, H.L.; Kwok, C.K. Capture-SELEX: Selection Strategy, Aptamer Identification, and Biosensing Application. Biosensors 2022, 12, 1142. https://doi.org/10.3390/bios12121142
Lam SY, Lau HL, Kwok CK. Capture-SELEX: Selection Strategy, Aptamer Identification, and Biosensing Application. Biosensors. 2022; 12(12):1142. https://doi.org/10.3390/bios12121142
Chicago/Turabian StyleLam, Sin Yu, Hill Lam Lau, and Chun Kit Kwok. 2022. "Capture-SELEX: Selection Strategy, Aptamer Identification, and Biosensing Application" Biosensors 12, no. 12: 1142. https://doi.org/10.3390/bios12121142
APA StyleLam, S. Y., Lau, H. L., & Kwok, C. K. (2022). Capture-SELEX: Selection Strategy, Aptamer Identification, and Biosensing Application. Biosensors, 12(12), 1142. https://doi.org/10.3390/bios12121142





