Advanced Lab-on-Fiber Optrodes Assisted by Oriented Antibody Immobilization Strategy
Abstract
1. Introduction
2. Materials and Methods
2.1. Instrumentation and Reagents
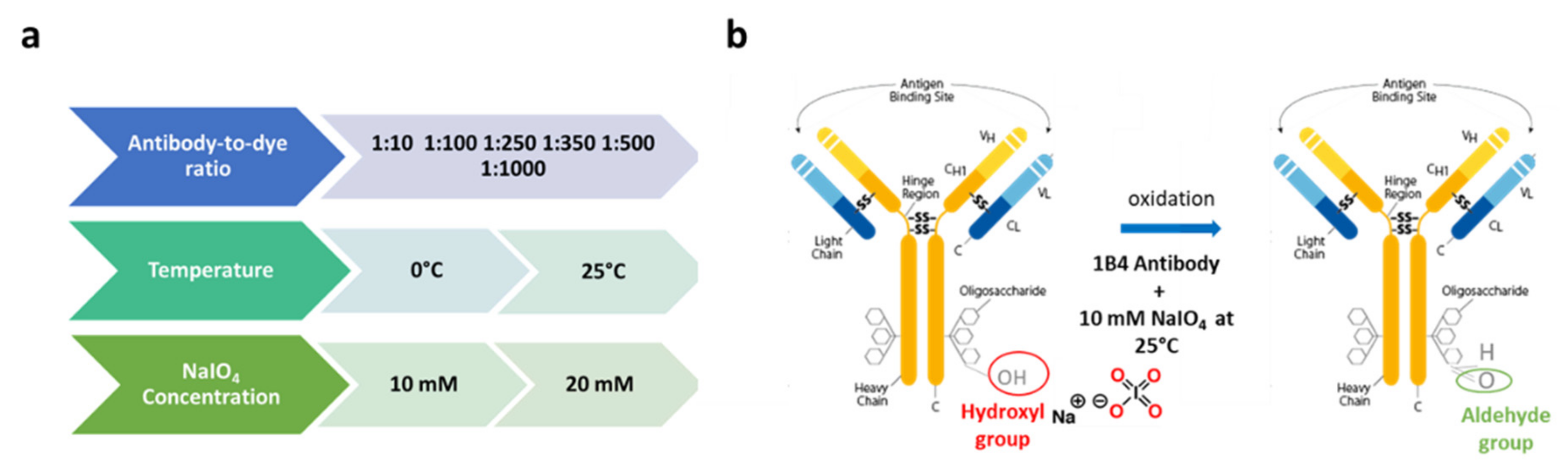
2.2. Biofunctionalization Protocol
2.2.1. Oxidized 1B4 Characterization
2.2.2. Immobilization on a Flat Gold Surface
2.2.3. SPR Assay: Analyses on Gold Chip (GC) Biosensor
2.3. Lab-on-Fiber Sensing Platforms
2.3.1. Bio-Layer Interferometry Analyses
2.3.2. Optical Fiber Meta-Tips (OFMTs)
2.3.3. OFMT Biofunctionalization
3. Results and Discussion
3.1. Biofunctionalization Protocol
3.1.1. Characterization of Ox-1B4
3.1.2. Recognition Efficiency and Quality Assessment of Ox-1B4
3.1.3. SPR Assay Optimization on GC Device
3.2. Lab-on-Fiber Sensing Platforms
- –
- A BLI-based platform exploiting a plastic optical fiber;
- –
- An advanced LoF biosensor based on highly sensitive OFMTs.
3.2.1. Cripto-1 Detection through BLI
3.2.2. Cripto-1 Detection through OFMT-Assisted Biosensors
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baig, N.; Kammakakam, I.; Falath, W. Nanomaterials: A Review of Synthesis Methods, Properties, Recent Progress, and Challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar] [CrossRef]
- Ali, J. Biosensors: Their Fundamentals, Designs, Types and Most Recent Impactful Applications: A Review. J. Biosens. Bioelectron. 2017, 8, 235. [Google Scholar] [CrossRef]
- Abid, S.A.; Ahmed Muneer, A.; Al-Kadmy, I.M.S.; Sattar, A.A.; Beshbishy, A.M.; Batiha, G.E.S.; Hetta, H.F. Biosensors as a Future Diagnostic Approach for COVID-19. Life Sci. 2021, 273, 119117. [Google Scholar] [CrossRef] [PubMed]
- Dkhar, D.S.; Kumari, R.; Mahapatra, S.; Divya; Kumar, R.; Tripathi, T.; Chandra, P. Antibody-Receptor Bioengineering and Its Implications in Designing Bioelectronic Devices. Int. J. Biol. Macromol. 2022, 218, 225–242. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Byrne, H.; O’kennedy, R.J. Antibodies and Antibody-Derived Analytical Biosensors. Essays Biochem. 2016, 60, 9–18. [Google Scholar] [CrossRef]
- Quaglio, D.; Mangiardi, L.; Venditti, G.; Del Plato, C.; Polli, F.; Ghirga, F.; Favero, G.; Pierini, M.; Botta, B.; Mazzei, F. Site- Directed Antibody Immobilization by Resorc[4]Arene-Based Immunosensors. Chemistry 2020, 26, 8400–8406. [Google Scholar] [CrossRef]
- Shen, M.; Rusling, J.F.; Dixit, C.K. Site-Selective Orientated Immobilization of Antibodies and Conjugates for Immunodiagnostics Development. Methods 2017, 116, 95. [Google Scholar] [CrossRef]
- Makaraviciute, A.; Ramanaviciene, A. Site-Directed Antibody Immobilization Techniques for Immunosensors. Biosens. Bioelectron. 2013, 50, 460–471. [Google Scholar] [CrossRef]
- Peluso, P.; Wilson, D.S.; Do, D.; Tran, H.; Venkatasubbaiah, M.; Quincy, D.; Heidecker, B.; Poindexter, K.; Tolani, N.; Phelan, M.; et al. Optimizing Antibody Immobilization Strategies for the Construction of Protein Microarrays. Anal. Biochem. 2003, 312, 113–124. [Google Scholar] [CrossRef]
- Welch, N.G.; Scoble, J.A.; Muir, B.W.; Pigram, P.J. Orientation and Characterization of Immobilized Antibodies for Improved Immunoassays (Review). Biointerphases 2017, 12, 02D301. [Google Scholar] [CrossRef]
- González-Martínez, M.A.; Morais, S.; Puchades, R.; Maquieira, A.; Abad, A.; Montoya, A. Monoclonal Antibody-Based Flow- Through Immunosensor for Analysis of Carbaryl. Anal. Chem. 1997, 69, 2812–2818. [Google Scholar] [CrossRef]
- Penalva, J.; Puchades, R.; Maquieira, Á. Analytical Properties of Immunosensors Working in Organic Media. Anal. Chem. 1999, 71, 3862–3872. [Google Scholar] [CrossRef] [PubMed]
- Lou, D.; Ji, L.; Fan, L.; Ji, Y.; Gu, N.; Zhang, Y. Antibody-Oriented Strategy and Mechanism for the Preparation of Fluorescent Nanoprobes for Fast and Sensitive Immunodetection. Langmuir 2019, 35, 4860–4867. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Anderson, H.; Myrskog, A.; Dunér, G.; Ingemarsson, B.; Aastrup, T. Optimizing Immobilization on Two-Dimensional Carboxyl Surface: PH Dependence of Antibody Orientation and Antigen Binding Capacity. Anal. Biochem. 2010, 398, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, G.; Tripathi, K.; Okyem, S.; Driskell, J.D. PH Impacts the Orientation of Antibody Adsorbed onto Gold Nanoparticles. Bioconjug. Chem. 2019, 30, 1182–1191. [Google Scholar] [CrossRef]
- O’Shannessy, D.J.; Quarles, R.H. Specific Conjugation Reactions of the Oligosaccharide Moieties of Immunoglobulins. J. Appl. Biochem. 1985, 7, 347–355. [Google Scholar]
- Hoffman, W.L.; O’Shannessy, D.J. Site-Specific Immobilization of Antibodies by Their Oligosaccharide Moieties to New Hydrazide Derivatized Solid Supports. J. Immunol. Methods 1988, 112, 113–120. [Google Scholar] [CrossRef]
- Adesina, A.; Mashazi, P. Oriented Antibody Covalent Immobilization for Label-Free Impedimetric Detection of C-Reactive Protein via Direct and Sandwich Immunoassays. Front. Chem. 2021, 9, 371. [Google Scholar] [CrossRef]
- Wong, H.Y.; Marcu, L.G.; Bezak, E.; Parange, N.A. Review of Health Economics of Point-of-Care Testing Worldwide and Its Efficacy of Implementation in the Primary Health Care Setting in Remote Australia. Risk Manag. Healthc. Policy 2020, 13, 379. [Google Scholar] [CrossRef]
- Chen, Y.T.; Lee, Y.C.; Lai, Y.H.; Lim, J.C.; Huang, N.T.; Lin, C.T.; Huang, J.J. Review of Integrated Optical Biosensors for Point-of-Care Applications. Biosensors 2020, 10, 209. [Google Scholar] [CrossRef]
- Ricciardi, A.; Crescitelli, A.; Vaiano, P.; Quero, G.; Consales, M.; Pisco, M.; Esposito, E.; Cusano, A. Lab-on-Fiber Technology: A New Vision for Chemical and Biological Sensing. Analyst 2015, 140, 8068–8079. [Google Scholar] [CrossRef] [PubMed]
- Pisco, M.; Cusano, A. Lab-On-Fiber Technology: A Roadmap toward Multifunctional Plug and Play Platforms. Sensors 2020, 20, 4705. [Google Scholar] [CrossRef] [PubMed]
- Cusano, A.; Consales, M.; Crescitelli, A.; Ricciardi, A. Book: Lab-on-Fiber Technology; Springer Series in Surface Sciences; Springer: Berlin/Heidelberg, Germany, 2015; Volume 56. [Google Scholar] [CrossRef]
- Quero, G.; Consales, M.; Severino, R.; Vaiano, P.; Boniello, A.; Sandomenico, A.; Ruvo, M.; Borriello, A.; Diodato, L.; Zuppolini, S.; et al. Long Period Fiber Grating Nano-Optrode for Cancer Biomarker Detection. Biosens. Bioelectron. 2016, 80, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Consales, M.; Quero, G.; Spaziani, S.; Principe, M.; Micco, A.; Galdi, V.; Cutolo, A.; Cusano, A. Metasurface-Enhanced Lab- on-Fiber Biosensors. Laser Photonics Rev. 2020, 14, 2000180. [Google Scholar] [CrossRef]
- Zuppolini, S.; Quero, G.; Consales, M.; Diodato, L.; Vaiano, P.; Venturelli, A.; Santucci, M.; Spyrakis, F.; Costi, M.P.; Giordano, M.; et al. Label-Free Fiber Optic Optrode for the Detection of Class C β-Lactamases Expressed by Drug Resistant Bacteria. Biomed. Opt. Express 2017, 8, 5191–5205. [Google Scholar] [CrossRef]
- Consales, M.; Ricciardi, A.; Crescitelli, A.; Esposito, E.; Cutolo, A.; Cusano, A. Lab-on-Fiber Technology: Toward Multifunctional Optical Nanoprobes. ACS Nano 2012, 6, 3163–3170. [Google Scholar] [CrossRef]
- Vaiano, P.; Carotenuto, B.; Pisco, M.; Ricciardi, A.; Quero, G.; Consales, M.; Crescitelli, A.; Esposito, E.; Cusano, A. Lab on Fiber Technology for Biological Sensing Applications. Laser Photonics Rev. 2016, 10, 858. [Google Scholar] [CrossRef]
- Managò, S.; Quero, G.; Zito, G.; Tullii, G.; Galeotti, F.; Pisco, M.; De Luca, A.C.; Cusano, A. Tailoring Lab-on-Fiber SERS Optrodes towards Biological Targets of Different Sizes. Sens. Actuators B Chem. 2021, 339, 129321. [Google Scholar] [CrossRef]
- Aliberti, A.; Ricciardi, A.; Giaquinto, M.; Micco, A.; Bobeico, E.; La Ferrara, V.; Ruvo, M.; Cutolo, A.; Cusano, A. Microgel Assisted Lab-on-Fiber Optrode. Sci. Rep. 2017, 7, 14459. [Google Scholar] [CrossRef]
- Huang, H.L.; Lu, P.H.; Yang, H.C.; Lee, G.D.; Li, H.R.; Liao, K.C. Fiber-Optic Triggered Release of Liposome in Vivo: Implication of Personalized Chemotherapy. Int. J. Nanomed. 2015, 10, 5171. [Google Scholar] [CrossRef]
- Principe, S.; Giaquinto, M.; Micco, A.; Cutolo, M.A.; Riccio, M.; Breglio, G.; Irace, A.; Ricciardi, A.; Cusano, A. Thermo- Plasmonic Lab-on-Fiber Optrodes. Opt. Laser Technol. 2020, 132, 106502. [Google Scholar] [CrossRef]
- Berruti, G.M.; Vaiano, P.; Boniello, A.; Principe, S.; Quero, G.; Persiano, G.V.; Consales, M.; Cusano, A. Highly Efficient Fiber Optic Thermal Heating Device Based on Turn-Around-Point Long Period Gratings. J. Lightwave Technol. 2022, 40, 797–804. [Google Scholar] [CrossRef]
- Principe, M.; Consales, M.; Castaldi, G.; Galdi, V.; Cusano, A. Evaluation of Fiber-Optic Phase-Gradient Meta-Tips for Sensing Applications. Nanomater. Nanotechnol. 2019, 9, 1847980419832724. [Google Scholar] [CrossRef]
- Principe, M.; Consales, M.; Micco, A.; Crescitelli, A.; Castaldi, G.; Esposito, E.; La Ferrara, V.; Cutolo, A.; Galdi, V.; Cusano, A. Optical Fiber Meta-Tips. Light Sci. Appl. 2017, 6, e16226-10. [Google Scholar] [CrossRef] [PubMed]
- Ucci, S.; Cicatiello, P.; Spaziani, S.; Cusano, A. (INVITED)Development of Custom Surface Plasmon Resonance Au Biosensor for Liver Cancer Biomarker Detection. Results Opt. 2021, 5, 100193. [Google Scholar] [CrossRef]
- Cusano, A.M.; Aliberti, A.; Cusano, A.; Ruvo, M. Detection of Small DNA Fragments by Biolayer Interferometry. Anal. Biochem. 2020, 607, 113898. [Google Scholar] [CrossRef]
- Strizzi, L.; Bianco, C.; Normanno, N.; Salomon, D. Cripto-1: A Multifunctional Modulator during Embryogenesis and Oncogenesis. Oncogene 2005, 24, 5731–5741. [Google Scholar] [CrossRef]
- Salomon, D.S.; Bianco, C.; Ebert, A.D.; Khan, N.I.; De Santis, M.; Normanno, N.; Wechselberger, C.; Seno, M.; Williams, K.; Sanicola, M.; et al. The EGF-CFC Family: Novel Epidermal Growth Factor-Related Proteins in Development and Cancer. Endocr.-Relat. Cancer 2000, 7, 199–226. [Google Scholar] [CrossRef]
- Bianco, C.; Strizzi, L.; Mancino, M.; Rehman, A.; Hamada, S.; Watanabe, K.; De Luca, A.; Jones, B.; Balogh, G.; Russo, J.; et al. Identification of Cripto-1 as a Novel Serologic Marker for Breast and Colon Cancer. Clin. Cancer Res. 2006, 12, 5158–5164. [Google Scholar] [CrossRef]
- Fontanini, G.; De Laurentiis, M.; Vignati, S.; Chinè, S.; Lucchi, M.; Silvestri, V.; Mussi, A.; De Placido, S.; Tortora, G.; Bianco, A.R.; et al. Evaluation of Epidermal Growth Factor-Related Growth Factors and Receptors and of Neoangiogenesis in Completely Resected Stage I-IIIA Non-Small-Cell Lung Cancer: Amphiregulin and Microvessel Count Are Independent Prognostic Indicators of Survival. Clin. Cancer Res. 1998, 4, 241–249. [Google Scholar]
- Xu, C.H.; Wang, Y.; Qian, L.H.; Yu, L.K.; Zhang, X.W.; Wang, Q.B. Serum Cripto-1 Is a Novel Biomarker for Non-Small Cell Lung Cancer Diagnosis and Prognosis. Clin. Respir. J. 2017, 11, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Bianco, C.; Strizzi, L.; Mancino, M.; Watanabe, K.; Gonzales, M.; Hamada, S.; Raafat, A.; Sahlah, L.; Chang, C.; Sotgia, F.; et al. Regulation of Cripto-1 Signaling and Biological Activity by Caveolin-1 in Mammary Epithelial Cells. Am. J. Pathol. 2008, 172, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Llovet, J.M.; Bruix, J. Hepatocellular Carcinoma. Lancet 2012, 379, 1245–1255. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.; Chi, X.; Fan, Y.; Shi, Y.; Niu, J. High Level of Serum Cripto-1 in Hepatocellular Carcinoma, Especially with Hepatitis B Virus Infection. Medicine 2018, 97, e11781. [Google Scholar] [CrossRef] [PubMed]
- Sandberg, C.J.; Altschuler, G.; Jeong, J.; Strømme, K.K.; Stangeland, B.; Murrell, W.; Grasmo-Wendler, U.-H.; Myklebost, O.; Helseth, E.; Vik-Mo, E.O.; et al. Comparison of Glioma Stem Cells to Neural Stem Cells from the Adult Human Brain Identifies Dysregulated Wnt- Signaling and a Fingerprint Associated with Clinical Outcome. Exp. Cell Res. 2013, 319, 2230–2243. [Google Scholar] [CrossRef]
- Nagaoka, T.; Karasawa, H.; Castro, N.P.; Rangel, M.C.; Salomon, D.S.; Bianco, C. An Evolving Web of Signaling Networks Regulated by Cripto-1. Rev. Growth Factors 2012, 30, 13–21. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Mi, X.G.; Song, Z.B.; Li, Y.X.; YingShi; Niu, J.Q. Cripto-1 Promotes Resistance to Drug-Induced Apoptosis by Activating the TAK-1/NF-ΚB/Survivin Signaling Pathway. Biomed. Pharmacother. 2018, 104, 729–737. [Google Scholar] [CrossRef]
- Watanabe, K.; Bianco, C.; Strizzi, L.; Hamada, S.; Mancino, M.; Bailly, V.; Mo, W.; Wen, D.; Miatkowski, K.; Gonzales, M.; et al. Growth Factor Induction of Cripto-1 Shedding by Glycosylphosphatidylinositol-Phospholipase D and Enhancement of Endothelial Cell Migration. J. Biol. Chem. 2007, 282, 31643–31655. [Google Scholar] [CrossRef]
- Sun, G.; Yan, S.S.; Shi, L.; Wan, Z.Q.; Jiang, N.; Fu, L.S.; Li, M.; Guo, J. MicroRNA-15b Suppresses the Growth and Invasion of Glioma Cells through Targeted Inhibition of Cripto-1 Expression. Mol. Med. Rep. 2016, 13, 4897–4903. [Google Scholar] [CrossRef]
- Sandomenico, A.; Ruvo, M. Targeting Nodal and Cripto-1: Perspectives Inside Dual Potential Theranostic Cancer Biomarkers. Curr. Med. Chem. 2018, 26, 1994–2050. [Google Scholar] [CrossRef]
- Klauzinska, M.; Castro, N.P.; Rangel, M.C.; Spike, B.T.; Gray, P.C.; Bertolette, D.; Cuttitta, F.; Salomon, D. The Multifaceted Role of the Embryonic Gene Cripto-1 in Cancer, Stem Cells and Epithelial-Mesenchymal Transition. Semin. Cancer Biol. 2014, 29, 51–58. [Google Scholar] [CrossRef]
- Xu, C.H.; Sheng, Z.H.; Hu, H.D.; Hao, K.K.; Wang, Q.B.; Yu, L.K. Elevated Expression of Cripto-1 Correlates with Poor Prognosis in Non-Small Cell Lung Cancer. Tumor Biol. 2014, 35, 8673–8678. [Google Scholar] [CrossRef] [PubMed]
- Kenney, N.J.; Smith, G.H.; Maroulakou, G.; Green, J.H.; Muller, W.J.; Callahan, R.; Salomon, D.; Dickson, R.B. Detection of Amphiregulin and Cripto-I in Mammary Tumors From Transgenic Mice. Mol. Carcinog. 1996, 15, 44–56. [Google Scholar] [CrossRef]
- Focà, G.; Iaccarino, E.; Focà, A.; Sanguigno, L.; Untiveros, G.; Cuevas-Nunez, M.; Strizzi, L.; Leonardi, A.; Ruvo, M.; Sandomenico, A. Development of Conformational Antibodies Targeting Cripto-1 with Neutralizing Effects in Vitro. Biochimie 2019, 158, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Love, J.C.; Estroff, L.A.; Kriebel, J.K.; Nuzzo, R.G.; Whitesides, G.M. Self-Assembled Monolayers of Thiolates on Metals as a Form of Nanotechnology. Chem. Rev. 2005, 105, 1103–1169. [Google Scholar] [CrossRef] [PubMed]
- Häkkinen, H. The Gold-Sulfur Interface at the Nanoscale. Nat. Chem. 2012, 4, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Rezaei, T. A New Optimal Diagnosis System for Coronavirus (COVID-19) Diagnosis Based on Archimedes Optimization Algorithm on Chest X-Ray Images. Comput. Intell. Neurosci. 2021, 2021, 7788491. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.A. IgG Structure and Function. Encycl. Immunobiol. 2016, 2, 15–22. [Google Scholar] [CrossRef]
- Kolenko, P.; Dohnálek, J.; Dušková, J.; Skálová, T.; Collard, R.; Hašek, J. New Insights into Intra- and Intermolecular Interactions of Immunoglobulins: Crystal Structure of Mouse IgG2b-Fc at 2.1-Å Resolution. Immunology 2009, 126, 378–385. [Google Scholar] [CrossRef]
- Wolfe, C.A.C.; Hage, D.S. Studies on the Rate and Control of Antibody Oxidation by Periodate. Anal. Biochem. 1995, 231, 123–130. [Google Scholar] [CrossRef]
- Hahn, C.D.; Leitner, C.; Weinbrenner, T.; Schlapak, R.; Tinazli, A.; Tampé, R.; Lackner, B.; Steindl, C.; Hinterdorfer, P.; Gruber, H.J.; et al. Self-Assembled Monolayers with Latent Aldehydes for Protein Immobilization. Bioconjug. Chem. 2007, 18, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Valdes, T.I.; Moussy, F. In Vitro and In Vivo Degradation of Glucose Oxidase Enzyme Used for an Implantable Glucose Biosensor. Comparative Study. Diabetes Technol. Ther. 2004, 2, 367–376. [Google Scholar] [CrossRef] [PubMed]




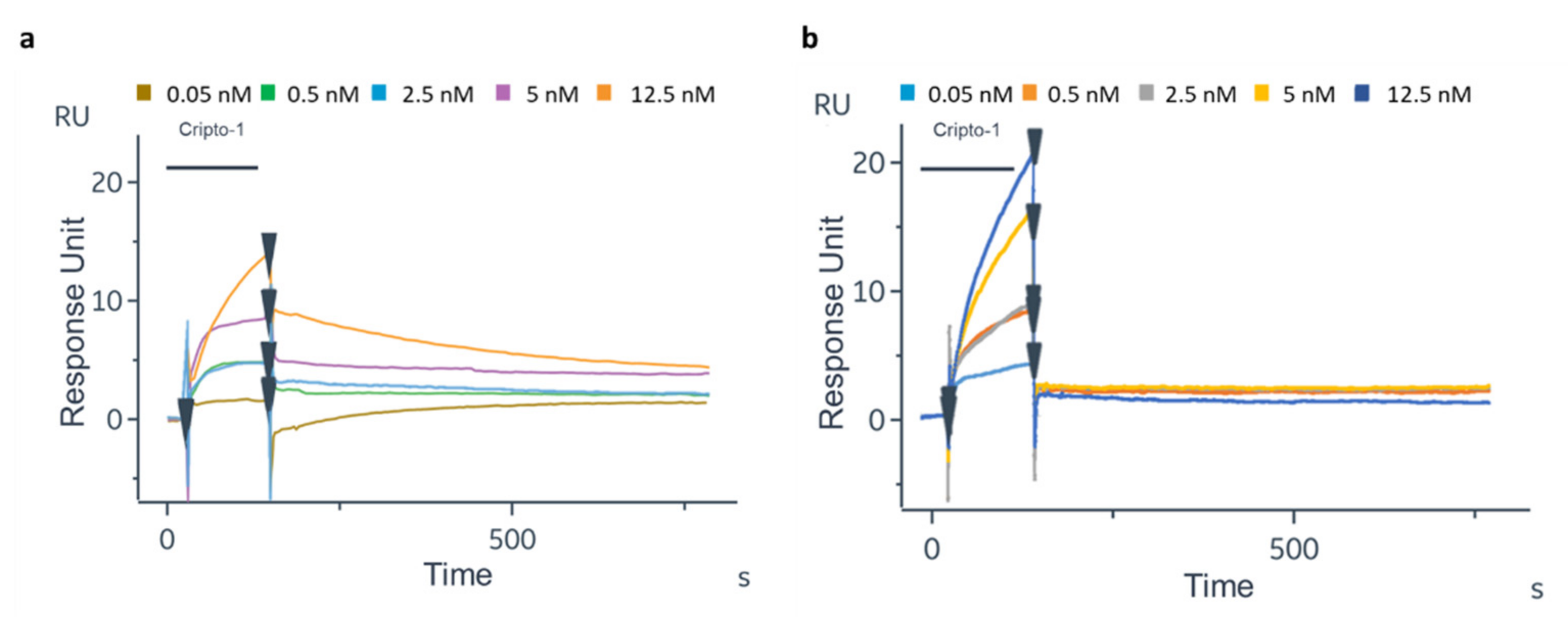



| Cripto-1 (nM) | Oriented 1B4 | Random 1B4 |
|---|---|---|
| ΔRU | ||
| 0.05 | 0.75 ± 0.07 | 0.35 ± 0.07 |
| 0.5 | 3.90 ± 0.40 | 0.35 ± 0.35 |
| 2.5 | 5.05 ± 0.20 | 0.35 ± 0.07 |
| 5 | 7.35 ± 0.07 | 0.76 ± 0.15 |
| 12.5 | 7.50 ± 1.97 | 1.86 ± 0.60 |
| Shift Ab (nm) | Cripto-1 (nM) | Shift Cripto-1 (nm) | ||
|---|---|---|---|---|
| Oriented 1B4 30 μg/mL | Random 1B4 30 μg/mL | Oriented 1B4 | Random 1B4 | |
| 2.06 ± 0.45 | 2.36 ± 0.62 | 0 | 0.001 ± 0.0006 | 0.001 ± 0.00025 |
| 12.5 | 0.33 ± 0.04 | 0.11 ± 0.08 | ||
| 50 | 0.49 ± 0.08 | 0.20 ± 0.07 | ||
| 500 | 0.84 ± 0.03 | 0.7 ± 0.17 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ucci, S.; Spaziani, S.; Quero, G.; Vaiano, P.; Principe, M.; Micco, A.; Sandomenico, A.; Ruvo, M.; Consales, M.; Cusano, A. Advanced Lab-on-Fiber Optrodes Assisted by Oriented Antibody Immobilization Strategy. Biosensors 2022, 12, 1040. https://doi.org/10.3390/bios12111040
Ucci S, Spaziani S, Quero G, Vaiano P, Principe M, Micco A, Sandomenico A, Ruvo M, Consales M, Cusano A. Advanced Lab-on-Fiber Optrodes Assisted by Oriented Antibody Immobilization Strategy. Biosensors. 2022; 12(11):1040. https://doi.org/10.3390/bios12111040
Chicago/Turabian StyleUcci, Sarassunta, Sara Spaziani, Giuseppe Quero, Patrizio Vaiano, Maria Principe, Alberto Micco, Annamaria Sandomenico, Menotti Ruvo, Marco Consales, and Andrea Cusano. 2022. "Advanced Lab-on-Fiber Optrodes Assisted by Oriented Antibody Immobilization Strategy" Biosensors 12, no. 11: 1040. https://doi.org/10.3390/bios12111040
APA StyleUcci, S., Spaziani, S., Quero, G., Vaiano, P., Principe, M., Micco, A., Sandomenico, A., Ruvo, M., Consales, M., & Cusano, A. (2022). Advanced Lab-on-Fiber Optrodes Assisted by Oriented Antibody Immobilization Strategy. Biosensors, 12(11), 1040. https://doi.org/10.3390/bios12111040












