Lab-on-Paper Devices for Diagnosis of Human Diseases Using Urine Samples—A Review
Abstract
:1. Introduction
2. Sample Pretreatment on Paper Devices
2.1. Sample Collection and Storage
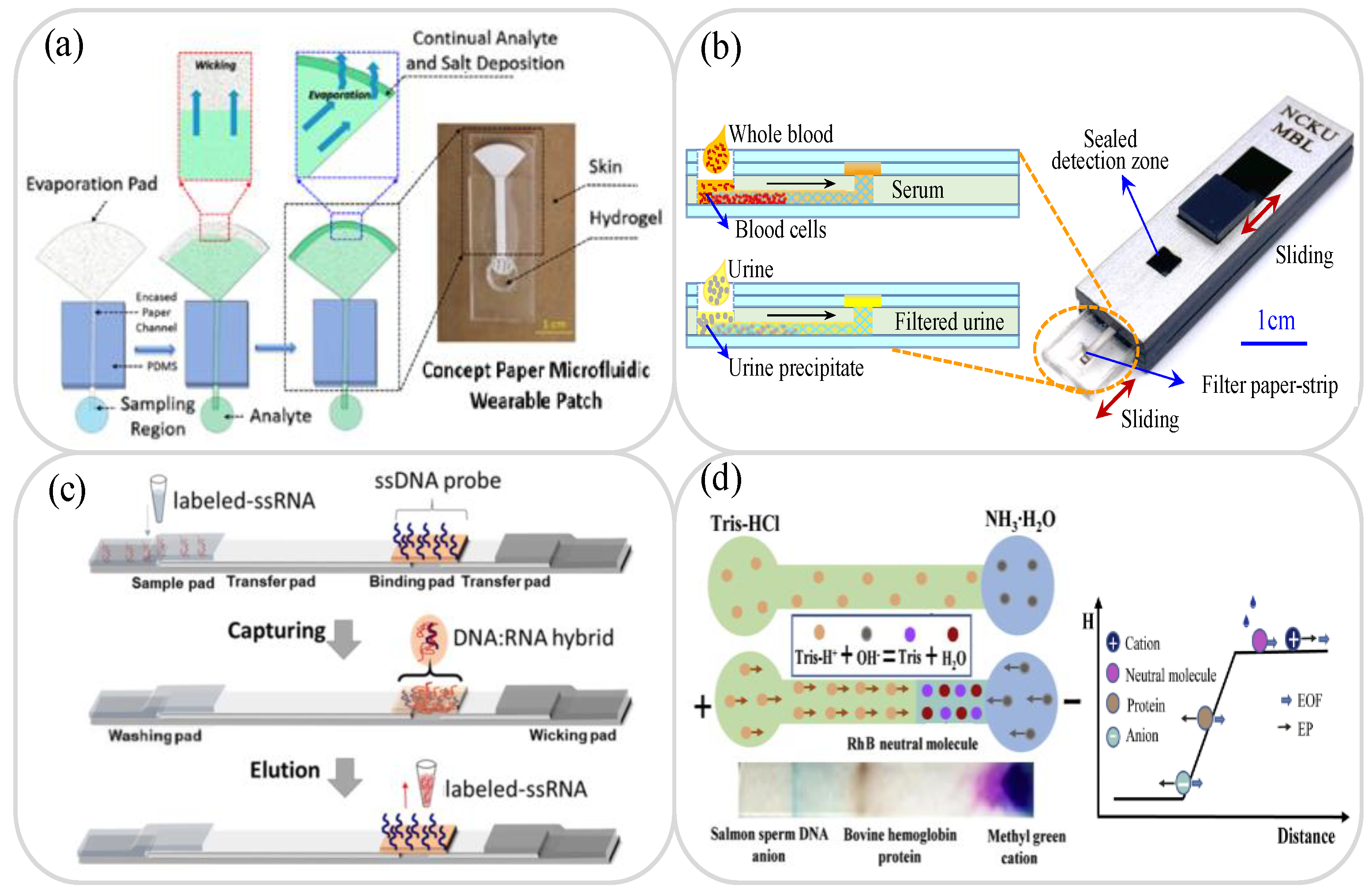
2.2. Sample Separation
2.3. Sample Extraction
2.4. Sample Concentration/Amplification
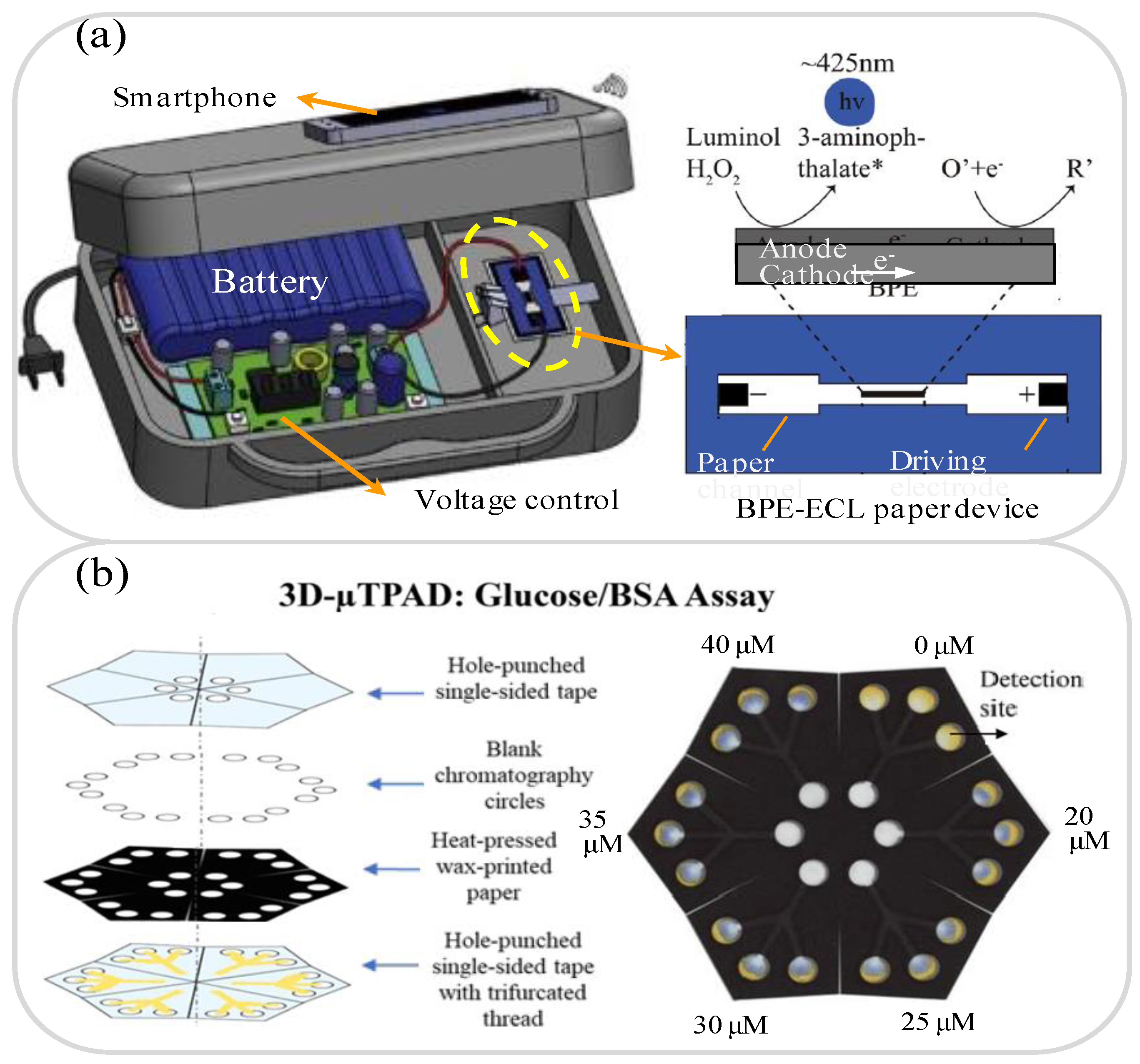
3. Application of Lab-on-Paper Platforms to Human Disease Diagnosis Using Urine Samples
3.1. Glucose Analysis
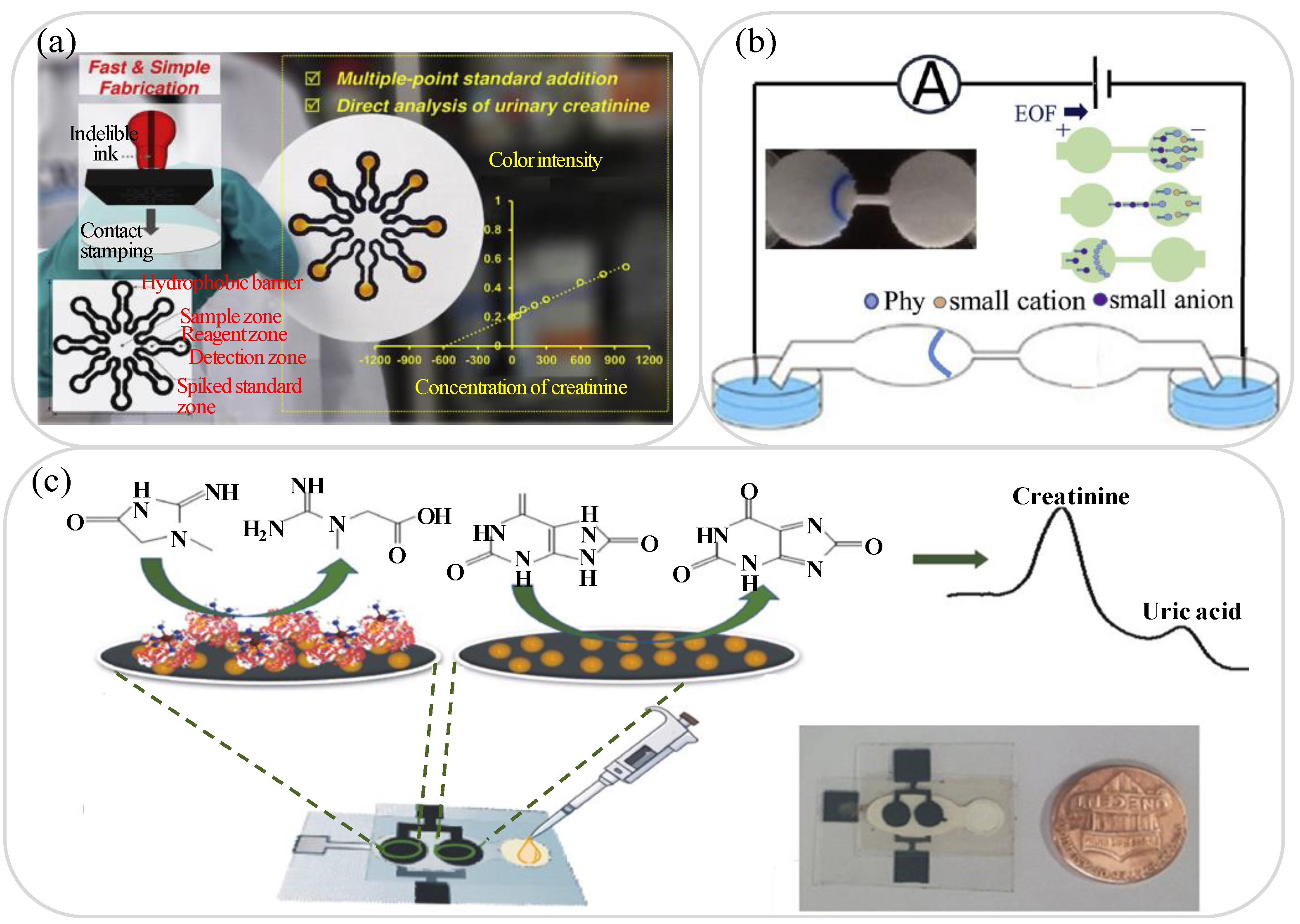
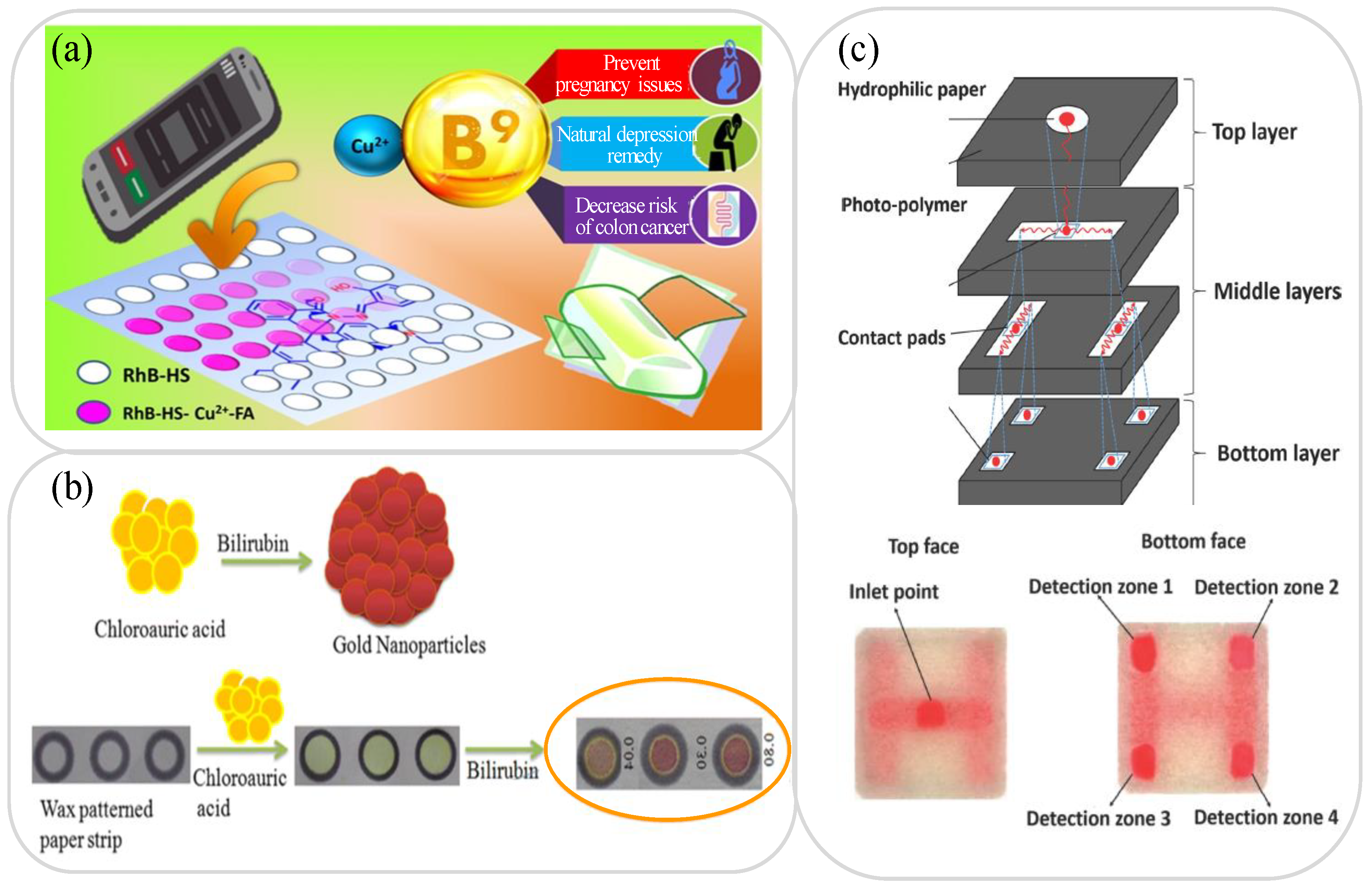
3.2. Creatinine, Albumin, Protein and Uric Acid Analysis
| Ref. | Materials and Structures | Fabrication Methods | Detection Methods | Target and Sample Matrices | Detection Range | Detection Limit |
|---|---|---|---|---|---|---|
| [87] | Filter paper, 3-D | Wax printing + screen printing | EC | Glucose | 0.1–40 mM | 0.03 mM |
| [89] | Filter paper, 3-D | Wax printing + screen printing | EC | Glucose | 0.5–5 mM | 0.5 mM |
| [90] | Filter paper, 2-D | Laser cutting | CM | Glucose | 0–150 μM | 2.5 μM |
| [95] | Filter paper, 3-D | Wax printing + screen printing | EC | Glucose Creatinine Uric acid | 0–5 mM | 0.12 μM 0.084 μM 0.012μM |
| [96] | Filter paper, 2-D | Cutting + coating | EC | Glucose | 0.1–1 mM | 25 μM |
| [97] | Filter paper, 2-D | Cutting | CM | Glucose | 0–56 mM | 0.54 mM |
| [98] | Thread + Chromatography paper, 3-D | Wax printing + cutting | CM | Glucose | 0–15 mM | 0.5 mM |
| [99] | Chromatography paper, 2-D | Inkjet printing | CM | Glucose | 0.01–4 mg/mL | 0.01 mg/mL |
| [100] | Filter paper, 3-D | Spray + punch | CM | Glucose Nitrite | 0.05–0.7 mM 0.02–0.9 mM | 25 μM 48 μM |
| [105] | Filter paper, 2-D | Cutting | CM | Creatinine | 10–60 mg/L | 4.2 mg/L |
| [107] | Filter paper, 2-D | Cutting | ISE | Sodium Potassium Creatinine | 0–36.99 mM 0–7.78 mM 0–15.65 mg/dL | 0.95 mM 0.47 mM 0.59 mg/dL |
| [117] | Filter paper, 2-D | Wax printing | CM | Creatinine Uric acid | 50–600 mg/L 50–500 mg/L | 15.7 mg/L 16.5 mg/L |
| [121] | Filter paper, 3-D | Inkjet printing + screen printing | EC | Dopamine Uric acid | 20–1000 μM | 2.19 μM 1.80 μM |
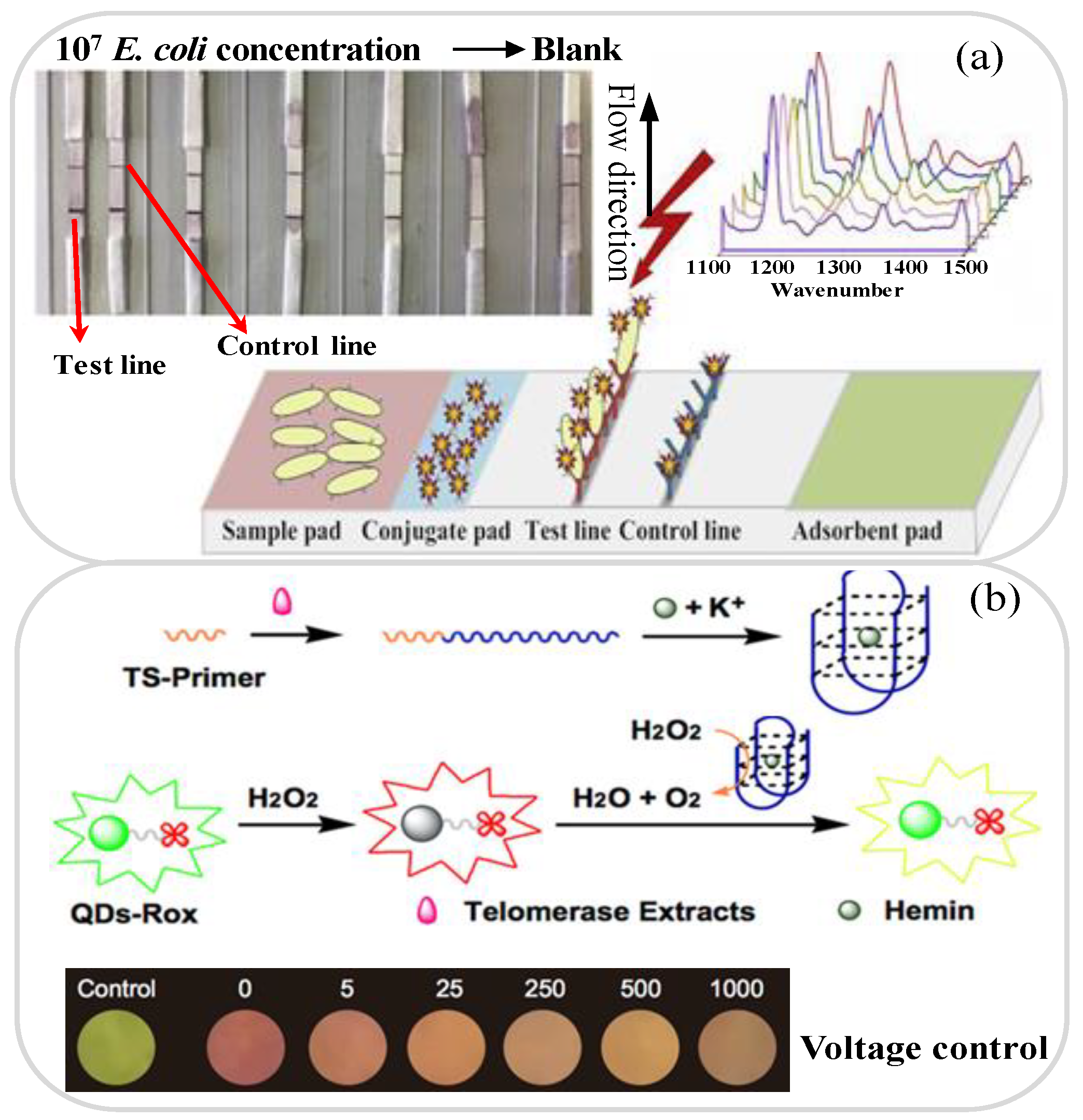
3.3. Bacteria and Cancer Analysis
3.4. Drugs and Ion Analysis
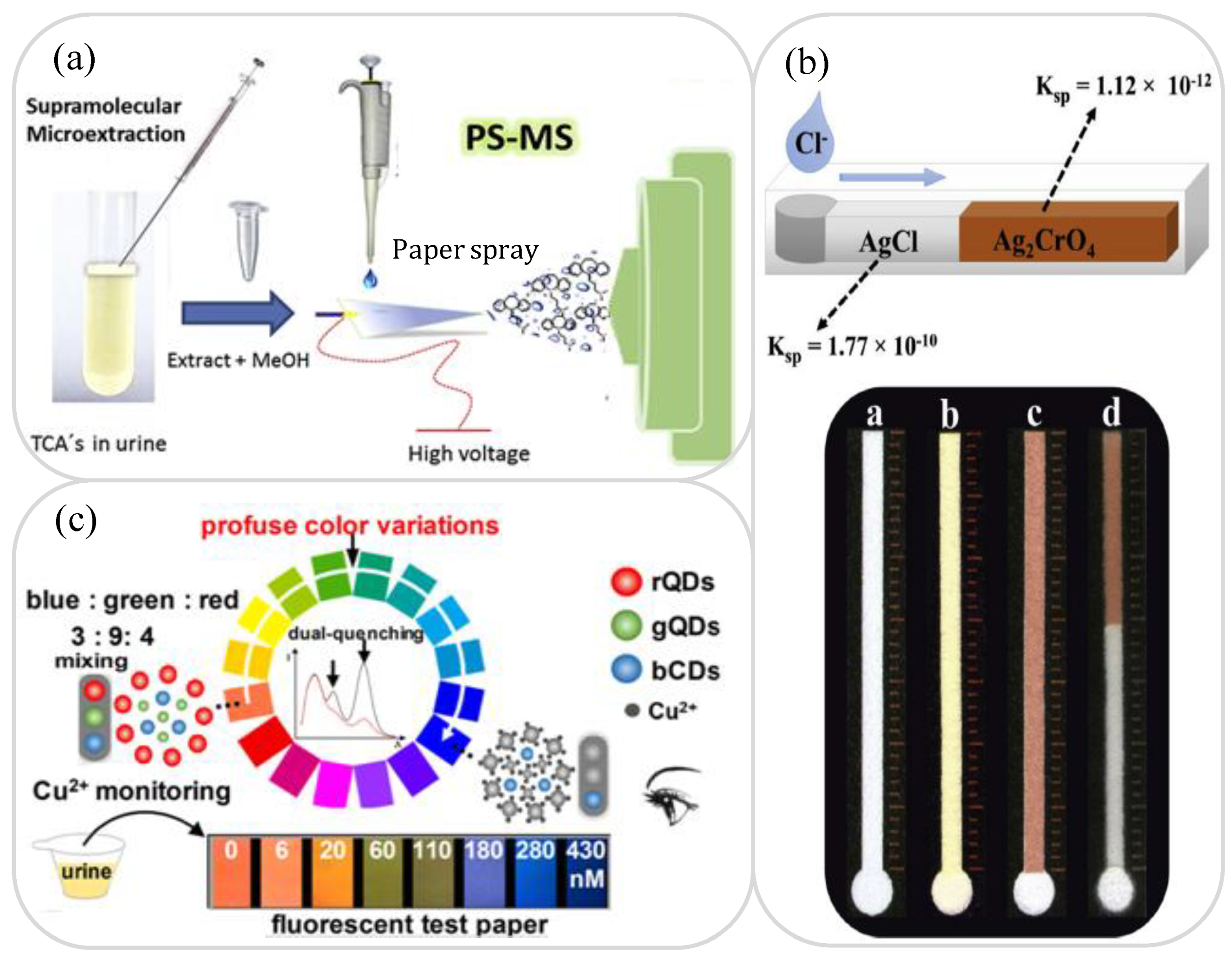
3.5. Other Analytes
| Ref. | Materials and Structures | Fabrication Methods | Detection Methods | Target and Sample Matrices | Detection Range | Detection Limit |
|---|---|---|---|---|---|---|
| [126] | Stripe paper, 3-D | Cutting + bonding | SERS | E. coil | 101–107 CFU/mL | 0.52 CFU/mL |
| [128] | Filter paper, 3-D | Wax printing + cutting | CM | Bacterial ATP | 3.3 × 10−9–3.3 × 10−15 mol | 3.8 × 10−14 mol |
| [130] | Filter paper, 2-D | Deposition | SERS | Adenosine | 3.8–4.9 μM | 3.8 μM |
| [134] | Filter paper, 2-D | Laser cutting | CM | miRNA-21 | 10–1000 pM | 4.1 pM |
| [138] | Chromatography paper, 2-D | Cutting | PS-MS | Caffeine CBD CBN Cocaine | 10–100 ng/mL | 7.8 ng/mL 11.4 ng/mL 1.4 ng/mL 2.1 ng/mL |
| [140] | Filter paper, 2-D | Cutting | MS |
Indomethacin
Diclofenac Tolmetin Ketoprofen Naproxen Ibuprofen |
25–1000 μg/L 25–1000 μg/L 25–1000 μg/L 50–1000 μg/L 50–1000 μg/L 50–1000 μg/L |
3.8 μg/L 7.2 μg/L 6.8 μg/L 9.4 μg/L 15.7 μg/L 5.1 μg/L |
| [142] | Filter paper, 2-D | Cutting | CM | Ketamine | 1–100 mM | 1 mM |
| [147] | Filter paper, 2-D | Wax printing | FLU | Au | 0–750 nM | 110 nM |
| [148] | Test card, 2-D | Wax printing | CM | Iodine | 50–300 μg I/L | 20 μg I/L |
| [150] | Filter paper, 2-D | Wax printing | FLU | Cu2+ | 5–1250 nM | 5 nM |
| [152] | Filter paper, 2-D | Screen printing | EC | Dopamine Serotonin | 30–800 μM 6–100 μM | 0.13 μM 0.39 μM |
| [153] | Filter paper, 2-D | Cutting | FLU | Folic acid | 1–300 μM | 0.28 μM |
| [158] | Stripe paper, 3-D | Cutting + bonding | CM | Biomarker 1 | 30–50 ng/mL | 5 ng/mL |
| [161] | Filter paper, 2-D | Immersed | CM | 17β-estradio | 0.1–1.0 μg/L | 0.25 μg/L |
| [162] | Filter paper, 2-D | Wax printing | CM | Cysteine | 0–1 mM | 10 μM |
| [163] | Filter paper, 2-D | Wax printing | CM | Salbutamol | 0.025–1 μg/L | 0.025 μg/L |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ma, L.; Zhang, Z.; Li, X. Non-invasive disease diagnosis using surface-enhanced Raman spectroscopy of urine and saliva. Appl. Spectros. Rev. 2020, 55, 197–219. [Google Scholar] [CrossRef]
- Aitekenov, S.; Gaipov, A.; Bukasov, R. Detection and quantification of proteins in human urine. Talanta 2020, 233, 121718. [Google Scholar] [CrossRef] [PubMed]
- Jemmeli, D.; Marcoccio, E.; Moscone, D.; Dridi, C.; Arduini, F. Highly sensitive paper-based electrochemical sensor for reagent free detection of bisphenol A. Talanta 2020, 216, 120924. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; You, M.; Li, S.; Hu, J.; Liu, C.; Gong, Y.; Yang, H.; Xu, F. Paper-based point-of-care immunoassays: Recent advances and emerging trends. Biotechnol. Adv. 2020, 39, 107442. [Google Scholar] [CrossRef]
- Mao, M.-X.; Zheng, R.; Peng, C.-F.; Wei, X.-L. DNA–Gold Nanozyme-Modified Paper Device for Enhanced Colorimetric Detection of Mercury Ions. Biosensors 2020, 10, 211. [Google Scholar] [CrossRef]
- Ng, H.Y.; Lee, W.C.; Kung, C.T.; Li, L.C.; Lee, C.T.; Fu, L.M. Recent Advances in Microfluidic Devices for Contamination Detection and Quality Inspection of Milk. Micromachines 2021, 12, 558. [Google Scholar] [CrossRef]
- Baek, S.H.; Park, C.; Jeon, J.; Park, S. Three-Dimensional Paper-Based Microfluidic Analysis Device for Simultaneous Detection of Multiple Biomarkers with a Smartphone. Biosensors 2020, 10, 187. [Google Scholar] [CrossRef]
- Tseng, C.C.; Kung, C.T.; Chen, R.F.; Tsai, M.H.; Chao, H.R.; Wang, Y.N.; Fu, L.M. Recent advances in microfluidic paper-based assay devices for diagnosis of human diseases using saliva, tears and sweat samples. Sens. Actuators B Chem. 2021, 342, 130078. [Google Scholar] [CrossRef]
- Ireta-Muñoz, L.A.; Morales-Narváez, E. Smartphone and Paper-Based Fluorescence Reader: A Do It Yourself Approach. Biosensors 2020, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.F.; Paixão, T.R.L.C.; Torres, M.D.T.; de Araujo, W.R. Simple and inexpensive electrochemical paper-based analytical device for sensitive detection of Pseudomonas aeruginosa. Sens. Actuators B Chem. 2020, 308, 127669. [Google Scholar] [CrossRef]
- Primpray, V.; Chailapakul, O.; Tokeshi, M.; Rojanarata, T.; Laiwattanapaisal, W. A paper-based analytical device coupled with electrochemical detection for the determination of dexamethasone and prednisolone in adulterated traditional medicines. Anal. Chim. Acta 2019, 1078, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Pan, S.C.; Cheng, C.M. Paper-based human neutrophil elastase detection device for clinical wound monitoring. Lab Chip 2020, 20, 2709–2716. [Google Scholar] [CrossRef] [PubMed]
- Amin, R.; Ghaderinezhad, F.; Bridge, C.; Temirel, M.; Jones, S.; Toloueinia, P.; Tasoglu, S. Pushing the limits of spatial assay resolution for paper-based microfluidics using low-cost and high-throughput pen plotter approach. Micromachines 2020, 11, 611. [Google Scholar] [CrossRef] [PubMed]
- Kamel, S.; Khattab, T.A. Recent Advances in Cellulose-Based Biosensors for Medical Diagnosis. Biosensors 2020, 10, 67. [Google Scholar] [CrossRef]
- Murray, L.P.; Mace, C.R. Usability as a guiding principle for the design of paper-based, point-of-care devices–A review. Anal. Chim. Acta 2020, 1140, 236–249. [Google Scholar] [CrossRef]
- Chen, P.; Gates-Hollingsworth, M.; Pandit, S.; Park, A.; Montgomery, D.; AuCoin, D.; Gu, J.; Zenhausern, F. Paper-based Vertical Flow Immunoassay (VFI) for detection of bio-threat pathogens. Talanta 2019, 191, 81–88. [Google Scholar] [CrossRef]
- Hecht, L.; Rager, K.; Davidonis, M.; Weber, P.; Gauglitz, G.; Dietzel, A. Blister-actuated LIFT printing for multiparametric functionalization of paper-like biosensors. Micromachines 2019, 10, 221. [Google Scholar] [CrossRef] [Green Version]
- Aydindogan, E.; Ceylan, A.E.; Timur, S. Paper-based colorimetric spot test utilizing smartphone sensing for detection of biomarkers. Talanta 2020, 208, 120446. [Google Scholar] [CrossRef]
- Jarujamrus, P.; Meelapsom, R.; Naksen, P.; Ditcharoen, N.; Anutrasakda, W.; Siripinyanond, A.; Amatatongchai, M.; Supasorn, S. Screen-printed microfluidic paper-based analytical device (μPAD) as a barcode sensor for magnesium detection using rubber latex waste as a novel hydrophobic reagent. Anal. Chim. Acta 2019, 1082, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, M.; Wheeler, A.R.; Paull, B.; Macka, M. Ion-Exchange Based Immobilization of Chromogenic Reagents on Microfluidic Paper Analytical Devices. Anal. Chem. 2019, 91, 8756–8761. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.H.; Wu, J.J.; Lee, G.B. Screening of highly-specific aptamers and their applications in paper-based microfluidic chips for rapid diagnosis of multiple bacteria. Sens. Actuators B Chem. 2019, 284, 395–402. [Google Scholar] [CrossRef]
- Batool, R.; Rhouati, A.; Nawaz, M.H.; Hayat, A.; Marty, J.L. A Review of the Construction of Nano-Hybrids for Electrochemical Biosensing of Glucose. Biosensors 2019, 9, 46. [Google Scholar] [CrossRef] [Green Version]
- Soum, V.; Park, S.; Brilian, A.I.; Kwon, O.S.; Shin, K. Programmable paper-based microfluidic devices for biomarker detections. Micromachines 2019, 10, 516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaeger, J.; Groher, F.; Stamm, J.; Spiehl, D.; Braun, J.; Dörsam, E.; Suess, B. Characterization and Inkjet Printing of an RNA Aptamer for Paper-Based Biosensing of Ciprofloxacin. Biosensors 2019, 9, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sousa, L.R.; Duarte, L.C.; Coltro, W.K. Instrument-free fabrication of microfluidic paper-based analytical devices through 3D pen drawing. Sens. Actuators B Chem. 2020, 312, 128018. [Google Scholar] [CrossRef]
- Noviana, E.; McCord, C.P.; Clark, K.M.; Jang, I.; Henry, C.S. Electrochemical paper-based devices: Sensing approaches and progress toward practical applications. Lab Chip 2020, 20, 9–34. [Google Scholar] [CrossRef]
- Antiochia, R. Paper-Based Biosensors: Frontiers in Point-of-Care Detection of COVID-19 Disease. Biosensors 2021, 11, 110. [Google Scholar] [CrossRef]
- Katelakha, K.; Nopponpunth, V.; Boonlue, W.; Laiwattanapaisal, W. A Simple Distance Paper-Based Analytical Device for the Screening of Lead in Food Matrices. Biosensors 2021, 11, 90. [Google Scholar] [CrossRef]
- Fu, L.M.; Wang, Y.N. Detection methods and applications of microfluidic paper-based analytical devices. TrAC Trends Anal. Chem. 2018, 107, 196–211. [Google Scholar] [CrossRef]
- Rossini, E.L.; Milani, M.I.; Pezza, H.R. Green synthesis of fluorescent carbon dots for determination of glucose in biofluids using a paper platform. Talanta 2019, 201, 503–510. [Google Scholar] [CrossRef]
- Yang, J.J.; Oh, H.B.; Hwang, S.H. Paper-based speedy separation of amplified DNA (PASS-DNA): Potential for molecular point-of-care testing. Sens. Actuators B Chem. 2019, 286, 101–103. [Google Scholar] [CrossRef]
- Kung, C.T.; Hou, C.Y.; Wang, Y.N.; Fu, L.M. Microfluidic paper-based analytical devices for environmental analysis of soil, air, ecology and river water. Sens. Actuators B Chem. 2019, 301, 126855. [Google Scholar] [CrossRef]
- Fu, L.M.; Liu, C.C.; Yang, C.E.; Wang, Y.N.; Ko, C.H. A PET/paper chip platform for high resolution sulphur dioxide detection in foods. Food Chem. 2019, 286, 316–321. [Google Scholar] [CrossRef]
- Hou, C.Y.; Fu, L.M.; Ju, W.J.; Wu, P.Y. Microfluidic colorimetric system for nitrite detection in foods. Chem. Eng. J. 2020, 398, 125573. [Google Scholar] [CrossRef]
- Ng, J.S.; Hashimoto, M. 3D-PAD: Paper-Based Analytical Devices with Integrated Three-Dimensional Features. Biosensors 2021, 11, 84. [Google Scholar] [CrossRef]
- Tseng, C.C.; Ko, C.H.; Lu, S.Y.; Yang, C.E.; Fu, L.M.; Li, C.Y. Rapid electrochemical-biosensor microchip platform for determination of microalbuminuria in CKD patients. Anal. Chim. Acta 2021, 1146, 70–76. [Google Scholar] [CrossRef] [PubMed]
- LIANG, S.J.; Ji-Kai, M.; Chen, G.; Dong-Dong, Y.; Jian-Guang, Z. Research and application progress of paper-based microfluidic sample preconcentration. Chin. J. Anal. Chem. 2019, 47, 1878–1886. [Google Scholar] [CrossRef]
- Tang, R.H.; Liu, L.N.; Zhang, S.F.; He, X.C.; Li, X.J.; Xu, F.; Ni, Y.H.; Li, F. A review on advances in methods for modification of paper supports for use in point-of-care testing. Microchim. Acta 2019, 186, 521. [Google Scholar] [CrossRef] [PubMed]
- Tang, R.H.; Yang, H.; Choi, J.R.; Gong, Y.; Feng, S.S.; Pingguan-Murphy, B.; Huang, Q.S.; Shi, J.L.; Mei, Q.B.; Xu, F. Advances in paper-based sample pretreatment for point-of-care testing. Crit. Rev. Biotechnol. 2017, 37, 411–428. [Google Scholar] [CrossRef]
- Frey, B.S.; Damon, D.E.; Badu-Tawiah, A.K. Emerging trends in paper spray mass spectrometry: Microsampling, storage, direct analysis, and applications. Mass Spectrom. Rev. 2020, 39, 336–370. [Google Scholar] [CrossRef]
- Trinh, K.T.L.; Trinh, T.N.D.; Lee, N.Y. Fully integrated and slidable paper-embedded plastic microdevice for point-of-care testing of multiple foodborne pathogens. Biosens. Bioelectron 2019, 135, 120–128. [Google Scholar] [CrossRef]
- Srisiri, K.; Jaroenwattana, R.; Panvisavas, N.; Bandhaya, A. Optimisation of DNA recovery and analysis of urine samples stored on FTA® card. Forensic Sci. Int. Genet. Suppl. Ser. 2017, 6, e520–e522. [Google Scholar] [CrossRef] [Green Version]
- Karthikeyan, K.; Saranya, R.; Bharath, R.; Vidya, R.; Itami, T.; Sudhakaran, R. A simple filter paper-based method for transporting and storing Enterocytozoon hepatopenaei DNA from infected Litopenaeus vannamei tissues. J. Invertebr. Pathol. 2020, 169, 107305. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Shen, R.; Li, T.; Xiong, C.; Li, G.; Ling, L. The peroxidase-mimicking function of acetate and its application in single-enzyme-based glucose test paper. Talanta 2019, 196, 493–497. [Google Scholar] [CrossRef]
- Yetisen, A.K.; Jiang, N.; Tamayol, A.; Ruiz-Esparza, G.U.; Zhang, Y.S.; Medina-Pando, S.; Gupta, A.; Wolffsohn, J.S.; Butt, H.; Khademhosseini, A. Paper-based microfluidic system for tear electrolyte analysis. Lab Chip 2017, 17, 1137–1148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Auray-Blais, C.; Collerette-Tremblay, J.; Lavoie, P. UPLC–MS/MS analysis of keratan sulfate from urine samples collected on filter paper for monitoring & follow-up of Morquio A patients. Bioanalysis 2018, 10, 1181–1192. [Google Scholar]
- Das, D.; Dsouza, A.; Kaur, N.; Soni, S.; Toley, B.J. Paper Stacks for Uniform Rehydration of Dried Reagents in Paper Microfluidic Devices. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zangmo, T.; Siripinyanond, A. Exploring the applicability of nano-selenium for capture of mercury vapor: Paper based sorbent and a chemical modifier in graphite furnace atomic absorption spectrometry. Anal. Chim. Acta 2019, 1085, 29–38. [Google Scholar] [CrossRef]
- Shay, T.; Saha, T.; Dickey, M.D.; Velev, O.D. Principles of long-term fluids handling in paper-based wearables with capillary–evaporative transport. Biomicrofluidics 2020, 14, 034112. [Google Scholar] [CrossRef] [PubMed]
- Kolluri, N.; Albarran, N.; Fan, A.; Olson, A.; Sagar, M.; Young, A.; Gomez-Marquez, J.; Klapperich, C.M. SNAPflex: A paper-and-plastic device for instrument-free RNA and DNA extraction from whole blood. Lab Chip 2020, 20, 3386–3398. [Google Scholar] [CrossRef]
- Laurenciano, C.J.D.; Tseng, C.C.; Chen, S.J.; Lu, S.Y.; Tayo, L.L.; Fu, L.M. Microfluidic colorimetric detection platform with sliding hybrid PMMA/paper microchip for human urine and blood sample analysis. Talanta 2021, 231, 122362. [Google Scholar] [CrossRef]
- Tenda, K.; van Gerven, B.; Arts, R.; Hiruta, Y.; Merkx, M.; Citterio, D. Paper-based antibody detection devices using bioluminescent BRET-switching sensor proteins. Angew. Chem. Int. Ed. 2018, 57, 15369–15373. [Google Scholar] [CrossRef] [Green Version]
- Caratelli, V.; Ciampaglia, A.; Guiducci, J.; Sancesario, G.; Moscone, D.; Arduini, F. Precision medicine in Alzheimer’s disease: An origami paper-based electrochemical device for cholinesterase inhibitors. Biosens. Bioelectron 2020, 165, 112411. [Google Scholar] [CrossRef]
- Noviana, E.; Carrão, D.B.; Pratiwi, R.; Henry, C.S. Emerging applications of paper-based analytical devices for drug analysis: A review. Anal. Chim. Acta 2020, 1116, 70–90. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Hahn, Y.K.; Kim, M.S. Recent Advances of Fluid Manipulation Technologies in Microfluidic Paper-Based Analytical Devices (μPADs) toward Multi-Step Assays. Micromachines 2020, 11, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, T.; Bi, Y.; Xu, X.; Zhu, Z.; Yang, C. Integrated paper-based microfluidic devices for point-of-care testing. Anal. Methods 2018, 10, 3567–3581. [Google Scholar] [CrossRef]
- Guan, Y.; Zhang, K.; Xu, F.; Guo, R.; Fang, A.; Sun, B.; Meng, X.; Liu, Y.; Bai, M. An integrated platform for fibrinogen quantification on a microfluidic paper-based analytical device. Lab Chip 2020, 20, 2724–2734. [Google Scholar] [CrossRef]
- Salentijn, G.I.; Grajewski, M.; Verpoorte, E. Countercurrent liquid–liquid extraction on paper. Lab Chip 2017, 17, 3401–3404. [Google Scholar] [CrossRef] [PubMed]
- Dong, T.; Wang, G.A.; Li, F. Shaping up field-deployable nucleic acid testing using microfluidic paper-based analytical devices. Anal. Bioanal. Chem. 2019, 411, 4401–4414. [Google Scholar] [CrossRef]
- Tang, R.; Yang, H.; Choi, J.R.; Gong, Y.; Hu, J.; Wen, T.; Li, X.; Xu, B.; Mei, Q.; Xu, F. Paper-based device with on-chip reagent storage for rapid extraction of DNA from biological samples. Microchim. Acta 2017, 184, 2141–2150. [Google Scholar] [CrossRef]
- Batule, B.S.; Seok, Y.; Kim, M.G. An innovative paper-based device for DNA extraction from processed meat products. Food Chem. 2020, 321, 126708. [Google Scholar] [CrossRef] [PubMed]
- Seok, Y.; Batule, B.S.; Kim, M.G. Lab-on-paper for all-in-one molecular diagnostics (LAMDA) of zika, dengue, and chikungunya virus from human serum. Biosens. Bioelectron 2020, 165, 112400. [Google Scholar] [CrossRef]
- Batule, B.S.; Seok, Y.; Kim, M.G. Paper-based nucleic acid testing system for simple and early diagnosis of mosquito-borne RNA viruses from human serum. Biosens. Bioelectron 2020, 151, 111998. [Google Scholar] [CrossRef] [PubMed]
- Alahmad, W.; Varanusupakul, P.; Kaneta, T.; Varanusupakul, P. Chromium speciation using paper-based analytical devices by direct determination and with electromembrane microextraction. Anal. Chim. Acta 2019, 1085, 98–106. [Google Scholar] [CrossRef]
- Lin, S.; Lin, X.; Han, S.; Liu, Y.; Hasi, W.; Wang, L. Flexible fabrication of a paper-fluidic SERS sensor coated with a monolayer of core–shell nanospheres for reliable quantitative SERS measurements. Anal. Chim. Acta 2020, 1108, 167–176. [Google Scholar] [CrossRef]
- Hedeshi, M.H.; Rezvani, O.; Bagheri, H. Silane–based modified papers and their extractive phase roles in a microfluidic platform. Anal. Chim. Acta 2020, 1128, 31–41. [Google Scholar] [CrossRef]
- Guan, Y.; Sun, B. Detection and extraction of heavy metal ions using paper-based analytical devices fabricated via atom stamp printing. Microsyst. Nanoeng. 2020, 6, 14. [Google Scholar] [CrossRef]
- Jin, L.; Hao, Z.; Zheng, Q.; Chen, H.; Zhu, L.; Wang, C.; Liu, X.; Lu, C. A facile microfluidic paper-based analytical device for acetylcholinesterase inhibition assay utilizing organic solvent extraction in rapid detection of pesticide residues in food. Anal. Chim. Acta 2020, 1100, 215–224. [Google Scholar] [CrossRef]
- Liu, L.; Xie, M.-R.; Chen, Y.Z.; Wu, Z.Y. Simultaneous electrokinetic stacking and separation of anionic and cationic species on a paper fluidic channel. Lab Chip 2019, 19, 845–850. [Google Scholar] [CrossRef]
- Perera, A.T.K.; Pudasaini, S.; Ahmed, S.S.U.; Phan, D.T.; Liu, Y.; Yang, C. Rapid pre-concentration of Escherichia coli in a microfluidic paper-based device using ion concentration polarization. Electrophoresis 2020, 41, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Schaumburg, F.; Carrell, C.S.; Henry, C.S. Rapid bacteria detection at low concentrations using sequential immunomagnetic separation and paper-based isotachophoresis. Anal. Chem. 2019, 91, 9623–9630. [Google Scholar] [CrossRef]
- Guo, S.; Xu, J.; Estell, A.P.; Ivory, C.F.; Du, D.; Lin, Y.; Dong, W.J. Paper-based ITP technology: An application to specific cancer-derived exosome detection and analysis. Biosens. Bioelectron. 2020, 164, 112292. [Google Scholar] [CrossRef]
- Kim, H.; Lee, K.H.; Han, S.I.; Lee, D.; Chung, S.; Lee, D.; Lee, J.H. Origami-paper-based device for microvesicle/exosome preconcentration and isolation. Lab chip 2019, 19, 3917–3921. [Google Scholar] [CrossRef] [PubMed]
- Xi, L.; De, R.; Dutta, D. Electrokinetic stacking of particle zones in confined channels enabling their UV absorbance detection on microchips. Anal. Chim. Acta 2020, 1135, 83–90. [Google Scholar] [CrossRef]
- Costa-Rama, E.; Nouws, H.; Delerue-Matos, C.; Blanco-López, M.d.C.; Fernández-Abedul, M.T. Preconcentration and sensitive determination of the anti-inflammatory drug diclofenac on a paper-based electroanalytical platform. Anal. Chim. Acta 2019, 1074, 89–97. [Google Scholar] [CrossRef]
- Hiltunen, J.; Liedert, C.; Hiltunen, M.; Huttunen, O.H.; Hiitola-Keinänen, J.; Aikio, S.; Harjanne, M.; Kurkinen, M.; Hakalahti, L.; Lee, L.P. Roll-to-roll fabrication of integrated PDMS–paper microfluidics for nucleic acid amplification. Lab Chip 2018, 18, 1552–1559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pang, B.; Fu, K.; Liu, Y.; Ding, X.; Hu, J.; Wu, W.; Xu, K.; Song, X.; Wang, J.; Mu, Y. Development of a self-priming PDMS/paper hybrid microfluidic chip using mixed-dye-loaded loop-mediated isothermal amplification assay for multiplex foodborne pathogens detection. Anal. Chim. Acta 2018, 1040, 81–89. [Google Scholar] [CrossRef]
- Naik, P.; Jaitpal, S.; Shetty, P.; Paul, D. An integrated one-step assay combining thermal lysis and loop-mediated isothermal DNA amplification (LAMP) in 30 min from E. coli and M. smegmatis cells on a paper substrate. Sens. Actuators B Chem. 2019, 291, 74–80. [Google Scholar] [CrossRef]
- Chen, P.; Chen, C.; Liu, Y.; Du, W.; Feng, X.; Liu, B.F. Fully integrated nucleic acid pretreatment, amplification, and detection on a paper chip for identifying EGFR mutations in lung cancer cells. Sens. Actuators B Chem. 2019, 283, 472–477. [Google Scholar] [CrossRef]
- He, T.; Li, J.; Liu, L.; Ge, S.; Yan, M.; Liu, H.; Yu, J. Origami-based “Book” shaped three-dimensional electrochemical paper microdevice for sample-to-answer detection of pathogens. RSC Adv. 2020, 10, 25808–25816. [Google Scholar] [CrossRef]
- Luo, F.; Li, Z.; Dai, G.; Lu, Y.; He, P.; Wang, Q. Ultrasensitive biosensing pathogenic bacteria by combining aptamer-induced catalysed hairpin assembly circle amplification with microchip electrophoresis. Sens. Actuators B Chem. 2020, 306, 127577. [Google Scholar] [CrossRef]
- Zhang, D.; Wu, C.; Luan, C.; Gao, P.; Wang, H.; Chi, J.; Kong, T. Distance-based quantification of miRNA-21 by the coffee-ring effect using paper devices. Microchim. Acta 2020, 187, 513. [Google Scholar] [CrossRef]
- Yang, H.; Hu, M.; Li, Z.; Zhao, P.; Xie, L.; Song, X.; Yu, J. Donor/acceptor-induced ratiometric photoelectrochemical paper analytical device with a hollow double-hydrophilic-walls channel for microRNA quantification. Anal. Chem. 2019, 91, 14577–14585. [Google Scholar] [CrossRef]
- Cai, Y.; Niu, J.C.; Liu, Y.Q.; Du, X.L.; Wu, Z.Y. Online sample clean-up and enrichment of proteins from salty media with dynamic double gradients on a paper fluidic channel. Anal. Chim. Acta 2020, 1100, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Lepowsky, E.; Ghaderinezhad, F.; Knowlton, S.; Tasoglu, S. Paper-based assays for urine analysis. Biomicrofluidics 2017, 11, 051501. [Google Scholar] [CrossRef] [PubMed]
- Worramongkona, P.; Seeda, K.; Phansomboon, P.; Ratnarathorn, N.; Chailapakul, O.; Dungchai, W. A simple paper-based colorimetric device for rapid and sensitive urinary oxalate determinations. Anal. Sci. 2018, 34, 103–108. [Google Scholar] [CrossRef] [Green Version]
- Fava, E.L.; Silva, T.A.; do Prado, T.M.; de Moraes, F.C.; Faria, R.C.; Fatibello-Filho, O. Electrochemical paper-based microfluidic device for high throughput multiplexed analysis. Talanta 2019, 203, 280–286. [Google Scholar] [CrossRef]
- Roychoudhury, A.; Francis, K.A.; Patel, J.; Jha, S.K.; Basu, S. A decoupler-free simple paper microchip capillary electrophoresis device for simultaneous detection of dopamine, epinephrine and serotonin. RSC Adv. 2020, 10, 25487–25495. [Google Scholar] [CrossRef]
- Mohammadifar, M.; Tahernia, M.; Choi, S. An equipment-free, paper-based electrochemical sensor for visual monitoring of glucose levels in urine. SLAS Technol. 2019, 24, 499–505. [Google Scholar] [CrossRef]
- Ortiz-Gómez, I.; Salinas-Castillo, A.; García, A.G.; Álvarez-Bermejo, J.A.; de Orbe-Payá, I.; Rodríguez-Diéguez, A.; Capitán-Vallvey, L.F. Microfluidic paper-based device for colorimetric determination of glucose based on a metal-organic framework acting as peroxidase mimetic. Microchim. Acta 2018, 185, 47. [Google Scholar] [CrossRef]
- Shitanda, I.; Fujimura, Y.; Nohara, S.; Hoshi, Y.; Itagaki, M.; Tsujimura, S. Paper-based disk-type self-powered glucose biosensor based on screen-printed biofuel cell array. J. Electrochem. Soc. 2019, 166, B1063. [Google Scholar] [CrossRef]
- Li, Z.; Li, F.; Hu, J.; Wee, W.H.; Han, Y.L.; Pingguan-Murphy, B.; Lu, T.J.; Xu, F. Direct writing electrodes using a ball pen for paper-based point-of-care testing. Analyst 2015, 140, 5526–5535. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, C.; Xing, D. Paper-based bipolar electrode-electrochemiluminescence (BPE-ECL) device with battery energy supply and smartphone read-out: A handheld ECL system for biochemical analysis at the point-of-care level. Sens. Actuators B Chem. 2016, 237, 308–317. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, C. A novel screen-printed microfluidic paper-based electrochemical device for detection of glucose and uric acid in urine. Biomed. Microdevices 2016, 18, 92. [Google Scholar] [CrossRef] [PubMed]
- Fava, E.L.; Martimiano do Prado, T.; Almeida Silva, T.; Cruz de Moraes, F.; Censi Faria, R.; Fatibello-Filho, O. New Disposable Electrochemical Paper-based Microfluidic Device with Multiplexed Electrodes for Biomarkers Determination in Urine Sample. Electroanalysis 2020, 32, 1075–1083. [Google Scholar] [CrossRef]
- Vishnu, N.; Sahatiya, P.; Kong, C.Y.; Badhulika, S. Large area, one step synthesis of NiSe2 films on cellulose paper for glucose monitoring in bio-mimicking samples for clinical diagnostics. Nanotechnology 2019, 30, 355502. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.; Kim, I.; Roh, S.; Jung, H.G.; Lee, S.W.; Kim, H.S.; Yoon, D.S.; Hong, Y.; Lee, G. Selective colorimetric urine glucose detection by paper sensor functionalized with polyaniline nanoparticles and cell membrane. Anal. Chim. Acta 2021, 1158, 338387. [Google Scholar] [CrossRef]
- Lee, W.; Gonzalez, A.; Arguelles, P.; Guevara, R.; Gonzalez-Guerrero, M.J.; Gomez, F.A. Thread/paper-and paper-based microfluidic devices for glucose assays employing artificial neural networks. Electrophoresis 2018, 39, 1443–1451. [Google Scholar] [CrossRef]
- Zhang, H.; Smith, E.; Zhang, W.; Zhou, A. Inkjet printed microfluidic paper-based analytical device (μPAD) for glucose colorimetric detection in artificial urine. Biomed. Microdevices 2019, 21, 48. [Google Scholar] [CrossRef]
- Yu, P.; Deng, M.; Yang, Y. New single-layered paper-based microfluidic devices for the analysis of nitrite and glucose built via deposition of adhesive tape. Sensors 2019, 19, 4082. [Google Scholar] [CrossRef] [Green Version]
- Galanis, P.; He, P.; Katis, I.; Iles, A.; Kumar, A.J.; Eason, R.; Sones, C. Local photo-polymer deposition-assisted fabrication of multilayer paper-based devices. Sens. Actuators B Chem. 2020, 322, 128574. [Google Scholar] [CrossRef]
- De Oliveira, R.A.; Camargo, F.; Pesquero, N.C.; Faria, R.C. A simple method to produce 2D and 3D microfluidic paper-based analytical devices for clinical analysis. Anal. Chim. Acta 2017, 957, 40–46. [Google Scholar] [CrossRef] [Green Version]
- Neris, N.M.; Guevara, R.D.; Gonzalez, A.; Gomez, F.A. 3D Multilayered paper-and thread/paper-based microfluidic devices for bioassays. Electrophoresis 2019, 40, 296–303. [Google Scholar] [CrossRef]
- Hu, R.; Gupta, R.; Wang, Z.; Wang, C.; Sun, H.; Singamaneni, S.; Kharasch, E.D.; Morrissey, J.J. Bioplasmonic paper–based assay for perilipin-2 non-invasively detects renal cancer. Kidney Int. 2019, 96, 1417–1421. [Google Scholar] [CrossRef]
- Sittiwong, J.; Unob, F. Paper-based Platform for Urinary Creatinine Detection. Anal. Sci. 2016, 32, 639–643. [Google Scholar] [CrossRef] [Green Version]
- Sununta, S.; Rattanarat, P.; Chailapakul, O.; Praphairaksit, N. Microfluidic paper-based analytical devices for determination of creatinine in urine samples. Anal. Sci. 2018, 34, 109–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tarik, M.; Ramakrishnan, L.; Amarchand, R.; Salve, H.R.; Mathur, P.; Joshi, P.; Krishnan, A. Feasibility of measuring sodium, potassium and creatinine from urine sample dried on filter paper. Bioanalysis 2019, 11, 689–701. [Google Scholar] [CrossRef]
- Mathaweesansurn, A.; Thongrod, S.; Khongkaew, P.; Phechkrajang, C.M.; Wilairat, P.; Choengchan, N. Simple and fast fabrication of microfluidic paper-based analytical device by contact stamping for multiple-point standard addition assay: Application to direct analysis of urinary creatinine. Talanta 2020, 210, 120675. [Google Scholar] [CrossRef] [PubMed]
- Chaiyo, S.; Kalcher, K.; Apilux, A.; Chailapakul, O.; Siangproh, W. A novel paper-based colorimetry device for the determination of the albumin to creatinine ratio. Analyst 2018, 143, 5453–5460. [Google Scholar] [CrossRef]
- Hiraoka, R.; Kuwahara, K.; Wen, Y.C.; Yen, T.H.; Hiruta, Y.; Cheng, C.M.; Citterio, D. Paper-based Device for Naked Eye Urinary Albumin/Creatinine Ratio Evaluation. ACS Sen. 2020, 5, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Shojaeifard, Z.; Bordbar, M.M.; Aseman, M.D.; Nabavizadeh, S.M.; Hemmateenejad, B. Collaboration of cyclometalated platinum complexes and metallic nanoclusters for rapid discrimination and detection of biogenic amines through a fluorometric paper-based sensor array. Sens. Actuators B Chem. 2021, 334, 129582. [Google Scholar] [CrossRef]
- Cai, Y.; Niu, J.C.; Du, X.L.; Fang, F.; Wu, Z.Y. Novel field amplification for sensitive colorimetric detection of microalbuminuria on a paper-based analytical device. Anal. Chim. Acta 2019, 1080, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Imamura, A.H.; Segato, T.P.; de Oliveira, L.J.M.; Hassan, A.; Crespilho, F.N.; Carrilho, E. Monitoring cellulose oxidation for protein immobilization in paper-based low-cost biosensors. Microchim. Acta 2020, 187, 272. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, M.O.; Chang, L.Y.; Chen, C.H.; Wu, T.F.; Chuang, C.H. Paper-based immunosensor utilizing dielectrophoretic trapping of microprobes for quantitative and label free detection using electrochemical impedance spectroscopy. Biomicrofluidics 2018, 12, 064102. [Google Scholar] [CrossRef]
- Gao, H.; Liu, J.J.; Liu, Y.Q.; Wu, Z.Y. Detection of urine protein by a paper-based analytical device enhanced with ion concentration polarization effect. Microfluid. Nanofluid 2019, 23, 51. [Google Scholar] [CrossRef]
- Huang, X.; Shi, W.; Li, J.; Bao, N.; Yu, C.; Gu, H. Determination of salivary uric acid by using poly(3,4-ethylenedioxythipohene) and graphene oxide in a disposable paper-based analytical device. Anal. Chim. Acta 2020, 1103, 75–83. [Google Scholar] [CrossRef]
- Rossini, E.L.; Milani, M.I.; Carrilho, E.; Pezza, L.; Pezza, H.R. Simultaneous determination of renal function biomarkers in urine using a validated paper-based microfluidic analytical device. Anal. Chim. Acta 2018, 997, 16–23. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.; Ahmed, I.; Anik, M.I.; Ferdous, M.; Khan, M.S. Developing paper based diagnostic technique to detect uric acid in urine. Front. Chem. 2018, 6, 496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, M.; Khalid, M.A.U.; Shah, I.; Kim, S.W.; Kim, Y.S.; Lim, J.H.; Choi, K.H. Paper-based selective and quantitative detection of uric acid using citrate-capped Pt nanoparticles (PtNPs) as a colorimetric sensing probe through a simple and remote-based device. N. J. Chem. 2019, 43, 7636–7645. [Google Scholar] [CrossRef]
- Villa, J.E.; Poppi, R.J. A portable SERS method for the determination of uric acid using a paper-based substrate and multivariate curve resolution. Analyst 2016, 141, 1966–1972. [Google Scholar] [CrossRef] [Green Version]
- Nontawong, N.; Amatatongchai, M.; Wuepchaiyaphum, W.; Chairam, S.; Pimmongkol, S.; Panich, S.; Tamuang, S.; Jarujamrus, P. Fabrication of a three-dimensional electrochemical paper-based device (3D-ePAD) for individual and simultaneous detection of ascorbic acid, dopamine and uric acid. Int. J. Electrochem. Sci. 2018, 13, 6940–6957. [Google Scholar] [CrossRef]
- Cincotto, F.H.; Fava, E.L.; Moraes, F.C.; Fatibello-Filho, O.; Faria, R.C. A new disposable microfluidic electrochemical paper-based device for the simultaneous determination of clinical biomarkers. Talanta 2019, 195, 62–68. [Google Scholar] [CrossRef]
- Manbohi, A.; Ahmadi, S.H. Chitosan–Fe 3 O 4 nanoparticle enzymatic electrodes on paper as an efficient assay for glucose and uric acid detection in biological fluids. Chem. Pap. 2020, 74, 2675–2687. [Google Scholar] [CrossRef]
- Shih, C.M.; Chang, C.L.; Hsu, M.Y.; Lin, J.Y.; Kuan, C.M.; Wang, H.K.; Huang, C.T.; Chung, M.C.; Huang, K.C.; Hsu, C.E. Paper-based ELISA to rapidly detect Escherichia coli. Talanta 2015, 145, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Gumustas, A.; Caglayan, M.G.; Eryilmaz, M.; Suludere, Z.; Soykut, E.A.; Uslu, B.; Boyaci, I.H.; Tamer, U. Paper based lateral flow immunoassay for the enumeration of Escherichia coli in urine. Anal. Methods 2018, 10, 1213–1218. [Google Scholar] [CrossRef]
- Ilhan, H.; Guven, B.; Dogan, U.; Torul, H.; Evran, S.; Çetin, D.; Suludere, Z.; Saglam, N.; Boyaci, İ.H.; Tamer, U. The coupling of immunomagnetic enrichment of bacteria with paper-based platform. Talanta 2019, 201, 245–252. [Google Scholar] [CrossRef]
- Noiphung, J.; Laiwattanapaisal, W. Multifunctional paper-based analytical device for in situ cultivation and screening of Escherichia coli infections. Sci. Rep. 2019, 9, 1–10. [Google Scholar]
- Calabretta, M.M.; Álvarez-Diduk, R.; Michelini, E.; Roda, A.; Merkoçi, A. Nano-lantern on paper for smartphone-based ATP detection. Biosens. Bioelectron 2020, 150, 111902. [Google Scholar] [CrossRef] [PubMed]
- He, P.J.; Katis, I.N.; Kumar, A.J.; Bryant, C.A.; Keevil, C.W.; Somani, B.K.; Mahobia, N.; Eason, R.W.; Sones, C.L. Laser-patterned paper-based sensors for rapid point-of-care detection and antibiotic-resistance testing of bacterial infections. Biosens. Bioelectron 2020, 152, 112008. [Google Scholar] [CrossRef]
- Villa, J.E.; Pasquini, C.; Poppi, R.J. Surface-enhanced Raman spectroscopy and MCR-ALS for the selective sensing of urinary adenosine on filter paper. Talanta 2018, 187, 99–105. [Google Scholar] [CrossRef]
- Zhang, H.; Lei, Z.; Tian, R.; Wang, Z. Polyamidoamine starburst dendrimer-activated chromatography paper-based assay for sensitive detection of telomerase activity. Talanta 2018, 178, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Mao, G.; Zhong, Y.; Wu, G.; Wu, W.; Zhan, Y.; He, Z.; Huang, W. Highly sensitive ratiometric fluorescent paper sensor for the urine assay of cancer. Talanta 2019, 194, 199–204. [Google Scholar] [CrossRef]
- Abarghoei, S.; Fakhri, N.; Borghei, Y.S.; Hosseini, M.; Ganjali, M.R. A colorimetric paper sensor for citrate as biomarker for early stage detection of prostate cancer based on peroxidase-like activity of cysteine-capped gold nanoclusters. Spectrochim. Acta A 2019, 210, 251–259. [Google Scholar] [CrossRef]
- Fakhri, N.; Abarghoei, S.; Dadmehr, M.; Hosseini, M.; Sabahi, H.; Ganjali, M.R. Paper based colorimetric detection of miRNA-21 using Ag/Pt nanoclusters. Spectrochim. Acta A 2020, 227, 117529. [Google Scholar] [CrossRef]
- Petroni, J.M.; Lucca, B.G.; da Silva Júnior, L.C.; Barbosa Alves, D.C.; Souza Ferreira, V. Paper-based Electrochemical Devices Coupled to External Graphene-Cu Nanoparticles Modified Solid Electrode through Meniscus Configuration and their Use in Biological Analysis. Electroanalysis 2017, 29, 2628–2637. [Google Scholar] [CrossRef]
- Wei, X.; Tian, T.; Jia, S.; Zhu, Z.; Ma, Y.; Sun, J.; Lin, Z.; Yang, C.J. Microfluidic distance readout sweet hydrogel integrated paper-based analytical device (μDiSH-PAD) for visual quantitative point-of-care testing. Anal. Chem. 2016, 88, 2345–2352. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, F.M.; Scheel, G.L.; Augusti, R.; Tarley, C.R.T.; Nascentes, C.C. Supramolecular microextraction combined with paper spray ionization mass spectrometry for sensitive determination of tricyclic antidepressants in urine. Anal. Chim. Acta 2020, 1106, 52–60. [Google Scholar] [CrossRef]
- Borges, M.M.; Santos, H.; Vasconcelos, G.A.; Nascimento, T.A.; Dutra, F.V.; Pires, B.C.; Allochio Filho, J.F.; Aquije, G.M.; Borges, W.S.; Lacerda, V. The use of conductive polymers as a substrate for paper spray ionization mass spectrometry. Anal. Methods 2019, 11, 3388–3400. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, J.; Deng, J.; Yuan, K.; Chen, X.; Liu, N.; Wang, X.; Luan, T. Rapid and on-site analysis of amphetamine-type illicit drugs in whole blood and raw urine by slug-flow microextraction coupled with paper spray mass spectrometry. Anal. Chim. Acta 2018, 1032, 75–82. [Google Scholar] [CrossRef]
- Ríos-Gómez, J.; García-Valverde, M.T.; López-Lorente, Á.I.; Toledo-Neira, C.; Lucena, R.; Cárdenas, S. Polymeric ionic liquid immobilized onto paper as sorptive phase in microextraction. Anal. Chim. Acta 2020, 1094, 47–56. [Google Scholar] [CrossRef]
- Narang, J.; Malhotra, N.; Singhal, C.; Mathur, A.; Chakraborty, D.; Anil, A.; Ingle, A.; Pundir, C. Point of care with micro fluidic paper based device integrated with nano zeolite–graphene oxide nanoflakes for electrochemical sensing of ketamine. Biosen. Bioelectron 2017, 88, 249–257. [Google Scholar] [CrossRef]
- Narang, J.; Singhal, C.; Mathur, A.; Dubey, A.K.; Pn, A.K.; Anil, A.; Pundir, C. Naked-eye quantitative assay on paper device for date rape drug sensing via smart phone APP. Vacuum 2018, 153, 300–305. [Google Scholar] [CrossRef]
- Rahbar, M.; Paull, B.; Macka, M. Instrument-free argentometric determination of chloride via trapezoidal distance-based microfluidic paper devices. Anal. Chim. Acta 2019, 1063, 1–8. [Google Scholar] [CrossRef]
- Wei, X.; Tian, T.; Jia, S.; Zhu, Z.; Ma, Y.; Sun, J.; Lin, Z.; Yang, C.J. Target-responsive DNA hydrogel mediated “stop-flow” microfluidic paper-based analytic device for rapid, portable and visual detection of multiple targets. Anal. Chem. 2015, 87, 4275–4282. [Google Scholar] [CrossRef] [PubMed]
- Vijitvarasan, P.; Oaew, S.; Surareungchai, W. Paper-based scanometric assay for lead ion detection using DNAzyme. Anal. Chim. Acta 2015, 896, 152–159. [Google Scholar] [CrossRef]
- Cai, Y.; You, J.; You, Z.; Dong, F.; Du, S.; Zhang, L. Profuse color-evolution-based fluorescent test paper sensor for rapid and visual monitoring of endogenous Cu2+ in human urine. Biosens. Bioelectron 2018, 99, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.H.; Chen, P.H.; Lin, C.; Chen, C.F.; Lee, I.R.; Yeh, Y.C. Determination of gold ions in human urine using genetically engineered microorganisms on a paper device. ACS Sen. 2018, 3, 744–748. [Google Scholar] [CrossRef] [PubMed]
- Myers, N.M.; Leung, I.C.; McGee, S.W.; Eggleson, K.; Lieberman, M. Green design of a paper test card for urinary iodine analysis. PLoS ONE 2017, 12, e0179716. [Google Scholar] [CrossRef] [Green Version]
- Faham, S.; Khayatian, G.; Golmohammadi, H.; Ghavami, R. A paper-based optical probe for chromium by using gold nanoparticles modified with 2, 2′-thiodiacetic acid and smartphone camera readout. Microchim. Acta 2018, 185, 374. [Google Scholar] [CrossRef]
- Li, J.; Li, P.; Bian, M.; Huo, D.; Hou, C.; Qin, H.; Zhang, S.; Zhang, L. A Fluorescent detection method for copper ions based on a direct redox route and desk study of wax-printed paper-based probes. Anal. Methods 2018, 10, 1895–1901. [Google Scholar] [CrossRef]
- Amatatongchai, M.; Sitanurak, J.; Sroysee, W.; Sodanat, S.; Chairam, S.; Jarujamrus, P.; Nacapricha, D.; Lieberzeit, P.A. Highly sensitive and selective electrochemical paper-based device using a graphite screen-printed electrode modified with molecularly imprinted polymers coated Fe3O4@ Au@ SiO2 for serotonin determination. Anal. Chim. Acta 2019, 1077, 255–265. [Google Scholar] [CrossRef]
- Orzari, L.O.; de Freitas, R.C.; de Araujo Andreotti, I.A.; Gatti, A.; Janegitz, B.C. A novel disposable self-adhesive inked paper device for electrochemical sensing of dopamine and serotonin neurotransmitters and biosensing of glucose. Biosens. Bioelectron 2019, 138, 111310. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, X.; Chunyue, M.; Ruijun, L.; Yibing, J. Fluorescent paper–based sensor based on carbon dots for detection of folic acid. Anal. Bioanal. Chem. 2020, 412, 2805–2813. [Google Scholar]
- Nghia, N.N.; Huy, B.T.; Lee, Y.I. Paper-based colorimetric probe for highly sensitive detection of folic acid based on open-ring form amplification of rhodamine B derivative. J. Ind. Eng. Chem. 2020, 81, 352–359. [Google Scholar] [CrossRef]
- Amin, R.; Ghaderinezhad, F.; Li, L.; Lepowsky, E.; Yenilmez, B.; Knowlton, S.; Tasoglu, S. Continuous-ink, multiplexed pen-plotter approach for low-cost, high-throughput fabrication of paper-based microfluidics. Anal. Chem. 2017, 89, 6351–6357. [Google Scholar] [CrossRef]
- Edachana, R.P.; Kumaresan, A.; Balasubramanian, V.; Thiagarajan, R.; Nair, B.G.; Gopalakrishnan, S.B.T. Paper-based device for the colorimetric assay of bilirubin based on in-situ formation of gold nanoparticles. Microchim. Acta 2020, 187, 60. [Google Scholar] [CrossRef]
- Meng, X.; Liu, Q.; Ding, Y. Paper-based solid-phase microextraction for analysis of 8-hydroxy-2′-deoxyguanosine in urine sample by CE-LIF. Electrophoresis 2017, 38, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Hainsworth, D.P.; Gangula, A.; Ghoshdastidar, S.; Kannan, R.; Upendran, A. Diabetic retinopathy screening using a gold nanoparticle–based paper strip assay for the at-home detection of the urinary biomarker 8-hydroxy-2′-deoxyguanosine. Am. J. Ophthalmol. 2020, 213, 306–319. [Google Scholar] [CrossRef] [PubMed]
- Messina, M.; Meli, C.; Conoci, S.; Petralia, S. A facile method for urinary phenylalanine measurement on paper-based lab-on-chip for PKU therapy monitoring. Analyst 2017, 142, 4629–4632. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Pal, A.; Chaudhury, K.; Das, S. Polyaniline Functionalized Impedimetric Paper Sensor for Urine pH Measurement. IEEE Sens. J. 2021, 21, 14474–14482. [Google Scholar] [CrossRef]
- Xiao, L.; Zhang, Z.; Wu, C.; Han, L.; Zhang, H. Molecularly imprinted polymer grafted paper-based method for the detection of 17β-estradiol. Food Chem. 2017, 221, 82–86. [Google Scholar] [CrossRef]
- Mehta, S.M.; Mehta, S.; Muthurajan, H.; D’Souza, J.S. Vertical flow paper-based plasmonic device for cysteine detection. Biomed. Microdevices 2019, 21, 55. [Google Scholar] [CrossRef]
- Malahom, N.; Jarujamrus, P.; Anutrasakda, W.; Chawengkirttikul, R.; Siripinyanond, A.; Meelapsom, R.; Amatatongchai, M. Novel paper-based colorimetric immunoassay (PCI) for sensitive and specific detection of salbutamol residues in flesh of swine and urine using Ag3PO4/Ag nanocomposite as label. J. Food Sci. 2020, 85, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Buser, J.R.; Byrnes, S.A.; Anderson, C.E.; Howell, A.J.; Kauffman, P.C.; Bishop, J.D.; Wheeler, M.H.; Kumar, S.; Yager, P. Understanding partial saturation in paper microfluidics enables alternative device architectures. Anal. Methods 2019, 11, 336–345. [Google Scholar] [CrossRef]
- Jeong, S.G.; Kim, D.-H.; Kim, J.; Kim, J.H.; Song, S.; Lee, C.S. Programmable microfluidic flow for automatic multistep digital assay in a single-sheet 3-dimensional paper-based microfluidic device. Chem. Eng. J. 2021, 411, 128429. [Google Scholar] [CrossRef]
- Geng, Z.Q.; Zheng, J.J.; Li, Y.P.; Chen, Y.; Wang, P.; Han, C.Q.; Yang, G.H.; Qu, L.L. A disposable paper-based hydrophobic substrate for highly sensitive surface-enhanced Raman scattering detection. Talanta 2020, 220, 121340. [Google Scholar] [CrossRef] [PubMed]
- Walgama, C.; Nguyen, M.P.; Boatner, L.M.; Richards, I.; Crooks, R.M. Hybrid paper and 3D-printed microfluidic device for electrochemical detection of Ag nanoparticle labels. Lab Chip 2020, 20, 1648–1657. [Google Scholar] [CrossRef]
- Khoshbin, Z.; Housaindokht, M.R.; Izadyar, M.; Verdian, A.; Bozorgmehr, M.R. A simple paper-based aptasensor for ultrasensitive detection of lead (II) ion. Anal. Chim. Acta 2019, 1071, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Wang, J.; Wang, Q.; Tang, D.; Lin, Y. Distance-dependent visual fluorescence immunoassay on CdTe quantum dot–impregnated paper through silver ion-exchange reaction. Microchim. Acta 2020, 187, 563. [Google Scholar] [CrossRef]
- Khoshbin, Z.; Housaindokht, M.R.; Verdian, A. A low-cost paper-based aptasensor for simultaneous trace-level monitoring of mercury (II) and silver (I) ions. Anal. Biochem. 2020, 597, 113689. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.M.; Zhou, T.; Fang, F.; Wu, Z.Y. Colorimetric speciation of Cr on paper-based analytical devices based on field amplified stacking. Talanta 2020, 210, 120635. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Zhang, H.; Yu, X.; Wang, W. A microfluidic paper-based laser-induced fluorescence sensor based on duplex-specific nuclease amplification for selective and sensitive detection of miRNAs in cancer cells. Talanta 2020, 216, 120996. [Google Scholar] [CrossRef]
- Li, F.; Guo, L.; Hu, Y.; Li, Z.; Liu, J.; He, J.; Cui, H. Multiplexed chemiluminescence determination of three acute myocardial infarction biomarkers based on microfluidic paper-based immunodevice dual amplified by multifunctionalized gold nanoparticles. Talanta 2020, 207, 120346. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.M.; Mazon, T. Early diagnosis of Zika infection using a ZnO nanostructures-based rapid electrochemical biosensor. Talanta 2019, 206, 153–160. [Google Scholar] [CrossRef] [PubMed]
| Human Sample | Blood | Urine | Saliva | Sweat |
|---|---|---|---|---|
| Sample type | Invasive | Non-invasive | Non-invasive | Non-invasive |
| Sampling | Difficult | Easy | Easy | Easy |
| Sample volume | Lots | Lots | Little | Little |
| Sample pretreatment | Complicated | Easy | Easy | Easy |
| Detection accuracy | High | Medium | Medium | Medium |
| Detection cost | High | Low | Low | Low |
| Detectable items | Diversity | Diversity | Limited | Limited |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tai, W.-C.; Chang, Y.-C.; Chou, D.; Fu, L.-M. Lab-on-Paper Devices for Diagnosis of Human Diseases Using Urine Samples—A Review. Biosensors 2021, 11, 260. https://doi.org/10.3390/bios11080260
Tai W-C, Chang Y-C, Chou D, Fu L-M. Lab-on-Paper Devices for Diagnosis of Human Diseases Using Urine Samples—A Review. Biosensors. 2021; 11(8):260. https://doi.org/10.3390/bios11080260
Chicago/Turabian StyleTai, Wei-Chun, Yu-Chi Chang, Dean Chou, and Lung-Ming Fu. 2021. "Lab-on-Paper Devices for Diagnosis of Human Diseases Using Urine Samples—A Review" Biosensors 11, no. 8: 260. https://doi.org/10.3390/bios11080260
APA StyleTai, W.-C., Chang, Y.-C., Chou, D., & Fu, L.-M. (2021). Lab-on-Paper Devices for Diagnosis of Human Diseases Using Urine Samples—A Review. Biosensors, 11(8), 260. https://doi.org/10.3390/bios11080260