Medical Devices for Tremor Suppression: Current Status and Future Directions
Abstract
1. Introduction
2. Early Innovations
3. Electrical Stimulation Systems
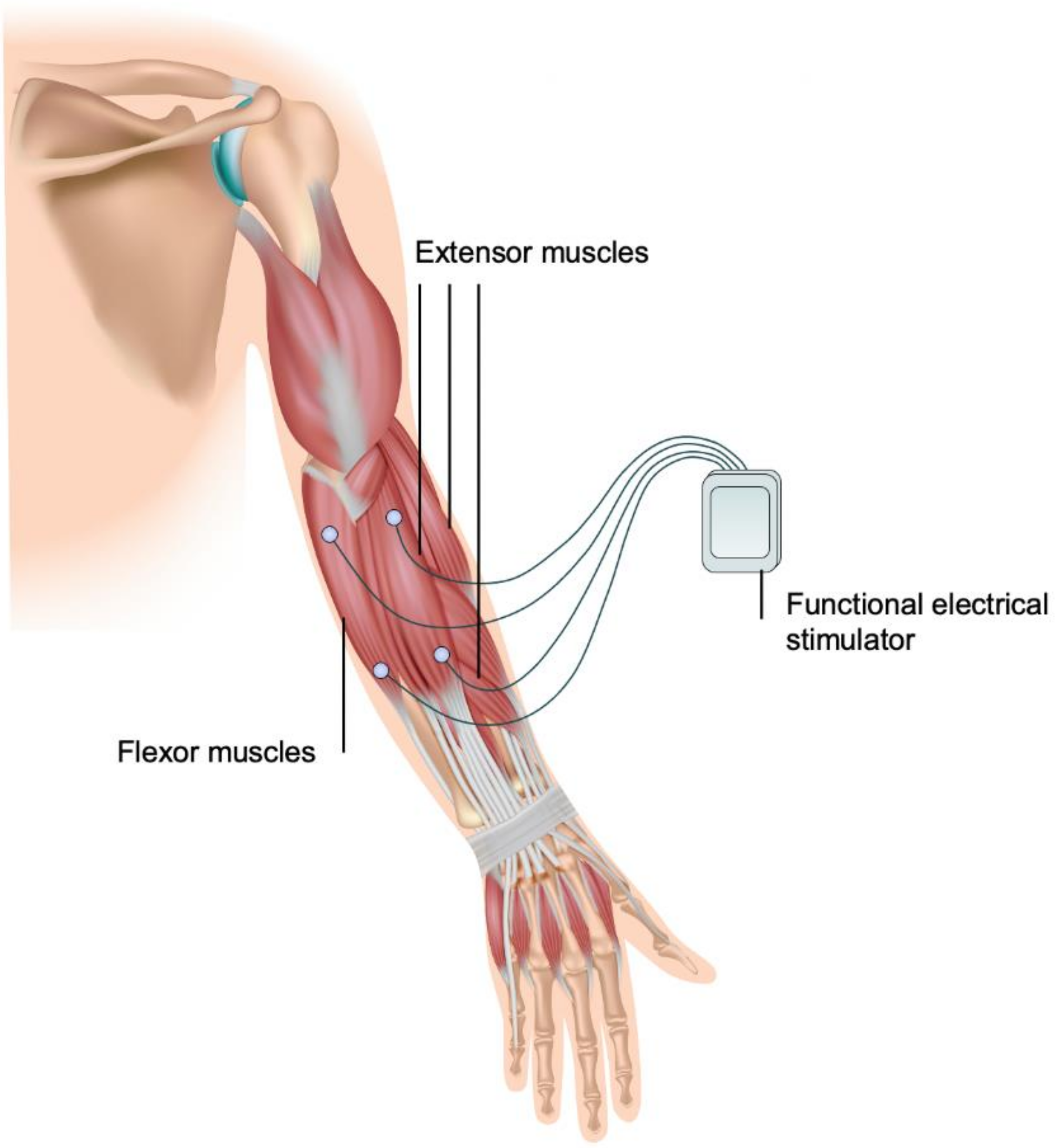
3.1. Median and Radial Nerve Excitation
3.2. Antagonistic Muscles Activation
4. Wearable Orthoses
4.1. Active Suppression
4.2. Semi-Active Suppression
4.3. Passive Suppression
4.4. Mechanism Underpinning the Efficacy of Wearable Orthoses in Tremor Suppression
4.5. Comparing Active, Semi-Active, and Passive Orthoses for Tremor Suppression
5. Assistive Feeding Devices
6. Other Devices
6.1. Gyroscopic Stabilization
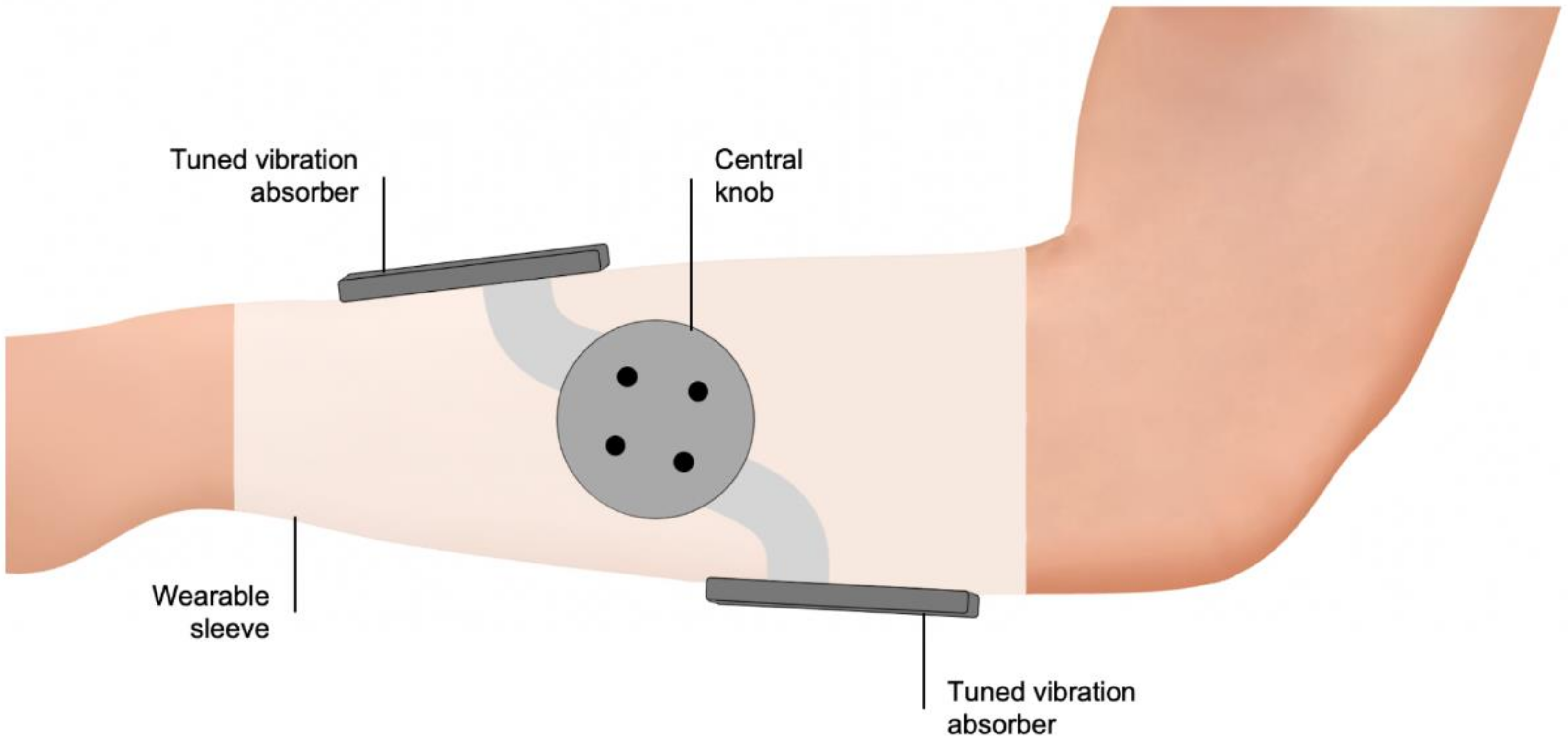
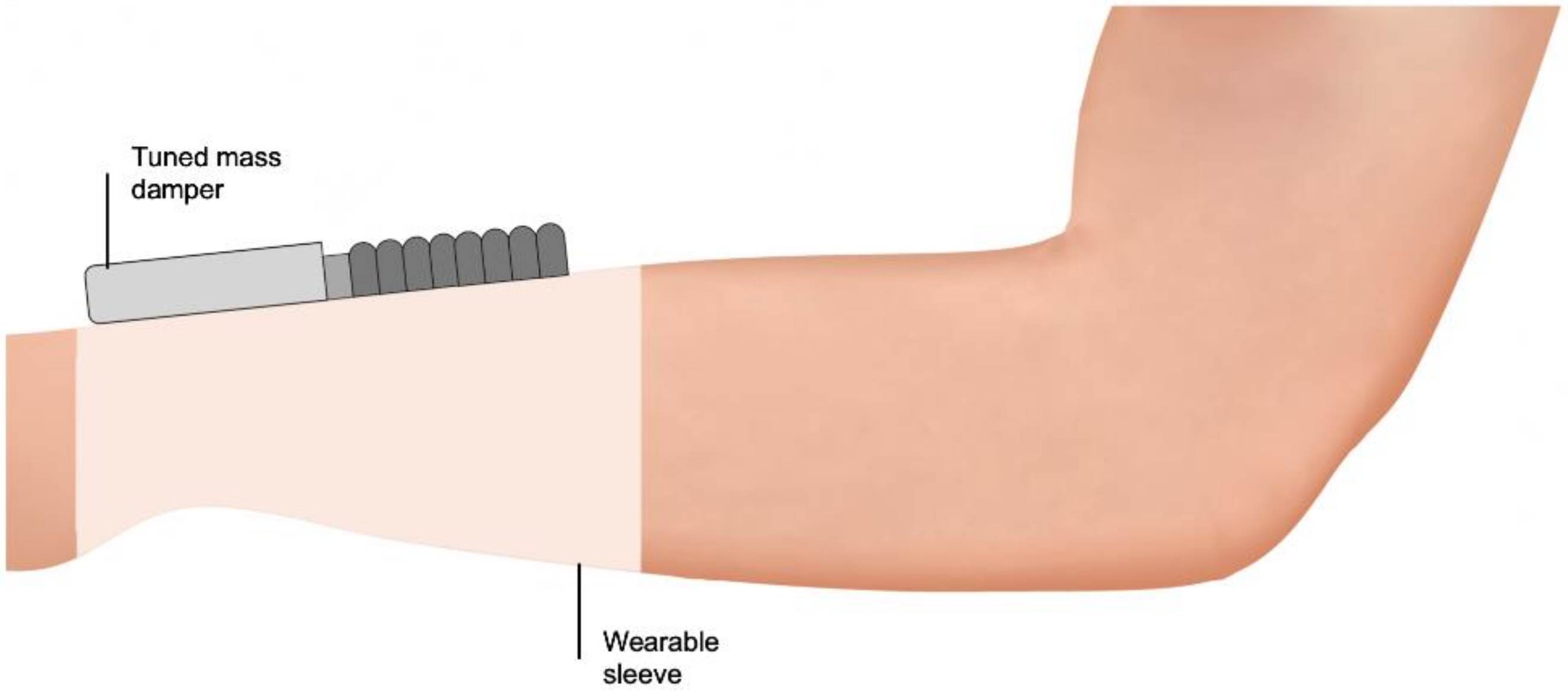
6.2. Haptic Stimulation Systems
7. Tremor Suppression Devices: Place in Therapy
8. Future Perspectives
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhatia, K.P.; Bain, P.; Bajaj, N.; Elble, R.J.; Hallett, M.; Louis, E.D.; Raethjen, J.; Stamelou, M.; Testa, C.M.; Deuschl, G.; et al. Consensus Statement on the classification of tremors. From the task force on tremor of the International Parkinson and Movement Disorder Society. Mov. Disord. 2018, 33, 75–87. [Google Scholar] [CrossRef]
- Louis, E.D.; Ferreira, J.J. How common is the most common adult movement disorder? Update on the worldwide prevalence of essential tremor. Mov. Disord. 2010, 25, 534–541. [Google Scholar] [CrossRef]
- Louis, E.D.; Ottman, R.; Hauser, W.A. How common is the most common adult movement disorder? Estimates of the prevalence of essential tremor throughout the world. Mov. Disord. 1998, 13, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Dorsey, E.R.; Elbaz, A.; Nichols, E.; Abd-Allah, F.; Abdelalim, A.; Adsuar, J.C.; Ansha, M.G.; Brayne, C.; Choi, J.-Y.J.; Collado-Mateo, D.; et al. Global, regional, and national burden of Parkinson’s disease, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 939–953. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (UK). Parkinson’s Disease in Adults: Diagnosis and Management; National Institute for Health and Care Excellence (UK): London, UK, 2017. [Google Scholar]
- Louis, E.D.; Barnes, L.; Albert, S.M.; Cote, L.; Schneier, F.R.; Pullman, S.L.; Yu, Q. Correlates of functional disability in essential tremor. Mov. Disord. 2001, 16, 914–920. [Google Scholar] [CrossRef]
- Elble, R.J.; Brilliant, M.; Leffler, K.; Higgins, C. Quantification of essential tremor in writing and drawing. Mov. Disord. 1996, 11, 70–78. [Google Scholar] [CrossRef]
- Héroux, M.E.; Parisi, S.L.; Larocerie-Salgado, J.; Norman, K.E. Upper-Extremity Disability in Essential Tremor. Arch. Phys. Med. Rehabil. 2006, 87, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.E.; D’Amboise, S.N.; Pari, G.; Héroux, M.E. Tremor during movement correlates well with disability in people with essential tremor. Mov. Disord. 2011, 26, 2088–2094. [Google Scholar] [CrossRef] [PubMed]
- Rajput, A.H.; Robinson, C.A. Essential tremor course and disability: A clinicopathologic study of 20 cases. Neurology 2004, 62, 932–936. [Google Scholar] [CrossRef]
- Louis, E.D.; Machado, D.G. Tremor-related quality of life: A comparison of essential tremor vs. Parkinson’s disease patients. Park. Relat. Disord. 2015, 21, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, D.; Schwieger, D.; Moises, H.; Deuschl, G. Quality of life and personality in essential tremor patients. Mov. Disord. 2006, 21, 1114–1118. [Google Scholar] [CrossRef]
- Monin, J.K.; Gutierrez, J.; Kellner, S.; Morgan, S.; Collins, K.; Rohl, B.; Migliore, F.; Cosentino, S.; Huey, E.; Louis, E.D. Psychological Suffering in Essential Tremor: A Study of Patients and Those Who Are Close to Them. Tremor Other Hyperkinetic Mov. 2017, 7, 526. [Google Scholar] [CrossRef]
- Schneier, F.R.; Barnes, L.F.; Albert, S.M.; Louis, E.D. Characteristics of Social Phobia among Persons with Essential Tremor. J. Clin. Psychiatry 2001, 62, 367–372. [Google Scholar] [CrossRef]
- Damen, J.A.A.G. Author’s reply to Woodward. BMJ 2016, 354, i4485. [Google Scholar] [CrossRef]
- Elias, W.J.; Shah, B.B. Tremor. JAMA 2014, 311, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Zesiewicz, T.A.; Elble, R.; Louis, E.D.; Hauser, R.A.; Sullivan, K.L.; Dewey, R.B.; Ondo, W.G.; Gronseth, G.S.; Weiner, W.J. Practice Parameter: Therapies for essential tremor: Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2005, 64, 2008–2020. [Google Scholar] [CrossRef]
- Zesiewicz, T.A.; Elble, R.J.; Louis, E.D.; Gronseth, G.S.; Ondo, W.G.; Dewey, R.B.; Okun, M.S.; Sullivan, K.L.; Weiner, W.J. Evidence-based guideline update: Treatment of essential tremor: Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2011, 77, 1752–1755. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.J.; Mestre, T.A.; Lyons, K.E.; Benito-León, J.; Tan, E.; Abbruzzese, G.; Hallett, M.; Haubenberger, D.; Elble, R.; Deuschl, G.; et al. MDS evidence-based review of treatments for essential tremor. Mov. Disord. 2019, 34, 950–958. [Google Scholar] [CrossRef]
- O’Connor, R.J.; Kini, M.U. Non-pharmacological and non-surgical interventions for tremor: A systematic review. Park. Relat. Disord. 2011, 17, 509–515. [Google Scholar] [CrossRef]
- Haubenberger, D.; Hallett, M. Essential Tremor. N. Engl. J. Med. 2018, 378, 1802–1810. [Google Scholar] [CrossRef]
- Deuschl, G.; Raethjen, J.; Hellriegel, H.; Elble, R. Treatment of patients with essential tremor. Lancet Neurol. 2011, 10, 148–161. [Google Scholar] [CrossRef]
- Diaz, N.L.; Louis, E.D. Survey of medication usage patterns among essential tremor patients: Movement disorder specialists vs. general neurologists. Park. Relat. Disord. 2010, 16, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Zappia, M.; Italian Movement Disorders Association (DISMOV-SIN) Essential Tremor Committee; Albanese, A.; Bruno, E.; Colosimo, C.; Filippini, G.; Martinelli, P.; Nicoletti, A.; Quattrocchi, G. Treatment of essential tremor: A systematic review of evidence and recommendations from the Italian Movement Disorders Association. J. Neurol. 2012, 260, 714–740. [Google Scholar] [CrossRef] [PubMed]
- Velisar, A.; Syrkin-Nikolau, J.; Blumenfeld, Z.; Trager, M.; Afzal, M.; Prabhakar, V.; Bronte-Stewart, H. Dual threshold neural closed loop deep brain stimulation in Parkinson disease patients. Brain Stimul. 2019, 12, 868–876. [Google Scholar] [CrossRef]
- Weerasinghe, G.; Duchet, B.; Cagnan, H.; Brown, P.; Bick, C.; Bogacz, R. Predicting the effects of deep brain stimulation using a reduced coupled oscillator model. PLoS Comput. Biol. 2019, 15, e1006575. [Google Scholar] [CrossRef] [PubMed]
- Kern, D.S.; Picillo, M.; Thompson, J.A.; Sammartino, F.; Di Biase, L.; Munhoz, R.P.; Fasano, A. Interleaving Stimulation in Parkinson’s Disease, Tremor, and Dystonia. Ster. Funct. Neurosurg. 2018, 96, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.S.; Chen, R. Invasive and Noninvasive Brain Stimulation in Parkinson’s Disease: Clinical Effects and Future Perspectives. Clin. Pharmacol. Ther. 2019, 106, 763–775. [Google Scholar] [CrossRef]
- Farokhniaee, A.; McIntyre, C.C. Theoretical principles of deep brain stimulation induced synaptic suppression. Brain Stimul. 2019, 12, 1402–1409. [Google Scholar] [CrossRef]
- Mao, Z.; Ling, Z.; Pan, L.; Xu, X.; Cui, Z.; Liang, S.; Yu, X. Comparison of Efficacy of Deep Brain Stimulation of Different Targets in Parkinson’s Disease: A Network Meta-Analysis. Front. Aging Neurosci. 2019, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Schuurman, P.R.; Bosch, D.A.; Bossuyt, P.M.; Bonsel, G.J.; Van Someren, E.J.; De Bie, R.M.; Merkus, M.P.; Speelman, J.D. A Comparison of Continuous Thalamic Stimulation and Thalamotomy for Suppression of Severe Tremor. N. Engl. J. Med. 2000, 342, 461–468. [Google Scholar] [CrossRef]
- Elble, R.J.; Shih, L.; Cozzens, J.W. Surgical treatments for essential tremor. Expert Rev. Neurother. 2018, 18, 303–321. [Google Scholar] [CrossRef]
- Ravikumar, V.K.; Parker, J.J.; Hornbeck, T.S.; Santini, V.E.; Pauly, K.B.; Wintermark, M.; Ghanouni, P.; Stein, S.C.; Halpern, C.H. Cost-effectiveness of focused ultrasound, radiosurgery, and DBS for essential tremor. Mov. Disord. 2017, 32, 1165–1173. [Google Scholar] [CrossRef]
- Lozano, A.M.; Lipsman, N.; Bergman, H.; Brown, P.; Chabardes, S.; Chang, J.W.; Matthews, K.; McIntyre, C.C.; Schlaepfer, T.E.; Schulder, M.; et al. Deep brain stimulation: Current challenges and future directions. Nat. Rev. Neurol. 2019, 15, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Walters, H.; Shah, B.B. Focused Ultrasound and Other Lesioning Therapies in Movement Disorders. Curr. Neurol. Neurosci. Rep. 2019, 19, 66. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.K.; McGovern, R.A.; Brown, L.T.; Sheehy, J.P.; Zacharia, B.E.; Mikell, C.B.; Bruce, S.S.; Ford, B.; McKhann, G.M. Disparities in Access to Deep Brain Stimulation Surgery for Parkinson Disease. JAMA Neurol. 2014, 71, 291–299. [Google Scholar] [CrossRef]
- Lange, M.; Mauerer, J.; Schlaier, J.; Janzen, A.; Zeman, F.; Bogdahn, U.; Brawanski, A.; Hochreiter, A. Underutilization of deep brain stimulation for Parkinson’s disease? A survey on possible clinical reasons. Acta Neurochir. 2017, 159, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.W.; Deeb, W.; Patel, B.; Ramirez-Zamora, A. Is deep brain stimulation therapy underutilized for movement disorders? Expert Rev. Neurother. 2018, 18, 899–901. [Google Scholar] [CrossRef]
- Kim, M.-R.; Yun, J.Y.; Jeon, B.; Lim, Y.H.; Kim, K.R.; Yang, H.-J.; Paek, S.H. Patients’ reluctance to undergo deep brain stimulation for Parkinson’s disease. Park. Relat. Disord. 2016, 23, 91–94. [Google Scholar] [CrossRef]
- Food and Drug Administration. Medical Devices; Neurological Devices; Classification of the External Upper Limb Tremor Stimulator. Final order. Fed. Regist. 2018, 83, 52315–52316. [Google Scholar]
- Beringhause, S.; Rosen, M.; Huang, S. Evaluation of a damped joystick for people disabled by intention tremor. In Proceedings of the 12th Annual Conference on Rehabilitation Technology, New Orleans, LA, USA, 25–30 June 1989; pp. 41–42. [Google Scholar]
- Hendriks, J.; Rosen, M.; Berube, N.; Aisen, M. A second-generation joystick for people disabled by tremor. In Proceedings of the 14th Annual RESNA Conference, Kansas City, MO, USA, 21–26 June 1991; pp. 248–251. [Google Scholar]
- Rosen, M.J. Tremor Suppressing Hand Controls. U.S. Patent 4,689,449, 25 August 1987. [Google Scholar]
- Rosen, M.J. Multiple Degree of Freedom Damped Hand Controls. U.S. Patent 5,107,080, 21 April 1992. [Google Scholar]
- Arnold, A.S.; Rosen, M.J.; Aisen, M.L. Evaluation of a controlled-energy-dissipation orthosis for tremor suppression. J. Electromyogr. Kinesiol. 1993, 3, 131–148. [Google Scholar] [CrossRef]
- Rosen, M.J.; Arnold, A.S.; Baiges, I.J.; Aisen, M.L.; Eglowstein, S.R. Design of a controlled-energy-dissipation orthosis (CEDO) for functional suppression of intention tremors. J. Rehabil. Res. Dev. 1995, 32, 1–16. [Google Scholar] [PubMed]
- Rosen, M.J.; Baiges, I.J. Whole-Arm Orthosis for Steadying Limb Motion. U.S. Patent 5,231,998, 3 August 1993. [Google Scholar]
- Maxwell, S.M. A Modulated-Energy-Dissipation Manipulator and Application to Suppressing Human Arm Tremor. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 1990. [Google Scholar]
- Maxwell, S.M. A System for Resisting Limb Movement. EP Patent 0,569,489 B1, 10 May 1995. [Google Scholar]
- Johnson, M. Transcutaneous Electrical Nerve Stimulation: Mechanisms, Clinical Application and Evidence. Rev. Pain 2007, 1, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Gibson, W.; Wand, B.M.; Meads, C.; Catley, M.J.; E O’Connell, N. Transcutaneous electrical nerve stimulation (TENS) for chronic pain—An overview of Cochrane Reviews. Cochrane Database Syst. Rev. 2019, 4, CD011890. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.I.; Paley, C.A.; Howe, T.E.; Sluka, K.A. Transcutaneous electrical nerve stimulation for acute pain. Cochrane Database Syst. Rev. 2015, 6, CD006142. [Google Scholar] [CrossRef]
- Gibson, W.; Wand, B.M.; E O’Connell, N. Transcutaneous electrical nerve stimulation (TENS) for neuropathic pain in adults. Cochrane Database Syst. Rev. 2017, 2017, 011976. [Google Scholar] [CrossRef]
- Toglia, J.U.; Izzo, K. Treatment of myoclonic dystonia with transcutaneous electrical nerve stimulation. Neurol. Sci. 1985, 6, 75–78. [Google Scholar] [CrossRef]
- Campbell, J.N.; Raja, S.N.; Meyer, R.A.; MacKinnon, S.E. Myelinated afferents signal the hyperalgesia associated with nerve injury. Pain 1988, 32, 89–94. [Google Scholar] [CrossRef]
- Truini, A.; Padua, L.; Biasiotta, A.; Caliandro, P.; Pazzaglia, C.; Galeotti, F.; Inghilleri, M.; Cruccu, G. Differential involvement of A-delta and A-beta fibres in neuropathic pain related to carpal tunnel syndrome. Pain 2009, 145, 105–109. [Google Scholar] [CrossRef]
- Xu, Z.-Z.; Kim, Y.H.; Bang, S.; Zhang, Y.; Berta, T.; Wang, F.; Oh, S.B.; Ji, R.-R. Inhibition of mechanical allodynia in neuropathic pain by TLR5-mediated A-fiber blockade. Nat. Med. 2015, 21, 1326–1331. [Google Scholar] [CrossRef]
- Nagi, S.S.; Marshall, A.G.; Makdani, A.; Jarocka, E.; Liljencrantz, J.; Ridderström, M.; Shaikh, S.; O’Neill, F.; Saade, D.; Donkervoort, S.; et al. An ultrafast system for signaling mechanical pain in human skin. Sci. Adv. 2019, 5, eaaw1297. [Google Scholar] [CrossRef]
- Garcia, K.; Wray, J.K.; Kumar, S. Spinal Cord Stimulation, StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Ferrara, J.; Stamey, W.; Strutt, A.M.; Adam, O.R.; Jankovic, J. Transcutaneous Electrical Stimulation (TENS) for Psychogenic Movement Disorders. J. Neuropsychiatry Clin. Neurosci. 2011, 23, 141–148. [Google Scholar] [CrossRef]
- Serrano-Muñoz, D.; Avendaño-Coy, J.; Simón-Martínez, C.; Taylor, J.; Gómez-Soriano, J. Effect of high-frequency alternating current transcutaneous stimulation over muscle strength: A controlled pilot study. J. Neuroeng. Rehabil. 2018, 15, 1–4. [Google Scholar] [CrossRef]
- Hao, M.-Z.; Xu, S.-Q.; Hu, Z.-X.; Xu, F.-L.; Niu, C.-X.M.; Xiao, Q.; Lan, N. Inhibition of Parkinsonian tremor with cutaneous afferent evoked by transcutaneous electrical nerve stimulation. J. Neuroeng. Rehabil. 2017, 14, 75. [Google Scholar] [CrossRef] [PubMed]
- Hao, M.-Z.; He, X.; Kipke, D.R.; Lan, N. Effects of electrical stimulation of cutaneous afferents on corticospinal transmission of tremor signals in patients with Parkinson’s disease. In Proceedings of the 2013 6th International IEEE/EMBS Conference on Neural Engineering (NER), San Diego, CA, USA, 6–8 November 2013. [Google Scholar]
- Munhoz, R.P.; Hanajima, R.; Ashby, P.; Lang, A.E. Acute effect of transcutaneous electrical nerve stimulation on tremor. Mov. Disord. 2002, 18, 191–194. [Google Scholar] [CrossRef] [PubMed]
- U.S. National Library of Medicine. Prospective Study for Symptomatic Relief of ET with Cala Therapy (PROSPECT). 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT03597100 (accessed on 20 July 2020).
- Hanajima, R. Somatosensory evoked potentials (SEPs) recorded from deep brain stimulation (DBS) electrodes in the thalamus and subthalamic nucleus (STN). Clin. Neurophysiol. 2004, 115, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Hanajima, R.; Chen, R.; Ashby, P.; Lozano, A.M.; Hutchison, W.D.; Davis, K.D.; Dostrovsky, J.O. Very Fast Oscillations Evoked by Median Nerve Stimulation in the Human Thalamus and Subthalamic Nucleus. J. Neurophysiol. 2004, 92, 3171–3182. [Google Scholar] [CrossRef] [PubMed]
- Bathien, N.; Rondot, P.; Toma, S. Inhibition and synchronisation of tremor induced by a muscle twitch. J. Neurol. Neurosurg. Psychiatry 1980, 43, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Dosen, S.; Muceli, S.; Dideriksen, J.L.; Romero, J.P.; Rocon, E.; Pons, J.; Farina, D. Online Tremor Suppression Using Electromyography and Low-Level Electrical Stimulation. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 23, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Pahwa, R.; Dhall, R.; Ostrem, J.; Gwinn, R.; Lyons, K.; Ro, S.; Dietiker, C.; Luthra, N.; Ms, P.C.; Hamner, S.; et al. An Acute Randomized Controlled Trial of Noninvasive Peripheral Nerve Stimulation in Essential Tremor. Neuromodulation 2018, 22, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Isaacson, S.H.; Peckham, E.; Tse, W.; Waln, O.; Way, C.; Petrossian, M.T.; Dahodwala, N.; Soileau, M.J.; Lew, M.; Dietiker, C.; et al. Prospective Home-use Study on Non-invasive Neuromodulation Therapy for Essential Tremor. Tremor Other Hyperkinetic Mov. 2020, 10, 29. [Google Scholar] [CrossRef]
- Elek, J.; Prochazka, A. Attenuation of wrist tremor with closed-loop electrical stimulation of muscles. J. Physiol. 1989, 414, 17P. [Google Scholar]
- Javidan, M.; Elek, J.; Prochazka, A. Tremor reduction by functional electrical stimulation. Neurology 1990, 40, 369. [Google Scholar]
- Prochazka, A.; Elek, J.; Javidan, M. Attenuation of pathological tremors by functional electrical stimulation I: Method. Ann. Biomed. Eng. 1992, 20, 205–224. [Google Scholar] [CrossRef]
- Javidan, M.; Elek, J.; Prochazka, A. Attenuation of pathological tremors by functional electrical stimulation II: Clinical evaluation. Ann. Biomed. Eng. 1992, 20, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Law, J.J. Reduction of Pathological Tremor by Functional Electrical Stimulation Using Digital Feedback Control. Master’s Thesis, University of Alberta, Edmonton, AB, Canada, 1995. [Google Scholar]
- Gillard, D.; Cameron, T.; Prochazka, A.; Gauthier, M. Tremor suppression using functional electrical stimulation: A comparison between digital and analog controllers. IEEE Trans. Rehabil. Eng. 1999, 7, 385–388. [Google Scholar] [CrossRef]
- Dideriksen, J.L.; Laine, C.M.; Dosen, S.; Muceli, S.; Rocon, E.; Pons, J.L.; Benito-Leon, J.; Farina, D. Electrical Stimulation of Afferent Pathways for the Suppression of Pathological Tremor. Front. Neurosci. 2017, 11, 178. [Google Scholar] [CrossRef]
- Maneski, L.P.; Jorgovanović, N.; Ilić, V.; Došen, S.; Keller, T.; Popović, M.B.; Popović, D.B. Electrical stimulation for the suppression of pathological tremor. Med. Biol. Eng. Comput. 2011, 49, 1187–1193. [Google Scholar] [CrossRef]
- Popović, D.; Popović-Maneski, L. The Instrumented Shoe Insole for Rule-Based Control of Gait in Persons with Hemiplegia. EasyChair Prepr. 2019, p. 1345. Available online: https://easychair.org/publications/preprint/ZF3Z (accessed on 26 March 2021).
- Gallego, J.Á.; Rocon, E.; Belda-Lois, J.M.; Pons, J.L. A neuroprosthesis for tremor management through the control of muscle co-contraction. J. Neuroeng. Rehabil. 2013, 10, 36. [Google Scholar] [CrossRef]
- Jitkritsadakul, O.; Thanawattano, C.; Anan, C.; Bhidayasiri, R. Tremor’s glove-an innovative electrical muscle stimulation therapy for intractable tremor in Parkinson’s disease: A randomized sham-controlled trial. J. Neurol. Sci. 2017, 381, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Meunier, S.; Pol, S.; Houeto, J.L.; Vidailhet, M. Abnormal reciprocal inhibition between antagonist muscles in Parkinson’s disease. Brain 2000, 123, 1017–1026. [Google Scholar] [CrossRef]
- Heo, J.-H.; Jeon, H.-M.; Choi, E.-B.; Kwon, D.-Y.; Eom, G.-M. Effect of Sensory Electrical Stimulation on Resting Tremors in Patients with Parkinson’s Disease and SWEDDS. J. Mech. Med. Biol. 2019, 19, 1940033. [Google Scholar] [CrossRef]
- Bickel, C.S.; Gregory, C.M.; Dean, J.C. Motor unit recruitment during neuromuscular electrical stimulation: A critical appraisal. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 111, 2399–2407. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Sadowsky, C.; Obst, K.; Meyer, B.; McDonald, J. Functional Electrical Stimulation in Spinal Cord Injury: From Theory to Practice. Top. Spinal Cord Inj. Rehabil. 2012, 18, 28–33. [Google Scholar] [CrossRef]
- Terry, T.E.; Hoyt, L.J., Sr. Cerebral Palsy Arm and Hand Brace. U.S. Patent 4,237,873, 9 December 1980. [Google Scholar]
- Kotovsky, J.; Rosen, M.J. A wearable tremor-suppression orthosis. J. Rehabil. Res. Dev. 1998, 35, 373–387. [Google Scholar] [PubMed]
- Rocon, E.; Ruiz, A.; Pons, J.L.; Belda-Lois, J.; Sánchez-Lacuesta, J. Rehabilitation Robotics: A Wearable Exo-Skeleton for Tremor Assessment and Suppression. In Proceedings of the Proceedings of the 2005 IEEE International Conference on Robotics and Automation, Barcelona, Spain, 18–22 April 2005. [Google Scholar]
- Rocon, E.; Ruiz, A.; Brunetti, F.; Pons, J.L.; Belda-Lois, J.; Sánchez-Lacuesta, J. On the use of an active wearable exoskeleton for tremor suppression via biomechanical loading. In Proceedings of the 2006 IEEE International Conference on Robotics and Automation, Orlando, FL, USA, 15–19 May 2006. [Google Scholar]
- Manto, M.; Rocon, E.; Pons, J.L.; Belda, J.M.; Camut, S. Evaluation of a wearable orthosis and an associated algorithm for tremor suppression. Physiol. Meas. 2007, 28, 415–425. [Google Scholar] [CrossRef]
- Rocón, E.; Belda-Lois, J.M.; Ruiz, A.; Manto, M.; Moreno, J.C.; Pons, J.L. Design and Validation of a Rehabilitation Robotic Exoskeleton for Tremor Assessment and Suppression. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 367–378. [Google Scholar] [CrossRef]
- Taheri, B.; Case, D.; Richer, E. Active Tremor Estimation and Suppression in Human Elbow Joint. In Proceedings of the ASME 2011 Dynamic Systems and Control Conference, Arlington, VI, USA, 31 October–2 November 2011. [Google Scholar]
- Taheri, B.; Case, D.; Richer, E. Robust Controller for Tremor Suppression at Musculoskeletal Level in Human Wrist. IEEE Trans. Neural Syst. Rehabil. Eng. 2013, 22, 379–388. [Google Scholar] [CrossRef]
- Taheri, B.; Case, D.; Richer, E. Adaptive Suppression of Severe Pathological Tremor by Torque Estimation Method. IEEE/ASME Trans. Mechatron. 2014, 20, 717–727. [Google Scholar] [CrossRef]
- Zamanian, A.H.; Richer, E. Adaptive disturbance rejection controller for pathological tremor suppression with permanent magnet linear motor. In Proceedings of the ASME 2017 Dynamic Systems and Control Conference, Tysons, VI, USA, 11–13 October 2017. [Google Scholar]
- Herrnstadt, G.; Menon, C. Voluntary-Driven Elbow Orthosis with Speed-Controlled Tremor Suppression. Front. Bioeng. Biotechnol. 2016, 4, 29. [Google Scholar] [CrossRef]
- Zhou, Y.; Naish, M.D.; Jenkins, M.E.; Trejos, A.L. Design and validation of a novel mechatronic transmission system for a wearable tremor suppression device. Robot. Auton. Syst. 2017, 91, 38–48. [Google Scholar] [CrossRef]
- Ando, T.; Watanabe, M.; Fujie, M.G. Extraction of voluntary movement for an EMG controlled exoskeltal robot of tremor patients. In Proceedings of the 2009 4th International IEEE/EMBS Conference on Neural Engineering, Antalya, Turkey, 29 April–2 May 2009. [Google Scholar]
- Seki, M.; Matsumoto, Y.; Ando, T.; Kobayashi, Y.; Fujie, M.G.; Iijima, H.; Nagaoka, M. Development of robotic upper limb orthosis with tremor suppressiblity and elbow joint movability. In Proceedings of the 2011 IEEE International Conference on Systems, Man, and Cybernetics, Anchorage, AK, USA, 9–12 October 2011. [Google Scholar]
- Seki, M.; Matsumoto, Y.; Ando, T.; Kobayashi, Y.; Iijima, H.; Nagaoka, M.; Fujie, M.G. The weight load inconsistency effect on voluntary movement recognition of essential tremor patient. In Proceedings of the 2011 IEEE International Conference on Robotics and Biomimetics, Karon Beach, Thailand, 7–11 December 2011. [Google Scholar]
- Ando, T.; Watanabe, M.; Nishimoto, K.; Matsumoto, Y.; Seki, M.; Fujie, M.G. Myoelectric-Controlled Exoskeletal Elbow Robot to Suppress Essential Tremor: Extraction of Elbow Flexion Movement Using STFTs and TDNN. J. Robot. Mechatron. 2012, 24, 141–149. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Amemiya, M.; Kaneishi, D.; Nakashima, Y.; Seki, M.; Ando, T.; Kobayashi, Y.; Iijima, H.; Nagaoka, M.; Fujie, M.G. Development of an elbow-forearm interlock joint mechanism toward an exoskeleton for patients with essential tremor. In Proceedings of the 2014 IEEE/RSJ International Conference on Intelligent Robots and Systems, Chicago, IL, USA, 14–18 September 2014. [Google Scholar]
- Matsumoto, Y.; Seki, M.; Ando, T.; Kobayashi, Y.; Nakashima, Y.; Iijima, H.; Nagaoka, M.; Fujie, M.G. Development of an Exoskeleton to Support Eating Movements in Patients with Essential Tremor. J. Robot. Mechatron. 2013, 25, 949–958. [Google Scholar] [CrossRef]
- Huen, D.; Liu, J.; Lo, B. An integrated wearable robot for tremor suppression with context aware sensing. In Proceedings of the 2016 IEEE 13th International Conference on Wearable and Implantable Body Sensor Networks (BSN), San Francisco, CA, USA, 14–17 June 2016. [Google Scholar]
- Fromme, N.P.; Camenzind, M.; Riener, R.; Rossi, R.M. Need for mechanically and ergonomically enhanced tremor-suppression orthoses for the upper limb: A systematic review. J. Neuroeng. Rehabil. 2019, 16, 1–15. [Google Scholar] [CrossRef]
- Loureiro, R.; Belda-Lois, J.M.; Lima, E.; Pons, J.; Sanchez-Lacuesta, J.; Harwin, W.S. Upper Limb Tremor Suppression in ADL Via an Orthosis Incorporating a Controllable Double Viscous Beam Actuator. In Proceedings of the 9th International Conference on Rehabilitation Robotics, Chicago, IL, USA, 28 June–1 July 2005. [Google Scholar]
- Case, D.; Taheri, B.; Richer, E. Dynamic Magnetorheological Damper for Othotic Tremor Suppression. In Proceedings of the 2011 Hawaii University International Conference on Mathematics and Engineering, Honolulu, HI, USA, 13–15 June 2011. [Google Scholar]
- Case, D.; Taheri, B.; Richer, E. Multiphysics modeling of magnetorheological dampers. Int. J. Multiphysics 2013, 7, 61–76. [Google Scholar] [CrossRef]
- Case, D.; Taheri, B.; Richer, E. A Lumped-Parameter Model for Adaptive Dynamic MR Damper Control. IEEE ASME Trans. Mechatron. 2014, 20, 1–8. [Google Scholar] [CrossRef]
- Case, D.; Taheri, B.; Richer, E. Active control of MR wearable robotic orthosis for pathological tremor suppression. In Proceedings of the ASME 2015 Dynamic Systems and Control Conference, Columbus, OH, USA, 28–30 October 2015. [Google Scholar]
- Zahedi, A.; Zhang, B.; Yi, A.; Zhang, D. A Soft Exoskeleton for Tremor Suppression Equipped with Flexible Semiactive Actuator. Soft Robot. 2020. [Google Scholar] [CrossRef] [PubMed]
- Herrnstadt, G.; Menon, C. On-Off Tremor Suppression Orthosis with Electromagnetic Brake. Int. J. Mech. Eng. Mechatron. 2013, 1, 7–14. [Google Scholar] [CrossRef]
- Kalaiarasi, A.; Kumar, L.A. Sensor Based Portable Tremor Suppression Device for Stroke Patients. Electrother. Res. 2018, 43, 29–37. [Google Scholar] [CrossRef]
- Rudraraju, S.; Nguyen, T. Wearable Tremor Reduction Device (TRD) for Human Hands and Arms. In Proceedings of the 2018 Design of Medical Devices Conference, Minneapolis, MI, USA, 9–12 April 2018. [Google Scholar]
- Elias, M.; Patel, S.; Maamary, E.; Araneta, L.; Obaid, N. Apparatus for Damping Involuntary Hand Motions. U.S. Patent 0,216,628 A1, 18 July 2019. [Google Scholar]
- Hunter, R.; Pivach, L.; Madere, K.; Van Gemmert, A.W.A. Potential benefits of the Readi-Steadi on essential tremor. In Proceedings of the 5th Annual LSU Discover Day, Baton Rouge, LA, USA, 10 April 2018. [Google Scholar]
- Paulig, J.; Jabusch, H.-C.; Groãÿbach, M.; Boullet, L.; Altenmüller, E. Sensory trick phenomenon improves motor control in pianists with dystonia: Prognostic value of glove-effect. Front. Psychol. 2014, 5, 1012. [Google Scholar] [CrossRef]
- Fromme, N.P.; Camenzind, M.; Riener, R.; Rossi, R.M. Design of a lightweight passive orthosis for tremor suppression. J. Neuroeng. Rehabil. 2020, 17, 1–15. [Google Scholar] [CrossRef]
- Lu, Z.; Huang, Z. Analytical and experimental studies on particle damper used for tremor suppression. J. Vib. Control 2020, 1–11. [Google Scholar] [CrossRef]
- Katz, R.; Buki, E.; Zacksenhouse, M. Attenuating Tremor Using Passive Devices. Stud. Health Technol. Inform. 2017, 242, 741–747. [Google Scholar]
- Buki, E.; Katz, R.; Zacksenhouse, M.; Schlesinger, I. Vib-bracelet: A passive absorber for attenuating forearm tremor. Med Biol. Eng. Comput. 2017, 56, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Takanokura, M.; Sugahara, R.; Miyake, N.; Ishiguro, K.; Muto, T.; Sakamoto, K. Upper-limb orthoses implemented with air dashpots for suppression of pathological tremor in daily activities. In Proceedings of the 23rd Congress of International Society of Biomechanics, Brussels, Belgium, 2–3 July 2011. [Google Scholar]
- Lusardi, M.M. Tremor, chorea and other involuntary movement. In Geriatric Rehabilitation Manual, 2nd ed.; Kauffman, T., Barr, J., Moran, M., Eds.; Elsevier: New York, NY, USA, 2007; pp. 215–225. [Google Scholar]
- Kelley, C.R.; Kauffman, J.L. Tremor-Active Controller for Dielectric Elastomer-Based Pathological Tremor Suppression. IEEE/ASME Trans. Mechatron. 2020, 25, 1143–1148. [Google Scholar] [CrossRef]
- Kelley, C.R.; Kauffman, J.L. Scaled Tremor Suppression with Folded Dielectric Elastomer Stack Actuators. In Electroactive Polymer Actuators and Devices (EAPAD) XXII; International Society for Optics and Photonics: Bellingham, WA, USA, 2020. [Google Scholar] [CrossRef]
- Michaelis, J. Introducing the neater eater. Action Res. 1988, 6, 2–3. [Google Scholar]
- Mandy, A.; Sims, T.; Stew, G.; Onions, D. Manual Feeding Device Experiences of People with a Neurodisability. Am. J. Occup. Ther. 2018, 72, 7203345010p1–7203345010p5. [Google Scholar] [CrossRef]
- Pathak, A.; Redmond, J.A.; Allen, M.; Chou, K.L. A noninvasive handheld assistive device to accommodate essential tremor: A pilot study. Mov. Disord. 2013, 29, 838–842. [Google Scholar] [CrossRef]
- Sabari, J.; Stefanov, D.G.; Chan, J.; Goed, L.; Starr, J. Adapted Feeding Utensils for People With Parkinson’s-Related or Essential Tremor. Am. J. Occup. Ther. 2019, 73, 7302205120p1–7302205120p9. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ren, K. Arm Vibration Damping Device. U.S. Patent 0,327,023 A1, 16 November 2017. [Google Scholar]
- De Panisse, P.; Ibrahim, Y.; Medeisis, J.; Tiarvando, L.; Vaklev, N.L.; Ong, J.F.; Gan, B.; Koh, B.; Soler, X.L.; Choong Ngan Lou, W. Tremor Stabilization Apparatus and Methods. U.S. Patent 0266820 A1, 20 September 2018. [Google Scholar]
- Hall, W.D. Hand-Held Gyroscopic Device. U.S. Patent 5,058,571 A, 22 October 1991. [Google Scholar]
- Kalvert, M.A. Adjustable and Tunable Hand Tremor Stabilizer. U.S. Patent 6,730,049 B2, 4 May 2004. [Google Scholar]
- Ferrington, D.G.; Nail, B.S.; Rowe, M. Human tactile detection thresholds: Modification by inputs from specific tactile receptor classes. J. Physiol. 1977, 272, 415–433. [Google Scholar] [CrossRef]
- Douglas, P.R.; Ferrington, D.G.; Rowe, M. Coding of information about tactile stimuli by neurones of the cuneate nucleus. J. Physiol. 1978, 285, 493–513. [Google Scholar] [CrossRef] [PubMed]
- Uemura, Y.; Haque, T.; Sato, F.; Tsutsumi, Y.; Ohara, H.; Oka, A.; Furuta, T.; Bae, Y.C.; Yamashiro, T.; Tachibana, Y.; et al. Proprioceptive thalamus receiving forelimb and neck muscle spindle inputs via the external cuneate nucleus in the rat. Brain Struct. Funct. 2020, 225, 2177–2192. [Google Scholar] [CrossRef]
- Tracey, D.J. The projection of joint receptors to the cuneate nucleus in the cat. J. Physiol. 1980, 305, 433–449. [Google Scholar] [CrossRef] [PubMed]
- Lora-Millán, J.S.; López-Blanco, R.; Gallego, J.Á.; Méndez-Guerrero, A.; de La Aleja, J.G.; Rocon, E. Mechanical vibration does not systematically reduce the tremor in essential tremor patients. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Louis, E.D. The Roles of Age and Aging in Essential Tremor: An Epidemiological Perspective. Neuroepidemiology 2019, 52, 111–118. [Google Scholar] [CrossRef]
- Louis, E.D.; Ford, B.; Barnes, L.F. Clinical subtypes of essential tremor. Arch. Neurol. 2000, 57, 1194–1198. [Google Scholar] [CrossRef]
- Benito-León, J.; Louis, E.D.; Bermejo-Pareja, F. Elderly-onset essential tremor is associated with dementia. Neurology 2006, 66, 1500–1505. [Google Scholar] [CrossRef] [PubMed]
- Hanagasi, H.A.; Tufekcioglu, Z.; Emre, M. Dementia in Parkinson’s disease. J. Neurol. Sci. 2017, 374, 26–31. [Google Scholar] [CrossRef]
- Park, I.-S.; Oh, Y.-S.; Lee, K.-S.; Yang, D.-W.; Song, I.-U.; Park, J.-W.; Kim, J.-S. Subtype of Mild Cognitive Impairment in Elderly Patients with Essential Tremor. Alzheimer Dis. Assoc. Disord. 2015, 29, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Benito-León, J.; Louis, E.D.; Mitchell, A.J.; Bermejo-Pareja, F. Elderly-Onset Essential Tremor and Mild Cognitive Impairment: A Population-Based Study (NEDICES). J. Alzheimer’s Dis. 2011, 23, 727–735. [Google Scholar] [CrossRef]
- Monastero, R.; Cicero, C.E.; Baschi, R.; Davì, M.; Luca, A.; Restivo, V.; Zangara, C.; Fierro, B.; Zappia, M.; Nicoletti, A. Mild cognitive impairment in Parkinson’s disease: The Parkinson’s disease cognitive study (PACOS). J. Neurol. 2018, 265, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Frost & Sullivan. Assessing the Full Impact of Essential Tremor on Patient Quality of Life and Finances in the United States. 2018. Available online: https://www.insightec.com/media/1550/fs_wp_insightec-et_010819.pdf (accessed on 20 September 2020).
- Axelrad, J.E.; Louis, E.D.; Honig, L.S.; Flores, I.; Ross, G.W.; Pahwa, R.; Lyons, K.E.; Faust, P.L.; Vonsattel, J.P.G. Reduced Purkinje Cell Number in Essential Tremor. Arch. Neurol. 2008, 65, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Louis, E.D.; Lee, M.; Babij, R.; Ma, K.; Cortés, E.; Vonsattel, J.-P.G.; Faust, P.L. Reduced Purkinje cell dendritic arborization and loss of dendritic spines in essential tremor. Brain 2014, 137, 3142–3148. [Google Scholar] [CrossRef]
- Helmich, R.C.; Janssen, M.J.R.; Oyen, W.J.G.; Bloem, B.R.; Toni, I. Pallidal dysfunction drives a cerebellothalamic circuit into Parkinson tremor. Ann. Neurol. 2011, 69, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Espay, A.J.; Lang, A.E.; Erro, R.; Merola, A.; Fasano, A.; Berardelli, A.; Bhatia, K.P. Essential pitfalls in “essential” tremor. Mov. Disord. 2017, 32, 325–331. [Google Scholar] [CrossRef] [PubMed]

| Type of Device | Study Participants (n) | Efficacy | Risks | Refs |
|---|---|---|---|---|
| Electrical Stimulation Systems: Transcutaneous Electrical Nerve Stimulators | ||||
| Cala ONE ‡ | ET (77) |
|
| [70] |
| Cala Trio * | ET (205) |
| [71] | |
| Electrical Stimulation Systems: Functional Electrical Stimulators | ||||
| MOTIMOVE | ET (3); PD tremor (4) | 67% tremor suppression | Muscle fatigue | [79] |
| TREMOR neurorobot | ET (4); PD tremor (2) | 52% tremor suppression | [81] | |
| Tremor’s glove | PD tremor (30) | Reduced UPDRS score (p = 0.001) | [82] | |
| Wearable Orthoses: Active Orthoses | ||||
| WOTAS exoskeleton | ET (7); MS tremor (1); Posttraumatic tremor (1); Mixed tremor (1) | 40% tremor suppression [92] | Not reported | [89,90,91,92] |
| Pneumatic actuator-based orthosis | ET (5) §; PD tremor (5) § | 98.1% tremor suppression [95] | [93,94,95] | |
| PMLM-based orthosis | PD tremor (5) § | 97.6% tremor suppression | [96] | |
| Voluntary-driven elbow orthosis | ET (1) § | 99.8% tremor suppression | [97] | |
| MMS-based WTSG | Not reported | Not reported | [98] | |
| Myoelectric-controlled orthosis | ET (2); Healthy (4) | Not reported | [99,100,101,102] | |
| Myoelectric-controlled orthosis (ver. 2) | Healthy (1) | 50–80% tremor suppression [104] | [103,104] | |
| BSN-based orthosis | Healthy (6) § | 77% tremor suppression | [105] | |
| Wearable Orthoses: Semi-Active Orthoses | ||||
| Double viscous beam orthosis | Not reported | Not reported | Not reported | [107] |
| MR damper-based orthosis | Not reported | Not reported | [108,109,110,111] | |
| SETS system | Not reported | Not reported | [112] | |
| Electromagnetic brake-based orthosis | Healthy (3) § | 88% tremor suppression | [113] | |
| Pneumatic hand cuff | ET (1) | 30% tremor suppression | [114] | |
| Wearable Orthoses: Passive Orthoses | ||||
| Tremelo * | PD tremor (1) | 85% tremor suppression | Not reported | [115] |
| Steadi-One * | Lab simulation | 85–90% tremor suppression | [116] | |
| Readi-Steadi * | ET (20); Healthy (40) | 50% tremor suppression | [117] | |
| Task-Adjustable Passive Orthosis | PD tremor (1) |
| [119] | |
| Particle Damper | Not reported | Not reported | [120] | |
| Vib-Bracelet | PD tremor (1) § | 85% tremor suppression | [121,122] | |
| Air-dashpot-based orthosis | Healthy (1) ¶ |
| [123] | |
| Assistive Feeding Devices | ||||
| Neater Eater * | Not reported | Not reported | Not reported | [127] |
| Liftware Steady * | ET (15) |
| [129] | |
| Gyenno Spoon * | Not reported | 85% tremor suppression (claimed) | [131] | |
| Gyroscopic Stabilizers | ||||
| GyroGlove * | Not reported | Not reported | Not reported | [132] |
| Haptic Stimulation Systems | ||||
| Emme Watch | Not reported | Not reported | Not reported | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mo, J.; Priefer, R. Medical Devices for Tremor Suppression: Current Status and Future Directions. Biosensors 2021, 11, 99. https://doi.org/10.3390/bios11040099
Mo J, Priefer R. Medical Devices for Tremor Suppression: Current Status and Future Directions. Biosensors. 2021; 11(4):99. https://doi.org/10.3390/bios11040099
Chicago/Turabian StyleMo, Jiancheng, and Ronny Priefer. 2021. "Medical Devices for Tremor Suppression: Current Status and Future Directions" Biosensors 11, no. 4: 99. https://doi.org/10.3390/bios11040099
APA StyleMo, J., & Priefer, R. (2021). Medical Devices for Tremor Suppression: Current Status and Future Directions. Biosensors, 11(4), 99. https://doi.org/10.3390/bios11040099






