1. Introduction
The heart is a vital part of the muscular system, which keeps blood circulating. Heart rhythm and heart rate are two fundamental indicators to assess whether the heart is working orderly [
1]. Heart rhythm is usually rhythmic, and its clinical significance is more important than the heart rate. However, suppose the heart’s four chambers, including the right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV), cannot alternately contract and relax to pump blood through the heart. In that case, the heartbeat will be abnormal in speed and rhythm. The irregular heartbeat typifies arrhythmia and harms the body’s organs and tissues, such as the lungs and brain [
2].
Table 1 lists the most common types of arrhythmia.
Arrhythmias are closely related to electrical irregulars of the pumping heart [
3]. Precisely, the heart’s electrical system controls the heartbeat by the electrical signal. However, when these electrical signals that should have traveled on a fixed path change or the heart tissue changes, arrhythmias occur. For most arrhythmias, the electrocardiogram (ECG) is a handy and visual tool and has the advantages of being simple, fast, and accurate [
4]. ECG can record the heart’s electrical signals and is non-invasive and affordable for ordinary people. Moreover, a normal heartbeat in ECG has four main entities: A P wave, a QRS complex (a combination of the Q wave, R wave and S wave), a T wave, and a U wave, as shown in
Figure 1.
Table 2 shows the cause of generating these waves.
However, ECG is powerless for some particular arrhythmias, such as premature ventricular contraction (PVC), because the patient has a limited time for testing on the ECG machine during a standard ECG recording. PVC is a common arrhythmia initiated in the ventricles and often occurs in repeating patterns, as stated in
Table 3. Specifically, PVC is ubiquitous in healthy individuals and patients and is associated with many diseases. There is a study evaluating the prevalence of frequent PVCs in Guangzhou, China [
5]. Above 1.5% of the residents who received 12-lead ECG had PVCs, and nearly 1/6 of subjects who received 24-h Holter ECG were diagnosed with PVCs. According to the report provided by the American College of Cardiology Electrophysiology Council, PVC is related to left ventricular dysfunction and cardiomyopathy [
6].
Furthermore, PVC is also associated with some disorders, such as ventricular tachycardia (VT), ventricular fibrillation (VF), underlying coronary artery disease, hypertension, and myocardial infarction (MI) [
7,
8,
9]. Because PVC usually causes few or no symptoms, self-diagnosis is not accessible. Most people go to the hospital for help only after they notice severe symptoms.
Since the Holter monitor is a small wearable device and can record the heart’s behavior in the patient’s everyday life, cardiologists usually use the Holter monitor as a medium to obtain long-term ECG and diagnose PVC in clinical practice. However, analyzing so many long-term ECGs takes a lot of time and energy for cardiologists. Therefore, it is crucial to improve the efficiency of cardiologists regarding reliable and automatic searching for PVC from the long-term ECG.
With the continuous advancement of technology for collecting and processing physiological signals in recent years, many researchers have developed various algorithms to detect PVC from the long-term ECG automatically, as summarized in
Table 4. In general, these algorithms are mainly of two types: Morphology-based methods and deep learning-based methods. In these morphology-based methods, extracting features relies on strong expertise, and most researchers have to manually design each feature to ensure that the features are practical. In the deep learning-based methods, extracting features is automatic, which is the most significant difference between the two methods.
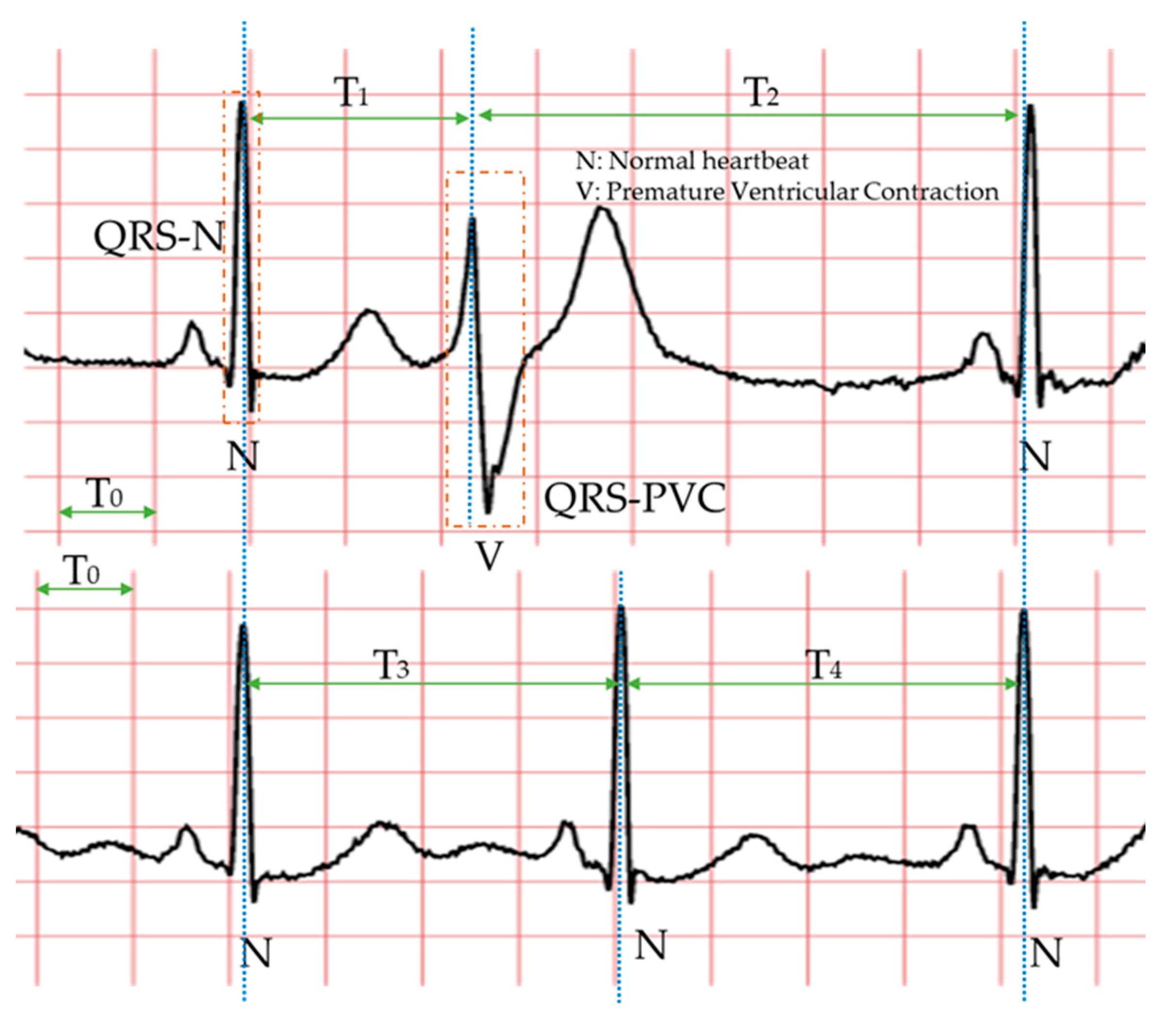
Specifically, the morphology-based method’s core is designing a series of trustworthy features manually with professional knowledge and experience. Compared with the normal heartbeat, PVC’s waveform usually has three main characteristics, as shown in
Figure 2: The QRS complex is broad and has an abnormal morphology (QRS-N and QRS-V); it occurs earlier than expected for the next sinus impulse (T
1 < T
3 < T
2); full compensatory pause (T
1 + T
2 = T
3 + T
4). Therefore, in the morphology-based methods, some classic features mostly come from the time-domain or frequency-domain of the ECG. Due to the continuous development of machine learning algorithms and the advancement of professional knowledge related to signal processing and ECG, most researchers have favored the morphology-based methods. Moreover, these approaches have occupied an unshakable status for a long time.
The signals, collected directly from wearable devices, are always noisy. These noises mainly include baseline wander, 60 Hz power-line noise, electromagnetic interference, 100 Hz interference from fluorescence lights, and motion artifacts. Therefore, many morphology-based methods usually denoise the long-term ECG to extract features more accurately. These popular denoising algorithms are usually based on filters [
10,
11,
12] or wavelet transforms [
13,
14].
Secondly, the morphology-based methods design and extract a series of features according to the expertise related to ECG and signal processing. Adnane et al. proposed a vital feature based on the Haar wavelet transform coefficients [
15]. Du et al. also recommended an essential feature obtained by the chaotic analysis and Lyapunov exponent, named the chaotic feature [
16]. Lek-uthai et al. extracted the four features based on cardiac electrophysiology: R-R interval, pattern of QRS complex, width of QRS complex, and ST-segment (the end of the QRS complex to the beginning of the T wave) level [
17]. Jenny et al. suggested using the independent component analysis (ICA) algorithm to extract features and applying
t-test analysis to evaluate these features [
18]. Nuryani et al. redefine the width and the gradient of the QRS wave and regarded them as features [
19].
Another factor determining the PVC detection method’s performance is the classifier, which classifies samples with these extracted features. The essence of the classifier is a hypothesis or discrete-valued function. There are some popular classifiers used to distinguish regular and PVC beats: Artificial neural networks (ANN) [
20,
21,
22], learning vector quantization neural network (LVQNN) [
23], k-nearest neighbours (k-NN) algorithm [
24,
25], discrete hidden Markov model (DHMM) [
26], support vector machine (SVM) [
27,
28], Bayesian classification algorithms [
29], and random forest (RF) [
30].
In summary, the morphology-based methods include three essential components: Denoising, designed features, and classifiers. Noise reduction is a prerequisite for accurately extracting features. Feature extraction is the core. The classifier directly plays a decisive role in the performance of these methods. Although the morphology-based methods have achieved significant success on this project after many researchers’ efforts, these methods still have some limitations. First, the process of feature extraction relies heavily on preprocessing, such as wavelet transform and QRS detection. Preprocessing undoubtedly increases computational overhead. Further, extracting features is a complex and professional process. In this process, features are not imagined out of thin air but based on knowledge and experience. The features in each literature are often different from person to person, which makes it biased. Therefore, some scholars have proposed deep learning-based methods, which can detect PVC without manually designing features.
Deep learning-based methods are also inseparable from denoising, designed features, and classifiers. Compared with the morphology-based methods, the deep learning-based methods usually do not require professional knowledge and experience related to ECG or signal processing to design features automatically. Although these features are challenging to understand intuitively, these features are useful. That is to say, in most cases, we do not know the meaning of these features, but these features can be used to distinguish between a normal heartbeat and PVC.
Conway et al. used an ANN to detect PVC without manually extracting features [
31]. The ANN’s input corresponds to the 30 points of the QRS complex. Yang et al. proposed an innovative algorithm based on sparse auto-encoder (SAE) to extract features [
32]. SAE is an unsupervised learning algorithm, including two processes of encoding and decoding. The encoding process performs the features’ extraction, and the decoding process ensures the effectiveness of the features. Zhou et al. suggested an approach based on the lead convolutional neural network (LCNN) and long short-term memory (LSTM) network to extract features [
33]. Liu et al. proposed a PVC detection method, which can directly analyze and process the ECG waveform images [
34]. The finetuned Inception V3 model, developed by Google, is the core component of the method [
35].
It is worth noting that feature extraction and classification are closely connected and inseparable. Liu et al. also recommend using a one-dimensional convolutional neural network (1D CNN) to classify the ECG time-series data obtained from ECG waveform images. Zhou et al. reported a PVC detection method based on the recurrent neural network (RNN) [
36], which has natural and inherent advantages in processing time-series signals because of its internal memory. Hoang et al. proposed a PVC detection model deployed in wearable devices [
37]. The model is based on a CNN and can be scalable from 3-lead to 16-lead ECG systems.
The deep learning-based methods alleviate the limitations of morphology-based methods and have the following three advantages. (1) The deep learning-based methods can use specific network structures to extract features, such as the convolutional kernel. This process does not require human intervention. (2) In extracting features, the deep learning-based methods can continuously optimize features to ensure that the features are practical and non-redundant, such as pooling operation. (3) The deep learning-based methods are less affected by preprocessing, such as detecting and locating the QRS waveform.
However, these existing deep learning-based methods are not without flaws. Most of the features extracted by deep learning algorithms are difficult to understand intuitively. The performance of the deep learning-based methods is slightly inferior to the morphology-based methods, as shown in
Table 4. Some deep learning-based methods need to preprocess the ECG. In the literature [
36], much preprocessing is required before the model training, such as resampling, signature detection, and normalization. In addition, the research [
37] takes 2D time-frequency images obtained by wavelet transform on the ECG as the proposed network’s input. No doubt preprocessing increases the computational overhead.
In summary, we can quickly draw the following conclusions according to the above discussion and
Table 4. (1) Most of the methods mentioned in the literature are based on morphology.
Table 4 lists 27 references, of which 22 belong to the morphology-based method, and only five belong to the deep learning-based method. (2) Most researchers prefer to use ANN, KNN, and SVM to identify PVC after completing the feature extraction. Six pieces of literature in
Table 4 use ANN as a classifier. (3) The R-R interval is an excellent feature, which has been recognized by the majority of researchers. Nearly one-third of morphology-based methods have used this feature. (4) In terms of accuracy, sensitivity, and specificity, these three classifiers, FNN, BCM, and SSVM, achieved the best results, respectively. Overall, the morphology-based method’s performances were slightly better than deep learning, due to the expert’s knowledge and experience.
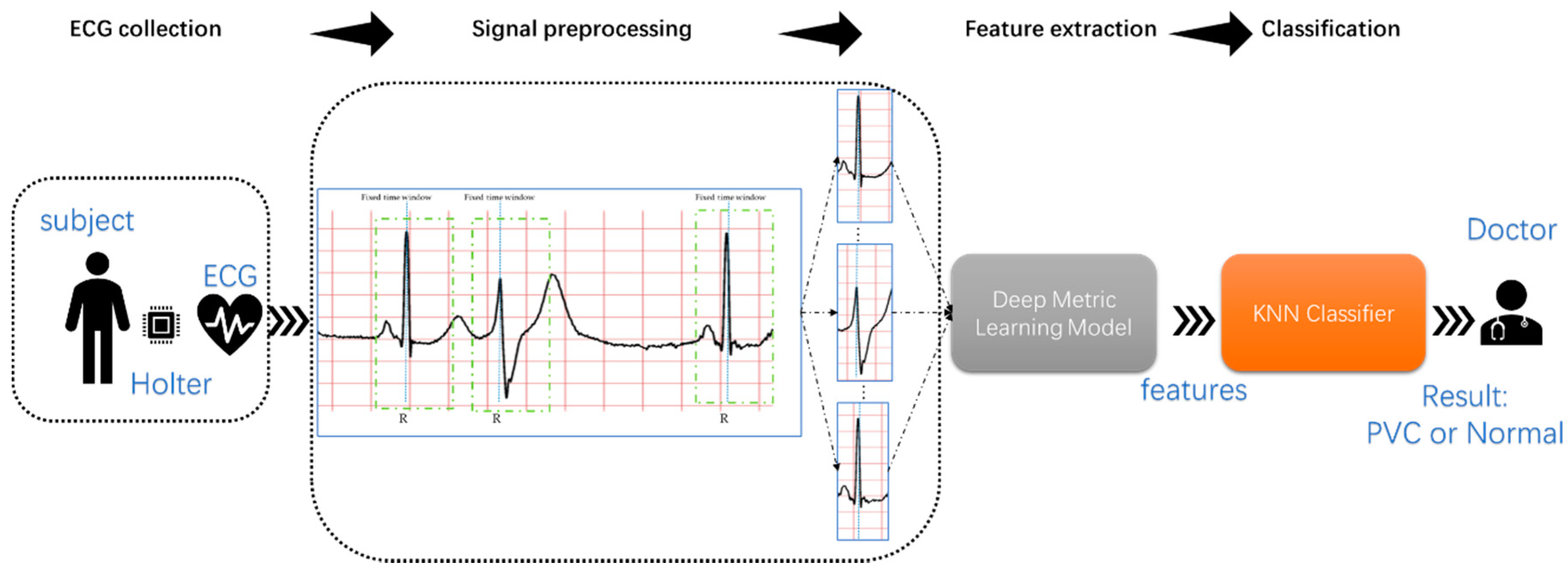
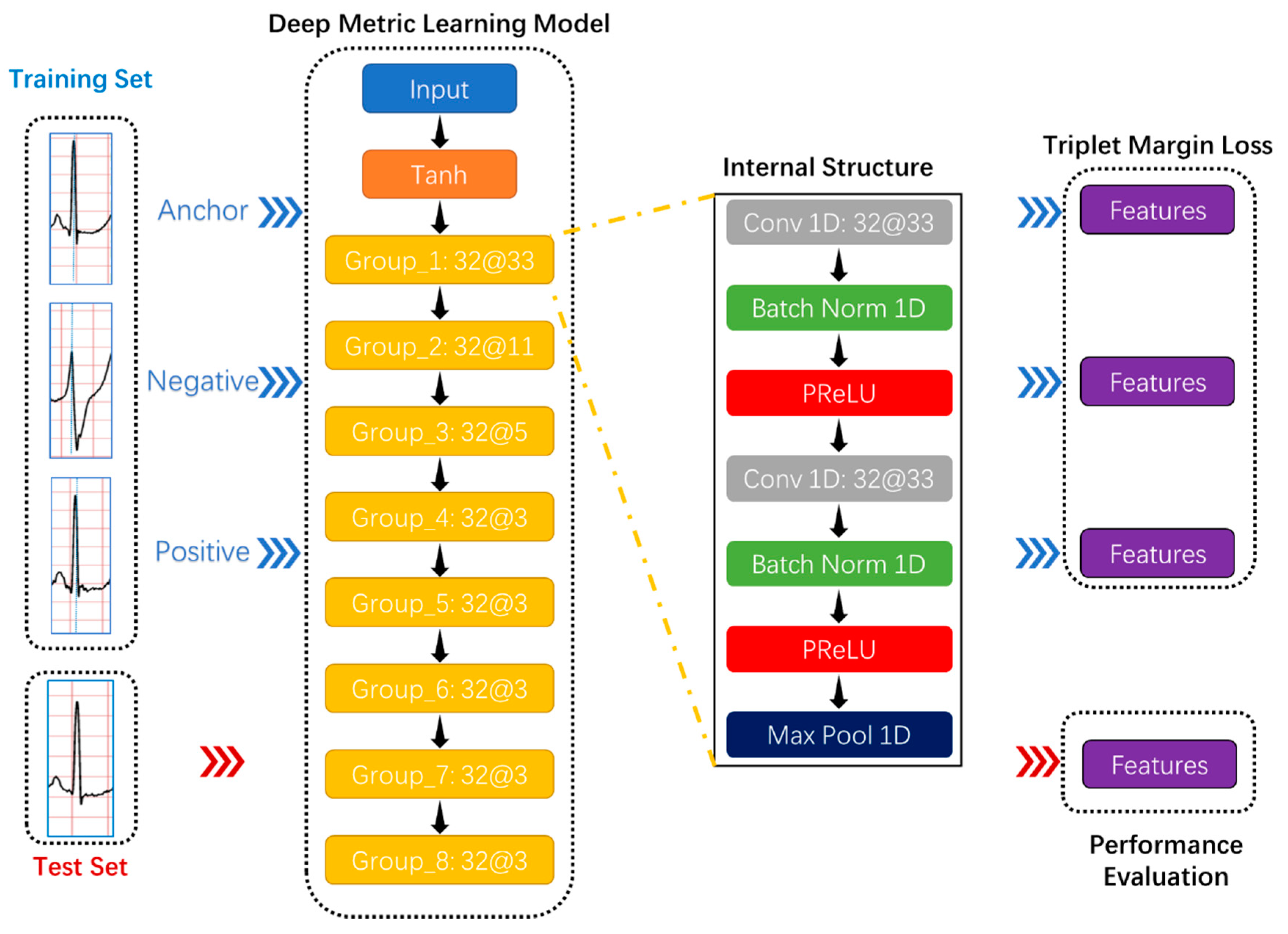
Consider the following: On the one hand, it is easy to understand the features extracted by the morphology-based methods, but feature engineering is the most significant limitation of this method; on the other hand, it is very difficult or even impossible to understand intuitively the features extracted by the deep learning-based methods, but deep learning algorithms can automatically extract and optimize features. This research proposed a novel approach based on deep metric learning and KNN to ensure that the features used to detect PVC can be extracted automatically and understood intuitively.
Specifically, the proposed method introduced deep metric learning into PVC inspection projects for the first time. It is worth mentioning that deep metric learning can automatically extract features, and these features are usually in the high-dimensional embedding space. In this case, the KNN classifier is undoubtedly an optimal choice. Second, the proposed method did not rely on expert knowledge and experience related to ECG, significantly reducing the threshold for studying physiological signals. In theory, the proposed method is suitable for the most physiological signals. Third, to improve the efficiency of detecting PVC from long-term ECG, this method can directly classify heartbeats. Preprocessing, such as denoising, is unnecessary. Finally, clinical ECG from the MIT-BIH (Massachusetts Institute of Technology-Beth Israel Hospital) Arrhythmia Database [
38,
39] evaluated and verified the proposed method’s performance and effectiveness. The following is the remainder’s arrangement:
Section 2 describes the dataset, proposed framework, and evaluation measures;
Section 3 presents and discusses the results;
Section 4 gives the conclusion and directions.
3. Results and Discussion
In this study, the main factors affecting the proposed system’s performance were as follows: The denoising method, the number of features, type of pooling layer, the loss function configuration, and type of classifier. First, denoising is a double-edged sword in the signal preprocessing stage. Denoising can improve the signal’s quality, reducing the difficulty of training a deep metric learning model. However, the signal may also lose some valuable information because of denoising.
Second, as a bridge between the deep metric learning model and classifier, the number of features is an essential hyper-parameter. This value cannot be too large or too small. The greater the number of features, the easier the features become redundant. Conversely, if there are too few features, the less information the features contain cause the classifier’s performance to deteriorate. Third, the type of pooling layer determines how features are summarized and retained and has the effect of de-redundancy. A proper pooling layer can select the most practical features to speed up the deep metric learning model’s training speed.
Fourth, the loss function configuration is the top priority of training the deep metric learning model. The loss function and the miner based on multi-similarity cooperated in the proposed system. In the loss function, the margin should be within a reasonable range. The larger the margin, the more valuable the feature, but the harder it is to train the deep metric learning model. Conversely, the smaller the margin, the easier it is to train the model, but the less practical the features. Finally, the KNN classifier is hugely suitable for processing the deep metric learning model’s features. However, the choice of K value is highly dependent on the distribution of features.
In this section, this study strictly divided the training set and the test set according to
Table 5 and used them in each experiment. Before anything else, we evaluated the necessity of signal denoising. Secondly, we assessed the impact of the number of features on the proposed model. Immediately afterward, we tested pooling layers’ influence on the feature extraction of deep metric learning models. To improve the proposed system’s performance, we have adjusted the loss function and the miner parameters many times. Subsequently, we checked the performance of the KNN classifier and further optimized the classifier. Finally, we compared the proposed method with other research literature on multiple evaluation indicators, such as accuracy, sensitivity, and specificity. We carried out the simulation process on a Linux server with an Nvidia GeForce RTX 2070 GPU.
3.1. Experiment 1: Evaluation of the Necessity for Signal Denoising
In collecting ECG, wearable devices also collect noises. These noises can affect the quality of the signal and even distort the signal. The analog-to-digital conversion chip is a critical hardware component in wearable devices, directly determining the signal quality. Therefore, in the signal acquisition phase, researchers usually improve the sensor’s hardware equipment to suppress noise as much as possible. On the other hand, most scholars use software algorithms in the signal preprocessing stage to remove noise further. However, it is worth mentioning that the noise reduction algorithm inevitably changes the signal more or less. For the metric learning model used in this paper, the convolutional layer can automatically extract useful features and ignore useless information, such as noise. Therefore, the necessity of denoising the signal in the preprocessing stage is worth exploring.
Considering that the data used in this article were all from the MIT-BIH Arrhythmia Database and the method proposed in this paper focused on signal analysis, the denoising methods only involve software algorithms in the signal preprocessing. Expressly, we set up a set of comparative experiments to evaluate the necessity of signal denoising. This comparative experiment first processes and classifies the ECG directly according to the method proposed in this article, without applying any denoising means. Secondly, based on the first experiment, we only added some denoising algorithms in the signal preprocessing stage.
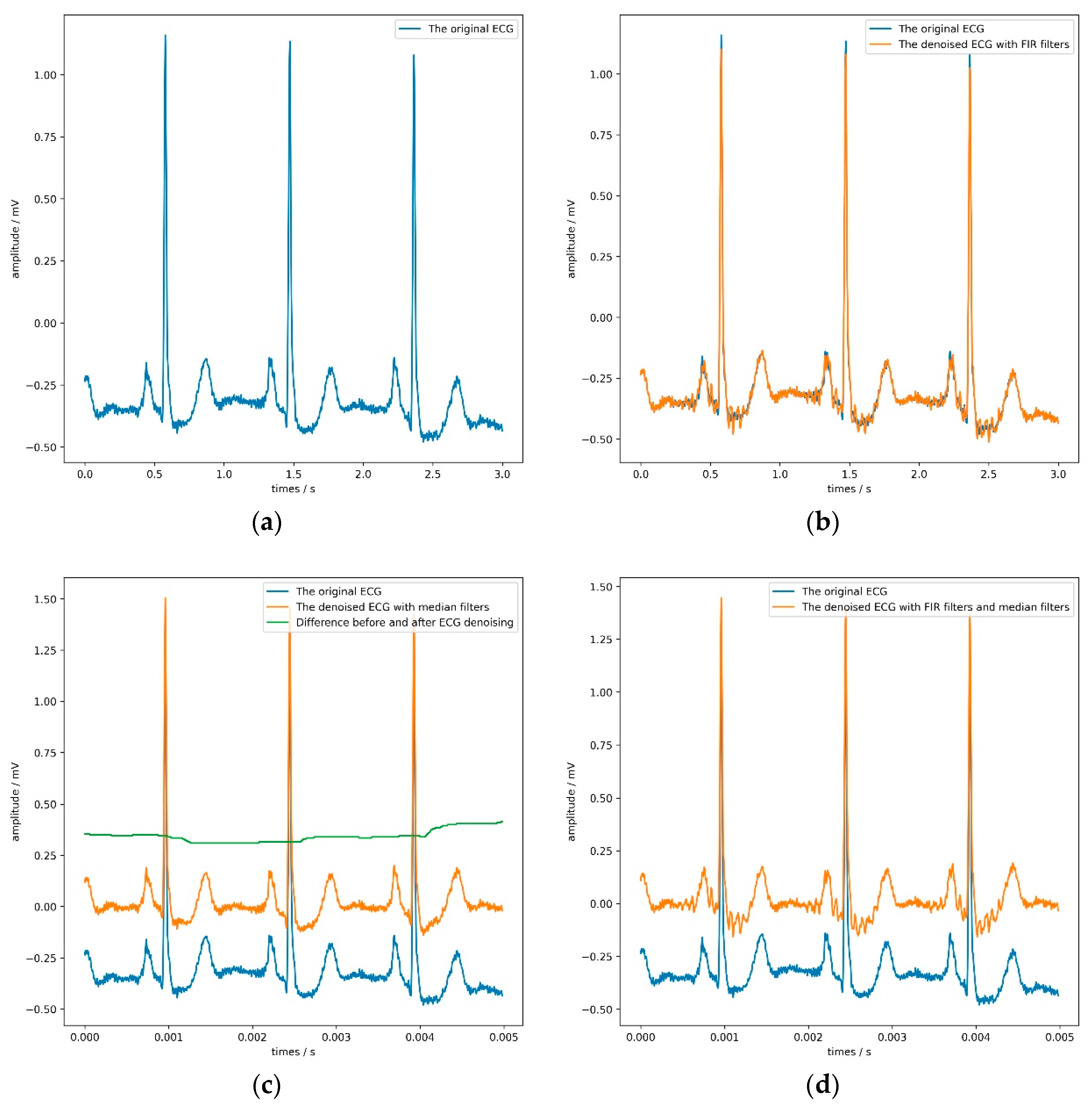
These denoising algorithms include two finite impulse response (FIR) filters with a sampling rate of 1000 Hz and two median filters.
Figure 5 shows the denoising effect of the ECG. The former can filter 60 Hz power-line noise and 100 Hz interference from fluorescence lights, and the latter can remove the baseline of the signal and some noise. It is worth noting that the sizes of these two median filters window are 71 and 215, respectively, which is the same as the setting in literature [
56].
Table 7 and
Table 8 record the parameters and results of the comparative experiment in detail. In
Table 7, the LR, WD, and
K refer to the learning rate, weight decay, and the KNN classifier’s parameter.
It is not difficult to find from
Table 8 that both the FIR filter and the median filter interfered with the model’s judgment to a certain extent, especially when applying both filters at the same time. Adding FIR filters and median filters in the signal preprocessing stage reduces each evaluation index of the model. The median filter can maximize the model’s sensitivity, but the model’s accuracy would drop slightly. According to the model’s overall performance, the most appropriate choice was not to use FIR filters or median filters.
Figure 5 directly confirms this conclusion.
By observing the four sub-pictures in
Figure 5, we can quickly and intuitively discover two phenomena. First of all, the FIR filters could filter out specific frequency components but make the ECG show more obvious glitches simultaneously, which would be counterproductive. Second, the median filters could effectively remove the baseline but slightly change the ECG’s contour, which would be hidden danger for the model’s judgment.
According to
Table 8 another thing worth noting is that the time required to process a half-hour-long ECG using the FIR filter and the median filter was 0.23 and 6.58 s, respectively, in this experiment. If this experiment used the computer hardware equipment with a lower frequency, the time spent on noise removal would become longer.
Considering the actual situation and experimental results, the method proposed in this paper had a particular anti-noise ability. Therefore, denoising was an option in this experiment’s signal preprocessing stage, though not a necessary option. Since this article focused on the classification of electrocardiograms, no more detailed research was done on noise reduction methods.
3.2. Experiment 2: The Choice of the Number of Features
There is no doubt that features are essential and directly determine the performance of the classifier. In theory, practical features should be informative, differentiated, and independent. The deep metric learning model can automatically extract features. In the process of producing high-quality features, the number of features is a critical parameter.
Suppose the number of features is too small. In that case, the deep metric learning model’s training process would be challenging. The acquired features are indistinguishable, and the information contained in the features is not enough to smoothly train the classifier.
On the contrary, too many features are redundant and increase the deep metric learning model’s training time. Further, the excessive features have the following shortcomings for the classifier: Expanding the classifier’s complexity, causing the dimensional disaster, and resulting in ill-posed problems and sparse features problems. These disadvantages eventually lead to a decline in the performance of the classifier.
Considering the above points, we conducted a series of experiments to find the appropriate number of features. We set different values for the number of kernels in the last convolutional layer to adjust the features.
Table 9 provides the detailed results. Moreover, this experiment also adopted the basic configuration in
Table 7.
According to the experimental results in
Table 9, we found that the features extracted by the deep metric learning model could make the KNN classifier perform best when the number of features was 32. Further, the number of features and sensitivity were positively correlated. In other words, the more features, the more confident the proposed system was in PVC predictions. To better analyze these results, we used
t-distributed stochastic neighbor embedding (t-SNE) [
57] to reduce the features’ dimension and then visualize the features in
Figure 6. The t-SNE is a machine learning algorithm for dimension reduction, which is very suitable for reducing high-dimensional data to 2 or 3 dimensions for visualization.
Suppose we used the deep metric learning model to extract only two features. In that case, we could directly draw the features in a two-dimensional coordinate system without dimension reduction by t-SNE. When the number of features was greater than 2, we would use the t-SNE algorithm to reduce the features’ dimensions and display them on a two-dimensional plane. The four sub-images in
Figure 6 show the distribution of different quantity features, extracted from all training data through the deep metric learning model, on a two-dimensional plane.
First, as shown in subfigure (a), the normal heartbeats and PVC were distributed on two parallel straight lines. However, when the first feature was around 2.1 and the second feature was around 0.26, the boundary between the normal heartbeat and PVC was not stark. Secondly, the other three subfigures showed that these features had obvious boundaries on the two-dimensional plane, distinguishing between the normal heartbeats and PVC. Finally, although the results in
Table 9 are not much different, it is better to use the deep metric learning model to extract 32 features after comparing evaluation indicators such as accuracy and sensitivity.
3.3. Experiment 3: Assess the Impact of Pooling Type
In the CNN architecture, most researchers tend to insert a pooling layer in-between consecutive convolutional layers periodically. On the one hand, the pooling layer reduces the number of parameters to learn, avoiding over-fitting, and accelerating the deep metric learning model’s training speed. On the other hand, unlike the convolutional layer that extracts features with precise positioning, the pooling layer summarizes the features generated by a convolution layer, making the deep metric learning model more robust to variations in the position of the features in the input ECG. In other words, the pooling layer has a natural advantage in analyzing heartbeats of different cycles, even if these heartbeats come from different people.
Generally speaking, the core of the pooling layer is a fixed-shaped window. According to a set stride, this window slid overall feature regions and computed a single output for each location. It is worth noting that the way the pooling layer computes the output has no kernel and is deterministic, typically based on the maximum or average value of the features in the pooling window.
Specifically, the output after the max-pooling layer would contain the previous feature map’s most prominent features, which guarantees that each feature used to transmit to the next layer is practical. The average-pooling gives the average of features, taking into account global features in the pooling window. Therefore, in this experiment, we tested these two pooling layers’ performances in feature extraction with the configuration in
Table 7.
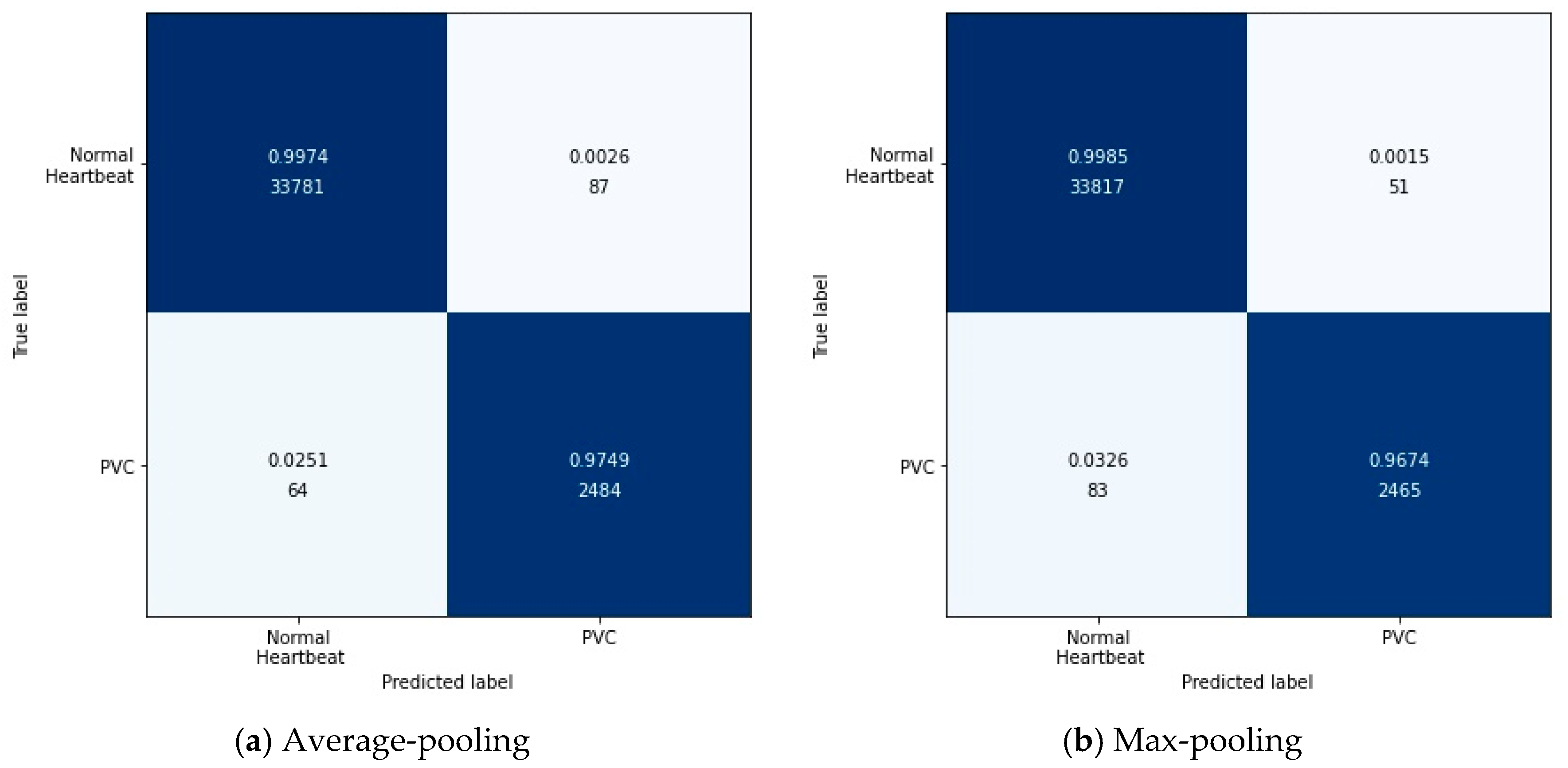
Figure 7 shows the results of this experiment in the form of a confusion matrix.
Table 10 illustrates the detailed results in each evaluation index.
According to
Figure 7, it can be found intuitively that the deep metric learning model with the max-pooling layer misjudged 134 test data, 17 fewer than the model with the average-pooling layer. Although the two models’ performances were similar, the max-pooling layer model was better at predicting normal heartbeats. The model with the average-pooling layer was more confident in predicting PVC, as shown in
Table 10.
In the proposed method, feature extraction’s error mainly came from two aspects: The pooling window size and the feature shift caused by convolutional layer parameters. Generally speaking, the average-pooling operation could reduce the former error to preserve more information in the pooling window. The max-pooling operation can reduce the latter error to focus on the highest intensity information.
Since the loss function was based on cosine similarity, the desired model used to extract features should make the cosine similarity between samples of different classes as small as possible. Suppose the number of features was 2. The PVC and normal heartbeat features should be as close as possible to the two coordinate axes, respectively, in a two-dimensional coordinate system. Under careful consideration, the max-pooling layer was better than the average-pooling layer.
3.4. Experiment 4: Configure the Parameters of the Loss Function and Miner
In the triplet margin loss, the margin is an indispensable parameter that directly affects training the deep metric learning model. The definition of margin is the desired difference between the anchor-positive distance and the anchor-negative distance. Generally speaking, the larger the margin, the higher the quality of the extracted features. However, a large margin makes the model’s training process very unstable, and the loss makes it challenging to approach zero.
Secondly, in this paper, when we trained the model using the triples format’s training data, there were countless triples. However, since some triples met the margin requirements in the loss function, these triples did not contribute to the training model. There is no doubt that blindly and directly using all triples is time-consuming and inefficient for training models.
Fortunately, the miner based on multi-similarity can solve this problem. In this miner, epsilon is an important parameter that controls which triples are selected to train the model. Generally speaking, the larger the epsilon, the more triples are involved in training the model. To maximize the deep metric learning model’s performance, we conducted a series of experiments on margin and epsilon values with the configuration in
Table 7.
Table 11 lists the results for different values of margin and epsilon.
First of all,
Table 11 shows that specificity and margin are negatively correlated, provide epsilon is 0. When the margins were 0.2, 0.4, 0.8, the proposed PVC detection system reached an accuracy of about 99.64% in these three experiments. However, when the margin was 0.1, the proposed PVC detection system performed best in the following indicators: Accuracy, specificity, and positive prediction. Secondly, increasing epsilon made the system’s overall performance worse, especially accuracy and positive prediction.
For the same batch of training data, the greater the margin, the greater the loss. In the case of a fixed learning rate, an enormous loss makes it difficult for the optimizer to find the best point, which leads to a decline in the quality of the extracted features. On the other hand, epsilon determines the number of triples involved in training. The larger the epsilon, the greater the number of triples in the same batch of training data, which undoubtedly increases the computational load. Furthermore, although the larger epsilon increases the number of triples, most of the triples can only produce a minimal loss, which leads to a reduction in the batch loss. A small loss may cause the optimizer to fall into a local optimum. Therefore, according to the experimental results, it is suitable to set the margin and epsilon to 0.1 and 0, respectively.
3.5. Experiment 5: Optimization of KNN Classifier and Comparison with Other Literature
In this article, the KNN classifier is suitable thanks to the spatiality of the features extracted by the deep metric learning model. Nevertheless, the performance of the KNN classifier is very dependent on the
K value. A small
K value is likely to cause overfitting, while an immense
K value is likely to overlook some useful information in the training data. Therefore, it is necessary to test the
K value.
Table 12 lists the performance of the KNN classifier under different
K values.
Overall, the best value of
K was 1, which made the classifier obtain the highest accuracy. Secondly, as the
K value continued to increase, the number of misjudgments by the KNN classifier for PVC was rising since the number of normal heartbeats was much larger than that of PVC. Finally, all the experimental results in
Table 12 confirmed the effectiveness of the PVC detection method proposed in this article. Finally, we compared the proposed method with other literature, as shown in
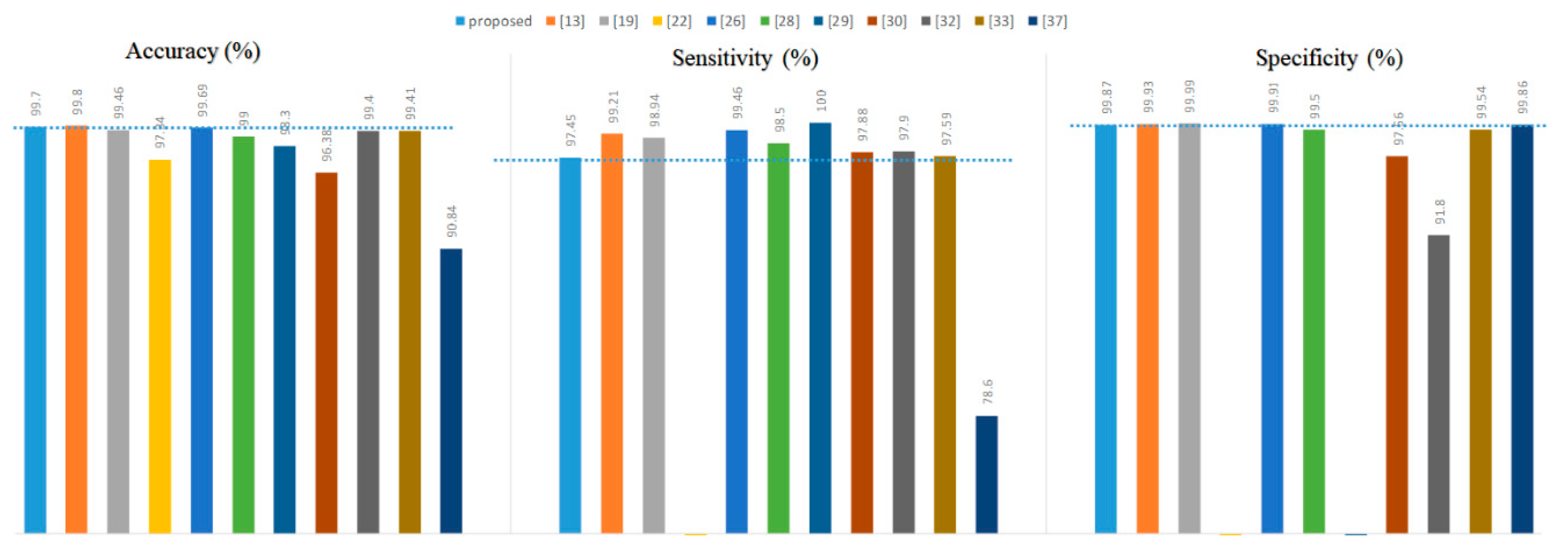
Figure 8.
As a whole, the proposed method was not superior in terms of accuracy, specificity, or sensitivity compared to the references [
13,
19,
22,
26]. However, they used long-term ECGs with no more than ten records from the MIT-BIH Arrhythmia Database to experiment. For example, references [
19,
22] used only six and five patient ECGs, respectively. In addition to this, randomly dividing the training set and test set should attract our attention and vigilance. For example, reference [
26] randomly divided the training set and the test set at a ratio of 2:1. References [
19,
22] are no exception to this problem. It is particularly noteworthy that the training set and the test set were the same in reference [
13], making their results unconvincing.
Second, the proposed method was only 0.1% lower than the reference [
13] in accuracy and outperformed the others. In terms of specificity, the proposed method was also only inferior to reference [
13,
19,
22,
26]. However, this paper’s proposed PVC detection system did not perform exceptionally well in terms of sensitivity.
It is worth mentioning that the results of reference [
28] were based on five-fold cross-validation. Suppose the division scheme of the training and test sets mentioned in this paper were used in reference [
28]. In that case, the accuracy, sensitivity, and specificity of reference [
28] would be 97.6%, 72.1%, and 99.9%, respectively. Therefore, our proposed method was superior to reference [
28,
29] used ten-fold cross-validation to select the classifier. Cross-validation is not suitable in the PVC detection task because it lays a hidden danger for label leakage. Further, our method did not rely on complex preprocessing and was superior to reference [
37] in all metrics. Finally, the proposed system’s sensitivity was similar to that of reference [
30,
32,
33]. Our method was superior to the methods presented in these three literature pieces in terms of accuracy and specificity.
In summary, our method outperformed other studies. Further, applying deep metric learning can automatically extract features and ensure that the features are spatially informative. Finally, the PVC detection system proposed in this paper was highly portable. The system could be directly applied to analyze many other physiological signals.