Activatable Second Near-Infrared Fluorescent Probes: A New Accurate Diagnosis Strategy for Diseases
Abstract
:1. Introduction
2. NIR-II Fluorescent Probes
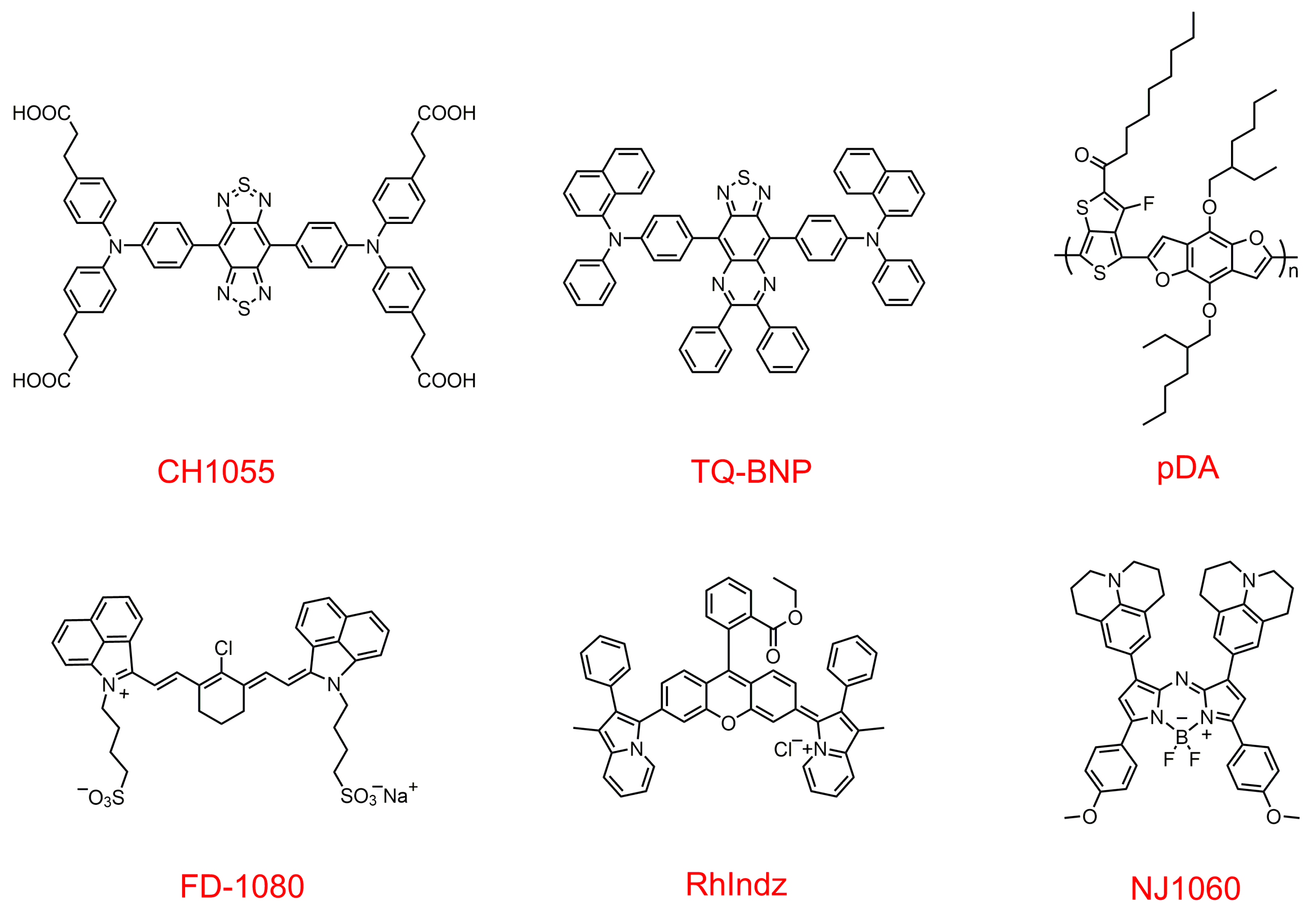
2.1. Organic NIR-II Fluorescent Probes
2.2. Inorganic NIR-II Fluorophores
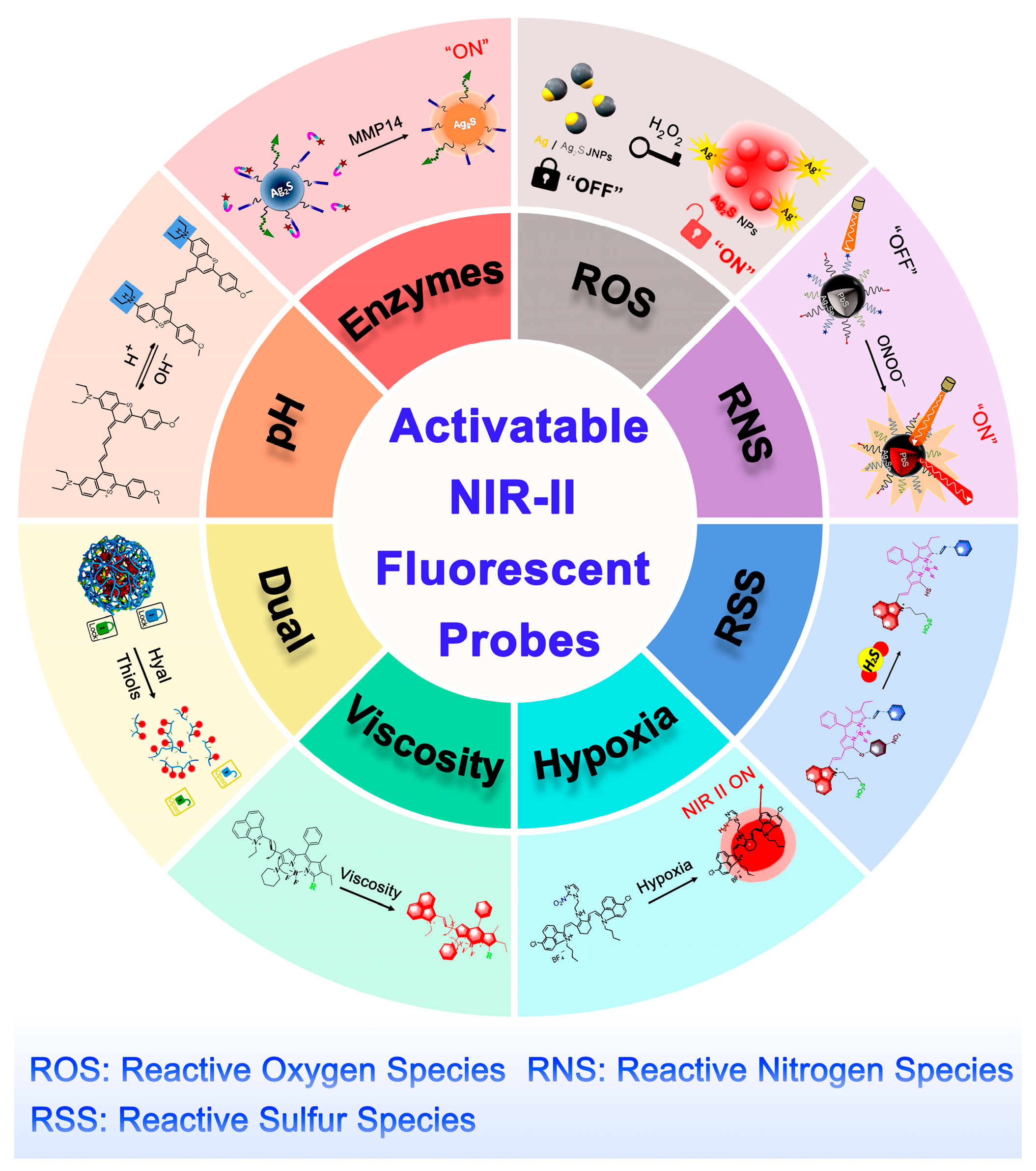
3. Activatable NIR-II Fluorescent Probes
3.1. pH
3.2. Enzymes
3.3. ROS
3.4. RNS
3.5. RSS
3.6. Hypoxia
3.7. Viscosity
3.8. Dual-Responsive
4. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hong, G.; Antaris, A.L.; Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 2017, 1, 1–22. [Google Scholar] [CrossRef]
- Antaris, A.L.; Chen, H.; Cheng, K.; Sun, Y.; Hong, G.; Qu, C.; Diao, S.; Deng, Z.; Hu, X.; Zhang, B.; et al. A small-molecule dye for NIR-II imaging. Nat. Mater. 2016, 15, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Cui, Y.; Levenson, R.M.; Chung, L.W.; Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Mancini, M.C.; Nie, S. Bioimaging: Second window for in vivo imaging. Nat. Nanotechnol. 2009, 4, 710–711. [Google Scholar] [CrossRef] [Green Version]
- Hong, G.; Zou, Y.; Antaris, A.L.; Diao, S.; Wu, D.; Cheng, K.; Zhang, X.; Chen, C.; Liu, B.; He, Y.; et al. Ultrafast fluorescence imaging in vivo with conjugated polymer fluorophores in the second near-infrared window. Nat. Commun. 2014, 5, 4206. [Google Scholar] [CrossRef] [Green Version]
- Sheng, Z.; Guo, B.; Hu, D.; Xu, S.; Wu, W.; Liew, W.H.; Yao, K.; Jiang, J.; Liu, C.; Zheng, H.; et al. Bright aggregation-induced-emission dots for targeted synergetic NIR-II fluorescence and NIR-I photoacoustic imaging of orthotopic brain tumors. Adv. Mater. 2018, 30, 1800766. [Google Scholar] [CrossRef]
- He, S.; Song, J.; Qu, J.; Cheng, Z. Crucial breakthrough of second near-infrared biological window fluorophores: Design and synthesis toward multimodal imaging and theranostics. Chem. Soc. Rev. 2018, 47, 4258–4278. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, S.; Lu, L.; Zhang, Q.; Yu, P.; Fan, Y.; Zhang, F. NIR-II chemiluminescence molecular sensor for in vivo high-contrast inflammation imaging. Angew. Chem. Int. Ed. 2020, 59, 18380–18385. [Google Scholar] [CrossRef]
- Antaris, A.L.; Chen, H.; Diao, S.; Ma, Z.; Zhang, Z.; Zhu, S.; Wang, J.; Lozano, A.X.; Fan, Q.; Chew, L.; et al. A high quantum yield molecule-protein complex fluorophore for near-infrared II imaging. Nat. Commun. 2017, 8, 15269. [Google Scholar] [CrossRef] [Green Version]
- Hu, Z.; Fang, C.; Li, B.; Zhang, Z.; Cao, C.; Cai, M.; Su, S.; Sun, X.; Shi, X.; Li, C.; et al. First-in-human liver-tumour surgery guided by multispectral fluorescence imaging in the visible and near-infrared-I/II windows. Nat. Biomed. Eng. 2020, 4, 259–271. [Google Scholar] [CrossRef]
- Yang, Q.; Hu, Z.; Zhu, S.; Ma, R.; Ma, H.; Ma, Z.; Wan, H.; Zhu, T.; Jiang, Z.; Liu, W.; et al. Donor engineering for NIR-II molecular fluorophores with enhanced fluorescent performance. J. Am. Chem. Soc. 2018, 140, 1715–1724. [Google Scholar] [CrossRef]
- Lei, Z.; Zhang, F. Molecular engineering of NIR-II fluorophores for improved biomedical detection. Angew. Chem. Int. Ed. 2021, 60, 16294–16308. [Google Scholar] [CrossRef]
- Huang, J.; Pu, K. Activatable molecular probes for second near-infrared fluorescence, chemiluminescence, and photoacoustic imaging. Angew. Chem. Int. Ed. 2020, 59, 11717–11731. [Google Scholar] [CrossRef]
- Chen, C.; Tian, R.; Zeng, Y.; Chu, C.; Liu, G. Activatable fluorescence probes for “turn-on” and ratiometric biosensing and bioimaging: From NIR-I to NIR-II. Bioconjug. Chem. 2020, 31, 276–292. [Google Scholar] [CrossRef] [Green Version]
- Zheng, H.; Ma, B.; Shi, Y.; Dai, Q.; Li, D.; Ren, E.; Zhu, J.; Liu, J.; Chen, H.; Yin, Z.; et al. Tumor microenvironment-triggered MoS2@GA-Fe nanoreactor: A self-rolling enhanced chemodynamic therapy and hydrogen sulfide treatment for hepatocellular carcinoma. Chem. Eng. J. 2021, 406, 126888. [Google Scholar] [CrossRef]
- Chu, C.; Yu, J.; Ren, E.; Ou, S.; Zhang, Y.; Wu, Y.; Wu, H.; Zhang, Y.; Zhu, J.; Dai, Q.; et al. Multimodal photoacoustic imaging-guided regression of corneal neovascularization: A non-invasive and safe strategy. Adv. Sci. 2020, 7, 2000346. [Google Scholar] [CrossRef]
- Shi, X.; Zhang, Y.; Tian, Y.; Xu, S.; Ren, E.; Bai, S.; Chen, X.; Chu, C.; Xu, Z.; Liu, G. Multi-responsive bottlebrush-like unimolecules self-assembled nano-riceball for synergistic sono-chemotherapy. Small Methods 2020, 5, 2000416. [Google Scholar] [CrossRef]
- He, T.; Jiang, C.; He, J.; Zhang, Y.; He, G.; Wu, J.; Lin, J.; Zhou, X.; Huang, P. Manganese-dioxide-coating-instructed plasmonic modulation of gold nanorods for activatable duplex-imaging-guided NIR-II photothermal-chemodynamic therapy. Adv. Mater. 2021, 33, 2008540. [Google Scholar] [CrossRef]
- Feng, B.; Hou, B.; Xu, Z.; Saeed, M.; Yu, H.; Li, Y. Self-amplified drug delivery with light-inducible nanocargoes to enhance cancer immunotherapy. Adv. Mater. 2019, 31, 1902960. [Google Scholar] [CrossRef]
- Li, X.; Zheng, B.Y.; Ke, M.R.; Zhang, Y.; Huang, J.D.; Yoon, J. A tumor-pH-responsive supramolecular photosensitizer for activatable photodynamic therapy with minimal in vivo skin phototoxicity. Theranostics 2017, 7, 2746–2756. [Google Scholar] [CrossRef]
- Li, D.; Wang, S.; Lei, Z.; Sun, C.; El-Toni, A.M.; Alhoshan, M.S.; Fan, Y.; Zhang, F. Peroxynitrite activatable NIR-II fluorescent molecular probe for drug-induced hepatotoxicity monitoring. Anal. Chem. 2019, 91, 4771–4779. [Google Scholar] [CrossRef]
- Danko, M.; Hrdlovic, P.; Martinicka, A.; Benda, A.; Cigan, M. Spectral properties of ionic benzotristhiazole based donor-acceptor NLO-phores in polymer matrices and their one- and two-photon cellular imaging ability. Photochem. Photobiol. Sci. 2017, 16, 1832–1844. [Google Scholar] [CrossRef]
- Zhou, C.; Zhang, L.; Sun, T.; Zhang, Y.; Liu, Y.; Gong, M.; Xu, Z.; Du, M.; Liu, Y.; Liu, G.; et al. Activatable NIR-II plasmonic nanotheranostics for efficient photoacoustic imaging and photothermal cancer therapy. Adv. Mater. 2021, 33, 2006532. [Google Scholar] [CrossRef]
- Zhao, M.; Li, B.; Zhang, H.; Zhang, F. Activatable fluorescence sensors for in vivo bio-detection in the second near-infrared window. Chem. Sci. 2020, 12, 3448–3459. [Google Scholar] [CrossRef]
- Zheng, B.D.; Ye, J.; Zhang, X.Q.; Zhang, N.; Xiao, M.T. Recent advances in supramolecular activatable phthalocyanine-based photosensitizers for anti-cancer therapy. Coord. Chem. Rev. 2021, 447, 214155. [Google Scholar] [CrossRef]
- Li, X.; Kolemen, S.; Yoon, J.; Akkaya, E.U. Activatable photosensitizers: Agents for selective photodynamic therapy. Adv. Funct. Mater. 2017, 27, 1604053. [Google Scholar] [CrossRef]
- Li, D.; Wang, X.Z.; Yang, L.F.; Li, S.C.; Hu, Q.Y.; Li, X.; Zheng, B.Y.; Ke, M.R.; Huang, J.D. Size-tunable targeting-triggered nanophotosensitizers based on self-assembly of a phthalocyanine-biotin conjugate for photodynamic therapy. ACS Appl. Mater. Interf. 2019, 11, 36435–36443. [Google Scholar] [CrossRef]
- Li, X.; Yu, S.; Lee, Y.; Guo, T.; Kwon, N.; Lee, D.; Yeom, S.C.; Cho, Y.; Kim, G.; Huang, J.D.; et al. In vivo albumin traps photosensitizer monomers from self-assembled phthalocyanine nanovesicles: A facile and switchable theranostic approach. J. Am. Chem. Soc. 2019, 141, 1366–1372. [Google Scholar] [CrossRef]
- Li, X.; Yu, S.; Lee, D.; Kim, G.; Lee, B.; Cho, Y.; Zheng, B.Y.; Ke, M.R.; Huang, J.D.; Nam, K.T.; et al. Facile supramolecular approach to nucleic-acid-driven activatable nanotheranostics that overcome drawbacks of photodynamic therapy. ACS Nano 2018, 12, 681–688. [Google Scholar] [CrossRef]
- Ding, F.; Zhan, Y.; Lu, X.; Sun, Y. Recent advances in near-infrared II fluorophores for multifunctional biomedical imaging. Chem. Sci. 2018, 9, 4370–4380. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Dong, X.; Li, J.; Wei, J. A short review on NIR-II organic small molecule dyes. Dyes Pigments 2020, 183, 108756. [Google Scholar] [CrossRef]
- Zhou, H.; Xiao, Y.; Hong, X. New NIR-II dyes without a benzobisthiadiazole core. Chin. Chem. Lett. 2018, 29, 1425–1428. [Google Scholar] [CrossRef]
- Zhou, H.; Yi, W.; Li, A.; Wang, B.; Ding, Q.; Xue, L.; Zeng, X.; Feng, Y.; Li, Q.; Wang, T.; et al. Specific small-molecule NIR-II fluorescence imaging of osteosarcoma and lung metastasis. Adv. Healthc. Mater. 2020, 9, 1901224. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Sun, C.; Zebibula, A.; Zhang, H.; Kwok, R.T.K.; Zhao, X.; Xi, W.; Lam, J.W.Y.; Qian, J.; Tang, B.Z. Real-time and high-resolution bioimaging with bright aggregation-induced emission dots in short-wave infrared region. Adv. Mater. 2018, 30, 1706856. [Google Scholar] [CrossRef]
- Samanta, S.; Huang, M.; Li, S.; Yang, Z.; He, Y.; Gu, Z.; Zhang, J.; Zhang, D.; Liu, L.; Qu, J. AIE-active two-photon fluorescent nanoprobe with NIR-II light excitability for highly efficient deep brain vasculature imaging. Theranostics 2021, 11, 2137–2148. [Google Scholar] [CrossRef]
- Li, B.; Lu, L.; Zhao, M.; Lei, Z.; Zhang, F. An efficient 1064 nm NIR-II excitation fluorescent molecular dye for deep-tissue high-resolution dynamic bioimaging. Angew. Chem. Int. Ed. 2018, 57, 7483–7487. [Google Scholar] [CrossRef]
- Bai, L.; Sun, P.; Liu, Y.; Zhang, H.; Hu, W.; Zhang, W.; Liu, Z.; Fan, Q.; Li, L.; Huang, W. Novel aza-BODIPY based small molecular NIR-II fluorophores for in vivo imaging. Chem. Commun. 2019, 55, 10920–10923. [Google Scholar] [CrossRef]
- Rathnamalala, C.S.L.; Gayton, J.N.; Dorris, A.L.; Autry, S.A.; Meador, W.; Hammer, N.I.; Delcamp, J.H.; Scott, C.N. Donor-acceptor-donor NIR II emissive rhodindolizine dye synthesized by C-H bond functionalization. J. Org. Chem. 2019, 84, 13186–13193. [Google Scholar] [CrossRef]
- Zeng, X.; Xue, L.; Chen, D.; Li, S.; Nong, J.; Wang, B.; Tang, L.; Li, Q.; Li, Y.; Deng, Z.; et al. A bright NIR-II fluorescent probe for breast carcinoma imaging and image-guided surgery. Chem. Commun. 2019, 55, 14287–14290. [Google Scholar] [CrossRef]
- Yang, R.Q.; Lou, K.L.; Wang, P.Y.; Gao, Y.Y.; Zhang, Y.Q.; Chen, M.; Huang, W.H.; Zhang, G.J. Surgical navigation for malignancies guided by near-infrared-II fluorescence imaging. Small Methods 2021, 5, 2001066. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, Q.; Huang, S.; Ma, D. Recent advances of near infrared inorganic fluorescent probes for biomedical applications. J. Mater. Chem. B 2020, 8, 7856–7879. [Google Scholar] [CrossRef]
- Zhu, S.; Tian, R.; Antaris, A.L.; Chen, X.; Dai, H. Near-infrared-II molecular dyes for cancer imaging and surgery. Adv. Mater. 2019, 31, 1900321. [Google Scholar] [CrossRef]
- Li, C.; Zhang, Y.; Wang, M.; Zhang, Y.; Chen, G.; Li, L.; Wu, D.; Wang, Q. In vivo real-time visualization of tissue blood flow and angiogenesis using Ag2S quantum dots in the NIR-II window. Biomaterials 2014, 35, 393–400. [Google Scholar] [CrossRef]
- Takeuchi, T.; Iizumi, Y.; Yudasaka, M.; Kizaka-Kondoh, S.; Okazaki, T. Characterization and biodistribution analysis of oxygen-doped single-walled carbon nanotubes used as in vivo fluorescence imaging probes. Bioconjug. Chem. 2019, 30, 1323–1330. [Google Scholar] [CrossRef]
- Ghosh, D.; Bagley, A.F.; Na, Y.J.; Birrer, M.J.; Bhatia, S.N.; Belcher, A.M. Deep, noninvasive imaging and surgical guidance of submillimeter tumors using targeted M13-stabilized single-walled carbon nanotubes. Proc. Natl. Acad. Sci. USA 2014, 111, 13948–13953. [Google Scholar] [CrossRef] [Green Version]
- Lei, X.; Li, R.; Tu, D.; Shang, X.; Liu, Y.; You, W.; Sun, C.; Zhang, F.; Chen, X. Intense near-infrared-II luminescence from NaCeF4:Er/Yb nanoprobes for in vitro bioassay and in vivo bioimaging. Chem. Sci. 2018, 9, 4682–4688. [Google Scholar] [CrossRef] [Green Version]
- Xue, Z.; Zeng, S.; Hao, J. Non-invasive through-skull brain vascular imaging and small tumor diagnosis based on NIR-II emissive lanthanide nanoprobes beyond 1500nm. Biomaterials 2018, 171, 153–163. [Google Scholar] [CrossRef]
- Su, M.; Dai, Q.; Chen, C.; Zeng, Y.; Chu, C.; Liu, G. Nano-medicine for thrombosis: A precise diagnosis and treatment strategy. Nano-Micro Lett. 2020, 12, 96. [Google Scholar] [CrossRef] [Green Version]
- Ke, M.R.; Chen, S.F.; Peng, X.H.; Zheng, Q.F.; Zheng, B.Y.; Yeh, C.K.; Huang, J.D. A tumor-targeted activatable phthalocyanine-tetrapeptide-doxorubicin conjugate for synergistic chemo-photodynamic therapy. Eur. J. Med. Chem. 2017, 127, 200–209. [Google Scholar] [CrossRef]
- Zhou, K.; Liu, H.; Zhang, S.; Huang, X.; Wang, Y.; Huang, G.; Sumer, B.D.; Gao, J. Multicolored pH-tunable and activatable fluorescence nanoplatform responsive to physiologic pH stimuli. J. Am. Chem. Soc. 2012, 134, 7803–7811. [Google Scholar] [CrossRef] [Green Version]
- Yue, Y.; Huo, F.; Lee, S.; Yin, C.; Yoon, J. A review: The trend of progress about pH probes in cell application in recent years. Analyst 2017, 142, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Yang, N.; Wang, X.; Zhao, Q.; Chen, Q.; Liu, Z.; Cheng, L. Tumor microenvironment-responsive intelligent nanoplatforms for cancer theranostics. Nano Today 2020, 32, 100851. [Google Scholar] [CrossRef]
- Raish, M.; Shahid, M.; Bin Jardan, Y.A.; Ansari, M.A.; Alkharfy, K.M.; Ahad, A.; Abdelrahman, I.A.; Ahmad, A.; Al-Jenoobi, F.I. Gastroprotective effect of sinapic acid on ethanol-induced gastric ulcers in rats: Involvement of Nrf2/HO-1 and NF-kappaB signaling and antiapoptotic role. Front. Pharmacol. 2021, 12, 622815. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Fan, Y.; Li, D.; Sun, C.; Lei, Z.; Lu, L.; Wang, T.; Zhang, F. Anti-quenching NIR-II molecular fluorophores for in vivo high-contrast imaging and pH sensing. Nat. Commun. 2019, 10, 1058. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wang, J.; Lei, Z.; Lu, L.; Wang, S.; Zhang, H.; Li, B.; Zhang, F. NIR-II pH sensor with a FRET adjustable transition point for in situ dynamic tumor microenvironment visualization. Angew. Chem. Int. Ed. 2021, 60, 5091–5095. [Google Scholar] [CrossRef]
- Wu, J.; You, L.; Chaudhry, S.T.; He, J.; Cheng, J.X.; Mei, J. Ambient oxygen-doped conjugated polymer for pH-activatable aggregation-enhanced photoacoustic imaging in the second near-infrared window. Anal. Chem. 2021, 93, 3189–3195. [Google Scholar] [CrossRef]
- Ren, E.; Chu, C.; Zhang, Y.; Wang, J.; Pang, X.; Lin, X.; Liu, C.; Shi, X.; Dai, Q.; Lv, P.; et al. Mimovirus vesicle-based biological orthogonal reaction for cancer diagnosis. Small Methods 2020, 4, 2000291. [Google Scholar] [CrossRef]
- Li, J.; Zheng, L.; Li, C.; Xiao, Y.; Liu, J.; Wu, S.; Zhang, B. Mannose modified zwitterionic polyester-conjugated second near-infrared organic fluorophore for targeted photothermal therapy. Biomater. Sci. 2021, 9, 4648–4661. [Google Scholar] [CrossRef]
- Bai, S.; Zhang, Y.; Li, D.; Shi, X.; Lin, G.; Liu, G. Gain an advantage from both sides: Smart size-shrinkable drug delivery nanosystems for high accumulation and deep penetration. Nano Today 2021, 36, 101038. [Google Scholar] [CrossRef]
- Wang, X.; Li, C.; Qian, J.; Lv, X.; Li, H.; Zou, J.; Zhang, J.; Meng, X.; Liu, H.; Qian, Y.; et al. NIR-II responsive hollow magnetite nanoclusters for targeted magnetic resonance imaging-guided photothermal/chemo-therapy and chemodynamic therapy. Small 2021, 17, 2100794. [Google Scholar] [CrossRef]
- Zhao, P.H.; Ma, S.T.; Hu, J.Q.; Zheng, B.Y.; Ke, M.R.; Huang, J.D. Artesunate-based multifunctional nanoplatform for photothermal/photoinduced thermodynamic synergistic anticancer therapy. ACS Appl. Bio. Mater. 2020, 3, 7876–7885. [Google Scholar] [CrossRef]
- Ling, S.; Yang, X.; Li, C.; Zhang, Y.; Yang, H.; Chen, G.; Wang, Q. Tumor microenvironment-activated NIR-II nanotheranostic system for precise diagnosis and treatment of peritoneal metastasis. Angew. Chem. Int. Ed. 2020, 59, 7219–7223. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, X.; Liu, D.; Chen, B.; Ge, X.; Gao, S.; Song, J. Self-assembled Ag2S-QD vesicles for in situ responsive NIR-II fluorescence imaging-guided photothermal cancer therapy. Adv. Optl. Mater. 2021, 9, 2100233. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Chu, C.; Zhou, Z.; Chen, B.; Pang, X.; Lin, G.; Lin, H.; Guo, Y.; Ren, E.; et al. Genetically engineered magnetic nanocages for cancer magneto-catalytic theranostics. Nat. Commun. 2020, 11, 5421. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, J.; Liu, J.; Lin, G.; Xie, F.; Pang, X.; Pei, Y.; Cheng, Y.; Zhang, Y.; Lin, Z.; et al. Oxidative stress-driven DR5 upregulation restores TRAIL/Apo2L sensitivity induced by iron oxide nanoparticles in colorectal cancer. Biomaterials 2020, 233, 119753. [Google Scholar] [CrossRef]
- Gong, L.; Shan, X.; Zhao, X.H.; Tang, L.; Zhang, X.B. Activatable NIR-II fluorescent probes applied in biomedicine: Progress and perspectives. ChemMedChem 2021, 16, 2426–2440. [Google Scholar] [CrossRef]
- Suzuki, H.; Ohto, U.; Higaki, K.; Mena-Barragan, T.; Aguilar-Moncayo, M.; Ortiz Mellet, C.; Nanba, E.; Garcia Fernandez, J.M.; Suzuki, Y.; Shimizu, T. Structural basis of pharmacological chaperoning for human beta-galactosidase. J. Biol. Chem. 2014, 289, 14560–14568. [Google Scholar] [CrossRef] [Green Version]
- Ohto, U.; Usui, K.; Ochi, T.; Yuki, K.; Satow, Y.; Shimizu, T. Crystal structure of human beta-galactosidase: Structural basis of Gm1 gangliosidosis and morquio B diseases. J. Biol. Chem. 2012, 287, 1801–1812. [Google Scholar] [CrossRef] [Green Version]
- Asanuma, D.; Sakabe, M.; Kamiya, M.; Yamamoto, K.; Hiratake, J.; Ogawa, M.; Kosaka, N.; Choyke, P.L.; Nagano, T.; Kobayashi, H.; et al. Sensitive beta-galactosidase-targeting fluorescence probe for visualizing small peritoneal metastatic tumours in vivo. Nat. Commun. 2015, 6, 6463. [Google Scholar] [CrossRef] [Green Version]
- Kim, E.J.; Kumar, R.; Sharma, A.; Yoon, B.; Kim, H.M.; Lee, H.; Hong, K.S.; Kim, J.S. In vivo imaging of beta-galactosidase stimulated activity in hepatocellular carcinoma using ligand-targeted fluorescent probe. Biomaterials 2017, 122, 83–90. [Google Scholar] [CrossRef]
- Zhen, X.; Zhang, J.; Huang, J.; Xie, C.; Miao, Q.; Pu, K. Macrotheranostic probe with disease-activated near-infrared fluorescence, photoacoustic, and photothermal signals for imaging-guided therapy. Angew. Chem. Int. Ed. 2018, 57, 7804–7808. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.A.; Pan, H.; Wang, Z.; Gao, J.; Tan, J.; Ouyang, Z.; Guo, W.; Gu, X. Imaging of ovarian cancers using enzyme activatable probes with second near-infrared window emission. Chem. Commun. 2020, 56, 2731–2734. [Google Scholar] [CrossRef] [PubMed]
- Gimeno, A.Z.; Santana, A.; Jimenez, A.; Parra, A.D.; Nicolas, D.; Paz, C.; Diaz, F.; Medina, C.; Diaz, L.; Quintero, E. Up-regulation of gelatinases in the colorectal adenoma-carcinoma sequence. Eur. J. Cancer 2006, 42, 3246–3252. [Google Scholar] [CrossRef] [PubMed]
- Xiang, X.; Zhao, X.; Qu, H.; Li, D.; Yang, D.; Pu, J.; Mei, H.; Zhao, J.; Huang, K.; Zheng, L.; et al. Hepatocyte nuclear factor 4 alpha promotes the invasion, metastasis and angiogenesis of neuroblastoma cells via targeting matrix metalloproteinase 14. Cancer Lett. 2015, 359, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.R.; Binatti, A.; Pantile, M.; Coppe, A.; Mazzocco, K.; Longo, L.; Capasso, M.; Lasorsa, V.A.; Luksch, R.; Bortoluzzi, S.; et al. Somatic mutations in specific and connected subpathways are associated with short neuroblastoma patients’ survival and indicate proteins targetable at onset of disease. Int. J. Cancer 2018, 143, 2525–2536. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Song, J.; Lee, W.; Ryu, Y.M.; Jung, Y.; Kim, S.Y.; Kim, K.; Hong, S.C.; Myung, S.J.; Kim, S. Cancer-microenvironment-sensitive activatable quantum dot probe in the second near-infrared window. Nano Lett. 2017, 17, 1378–1386. [Google Scholar] [CrossRef]
- Zhan, Y.; Ling, S.; Huang, H.; Zhang, Y.; Chen, G.; Huang, S.; Li, C.; Guo, W.; Wang, Q. Rapid unperturbed-tissue analysis for intraoperative cancer diagnosis using an enzyme-activated NIR-II nanoprobe. Angew. Chem. Int. Ed. 2021, 60, 2637–2642. [Google Scholar] [CrossRef]
- Li, D.; Hu, Q.Y.; Wang, X.Z.; Li, X.; Hu, J.Q.; Zheng, B.Y.; Ke, M.R.; Huang, J.D. A non-aggregated silicon(IV) phthalocyanine-lactose conjugate for photodynamic therapy. Bioorg. Med. Chem. Lett. 2020, 30, 127164. [Google Scholar] [CrossRef]
- Zhao, Y.Y.; Chen, J.Y.; Hu, J.Q.; Zhang, L.; Lin, A.L.; Wang, R.; Zheng, B.Y.; Ke, M.R.; Li, X.; Huang, J.D. The substituted zinc(II) phthalocyanines using “sulfur bridge” as the linkages. synthesis, red-shifted spectroscopic properties and structure-inherent targeted photodynamic activities. Dyes Pigments 2021, 189, 109270. [Google Scholar] [CrossRef]
- He, X.J.; Chen, H.; Xu, C.C.; Fan, J.Y.; Xu, W.; Li, Y.H.; Deng, H.; Shen, J.L. Ratiometric and colorimetric fluorescent probe for hypochlorite monitor and application for bioimaging in living cells, bacteria and zebrafish. J. Hazard. Mater. 2020, 388, 122029. [Google Scholar] [CrossRef]
- Ye, J.; Li, Z.; Fu, Q.; Li, Q.; Zhang, X.; Su, L.; Yang, H.; Song, J. Quantitative photoacoustic diagnosis and precise treatment of inflammation in vivo using activatable theranostic nanoprobe. Adv. Funct. Mater. 2020, 30, 2001771. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Su, L.; Ge, X.; Ye, J.; Zhao, C.; He, Y.; Yang, H.; Song, J.; Duan, H. Plasmonic-fluorescent janus Ag/Ag2S nanoparticles for in situ H2O2-activated NIR-II fluorescence imaging. Nano Lett. 2021, 21, 2625–2633. [Google Scholar] [CrossRef]
- Khojah, H.M.; Ahmed, S.; Abdel-Rahman, M.S.; Hamza, A.B. Reactive oxygen and nitrogen species in patients with rheumatoid arthritis as potential biomarkers for disease activity and the role of antioxidants. Free Radic. Biol. Med. 2016, 97, 285–291. [Google Scholar] [CrossRef]
- Sayre, L.M.; Perry, G.; Smith, M.A. Oxidative stress and neurotoxicity. Chem. Res. Toxicol. 2008, 21, 172–188. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Li, P.; Ding, Q.; Wu, C.; Zhang, W.; Tang, B. Observation of acetylcholinesterase in stress-induced depression phenotypes by two-photon fluorescence imaging in the mouse brain. J. Am. Chem. Soc. 2019, 141, 2061–2068. [Google Scholar] [CrossRef]
- Oka, T.; Yamashita, S.; Midorikawa, M.; Saiki, S.; Muroya, Y.; Kamibayashi, M.; Yamashita, M.; Anzai, K.; Katsumura, Y. Spin-trapping reactions of a novel gauchetype radical trapper G-CYPMPO. Anal. Chem. 2011, 83, 9600–9604. [Google Scholar] [CrossRef]
- Sun, W.; Guo, S.; Hu, C.; Fan, J.; Peng, X. Recent development of chemosensors based on cyanine platforms. Chem. Rev. 2016, 116, 7768–7817. [Google Scholar] [CrossRef]
- Feng, W.; Zhang, Y.; Li, Z.; Zhai, S.; Lv, W.; Liu, Z. Lighting up NIR-II fluorescence in vivo: An activable probe for noninvasive hydroxyl radical imaging. Anal. Chem. 2019, 91, 15757–15762. [Google Scholar] [CrossRef]
- Zhu, H.; Fan, J.; Wang, J.; Mu, H.; Peng, X. An “enhanced PET”-based fluorescent probe with ultrasensitivity for imaging basal and elesclomol-induced HClO in cancer cells. J. Am. Chem. Soc. 2014, 136, 12820–12823. [Google Scholar] [CrossRef]
- Wu, L.; Wu, I.C.; DuFort, C.C.; Carlson, M.A.; Wu, X.; Chen, L.; Kuo, C.T.; Qin, Y.; Yu, J.; Hingorani, S.R.; et al. Photostable ratiometric pdot probe for in vitro and in vivo imaging of hypochlorous acid. J. Am. Chem. Soc. 2017, 139, 6911–6918. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Rao, J.; Pu, K. Recent progress on semiconducting polymer nanoparticles for molecular imaging and cancer phototherapy. Biomaterials 2018, 155, 217–235. [Google Scholar] [CrossRef]
- Wang, S.; Liu, L.; Fan, Y.; El-Toni, A.M.; Alhoshan, M.S.; Li, D.; Zhang, F. In vivo high-resolution ratiometric fluorescence imaging of inflammation using NIR-II nanoprobes with 1550 nm emission. Nano Lett. 2019, 19, 2418–2427. [Google Scholar] [CrossRef]
- Miao, Q.; Xie, C.; Zhen, X.; Lyu, Y.; Duan, H.; Liu, X.; Jokerst, J.V.; Pu, K. Molecular afterglow imaging with bright, biodegradable polymer nanoparticles. Nat. Biotechnol. 2017, 35, 1102–1110. [Google Scholar] [CrossRef]
- Ge, X.; Lou, Y.; Su, L.; Chen, B.; Guo, Z.; Gao, S.; Zhang, W.; Chen, T.; Song, J.; Yang, H. Single wavelength laser excitation ratiometric NIR-II fluorescent probe for molecule imaging in vivo. Anal. Chem. 2020, 92, 6111–6120. [Google Scholar] [CrossRef]
- Tang, Y.; Li, Y.; Lu, X.; Hu, X.; Zhao, H.; Hu, W.; Lu, F.; Fan, Q.; Huang, W. Bio-erasable intermolecular donor–acceptor interaction of organic semiconducting nanoprobes for activatable NIR-II fluorescence imaging. Adv. Funct. Mater. 2019, 29, 1807376. [Google Scholar] [CrossRef]
- Kwon, N.; Kim, D.; Swamy, K.M.K.; Yoon, J. Metal-coordinated fluorescent and luminescent probes for reactive oxygen species (ROS) and reactive nitrogen species (RNS). Coord. Chem. Rev. 2021, 427, 213581. [Google Scholar] [CrossRef]
- Tang, Y.; Pei, F.; Lu, X.; Fan, Q.; Huang, W. Recent advances on activatable NIR-II fluorescence probes for biomedical imaging. Adv. Opt. Mater. 2019, 7, 1900917. [Google Scholar] [CrossRef]
- Szabo, C.; Ischiropoulos, H.; Radi, R. Peroxynitrite: Biochemistry, pathophysiology and development of therapeutics. Nat. Rev. Drug Discov. 2007, 6, 662–680. [Google Scholar] [CrossRef]
- Ai, X.; Wang, Z.; Cheong, H.; Wang, Y.; Zhang, R.; Lin, J.; Zheng, Y.; Gao, M.; Xing, B. Multispectral optoacoustic imaging of dynamic redox correlation and pathophysiological progression utilizing upconversion nanoprobes. Nat. Commun. 2019, 10, 1087. [Google Scholar] [CrossRef] [Green Version]
- Zhao, M.; Li, B.; Wu, Y.; He, H.; Zhu, X.; Zhang, H.; Dou, C.; Feng, L.; Fan, Y.; Zhang, F. A tumor-microenvironment-responsive lanthanide-cyanine FRET sensor for NIR-II luminescence-lifetime in situ imaging of hepatocellular carcinoma. Adv. Mater. 2020, 32, 2001172. [Google Scholar] [CrossRef]
- Zhang, J.; Zhen, X.; Upputuri, P.K.; Pramanik, M.; Chen, P.; Pu, K. Activatable photoacoustic nanoprobes for in vivo ratiometric imaging of peroxynitrite. Adv. Mater. 2017, 29, 1604764. [Google Scholar] [CrossRef] [PubMed]
- Uteshev, V.V. Allosteric modulation of nicotinic acetylcholine receptors: The concept and therapeutic trends. Curr. Pharm. Des. 2016, 22, 1986–1997. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tao, R.R.; Hong, L.J.; Cheng, J.; Jiang, Q.; Lu, Y.M.; Liao, M.H.; Ye, W.F.; Lu, N.N.; Han, F.; et al. Visualizing peroxynitrite fluxes in endothelial cells reveals the dynamic progression of brain vascular injury. J. Am. Chem. Soc. 2015, 137, 12296–12303. [Google Scholar] [CrossRef] [PubMed]
- Adibhatla, R.M.; Hatcher, J.F. Lipid oxidation and peroxidation in CNS health and disease: From molecular mechanisms to therapeutic opportunities. Antioxid. Redox Signal. 2010, 12, 125–169. [Google Scholar] [CrossRef]
- Li, C.; Li, W.; Liu, H.; Zhang, Y.; Chen, G.; Li, Z.; Wang, Q. An activatable NIR-II nanoprobe for in vivo early real-time diagnosis of traumatic brain injury. Angew. Chem. Int. Ed. 2020, 59, 247–252. [Google Scholar] [CrossRef]
- Zhang, K.; Tu, M.; Gao, W.; Cai, X.; Song, F.; Chen, Z.; Zhang, Q.; Wang, J.; Jin, C.; Shi, J.; et al. Hollow prussian blue nanozymes drive neuroprotection against ischemic stroke via attenuating oxidative stress, counteracting inflammation, and suppressing cell apoptosis. Nano Lett. 2019, 19, 2812–2823. [Google Scholar] [CrossRef]
- Li, S.; Jiang, D.; Ehlerding, E.B.; Rosenkrans, Z.T.; Engle, J.W.; Wang, Y.; Liu, H.; Ni, D.; Cai, W. Intrathecal administration of nanoclusters for protecting neurons against oxidative stress in cerebral ischemia/reperfusion injury. ACS Nano 2019, 13, 13382–13389. [Google Scholar] [CrossRef]
- Liu, Y.; Ai, K.; Ji, X.; Askhatova, D.; Du, R.; Lu, L.; Shi, J. Comprehensive insights into the multi-antioxidative mechanisms of melanin nanoparticles and their application to protect brain from injury in ischemic stroke. J. Am. Chem. Soc. 2017, 139, 856–862. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Wang, Z.; Huang, H.; Ling, S.; Zhang, R.; Zhang, Y.; Chen, G.; Li, C.; Wang, Q. A targeted activatable NIR-IIb nanoprobe for highly sensitive detection of ischemic stroke in a photothrombotic stroke model. Adv. Healthc. Mater. 2021, 10, 2001544. [Google Scholar] [CrossRef]
- Wang, R.; Zhou, L.; Wang, W.; Li, X.; Zhang, F. In vivo gastrointestinal drug-release monitoring through second near-infrared window fluorescent bioimaging with orally delivered microcarriers. Nat. Commun. 2017, 8, 14702. [Google Scholar] [CrossRef]
- Xie, C.; Zhen, X.; Lyu, Y.; Pu, K. Nanoparticle regrowth enhances photoacoustic signals of semiconducting macromolecular probe for in vivo imaging. Adv. Mater. 2017, 29, 1703693. [Google Scholar] [CrossRef]
- Peng, J.; Samanta, A.; Zeng, X.; Han, S.; Wang, L.; Su, D.; Loong, D.T.; Kang, N.Y.; Park, S.J.; All, A.H.; et al. Real-time in vivo hepatotoxicity monitoring through chromophore-conjugated photon-upconverting nanoprobes. Angew. Chem. Int. Ed. 2017, 56, 4165–4169. [Google Scholar] [CrossRef]
- Shuhendler, A.J.; Pu, K.; Cui, L.; Uetrecht, J.P.; Rao, J. Real-time imaging of oxidative and nitrosative stress in the liver of live animals for drug-toxicity testing. Nat. Biotechnol. 2014, 32, 373380. [Google Scholar] [CrossRef]
- Iverson, N.M.; Barone, P.W.; Shandell, M.; Trudel, L.J.; Sen, S.; Sen, F.; Ivanov, V.; Atolia, E.; Farias, E.; McNicholas, T.P.; et al. In vivo biosensing via tissue-localizable near-infrared-fluorescent single-walled carbon nanotubes. Nat. Nanotechnol. 2013, 8, 873–880. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.; Li, Y.; Wang, Z.; Pei, F.; Hu, X.; Ji, Y.; Li, X.; Zhao, H.; Hu, W.; Lu, X.; et al. Organic semiconducting nanoprobe with redox-activatable NIR-II fluorescence for in vivo real-time monitoring of drug toxicity. Chem. Commun. 2019, 55, 27–30. [Google Scholar] [CrossRef]
- Shi, B.; Yan, Q.; Tang, J.; Xin, K.; Zhang, J.; Zhu, Y.; Xu, G.; Wang, R.; Chen, J.; Gao, W.; et al. Hydrogen sulfide-activatable second near-infrared fluorescent nanoassemblies for targeted photothermal cancer therapy. Nano Lett. 2018, 18, 6411–6416. [Google Scholar] [CrossRef]
- Xu, G.; Yan, Q.; Lv, X.; Zhu, Y.; Xin, K.; Shi, B.; Wang, R.; Chen, J.; Gao, W.; Shi, P.; et al. Imaging of colorectal cancers using activatable nanoprobes with second near-infrared window emission. Angew. Chem. Int. Ed. 2018, 57, 3626–3630. [Google Scholar] [CrossRef]
- Bai, S.; Jia, D.; Ma, X.; Liang, M.; Xue, P.; Kang, Y.; Xu, Z. Cylindrical polymer brushes-anisotropic unimolecular micelle drug delivery system for enhancing the effectiveness of chemotherapy. Bioact. Mater. 2021, 6, 2894–2904. [Google Scholar] [CrossRef]
- Li, Z.; Wu, J.; Wang, Q.; Liang, T.; Ge, J.; Wang, P.; Liu, Z. A universal strategy to construct lanthanide-doped nanoparticles-based activable NIR-II luminescence probe for bioimaging. iScience 2020, 23, 100962. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Luo, Y.; Hu, Y.; Liang, K.; He, G.; Chen, Q.; Wang, Q.; Chen, H. Photothermo-promoted nanocatalysis combined with H2S-mediated respiration inhibition for efficient cancer therapy. Adv. Funct. Mater. 2021, 31, 2007991. [Google Scholar] [CrossRef]
- Dou, K.; Feng, W.; Fan, C.; Cao, Y.; Xiang, Y.; Liu, Z. Flexible designing strategy to construct activatable NIR-II fluorescent probes with emission maxima beyond 1200 nm. Anal. Chem. 2021, 93, 4006–4014. [Google Scholar] [CrossRef]
- Liu, Q.; Zhong, Y.; Su, Y.; Zhao, L.; Peng, J. Real-time imaging of hepatic inflammation using hydrogen sulfide-activatable second near-infrared luminescent nanoprobes. Nano Lett. 2021, 21, 4606–4614. [Google Scholar] [CrossRef]
- Xu, M.; Wang, P.; Sun, S.; Gao, L.; Sun, L.; Zhang, L.; Zhang, J.; Wang, S.; Liang, X. Smart strategies to overcome tumor hypoxia toward the enhancement of cancer therapy. Nanoscale 2020, 12, 21519–21533. [Google Scholar] [CrossRef]
- Brown, J.M.; Wilson, W.R. Exploiting tumour hypoxia in cancer treatment. Nat. Rev. Cancer 2004, 4, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, A.; Peng, L.; Chai, L.; Xie, Z.; Ponraj, J.S.; Wang, X.; Wang, G.; Zhang, B.; Nie, G.; Xie, N.; et al. Advanced nanomaterials for hypoxia tumor therapy: Challenges and solutions. Nanoscale 2020, 12, 21497–21518. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Zhang, J.; Sun, Z.; Zhou, L.; Deng, G.; Li, S.; Li, W.; Gong, P.; Cai, L. Hypoxia-triggered single molecule probe for high-contrast NIR II/PA tumor imaging and robust photothermal therapy. Theranostics 2018, 8, 6025–6034. [Google Scholar] [CrossRef] [PubMed]
- Qiu, G.Z.; Jin, M.Z.; Dai, J.X.; Sun, W.; Feng, J.H.; Jin, W.L. Reprogramming of the tumor in the hypoxic niche: The emerging concept and associated therapeutic strategies. Trends Pharmacol. Sci. 2017, 38, 669–686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, D.C. Mitochondria and cancer. Nat. Rev. Cancer 2012, 12, 685–698. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; He, Y.; Lee, J.H.; Park, N.; Suh, M.; Chae, W.S.; Cao, J.; Peng, X.; Jung, H.; Kang, C.; et al. A self-calibrating bipartite viscosity sensor for mitochondria. J. Am. Chem. Soc. 2013, 135, 9181–9185. [Google Scholar] [CrossRef]
- Liu, F.; Yuan, Z.; Sui, X.; Wang, C.; Xu, M.; Li, W.; Chen, Y. Viscosity sensitive near-infrared fluorescent probes based on functionalized single-walled carbon nanotubes. Chem. Commun. 2020, 56, 8301–8304. [Google Scholar] [CrossRef]
- Dou, K.; Huang, W.; Xiang, Y.; Li, S.; Liu, Z. Design of activatable NIR-II molecular probe for in vivo elucidation of disease-related viscosity variations. Anal. Chem. 2020, 92, 4177–4181. [Google Scholar] [CrossRef] [Green Version]
- Kwon, H.; Kim, M.; Meany, B.; Piao, Y.; Powell, L.R.; Wang, Y. Optical probing of local pH and temperature in complex fluids with covalently functionalized, semiconducting carbon nanotubes. J. Phys. Chem. C 2015, 119, 3733–3739. [Google Scholar] [CrossRef]
- Lau, J.T.; Lo, P.C.; Jiang, X.J.; Wang, Q.; Ng, D.K. A dual activatable photosensitizer toward targeted photodynamic therapy. J. Med. Chem. 2014, 57, 4088–4097. [Google Scholar] [CrossRef]
- Zheng, Z.; Chen, Q.; Dai, R.; Jia, Z.; Yang, C.; Peng, X.; Zhang, R. A continuous stimuli-responsive system for NIR-II fluorescence/photoacoustic imaging guided photothermal/gas synergistic therapy. Nanoscale 2020, 12, 11562–11572. [Google Scholar] [CrossRef]
- Xu, Q.; Lee, K.A.; Lee, S.; Lee, K.M.; Lee, W.J.; Yoon, J. A highly specific fluorescent probe for hypochlorous acid and its application in imaging microbe-induced HOCl production. J. Am. Chem. Soc. 2013, 135, 9944–9949. [Google Scholar] [CrossRef]
- He, Y.; Wang, S.; Yu, P.; Yan, K.; Ming, J.; Yao, C.; He, Z.; El-Toni, A.M.; Khan, A.; Zhu, X.; et al. NIR-II cell endocytosis-activated fluorescent probes for in vivo high-contrast bioimaging diagnostics. Chem. Sci. 2021, 12, 10474–10482. [Google Scholar] [CrossRef]
- Teng, L.; Song, G.; Liu, Y.; Han, X.; Li, Z.; Wang, Y.; Huan, S.; Zhang, X.B.; Tan, W. Nitric oxide-activated “dual-key-one-lock” nanoprobe for in vivo molecular imaging and high-specificity cancer therapy. J. Am. Chem. Soc. 2019, 141, 13572–13581. [Google Scholar] [CrossRef]
- Teng, K.X.; Niu, L.Y.; Kang, Y.F.; Yang, Q.Z. Rational design of a “dual lock-and-key” supramolecular photosensitizer based on aromatic nucleophilic substitution for specific and enhanced photodynamic therapy. Chem. Sci. 2020, 11, 9703–9711. [Google Scholar] [CrossRef]
- Tang, Y.; Li, Y.; Hu, X.; Zhao, H.; Ji, Y.; Chen, L.; Hu, W.; Zhang, W.; Li, X.; Lu, X.; et al. “Dual lock-and-key”-controlled nanoprobes for ultrahigh specific fluorescence imaging in the second near-infrared window. Adv. Mater. 2018, 30, 1801140. [Google Scholar] [CrossRef]









| Activation Mode | Probe Name | Probe Fluorophores | Activation Mechanism | Excitation/ Emission (nm) | Biological Model | Refs |
|---|---|---|---|---|---|---|
| pH | BTC1070 | Pentamethine cyanine | Protonation, inhibition of ICT effect | 808/1000–1300 | Simulated gastric fluid with different pH | [54] |
| pH | Ag2S Ve | Ag2S | Protonation, increased hydrophilicity | 808/1250 | 4T1 breast cancer | [63] |
| β-Gal | BOD-M-βGal | BODPY derivatives | Hydrolyzation of β-galactose residues | 808/900–1300 | SKOV3 ovarian cancer | [72] |
| MMP14 | A&MMP@Ag2S-AF7P | Ag2S | Consumption of NIR absorber A1094 | 785/1050 | NB | [77] |
| H2O2 | Ag/Ag2S JNP | Ag2S | Oxidizing the plasmonic Ag parts | 825/1250 | MCF-7 breast cancer | [82] |
| ·OH | Hydro-1080 | cyanine dyes | Conjugated system recovery | 980/1100 | Liver injury | [88] |
| HClO | SPNPs | Degradation of the receptor ITTC | 808/1010 | Inflamed paws | [95] | |
| ONOO− | V&A@Ag2S | Ag2S | Consumption of A1094 chromophore | 825/1050 | TBI | [105] |
| ONOO− | V&C/PbS@Ag2Se | PbS@Ag2Se | Consumption of absorber Cy7.5 | 808/1616 | Ischemic stroke | [109] |
| NO | AOSNP | Ag2S | Energy receptor units are converted | 808/900–1150 | Liver injury | [115] |
| H2S | WH-3 | BODIPY derivatives | Release fluorescence quencher | 980/1140 | HCT-116 colon cancer | [121] |
| Hypoxia (NTR) | IR-1048-MZ | IR-1048 | MZ group was reduced | 980/1046 | A549 lung cancer | [126] |
| Viscosity | WD-NO2 | BODIPY derivatives | Intramolecular rotation of chemical bonds is limited | 808/900–1400 | Abnormal liver viscosity | [131] |
| Hyal and GSH | HISSNPs | IR-1061 | Fracture of HA chains and disulfide bonds | 808/1070 | MCF-7 breast cancer | [139] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, D.; Pan, J.; Xu, S.; Fu, S.; Chu, C.; Liu, G. Activatable Second Near-Infrared Fluorescent Probes: A New Accurate Diagnosis Strategy for Diseases. Biosensors 2021, 11, 436. https://doi.org/10.3390/bios11110436
Li D, Pan J, Xu S, Fu S, Chu C, Liu G. Activatable Second Near-Infrared Fluorescent Probes: A New Accurate Diagnosis Strategy for Diseases. Biosensors. 2021; 11(11):436. https://doi.org/10.3390/bios11110436
Chicago/Turabian StyleLi, Dong, Jie Pan, Shuyu Xu, Shiying Fu, Chengchao Chu, and Gang Liu. 2021. "Activatable Second Near-Infrared Fluorescent Probes: A New Accurate Diagnosis Strategy for Diseases" Biosensors 11, no. 11: 436. https://doi.org/10.3390/bios11110436
APA StyleLi, D., Pan, J., Xu, S., Fu, S., Chu, C., & Liu, G. (2021). Activatable Second Near-Infrared Fluorescent Probes: A New Accurate Diagnosis Strategy for Diseases. Biosensors, 11(11), 436. https://doi.org/10.3390/bios11110436







